Impedance-Based Biosensing of Pseudomonas putida via Solution Blow Spun PLA: MWCNT Composite Nanofibers
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Cultures
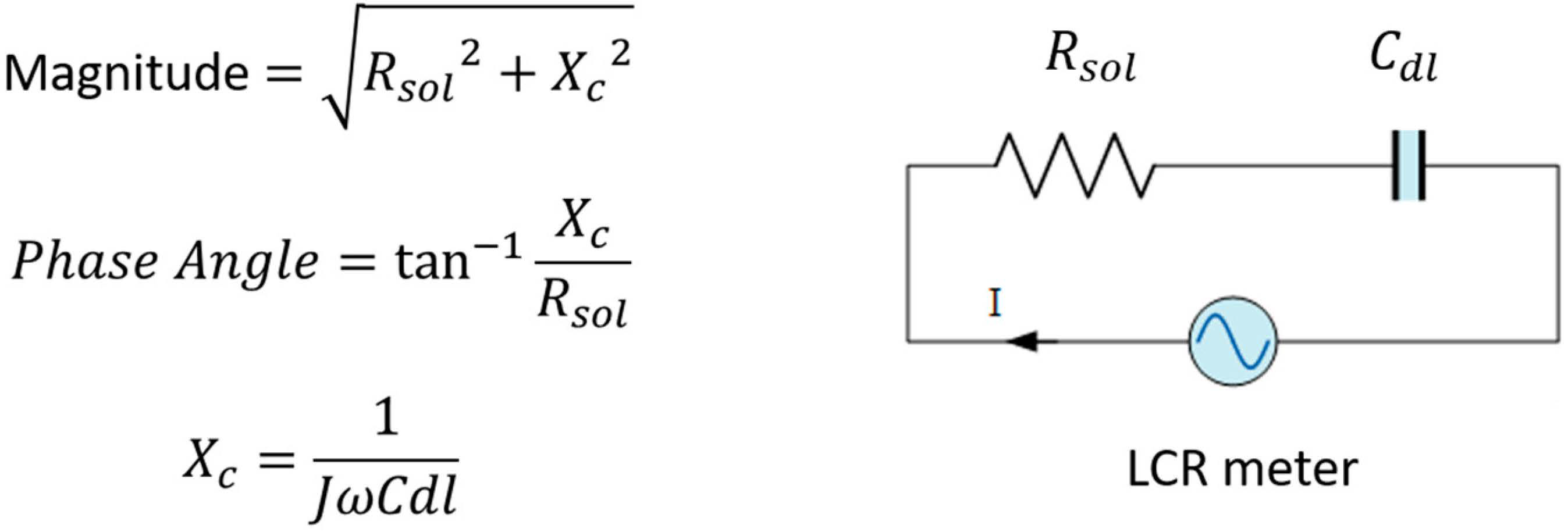
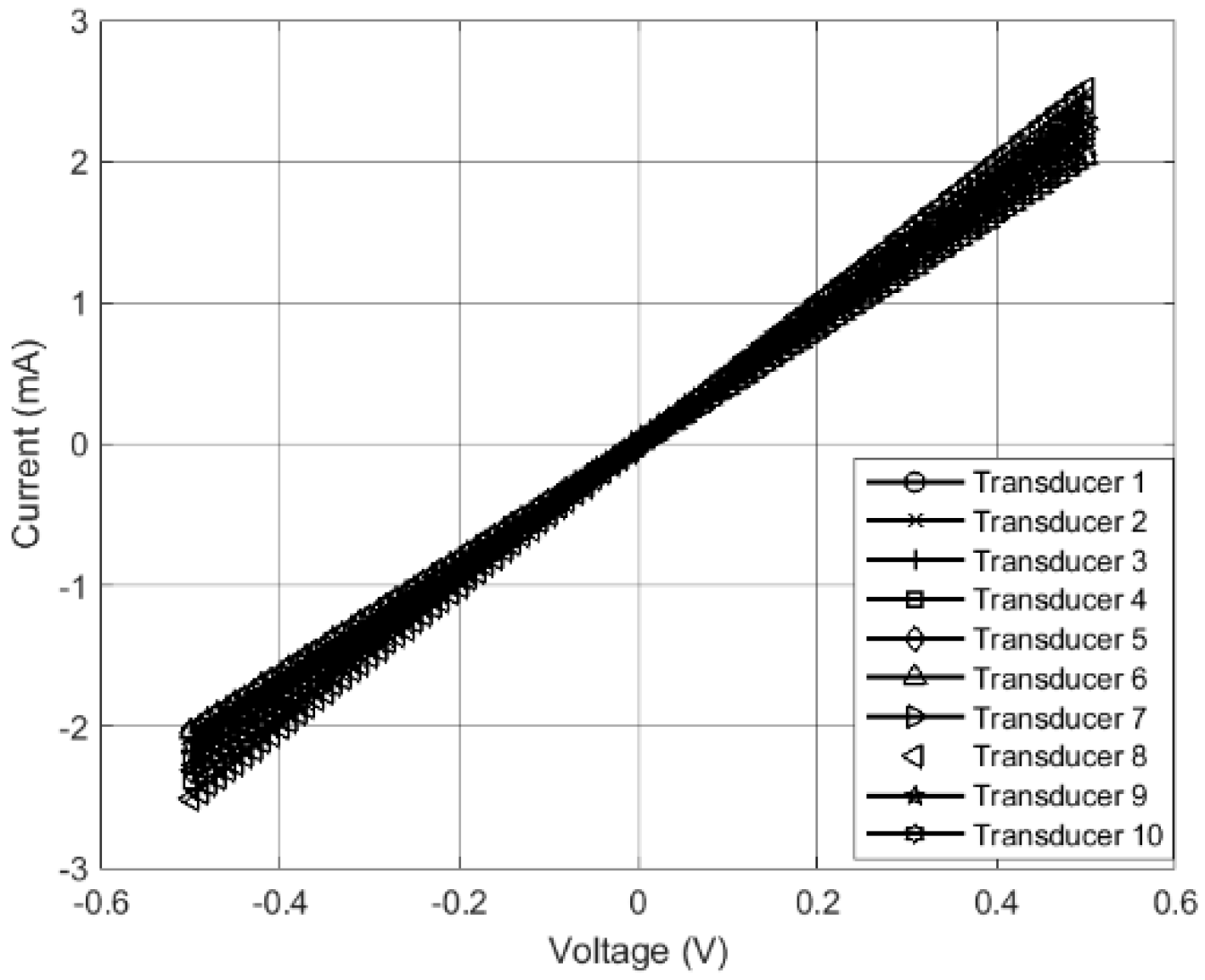
2.2. Nanofiber Sensor Fabrication and Multi-Walled Carbon Nanotube-Solution Blow Spun-Printed Circuit Board (MWCNT-SBS-PCB) Board Characterization
3. Results
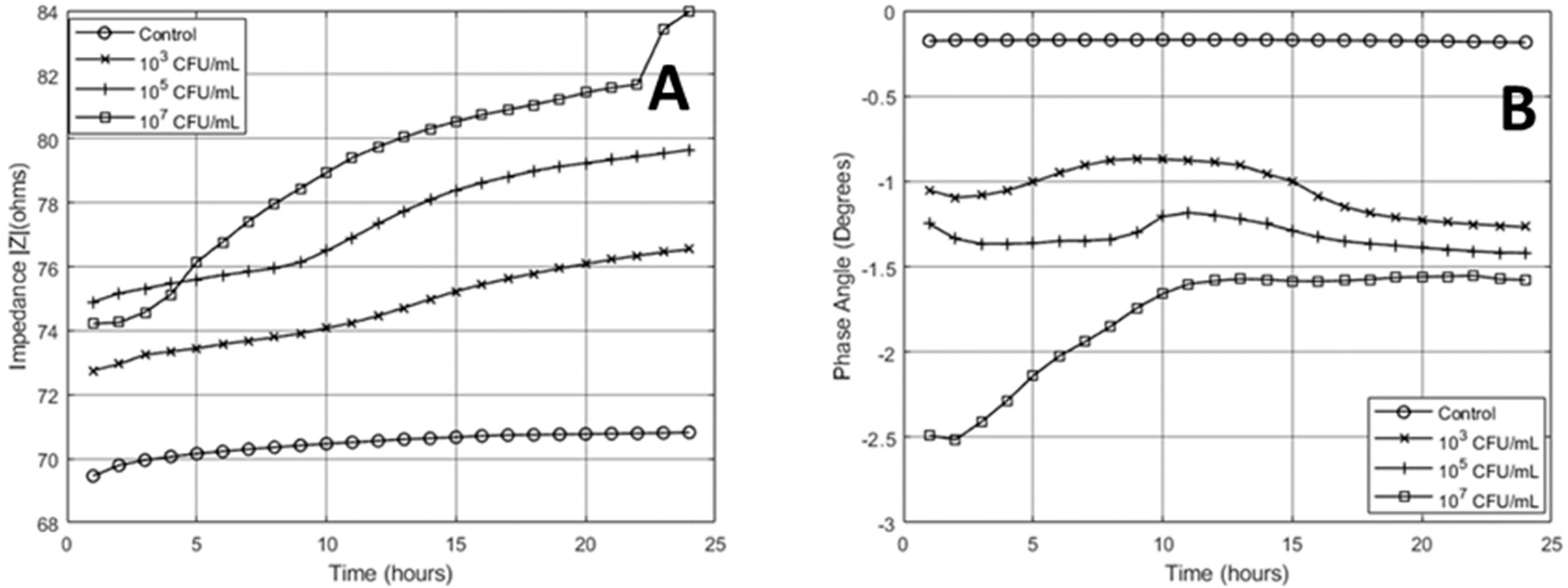
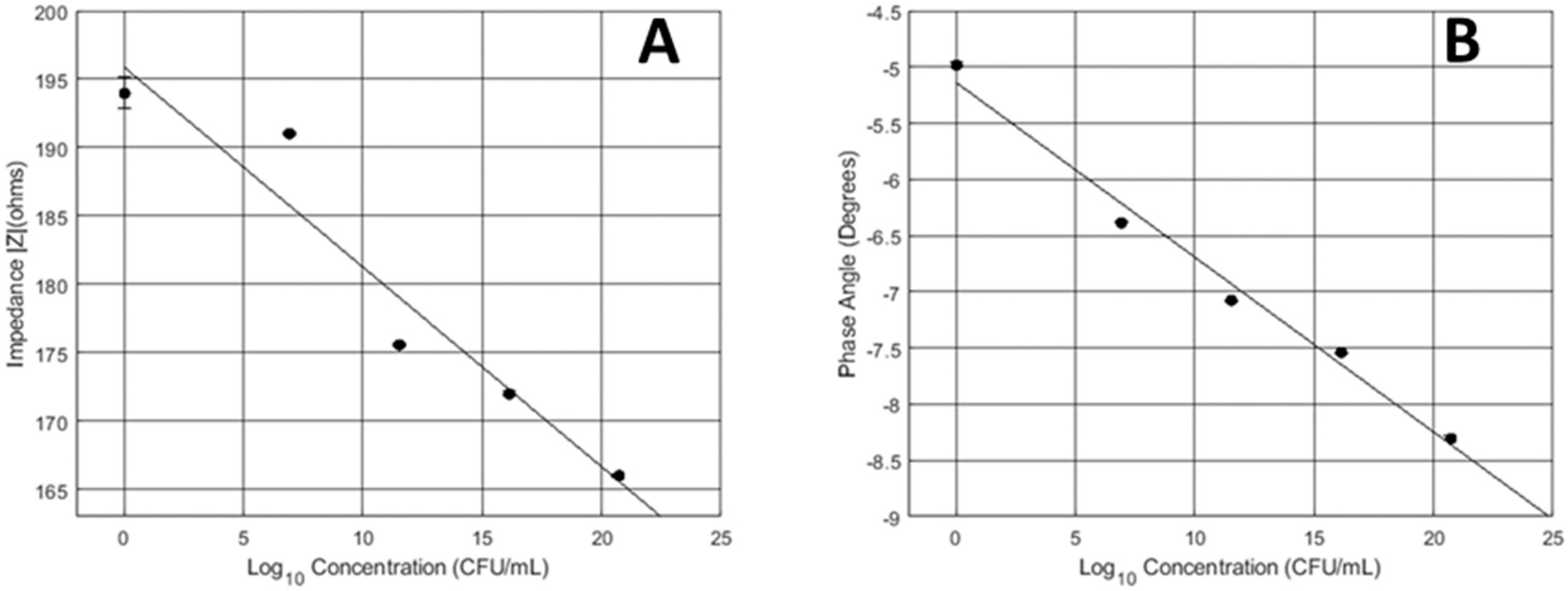
3.1. 24-Hour Continuous Impedance Sensing
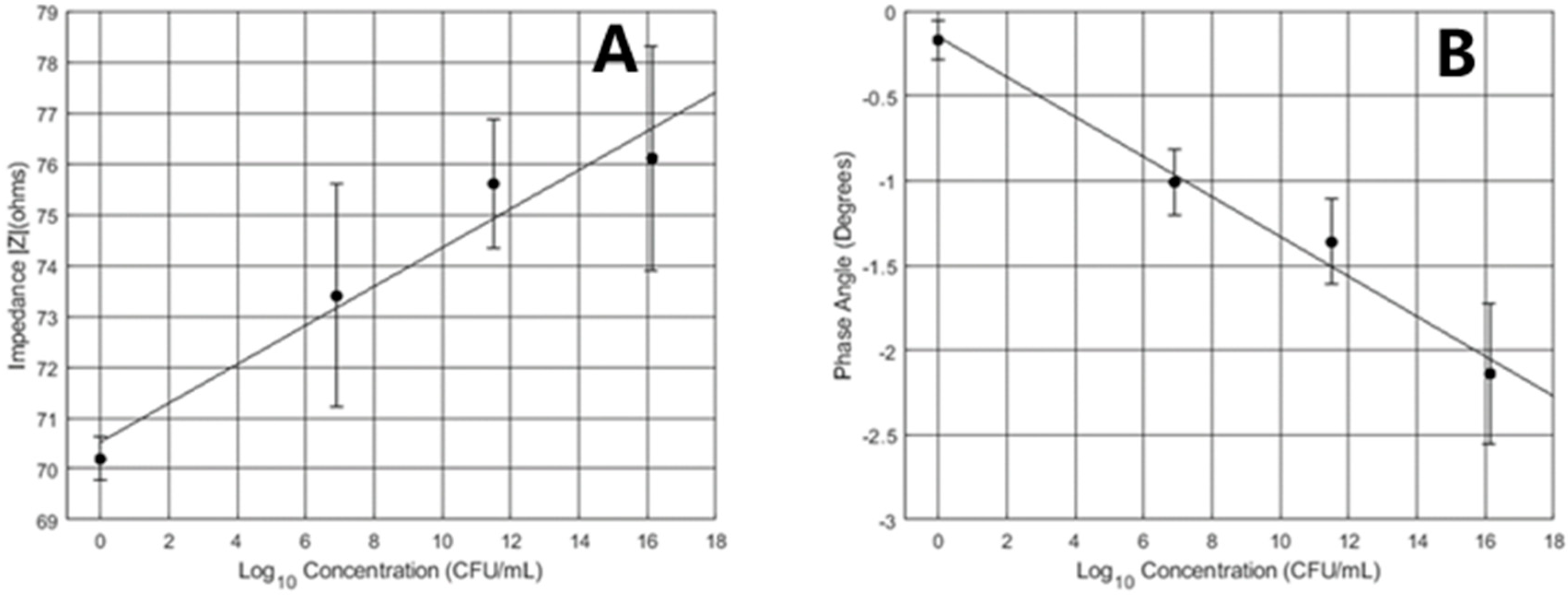
3.2. Instantaneous Metabolic Sensing
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Bader, M.S. Diabetic foot infections. Am. Fam. Physician 2008, 78, 71–79. [Google Scholar] [PubMed]
- Siddiqui, A.R.; Bernstein, J.M. Chronic wound infection: Facts and controversies. Clin. Dermatol. 2010, 28, 519–526. [Google Scholar] [CrossRef] [PubMed]
- Kalia, V.C. Quorum Sensing vs Quorum Quenching: A Battle with No End in Sight; Springer: New Delhi, India, 2015. [Google Scholar]
