In Vivo Tracking of Tissue Engineered Constructs
Abstract
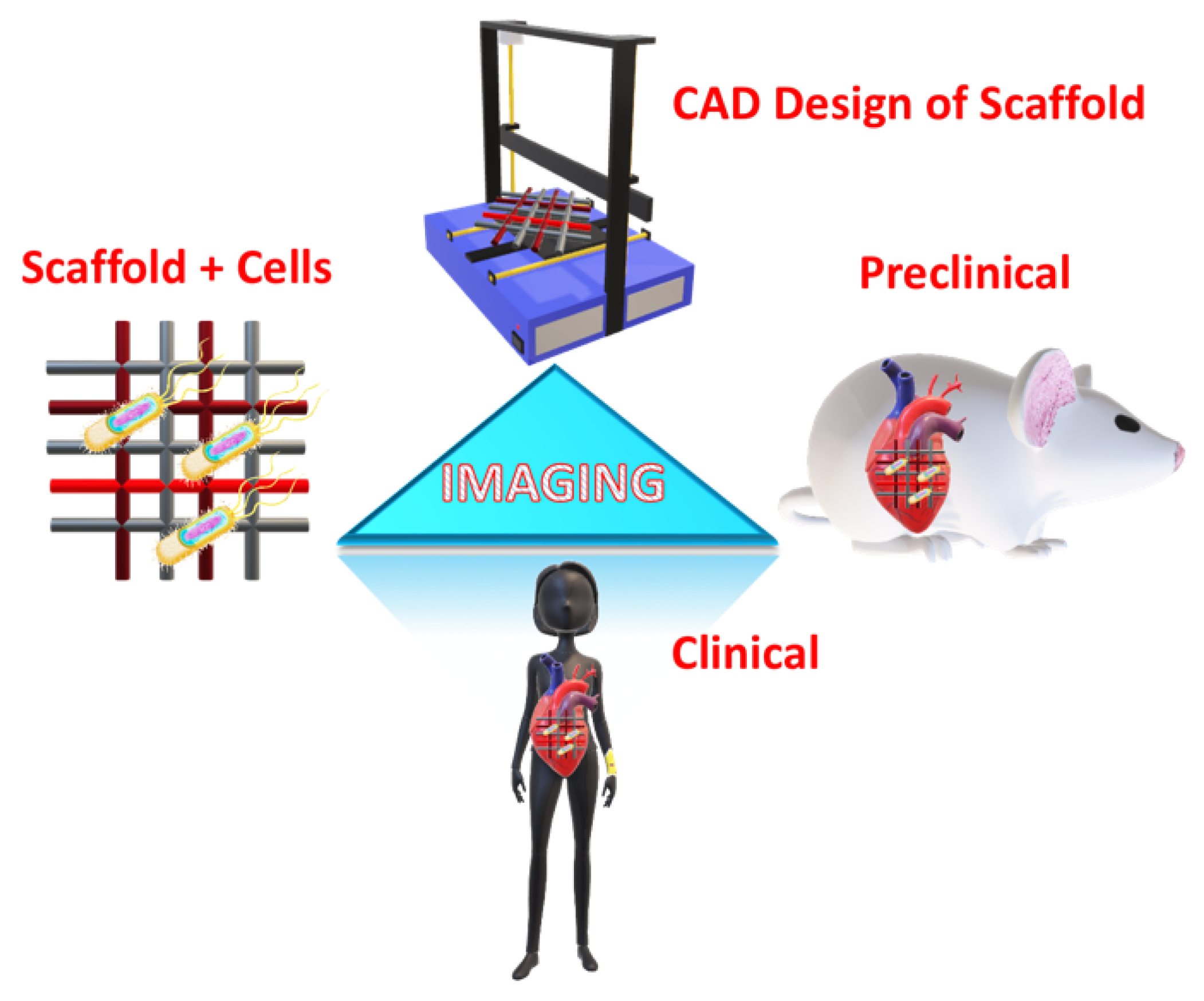
1. Introduction
2. Scaffold Tracking Techniques
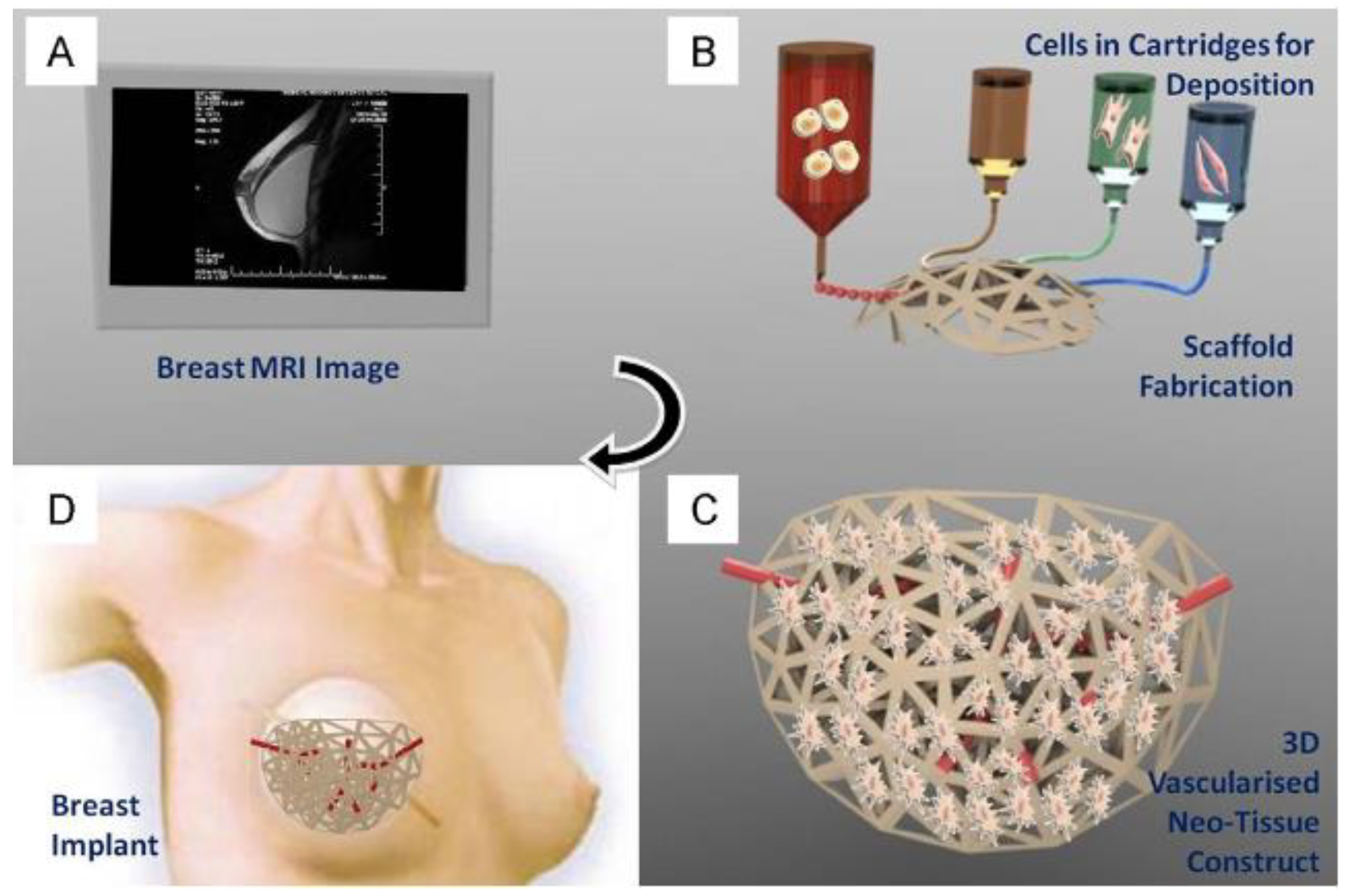
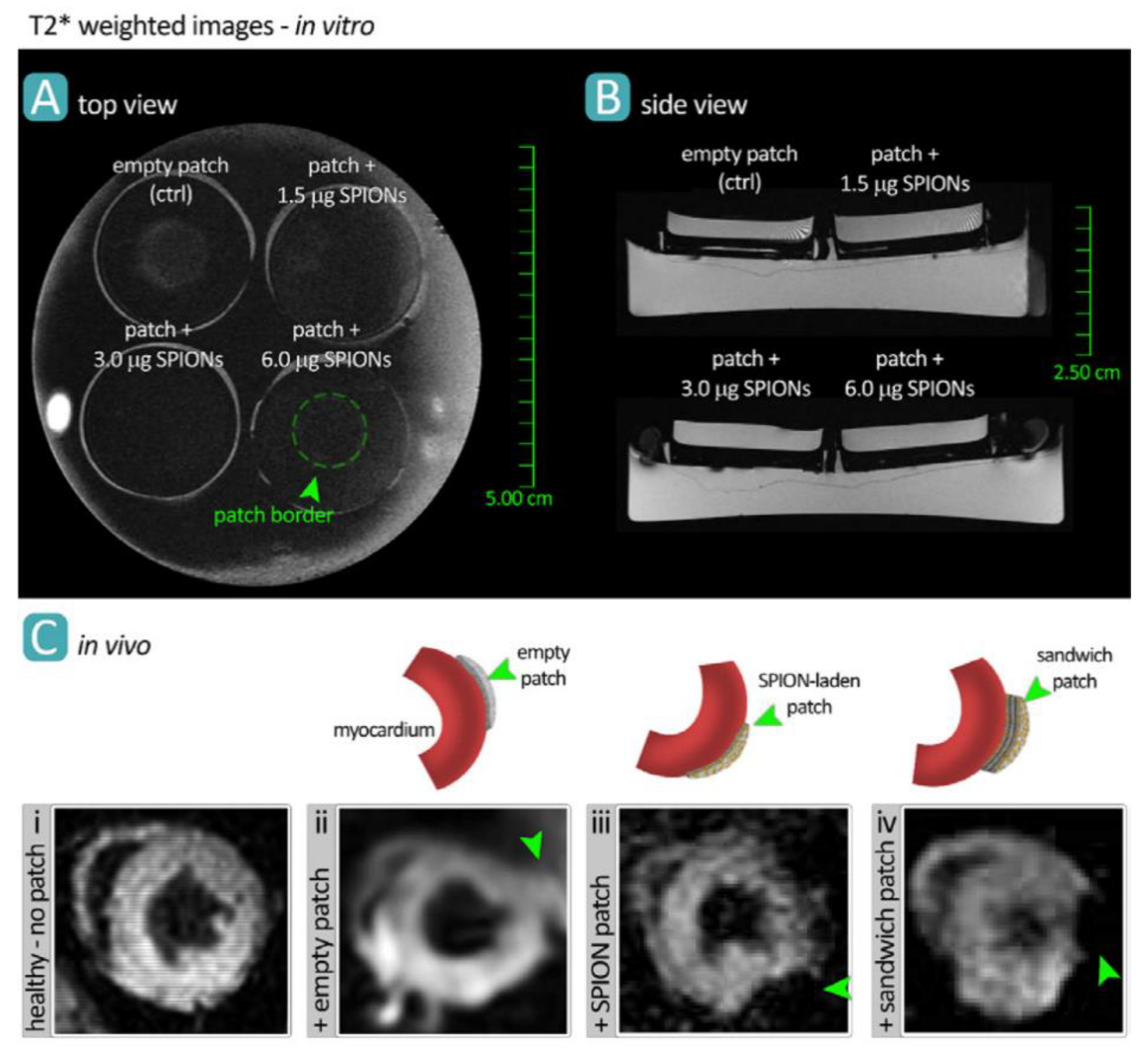
2.1. Magnetic Resonance Imaging (MRI)
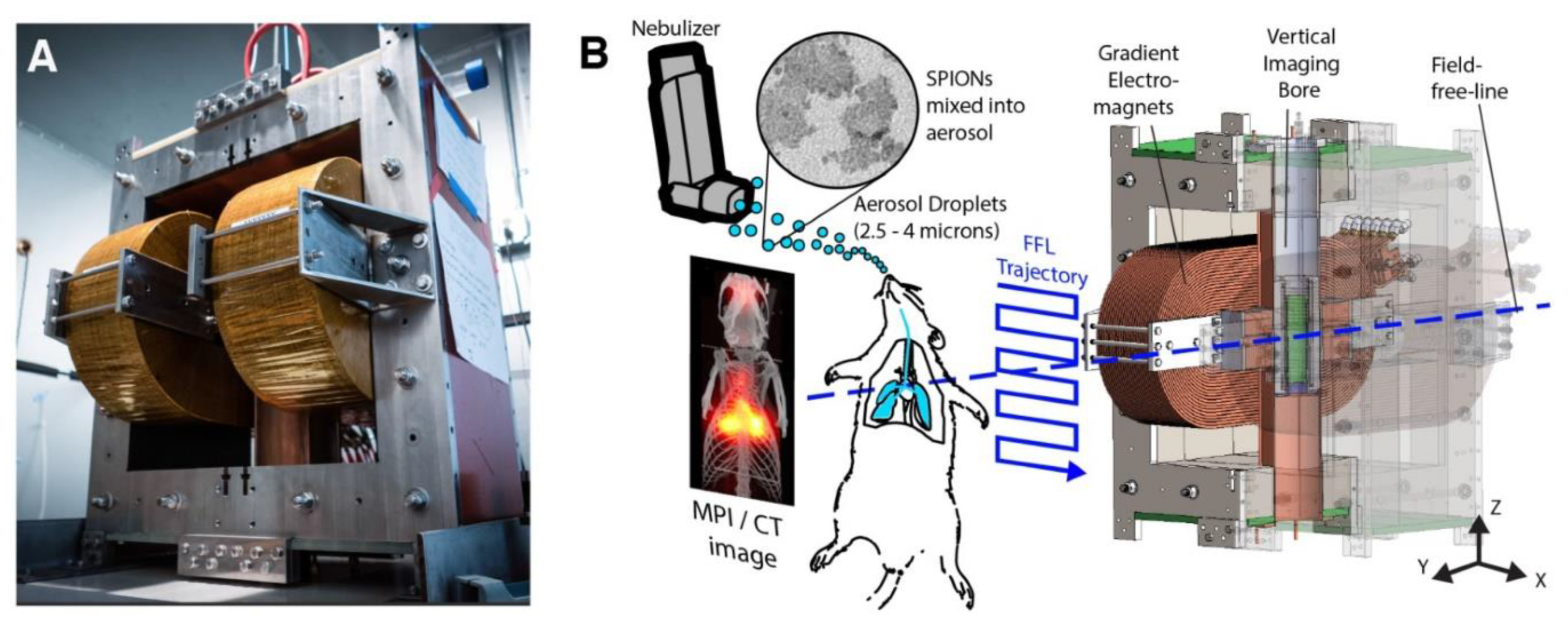
2.2. Magnetic Particle Imaging (MPI)
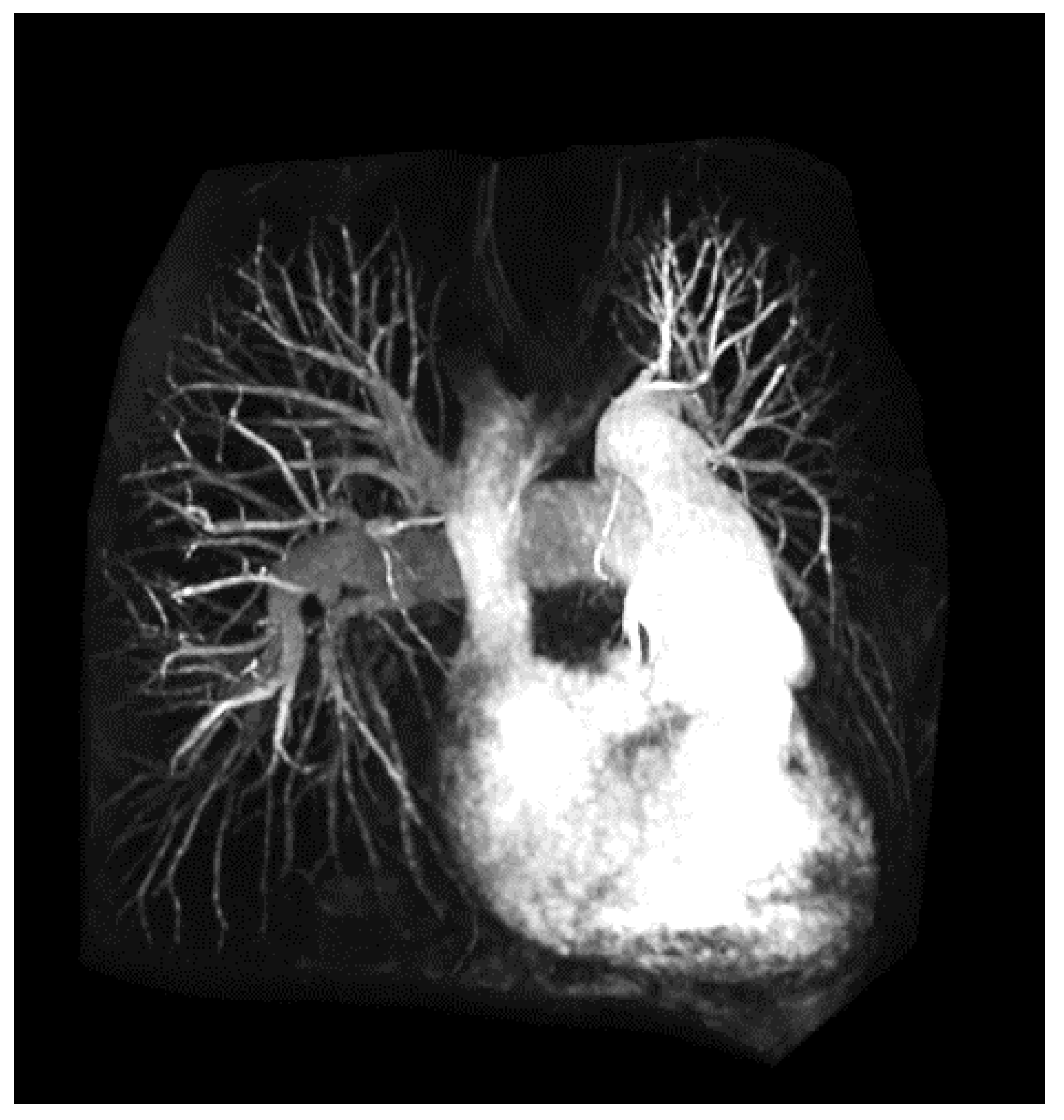
2.3. Angiography
2.4. Computed Tomography (CT)
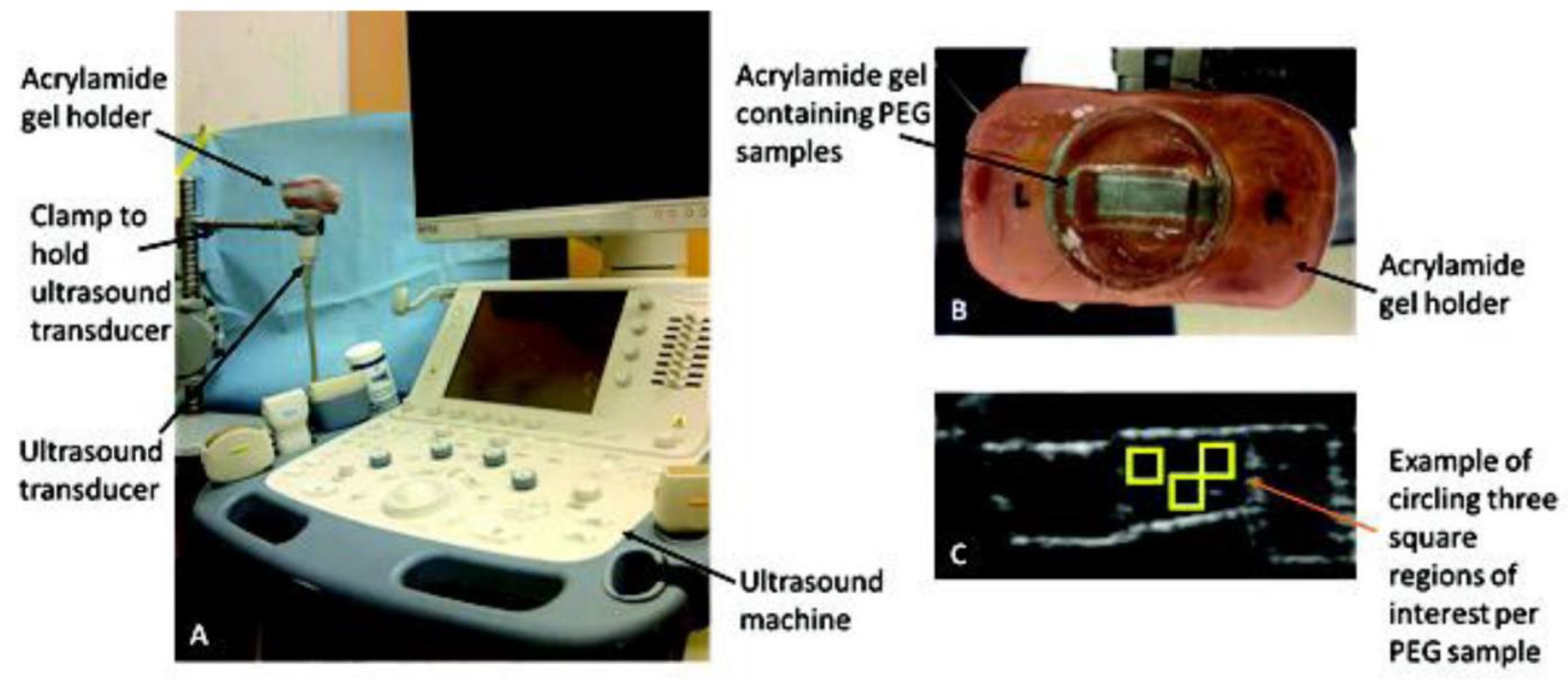
2.5. Ultrasound
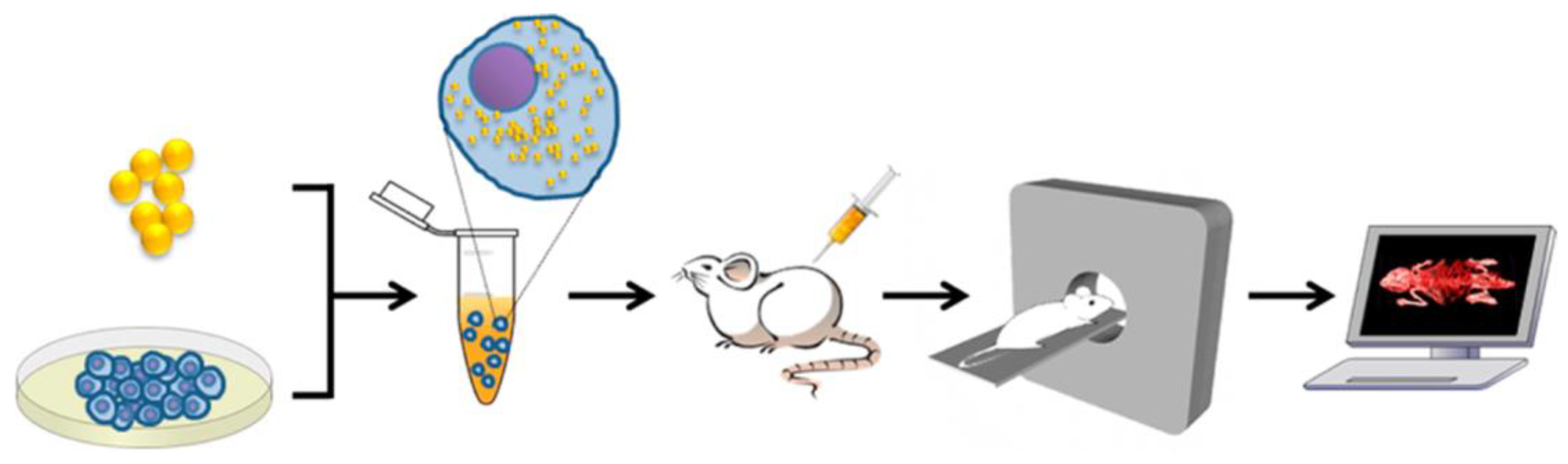
2.6. Bioluminescence
2.7. Fluorescence Spectroscopy
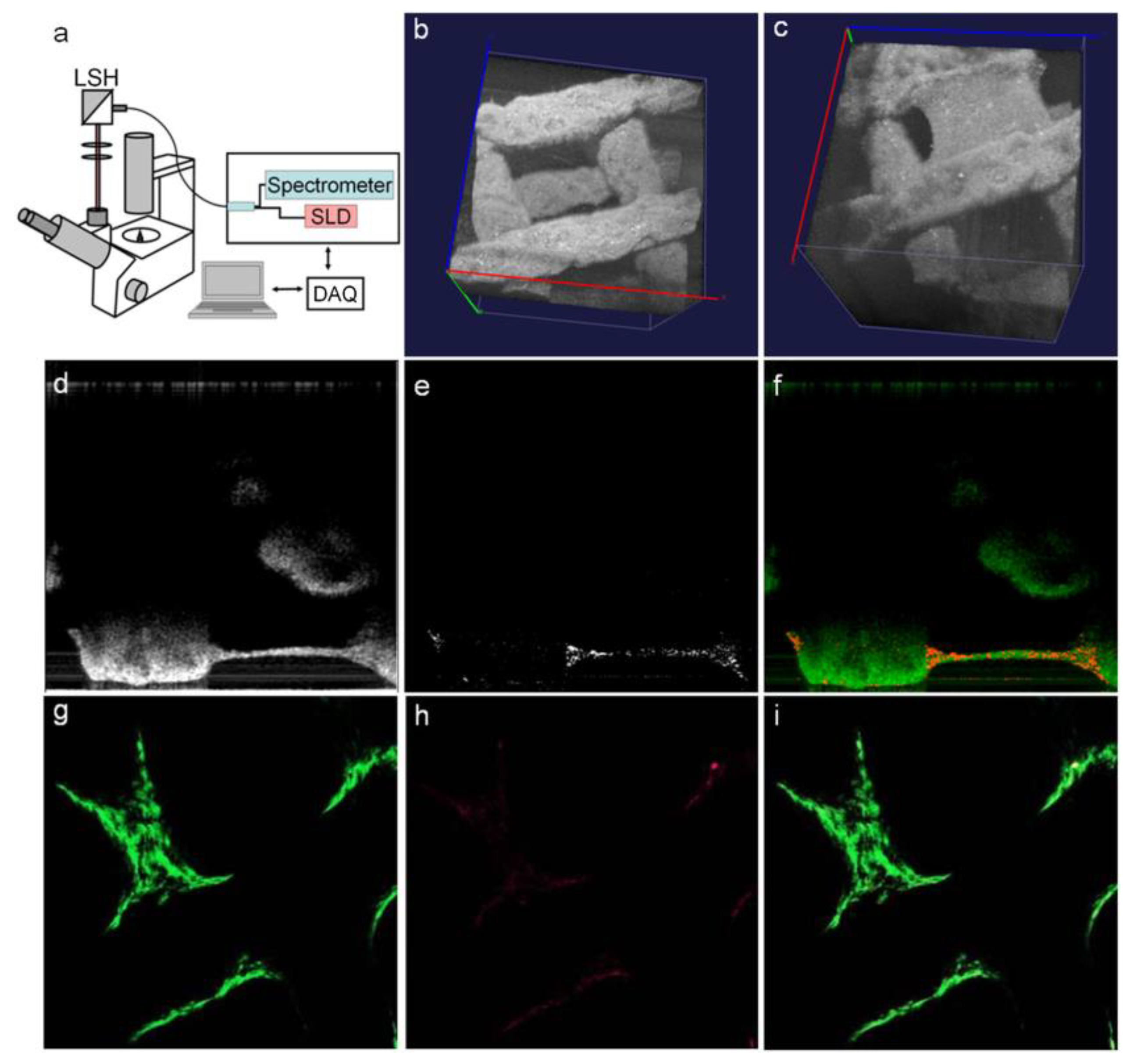
2.8. Optical Coherence Tomography
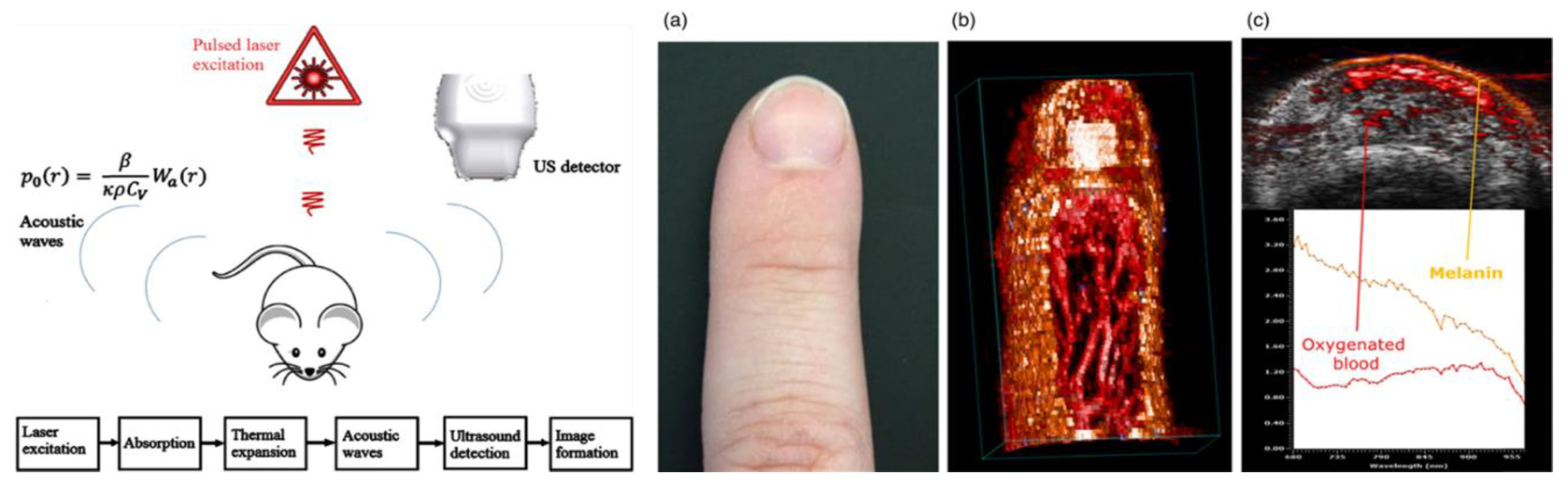
2.9. Photoacoustic Imaging (PAI)
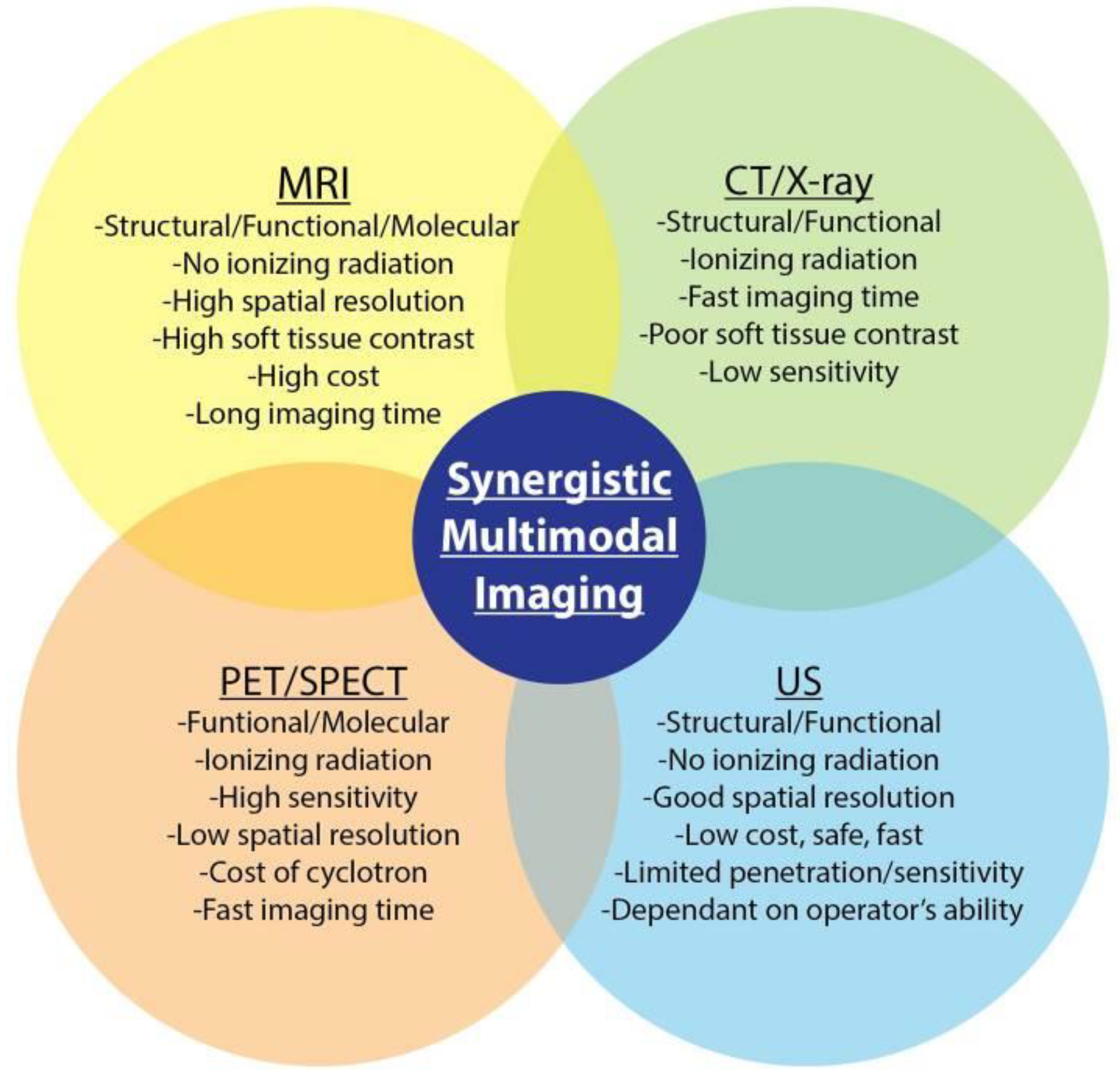
2.10. Multimodal Imaging
3. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zhang, Y.S.; Yao, J. Imaging Biomaterial-Tissue Interactions. Trends Biotechnol. 2015, 40, 1291–1296. [Google Scholar] [CrossRef] [PubMed]
- Khan, F.; Tanaka, M. Designing Smart Biomaterials for Tissue Engineering. Int. J. Mol. Sci. 2018, 19, 17. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.J.; Kasper, F.K.; Mikos, A.G. Biomaterials for Tissue Engineering. Ann. Biomed. Eng. 2014, 42, 323–337. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, Y. Tissue Engineering Applications of Three-Dimensional Bioprinting. Cell Biochem. Biophys. 2015, 72, 777–782. [Google Scholar] [CrossRef] [PubMed]
- Hunsberger, J.; Harrysson, O.; Shirwaiker, R.; Starly, B.; Wysk, R.; Cohen, P.; Allickson, J.; Yoo, J.; Atala, A. Manufacturing Road Map for Tissue Engineering and Regenerative Medicine Technologies. Stem Cells Transl. Med. 2015, 4, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.B.; Tomov, M.L.; Buikema, J.W.; Chen, C.; Mahmoudi, M.; Wu, S.M.; Serpooshan, V. Cardiovascular tissue bioprinting: Physical and chemical processes. Appl. Phys. Rev. 2018, 5, 041106. [Google Scholar] [CrossRef]
- Serpooshan, V.; Mahmoudi, M.; Hu, D.A.; Hu, J.B.; Wu, S.M. Bioengineering cardiac constructs using 3D printing. J. 3D Print. Med. 2017, 1, 123–139. [Google Scholar] [CrossRef]
- Serpooshan, V.; Hu, J.B.; Chirikian, O.; Hu, D.A.; Mahmoudi, M.; Wu, S.M. Chapter 8—4D Printing of Actuating Cardiac Tissue. In 3D Printing Applications in Cardiovascular Medicine; Al’Aref, S.J., Mosadegh, B., Dunham, S., Min, J.K., Eds.; Academic Press: Boston, MA, USA, 2018; pp. 153–162. [Google Scholar]
- Appel, A.A.; Anastasio, M.A.; Larson, J.C.; Brey, E.M. Imaging challenges in biomaterials and tissue engineering. Biomaterials 2013, 34, 6615–6630. [Google Scholar] [CrossRef] [PubMed]
- Nam, S.Y.; Ricles, L.M.; Suggs, L.J.; Emelianov, S.Y. Imaging strategies for tissue engineering applications. Tissue Eng. Part B Rev. 2014, 21, 88–102. [Google Scholar] [CrossRef]
- Teodori, L.; Crupi, A.; Costa, A.; Diaspro, A.; Melzer, S.; Tarnok, A. Three-dimensional imaging technologies: A priority for the advancement of tissue engineering and a challenge for the imaging community. J. Biophotonics 2017, 10, 24–45. [Google Scholar] [CrossRef]
- Stacy, M.R.; Sinusas, A.J. Emerging Imaging Modalities in Regenerative Medicine. Curr. Pathobiol. Rep. 2015, 3, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Willadsen, M.; Chaise, M.; Yarovoy, I.; Zhang, A.Q.; Parashurama, N. Engineering molecular imaging strategies for regenerative medicine. Bioeng. Transl. Med. 2018, 3, 232–255. [Google Scholar] [CrossRef] [PubMed]
- Maniam, S.; Szklaruk, J. Magnetic resonance imaging: Review of imaging techniques and overview of liver imaging. World J. Radiol. 2010, 2, 309–322. [Google Scholar] [CrossRef] [PubMed]
- Hartwig, V.; Giovannetti, G.; Vanello, N.; Lombardi, M.; Landini, L.; Simi, S. Biological effects and safety in magnetic resonance imaging: A review. Int. J. Environ. Res. Public Health 2009, 6, 1778–1798. [Google Scholar] [CrossRef] [PubMed]
- Fu, F.; Qin, Z.; Xu, C.; Chen, X.Y.; Li, R.X.; Wang, L.N.; Peng, D.W.; Sun, H.T.; Tu, Y.; Chen, C.; et al. Magnetic resonance imaging-three-dimensional printing technology fabricates customized scaffolds for brain tissue engineering. Neural Regener. Res. 2017, 12, 614–622. [Google Scholar]
- Mahmoudi, M.; Zhao, M.; Matsuura, Y.; Laurent, S.; Yang, P.C.; Bernstein, D.; Ruiz-Lozano, P.; Serpooshan, V. Infection-resistant MRI-visible scaffolds for tissue engineering applications. Bioimpacts 2016, 6, 111–115. [Google Scholar] [CrossRef] [PubMed]
- Wei, K.; Serpooshan, V.; Hurtado, C.; Diez-Cunado, M.; Zhao, M.; Maruyama, S.; Zhu, W.; Fajardo, G.; Noseda, M.; Nakamura, K.; et al. Epicardial FSTL1 reconstitution regenerates the adult mammalian heart. Nature 2015, 525, 479–485. [Google Scholar] [CrossRef] [PubMed]
- Melchels, F.P.W.; Domingos, M.A.N.; Klein, T.J.; Malda, J.; Bartolo, P.J.; Hutmacher, D.W. Additive manufacturing of tissues and organs. Prog. Polym. Sci. 2012, 37, 1079–1104. [Google Scholar] [CrossRef]
