Synthesis and a Photo-Stability Study of Organic Dyes for Electro-Fluidic Display
Abstract
1. Introduction
2. Experimental
2.1. Materials
2.2. Analysis Methods
2.3. EFD Oil Formulation
2.4. Electro-Fluidic Display (EFD) Device Fabrication
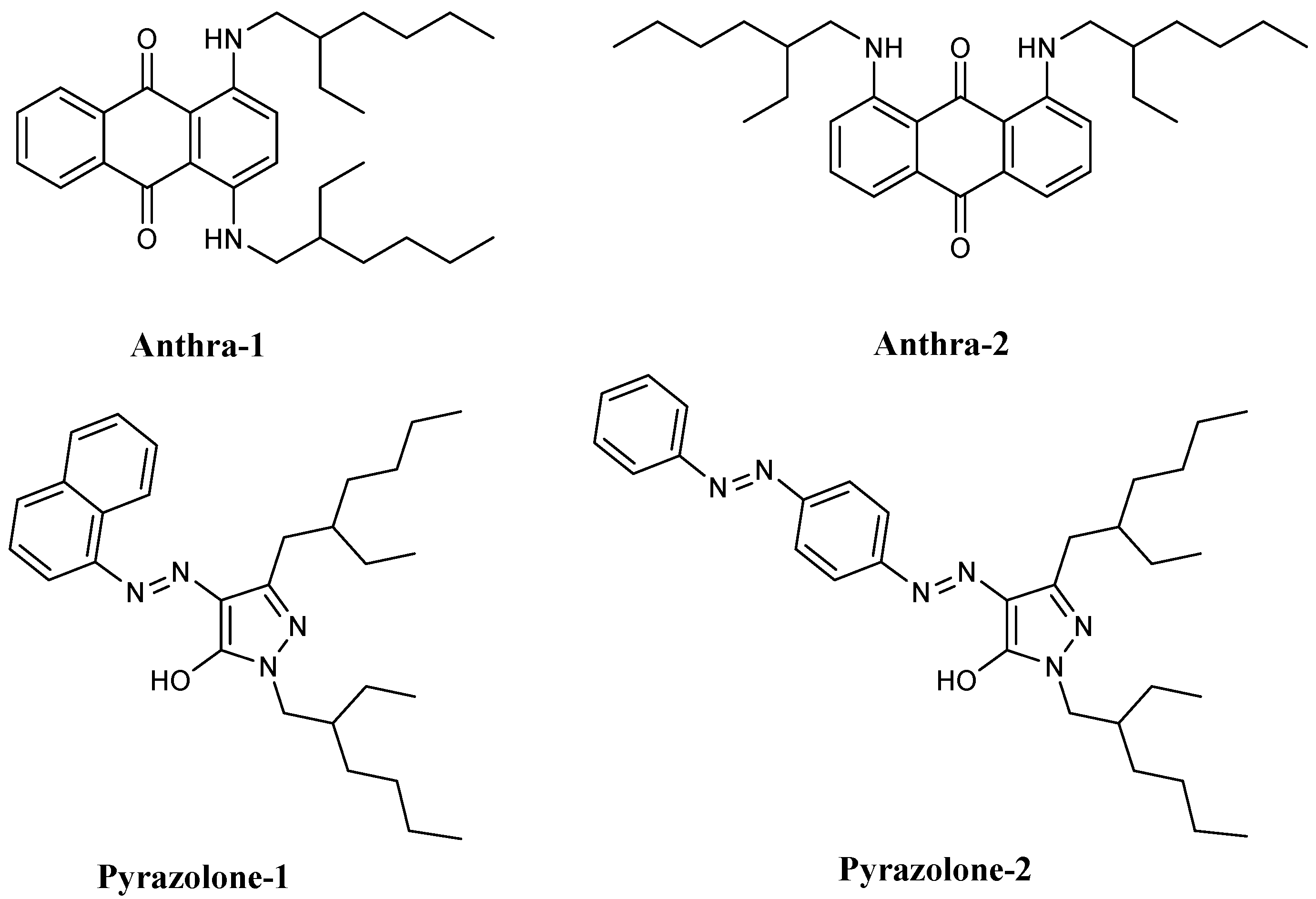
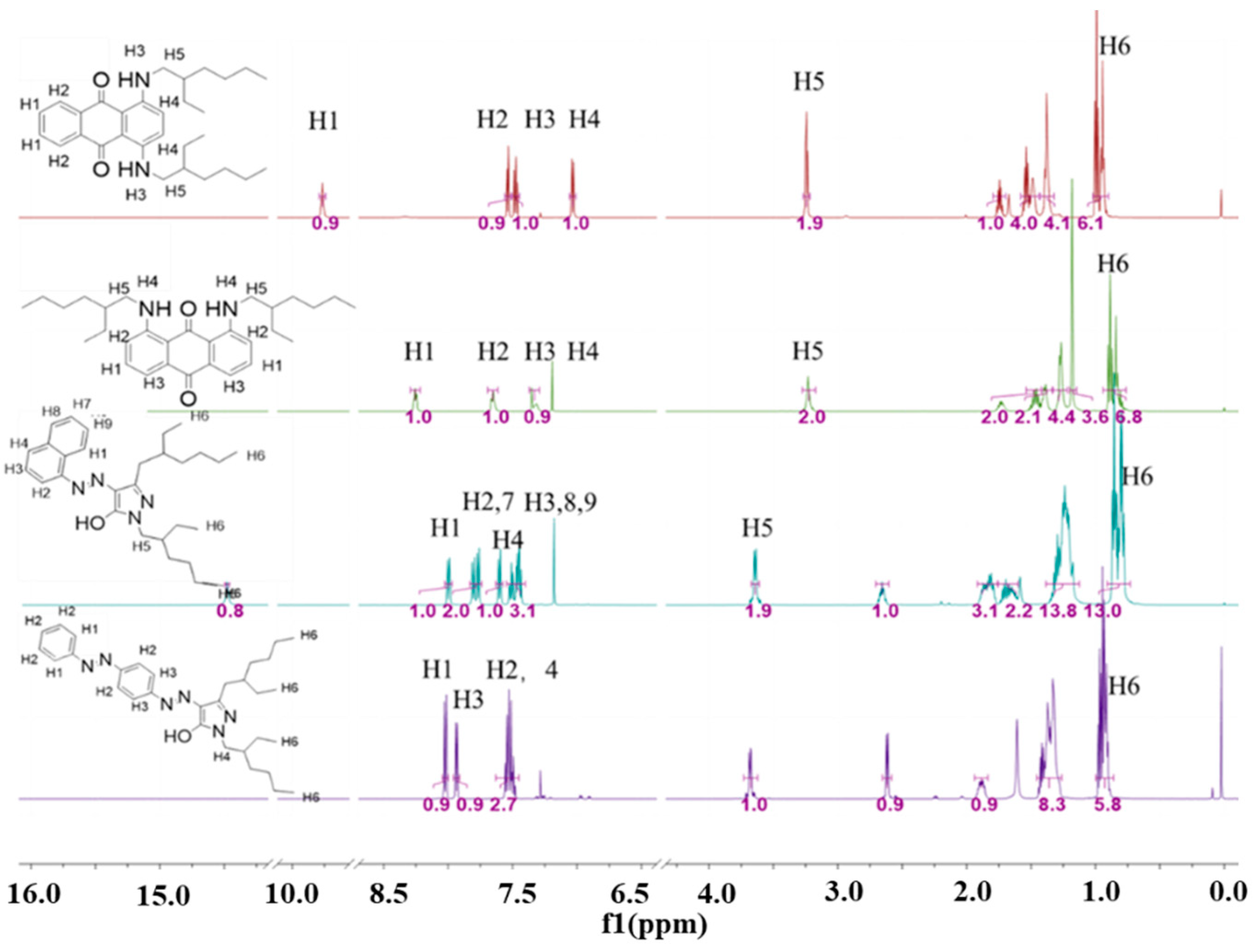
3. Dye Synthesis and Characterization
4. Results and Discussion
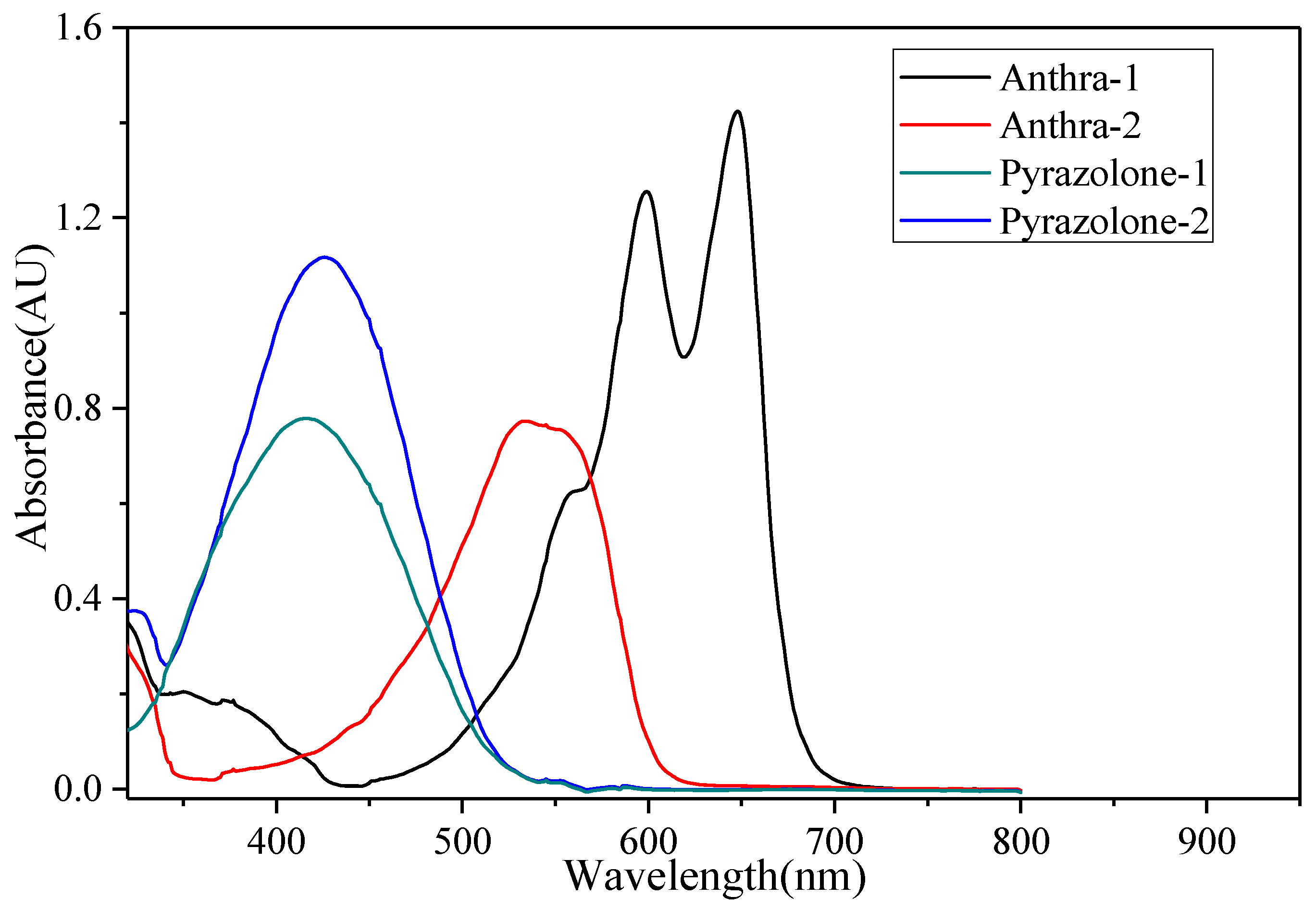
4.1. Absorption Properties of Four Dyes
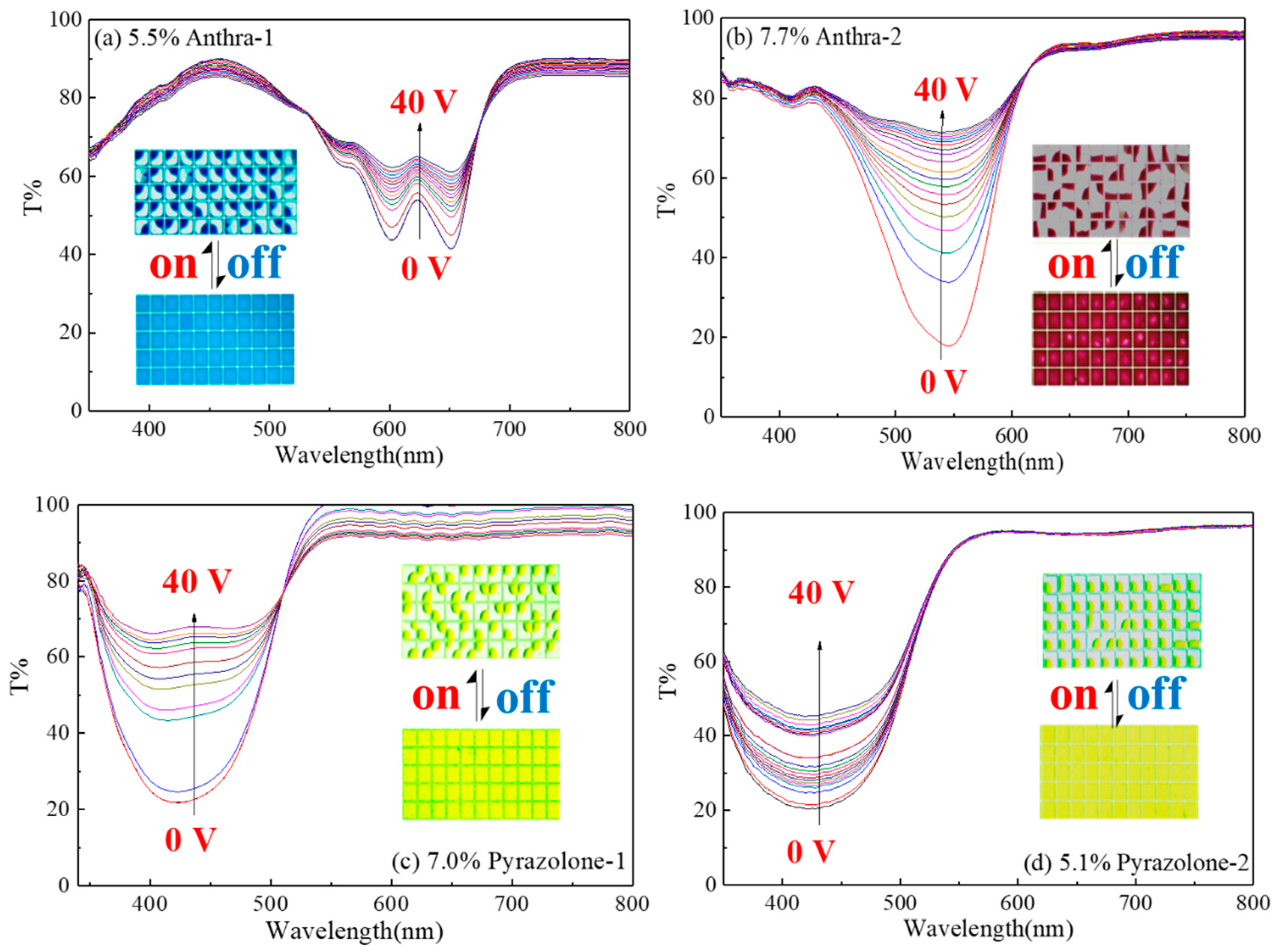
4.2. Photoelectric Response Properties of Dyes
4.3. Photo-Stability Research of Dyes
4.4. Effect of Irradiation on Backflow Property of EFD Devices
5. Conclusion
Author Contributions
Funding
Conflicts of Interest
References
