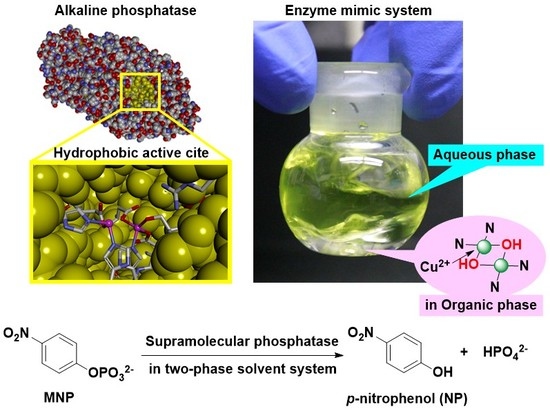
Catalytic Hydrolysis of Phosphate Monoester by Supramolecular Complexes Formed by the Self-Assembly of a Hydrophobic Bis(Zn2+-cyclen) Complex, Copper, and Barbital Units That Are Functionalized with Amino Acids in a Two-Phase Solvent System
Abstract
1. Introduction
2. Materials and Methods
2.1. General Information
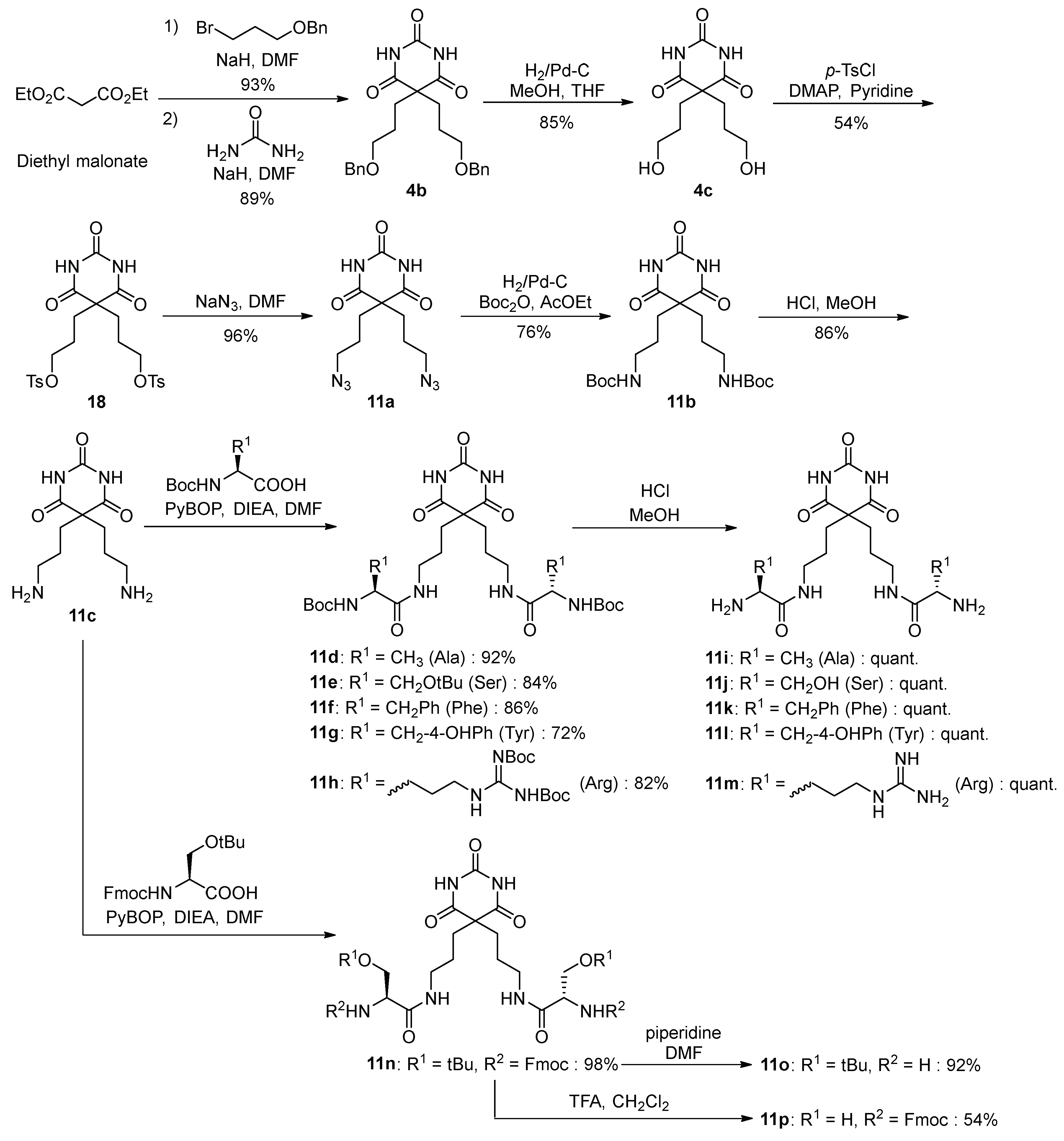
2.2. Synthesis of Compounds
2.3. Hydrolysis of MNP
3. Results and Discussion
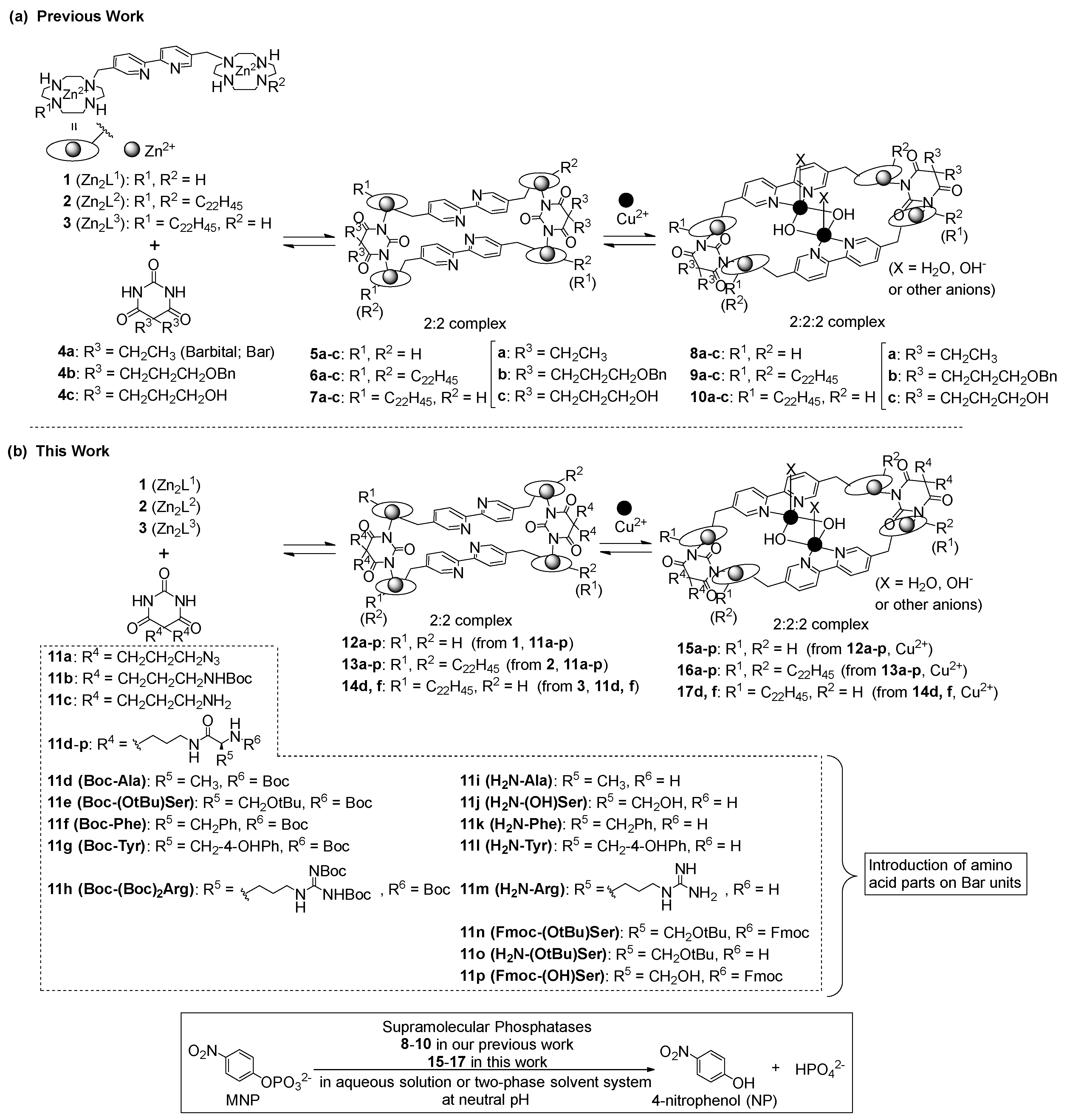
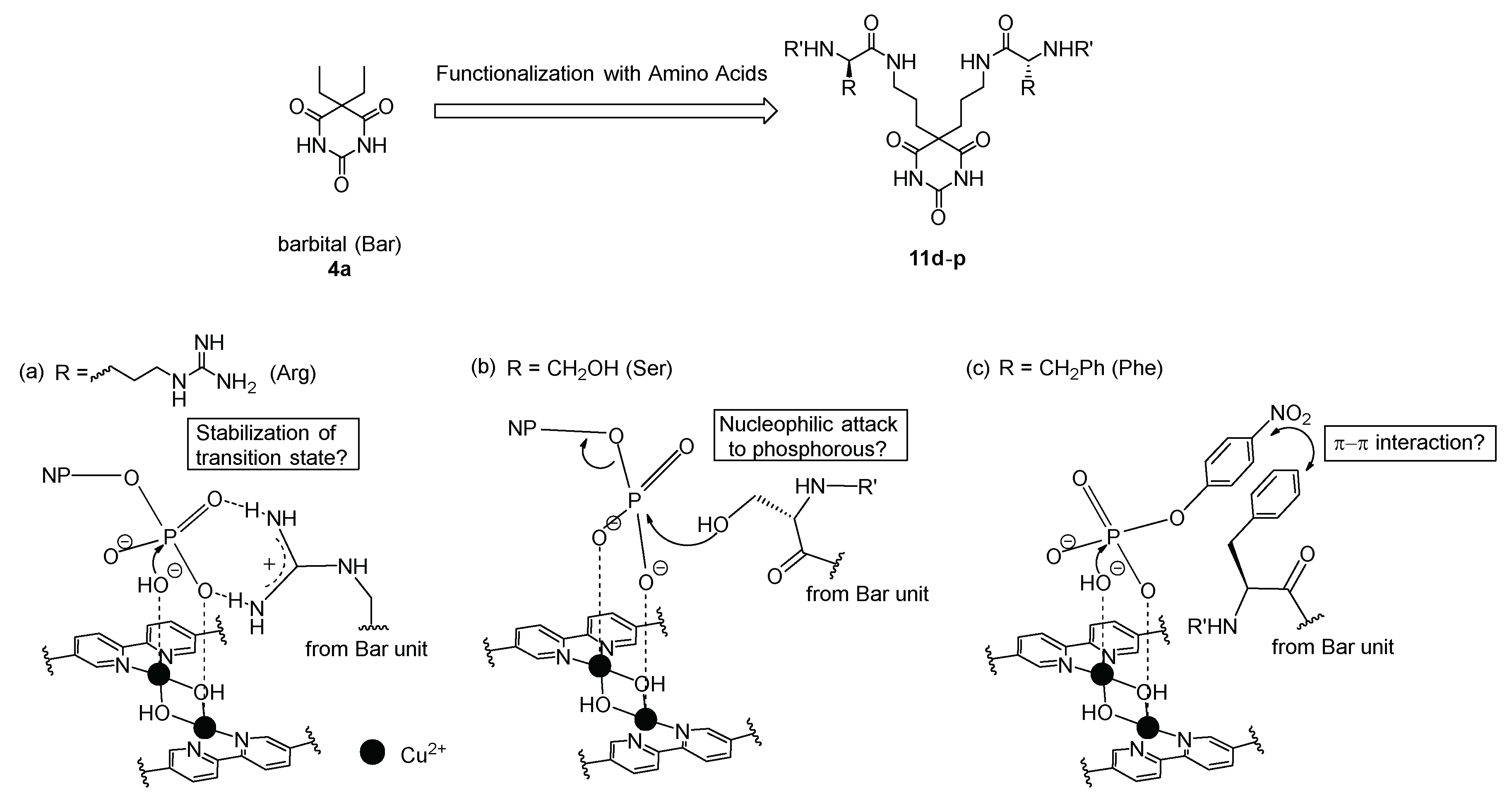
3.1. Synthesis of Barbital Derivatives
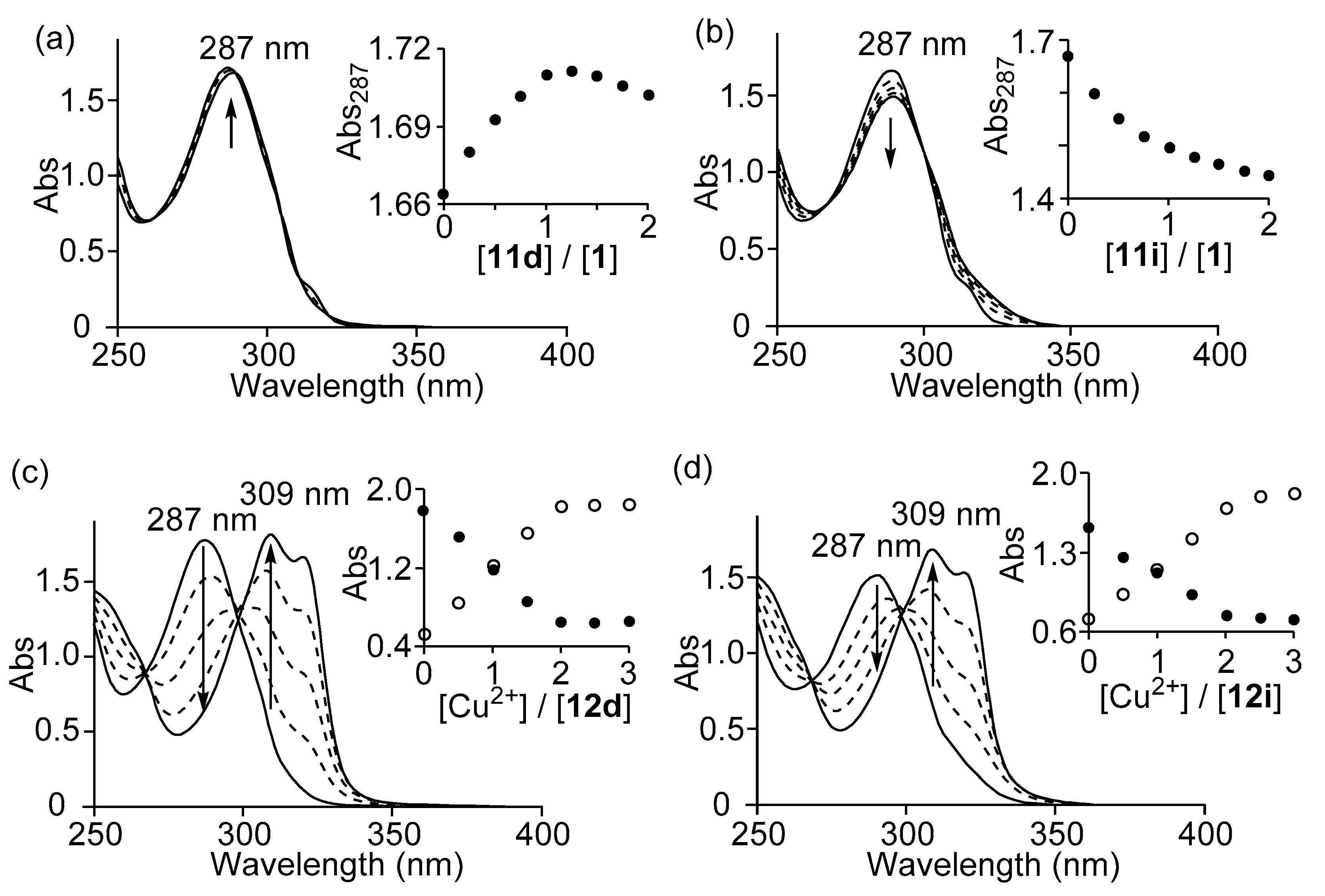
3.2. Complexation Behavior of 1 (Zn2L1) with Barbital Derivatives and Cu2+ by UV/Vis Titrations
3.3. Location of Complexes 13 and 16 in the Two-Phase Solvent System, as Determined by UV/Vis Spectra
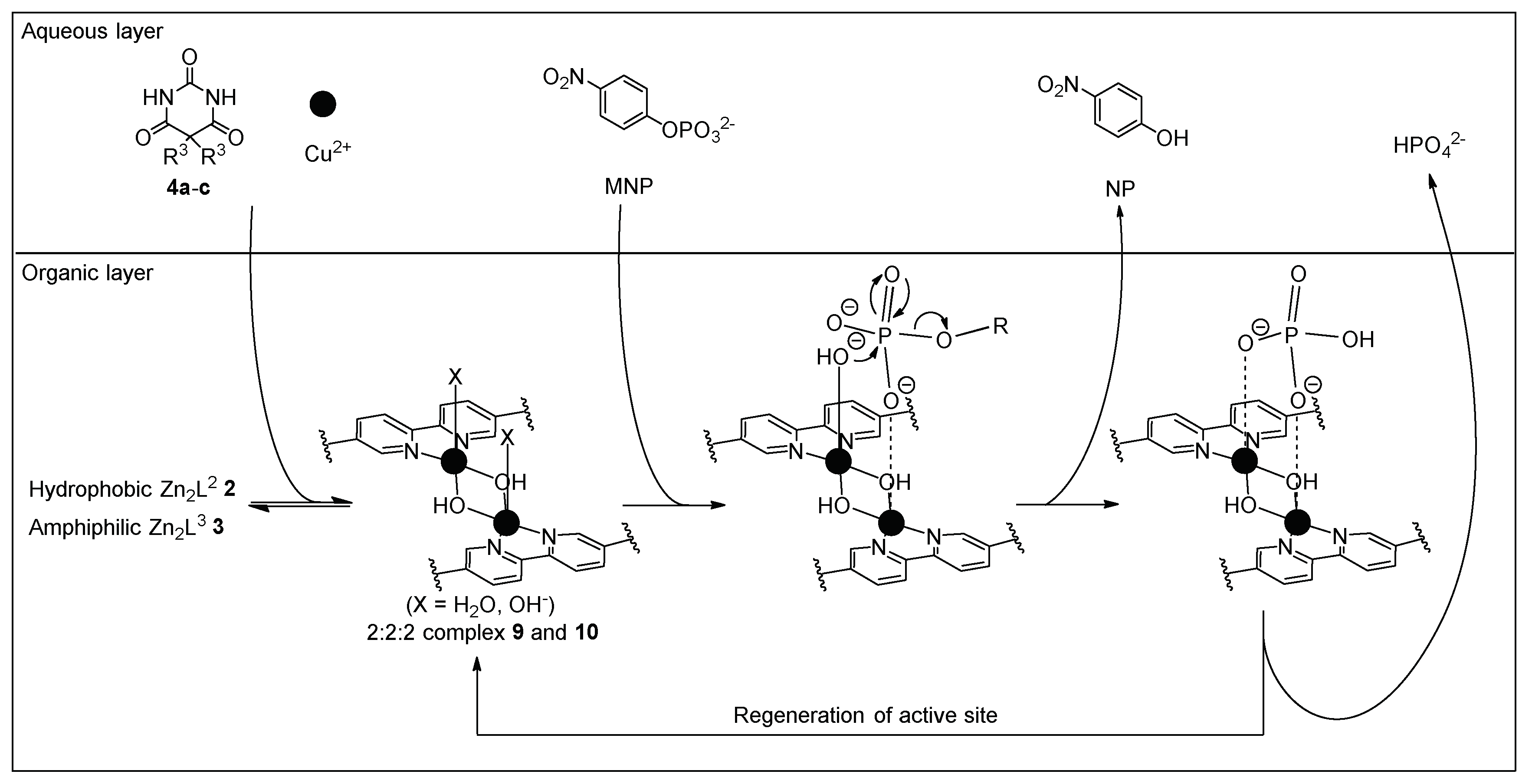
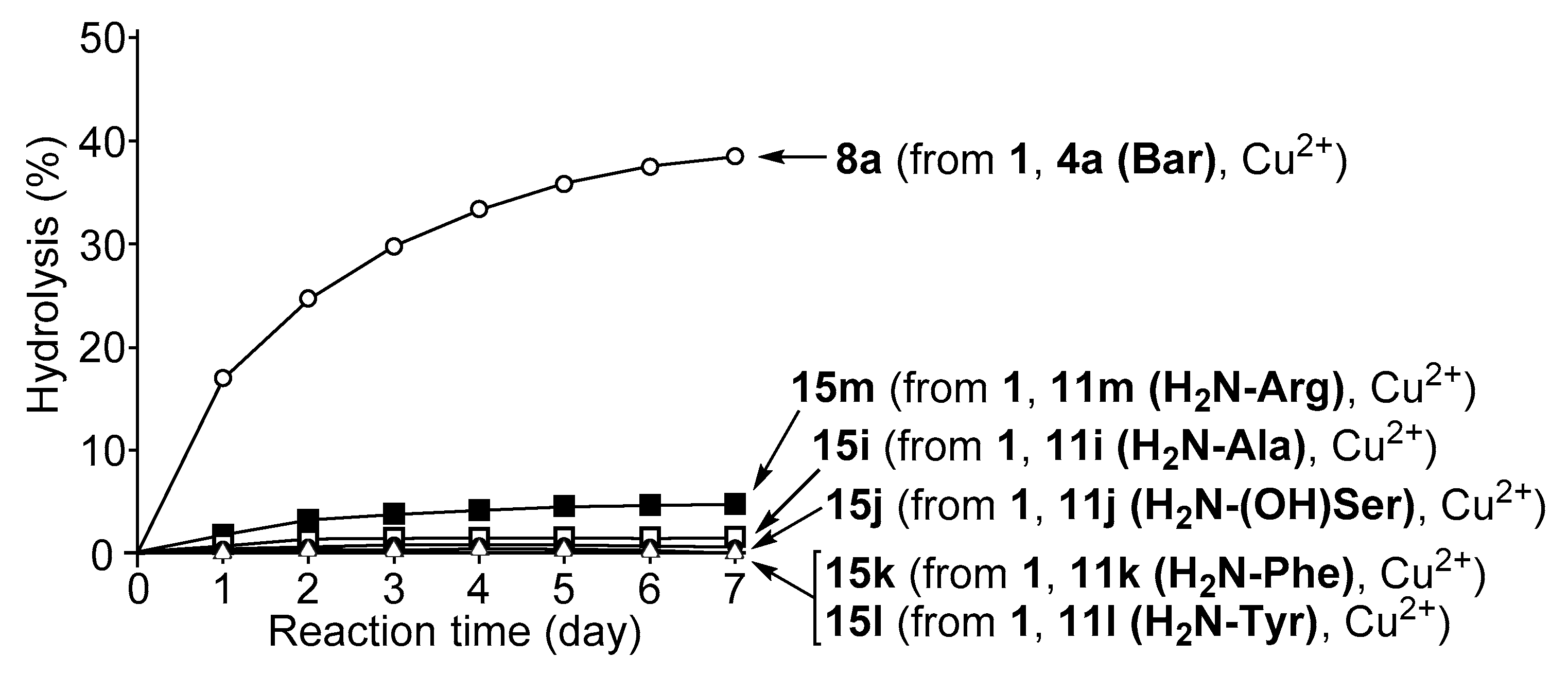
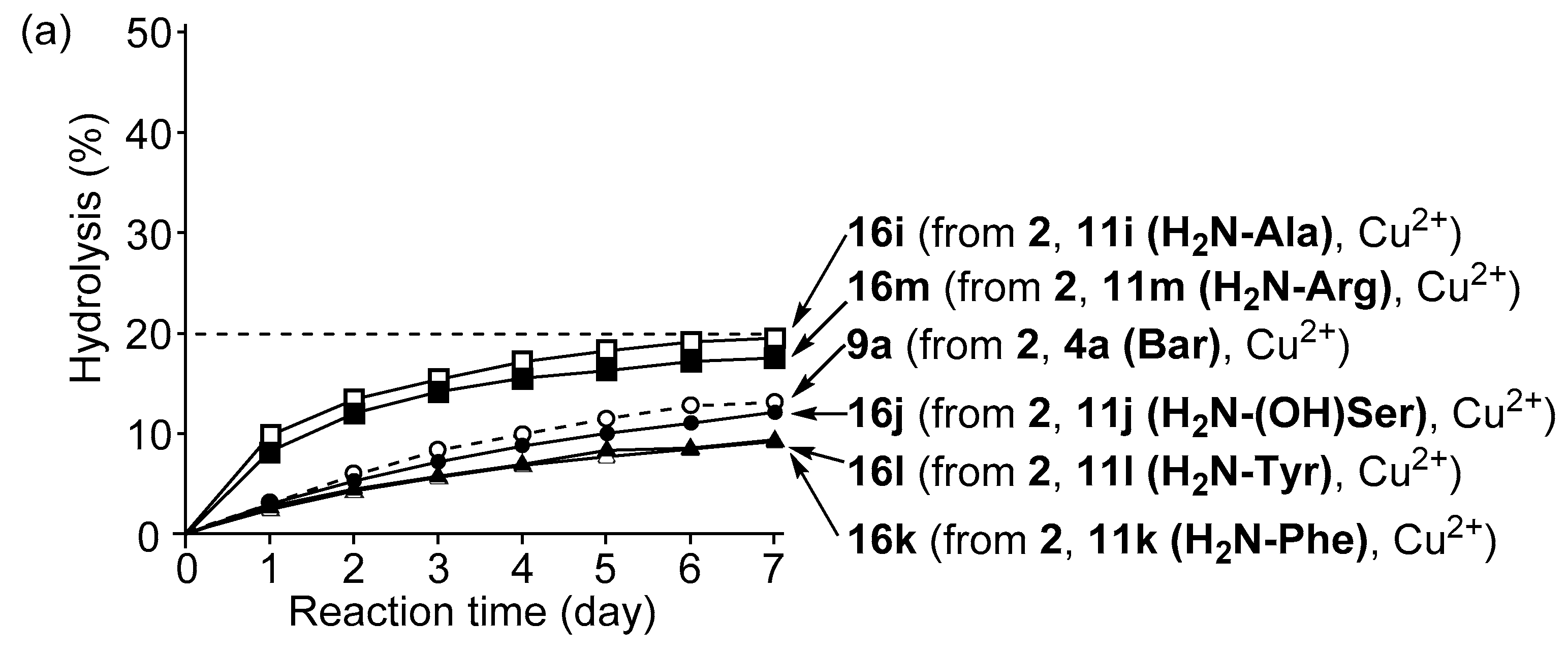
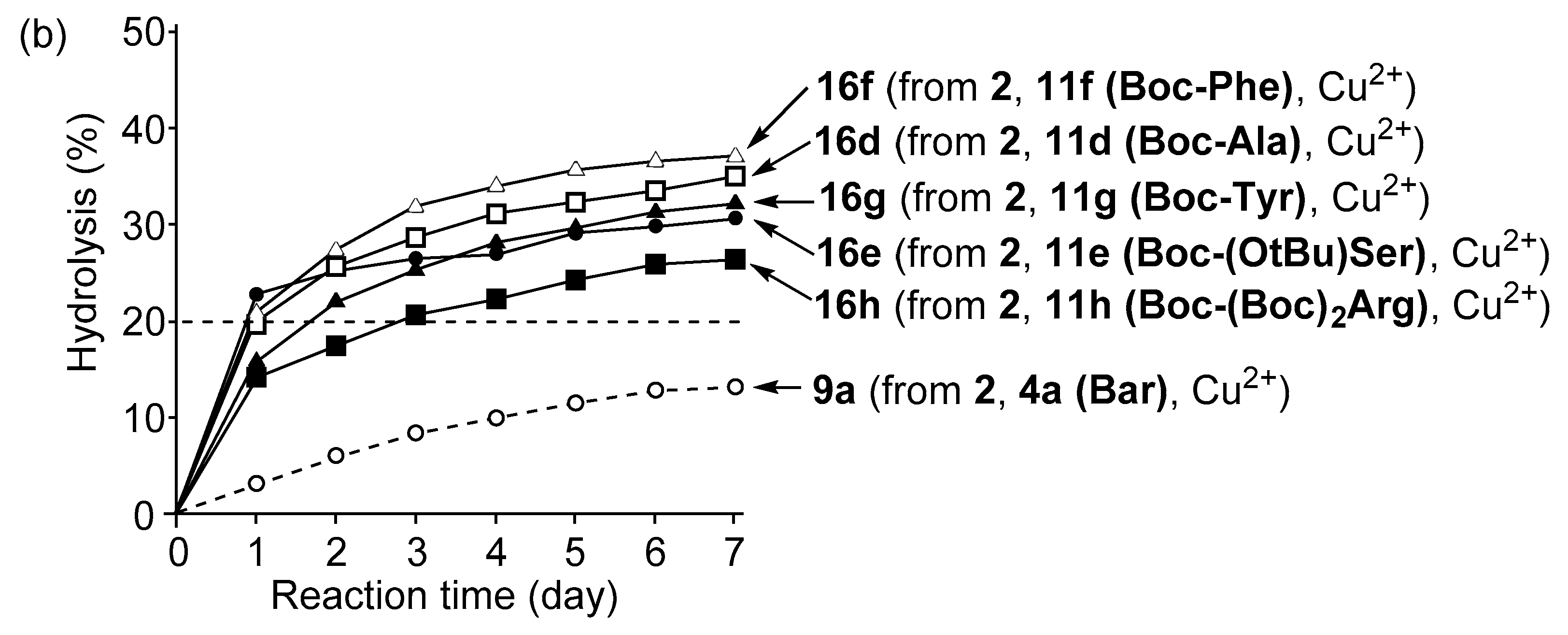
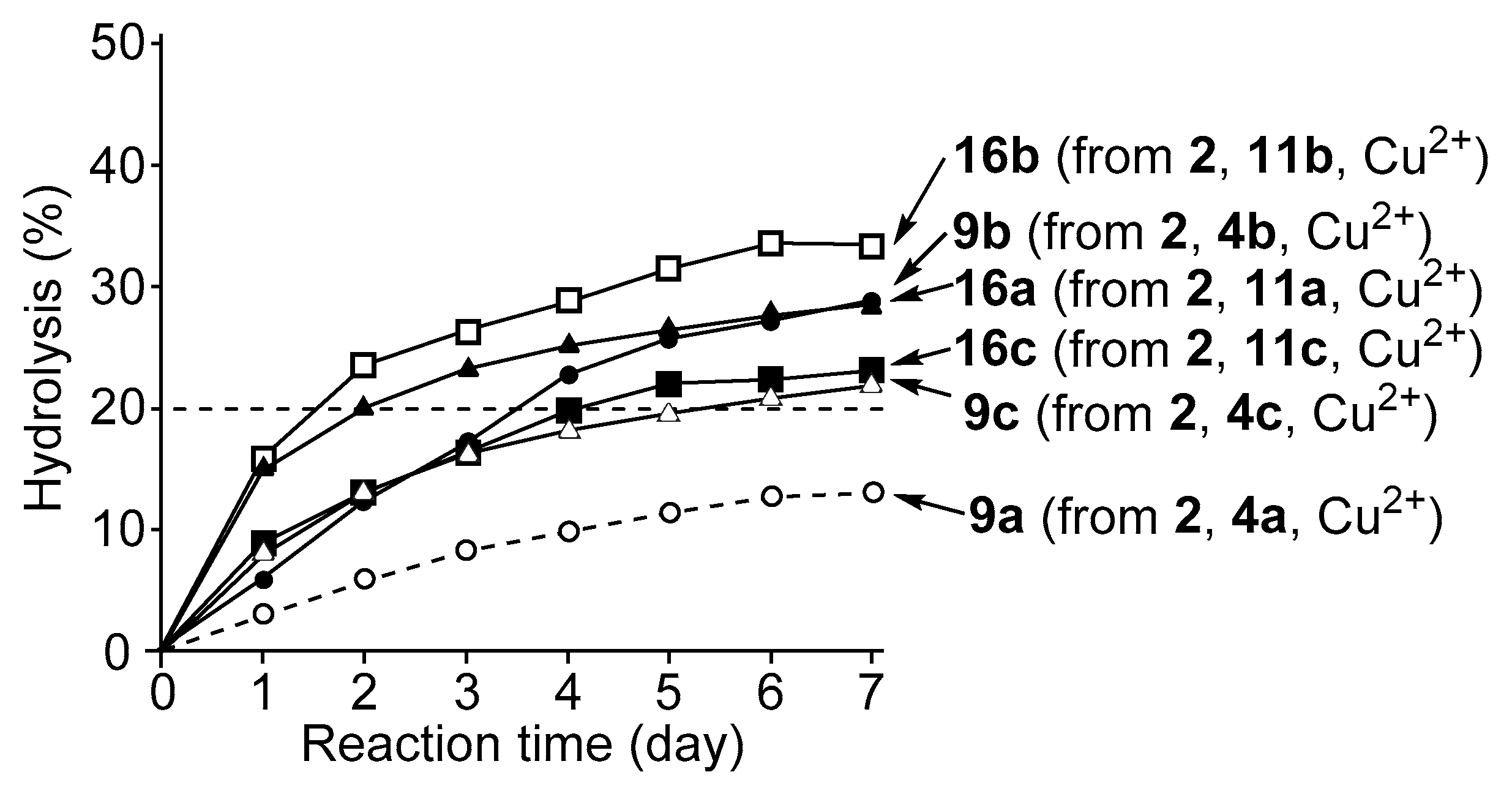
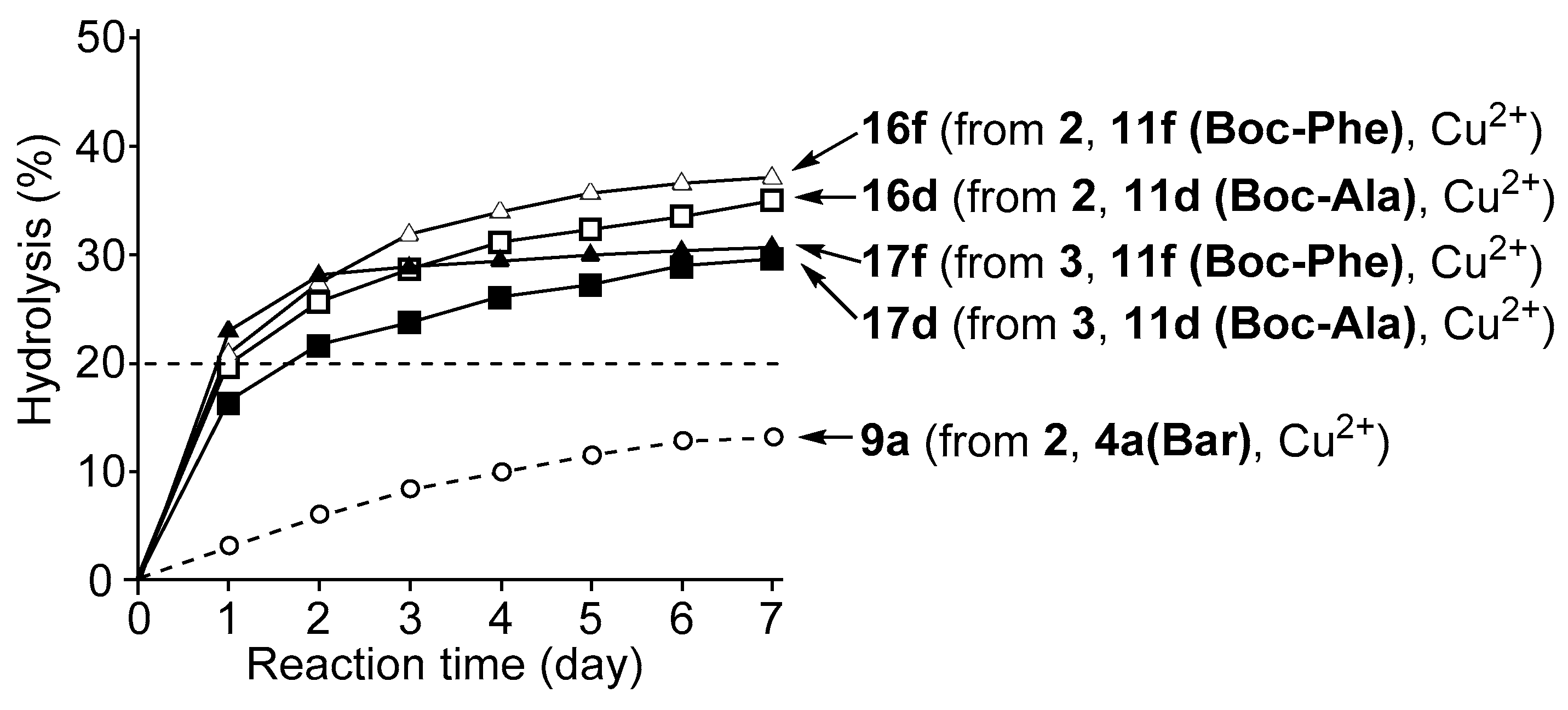
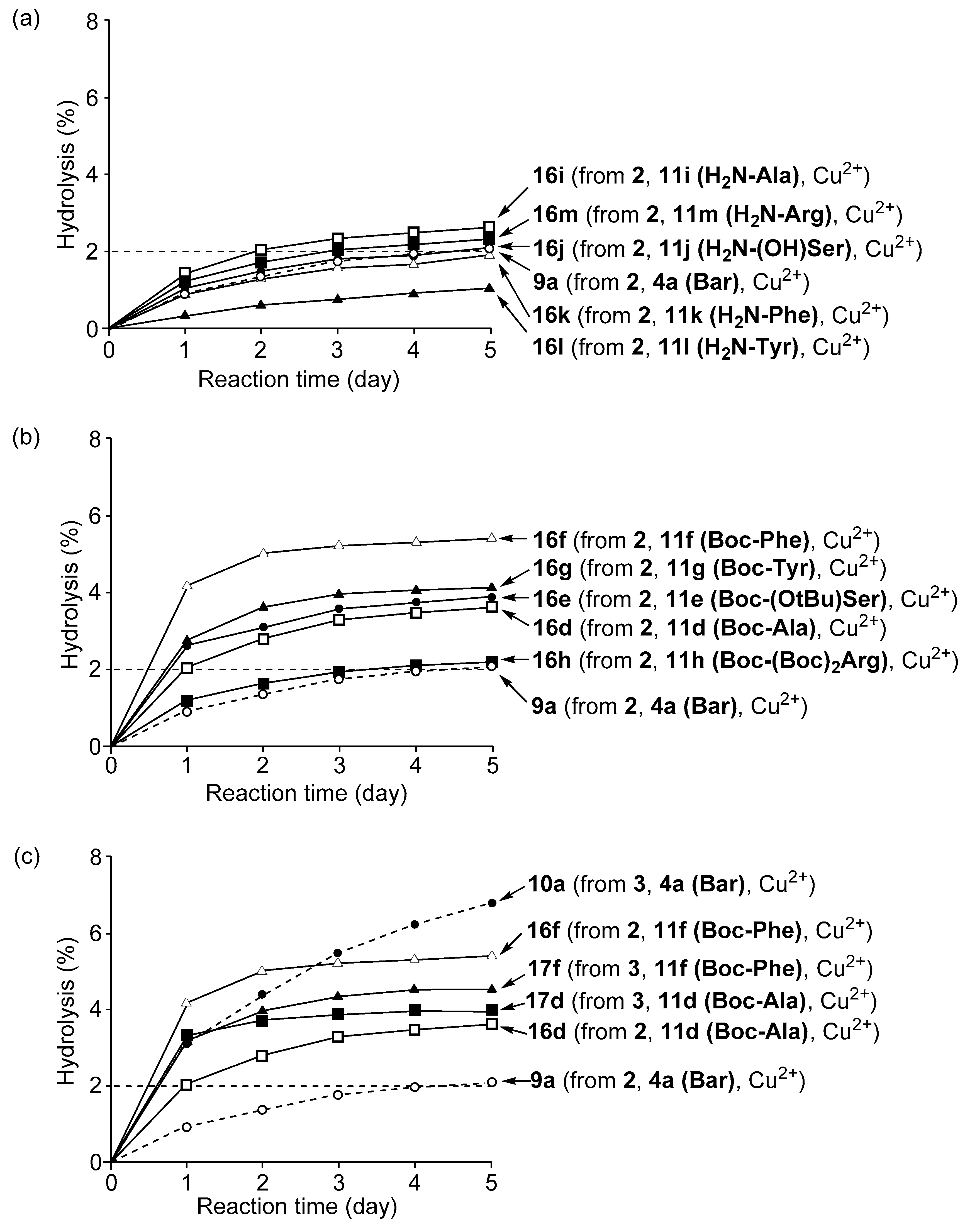
3.4. Hydrolysis of MNP by 2:2:2 Complexes in a Two-Phase Solvent System
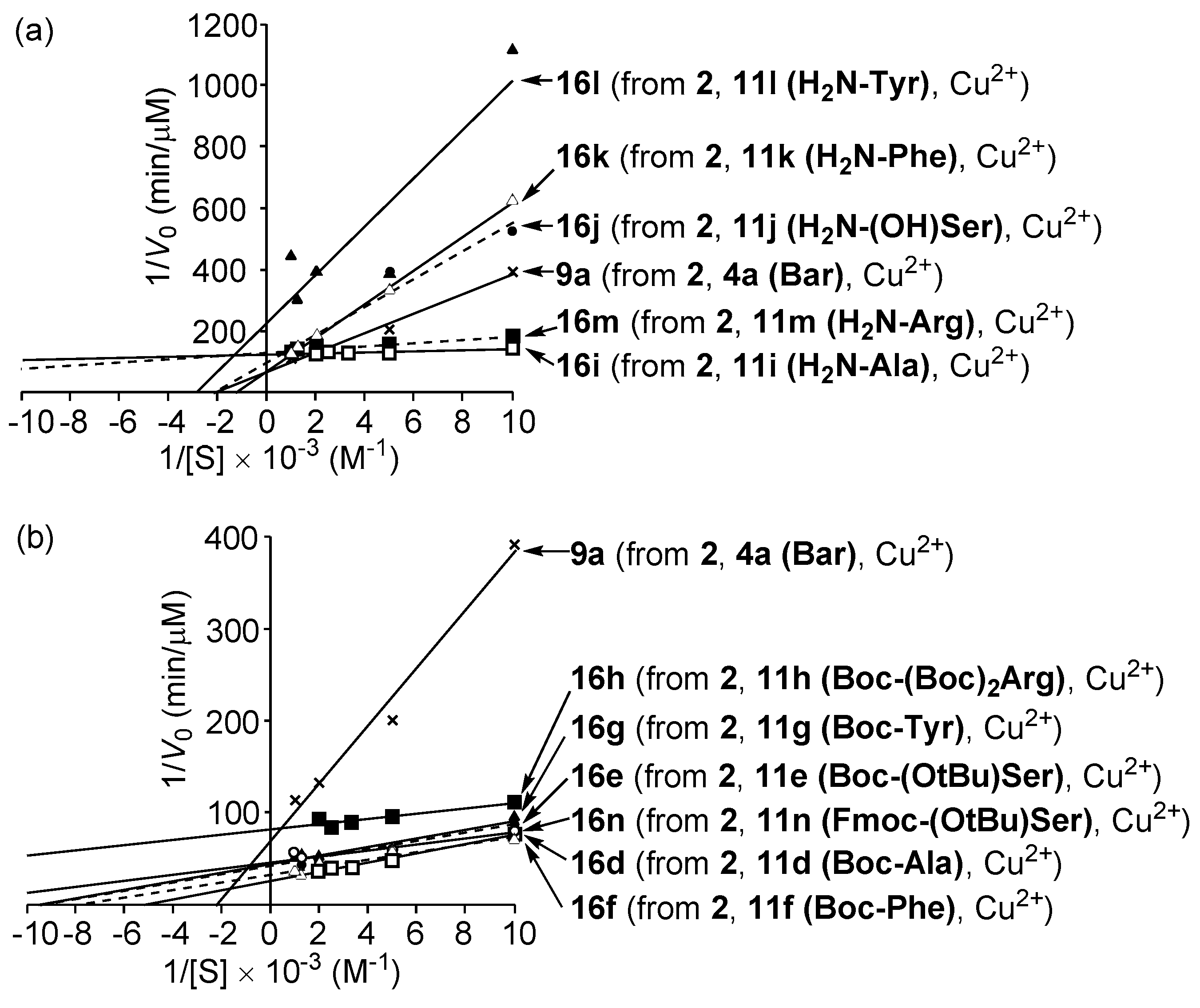
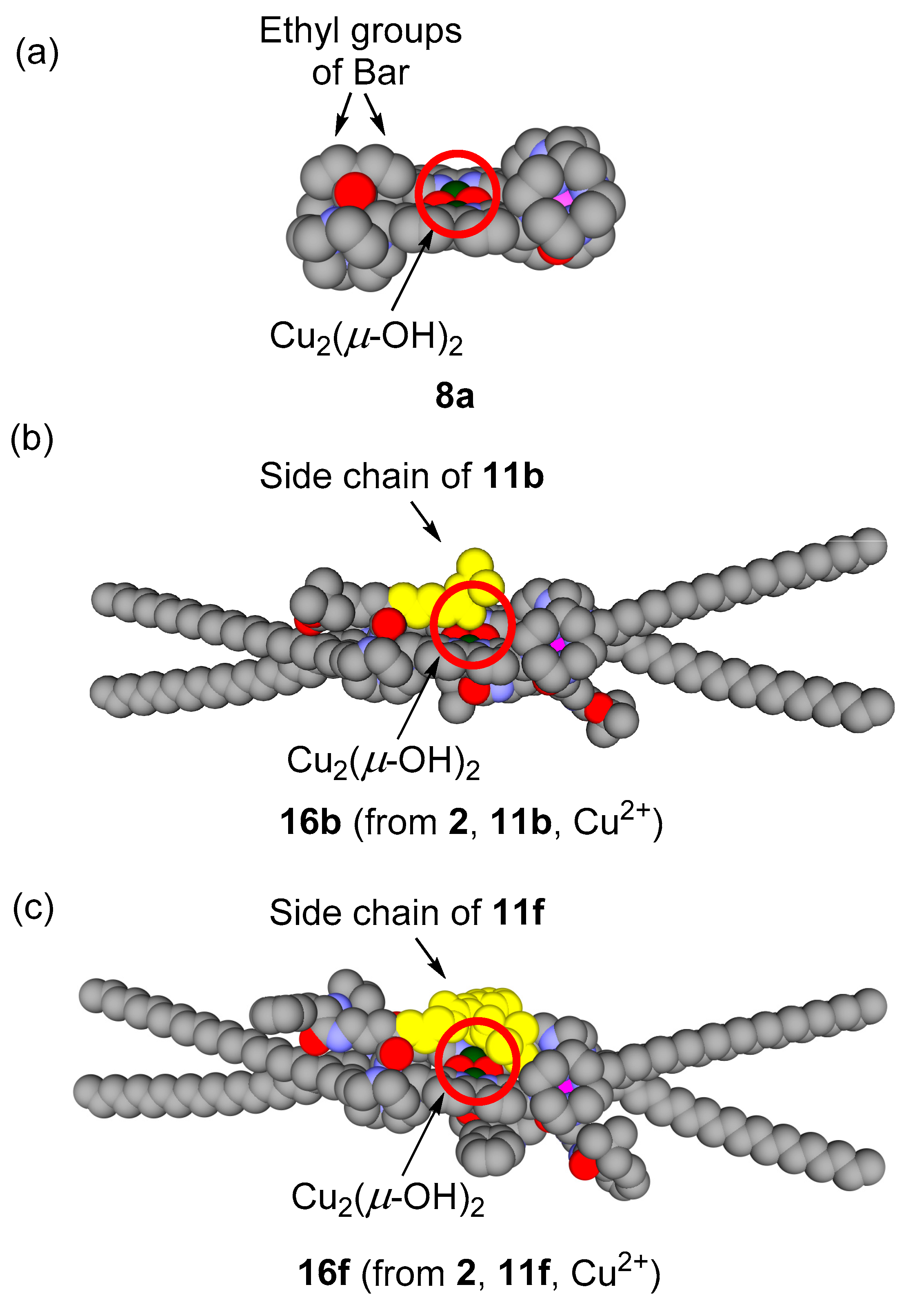
3.5. Michaelis–Menten Kinetics for Hydrolysis of MNP by 9, 16, and 17 in the Two-Phase Solvent System
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Woodgett, J. Protein Kinase Functions, Frontiers in Molecular Biology; Oxford University Press: Oxford, UK, 2000. [Google Scholar]
- Tibes, R.; Trent, J.; Kurzrock, R. Tyrosine kinase inhibitors and the dawn of molecular cancer therapeutics. Annu. Rev. Pharmacol. Toxicol. 2005, 45, 357–384. [Google Scholar] [CrossRef] [PubMed]
- Jiao, Q.; Bi, L.; Ren, Y.; Song, S.; Wang, Q.; Wang, Y. Advances in studies of tyrosine kinase inhibitors and their acquired resistance. Mol. Cancer 2018, 17, 36. [Google Scholar] [CrossRef] [PubMed]
- Coleman, J.E. Structure and mechanism of alkaline phosphatase. Annu. Rev. Biophys. Biomol. Struct. 1992, 21, 441–483. [Google Scholar] [CrossRef] [PubMed]
