Rapid Microfluidic Mixer Based on Ferrofluid and Integrated Microscale NdFeB-PDMS Magnet
Abstract
1. Introduction
2. Work Concept and Materials
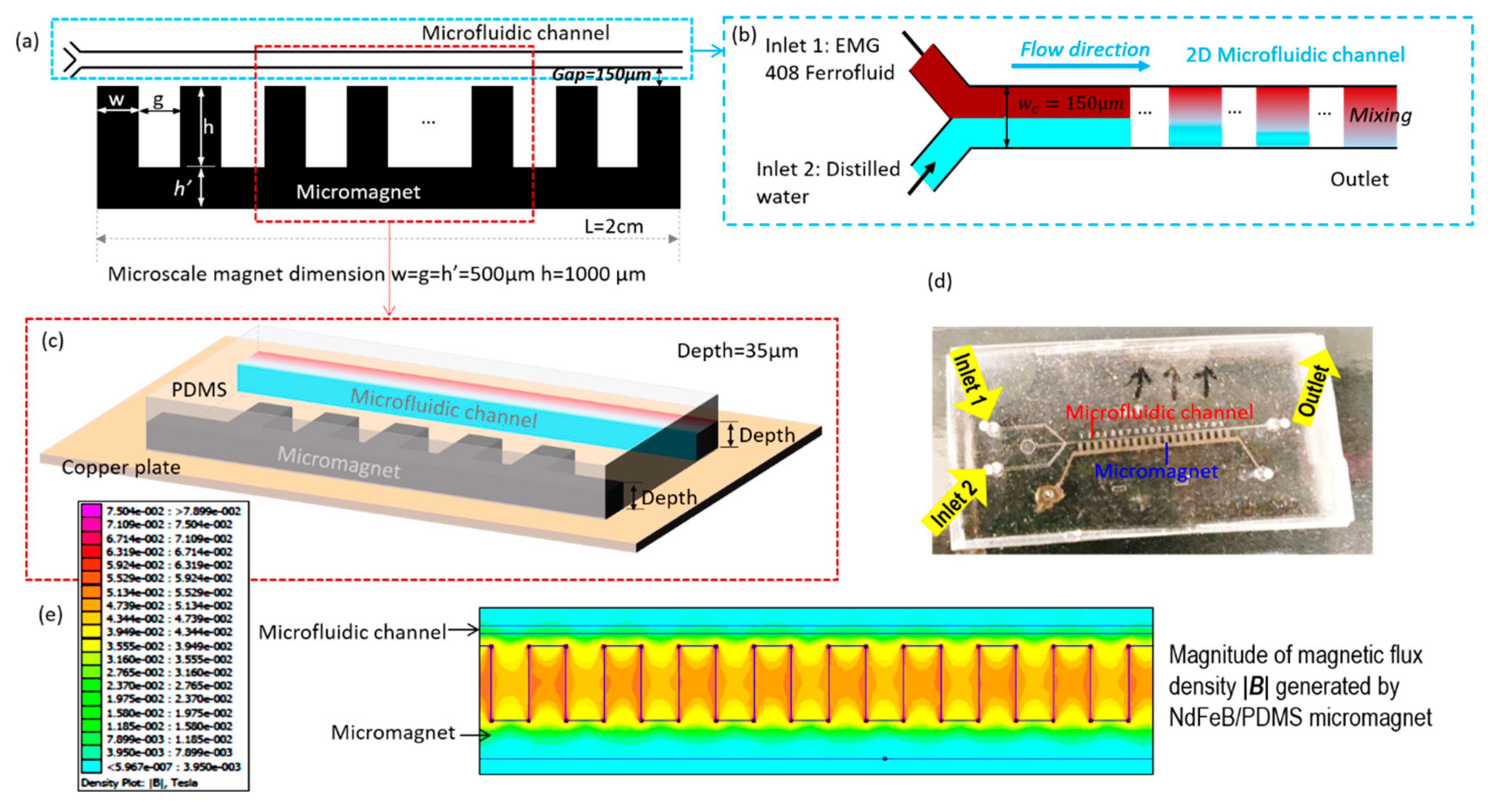
2.1. Work Concept
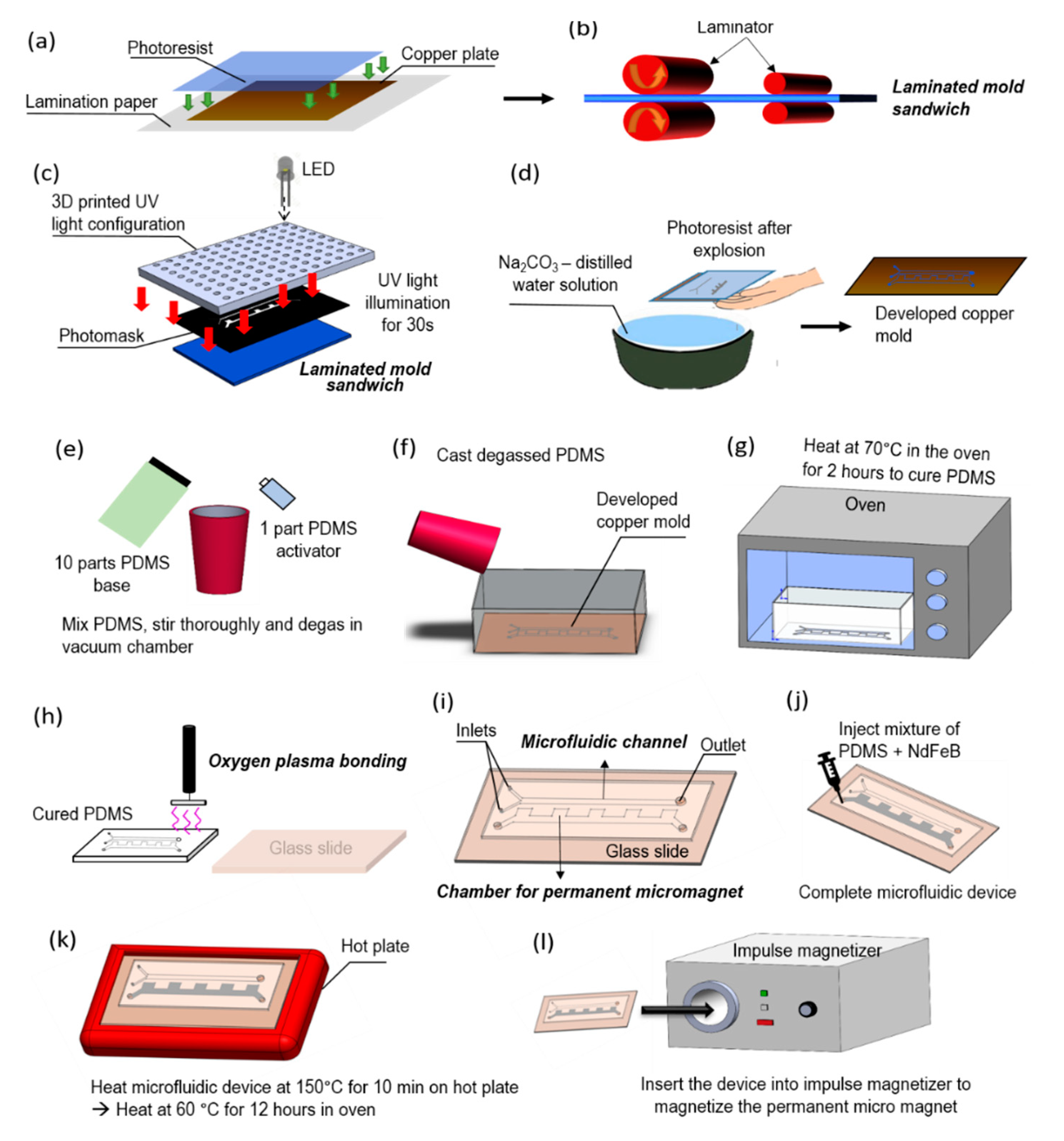
2.2. Microfluidic Device Fabrication
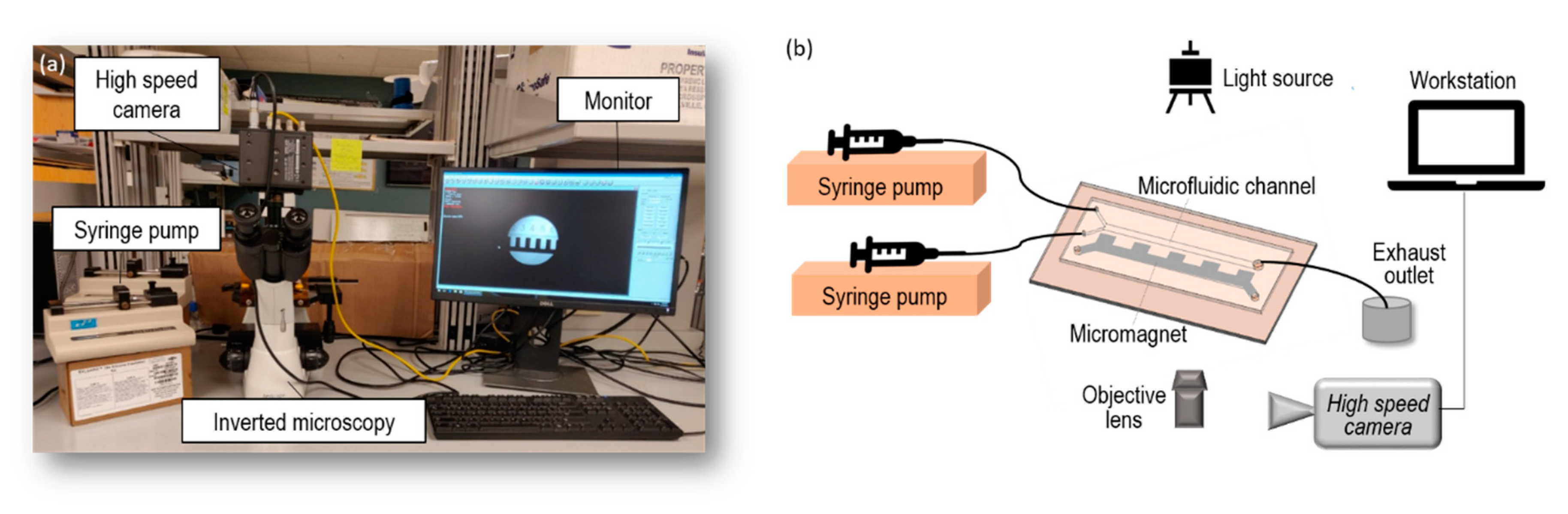
2.3. Experiment Setup and Materials
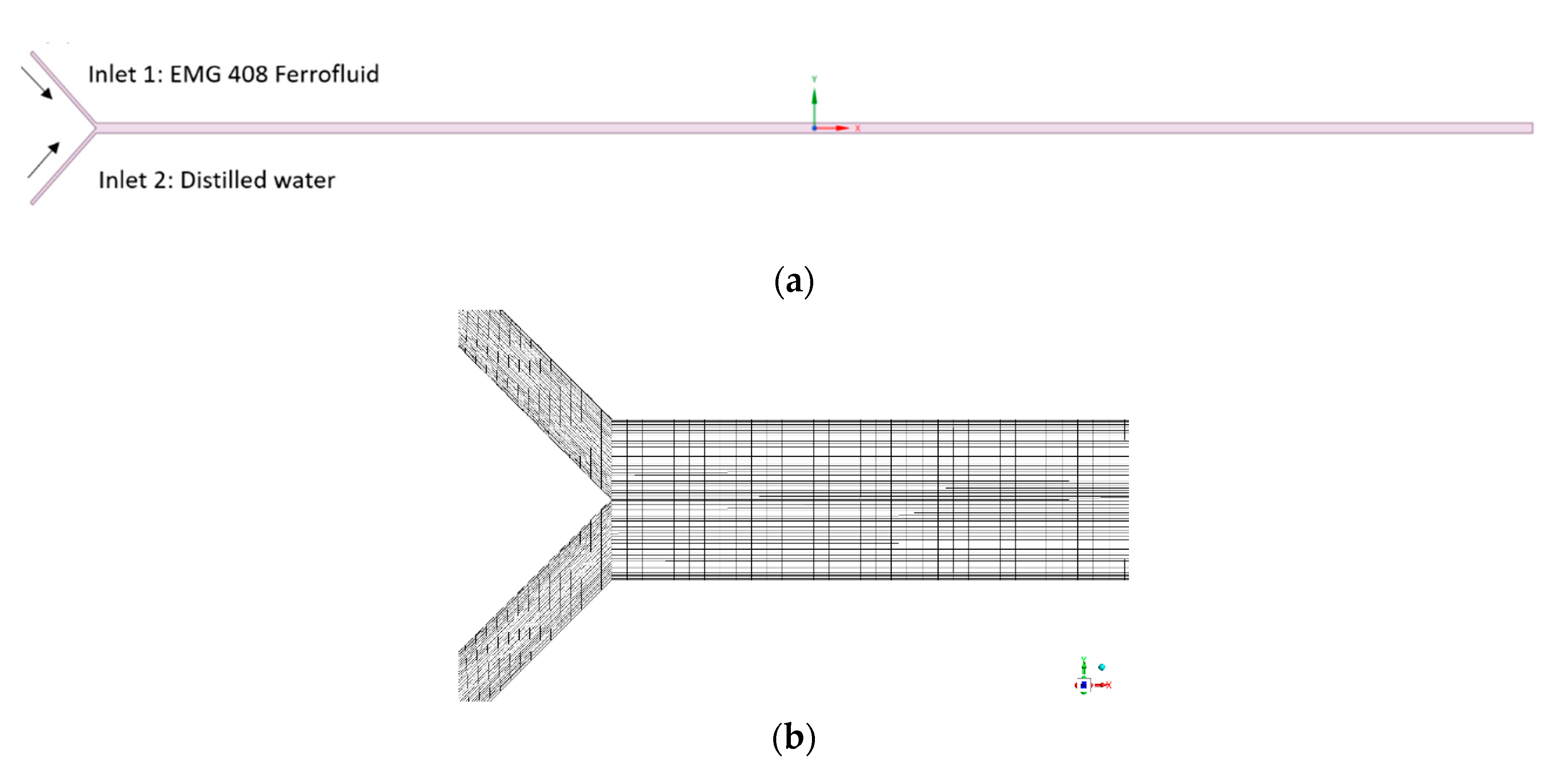
3. Numerical Simulation
4. Results and Discussion
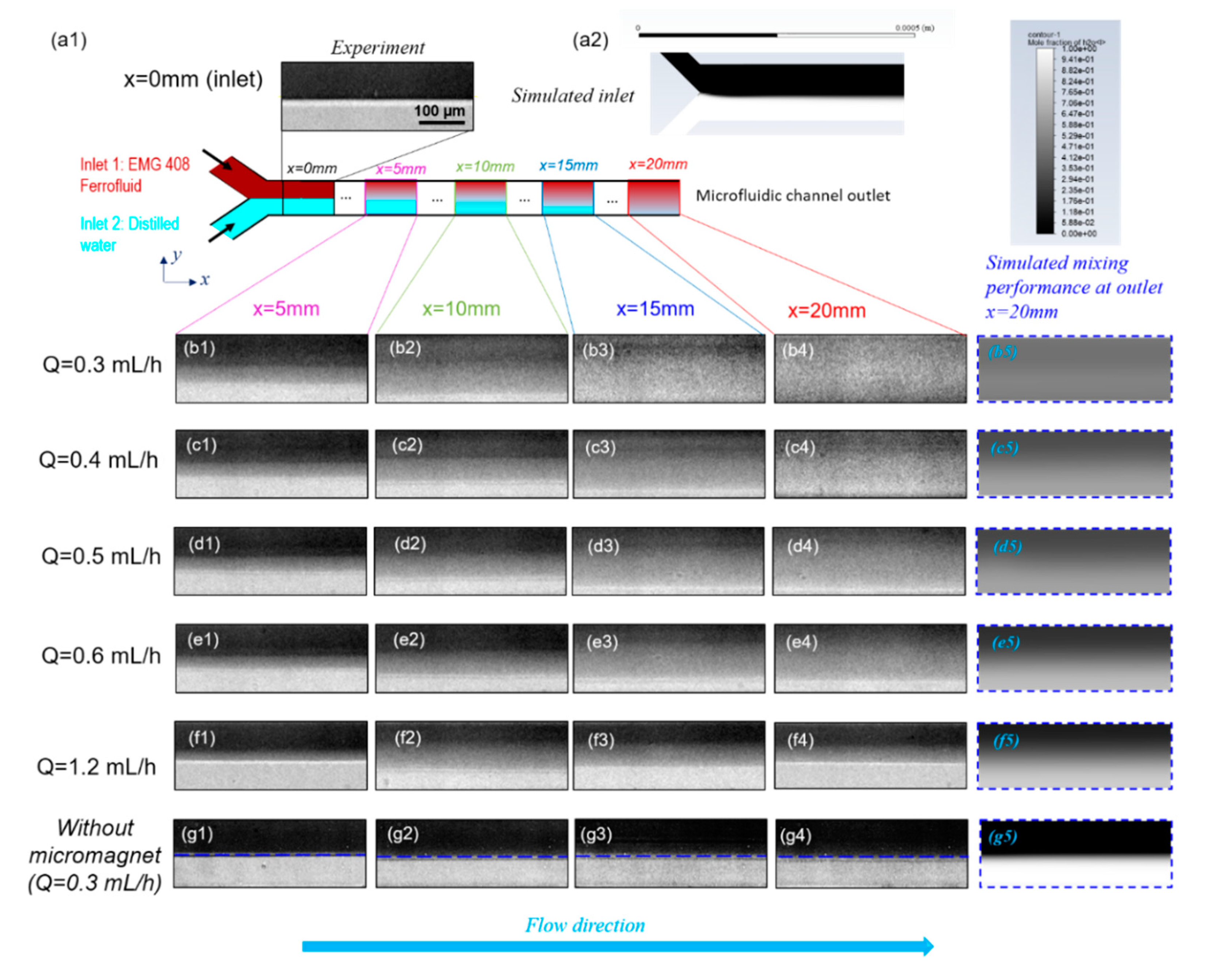
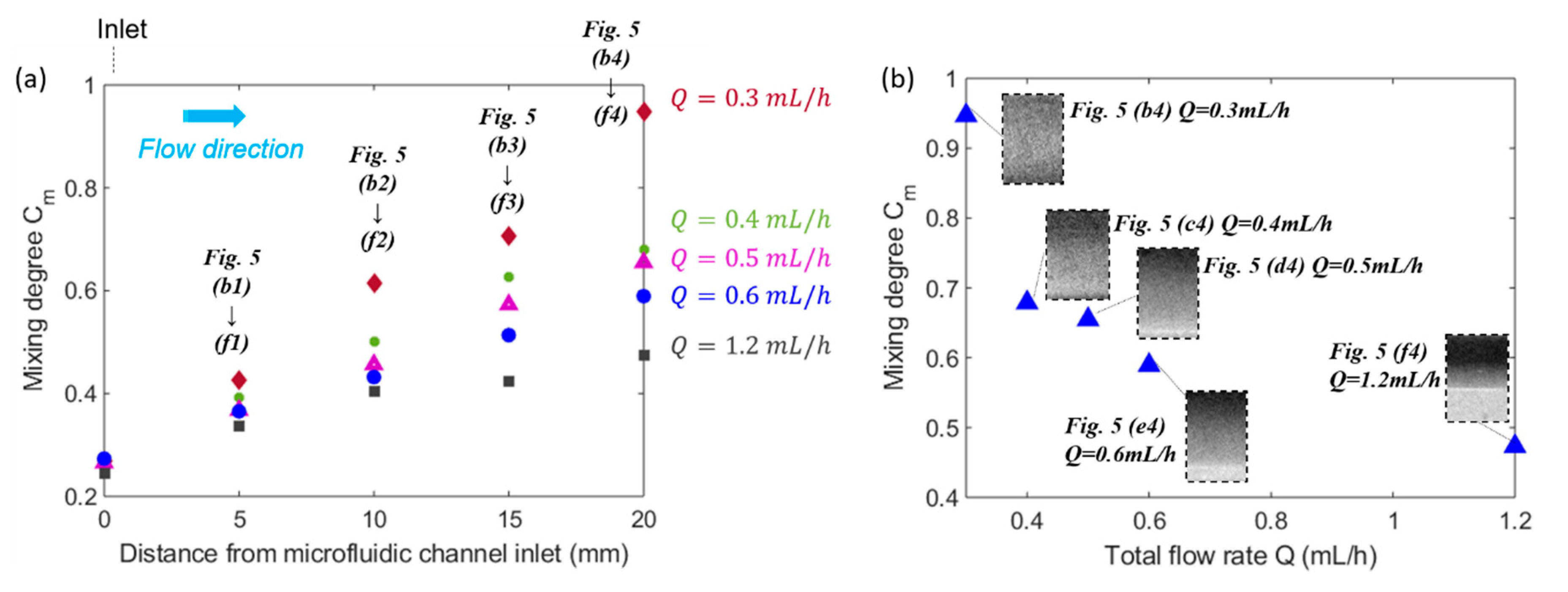
4.1. Effect of Total Flow Rate on Mixing Efficiency
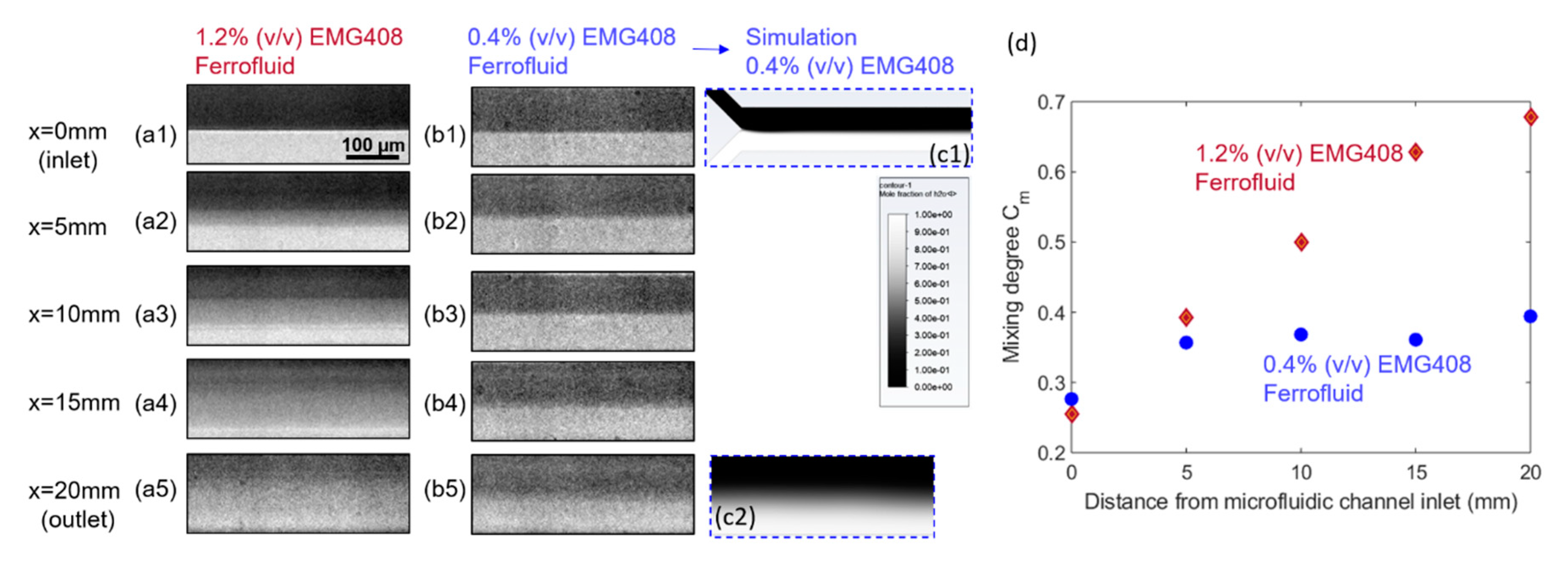
4.2. Effect of Ferrofluid Concentration on the Mixing Efficiency
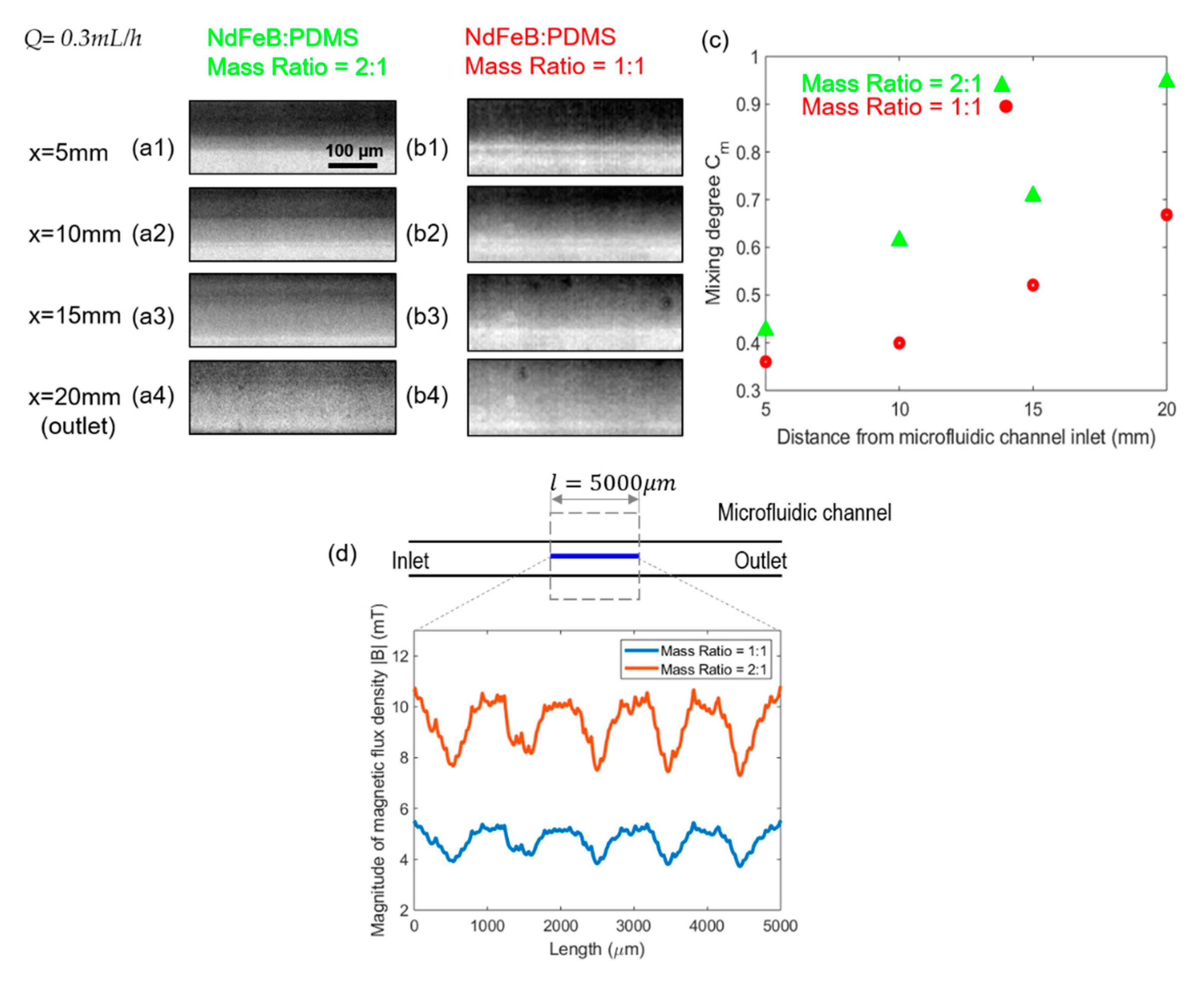
4.3. Effect of Micromagnet Mass Ratio of NdFeB:PDMS on the Mixing Efficiency
5. Conclusions
- (1)
- A 2-D steady-state CFD model was developed to simulate laminar flow of fluids and their mixing behavior in a micromixer channel, with and without the effect of externally imposed magnetic field. The magnetic field generated by the array of fabricated magnets were calculated in prior with an open-source package of FEMM, which was then imported in the CFD model for coupled simulations of fluid flow and mixing in the microchannel. With the numerical simulation results matching closely with experimental measurements, this modeling workflow is validated.
