Gaining Micropattern Fidelity in an NOA81 Microsieve Laser Ablation Process
Abstract
1. Introduction
2. Materials and Methods
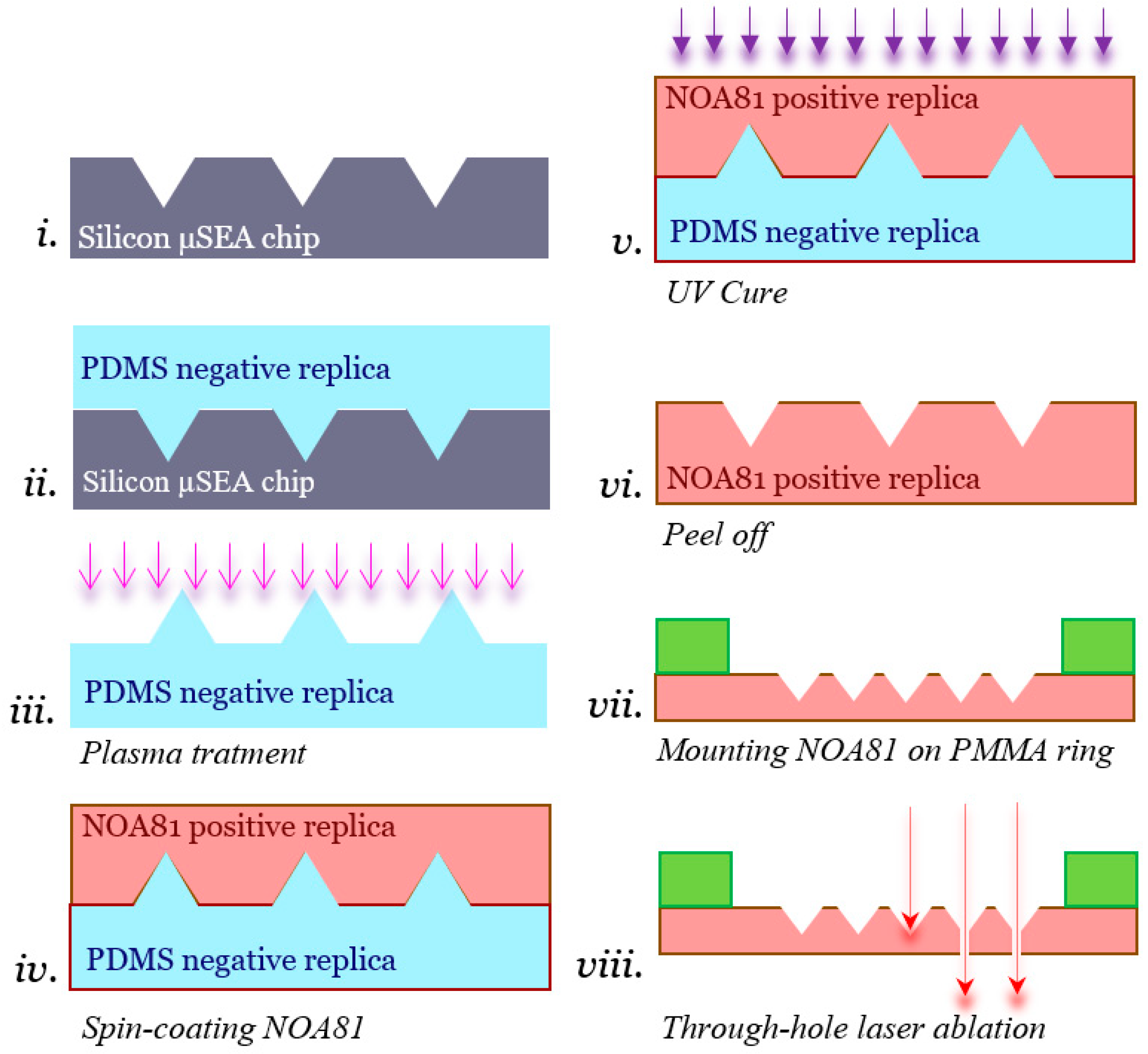
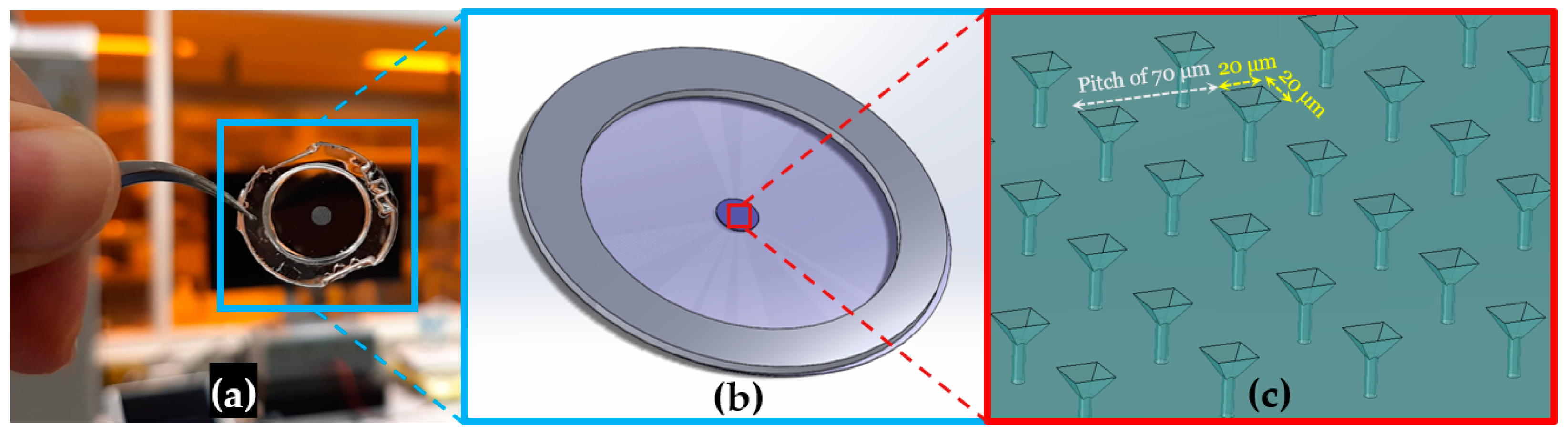
2.1. NOA81 Replica Molding from Silicon Microsieve Electrode Array
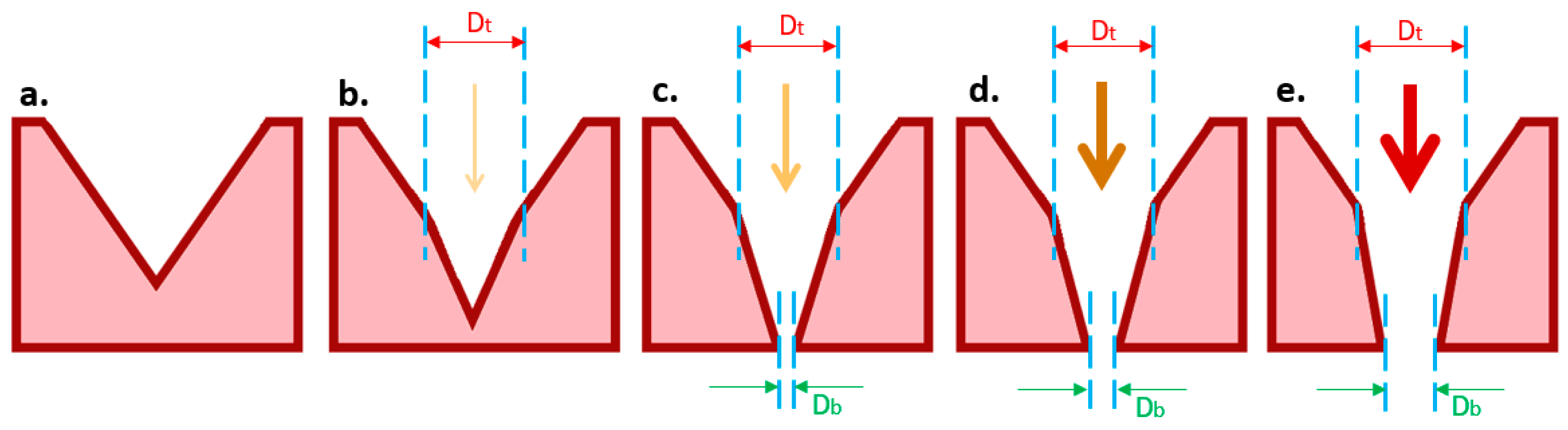
2.2. Laser Ablation of the NOA81 Microsieve
3. Results and Discussion
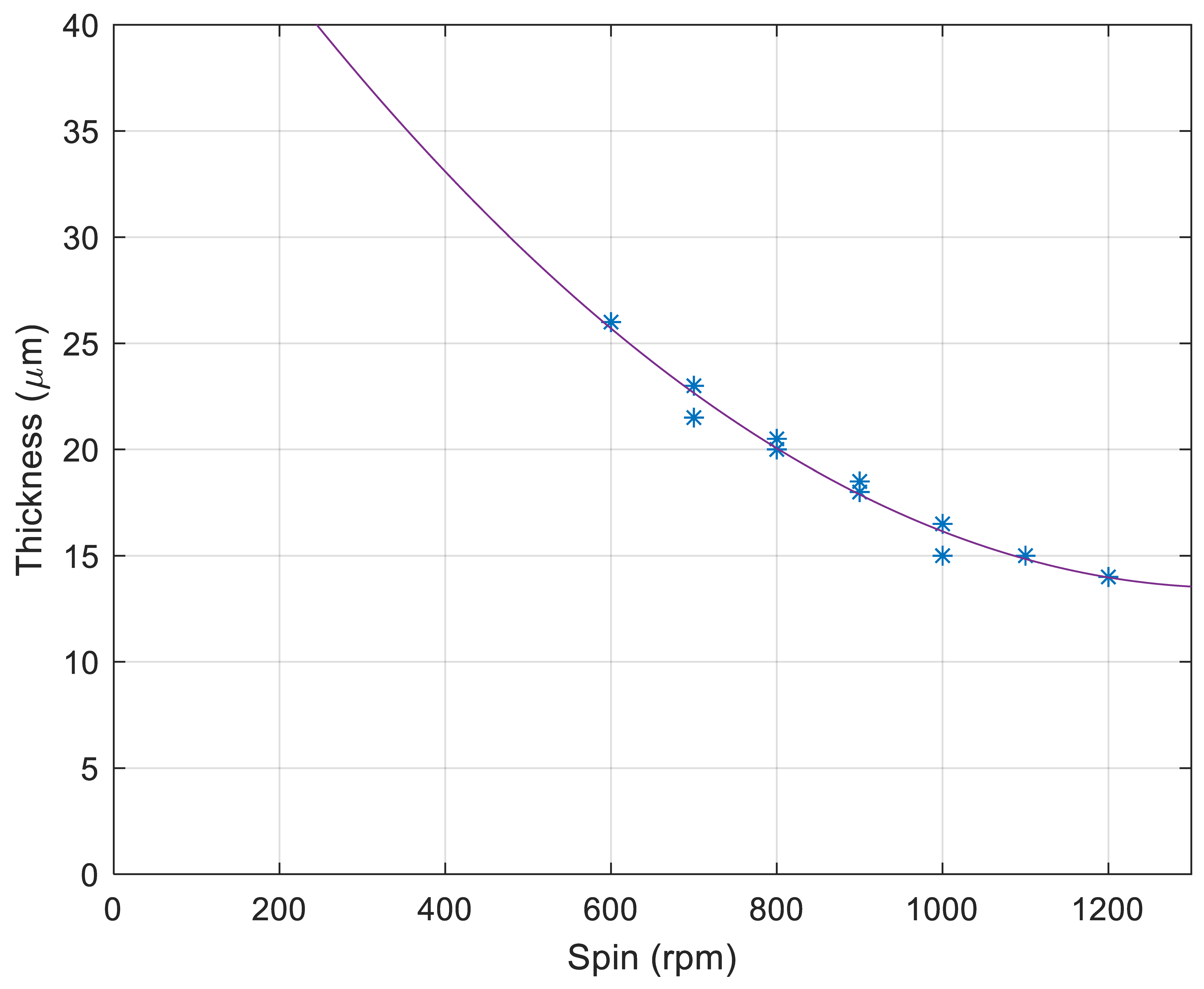
3.1. NOA81 Device Replication
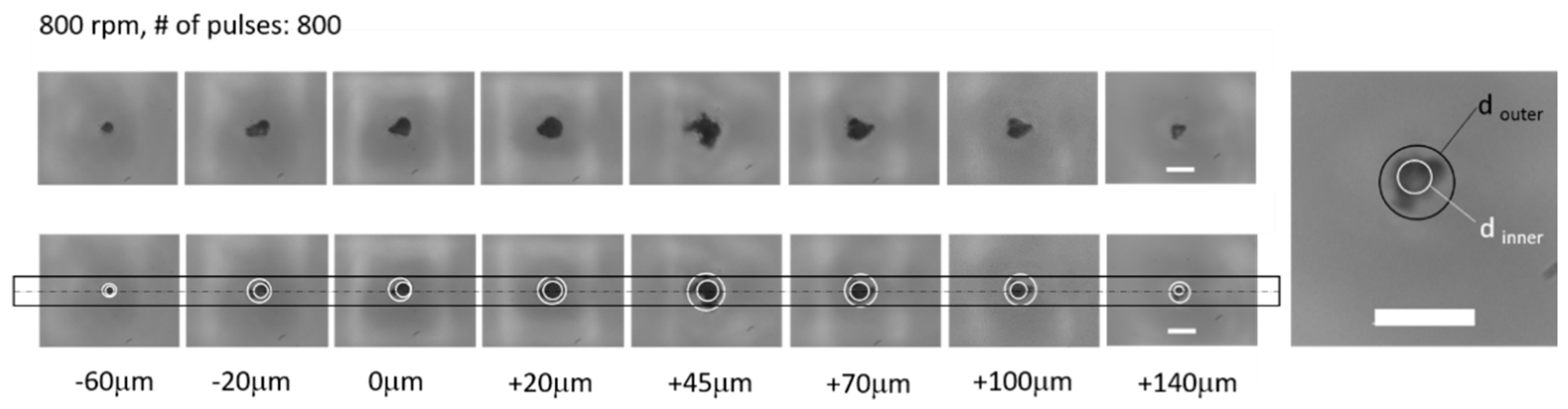
3.2. Laser Ablation of NOA81
3.3. Microsieves Characterization by Replica and FIB-Cut Sectioning
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Knowles, M.R.H.; Rutterford, G.; Karnakis, D.; Ferguson, A. Micro-machining of metals, ceramics and polymers using nanosecond lasers. Int. J. Adv. Manuf. Technol. 2007, 33, 95–102. [Google Scholar] [CrossRef]
- Mishra, S.; Yadava, V. Laser Beam MicroMachining (LBMM)-A review. Opt. Lasers Eng. 2015, 73, 89–122. [Google Scholar] [CrossRef]
- Van Pelt, S.; Frijns, A.J.H.; Mandamparambil, R.; Toonder, J.D. Local wettability tuning with laser ablation redeposits on PDMS. Appl. Surf. Sci. 2014, 303, 456–464. [Google Scholar] [CrossRef]
- Hart, C.; Rajaraman, S. Low-Power, Multimodal Laser Micromachining of Materials for Applications in sub-5 µm Shadow Masks and sub-10 µm Interdigitated Electrodes (IDEs) Fabrication. Micromachines 2020, 11, 178. [Google Scholar] [CrossRef] [PubMed]
- Chien, H.-L.; Lee, Y.-C. A ball valve micro-pump based on axially symmetrical nozzle fabricated by excimer laser micromachining technology. Int. J. Precis. Eng. Manuf. 2017, 18, 1315–1320. [Google Scholar] [CrossRef]