- Tegafaw, T.; Xu, W.; Ahmad, M.W.; Baeck, J.S.; Chang, Y.; Bae, J.E.; Chae, K.S.; Kim, T.J.; Lee, G.H. Dual-mode T1 and T2 magnetic resonance imaging contrast agent based on ultrasmall mixed gadolinium-dysprosium oxide nanoparticles: Synthesis, characterization, and in vivo application. Nanotechnology 2016, 26, 365102. [Google Scholar] [CrossRef]
- Krourdan, O.; Ribot, E.; Fricain, J.C.; Devillard, R.; Miraux, S. Magnetic Resonance Imaging for tracking cellular patterns obtained by Laser-Assisted Bioprinting. Sci. Rep. 2018, 8, 15777. [Google Scholar] [CrossRef]
- Choi, J.; Kim, K.; Kim, T.; Liu, G.; Bar-Shir, A.; Hyeon, T.; McMahon, M.T.; Bulte, J.W.; Fisher, J.P.; Gilad, A.A. Multimodal imaging of sustained drug release from 3-D poly(propylene fumarate) (PPF) scaffolds. J. Control. Release 2011, 156, 239–245. [Google Scholar] [CrossRef]
- Kang, X.; Yang, D.; Dai, Y.; Shang, M.; Cheng, Z.; Zhang, X.; Lian, H.; Ma, P.; Lin, J. Poly(acrylic acid) modified lanthanide-doped GdVO4 hollow spheres for up-conversion cell imaging, MRI and pH-dependent drug release. Nanoscale 2013, 5, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Berdichevski, A.; Yameen, H.S.; Dafni, H.; Neeman, M.; Seliktar, D. Using bimodal MRI/fluorescence imaging to identify host angiogenic response to implants. Proc. Natl. Acad. Sci. USA 2015, 112, 5147–5152. [Google Scholar] [CrossRef] [PubMed]
- Zadpoor, A.A.; Malda, J. Additive Manufacturing of Biomaterials, Tissues, and Organs. Ann. Biomed. Eng. 2017, 45, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Ye, Q.; Wu, Y.; Hsieh, W.Y.; Chen, C.L.; Shen, H.H.; Wang, S.J.; Zhang, H.; Hitchens, T.K.; Ho, C. Tracking T-cells in vivo with a new nano-sized MRI contrast agent. Nanomedicine 2012, 8, 1345–1354. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, M.; Yu, M.; Serpooshan, V.; Wu, J.C.; Langer, R.; Lee, R.T.; Karp, J.M.; Farokhzad, O.C. Multiscale technologies for treatment of ischemic cardiomyopathy. Nat. Nanotechnol. 2017, 12, 845–855. [Google Scholar] [CrossRef] [PubMed]
- Serpooshan, V.; Zhao, M.; Metzler, S.A.; Wei, K.; Shah, P.B.; Wang, A.; Mahmoudi, M.; Malkovskiy, A.V.; Rajadas, J.; Butte, M.J.; et al. The effect of bioengineered acellular collagen patch on cardiac remodeling and ventricular function post myocardial infarction. Biomaterials 2013, 34, 9048–9055. [Google Scholar] [CrossRef]
- Chen, Z.; Yan, C.; Yan, S.; Liu, Q.; Hou, M.; Xu, Y.; Guo, R. Non-invasive monitoring of in vivo hydrogel degradation and cartilage regeneration by multiparametric MR imaging. Theranostics 2018, 8, 1146–1158. [Google Scholar] [CrossRef] [PubMed]
- Proulx, M.; Aubin, K.; Lagueux, J.; Audet, P.; Auger, M.; Fortin, M.-A.; Fradette, J. Magnetic Resonance Imaging of Human Tissue-Engineered Adipose Substitutes. Tissue Eng. Part C Methods 2014, 21, 693–704. [Google Scholar] [CrossRef] [PubMed]
- Constantinides, C.; Basnett, P.; Lukasiewicz, B.; Carnicer, R.; Swider, E.; Majid, Q.A.; Srinivas, M.; Carr, C.A.; Roy, I. In Vivo Tracking and 1H/19F Magnetic Resonance Imaging of Biodegradable Polyhydroxyalkanoate/Polycaprolactone Blend Scaffolds Seeded with Labeled Cardiac Stem Cells. ACS Appl. Mater. Interfaces 2018, 10, 25056–25068. [Google Scholar] [CrossRef]
- Kedziorek, D.A.; Kraitchman, D.L. Superparamagnetic iron oxide labeling of stem cells for MRI tracking and delivery in cardiovascular disease. Methods Mol. Biol. 2010, 660, 171–183. [Google Scholar] [PubMed]
- Bulte, J.W. In vivo MRI cell tracking: Clinical studies. Am. J. Roentgenol. 2009, 193, 314–325. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, M.L.; Davis, C.; Bowen, C.V.; Stanley, O.; Parsons, C.; Weir, G.; Karkada, M.; Stanford, M.M.; Brewer, K.D. Using MRI cell tracking to monitor immune cell recruitment in response to a peptide-based cancer vaccine. Magn. Reson. Med. 2018, 80, 304–316. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.; Lai, P.T.; Leung, C.H.; Pong, P.W.T. Design of superparamagnetic nanoparticles for magnetic particle imaging (MPI). Int. J. Mol. Sci. 2013, 14, 18682–18710. [Google Scholar] [CrossRef] [PubMed]
- Bauer, L.M.; Situ, S.F.; Griswold, M.A.; Samia, A.C.S. Magnetic Particle Imaging Tracers: State-of-the-Art and Future Directions. J. Phys. Chem. Lett. 2015, 6, 2509–2517. [Google Scholar] [CrossRef]
- Panagiotopoulos, N.; Vogt, F.; Barkhausen, J.; Buzug, T.M.; Duschka, R.L.; Ldtke-Buzug, K.; Ahlborg, M.; Bringout, G.; Debbeler, C.; Grser, M.; et al. Magnetic particle imaging: Current developments and future directions. Int. J. Nanomed. 2015, 10, 3097. [Google Scholar] [CrossRef]
- Bietenbeck, M.; Florian, A.; Faber, C.; Sechtem, U.; Yilmaz, A. Remote magnetic targeting of iron oxide nanoparticles for cardiovascular diagnosis and therapeutic drug delivery: Where are we now? Int. J. Nanomed. 2016, 11, 3191–3203. [Google Scholar]
- Haegele, J.; Vaalma, S.; Panagiotopoulos, N.; Barkhausen, J.; Vogt, F.M.; Borgert, J.; Rahmer, J. Multi-color magnetic particle imaging for cardiovascular interventions. Phys. Med. Biol. 2016, 61, N415–N426. [Google Scholar] [CrossRef]
- Vogel, P.; Rckert, M.A.; Klauer, P.; Kullmann, W.H.; Jakob, P.M.; Behr, V.C. First in vivo traveling wave magnetic particle imaging of a beating mouse heart. Phys. Med. Biol. 2016, 61, 6620–6634. [Google Scholar] [CrossRef]