- (2)
- By decreasing the total flow rate, the residence time increased, and the ferrofluid and distilled water had longer time to mix thoroughly with each other inside the microfluidic channel.
- (3)
- As the ferrofluid concentration and the strength of the magnet increased, the mixing efficiency also increased due to the stronger magnetic force. These results show that the mixing in the microfluidic channel can be done with the help of a magnet without increasing the length of the channel.
- (4)
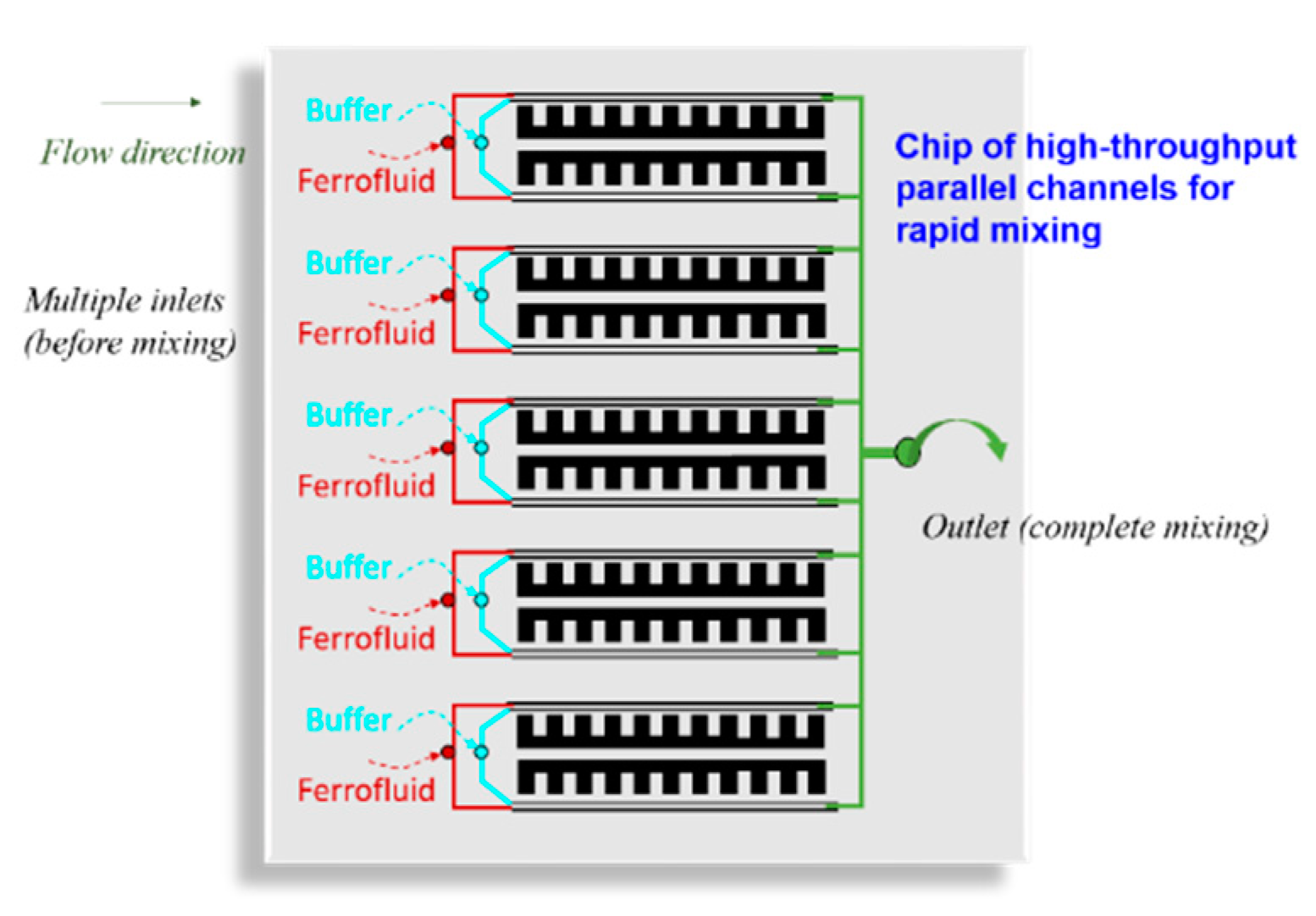
- The simple yet powerful technique proposed in this work significantly reduces the size of the integrated device and is obviously less expensive fabrication approach. In the meanwhile, the microscale permanent magnets can also be easily adapted to high throughput systems as shown in Figure 9.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Whitesides, G.M. The origins and the future of microfluidics. Nature 2006, 442, 368. [Google Scholar] [CrossRef] [PubMed]
- Mansur, E.A.; Mingxing, Y.E.; Yundong, W.A.N.G.; Youyuan, D.A.I. A state-of-the-art review of mixing in microfluidic mixers. Chin. J. Chem. Eng. 2008, 16, 503–516. [Google Scholar] [CrossRef]