- Millan, J.L. Alkaline phosphatases structure, substrate specificity and functional relatedness to other members of a large superfamily of enzymes. Purinergic Signal. 2006, 2, 335–341. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, P.J.; Herschlag, D. Functional interrelationships in the alkaline phosphatase superfamily: Phosphodiesterase activity of Escherichia coli alkaline phosphatase. Biochemistry 2001, 40, 5691–5699. [Google Scholar] [PubMed]
- Denu, J.M.; Stuckey, J.A.; Saper, M.A.; Dixon, J.E. Form and function in protein dephosphorylation. Cell 1996, 87, 361–364. [Google Scholar] [CrossRef]
- Kramer, R. Bioinorganic models for the catalytic cooperation of metal ions and functional groups in nuclease and peptidase enzymes. Coord. Chem. Rev. 1999, 182, 243–261. [Google Scholar] [CrossRef]
- Cleland, W.W.; Hengge, A.C. Enzymatic mechanisms of phosphate and sulfate transfer. Chem. Rev. 2006, 106, 3252–3278. [Google Scholar] [CrossRef]
- Kim, E.E.; Wyckoff, H.W. Reaction mechanism of alkaline phosphatase based on crystal structures. J. Mol. Biol. 1991, 218, 449–464. [Google Scholar] [CrossRef]
- Kimura, E. Dimetallic hydrolases and their models. Curr. Opin. Chem. Biol. 2000, 4, 207–213. [Google Scholar] [CrossRef]
- Aoki, S.; Kimura, E. Bio-Coordination Chemistry in Comprehensive Coordination Chemistry II; Que, L., Jr., Tolman, W.B., Eds.; Elsevier: Amsterdam, The Netherlands, 2003; Volume 8, pp. 601–640. [Google Scholar]
- Seo, J.S.; Sung, N.-D.; Hynes, R.C.; Chin, J. Structure and reactivity of a dinuclear cobalt(III) complex with a bridging phosphate monoester. Inorg. Chem. 1996, 35, 7472–7473. [Google Scholar] [CrossRef]
- Williams, N.H.; Lebuis, A.-M.; Chin, J. A structural and functional model of dinuclear metallophosphatases. J. Am. Chem. Soc. 1999, 121, 3341–3348. [Google Scholar] [CrossRef]
- Williams, N.H.; Takasaki, B.; Wall, M.; Chin, J. Structure and nuclease activity of simple dinuclear metal complexes: Quantitative dissection of the role of metal ions. Acc. Chem. Res. 1999, 32, 485–493. [Google Scholar] [CrossRef]
- Vance, D.H.; Czarnik, A.W. Functional group convergency in a binuclear dephosphorylation reagent. J. Am. Chem. Soc. 1993, 115, 12165–12166. [Google Scholar] [CrossRef]