- Vaalma, S.; Rahmer, J.; Panagiotopoulos, N.; Duschka, R.L.; Borgert, J.; Barkhausen, J.; Vogt, F.M.; Haegele, J. Magnetic Particle Imaging (MPI): Experimental quantification of vascular stenosis using stationary stenosis phantoms. PLoS ONE 2017, 12, e0168902. [Google Scholar]
- Zhou, X.Y.; Tay, Z.W.; Chandrasekharan, P.; Yu, E.Y.; Hensley, D.W.; Orendorff, R.; Jeffris, K.E.; Mai, D.; Zheng, B.; Goodwill, P.W.; et al. Magnetic particle imaging for radiation-free, sensitive and high-contrast vascular imaging and cell tracking. Curr. Opin. Chem. Biol. 2018, 45, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Hachani, R.; Lowdell, M.; Birchall, M.; Thanh, N.T.K. Tracking stem cells in tissue-engineered organs using magnetic nanoparticles. Nanoscale 2013, 5, 11362–11373. [Google Scholar] [CrossRef]
- Zheng, B.; Vazin, T.; Goodwill, P.W.; Conway, A.; Verma, A.; Saritas, E.U.; Schaffer, D.; Conolly, S.M. Magnetic particle imaging tracks the long-term fate of in vivo neural cell implants with high image contrast. Sci. Rep. 2015, 5, 14055. [Google Scholar] [CrossRef] [PubMed]
- Zheng, B.; von See, M.P.; Yu, E.; Gunel, B.; Lu, K.; Vazin, T.; Schaffer, D.V.; Goodwill, P.W.; Conolly, S.M. Quantitative magnetic particle imaging monitors the transplantation, biodistribution, and clearance of stem cells in vivo. Theranostics 2016, 6, 291–301. [Google Scholar] [CrossRef] [PubMed]
- Jasmin; de Souza, G.T.; Louzada, R.A.; Rosado-de-Castro, P.H.; Mendez-Otero, R.; de Carvalho, A.C.C. Tracking stem cells with superparamagnetic iron oxide nanoparticles: Perspectives and considerations. Int. J. Nanomed. 2017, 12, 779–793. [Google Scholar] [CrossRef] [PubMed]
- Gu, L.; Li, X.; Jiang, J.; Guo, G.; Wu, H.; Wu, M.; Zhu, H. Stem cell tracking using effective self-assembled peptide-modified superparamagnetic nanoparticles. Nanoscale 2018, 10, 15967–15979. [Google Scholar] [CrossRef] [PubMed]
- Tay, Z.W.; Chandrasekharan, P.; Zhou, X.Y.; Yu, E.; Zheng, B.; Conolly, S. In vivo tracking and quantification of inhaled aerosol using magnetic particle imaging towards inhaled therapeutic monitoring. Theranostics 2018, 8, 3676–3687. [Google Scholar] [CrossRef]
- Schwenk, M.H. Ferumoxytol: A new intravenous iron preparation for the treatment of iron deficiency anemia in patients with chronic kidney disease. Pharmacotherapy 2010, 30, 70–79. [Google Scholar] [CrossRef]
- Reimer, P.; Balzer, T. Ferucarbotran (Resovist): A new clinically approved RES-specific contrast agent for contrast-enhanced MRI of the liver: Properties, clinical development, and applications. Eur. Radiol. 2003, 13, 1266–1276. [Google Scholar]
- Wang, Y.X. Current status of superparamagnetic iron oxide contrast agents for liver magnetic resonance imaging. World J. Gastroenterol. 2015, 21, 13400–13402. [Google Scholar] [CrossRef]
- Teshome, M.; Wei, C.; Hunt, K.K.; Thompson, A.; Rodriguez, K.; Mittendorf, E.A. Use of a Magnetic Tracer for Sentinel Lymph Node Detection in Early-Stage Breast Cancer Patients: A Meta-analysis. Ann. Surg. Oncol. 2016, 23, 1508–1514. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.; Liu, X.L.; di Fan, D.; Miao, Y.Q.; Zhang, H.; Ma, H.P.; Liu, Q.Y.; Ma, P.; Xue, W.M.; Luo, Y.E.; et al. The efficiency of magnetic hyperthermia and in vivo histocompatibility for human-like collagen protein-coated magnetic nanoparticles. Int. J. Nanomed. 2016, 11, 1175–1185. [Google Scholar]
- Bulte, J.W.M.; Walczak, P.; Janowski, M.; Krishnan, K.M.; Arami, H.; Halkola, A.; Gleich, B.; Rahmer, J. Quantitative “Hot-Spot” Imaging of Transplanted Stem Cells Using Superparamagnetic Tracers and Magnetic Particle Imaging. Tomography 2015, 1, 91–97. [Google Scholar] [PubMed]
- Thakor, A.S.; Jokerst, J.V.; Ghanouni, P.; Campbell, J.L.; Mittra, E.; Gambhir, S.S. Clinically Approved Nanoparticle Imaging Agents. J. Nucl. Med. 2016, 57, 1833–1837. [Google Scholar] [CrossRef] [PubMed]
- Laperle, C.M.; Hamilton, T.J.; Wintermeyer, P.; Walker, E.J.; Shi, D.; Anastasio, M.A.; Derdak, Z.; Wands, J.R.; Diebold, G.; Rose-Petruck, C. Low density contrast agents for x-ray phase contrast imaging: The use of ambient air for x-ray angiography of excised murine liver tissue. Phys. Med. Biol. 2008, 53, 6911–6923. [Google Scholar] [CrossRef]
- Murata, H.; Oka, Y.; Aoki, K.; Maeda, S.; Yamashita, K.; Terai, H.; Miyamoto, T. Digital subtraction angiography of cardiovascular abnormalities using FCR, a new X-ray diagnostic system. Kyobu Geka 1985, 38, 530–534. [Google Scholar]
- Rhee, T.K.; Park, J.K.; Cashen, T.A.; Shin, W.; Schirf, B.E.; Gehl, J.A.; Larson, A.C.; Carr, J.C.; Li, D.; Carroll, T.J.; et al. Comparison of intraarterial MR angiography at 3.0 T with X-ray digital subtraction angiography for detection of renal artery stenosis in swine. J. Vasc. Interv. Radiol. 2006, 17, 1131–1137. [Google Scholar] [CrossRef]
- Mu, C.L.; Luo, L.H.; Wang, B.P. Comparative study of color Doppler flow imaging with X-ray angiography in the diagnosis of subcutaneous soft-tissue hemangioma. Nan Fang Yi Ke Da Xue Xue Bao 2010, 30, 2770–2771. [Google Scholar]