- Liu, W.; Kim, H.J.; Lucchetta, E.M.; Du, W.; Ismagilov, R.F. Isolation, incubation, and parallel functional testing and identification by FISH of rare microbial single-copy cells from multi-species mixtures using the combination of chemistrode and stochastic confinement. Lab A Chip 2009, 9, 2153–2162. [Google Scholar] [CrossRef] [PubMed]
- Volfson, D.; Cookson, S.; Hasty, J.; Tsimring, L.S. Biomechanical ordering of dense cell populations. Proc. Natl. Acad. Sci. USA 2008, 105, 15346–15351. [Google Scholar] [CrossRef]
- Brouzes, E.; Medkova, M.; Savenelli, N.; Marran, D.; Twardowski, M.; Hutchison, J.B.; Rothberg, J.M.; Link, D.R.; Perrimon, N.; Samuels, M.L. Droplet microfluidic technology for single-cell high-throughput screening. Proc. Natl. Acad. Sci. USA 2009, 106, 14195–14200. [Google Scholar] [CrossRef]
- Ainslie, K.M.; Kraning, C.M.; Desai, T.A. Microfabrication of an asymmetric, multi-layered microdevice for controlled release of orally delivered therapeutics. Lab Chip 2008, 8, 1042–1047. [Google Scholar] [CrossRef]