- Koike, T.; Inoue, M.; Kimura, E.; Shiro, M. Novel properties of cooperative dinuclear zinc(II) ions: The selective recognition of phosphomonoesters and their P−O ester bond cleavage by a new dinuclear zinc(II) cryptate. J. Am. Chem. Soc. 1996, 118, 3091–3099. [Google Scholar] [CrossRef]
- Hettich, R.; Schneider, H.-J. Cobalt(III) polyamine complexes as catalysts for the hydrolysis of phosphate esters and of DNA. A measurable 10 million-fold rate increase. J. Am. Chem. Soc. 1997, 119, 5638–5647. [Google Scholar] [CrossRef]
- Zulkefeli, M.; Suzuki, A.; Shiro, M.; Hisamatsu, Y.; Kimura, E.; Aoki, S. Selective hydrolysis of phosphate monoester by supramolecular phosphatase formed by the self-assembly of a Bis(Zn2+-cyclen) complex, cyanuric acid, and copper in an aqueous solution (cyclen = 1, 4, 7, 10-Tetraazacyclododecane). Inorg. Chem. 2011, 50, 10113–10123. [Google Scholar] [CrossRef] [PubMed]
- Der, B.S.; Edwards, D.R.; Kuhlman, B. Catalysis by a de novo zinc-mediated protein interface: Implications for natural enzyme evolution and rational enzyme engineering. Biochemistry 2012, 51, 3933–3940. [Google Scholar] [CrossRef]
- Zhang, X.; Zhu, Y.; Zheng, X.; Phillips, D.L.; Zhao, C. Mechanistic investigation on the cleavage of phosphate monoester catalyzed by unsymmetrical macrocyclic dinuclear complexes: The selection of metal centers and intrinsic flexibility of the ligand. Inorg. Chem. 2014, 53, 3354–3361. [Google Scholar] [CrossRef]
- Xue, S.-S.; Zhao, M.; Ke, Z.-F.; Cheng, B.-C.; Su, H.; Cao, H.; Cao, K.C.; Wang, J.; Ji, L.-N.; Mao, Z.-W. Enantioselective hydrolysis of amino acid esters promoted by Bis(ß-cyclodextrin) copper complexes. Sci. Rep. 2016, 6, 22080. [Google Scholar] [CrossRef]
- Lin, Y.-W. Rational design of metalloenzymes: From single to multiple active sites. Coord. Chem. Rev. 2017, 336, 1–27. [Google Scholar] [CrossRef]
- Zulkefeli, M.; Hisamatsu, Y.; Suzuki, A.; Miyazawa, Y.; Shiro, M.; Aoki, S. Supramolecular phosphatases formed by the self-assembly of the Bis(Zn2+-cyclen) complex, copper(II), and barbital derivatives in water. Chem. Asian J. 2014, 9, 2831–2841. [Google Scholar] [CrossRef] [PubMed]