- Johansson, L.O.; Nolan, M.M.; Taniuchi, M.; Fischer, S.E.; Wickline, S.A.; Lorenz, C.H. High-resolution magnetic resonance coronary angiography of the entire heart using a new blood-pool agent, NC100150 injection: Comparison with invasive x-ray angiography in pigs. J. Cardiovasc. Magn. Reson. 1999, 1, 139–143. [Google Scholar] [CrossRef]
- Geva, T.; Greil, G.F.; Marshall, A.C.; Landzberg, M.; Powell, A.J. Gadolinium-enhanced 3-dimensional magnetic resonance angiography of pulmonary blood supply in patients with complex pulmonary stenosis or atresia: Comparison with x-ray angiography. Circulation 2002, 106, 473–478. [Google Scholar] [CrossRef]
- Anderson, C.M.; Saloner, D.; Lee, R.E.; Griswold, V.J.; Shapeero, L.G.; Rapp, J.H.; Nagarkar, S.; Pan, X.; Gooding, G.A. Assessment of carotid artery stenosis by MR angiography: Comparison with x-ray angiography and color-coded Doppler ultrasound. Am. J. Neuroradiol. 1992, 13, 989–1003. [Google Scholar] [PubMed]
- Wutke, R.; Lang, W.; Fellner, C.; Janka, R.; Denzel, C.; Lell, M.; Bautz, W.; Fellner, F.A. High-resolution, contrast-enhanced magnetic resonance angiography with elliptical centric k-space ordering of supra-aortic arteries compared with selective X-ray angiography. Stroke 2002, 33, 1522–1529. [Google Scholar] [CrossRef] [PubMed]
- Hartung, M.P.; Grist, T.M.; Francois, C.J. Magnetic resonance angiography: Current status and future directions. J. Cardiovasc. Magn. Reson. 2011, 13, 19. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.N.; Basheer, A.S.; Sukumaran, G.G.; Padmajan, S.; Praveen, S.; Velappan, P.; Nair, B.U.; Nair, S.G.; Kunjuraman, U.K.; Madthipat, U.; et al. Femoral artery pseudoaneurysm as a complication of angioplasty. How can it be prevented? Heart Asia 2013, 5, 144–147. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cox, S.C.; Thornby, J.A.; Gibbons, G.J.; Williams, M.A.; Mallick, K.K. 3D printing of porous hydroxyapatite scaffolds intended for use in bone tissue engineering applications. Mater. Sci. Eng. C 2015, 47, 237–247. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.T.; Milby, A.H.; Ikuta, K.; Poudel, S.; Pfeifer, C.G.; Elliott, D.M.; Smith, H.E.; Mauck, R.L. A radiopaque electrospun scaffold for engineering fibrous musculoskeletal tissues: Scaffold characterization and in vivo applications. Acta Biomater. 2015, 26, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Osorio, D.A.; Lee, B.E.J.; Kwiecien, J.M.; Wang, X.; Shahid, I.; Hurley, A.L.; Cranston, E.D.; Grandfield, K. Cross-linked cellulose nanocrystal aerogels as viable bone tissue scaffolds. Acta Biomater. 2019, 87, 152–165. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, V.P.; Pina, S.; Costa, J.B.; Cengiz, I.F.; Garca-Fernndez, L.; Fernndez-Gutirrez, M.D.M.; Paiva, O.C.; Oliveira, A.L.; San-Romn, J.; Oliveira, J.M.; et al. Enzymatically Cross-Linked Silk Fibroin-Based Hierarchical Scaffolds for Osteochondral Regeneration. ACS Appl. Mater. Interfaces 2019, 11, 3781–3799. [Google Scholar] [CrossRef]
- Shepherd, D.V.; Shepherd, J.H.; Best, S.M.; Cameron, R.E. 3D imaging of cells in scaffolds: Direct labelling for micro CT. J. Mater. Sci. Mater. Med. 2018, 29, 86. [Google Scholar] [CrossRef]
- Zidek, J.; Vojtova, L.; Abdel-Mohsen, A.M.; Chmelik, J.; Zikmund, T.; Brtnikova, J.; Jakubicek, R.; Zubal, L.; Jan, J.; Kaiser, J. Accurate micro-computed tomography imaging of pore spaces in collagen-based scaffold. J. Mater. Sci. Mater. Med. 2016, 27, 110. [Google Scholar] [CrossRef]
- Gmez, S.; Vlad, M.D.; Lpez, J.; Fernndez, E. Design and properties of 3D scaffolds for bone tissue engineering. Acta Biomater. 2016, 42, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Izadifar, Z.; Honaramooz, A.; Wiebe, S.; Belev, G.; Chen, X.; Chapman, D. Low-dose phase-based X-ray imaging techniques for in situ soft tissue engineering assessments. Biomaterials 2016, 82, 151–167. [Google Scholar] [CrossRef] [PubMed]
- Sonnaert, M.; Kerckhofs, G.; Papantoniou, I.; Van Vlierberghe, S.; Boterberg, V.; Dubruel, P.; Luyten, F.P.; Schrooten, J.; Geris, L. Multifactorial optimization of contrast-enhanced nanofocus computed tomography for quantitative analysis of neo-tissue formation in tissue engineering constructs. PLoS ONE 2015, 10, e0130227. [Google Scholar] [CrossRef] [PubMed]
- Appel, A.A.; Larson, J.C.; Garson, A.B.; Guan, H.; Zhong, Z.; Nguyen, B.N.B.; Fisher, J.P.; Anastasio, M.A.; Brey, E.M. X-ray phase contrast imaging of calcified tissue and biomaterial structure in bioreactor engineered tissues. Biotechnol. Bioeng. 2015, 112, 612–620. [Google Scholar] [CrossRef] [PubMed]
- Appel, A.A.; Larson, J.C.; Jiang, B.; Zhong, Z.; Anastasio, M.A.; Brey, E.M. X-ray Phase Contrast Allows Three Dimensional, Quantitative Imaging of Hydrogel Implants. Ann. Biomed. Eng. 2016, 44, 773–781. [Google Scholar] [CrossRef] [PubMed]