- Kastrup, C.J.; Boedicker, J.Q.; Pomerantsev, A.P.; Moayeri, M.; Bian, Y.; Pompano, R.R.; Kline, T.R.; Sylvestre, P.; Shen, F.; Leppla, S.H.; et al. Spatial localization of bacteria controls coagulation of human blood by ‘quorum acting’. Nat. Chem. Biol. 2008, 4, 742. [Google Scholar] [CrossRef]
- Lee, H.; Sun, E.; Ham, D.; Weissleder, R. Chip–NMR biosensor for detection and molecular analysis of cells. Nat. Med. 2008, 14, 869. [Google Scholar] [CrossRef]
- Jeong, G.S.; Chung, S.; Kim, C.B.; Lee, S.H. Applications of micromixing technology. Analyst 2010, 135, 460–473. [Google Scholar] [CrossRef]
- Fu, L.M.; Tsai, C.H.; Leong, K.P.; Wen, C.Y. Rapid micromixer via ferrofluids. Phys. Procedia 2010, 9, 270–273. [Google Scholar] [CrossRef]
- Groisman, A.; Steinberg, V. Efficient mixing at low Reynolds numbers using polymer additives. Nature 2001, 410, 905. [Google Scholar] [CrossRef]
- Valencia, P.M.; Basto, P.A.; Zhang, L.; Rhee, M.; Langer, R.; Farokhzad, O.C.; Karnik, R. Single-step assembly of homogenous lipid−polymeric and lipid−quantum dot nanoparticles enabled by microfluidic rapid mixing. ACS Nano 2010, 4, 1671–1679. [Google Scholar] [CrossRef] [PubMed]
- Johnson, T.J.; Ross, D.; Locascio, L.E. Rapid microfluidic mixing. Anal. Chem. 2002, 74, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.Y.; Chang, C.L.; Wang, Y.N.; Fu, L.M. Microfluidic mixing: A review. Int. J. Mol. Sci. 2011, 12, 3263–3287. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, N.T.; Wu, Z. Micromixers—A review. J. Micromech. Microeng. 2004, 15, R1. [Google Scholar] [CrossRef]