- Coon, M.M.; Rebek, J., Jr. Self-assembling capsules. Chem. Rev. 1997, 97, 1647–1668. [Google Scholar] [CrossRef]
- Fujita, M. Molecular Self-Assembly Organic Versus Inorganic Approaches; Springer: Berlin, Germany, 2000; Volume 96. [Google Scholar]
- Breit, B. Supramolecular approaches to generate libraries of chelating bidentate ligands for homogeneous catalysis. Angewandte Chem. Int. Ed. 2005, 44, 6816–6825. [Google Scholar] [CrossRef]
- Kleij, A.W.; Reek, J.N.H. Ligand-template directed assembly: An efficient approach for the supramolecular encapsulation of transition-metal catalysts. Chem. Eur. J. 2006, 12, 4218–4227. [Google Scholar] [CrossRef] [PubMed]
- Oshovsky, G.V.; Reinhoudt, D.N.; Verboom, W. Supramolecular chemistry in water. Angewandte Chem. Int. Ed. 2007, 46, 2366–2393. [Google Scholar] [CrossRef] [PubMed]
- Hannon, M.J. Supramolecular DNA recognition. Chem. Soc. Rev. 2007, 36, 280–295. [Google Scholar] [CrossRef]
- Van Leeuwen, P.W.N.M. Supramolecular Catalysis; Wiley-VCH: New York, NY, USA, 2008. [Google Scholar]
- Koblenz, T.S.; Wassenaar, J.; Reek, J.N.H. Reactivity within a confined self-assembled nanospace. Chem. Soc. Rev. 2008, 37, 247–262. [Google Scholar] [CrossRef]
- Suzuki, K.; Tominaga, M.; Kawano, M.; Fujita, M. Self-assembly of an M6L12 coordination cube. Chem. Commun. 2009, 1638–1640. [Google Scholar] [CrossRef]
- Northrop, B.H.; Zheng, Y.-R.; Chi, K.-W.; Stang, P.J. Self-organization in coordination-driven self-assembly. Acc. Chem. Res. 2009, 42, 1554–1563. [Google Scholar] [CrossRef]
- Leung, K.C.-F.; Chak, C.-P.; Lo, C.-M.; Wong, W.-Y.; Xuan, S.; Cheng, C.H.K. pH-Controllable supramolecular systems. Chem. Asian J. 2009, 4, 364–381. [Google Scholar] [CrossRef] [PubMed]
- Yoshizawa, M.; Klosterman, J.K.; Fujita, M. Functional molecular flasks: New properties and reactions within discrete, self-assembled hosts. Angewandte Chem. Int. Ed. 2009, 48, 3418–3438. [Google Scholar] [CrossRef] [PubMed]
- Yoshizawa, M.; Fujita, M. Development of unique chemical phenomena within nanometer-sized, self-assembled coordination hosts. Bull. Chem. Soc. Jpn. 2010, 83, 609–618. [Google Scholar] [CrossRef]