- Hayes, R.A.; Feenstra, B.J. Video-speed electronic paper based on electrowetting. Nature 2003, 425, 383–385. [Google Scholar] [CrossRef] [PubMed]
- Tang, B.; Groenewold, J.; Zhou, M.; Hayes, R.A.; Zhou, G.G. Interfacial electrofluidics in confined systems. Sci. Rep. 2016, 6, 26593. [Google Scholar] [CrossRef] [PubMed]
- Shui, L.; Hayes, R.A.; Jin, M.; Zhang, X.; Bai, P.; van den Berg, A.; Zhou, G. Microfluidics for electronic paper-like displays. Lab Chip 2014, 14, 2374–2384. [Google Scholar] [CrossRef] [PubMed]
- Riahi, M.; Brakke, K.A.; Alizadeh, E.; Shahroosvand, H. Fabrication and characterization of an electrowetting display based on the wetting–dewetting in a cubic structure. Opt. Int. J. Light Electron Opt. 2016, 127, 2703–2707. [Google Scholar] [CrossRef]
- Wang, M.; Guo, Y.; Hayes, R.; Liu, D.; Broer, D.; Zhou, G. Forming spacers in situ by photolithography to mechanically stabilize electrofluidic-based switchable optical elements. Materials 2016, 9, 250. [Google Scholar] [CrossRef] [PubMed]
- Smith, N.R.; Hou, L.; Zhang, J.; Heikenfeld, J. Fabrication and demonstration of electrowetting liquid lens arrays. J. Disp. Technol. 2009, 5, 411–413. [Google Scholar] [CrossRef]
- Heikenfeld, J.; Smith, N.; Dhindsa, M.; Zhou, K.; Kilaru, M.; Hou, L.; Zhang, J.; Kreit, E.; Raj, B. Recent progress in arrayed electrowetting optics. Opt. Photonics News 2009, 4, 20–26. [Google Scholar] [CrossRef]
- Heikenfeld, J.; Zhou, K.; Kreit, E.; Raj, B.; Yang, S.; Sun, B.; Milarcik, A.; Clapp, L.; Schwartz, R. Electrofluidic displays using Young-Laplace transposition of brilliant pigment dispersions. Nat. Photonics 2009, 3, 292–296. [Google Scholar] [CrossRef]