- Meijer, H.E.; Singh, M.K.; Kang, T.G.; Den Toonder, J.M.; Anderson, P.D. Passive and active mixing in microfluidic devices. In Macromolecular Symposia; WILEY-VCH Verlag: Weinheim, Germany, 2009; Volume 279, pp. 201–209. [Google Scholar]
- Deshmukh, A.A.; Liepmann, D.; Pisano, A.P. Continuous micromixer with pulsatile micropumps. In Proceedings of the Technical Digest of the IEEE Solid State Sensor and Actuator Workshop, Hilton Head Island, SC, USA, 13–16 June 2000; Volume 736. [Google Scholar]
- Fujii, T.; Sando, Y.; Higashino, K.; Fujii, Y. A plug and play microfluidic device. Lab Chip 2003, 3, 193–197. [Google Scholar] [CrossRef]
- El Moctar, A.O.; Aubry, N.; Batton, J. Electro-hydrodynamic micro-fluidic mixer. Lab Chip 2003, 3, 273–280. [Google Scholar] [CrossRef]
- Samiei, E.; de Leon Derby, M.D.; Van den Berg, A.; Hoorfar, M. An electrohydrodynamic technique for rapid mixing in stationary droplets on digital microfluidic platforms. Lab Chip 2017, 17, 227–234. [Google Scholar] [CrossRef]