- Sun, T.; Jackson, S.; Haycock, J.W.; MacNeil, S. Culture of skin cells in 3D rather than 2D improves their ability to survive exposure to cytotoxic agents. J. Biotechnol. 2006, 122, 372–381. [Google Scholar] [CrossRef]
- Tang, Q.Y.; Tong, W.Y.; Shi, J.; Shi, P.; Lam, Y.W.; Pang, S.W. Influence of engineered surface on cell directionality and motility. Biofabrication 2014, 6, 015011. [Google Scholar] [CrossRef]
- Braeken, D.; Li, Y.-C.E. Brain-on-a-chip Devices for Drug Screening and Disease Modeling Applications. Curr. Pharm. Des. 2019, 24, 5419–5436. [Google Scholar] [CrossRef]
- Van Engeland, N.C.A.; Pollet, A.M.A.O.; Toonder, J.M.J.D.; Bouten, C.V.; Stassen, O.M.J.A.; Sahlgren, C.M. A biomimetic microfluidic model to study signalling between endothelial and vascular smooth muscle cells under hemodynamic conditions. Lab Chip 2018, 18, 1607–1620. [Google Scholar] [CrossRef]
- Sano, H.; Watanabe, M.; Yamashita, T.; Tanishita, K.; Sudo, R. Control of vessel diameters mediated by flow-induced outward vascular remodeling in vitro. Biofabrication 2020, 12, 045008. [Google Scholar] [CrossRef]
- Lee, S.W.L.; Campisi, M.; Osaki, T.; Possenti, L.; Mattu, C.; Adriani, G.; Kamm, R.D.; Chiono, V. Modeling Nanocarrier Transport across a 3D In Vitro Human Blood-Brain–Barrier Microvasculature. Adv. Heal. Mater. 2020, 9, e1901486. [Google Scholar] [CrossRef] [PubMed]
- Tang, V.W. Collagen, stiffness, and adhesion: The evolutionary basis of vertebrate mechanobiology. Mol. Biol. Cell 2020, 31, 1823–1834. [Google Scholar] [CrossRef] [PubMed]
