Hemostasis-On-a-Chip: Impedance Spectroscopy Meets Microfluidics for Hemostasis Evaluation
Abstract
1. Introduction
2. Materials and Methods
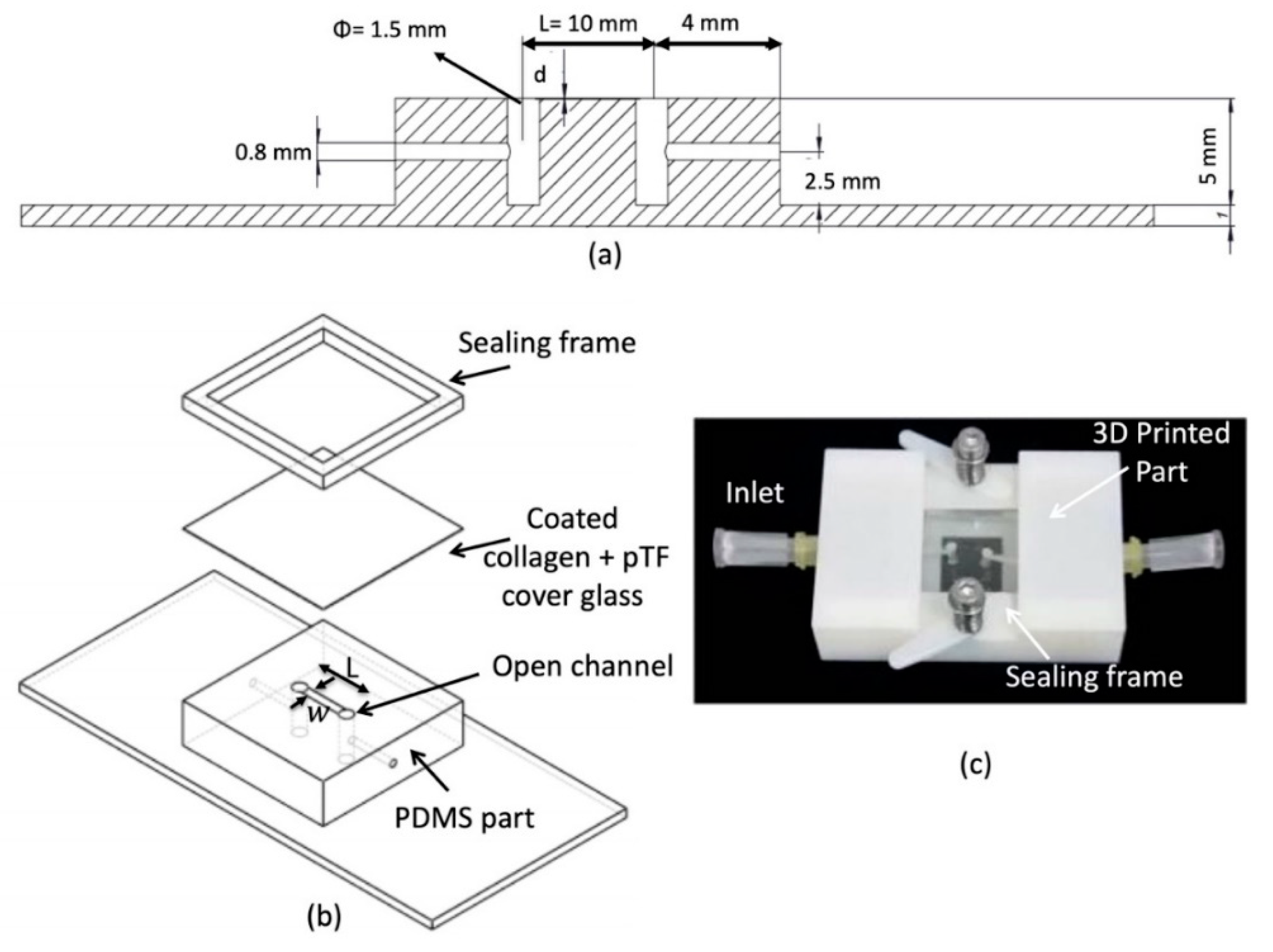
2.1. Microflow Chamber Design and Manufacturing
2.2. Biomimetic Coatings and Sample Preparation