- Trabolsi, A.; Khashab, N.; Fahrenbach, A.C.; Friedman, D.C.; Colvin, M.T.; Coti, K.K.; Benitez, D.; Tkatchouk, E.; Olsen, J.-C.; Belowich, M.E.; et al. Radically enhanced molecular recognition. Nat. Chem. 2010, 2, 42–49. [Google Scholar] [CrossRef]
- Meeuwissen, J.; Reek, J.N.H. Supramolecular catalysis beyond enzyme mimics. Nat. Chem. 2010, 2, 615–621. [Google Scholar] [CrossRef]
- Ma, Z.; Moulton, B. Recent advances of discrete coordination complexes and coordination polymers in drug delivery. Coord. Chem. Rev. 2011, 255, 1623–1641. [Google Scholar] [CrossRef]
- Aoki, S.; Zulkefeli, M.; Hisamatsu, Y.; Kitamura, M. Supramolecular host and catalysts formed by the synergistic molecular assembly of multinuclear zinc(II) complexes in aqueous solution. In Synergy in Supramolecular Chemistry; Nabeshima, T., Ed.; CRC: Boca Raton, FL, USA, 2015; pp. 33–56. [Google Scholar]
- Kimura, E.; Koike, T.; Aoki, S. Evolution of ZnII-macrocyclic polyamines to biological probes and supramolecular assembly elements. In Macrocyclic and Supramolecular Chemistry: How Izatt-Christensen Award Winners Shaped the Field; Izatt, R.M., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2016; pp. 417–445. [Google Scholar]
- Gibb, C.L.D.; Gibb, B.C. Well-defined, organic nanoenvironments in water: The hydrophobic effect drives a capsular assembly. J. Am. Chem. Soc. 2004, 126, 11408–11409. [Google Scholar] [CrossRef] [PubMed]
- Slagt, V.F.; Kamer, P.C.J.; Van Leeuwen, P.W.N.M.; Reek, J.N.H. Encapsulation of transition metal catalysts by ligand-template directed assembly. J. Am. Chem. Soc. 2004, 126, 1526–1536. [Google Scholar] [CrossRef] [PubMed]
- Kuil, M.; Soltner, T.; Van Leeuwen, P.W.N.M.; Reek, J.N.H. High-precision catalysts: Regioselective hydroformylation of internal alkenes by encapsulated rhodium complexes. J. Am. Chem. Soc. 2006, 128, 11344–11345. [Google Scholar] [CrossRef] [PubMed]
- Neelakandan, P.P.; Hariharan, M.; Ramaiah, D. A supramolecular ON−OFF−ON fluorescence assay for selective recognition of GTP. J. Am. Chem. Soc. 2006, 128, 11334–11335. [Google Scholar] [CrossRef] [PubMed]