- Ober, T.J.; Foresti, D.; Lewis, J.A. Active mixing of complex fluids at the microscale. Proc. Natl. Acad. Sci. USA 2015, 112, 12293–12298. [Google Scholar] [CrossRef]
- Lu, L.H.; Ryu, K.S.; Liu, C. A magnetic microstirrer and array for microfluidic mixing. J. Microelectromech. Syst. 2002, 11, 462–469. [Google Scholar]
- Mao, L.; Koser, H. Towards ferrofluidics for μ-TAS and lab on-a-chip applications. Nanotechnology 2006, 17, S34. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zolgharni, M.; Azimi, S.M.; Bahmanyar, M.R.; Balachandran, W. A microfluidic mixer for chaotic mixing of magnetic particles. In Proceedings of the 2007 NSTI Nanotechnology Conference and Trade Show, Santa Clara, CA, USA, 20–24 May 2007; pp. 20–24. [Google Scholar]
- Oh, D.W.; Jin, J.S.; Choi, J.H.; Kim, H.Y.; Lee, J.S. A microfluidic chaotic mixer using ferrofluid. J. Micromech. Microeng. 2007, 17, 2077. [Google Scholar] [CrossRef]
- Mao, L.; Koser, H. Overcoming the diffusion barrier: Ultra-fast micro-scale mixing via ferrofluids. In Proceedings of the TRANSDUCERS 2007–2007 International Solid-State Sensors, Actuators and Microsystems Conference, Lyon, France, 10–14 June 2007; IEEE: Piscataway, NJ, USA, 2007; pp. 1829–1832. [Google Scholar]
- Nouri, D.; Zabihi-Hesari, A.; Passandideh-Fard, M. Rapid mixing in micromixers using magnetic field. Sens. Actuators A Phys. 2017, 255, 79–86. [Google Scholar] [CrossRef]