- Meir, R.; Shamalov, K.; Betzer, O.; Motiei, M.; Horovitz-Fried, M.; Yehuda, R.; Popovtzer, A.; Popovtzer, R.; Cohen, C.J. Nanomedicine for Cancer Immunotherapy: Tracking Cancer-Specific T-Cells in Vivo with Gold Nanoparticles and CT Imaging. ACS Nano 2015, 9, 6363–6372. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, A.L.; Dhanantwari, A.; Jurcova, M.; Cheheltani, R.; Naha, P.C.; Ivanc, T.; Shefer, E.; Cormode, D.P. Improved sensitivity of computed tomography towards iodine and gold nanoparticle contrast agents via iterative reconstruction methods. Sci. Rep. 2016, 6, 26177. [Google Scholar] [CrossRef]
- Khademi, S.; Sarkar, S.; Shakeri-Zadeh, A.; Attaran, N.; Kharrazi, S.; Ay, M.R.; Ghadiri, H. Folic acid-cysteamine modified gold nanoparticle as a nanoprobe for targeted computed tomography imaging of cancer cells. Mater. Sci. Eng. C 2018, 89, 182–193. [Google Scholar] [CrossRef]
- Celikkin, N.; Mastrogiacomo, S.; Walboomers, X.; Swieszkowski, W. Enhancing X-ray Attenuation of 3D Printed Gelatin Methacrylate (GelMA) Hydrogels Utilizing Gold Nanoparticles for Bone Tissue Engineering Applications. Polymers 2019, 11, 367. [Google Scholar] [CrossRef]
- Finamore, T.A.; Curtis, T.E.; Tedesco, J.V.; Grandfield, K.; Roeder, R.K. Nondestructive, longitudinal measurement of collagen scaffold degradation using computed tomography and gold nanoparticles. Nanoscale 2019, 11, 4345–4354. [Google Scholar] [CrossRef]
- Wang, Y.; Xiong, Z.; He, Y.; Zhou, B.; Qu, J.; Shen, M.; Shi, X.; Xia, J. Optimization of the composition and dosage of PEGylated polyethylenimine-entrapped gold nanoparticles for blood pool, tumor, and lymph node CT imaging. Mater. Sci. Eng. C 2018, 83, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Cheheltani, R.; Ezzibdeh, R.M.; Chhour, P.; Pulaparthi, K.; Kim, J.; Jurcova, M.; Hsu, J.C.; Blundell, C.; Litt, H.I.; Ferrari, V.A.; et al. Tunable, biodegradable gold nanoparticles as contrast agents for computed tomography and photoacoustic imaging. Biomaterials 2016, 102, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Chhour, P.; Hsu, J.; Litt, H.I.; Ferrari, V.A.; Popovtzer, R.; Cormode, D.P. Use of Nanoparticle Contrast Agents for Cell Tracking with Computed Tomography. Bioconjugate Chem. 2017, 28, 1581–1597. [Google Scholar] [CrossRef] [PubMed]
- Lakin, B.A.; Patel, H.; Holland, C.; Freedman, J.D.; Shelofsky, J.S.; Snyder, B.D.; Stok, K.S.; Grinstaff, M.W. Contrast-enhanced CT using a cationic contrast agent enables non-destructive assessment of the biochemical and biomechanical properties of mouse tibial plateau cartilage. J. Orthop. Res. 2016, 34, 1130–1138. [Google Scholar] [CrossRef] [PubMed]
- Chae, K.S.; Ahmad, M.W.; Kim, T.J.; Lee, G.H.; Xu, W.; Baeck, J.S.; Kim, S.J.; Park, J.A.; Bae, J.E.; Chang, Y. Synthesis of nanoparticle CT contrast agents: In vitro and in vivo studies. Sci. Technol. Adv. Mater. 2015, 16, 055003. [Google Scholar]
- Torchilin, V.P. Polymeric contrast agents for medical imaging. Curr. Pharm. Biotechnol. 2000, 1, 183–215. [Google Scholar] [CrossRef] [PubMed]
- Boll, H.; Nittka, S.; Doyon, F.; Neumaier, M.; Marx, A.; Kramer, M.; Groden, C.; Brockmann, M.A. Micro-CT based experimental liver imaging using a nanoparticulate contrast agent: A longitudinal study in mice. PLoS One 2011, 6, e25692. [Google Scholar] [CrossRef] [PubMed]
- Emmert, M.Y.; Schmitt, B.A.; Loerakker, S.; Sanders, B.; Spriestersbach, H.; Fioretta, E.S.; Bruder, L.; Brakmann, K.; Motta, S.E.; Lintas, V.; et al. Computational modeling guides tissue-engineered heart valve design for long-term in vivo performance in a translational sheep model. Sci. Transl. Med. 2018, 10, eaan4587. [Google Scholar] [CrossRef]
- Kajbafzadeh, A.M.; Ahmadi Tafti, S.H.; Mokhber-Dezfooli, M.R.; Khorramirouz, R.; Sabetkish, S.; Sabetkish, N.; Rabbani, S.; Tavana, H.; Mohseni, M.J. Aortic valve conduit implantation in the descending thoracic aorta in a sheep model: The outcomes of pre-seeded scaffold. Int. J. Surg. 2016, 28, 97–105. [Google Scholar] [CrossRef]
- Zhu, J.; Wang, G.; Alves, C.S.; Toms, H.; Xiong, Z.; Shen, M.; Rodrigues, J.; Shi, X. Multifunctional Dendrimer-Entrapped Gold Nanoparticles Conjugated with Doxorubicin for pH-Responsive Drug Delivery and Targeted Computed Tomography Imaging. Langmuir 2018, 34, 12428–12435. [Google Scholar] [CrossRef]
- Shikhaliev, P.M. Soft tissue imaging with photon counting spectroscopic CT. Phys. Med. Biol. 2015, 60, 2453–2474. [Google Scholar] [CrossRef] [PubMed]
- Talukdar, Y.; Avti, P.; Sun, J.; Sitharaman, B. Multimodal ultrasound-photoacoustic imaging of tissue engineering scaffolds and blood oxygen saturation in and around the scaffolds. Tissue Eng. Part C Methods 2014, 20, 440–449. [Google Scholar] [CrossRef] [PubMed]
- Deng, C.X.; Hong, X.; Stegemann, J.P. Ultrasound Imaging Techniques for Spatiotemporal Characterization of Composition, Microstructure, and Mechanical Properties in Tissue Engineering. Tissue Eng. Part B Rev. 2016, 22, 311–321. [Google Scholar] [CrossRef]
- Kim, K.; Wagner, W.R. Non-invasive and Non-destructive Characterization of Tissue Engineered Constructs Using Ultrasound Imaging Technologies: A Review. Ann. Biomed. Eng. 2016, 44, 621–635. [Google Scholar] [CrossRef] [PubMed]