- Van De Weijer, M.M.H.; Massard, R.; Hayes, R.A. Electrowetting Elements. U.S. Patent 8,980,141, 17 March 2011. [Google Scholar]
- Ishida, M.; Shiga, Y.; Takeda, U.; Kadowaki, M. Ink Containing Anthraquinone Based Dye, Dye Used in the Ink, and Display. U.S. Patent 8,999,050, 7 April 2013. [Google Scholar]
- Shiga, Y.; Takeda, U.; Ichinosawa, S.; Ishida, M. Ink Containing Heterocyclic azo Dye, and Dye for Use in Said Ink. U.S. Patent 8,747,537, 10 June 2014. [Google Scholar]
- Shiga, Y.; Ishida, M. Pyrazole Disazo Dye and Ink Containing the Dye. U.S. Patent 8,143,382, 27 March 2012. [Google Scholar]
- Chiang, Y.; Chao, Y. Synthesis of dis-azo black dyes for electrowetting displays. Mater. Sci. Eng. B 2012, 177, 1672–1677. [Google Scholar] [CrossRef]
- Farrand, L.D.; Smith, N.; Corbett, A. and Lawrence, A.; Merck Patent GmbH. Electrowetting Fluids. U.S. Patent 20150355456 A1, 10 December 2015. [Google Scholar]
- Chiang, Y.F.; Chao, Y.C. Synthesis and Application of Oil-Soluble Red Dyes Derived from p-n-Alkyl Aniline. Mater. Sci. Appl. 2014, 5, 485–490. [Google Scholar] [CrossRef]
- Kato, T.; Higuchi, S.; Fukushige, Y.; Jimbo, Y.; Sasaki, D. Colored Composition for Electrowetting Display, Image Display Structure, and Electrowetting Display Device. U.S. Patent 9,494,789, 15 November 2014. [Google Scholar]
- Lee, P.T.; Chiu, C.W.; Chang, L.Y.; Chou, P.Y.; Lee, T.M.; Chang, T.Y.; Wu, M.T.; Cheng, W.Y.; Kuo, S.W.; Lin, J.J. Tailoring