Integrating Microfabrication into Biological Investigations: the Benefits of Interdisciplinarity
Abstract
1. Introduction
2. Development of Microfabrication for Biological Research: A Brief Historical Perspective
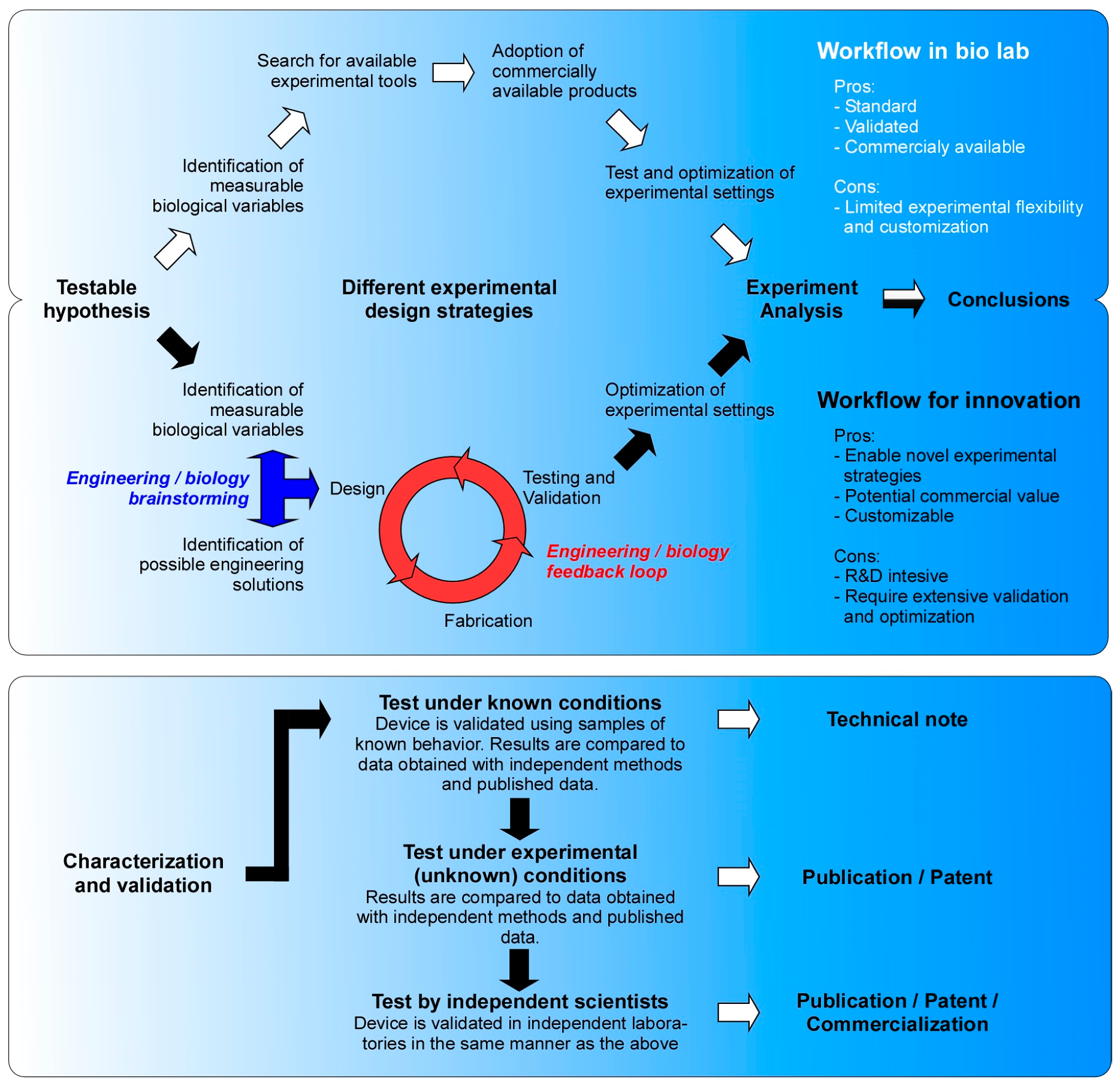
3. Working in an Interdisciplinary Environment: Bridging Biologists and Engineers
4. Microfabrication for Biological Investigations: A Few Examples from Our Toolbox
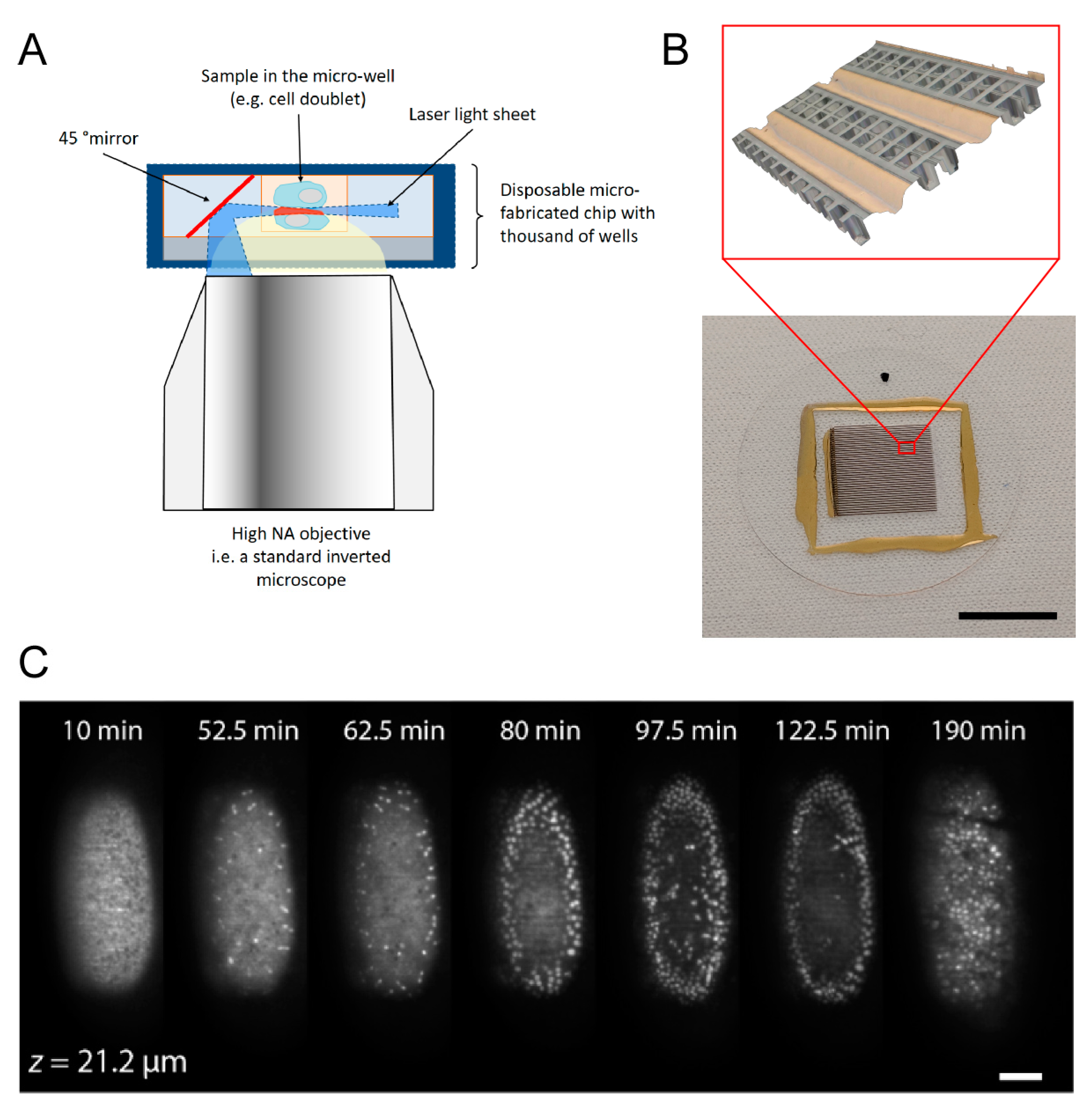
- soSPIM: a light sheet microscopy system that needs only one single objective with high numerical aperture owing to the use of microfabricated disposable chips;
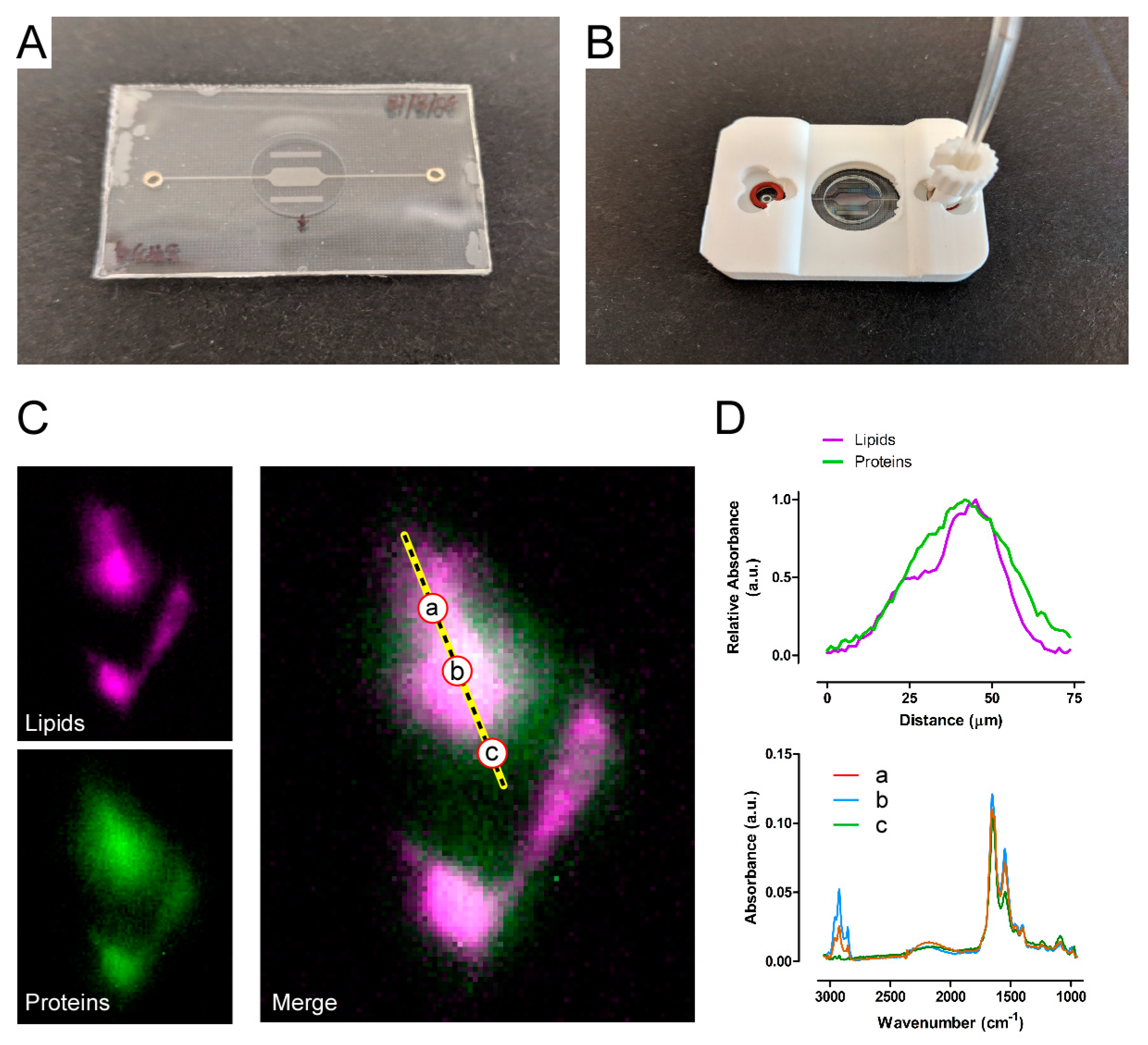
- IR-live: a microfluidic platform for label-free chemical imaging of live cells by infra-red absorption spectroscopy;
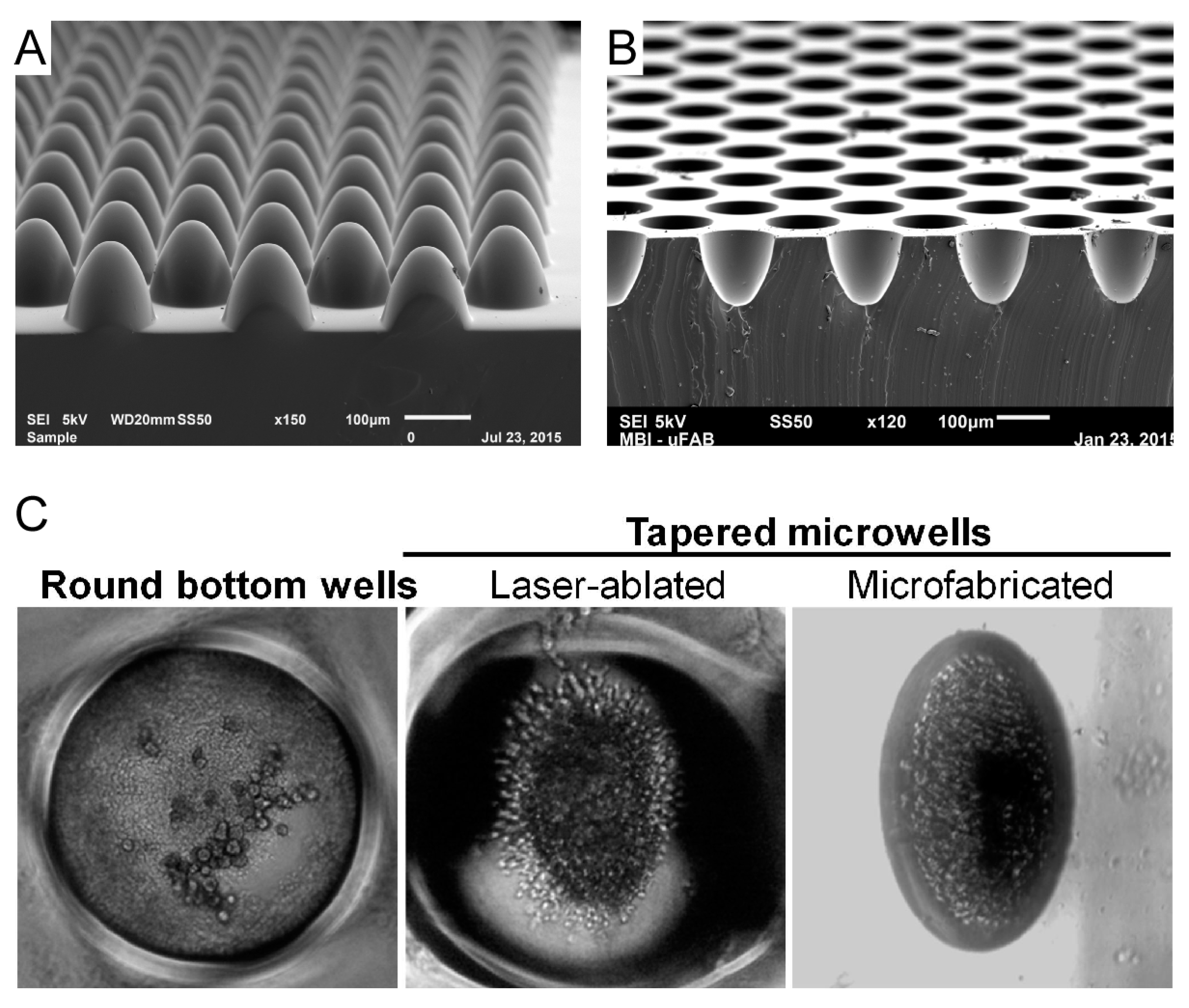
- Microfabricated microwells for expansion of circulating tumor cells (CTC).
4.1. soSPIM: Advanced Microscopy on A Chip
4.2. IR-live: Infra-red Spectro-microscopy On Live Cells
4.3. Microfabricated Microwells for Expansion of Circulating Tumor Cells (CTC)
5. Concluding Remarks
6. Patents
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Novikoff, A.B. The concept of integrative levels and biology. Science 1945, 101, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Regenmortel, M.H.V.V. Reductionism and complexity in molecular biology. EMBO Rep. 2004, 5, 1016. [Google Scholar] [CrossRef]