- Stukel, J.M.; Goss, M.; Zhou, H.; Zhou, W.; Willits, R.K.; Exner, A.A. Development of a High-Throughput Ultrasound Technique for the Analysis of Tissue Engineering Constructs. Ann. Biomed. Eng. 2016, 44, 793–802. [Google Scholar] [CrossRef] [PubMed]
- Shinohara, M.; Sabra, K.; Gennisson, J.L.; Fink, M.; Tanter, M. Real-time visualization of muscle stiffness distribution with ultrasound shear wave imaging during muscle contraction. Muscle Nerve 2010, 42, 438–441. [Google Scholar] [CrossRef] [PubMed]
- Arda, K.; Ciledag, N.; Aktas, E.; Aribas, B.K.; Kose, K. Quantitative assessment of normal soft-tissue elasticity using shear-wave ultrasound elastography. Am. J. Roentgenol. 2011, 197, 532–536. [Google Scholar] [CrossRef] [PubMed]
- Itoh, A.; Ueno, E.; Tohno, E.; Kamma, H.; Takahashi, H.; Shiina, T.; Yamakawa, M.; Matsumura, T. Breast disease: Clinical application of US elastography for diagnosis. Radiology 2006, 239, 341–350. [Google Scholar] [CrossRef]
- Walker, J.M.; Myers, A.M.; Schluchter, M.D.; Goldberg, V.M.; Caplan, A.I.; Berilla, J.A.; Mansour, J.M.; Welter, J.F. Nondestructive evaluation of hydrogel mechanical properties using ultrasound. Ann. Biomed. Eng. 2011, 39, 2521–2530. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y. The Application of Ultrasound in 3D Bio-Printing. Molecules 2016, 21, 590. [Google Scholar] [CrossRef]
- Garvin, K.A.; Dalecki, D.; Hocking, D.C. Vascularization of three-dimensional collagen hydrogels using ultrasound standing wave fields. Ultrasound Med. Biol. 2011, 37, 1853–1864. [Google Scholar] [CrossRef] [PubMed]
- Angele, P.; Yoo, J.U.; Smith, C.; Mansour, J.; Jepsen, K.J.; Nerlich, M.; Johnstone, B. Cyclic hydrostatic pressure enhances the chondrogenic phenotype of human mesenchymal progenitor cells differentiated in vitro. J. Orthop. Res. 2003, 21, 451–457. [Google Scholar] [CrossRef]
- Kim, J.E.; Kalimuthu, S.; Ahn, B.C. In Vivo Cell Tracking with Bioluminescence Imaging. Nucl. Med. Mol. Imaging 2015, 49, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Yun, S.H.; Kwok, S.J.J. Light in diagnosis, therapy and surgery. Nat. Biomed. Eng. 2017, 1, 8. [Google Scholar] [CrossRef] [PubMed]
- Arranz, A.; Ripoll, J. Advances in optical imaging for pharmacological studies. Front. Pharmacol. 2015, 6, 189. [Google Scholar] [CrossRef]
- Sharkey, J.; Scarfe, L.; Santeramo, I.; Garcia-Finana, M.; Park, B.K.; Poptani, H.; Wilm, B.; Taylor, A.; Murray, P. Imaging technologies for monitoring the safety, efficacy and mechanisms of action of cell-based regenerative medicine therapies in models of kidney disease. Eur. J. Pharmacol. 2016, 790, 74–82. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Conradi, L.; Schmidt, S.; Neofytou, E.; Deuse, T.; Peters, L.; Eder, A.; Hua, X.; Hansen, A.; Robbins, R.C.; Beygui, R.E.; et al. Immunobiology of Fibrin-Based Engineered Heart Tissue. Stem Cells Transl. Med. 2015, 4, 625–631. [Google Scholar] [CrossRef]
- Crowley, C.; Butler, C.R.; Camilli, C.; Hynds, R.E.; Kolluri, K.K.; Janes, S.M.; De Coppi, P.; Urbani, L. Non-Invasive Longitudinal Bioluminescence Imaging of Human Mesoangioblasts in Bioengineered Esophagi. Tissue Eng. Part C Methods 2019, 25, 103–113. [Google Scholar] [CrossRef]
- Kuchimaru, T.; Iwano, S.; Kiyama, M.; Mitsumata, S.; Kadonosono, T.; Niwa, H.; Maki, S.; Kizaka-Kondoh, S. A luciferin analogue generating near-infrared bioluminescence achieves highly sensitive deep-tissue imaging. Nat. Commun. 2016, 7, 11856. [Google Scholar] [CrossRef]
- Iwano, S.; Sugiyama, M.; Hama, H.; Watakabe, A.; Hasegawa, N.; Kuchimaru, T.; Tanaka, K.Z.; Takahashi, M.; Ishida, Y.; Hata, J.; et al. Single-cell bioluminescence imaging of deep tissue in freely moving animals. Science 2018, 359, 935–939. [Google Scholar] [CrossRef]
- Peeters, M.; Van Rijn, S.; Vergroesen, P.P.A.; Paul, C.P.; Noske, D.P.; Vandertop, W.P.; Wurdinger, T.; Helder, M.N. Bioluminescence-mediated longitudinal monitoring of adipose-derived stem cells in a large mammal ex vivo organ culture. Sci. Rep. 2015, 5, 13960. [Google Scholar] [CrossRef] [PubMed]
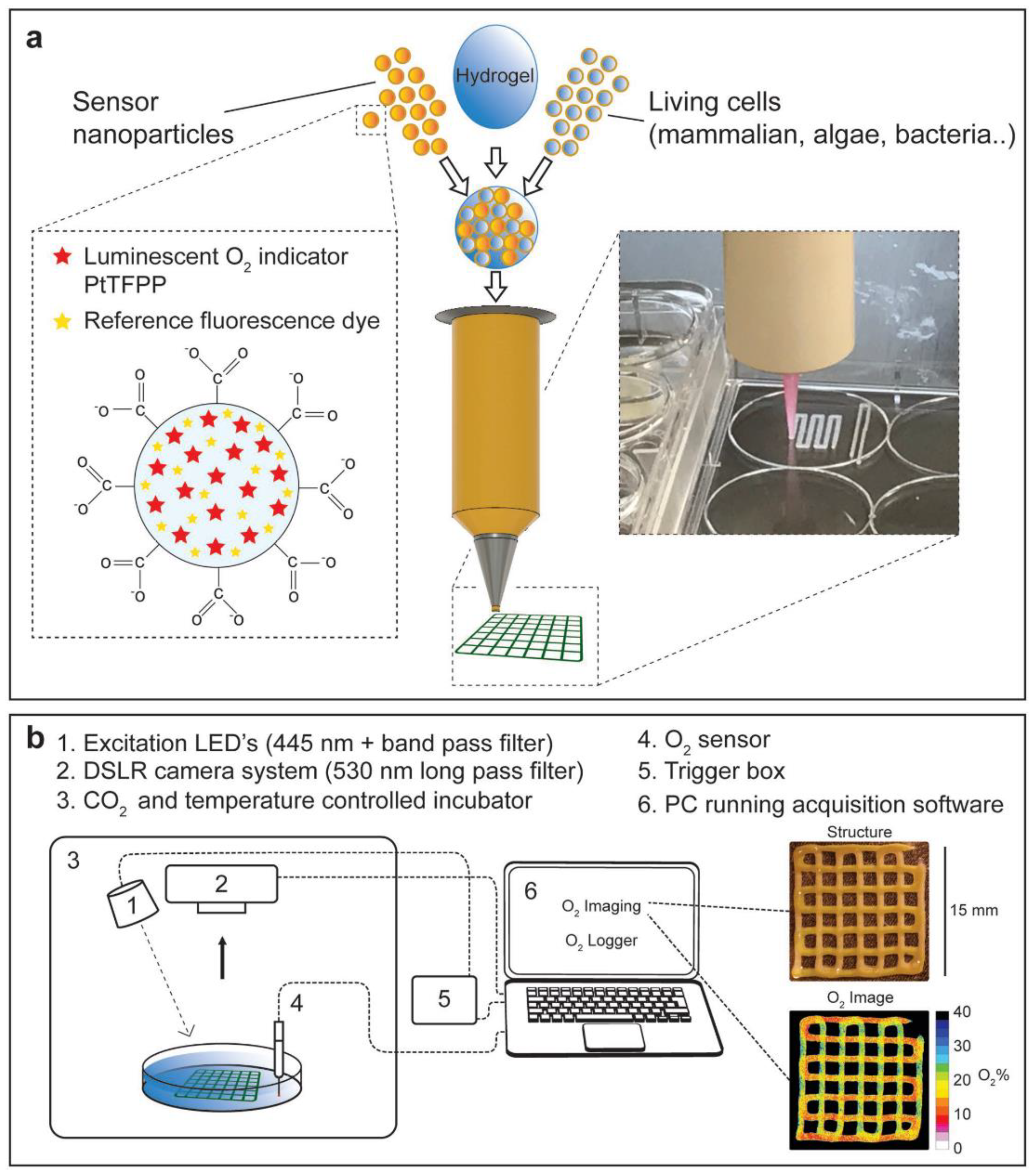
- Trampe, E.; Koren, K.; Akkineni, A.R.; Senwitz, C.; Krujatz, F.; Lode, A.; Gelinsky, M.; Kühl, M. Functionalized Bioink with Optical Sensor Nanoparticles for O2 Imaging in 3D-Bioprinted Constructs. Adv. Funct. Mater. 2018, 28, 1804411. [Google Scholar] [CrossRef]
- Keriquel, V.; Oliveira, H.; Remy, M.; Ziane, S.; Delmond, S.; Rousseau, B.; Rey, S.; Catros, S.; Amedee, J.; Guillemot, F.; et al. In situ printing of mesenchymal stromal cells, by laser-assisted bioprinting, for in vivo bone regeneration applications. Sci. Rep. 2017, 7, 1778. [Google Scholar] [CrossRef] [PubMed]
- Chinen, A.B.; Guan, C.M.; Ferrer, J.R.; Barnaby, S.N.; Merkel, T.J.; Mirkin, C.A. Nanoparticle Probes for the Detection of Cancer Biomarkers, Cells, and Tissues by Fluorescence. Chem. Rev. 2015, 115, 10530–10574. [Google Scholar] [CrossRef]