- Meeker, D. FEMM 4.2. User’s Manual; University of Virginia: Charlottesville, VA, USA, 2009. [Google Scholar]
- Zhou, R.; Yang, Q.; Bai, F.; Werner, J.A.; Shi, H.; Ma, Y.; Wang, C. Fabrication and integration of microscale permanent magnets for particle separation in microfluidics. Microfluid. Nanofluid. 2016, 20, 110. [Google Scholar] [CrossRef]
- Zhang, X.; Stefanick, S.; Villani, F.J. Application of microreactor technology in process development. Org. Process Res. Dev. 2004, 8, 455–460. [Google Scholar] [CrossRef]
- Wen, C.Y.; Liang, K.P.; Chen, H.; Fu, L.M. Numerical analysis of a rapid magnetic microfluidic mixer. Electrophoresis 2011, 32, 3268–3276. [Google Scholar] [CrossRef]
- Hashmi, A.; Xu, J. On the quantification of mixing in microfluidics. J. Lab. Autom. 2014, 19, 488–491. [Google Scholar] [CrossRef]
- Ma, Y.; Sun, C.P.; Fields, M.; Li, Y.; Haake, D.A.; Churchill, B.M.; Ho, C.M. An unsteady microfluidic T-form mixer perturbed by hydrodynamic pressure. J. Micromech. Microeng. 2008, 18. [Google Scholar] [CrossRef]
- Abràmoff, M.D.; Magalhães, P.J.; Ram, S.J. Image processing with ImageJ. Biophotonics Int. 2004, 11, 36–42. [Google Scholar]