2.3. Flow Assays
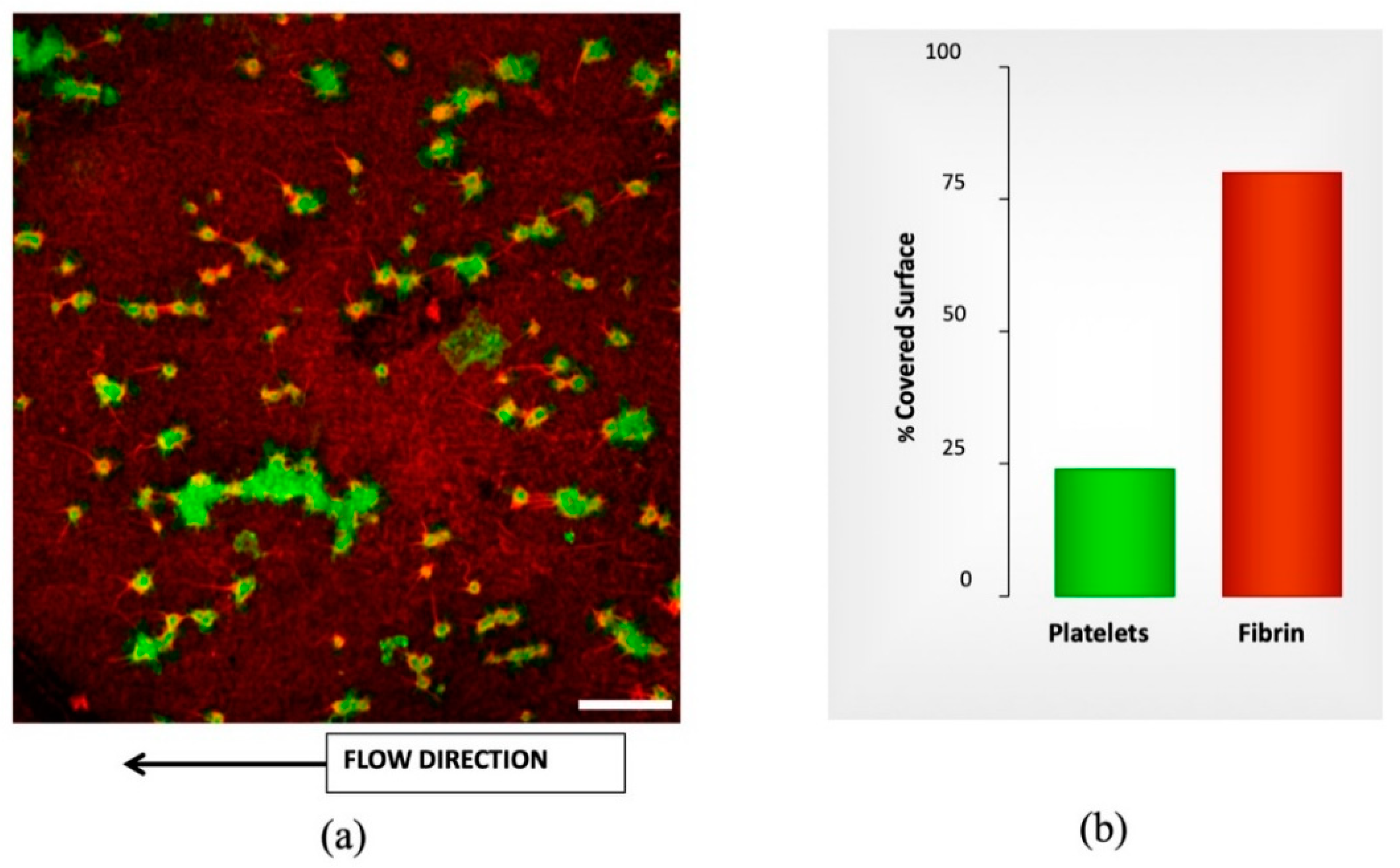
2.4. Image Capture and Analysis
2.5. Impedance Characterization
- For low frequencies , dominates the impedance measured.
- For frequencies , is the dominant impedance.
- For frequencies , is the dominant impedance.