- Ahn, A.C.; Tewari, M.; Poon, C.-S.; Phillips, R.S. The Limits of Reductionism in Medicine: Could Systems Biology Offer an Alternative? PLOS Med. 2006, 3, e208. [Google Scholar] [CrossRef] [PubMed]
- Hughes, J.H.; Kumar, S. Synthetic mechanobiology: Engineering cellular force generation and signaling. Curr. Opin. Biotechnol. 2016, 40, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Black, J.B.; Perez-Pinera, P.; Gersbach, C.A. Mammalian Synthetic Biology: Engineering Biological Systems. Annu. Rev. Biomed. Eng. 2017, 19, 249–277. [Google Scholar] [CrossRef]
- Iskratsch, T.; Wolfenson, H.; Sheetz, M.P. Appreciating force and shape-the rise of mechanotransduction in cell biology. Nat. Rev. Mol. Cell Biol. 2014, 15, 825–833. [Google Scholar] [CrossRef]
- Karczewski, K.J.; Snyder, M.P. Integrative omics for health and disease. Nat. Rev. Genet. 2018, 19, 299–310. [Google Scholar] [CrossRef]
- Sosa-Hernandez, J.E.; Villalba-Rodriguez, A.M.; Romero-Castillo, K.D.; Aguilar-Aguila-Isaias, M.A.; Garcia-Reyes, I.E.; Hernandez-Antonio, A.; Ahmed, I.; Sharma, A.; Parra-Saldivar, R.; Iqbal, H.M.N. Organs-on-a-Chip Module: A Review from the Development and Applications Perspective. Micromachines 2018, 9, 536. [Google Scholar] [CrossRef] [PubMed]