- Tajika, Y.; Murakami, T.; Iijima, K.; Gotoh, H.; Takahashi-Ikezawa, M.; Ueno, H.; Yoshimoto, Y.; Yorifuji, H. A novel imaging method for correlating 2D light microscopic data and 3D volume data based on block-face imaging. Sci. Rep. 2017, 7, 3645. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Hilderink, J.; Groothuis, T.A.M.; Otto, C.; van Blitterswijk, C.A.; de Boer, J. Monitoring nutrient transport in tissue-engineered grafts. J. Tissue Eng. Regener. Med. 2015, 9, 952–960. [Google Scholar] [CrossRef] [PubMed]
- Park, G.K.; I, H.; Kim, G.S.; Hwang, N.S.; Choi, H.S. Optical spectroscopic imaging for cell therapy and tissue engineering. Appl. Spectrosc. Rev. 2018, 53, 360–375. [Google Scholar] [CrossRef] [PubMed]
- Bilen, B.; Gokbulut, B.; Kafa, U.; Heves, E.; Inci, M.N.; Unlu, M.B. Scanning Acoustic Microscopy and Time-Resolved Fluorescence Spectroscopy for Characterization of Atherosclerotic Plaques. Sci. Rep. 2018, 8, 14378. [Google Scholar] [CrossRef]
- Sabapathy, V.; Mentam, J.; Jacob, P.M.; Kumar, S. Noninvasive Optical Imaging and In Vivo Cell Tracking of Indocyanine Green Labeled Human Stem Cells Transplanted at Superficial or In-Depth Tissue of SCID Mice. Stem Cells Int. 2015, 2015, 606415. [Google Scholar] [CrossRef]
- Artzi, N.; Oliva, N.; Puron, C.; Shitreet, S.; Artzi, S.; Bon Ramos, A.; Groothuis, A.; Sahagian, G.; Edelman, E.R. In vivo and in vitro tracking of erosion in biodegradable materials using non-invasive fluorescence imaging. Nat. Mater. 2011, 10, 704–709. [Google Scholar]
- Cen, P.; Chen, J.; Hu, C.; Fan, L.; Wang, J.; Li, L. Noninvasive in-vivo tracing and imaging of transplanted stem cells for liver regeneration. Stem Cell Res. Ther. 2016, 7, 143. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dong, Y.; Jin, G.; Ji, C.; He, R.; Lin, M.; Zhao, X.; Li, A.; Lu, T.J.; Xu, F. Non-invasive tracking of hydrogel degradation using upconversion nanoparticles. Acta Biomater. 2017, 55, 410–419. [Google Scholar] [CrossRef] [PubMed]
- Thoniyot, P.; Tan, M.J.; Karim, A.A.; Young, D.J.; Loh, X.J. Nanoparticle-Hydrogel Composites: Concept, Design, and Applications of These Promising, Multi-Functional Materials. Adv. Sci. 2015, 2, 1400010. [Google Scholar] [CrossRef] [PubMed]
- Shu, X.; Beckmann, L.; Zhang, H. Visible-light optical coherence tomography: A review. J. Biomed. Opt. 2017, 22, 121707. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Brown, W.; Maher, J.R.; Levinson, H.; Wax, A. Functional optical coherence tomography: Principles and progress. Phys. Med. Biol. 2015, 60, R211–R237. [Google Scholar] [CrossRef] [PubMed]
- Holmes, C.; Tabrizian, M.; Bagnaninchi, P.O. Motility imaging via optical coherence phase microscopy enables label-free monitoring of tissue growth and viability in 3D tissue-engineering scaffolds. J. Tissue Eng. Regener. Med. 2015, 9, 641–645. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Xu, M.; Zhang, L.; Zhou, Q.; Luo, L. Automated quantitative assessment of three-dimensional bioprinted hydrogel scaffolds using optical coherence tomography. Biomed. Opt. Express 2016, 7, 894–910. [Google Scholar] [CrossRef]
- Chen, W.; Yang, J.; Liao, W.; Zhou, J.; Zheng, J.; Wu, Y.; Li, D.; Lin, Z. In vitro remodeling and structural characterization of degradable polymer scaffold-based tissue-engineered vascular grafts using optical coherence tomography. Cell Tissue Res. 2017, 370, 417–426. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Xu, M.E.; Luo, L.; Zhou, Y.; Si, P. Iterative feedback bio-printing-derived cell-laden hydrogel scaffolds with optimal geometrical fidelity and cellular controllability. Sci. Rep. 2018, 8, 2802. [Google Scholar] [CrossRef] [PubMed]
- Guo, T.; Noshin, M.; Baker, H.B.; Taskoy, E.; Meredith, S.J.; Tang, Q.; Ringel, J.P.; Lerman, M.J.; Chen, Y.; Packer, J.D.; et al. 3D printed biofunctionalized scaffolds for microfracture repair of cartilage defects. Biomaterials 2018, 185, 219–231. [Google Scholar] [CrossRef] [PubMed]
- Avigo, C.; Flori, A.; Armanetti, P.; Di Lascio, N.; Kusmic, C.; Jose, J.; Losi, P.; Soldani, G.; Faita, F.; Menichetti, L. Strategies for non-invasive imaging of polymeric biomaterial in vascular tissue engineering and regenerative medicine using ultrasound and photoacoustic techniques. Polym. Int. 2016, 65, 734–740. [Google Scholar] [CrossRef]
- Wang, L.V.; Yao, J. A practical guide to photoacoustic tomography in the life sciences. Nat. Methods 2016, 13, 627–638. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; Paratala, B.S.; Hu, S.; Sitharaman, B.; Wang, L.V. Multiscale photoacoustic microscopy of single-walled carbon nanotube-incorporated tissue engineering scaffolds. Tissue Eng. Part C Methods 2012, 18, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Aliabouzar, M.; Zhang, G.L.; Sarkar, K. Acoustic and mechanical characterization of 3D-printed scaffolds for tissue engineering applications. Biomed. Mater. 2018, 13, 055013. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Wang, L.V. Photoacoustic imaging and characterization of the microvasculature. J. Biomed. Opt. 2010, 15, 011101. [Google Scholar] [CrossRef] [PubMed]