- Edwards, R.; Harding, K.G. Bacteria and wound healing. Curr. Opin. Infect. Dis. 2004, 17, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, R.J.; Hartzell, J.D.; Forsberg, J.A.; Babel, B.S.; Ganesan, A. Pseudomonas putida war wound infection in a US Marine: A case report and review of the literature. J. Infect. 2008, 56, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Mandell, G.L.; Douglas, R.G.; Bennett, J.E. Principles and Practice of Infectious Diseases. Volume 1 and 2; John Wiley & Sons: New York, NY, USA, 1979. [Google Scholar]
- Gianino, E.; Miller, C.; Gilmore, J. Smart Wound Dressings for Diabetic Chronic Wounds. Bioengineering 2018, 5, 51. [Google Scholar] [CrossRef] [PubMed]
- Swisher, S.L.; Lin, M.C.; Liao, A.; Leeflang, E.J.; Khan, Y.; Pavinatto, F.J.; Mann, K.; Naujokas, A.; Young, D.; Roy, S.; et al. Impedance sensing device enables early detection of pressure ulcers in vivo. Nat. Commun. 2015, 6, 6575. [Google Scholar] [CrossRef] [PubMed]
- Lukaski, H.C.; Moore, M. Bioelectrical impedance assessment of wound healing. J. Diabetes Sci. Technol. 2012, 6, 209–212. [Google Scholar] [CrossRef] [PubMed]