- Kimura, H.; Sakai, Y.; Fujii, T. Organ/body-on-a-chip based on microfluidic technology for drug discovery. Drug Metab. Pharmacokinet. 2018, 33, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhao, Z.; Abdul Rahim, N.A.; van Noort, D.; Yu, H. Towards a human-on-chip: Culturing multiple cell types on a chip with compartmentalized microenvironments. Lab Chip 2009, 9, 3185–3192. [Google Scholar] [CrossRef]
- Yum, K.; Hong, S.G.; Healy, K.E.; Lee, L.P. Physiologically relevant organs on chips. Biotechnol. J. 2014, 9, 16–27. [Google Scholar] [CrossRef]
- Sia, D.; Llovet, J.M. Translating ‘–omics’ results into precision medicine for hepatocellular carcinoma. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 571. [Google Scholar] [CrossRef] [PubMed]
- Reddy, B.; Hassan, U.; Seymour, C.; Angus, D.C.; Isbell, T.S.; White, K.; Weir, W.; Yeh, L.; Vincent, A.; Bashir, R. Point-of-care sensors for the management of sepsis. Nat. Biomed. Eng. 2018, 2, 640–648. [Google Scholar] [CrossRef]
- Food Drug Administration Center for Drugs Evaluation Research. Self-MonitoringBlood Glucose Test Systems for Over-the-Counter Use; Food Drug Administration Center for Drugs Evaluation Research: Silver Spring, MD, USA, 2016.