- Pieuchot, L.; Marteau, J.; Guignandon, A.; Dos Santos, T.; Brigaud, I.; Chauvy, P.-F.; Cloatre, T.; Ponche, A.; Petithory, T.; Rougerie, P.; et al. Curvotaxis directs cell migration through cell-scale curvature landscapes. Nat. Commun. 2018, 9, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Ramaswamy, Y.; Roohani, I.; No, Y.J.; Madafiglio, G.; Chang, F.; Zhao, F.; Lu, Z.; Zreiqat, H. Nature-inspired topographies on hydroxyapatite surfaces regulate stem cells behaviour. Bioact. Mater. 2021, 6, 1107–1117. [Google Scholar] [CrossRef]
- Knight, E.; Przyborski, S. Advances in 3D cell culture technologies enabling tissue-like structures to be created in vitro. J. Anat. 2015, 227, 746–756. [Google Scholar] [CrossRef]
- Callens, S.J.; Uyttendaele, R.J.; Fratila-Apachitei, L.E.; Zadpoor, A.A. Substrate curvature as a cue to guide spatiotemporal cell and tissue organization. Biomaterials 2020, 232, 119739. [Google Scholar] [CrossRef]
- Centeno, E.G.Z.; Cimarosti, H.; Bithell, A. 2D versus 3D human induced pluripotent stem cell-derived cultures for neurodegenerative disease modelling. Mol. Neurodegener. 2018, 13, 1–15. [Google Scholar] [CrossRef]
- Bosi, S.; Rauti, R.; Laishram, J.; Turco, A.; Lonardoni, D.; Nieus, T.; Prato, M.; Scaini, D.; Ballerini, L. From 2D to 3D: Novel nanostructured scaffolds to investigate signalling in reconstructed neuronal networks. Sci. Rep. 2015, 5, srep09562. [Google Scholar] [CrossRef]
- Xie, S.; Luttge, R. Imprint lithography provides topographical nanocues to guide cell growth in primary cortical cell culture. Microelectron. Eng. 2014, 124, 30–36. [Google Scholar] [CrossRef]