3. Results
3.1. Flow Assay Results
3.2. Impedance Spectroscopy Assay Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Neeves, K.B.; Onasoga, A.A.; Wufsus, A.R. The use of microfluidics in hemostasis: Clinical diagnostics and biomimetic models of vascular injury. Curr. Opin. Hematol. 2013, 20, 417–423. [Google Scholar] [CrossRef] [PubMed]
- Shen, F.; Kastrup, C.J.; Liu, Y.; Ismagilov, R.F. Threshold Response of Initiation of Blood Coagulation by Tissue Factor in Patterned Microfluidic Capillaries Is Controlled by Shear Rate. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 2035–2041. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Ku, D.N.; Forest, C.R. Microfluidic system for simultaneous optical measurement of platelet aggregation at multiple shear rates in whole blood. Lab Chip 2012, 12, 1355. [Google Scholar] [CrossRef] [PubMed]
- Michelson, A.D. Methods for the measurement of platelet function. Am. J. Cardiol. 2009, 103, 20A–26A. [Google Scholar] [CrossRef] [PubMed]
- Sakariassen, K.S.; Houdijk, W.P.; Sixma, J.J.; Aarts, P.A.; de Groot, P.G. A perfusion chamber developed to investigate platelet interaction in flowing blood with human vessel wall cells, their extracellular matrix, and purified components. J. Lab Clin. Med. 1983, 102, 522–535. [Google Scholar] [PubMed]
- Escolar, G.; Mazzara, R.; Castillo, R.; Ordinas, A. The role of the Baumgartner technique in transfusion medicine: Research and clinical applications. Transfusion 1994, 34, 542–549. [Google Scholar] [CrossRef]
- Nagy, M.; Heemskerk, J.W.; Swieringa, F. Use of microfluidics to assess the platelet-based control of coagulation. Platelets 2017, 28, 441–448. [Google Scholar] [CrossRef] [PubMed]
- Onasoga-Jarvis, A.A.; Leiderman, K.; Fogelson, A.L.; Wang, M.; Manco-Johnson, M.J.; Di Paola, J.A.; Neeves, K.B. The Effect of Factor VIII Deficiencies and Replacement and Bypass Therapies on Thrombus Formation under Venous Flow Conditions in Microfluidic and Computational Models. PLoS ONE 2013, 8, e78732. [Google Scholar] [CrossRef]
- Fedosov, D.A.; Dao, M.; Karniadakis, G.E.; Suresh, S. Computational Biorheology of Human Blood Flow in Health and Disease. Ann. Biomed. Eng. 2014, 42, 368–387. [Google Scholar] [CrossRef]
- Jain, A.; van der Meer, A.D.; Papa, A.L.; Barrile, R.; Lai, A.; Schlechter, B.L.; Otieno, M.A.; Louden, C.S.; Hamilton, G.A.; Frelinger, A.L.; et al. Assessment of whole blood thrombosis in a microfluidic device lined by fixed human endothelium. Biomed. Microdevices 2016, 18, 73. [Google Scholar] [CrossRef]
- Zhang, Y.S.; Davoudi, F.; Walch, P.; Manbachi, A.; Luo, X.; Dell’Erba, V.; Miri, A.K.; Albadawi, H.; Arneri, A.; Wang, X.; et al. Bioprinted thrombosis-on-a-chip. Lab Chip 2016, 16, 4097–4105. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.; Graveline, A.; Waterhouse, A.; Vernet, A.; Flaumenhaft, R.; Ingber, D.E. A shear gradient-activated microfluidic device for automated monitoring of whole blood haemostasis and platelet function. Nat. Commun. 2016, 7, 10176. [Google Scholar] [CrossRef] [PubMed]
- Patel, M.R.; Hellkamp, A.S.; Fox, K.A. Point-of-Care Warfarin Monitoring in the ROCKET AF Trial. N. Engl. J. Med. 2016, 374, 785–788. [Google Scholar] [CrossRef] [PubMed]
- Hori, M.; Ohashi, Y.; Pan, G.; Kato, M.; Kajikawa, M. Point-of-Care Device for Warfarin Monitoring Used in the J-ROCKET AF Study. Circ. J. 2016, 80, 1488–1490. [Google Scholar] [PubMed]
- Cattaneo, M. Resistance to antiplatelet drugs: Molecular mechanisms and laboratory detection. J. Thromb. Haemost. 2007, 5, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Lenk, E.; Spannagl, M. Platelet Function Testing—Guided Antiplatelet Therapy. EJIFCC 2014, 24, 90. [Google Scholar]