- Moore, G.E. Cramming more components onto integrated circuits, Reprinted from Electronics, volume 38, number 8, April 19, 1965, pp.114 ff. IEEE Solid-State Circuits Soc. Newsl. 2006, 11, 33–35. [Google Scholar] [CrossRef]
- Ronse, K. Optical lithography—A historical perspective. C. R. Phys. 2006, 7, 844–857. [Google Scholar] [CrossRef]
- Cerrina, F. X-ray imaging: Applications to patterning and lithography. J. Phys. D Appl. Phys. 2000, 33, R103–R116. [Google Scholar] [CrossRef]
- Tormen, M.; Grenci, G.; Marmiroli, B.; Romanato, F. X-ray Lithography. In Nano-Lithography; Landis, S., Ed.; Wiley Online Books: Hoboken, NJ, USA, 2013; Volume 1, pp. 1–81. [Google Scholar]
- Xia, Y.; Whitesides, G.M. Soft Lithography. Angew. Chem. 1998, 37, 550–575. [Google Scholar] [CrossRef]
- Duffy, D.C.; McDonald, J.C.; Schueller, O.J.; Whitesides, G.M. Rapid Prototyping of Microfluidic Systems in Poly(dimethylsiloxane). Anal. Chem. 1998, 70, 4974–4984. [Google Scholar] [CrossRef]
- Kane, R.S.; Takayama, S.; Ostuni, E.; Ingber, D.E.; Whitesides, G.M. Patterning proteins and cells using soft lithography. Biomaterials 1999, 20, 2363–2376. [Google Scholar] [CrossRef]
- Xia, Y.; Rogers, J.A.; Paul, K.E.; Whitesides, G.M. Unconventional Methods for Fabricating and Patterning Nanostructures. Chem. Rev. 1999, 99, 1823–1848. [Google Scholar] [CrossRef] [PubMed]
- Whitesides, G.M.; Ostuni, E.; Takayama, S.; Jiang, X.; Ingber, D.E. Soft lithography in biology and biochemistry. Annu. Rev. Biomed. Eng. 2001, 3, 335–373. [Google Scholar] [CrossRef]
- Verpoorte, E.; Rooij, N.F.D. Microfluidics meets MEMS. Proc. IEEE 2003, 91, 930–953. [Google Scholar] [CrossRef]
- Zemel, A.; Rehfeldt, F.; Brown, A.E.; Discher, D.E.; Safran, S.A. Optimal matrix rigidity for stress fiber polarization in stem cells. Nature physics 2010, 6, 468–473. [Google Scholar] [CrossRef] [PubMed]
- Quail, D.F.; Joyce, J.A. Microenvironmental regulation of tumor progression and metastasis. Nat. Med. 2013, 19, 1423–1437. [Google Scholar] [CrossRef]
- Lv, D.; Hu, Z.; Lu, L.; Lu, H.; Xu, X. Three-dimensional cell culture: A powerful tool in tumor research and drug discovery. Oncol. Lett. 2017, 14, 6999–7010. [Google Scholar] [CrossRef]
- Bao, M.; Xie, J.; Piruska, A.; Huck, W.T.S. 3D microniches reveal the importance of cell size and shape. Nat. Commun. 2017, 8, 1962. [Google Scholar] [CrossRef]
- Liu, F.D.; Tam, K.; Pishesha, N.; Poon, Z.; Van Vliet, K.J. Improving hematopoietic recovery through modeling and modulation of the mesenchymal stromal cell secretome. Stem Cell Res. Ther. 2018, 9, 268. [Google Scholar] [CrossRef]
- Adriani, G.; Pavesi, A.; Tan, A.T.; Bertoletti, A.; Thiery, J.P.; Kamm, R.D. Microfluidic models for adoptive cell-mediated cancer immunotherapies. Drug Discov. Today 2016, 21, 1472–1478. [Google Scholar] [CrossRef]
- Wang, X.; Sun, Q.; Pei, J. Microfluidic-Based 3D Engineered Microvascular Networks and Their Applications in Vascularized Microtumor Models. Micromachines 2018, 9, 493. [Google Scholar] [CrossRef] [PubMed]
- Jeon, J.S.; Bersini, S.; Whisler, J.A.; Chen, M.B.; Dubini, G.; Charest, J.L.; Moretti, M.; Kamm, R.D. Generation of 3D functional microvascular networks with human mesenchymal stem cells in microfluidic systems. Integr. Biol. Quant. Biosci. Nano Macro 2014, 6, 555–563. [Google Scholar] [CrossRef]
- Bersini, S.; Miermont, A.; Pavesi, A.; Kamm, R.D.; Thiery, J.P.; Moretti, M.; Adriani, G. A combined microfluidic-transcriptomic approach to characterize the extravasation potential of cancer cells. Oncotarget 2018, 9, 36110–36125. [Google Scholar] [CrossRef]
- Cho, Y.M.; Moon, W.K.; Kim, H.S.; Na, K.; Yang, J.H.; Huh, Y.H.; Kim, J.A.; Chung, S.; Lee, S.H. Construction of a 3D mammary duct based on spatial localization of the extracellular matrix. NPG Asia Mater. 2018, 10. [Google Scholar] [CrossRef]
- Baker, M. A living system on a chip. Nature 2011, 471, 661. [Google Scholar] [CrossRef]
- Willyard, C. Channeling chip power: Tissue chips are being put to the test by industry. Nat. Med. 2017, 23, 138. [Google Scholar] [CrossRef]
- Zaretsky, I.; Polonsky, M.; Shifrut, E.; Reich-Zeliger, S.; Antebi, Y.; Aidelberg, G.; Waysbort, N.; Friedman, N. Monitoring the dynamics of primary T cell activation and differentiation using long term live cell imaging in microwell arrays. Lab Chip 2012, 12, 5007–5015. [Google Scholar] [CrossRef]
- Jung, J.H.; Han, C.; Lee, S.A.; Kim, J.; Yang, C. Microfluidic-integrated laser-controlled microactuators with on-chip microscopy imaging functionality. Lab Chip 2014, 14, 3781–3789. [Google Scholar] [CrossRef]
- Levario, T.J.; Zhan, M.; Lim, B.; Shvartsman, S.Y.; Lu, H. Microfluidic trap array for massively parallel imaging of Drosophila embryos. Nat. Protoc. 2013, 8, 721. [Google Scholar] [CrossRef]
- Holzner, G.; Du, Y.; Cao, X.; Choo, J.; deMello, A.J.; Stavrakis, S. An optofluidic system with integrated microlens arrays for parallel imaging flow cytometry. Lab Chip 2018, 18, 3631–3637. [Google Scholar] [CrossRef]
- Vaidyanathan, R.; Yeo, T.; Lim, C.T. Microfluidics for cell sorting and single cell analysis from whole blood. Methods Cell Biol. 2018, 147, 151–173. [Google Scholar] [CrossRef]