- Lyu, Y.; Fang, Y.; Miao, Q.; Zhen, X.; Ding, D.; Pu, K. Intraparticle Molecular Orbital Engineering of Semiconducting Polymer Nanoparticles as Amplified Theranostics for in Vivo Photoacoustic Imaging and Photothermal Therapy. ACS Nano 2016, 10, 4472–4481. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.; Kim, H.; Kumar, D.; Kim, H.; Sim, C.; Chang, J.H.; Kim, J.M.; Lim, D.K. Amplified photoacoustic performance and enhanced photothermal stability of reduced graphene oxide coated gold nanorods for sensitive photoacoustic imaging. ACS Nano 2015, 9, 2711–2719. [Google Scholar] [CrossRef] [PubMed]
- Antaris, A.L.; Chen, H.; Cheng, K.; Sun, Y.; Hong, G.; Qu, C.; Diao, S.; Deng, Z.; Hu, X.; Zhang, B.; et al. A small-molecule dye for NIR-II imaging. Nat. Mater. 2016, 15, 235–242. [Google Scholar] [CrossRef]
- Jiang, Y.; Pu, K. Advanced Photoacoustic Imaging Applications of Near-Infrared Absorbing Organic Nanoparticles. Small 2017, 13, 1700710. [Google Scholar] [CrossRef]
- Huang, Q.; Zhang, S.; Zhang, H.; Han, Y.; Liu, H.; Ren, F.; Sun, Q.; Li, Z.; Gao, M. Boosting the Radiosensitizing and Photothermal Performance of Cu2-x Se Nanocrystals for Synergetic Radiophotothermal Therapy of Orthotopic Breast Cancer. ACS Nano 2019, 13, 1342–1353. [Google Scholar]
- Van der Horst, G.; Buijs, J.T.; van der Pluijm, G. Chapter 46—Pre-clinical molecular imaging of “the seed and the soil” in bone metastasis. In Bone Cancer, 2nd ed.; Heymann, D., Ed.; Academic Press: San Diego, CA, USA, 2015; pp. 557–570. [Google Scholar]
- Rieffel, J.; Chitgupi, U.; Lovell, J.F. Recent Advances in Higher-Order, Multimodal, Biomedical Imaging Agents. Small 2015, 11, 4445–4461. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Dougherty, C.A.; Yang, D.; Wu, H.; Hong, H. Radioactive Nanomaterials for Multimodality Imaging. Tomography 2016, 2, 3–16. [Google Scholar] [PubMed]
- Ametamey, S.M.; Honer, M.; Schubiger, P.A. Molecular imaging with PET. Chem. Rev. 2008, 108, 1501–1516. [Google Scholar] [CrossRef] [PubMed]
- Kempen, D.H.; Yaszemski, M.J.; Heijink, A.; Hefferan, T.E.; Creemers, L.B.; Britson, J.; Maran, A.; Classic, K.L.; Dhert, W.J.; Lu, L. Non-invasive monitoring of BMP-2 retention and bone formation in composites for bone tissue engineering using SPECT/CT and scintillation probes. J. Control. Release 2009, 134, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Peng, X.; Mao, C.; Tian, J.H.; Zhang, S.W.; Xu, F.; Tu, J.J.; Liu, S.; Hu, M.; Yu, G.Y. The Value of SPECT/CT in Monitoring Prefabricated Tissue-Engineered Bone and Orthotopic rhBMP-2 Implants for Mandibular Reconstruction. PLoS ONE 2015, 10, e0137167. [Google Scholar] [CrossRef] [PubMed]
- Abou, D.S.; Thorek, D.L.; Ramos, N.N.; Pinkse, M.W.; Wolterbeek, H.T.; Carlin, S.D.; Beattie, B.J.; Lewis, J.S. (89)Zr-labeled paramagnetic octreotide-liposomes for PET-MR imaging of cancer. Pharm. Res. 2013, 30, 878–888. [Google Scholar] [CrossRef] [PubMed]
- Alex, S.; Tiwari, A. Functionalized Gold Nanoparticles: Synthesis, Properties and Applications—A Review. J. Nanosci. Nanotechnol. 2015, 15, 1869–1894. [Google Scholar] [CrossRef] [PubMed]
- Nagao, R.J.; Ouyang, Y.; Keller, R.; Nam, S.Y.; Malik, G.R.; Emelianov, S.Y.; Suggs, L.J.; Schmidt, C.E. Ultrasound-guided photoacoustic imaging-directed re-endothelialization of acellular vasculature leads to improved vascular performance. Acta Biomater. 2016, 32, 35–45. [Google Scholar] [CrossRef]

| Method | Spatial Resolution | Imaging Depth | Information Type | Cost | External Material | Applications |
|---|---|---|---|---|---|---|
| CT | 5 µm | No limit | 3D | Medium | Yes | Scaffold + cells + Pre/Clinical |
| MRI | 5–200 µm | No limit | 3D | High | No | Scaffold + cells + Pre/Clinical |
| MPI | 1 µm | No limit | 2D/3D | Medium | Yes | Scaffold + cells + Pre/Clinical |
| Ultrasound | 20–100 µm | 10 mm | 3D | Medium | No | Scaffold + cells + Pre/Clinical |
| Fluorescence | 0.2–1 µm | 0.3–1 mm | 2D | Low | Yes | Scaffold + cells |
| Bioluminescence | 2–3 mm | 10 mm | 2D | Low | Yes | Scaffold + cells |
| Optical coherence tomography (OCT) | 1–15 µm | 1–3 mm | 2D | Low | No | Scaffold + cells + Pre/Clinical |
| Photoacoustic | 50–150 µm | 20 mm | 2D/3D | Medium | No | Scaffold + cells + Pre/Clinical |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gil, C.J.; Tomov, M.L.; Theus, A.S.; Cetnar, A.; Mahmoudi, M.; Serpooshan, V. In Vivo Tracking of Tissue Engineered Constructs. Micromachines 2019, 10, 474. https://doi.org/10.3390/mi10070474
Gil CJ, Tomov ML, Theus AS, Cetnar A, Mahmoudi M, Serpooshan V. In Vivo Tracking of Tissue Engineered Constructs. Micromachines. 2019; 10(7):474. https://doi.org/10.3390/mi10070474
Chicago/Turabian StyleGil, Carmen J., Martin L. Tomov, Andrea S. Theus, Alexander Cetnar, Morteza Mahmoudi, and Vahid Serpooshan. 2019. "In Vivo Tracking of Tissue Engineered Constructs" Micromachines 10, no. 7: 474. https://doi.org/10.3390/mi10070474
APA StyleGil, C. J., Tomov, M. L., Theus, A. S., Cetnar, A., Mahmoudi, M., & Serpooshan, V. (2019). In Vivo Tracking of Tissue Engineered Constructs. Micromachines, 10(7), 474. https://doi.org/10.3390/mi10070474