- Lei, K.F. Review on impedance detection of cellular responses in micro/nano environment. Micromachines 2014, 5, 1–12. [Google Scholar] [CrossRef]
- Bancalari, E.; Bernini, V.; Bottari, B.; Neviani, E.; Gatti, M. Application of impedance microbiology for evaluating potential acidifying performances of starter lactic acid bacteria to employ in milk transformation. Front. Microbiol. 2016, 7, 1628. [Google Scholar] [CrossRef] [PubMed]
- Szulcek, R.; Bogaard, H.J.; Amerongen, G.P.V. Electric cell-substrate impedance sensing for the quantification of endothelial proliferation, barrier function, and motility. J. Vis. Exp. 2014, 85, e51300. [Google Scholar] [CrossRef] [PubMed]
- Safavieh, M.; Pandya, H.J.; Venkataraman, M.; Thirumalaraju, P.; Kanakasabapathy, M.K.; Singh, A.; Prabhakar, D.; Chug, M.K.; Shafiee, H. Rapid Real-Time Antimicrobial Susceptibility Testing with Electrical Sensing on Plastic Microchips with Printed Electrodes. ACS Appl. Mater. Interfaces 2017, 9, 12832–12840. [Google Scholar] [CrossRef] [PubMed]
- Wegener, J.; Keese, C.R.; Giaever, I. Electric cell-substrate impedance sensing (ECIS) as a noninvasive means to monitor the kinetics of cell spreading to artificial surfaces. Exp. Cell Res. 2000, 259, 158–166. [Google Scholar] [CrossRef] [PubMed]
- Maalouf, R.; Fournier-Wirth, C.; Coste, J.; Chebib, H.; Saikali, Y.; Vittori, O.; Errachid, A.; Cloarec, J.P.; Martelet, C.; Jaffrezic-Renault, N. Label-free detection of bacteria by electrochemical impedance spectroscopy: Comparison to surface plasmon resonance. Anal. Chem. 2007, 79, 4879–4886. [Google Scholar] [CrossRef] [PubMed]
- Bellitti, P.; Bona, M.; Fontana, S.; Sardini, E.; Serpelloni, M. Lecture Notes in Electrical Engineering; Springer: Cham, Switzerland, 2019; pp. 445–454. [Google Scholar]
- Cristina, P.; Javier, M.; Sandra, C.; Rafael, V.G. Implications of Electrical Impedance-Based Microbiological Technology in Pork Meat Processing Industry for the Rapid Detection and Quantification of Salmonella spp. J. Food Sci. Eng. 2017, 7, 1–16. [Google Scholar] [CrossRef][Green Version]
- Wawerla, M.; Stolle, A.; Schalch, B.; Eisgruber, H. Impedance microbiology: Applications in Food Hygiene. J. Food Prot. 1999, 62, 1488–1496. [Google Scholar] [CrossRef] [PubMed]