- Wasserberg, D.; Zhang, X.; Breukers, C.; Connell, B.J.; Baeten, E.; van den Blink, D.; Èlia, S.; Bloem, A.C.; Nijhuis, M.; Wensing, A.M.J.; et al. All-printed cell counting chambers with on-chip sample preparation for point-of-care CD4 counting. Biosens. bioelectron. 2018, 117, 659–668. [Google Scholar] [CrossRef]
- Shields, C.W., IV; Reyes, C.D.; López, G.P. Microfluidic cell sorting: A review of the advances in the separation of cells from debulking to rare cell isolation. Lab Chip 2015, 15, 1230–1249. [Google Scholar] [CrossRef] [PubMed]
- Kung, Y.C.; Huang, K.W.; Chong, W.; Chiou, P.Y. Tunnel Dielectrophoresis for Tunable, Single-Stream Cell Focusing in Physiological Buffers in High-Speed Microfluidic Flows. Small 2016, 12, 4343–4348. [Google Scholar] [CrossRef] [PubMed]
- Kung, Y.-C.; Huang, K.-W.; Fan, Y.-J.; Chiou, P.-Y. Fabrication of 3D high aspect ratio PDMS microfluidic networks with a hybrid stamp. Lab Chip 2015, 15, 1861–1868. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.J.; Wu, Y.C.; Chen, Y.; Kung, Y.C.; Wu, T.H.; Huang, K.W.; Sheen, H.J.; Chiou, P.Y. Three dimensional microfluidics with embedded microball lenses for parallel and high throughput multicolor fluorescence detection. Biomicrofluidics 2013, 7, 044121. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wu, T.-H.; Kung, Y.-C.; Teitell, M.A.; Chiou, P.-Y. 3D pulsed laser-triggered high-speed microfluidic fluorescence-activated cell sorter. Analyst 2013, 138, 7308–7315. [Google Scholar] [CrossRef]
- Chang, Y.H.; Wu, M.H. Microbioreactors for Cartilage Tissue Engineering. Methods Mol. Biol. 2015, 1340, 235–244. [Google Scholar] [CrossRef]
- Huang, S.B.; Wang, S.S.; Hsieh, C.H.; Lin, Y.C.; Lai, C.S.; Wu, M.H. An integrated microfluidic cell culture system for high-throughput perfusion three-dimensional cell culture-based assays: Effect of cell culture model on the results of chemosensitivity assays. Lab Chip 2013, 13, 1133–1143. [Google Scholar] [CrossRef] [PubMed]
- Abaci, H.E.; Devendra, R.; Smith, Q.; Gerecht, S.; Drazer, G. Design and development of microbioreactors for long-term cell culture in controlled oxygen microenvironments. Biomed. Microdev. 2012, 14, 145–152. [Google Scholar] [CrossRef]
- Yager, P.; Edwards, T.; Fu, E.; Helton, K.; Nelson, K.; Tam, M.R.; Weigl, B.H. Microfluidic diagnostic technologies for global public health. Nature 2006, 442, 412–418. [Google Scholar] [CrossRef]
- Fan, R.; Vermesh, O.; Srivastava, A.; Yen, B.K.; Qin, L.; Ahmad, H.; Kwong, G.A.; Liu, C.C.; Gould, J.; Hood, L.; et al. Integrated barcode chips for rapid, multiplexed analysis of proteins in microliter quantities of blood. Nat. Biotechnol. 2008, 26, 1373–1378. [Google Scholar] [CrossRef]
- McHugh, K.J.; Nguyen, T.D.; Linehan, A.R.; Yang, D.; Behrens, A.M.; Rose, S.; Tochka, Z.L.; Tzeng, S.Y.; Norman, J.J.; Anselmo, A.C.; et al. Fabrication of fillable microparticles and other complex 3D microstructures. Science 2017, 357, 1138–1142. [Google Scholar] [CrossRef]
- Rolland, J.P.; Maynor, B.W.; Euliss, L.E.; Exner, A.E.; Denison, G.M.; DeSimone, J.M. Direct Fabrication and Harvesting of Monodisperse, Shape-Specific Nanobiomaterials. J. Am. Chem. Soc. 2005, 127, 10096–10100. [Google Scholar] [CrossRef] [PubMed]
- Birarda, G.; Ravasio, A.; Suryana, M.; Maniam, S.; Holman, H.Y.N.; Grenci, G. IR-Live: Fabrication of a low-cost plastic microfluidic device for infrared spectromicroscopy of living cells. Lab Chip 2016, 16, 1644–1651. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.; Brink, H.; Blanche, S.; DiPrete, C.; Bongiorno, T.; Stone, N.; Liu, A.; Philip, A.; Wang, G.; Lam, W.; et al. Microfluidic Sorting of Cells by Viability Based on Differences in Cell Stiffness. Sci. Rep. 2017, 7, 1997. [Google Scholar] [CrossRef] [PubMed]
- Weibel, D.B.; DiLuzio, W.R.; Whitesides, G.M. Microfabrication meets microbiology. Nat. Rev. Microbiol. 2007, 5, 209. [Google Scholar] [CrossRef] [PubMed]
- Rothbauer, M.; Zirath, H.; Ertl, P. Recent advances in microfluidic technologies for cell-to-cell interaction studies. Lab Chip 2018, 18, 249–270. [Google Scholar] [CrossRef] [PubMed]
- Luo, T.; Fan, L.; Zhu, R.; Sun, D. Microfluidic Single-Cell Manipulation and Analysis: Methods and Applications. Micromachines 2019, 10, 104. [Google Scholar] [CrossRef] [PubMed]
- Dirscherl, C.; Springer, S. Protein micropatterns printed on glass: Novel tools for protein-ligand binding assays in live cells. Eng. Life Sci. 2018, 18, 124–131. [Google Scholar] [CrossRef]
- Von Philipsborn, A.C.; Lang, S.; Bernard, A.; Loeschinger, J.; David, C.; Lehnert, D.; Bastmeyer, M.; Bonhoeffer, F. Microcontact printing of axon guidance molecules for generation of graded patterns. Nat. Protoc. 2006, 1, 1322. [Google Scholar] [CrossRef]
- Chen, C.S.; Mrksich, M.; Huang, S.; Whitesides, G.M.; Ingber, D.E. Geometric Control of Cell Life and Death. Science 1997, 276, 1425–1428. [Google Scholar] [CrossRef]
- Jain, N.; Iyer, K.V.; Kumar, A.; Shivashankar, G.V. Cell geometric constraints induce modular gene-expression patterns via redistribution of HDAC3 regulated by actomyosin contractility. Proc. Natl. Acad. Sci. USA 2013, 110, 11349. [Google Scholar] [CrossRef]