Pigment Dispersants with Polyisobutylene Twin-Tail Structures for Electrowetting Display Application. ACS Appl. Mater. Interfaces 2014, 6, 14345–14352. [Google Scholar] [CrossRef]
- Lee, P.T.; Chiu, C.W.; Lee, T.M.; Chang, T.Y.; Wu, M.T.; Cheng, W.Y.; Kuo, S.W.; Lin, J.J. First Fabrication of Electrowetting Display by Using Pigment-in-Oil Driving Pixels. ACS Appl. Mater. Interfaces 2013, 5, 5914–5920. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Jiang, H.; Ye, D.; Zhou, R.; Li, H.; Tang, B.; Jin, M.; Li, N.; Guo, Y.; Zhou, G. Synthesis and application of an alkylated pyrazole-based azo dye for Electro-fluidic display. J. Soc. Inf. Disp. 2018, 26, 369–375. [Google Scholar] [CrossRef]
- Van de Weijer, W.; Melanie, M.; Massard, R.; Hayes Robert, A. Improvements in Relation to Electrowetting Elements. U.S. Patent WO2010031860 A2, 25 March 2010. [Google Scholar]
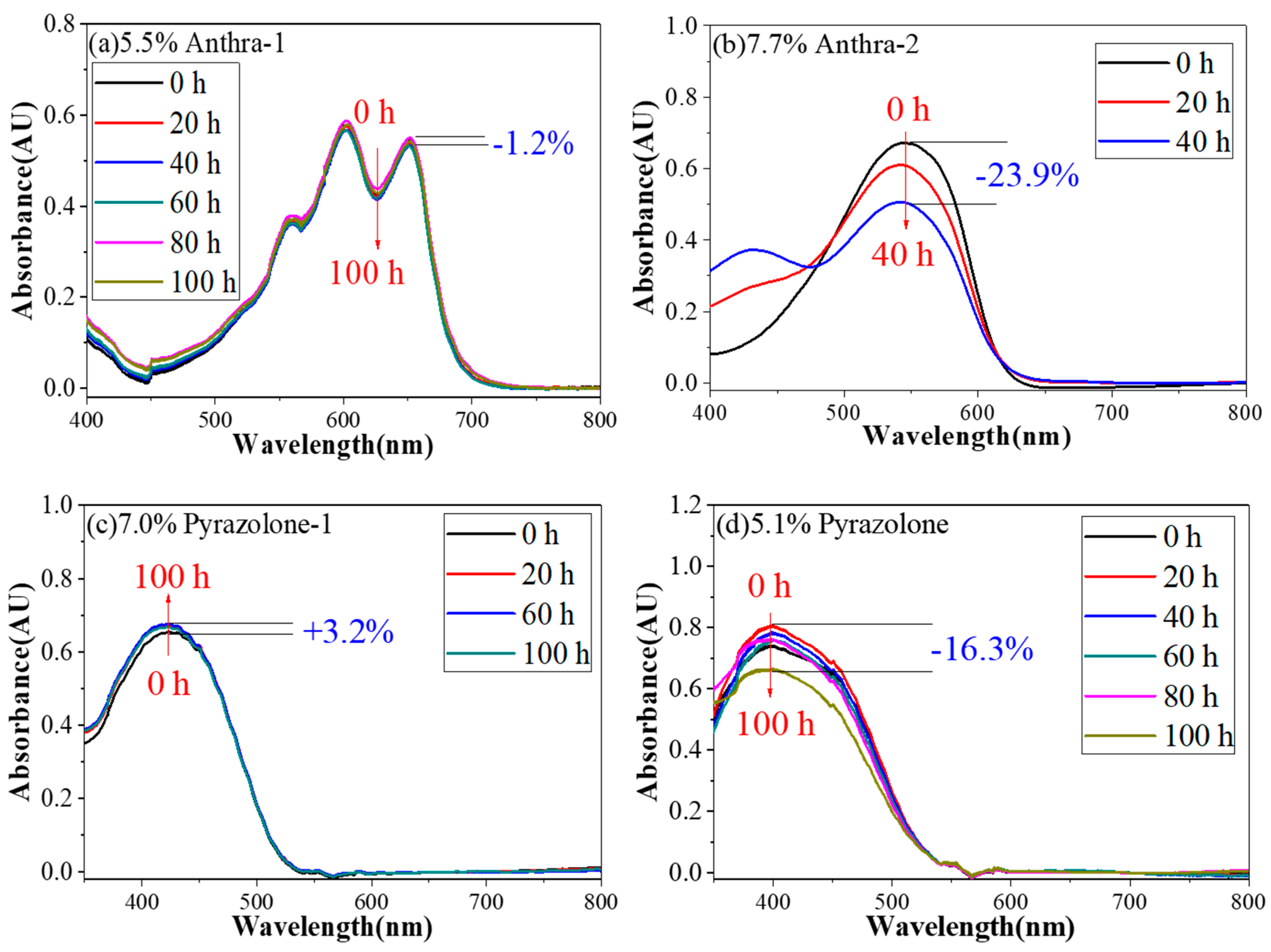
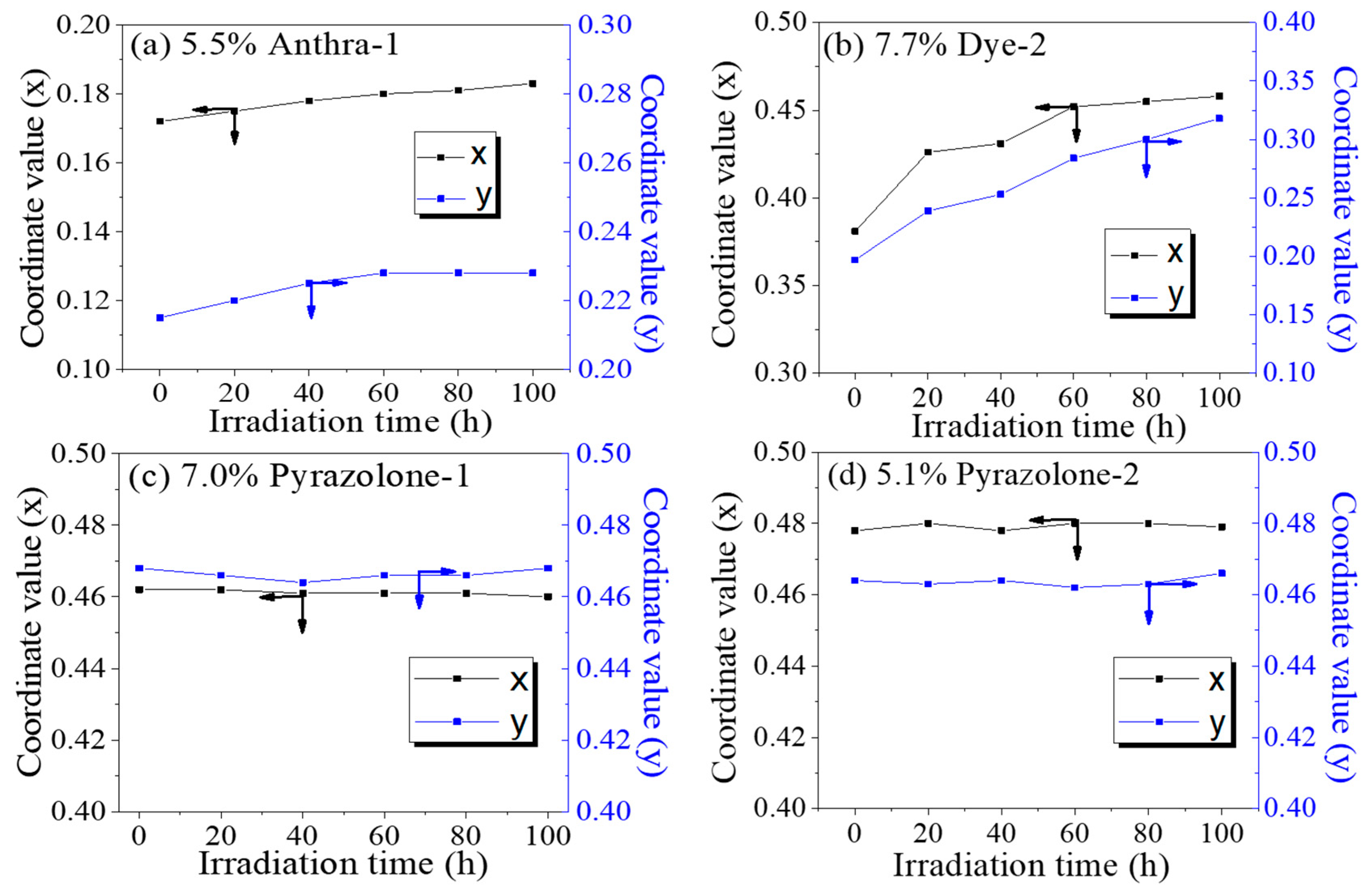
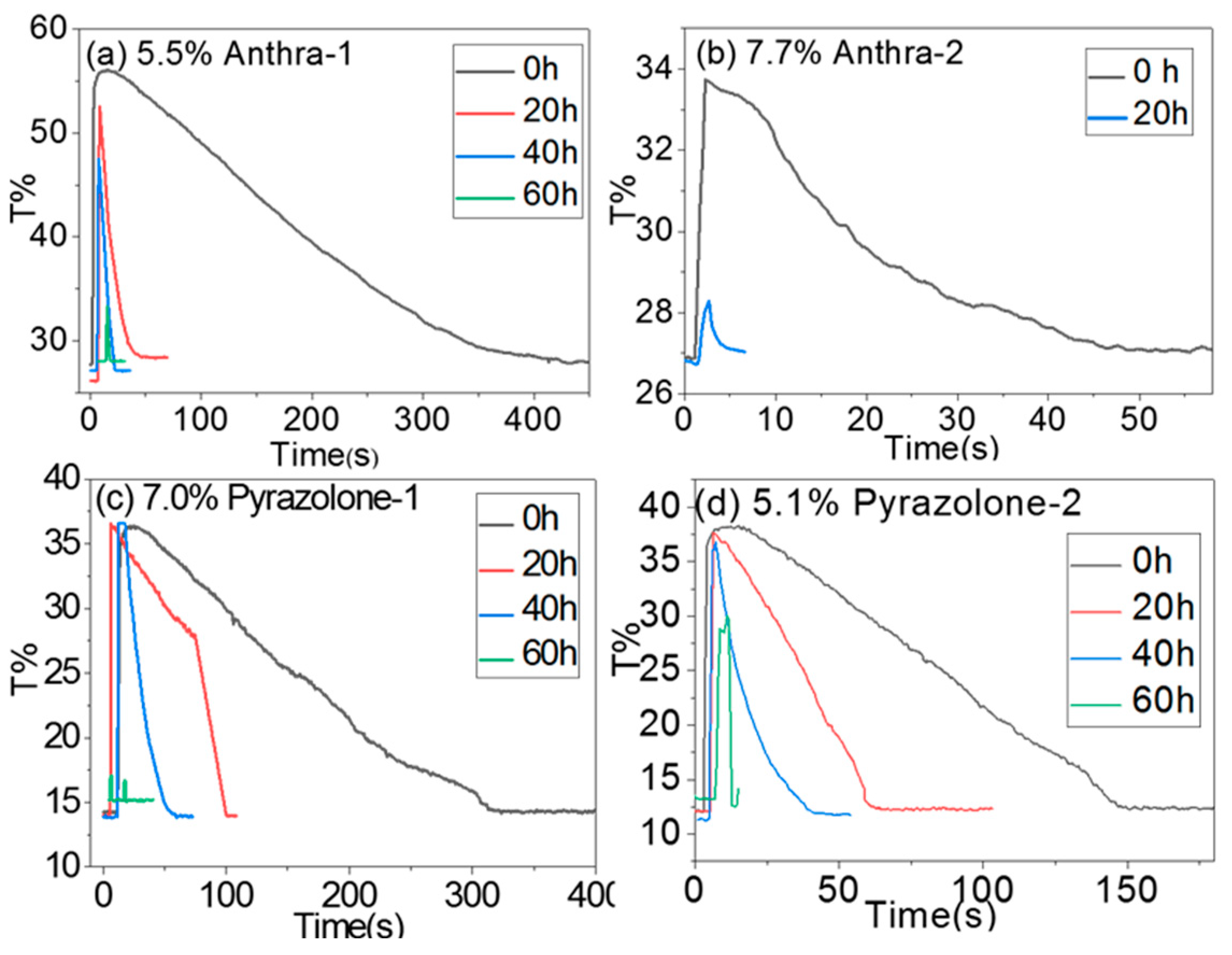
| Dye | Before Irradiation | After Irradiation | ΔE | ||||
|---|---|---|---|---|---|---|---|
| L | a | b | L | a | b | ||
| Anthra-1 | 174.9 | −45.5 | −123.6 | 171.2 | −43.5 | −117.1 | 7.7 |
| Anthra-2 | 140.8 | 181.0 | −68.2 | 160.7 | 113.2 | 41.6 | 130.5 |
| Pyrazolone-1 | 236.2 | 14.0 | 210.5 | 236.3 | 12.3 | 209.3 | 2.0 |
| Pyrazolone-2 | 233.3 | 29.7 | 221.4 | 231.9 | 28.1 | 224.6 | 3.7 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Deng, Y.; Li, S.; Ye, D.; Jiang, H.; Tang, B.; Zhou, G. Synthesis and a Photo-Stability Study of Organic Dyes for Electro-Fluidic Display. Micromachines 2020, 11, 81. https://doi.org/10.3390/mi11010081
Deng Y, Li S, Ye D, Jiang H, Tang B, Zhou G. Synthesis and a Photo-Stability Study of Organic Dyes for Electro-Fluidic Display. Micromachines. 2020; 11(1):81. https://doi.org/10.3390/mi11010081
Chicago/Turabian StyleDeng, Yong, Shi Li, Dechao Ye, Hongwei Jiang, Biao Tang, and Guofu Zhou. 2020. "Synthesis and a Photo-Stability Study of Organic Dyes for Electro-Fluidic Display" Micromachines 11, no. 1: 81. https://doi.org/10.3390/mi11010081
APA StyleDeng, Y., Li, S., Ye, D., Jiang, H., Tang, B., & Zhou, G. (2020). Synthesis and a Photo-Stability Study of Organic Dyes for Electro-Fluidic Display. Micromachines, 11(1), 81. https://doi.org/10.3390/mi11010081