- Zhu, T.; Pei, Z.H.; Huang, J.Y.; Xiong, C.Y.; Shi, S.G.; Fang, J. Detection of bacterial cells by impedance spectra via fluidic electrodes in a microfluidic device. Lab Chip 2010, 10, 1557–1560. [Google Scholar] [CrossRef] [PubMed]
- Miller, C.L.; Stafford, G.; Sigmon, N.; Gilmore, J.A. Conductive nonwoven carbon nanotube-PLA composite nanofibers towards wound sensors via solution blow spinning. IEEE Trans. Nanobiosci. 2019, 18, 244–247. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.J.; Li, Y.B.; Griffis, C.L.; Johnson, M.G. Interdigitated microelectrode (IME) impedance sensor for the detection of viable Salmonella typhimurium. Biosens. Bioelectron. 2004, 19, 1139–1147. [Google Scholar] [CrossRef] [PubMed]
- Sanchis, A.; Brown, A.P.; Sancho, M.; Martinez, G.; Sebastian, J.L.; Munoz, S.; Miranda, J.M. Dielectric characterization of bacterial cells using dielectrophoresis. Bioelectromagnetic 2007, 28, 393–401. [Google Scholar] [CrossRef] [PubMed]
- Couniot, N.; Afzalian, A.; Van Overstraeten-Schlögel, N.; Francis, L.A.; Flandre, D. Capacitive biosensing of bacterial cells: Analytical model and numerical simulations. Sens. Actuators B Chem. 2015, 211, 428–438. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miller, C.; Stiglich, M.; Livingstone, M.; Gilmore, J. Impedance-Based Biosensing of Pseudomonas putida via Solution Blow Spun PLA: MWCNT Composite Nanofibers. Micromachines 2019, 10, 876. https://doi.org/10.3390/mi10120876
Miller C, Stiglich M, Livingstone M, Gilmore J. Impedance-Based Biosensing of Pseudomonas putida via Solution Blow Spun PLA: MWCNT Composite Nanofibers. Micromachines. 2019; 10(12):876. https://doi.org/10.3390/mi10120876
Chicago/Turabian StyleMiller, Craig, Madison Stiglich, Mark Livingstone, and Jordon Gilmore. 2019. "Impedance-Based Biosensing of Pseudomonas putida via Solution Blow Spun PLA: MWCNT Composite Nanofibers" Micromachines 10, no. 12: 876. https://doi.org/10.3390/mi10120876
APA StyleMiller, C., Stiglich, M., Livingstone, M., & Gilmore, J. (2019). Impedance-Based Biosensing of Pseudomonas putida via Solution Blow Spun PLA: MWCNT Composite Nanofibers. Micromachines, 10(12), 876. https://doi.org/10.3390/mi10120876