- Sahni, G.; Yuan, J.; Toh, Y.-C. Stencil Micropatterning of Human Pluripotent Stem Cells for Probing Spatial Organization of Differentiation Fates. J. Vis. Exp. JoVE 2016. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Tian, J.; Wu, J.; Cao, W.; Zhou, B.; Shen, R.; Wen, W. Digital microfluidic programmable stencil (dMPS) for protein and cell patterning. RSC Adv. 2016, 6, 101760–101769. [Google Scholar] [CrossRef]
- Wright, D.; Rajalingam, B.; Selvarasah, S.; Dokmeci, M.R.; Khademhosseini, A. Generation of static and dynamic patterned co-cultures using microfabricated parylene-C stencils. Lab Chip 2007, 7, 1272–1279. [Google Scholar] [CrossRef] [PubMed]
- Masters, T.; Engl, W.; Weng, Z.L.; Arasi, B.; Gauthier, N.; Viasnoff, V. Easy Fabrication of Thin Membranes with Through Holes. Application to Protein Patterning. PLoS ONE 2012, 7, e44261. [Google Scholar] [CrossRef] [PubMed]
- Azioune, A.; Carpi, N.; Tseng, Q.; Théry, M.; Piel, M. Protein Micropatterns: A Direct Printing Protocol Using Deep UVs. In Methods in Cell Biology; Cassimeris, L., Tran, P., Eds.; Academic Press: Waltham, MA, USA, 2010; Chapter 8; Volume 97, pp. 133–146. [Google Scholar]
- Azioune, A.; Storch, M.; Bornens, M.; Théry, M.; Piel, M. Simple and rapid process for single cell micro-patterning. Lab Chip 2009, 9, 1640–1642. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Chu, Y.-S.; Thiery, J.P.; Lim, C.T.; Rodriguez, I. Microfluidic cell trap array for controlled positioning of single cells on adhesive micropatterns. Lab Chip 2013, 13, 714–721. [Google Scholar] [CrossRef] [PubMed]
- Gupta, M.; Kocgozlu, L.; Sarangi, B.R.; Margadant, F.; Ashraf, M.; Ladoux, B. Micropillar substrates: A tool for studying cell mechanobiology. Methods Cell Biol. 2015, 125, 289–308. [Google Scholar] [CrossRef]
- Ravasio, A.; Vaishnavi, S.; Ladoux, B.; Viasnoff, V. High-resolution imaging of cellular processes across textured surfaces using an indexed-matched elastomer. Acta Biomater. 2015, 14, 53–60. [Google Scholar] [CrossRef]
- Moe, A.A.; Suryana, M.; Marcy, G.; Lim, S.K.; Ankam, S.; Goh, J.Z.; Jin, J.; Teo, B.K.; Law, J.B.; Low, H.Y.; et al. Microarray with micro- and nano-topographies enables identification of the optimal topography for directing the differentiation of primary murine neural progenitor cells. Small 2012, 8, 3050–3061. [Google Scholar] [CrossRef]
- Yim, E.K.; Darling, E.M.; Kulangara, K.; Guilak, F.; Leong, K.W. Nanotopography-induced changes in focal adhesions, cytoskeletal organization, and mechanical properties of human mesenchymal stem cells. Biomaterials 2010, 31, 1299–1306. [Google Scholar] [CrossRef]
- Migliorini, E.; Grenci, G.; Ban, J.; Pozzato, A.; Tormen, M.; Lazzarino, M.; Torre, V.; Ruaro, M.E. Acceleration of neuronal precursors differentiation induced by substrate nanotopography. Biotechnol. Bioeng. 2011, 108, 2736–2746. [Google Scholar] [CrossRef]
- Sperling, L.E.; Reis, K.P.; Pozzobon, L.G.; Girardi, C.S.; Pranke, P. Influence of random and oriented electrospun fibrous poly(lactic-co-glycolic acid) scaffolds on neural differentiation of mouse embryonic stem cells. J. Biomed. Mater. Res. Part A 2017, 105, 1333–1345. [Google Scholar] [CrossRef]
- Abagnale, G.; Steger, M.; Nguyen, V.H.; Hersch, N.; Sechi, A.; Joussen, S.; Denecke, B.; Merkel, R.; Hoffmann, B.; Dreser, A.; et al. Surface topography enhances differentiation of mesenchymal stem cells towards osteogenic and adipogenic lineages. Biomaterials 2015, 61, 316–326. [Google Scholar] [CrossRef] [PubMed]
- Ravasio, A.; Cheddadi, I.; Chen, T.; Pereira, T.; Ong, H.T.; Bertocchi, C.; Brugues, A.; Jacinto, A.; Kabla, A.J.; Toyama, Y.; et al. Gap geometry dictates epithelial closure efficiency. Nat. Commun. 2015, 6, 7683. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Callan-Jones, A.; Fedorov, E.; Ravasio, A.; Brugués, A.; Ong, H.T.; Toyama, Y.; Low, B.C.; Trepat, X.; Shemesh, T.; et al. Large-scale curvature sensing by directional actin flow drives cellular migration mode switching. Nat. Phys. 2019. [Google Scholar] [CrossRef] [PubMed]
- Xi, W.; Sonam, S.; Beng Saw, T.; Ladoux, B.; Teck Lim, C. Emergent patterns of collective cell migration under tubular confinement. Nat. Commun. 2017, 8, 1517. [Google Scholar] [CrossRef] [PubMed]
- Vedula, S.R.K.; Ravasio, A.; Anon, E.; Chen, T.; Peyret, G.; Ashraf, M.; Ladoux, B. Microfabricated Environments to Study Collective Cell Behaviors. In Methods in Cell Biolology; Piel, M., Théry, M., Eds.; Academic Press: Waltham, MA, USA, 2014; Chapter 16; Volume 120, pp. 235–252. [Google Scholar]
- Gao, X.; Stoecklin, C.; Zhang, Y.; Weng, Z.; De Mets, R.; Grenci, G.; Viasnoff, V. Artificial Microniche Array with Spatially Structured Biochemical Cues. Methods Mol. Biol. 2018, 1771, 55–66. [Google Scholar] [CrossRef]
- Folch, A.; Jo, B.-H.; Hurtado, O.; Beebe, D.J.; Toner, M. Microfabricated elastomeric stencils for micropatterning cell cultures. J. Biomed. Mater. Res. 2000, 52, 346–353. [Google Scholar] [CrossRef]
- Huisken, J.; Swoger, J.; Del Bene, F.; Wittbrodt, J.; Stelzer, E.H.K. Optical Sectioning Deep Inside Live Embryos by Selective Plane Illumination Microscopy. Science 2004, 305, 1007. [Google Scholar] [CrossRef]
- Planchon, T.A.; Gao, L.; Milkie, D.E.; Davidson, M.W.; Galbraith, J.A.; Galbraith, C.G.; Betzig, E. Rapid three-dimensional isotropic imaging of living cells using Bessel beam plane illumination. Nat. Methods 2011, 8, 417–423. [Google Scholar] [CrossRef] [PubMed]