- Bastiaens, A.; Luttge, R. Nanogroove-Enhanced Hydrogel Scaffolds for 3D Neuronal Cell Culture: An Easy Access Brain-on-Chip Model. Micromachines 2019, 10, 638. [Google Scholar] [CrossRef]
- Bastiaens, A.; Sabahi-Kaviani, R.; Luttge, R. Nanogrooves for 2D and 3D Microenvironments of SH-SY5Y Cultures in Brain-on-Chip Technology. Front. Neurosci. 2020, 14, 666. [Google Scholar] [CrossRef] [PubMed]
- Schurink, B.; Berenschot, E.J.W.; Tiggelaar, R.M.; Luttge, R. Highly uniform sieving structure by corner lithography and silicon wet etching. Microelectron. Eng. 2015, 144, 12–18. [Google Scholar] [CrossRef]
- Frimat, J.-P.; Schurink, B.; Luttge, R. Passive pumping for the parallel trapping of single neurons onto a microsieve electrode array. J. Vac. Sci. Technol. B 2017, 35, 06GA01. [Google Scholar] [CrossRef]
- Sollier, E.; Murray, C.; Maoddi, P.; Di Carlo, D. Rapid prototyping polymers for microfluidic devices and high pressure injections. Lab Chip 2011, 11, 3752–3765. [Google Scholar] [CrossRef]
- Mendes-Felipe, C.; Oliveira, J.; Etxebarria, I.; Vilas-Vilela, J.L.; Lanceros-Mendez, S. State-of-the-Art and Future Challenges of UV Curable Polymer-Based Smart Materials for Printing Technologies. Adv. Mater. Technol. 2019, 4, 1–16. [Google Scholar] [CrossRef]
- Han, D.; Lee, H. Recent advances in multi-material additive manufacturing: Methods and applications. Curr. Opin. Chem. Eng. 2020, 28, 158–166. [Google Scholar] [CrossRef]
- Carve, M.; Wlodkowic, D. 3D-Printed Chips: Compatibility of Additive Manufacturing Photopolymeric Substrata with Biological Applications. Micromachines 2018, 9, 91. [Google Scholar] [CrossRef]