- Nishioka, Y.; Yamaguchi, T.; Yoshizawa, M.; Fujita, M. Unusual [2+4] and [2+2] cycloadditions of arenes in the confined cavity of self-assembled cages. J. Am. Chem. Soc. 2007, 129, 7000–7001. [Google Scholar] [CrossRef] [PubMed]
- Natarajan, A.; Kaanumalle, L.S.; Jockusch, S.; Gibb, C.L.D.; Gibb, B.C.; Turro, N.J.; Ramamurthy, V. Controlling photoreactions with restricted spaces and weak intermolecular forces: Exquisite selectivity during oxidation of olefins by singlet oxygen. J. Am. Chem. Soc. 2007, 129, 4132–4133. [Google Scholar] [CrossRef] [PubMed]
- Sawada, T.; Yoshizawa, M.; Sato, S.; Fujita, M. Minimal nucleotide duplex formation in water through enclathration in self-assembled hosts. Nat. Chem. 2009, 1, 53–56. [Google Scholar] [CrossRef] [PubMed]
- Gianneschi, N.C.; Nguyen, S.T.; Mirkin, C.A. Signal amplification and detection via a supramolecular allosteric catalyst. J. Am. Chem. Soc. 2005, 127, 1644–1645. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Cho, S.H.; Mulfort, K.L.; Tiede, D.M.; Hupp, J.T.; Nguyen, S.T. Cavity-tailored, self-sorting supramolecular catalytic boxes for selective oxidation. J. Am. Chem. Soc. 2008, 130, 16828–16829. [Google Scholar] [CrossRef]
- Murase, T.; Horiuchi, S.; Fujita, M. Naphthalene Diels−Alder in a self-assembled molecular flask. J. Am. Chem. Soc. 2010, 132, 2866–2867. [Google Scholar] [CrossRef]
- Hisamatsu, Y.; Miyazawa, Y.; Yoneda, K.; Miyauchi, M.; Zulkefeli, M.; Aoki, S. Supramolecular complexes formed by the self-assembly of hydrophobic Bis(Zn2+-cyclen) complexes, copper, and Di- or Triimide units for the hydrolysis of phosphate Mono- and diesters in two-phase solvent systems (Cyclen = 1, 4, 7, 10-Tetraazacyclododecane). Chem. Pharm. Bull. 2016, 64, 451–464. [Google Scholar] [CrossRef][Green Version]
- Rahman, A.B.; Imafuku, Y.; Miyazawa, Y.; Kalfe, A.; Sakai, H.; Saga, Y.; Aoki, S. Catalytic hydrolysis of phosphate monoester by supramolecular phosphatases formed from a monoalkylated dizinc(II) complex, cyclic diimide units, and copper(II) in two-phase solvent system. Inorg. Chem. 2019, 58, 5603–5616. [Google Scholar] [CrossRef]