- Wen, C.Y.; Yeh, C.P.; Tsai, C.H.; Fu, L.M. Rapid magnetic microfluidic mixer utilizing AC electromagnetic field. Electrophoresis 2009, 30, 4179–4186. [Google Scholar] [CrossRef]
- Izadi, D.; Nguyen, T.; Lapidus, L. Complete Procedure for Fabrication of a Fused Silica Ultrarapid Microfluidic Mixer Used in Biophysical Measurements. Micromachines 2017, 8, 16. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, R.; Surendran, A.N.; Mejulu, M.; Lin, Y. Rapid Microfluidic Mixer Based on Ferrofluid and Integrated Microscale NdFeB-PDMS Magnet. Micromachines 2020, 11, 29. https://doi.org/10.3390/mi11010029
Zhou R, Surendran AN, Mejulu M, Lin Y. Rapid Microfluidic Mixer Based on Ferrofluid and Integrated Microscale NdFeB-PDMS Magnet. Micromachines. 2020; 11(1):29. https://doi.org/10.3390/mi11010029
Chicago/Turabian StyleZhou, Ran, Athira N. Surendran, Marcel Mejulu, and Yang Lin. 2020. "Rapid Microfluidic Mixer Based on Ferrofluid and Integrated Microscale NdFeB-PDMS Magnet" Micromachines 11, no. 1: 29. https://doi.org/10.3390/mi11010029
APA StyleZhou, R., Surendran, A. N., Mejulu, M., & Lin, Y. (2020). Rapid Microfluidic Mixer Based on Ferrofluid and Integrated Microscale NdFeB-PDMS Magnet. Micromachines, 11(1), 29. https://doi.org/10.3390/mi11010029