- Lopez-Vilchez, I.; Tonda, R.; Hernandez, R.M.; Navalon, F.; Diaz-Ricart, M.; Galan, A.M.; Escolar, G. Relative contributions of collagen and tissue factor to thrombus formation on damaged vascular vessels: In-vitro studies with circulating blood. Coron. Artery Dis. 2009, 20, 392–399. [Google Scholar] [CrossRef] [PubMed]
- Papaioannou, T.G.; Stefanadis, C. Vascular Wall shear stress: Basic principles and Methods. Hell. J. Cardiol 2005, 46, 9–15. [Google Scholar]
- Bruus, H. Theoretical Microfluidics; Oxford university press: College Park, MD, USA, 2008; Volume 18, p. 363. [Google Scholar]
- Yeom, E.; Kang, Y.J.; Lee, S.J. Changes in velocity profile according to blood viscosity in a microchannel. Biomicrofluidics 2014, 8, 034110. [Google Scholar] [CrossRef]
- Dak, P.; Ebrahimi, A.; Alam, M.A. Non-Faradaic Impedance Characterization of an Evaporating Droplet for Microfluidic and Biosensing Applications. Lab Chip 2015, 14, 2469–2479. [Google Scholar] [CrossRef]
- Ting, L.; Feghhi, S.; Karchin, A.; Tooley, W.; White, N.J. Clot-On-A-Chip: A Microfluidic Device to Study Platelet Aggregation and Contractility Under Shear. Blood 2013, 122, 2363. [Google Scholar]
- Lam, W.A. Thrombosis-on-a-Chip: A New Way to Model a Complex Process. Blood 2017, 130, SCI-10. [Google Scholar]
- Mallén-Alberdi, M.; Vigués, N.; Mas, J.; Fernández-Sánchez, C.; Baldi, A. Impedance spectral fingerprint of E. coli cells on interdigitated electrodes: A new approach for label free and selective detection. Sens. Bio-Sens. Res. 2016, 7, 100–106. [Google Scholar] [CrossRef]
- Van Duuren, J.B.; Müsken, M.; Karge, B.; Tomasch, J.; Wittmann, C.; Häussler, S.; Brönstrup, M. Use of Single-Frequency Impedance Spectroscopy to Characterize the Growth Dynamics of Biofilm Formation in Pseudomonas aeruginosa. Sci. Rep. 2017, 7, 5223. [Google Scholar] [CrossRef] [PubMed]
- Clausen, C.; Dimaki, M.; Bertelsen, C.; Skands, G.; Rodriguez-Trujillo, R.; Thomsen, J.; Svendsen, W. Bacteria Detection and Differentiation Using Impedance Flow Cytometry. Sensors (Basel) 2018, 18, 3496. [Google Scholar] [CrossRef] [PubMed]
- Eldarrat, A.; High, A.; Kale, G. Influence of sodium chloride content in electrolyte solution on electrochemical impedance measurements of human dentin. Dent. Res. J. (Isfahan) 2017, 14, 25–31. [Google Scholar] [CrossRef]
- Boumya, W.; Laghrib, F.; Lahrich, S.; Farahi, A.; Achak, M.; Bakasse, M.; El Mhammedi, M.A. Electrochemical impedance spectroscopy measurements for determination of derivatized aldehydes in several matrices. Heliyon 2017, 3, e00392. [Google Scholar] [CrossRef] [PubMed]



| [APIX] ng/mL | Platelets | Fibrin |
|---|---|---|
| 0 | 23.0 ± 3.0 | 43.4 ± 4.8 |
| 10 | 17.9 ± 0.9 | 42.1 ± 1.9 |
| 40 | 14.0 ± 5.3 | 23.4 ± 7.7 |
| 160 | 5.4 ± 2.2 *# | 14.1 ± 4.9 *# |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karimi, S.; Farré-Lladós, J.; Mir, E.; Escolar, G.; Casals-Terré, J. Hemostasis-On-a-Chip: Impedance Spectroscopy Meets Microfluidics for Hemostasis Evaluation. Micromachines 2019, 10, 534. https://doi.org/10.3390/mi10080534
Karimi S, Farré-Lladós J, Mir E, Escolar G, Casals-Terré J. Hemostasis-On-a-Chip: Impedance Spectroscopy Meets Microfluidics for Hemostasis Evaluation. Micromachines. 2019; 10(8):534. https://doi.org/10.3390/mi10080534
Chicago/Turabian StyleKarimi, Shadi, Josep Farré-Lladós, Enrique Mir, Ginés Escolar, and Jasmina Casals-Terré. 2019. "Hemostasis-On-a-Chip: Impedance Spectroscopy Meets Microfluidics for Hemostasis Evaluation" Micromachines 10, no. 8: 534. https://doi.org/10.3390/mi10080534
APA StyleKarimi, S., Farré-Lladós, J., Mir, E., Escolar, G., & Casals-Terré, J. (2019). Hemostasis-On-a-Chip: Impedance Spectroscopy Meets Microfluidics for Hemostasis Evaluation. Micromachines, 10(8), 534. https://doi.org/10.3390/mi10080534