- Nilsson, B.L.; Soellner, M.B.; Raines, R.T. Chemical synthesis of proteins. Annu. Rev. Biophys. Biomol. Struct. 2005, 34, 91–118. [Google Scholar] [CrossRef]
- Van Berkel, S.S.; Van Eldijk, M.B.; Van Hest, J.C.M. Staudinger ligation as a method for bioconjugation. Angewandte Chem. Int. Ed. 2011, 50, 8806–8827. [Google Scholar] [CrossRef]
- Sagel, I.H. Biochemical Calculations, 2nd ed.; John Wiley & Sons: New York, NY, USA, 1976. [Google Scholar]



| - | In H2O Layer (%) | In CHCl3 Layer (%) |
|---|---|---|
| 5aa | >98 | <2 |
| 6a | 1 | 99 |
| 7aa | 35 | 65 |
| 13d | 2 | 98 |
| 13f | 4 | 96 |
| 13i | <1 | >99 |
| 13k | <1 | >99 |
| Entry | Cat. a | Vmax (μM min−1) | Km (μM) | k (min−1) b | Ki (μM) | Km/Ki | CTN c |
|---|---|---|---|---|---|---|---|
| 1 | 8ad | (8.9 ± 0.2) × 10−2 d | (4.1 ± 0.3) × 102 d | (2.2 ± 0.2) × 10−4 d | ca. 15 (mixed-type) d | ca. 27 | 0.4 |
| 2 | APe | 1.3 ± 0.1 e | 7 ± 4 e | (2.9 ± 1.8) × 10−1 e | 3 ± 1 (competitive) e | ca. 2.3 | >103 f |
| 3 | 9ag | (1.4 ± 0.4) × 10−2 g | (5.4 ± 0.5) × 102 g | (2.7 ± 1.0) × 10−5 g | ca. 15 (competitive) g,h | ca. 36 | 1.0 |
| 4 | 10ah | (6.8 ± 0.3) × 10−2 h | (3.8 ± 0.2) × 102 h | (1.8 ± 0.2) × 10−4 h | ca. 80 (mixed-type) h | ca. 4.8 h | ~4 |
| 5 | 16bi | (3.6 ± 0.2) × 10−2 | (2.5 ± 0.3) × 102 | (1.4 ± 0.3) × 10−4 | n.d. j | n.d. j | 2.1 |
| 6 | 16di | (3.9 ± 0.2) × 10−2 | (1.9 ± 0.1) × 102 | (2.1 ± 0.2) × 10−4 | 16 (competitive) | ca. 12 | 1.8 |
| 7 | 16ei | (2.4 ± 0.2) × 10−2 | (1.1 ± 0.1) × 102 | (2.2 ± 0.4) × 10−4 | n.d. j | n.d. j | 2.0 |
| 8 | 16fi | (2.9 ± 0.1) × 10−2 | (1.2 ± 0.1) × 102 | (2.4 ± 0.3) × 10−4 | 23 (competitive) | ca. 5.4 | 2.7 |
| 9 | 16gi | (2.3 ± 0.2) × 10−2 | (1.0 ± 0.1) × 102 | (2.4 ± 0.4) × 10−4 | n.d. j | n.d. j | 2.1 |
| 10 | 16hi | (1.2 ± 0.1) × 10−2 | 33 ± 1 | (3.6 ± 0.5) × 10−4 | n.d. j | n.d. j | 1.1 |
| 11 | 16ii | (8.1 ± 0.2) × 10−3 | 13 ± 1 | (6.0 ± 0.4) × 10−4 | 0.67 (competitive) | ca. 20 | 1.3 |
| 12 | 16ji | (1.0 ± 0.1) × 10−2 | (4.7 ± 0.2) × 102 | (2.2 ± 0.2) × 10−5 | n.d. j | n.d. j | 1.2 |
| 13 | 16ki | (1.4 ± 0.1) × 10−2 | (7.6 ± 0.2) × 102 | (1.9 ± 0.2) × 10−5 | n.d. j | n.d. j | 1.0 |