- Kwak, T.J.; Lee, E. Rapid multilayer microfabrication for modeling organotropic metastasis in breast cancer. Biofabrication 2021, 13, 015002. [Google Scholar] [CrossRef]
- Rimington, R.P.; Capel, A.J.; Player, D.J.; Bibb, R.; Christie, S.D.R.; Lewis, M.P. Feasibility and Biocompatibility of 3D-Printed Photopolymerized and Laser Sintered Polymers for Neuronal, Myogenic, and Hepatic Cell Types. Macromol. Biosci. 2018, 18, e1800113. [Google Scholar] [CrossRef]
- Koroleva, A.; Gill, A.A.; Ortega, I.; Haycock, J.W.; Schlie, S.; Gittard, S.D.; Chichkov, B.N.; Claeyssens, F. Two-photon polymerization-generated and micromolding-replicated 3D scaffolds for peripheral neural tissue engineering applications. Biofabrication 2012, 4, 025005. [Google Scholar] [CrossRef]
- Moonen, E.; Luttge, R.; Frimat, J.-P. Single cell trapping by capillary pumping using NOA81 replica moulded stencils. Microelectron. Eng. 2018, 197, 1–7. [Google Scholar] [CrossRef]
- Levental, I.; Georgesa, P.C.; Janmey, P.A. Soft biological materials and their impact on cell function. Soft Matter 2007, 3, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Schurink, B. Microfabrication and Microfluidics for 3D Brain-on-Chip; University Library/University of Twente: Enschede, The Netherlands, 2016. [Google Scholar]
- Optec, S.A. MicroMaster User Instruction Manual, 6th ed.; Optec, S.A.: Frameries, Belgium, 2005. [Google Scholar]
- Norland Products Inc., Norland Optical Adhesive 81. Available online: http://www.norlandprod.com/adhesives/NOA81.html (accessed on 10 December 2020).
- Bartolo, D.; Degré, G.; Nghe, P.; Studer, V. Microfluidic stickers. Lab Chip 2008, 8, 274–279. [Google Scholar] [CrossRef] [PubMed]