- Gebhardt, J.C.; Suter, D.M.; Roy, R.; Zhao, Z.W.; Chapman, A.R.; Basu, S.; Maniatis, T.; Xie, X.S. Single-molecule imaging of transcription factor binding to DNA in live mammalian cells. Nat. Methods 2013, 10, 421–426. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.C.; Legant, W.R.; Wang, K.; Shao, L.; Milkie, D.E.; Davidson, M.W.; Janetopoulos, C.; Wu, X.S.; Hammer, J.A., 3rd; Liu, Z.; et al. Lattice light-sheet microscopy: Imaging molecules to embryos at high spatiotemporal resolution. Science 2014, 346, 1257998. [Google Scholar] [CrossRef] [PubMed]
- Galland, R.; Grenci, G.; Aravind, A.; Viasnoff, V.; Studer, V.; Sibarita, J.B. 3D high- and super-resolution imaging using single-objective SPIM. Nat. Methods 2015, 12, 641–644. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.P.; Galland, R.; Finch-Edmondson, M.L.; Grenci, G.; Sibarita, J.-B.; Studer, V.; Viasnoff, V.; Saunders, T.E. 3D Protein Dynamics in the Cell Nucleus. Biophys. J. 2017, 112, 133–142. [Google Scholar] [CrossRef]
- Mantsch, H.H.; Chapman, A.R. Infrared Spectroscopy of Biomolecules; Wiley-Liss: New York, NY, USA, 1996; 359p. [Google Scholar]
- Nasse, M.J.; Walsh, M.J.; Mattson, E.C.; Reininger, R.; Kajdacsy-Balla, A.; Macias, V.; Bhargava, R.; Hirschmugl, C.J. High-resolution Fourier-transform infrared chemical imaging with multiple synchrotron beams. Nat. Methods 2011, 8, 413. [Google Scholar] [CrossRef]
- Kröger-Lui, N.; Gretz, N.; Haase, K.; Kränzlin, B.; Neudecker, S.; Pucci, A.; Regenscheit, A.; Schönhals, A.; Petrich, W. Rapid identification of goblet cells in unstained colon thin sections by means of quantum cascade laser-based infrared microspectroscopy. Analyst 2015, 140, 2086–2092. [Google Scholar] [CrossRef]
- Mitri, E.; Birarda, G.; Vaccari, L.; Kenig, S.; Tormen, M.; Grenci, G. SU-8 bonding protocol for the fabrication of microfluidic devices dedicated to FTIR microspectroscopy of live cells. Lab Chip 2014, 14, 210–218. [Google Scholar] [CrossRef]
- Marcsisin, E.J.; Uttero, C.M.; Miljković, M.; Diem, M. Infrared microspectroscopy of live cells in aqueous media. Analyst 2010, 135, 3227–3232. [Google Scholar] [CrossRef]
- Holman, H.-Y.N.; Bechtel, H.A.; Hao, Z.; Martin, M.C. Synchrotron IR Spectromicroscopy: Chemistry of Living Cells. Anal. Chem. 2010, 82, 8757–8765. [Google Scholar] [CrossRef]
- Nasse, M.J.; Ratti, S.; Giordano, M.; Hirschmugl, C.J. Demountable Liquid/Flow Cell for in vivo Infrared Microspectroscopy of Biological Specimens. Appl. Spectrosc. 2009, 63, 1181–1186. [Google Scholar] [CrossRef] [PubMed]
- Birarda, G.; Bedolla, D.E.; Mitri, E.; Pacor, S.; Grenci, G.; Vaccari, L. Apoptotic pathways of U937 leukemic monocytes investigated by infrared microspectroscopy and flow cytometry. Analyst 2014, 139, 3097–3106. [Google Scholar] [CrossRef] [PubMed]
- Bedolla, D.E.; Kenig, S.; Mitri, E.; Ferraris, P.; Marcello, A.; Grenci, G.; Vaccari, L. Determination of cell cycle phases in live B16 melanoma cells using IRMS. Analyst 2013, 138, 4015–4021. [Google Scholar] [CrossRef]
- Sandt, C.; Frederick, J.; Dumas, P. Profiling pluripotent stem cells and organelles using synchrotron radiation infrared microspectroscopy. J. Biophotonics 2013, 6, 60–72. [Google Scholar] [CrossRef]
- Hoffner, G.; André, W.; Sandt, C.; Djian, P. Synchrotron-based infrared spectroscopy brings to light the structure of protein aggregates in neurodegenerative diseases. Rev. Anal. Chem. 2014, 33, 231. [Google Scholar] [CrossRef]
- Miller, L.M.; Bourassa, M.W.; Smith, R.J. FTIR spectroscopic imaging of protein aggregation in living cells. Biochim. Biophys. Acta (BBA) Biomembr. 2013, 1828, 2339–2346. [Google Scholar] [CrossRef] [PubMed]
- Tobin, M.J.; Puskar, L.; Barber, R.L.; Harvey, E.C.; Heraud, P.; Wood, B.R.; Bambery, K.R.; Dillon, C.T.; Munro, K.L. FTIR spectroscopy of single live cells in aqueous media by synchrotron IR microscopy using microfabricated sample holders. Vib. Spectrosc. 2010, 53, 34–38. [Google Scholar] [CrossRef]
- Suryana, M.; Shanmugarajah, J.V.; Maniam, S.M.; Grenci, G. Soft Lithographic Procedure for Producing Plastic Microfluidic Devices with View-ports Transparent to Visible and Infrared Light. JoVE 2017. [Google Scholar] [CrossRef] [PubMed]
- Vaccari, L.; Birarda, G.; Businaro, L.; Pacor, S.; Grenci, G. Infrared Microspectroscopy of Live Cells in Microfluidic Devices (MD-IRMS): Toward a Powerful Label-Free Cell-Based Assay. Anal. Chem. 2012, 84, 4768–4775. [Google Scholar] [CrossRef]
- Birarda, G.; Grenci, G.; Businaro, L.; Marmiroli, B.; Pacor, S.; Vaccari, L. Fabrication of a microfluidic platform for investigating dynamic biochemical processes in living samples by FTIR microspectroscopy. Microelectron. Eng. 2010, 87, 806–809. [Google Scholar] [CrossRef]
- Valastyan, S.; Weinberg, R.A. Tumor metastasis: Molecular insights and evolving paradigms. Cell 2011, 147, 275–292. [Google Scholar] [CrossRef] [PubMed]
- Fehm, T.; Braun, S.; Muller, V.; Janni, W.; Gebauer, G.; Marth, C.; Schindlbeck, C.; Wallwiener, D.; Borgen, E.; Naume, B.; et al. A concept for the standardized detection of disseminated tumor cells in bone marrow from patients with primary breast cancer and its clinical implementation. Cancer 2006, 107, 885–892. [Google Scholar] [CrossRef]