| 14 | 16li | (4.5 ± 0.2) × 10−3 | (3.5 ± 0.1) × 102 | (1.3 ± 0.1) × 10−5 | n.d. j | n.d. j | 0.6 |
| 15 | 16mi | (7.8 ± 0.3) × 10−3 | 40 ± 1 | (2.0 ± 0.1) × 10−4 | n.d. j | n.d. j | 1.2 |
| 16 | 16ni | (2.2 ± 0.2) × 10−2 | 72 ± 2 | (3.0 ± 0.3) × 10−4 | n.d. j | n.d. j | 1.7 |
| 17 | 17di | (2.5 ± 0.2) × 10−2 | (1.2 ± 0.1) × 102 | (2.2 ± 0.4) × 10−4 | 17 (mixed-type) | ca. 7.4 | 2.0 |
| 18 | 17fi | (2.3 ± 0.2) × 10−2 | 47 ± 2 | (5.0 ± 0.6) × 10−4 | 5.7 (competitive) | ca. 8.3 | 2.3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miyazawa, Y.; Rahman, A.B.; Saga, Y.; Imafuku, H.; Hisamatsu, Y.; Aoki, S. Catalytic Hydrolysis of Phosphate Monoester by Supramolecular Complexes Formed by the Self-Assembly of a Hydrophobic Bis(Zn2+-cyclen) Complex, Copper, and Barbital Units That Are Functionalized with Amino Acids in a Two-Phase Solvent System. Micromachines 2019, 10, 452. https://doi.org/10.3390/mi10070452
Miyazawa Y, Rahman AB, Saga Y, Imafuku H, Hisamatsu Y, Aoki S. Catalytic Hydrolysis of Phosphate Monoester by Supramolecular Complexes Formed by the Self-Assembly of a Hydrophobic Bis(Zn2+-cyclen) Complex, Copper, and Barbital Units That Are Functionalized with Amino Acids in a Two-Phase Solvent System. Micromachines. 2019; 10(7):452. https://doi.org/10.3390/mi10070452
Chicago/Turabian StyleMiyazawa, Yuya, Akib Bin Rahman, Yutaka Saga, Hiroki Imafuku, Yosuke Hisamatsu, and Shin Aoki. 2019. "Catalytic Hydrolysis of Phosphate Monoester by Supramolecular Complexes Formed by the Self-Assembly of a Hydrophobic Bis(Zn2+-cyclen) Complex, Copper, and Barbital Units That Are Functionalized with Amino Acids in a Two-Phase Solvent System" Micromachines 10, no. 7: 452. https://doi.org/10.3390/mi10070452
APA StyleMiyazawa, Y., Rahman, A. B., Saga, Y., Imafuku, H., Hisamatsu, Y., & Aoki, S. (2019). Catalytic Hydrolysis of Phosphate Monoester by Supramolecular Complexes Formed by the Self-Assembly of a Hydrophobic Bis(Zn2+-cyclen) Complex, Copper, and Barbital Units That Are Functionalized with Amino Acids in a Two-Phase Solvent System. Micromachines, 10(7), 452. https://doi.org/10.3390/mi10070452