- Wägli, P.; Homsy, A.; De Rooij, N. Norland optical adhesive (NOA81) microchannels with adjustable wetting behavior and high chemical resistance against a range of mid-infrared-transparent organic solvents. Sens. Actuators B Chem. 2011, 156, 994–1001. [Google Scholar] [CrossRef]
- Peurrung, L.; Graves, D. Spin coating over topography. IEEE Trans. Semicond. Manuf. 1993, 6, 72–76. [Google Scholar] [CrossRef]
- Wu, T.; Zhu, F.; Wang, Q. The Global Variation of Photoresist Topography and CD Uniformity due to Local High Step. In Proceedings of the 2020 IEEE International Conference on Artificial Intelligence and Computer Applications (ICAICA), Dalian, China, 27–29 June 2020; pp. 1005–1009. [Google Scholar]
- Zhang, Z.; Xu, J.; Drapaca, C. Particle squeezing in narrow confinements. Microfluid. Nanofluidics 2018, 22, 120. [Google Scholar] [CrossRef]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sabahi-Kaviani, R.; Luttge, R. Gaining Micropattern Fidelity in an NOA81 Microsieve Laser Ablation Process. Micromachines 2021, 12, 21. https://doi.org/10.3390/mi12010021
Sabahi-Kaviani R, Luttge R. Gaining Micropattern Fidelity in an NOA81 Microsieve Laser Ablation Process. Micromachines. 2021; 12(1):21. https://doi.org/10.3390/mi12010021
Chicago/Turabian StyleSabahi-Kaviani, Rahman, and Regina Luttge. 2021. "Gaining Micropattern Fidelity in an NOA81 Microsieve Laser Ablation Process" Micromachines 12, no. 1: 21. https://doi.org/10.3390/mi12010021
APA StyleSabahi-Kaviani, R., & Luttge, R. (2021). Gaining Micropattern Fidelity in an NOA81 Microsieve Laser Ablation Process. Micromachines, 12(1), 21. https://doi.org/10.3390/mi12010021