- Nieva, J.J.; Kuhn, P. Fluid biopsy for solid tumors: A patient’s companion for lifelong characterization of their disease. Future Oncol. 2012, 8, 989–998. [Google Scholar] [CrossRef] [PubMed]
- Goldkorn, A.; Ely, B.; Quinn, D.I.; Tangen, C.M.; Fink, L.M.; Xu, T.; Twardowski, P.; Van Veldhuizen, P.J.; Agarwal, N.; Carducci, M.A.; et al. Circulating tumor cell counts are prognostic of overall survival in SWOG S0421: A phase III trial of docetaxel with or without atrasentan for metastatic castration-resistant prostate cancer. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2014, 32, 1136–1142. [Google Scholar] [CrossRef] [PubMed]
- Cui, L.; Kwong, J.; Wang, C.C. Prognostic value of circulating tumor cells and disseminated tumor cells in patients with ovarian cancer: A systematic review and meta-analysis. J. Ovarian Res. 2015, 8, 38. [Google Scholar] [CrossRef] [PubMed]
- Van der Toom, E.E.; Verdone, J.E.; Gorin, M.A.; Pienta, K.J. Technical challenges in the isolation and analysis of circulating tumor cells. Oncotarget 2016, 7, 62754–62766. [Google Scholar] [CrossRef]
- Zhang, L.; Ridgway, L.D.; Wetzel, M.D.; Ngo, J.; Yin, W.; Kumar, D.; Goodman, J.C.; Groves, M.D.; Marchetti, D. The Identification and Characterization of Breast Cancer CTCs Competent for Brain Metastasis. Sci. Transl. Med. 2013, 5, 180ra148. [Google Scholar] [CrossRef]
- Yu, M.; Bardia, A.; Aceto, N.; Bersani, F.; Madden, M.W.; Donaldson, M.C.; Desai, R.; Zhu, H.; Comaills, V.; Zheng, Z.; et al. Ex vivo culture of circulating breast tumor cells for individualized testing of drug susceptibility. Science 2014, 345, 216. [Google Scholar] [CrossRef] [PubMed]
- Gao, D.; Vela, I.; Sboner, A.; Iaquinta, P.J.; Karthaus, W.R.; Gopalan, A.; Dowling, C.; Wanjala, J.N.; Undvall, E.A.; Arora, V.K.; et al. Organoid cultures derived from patients with advanced prostate cancer. Cell 2014, 159, 176–187. [Google Scholar] [CrossRef]
- Maheswaran, S.; Haber, D.A. Ex Vivo Culture of CTCs: An Emerging Resource to Guide Cancer Therapy. Cancer Res. 2015. [Google Scholar] [CrossRef] [PubMed]
- Khoo, B.L.; Lee, S.C.; Kumar, P.; Tan, T.Z.; Warkiani, M.E.; Ow, S.G.; Nandi, S.; Lim, C.T.; Thiery, J.P. Short-term expansion of breast circulating cancer cells predicts response to anti-cancer therapy. Oncotarget 2015, 6, 15578–15593. [Google Scholar] [CrossRef] [PubMed]
- Khoo, B.L.; Grenci, G.; Jing, T.; Lim, Y.B.; Lee, S.C.; Thiery, J.P.; Han, J.; Lim, C.T. Liquid biopsy and therapeutic response: Circulating tumor cell cultures for evaluation of anticancer treatment. Sci. Adv. 2016, 2, e1600274. [Google Scholar] [CrossRef] [PubMed]
- Khoo, B.L.; Grenci, G.; Lim, Y.B.; Lee, S.C.; Han, J.; Lim, C.T. Expansion of patient-derived circulating tumor cells from liquid biopsies using a CTC microfluidic culture device. Nat. Protoc. 2018, 13, 34–58. [Google Scholar] [CrossRef] [PubMed]
- Khoo, B.L.; Grenci, G.; Lim, J.S.Y.; Lim, Y.P.; Fong, J.; Yeap, W.H.; Bin Lim, S.; Chua, S.L.; Wong, S.C.; Yap, Y.-S.; et al. Low-dose anti-inflammatory combinatorial therapy reduced cancer stem cell formation in patient-derived preclinical models for tumour relapse prevention. Br. J. Cancer 2019, 120, 407–423. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-H.; Choi, W.-S.; Lee, K.-H.; Yoon, J.-B. A simple and effective fabrication method for various 3D microstructures: Backside 3D diffuser lithography. J. Micromech. Microeng. 2008, 18, 125015. [Google Scholar] [CrossRef]
- Maslow, A.H. The Psychology of Science; Harper & Row: Manhattan, NY, USA, 1966. [Google Scholar]
| Application | Method(s) | Required Micro-Fabricated System | References |
|---|---|---|---|
| Protein micro-patterning | Micro-contact printing | PDMS stamps | [60,61,62,63] |
| Stenciling | Micro-stencils | [64,65,66,67] | |
| UV-patterning | UV mask | [68,69,70] | |
| Traction force microscopy | Micro-pillars deflection tracking | Soft micro-pillars | [71,72] |
| Cells response to topology | Cells culturing on substrates with different topologies | Micro/nano patterned substrates | [73,74,75,76,77] |
| Collective cells migration | Engineering of environmental cues for collective migration | Micro-stencils Micro-patterning | [78,79,80,81] |
| Cells response to micro-structured environment | Cells culturing in micro-patterned niches | Substrates with micro-wells | [82,83] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grenci, G.; Bertocchi, C.; Ravasio, A. Integrating Microfabrication into Biological Investigations: the Benefits of Interdisciplinarity. Micromachines 2019, 10, 252. https://doi.org/10.3390/mi10040252
Grenci G, Bertocchi C, Ravasio A. Integrating Microfabrication into Biological Investigations: the Benefits of Interdisciplinarity. Micromachines. 2019; 10(4):252. https://doi.org/10.3390/mi10040252
Chicago/Turabian StyleGrenci, Gianluca, Cristina Bertocchi, and Andrea Ravasio. 2019. "Integrating Microfabrication into Biological Investigations: the Benefits of Interdisciplinarity" Micromachines 10, no. 4: 252. https://doi.org/10.3390/mi10040252
APA StyleGrenci, G., Bertocchi, C., & Ravasio, A. (2019). Integrating Microfabrication into Biological Investigations: the Benefits of Interdisciplinarity. Micromachines, 10(4), 252. https://doi.org/10.3390/mi10040252





