An Implantable Cranial Window Using a Collagen Membrane for Chronic Voltage-Sensitive Dye Imaging
Abstract
1. Introduction
2. Materials and Methods
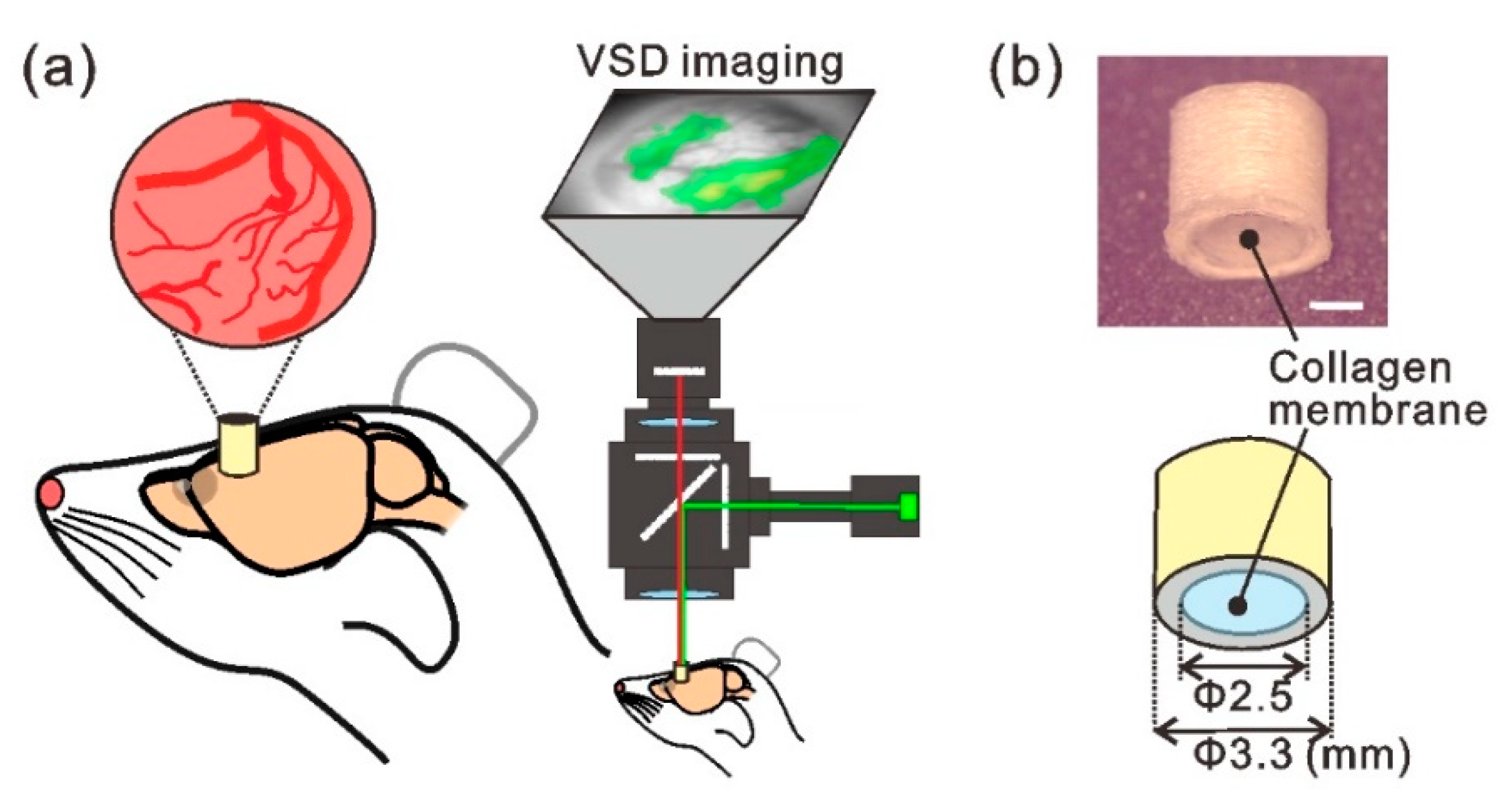
2.1. Implantable Cranial Window Device with Collagen Membrane
2.2. Animals and Surgical Procedures
2.3. Voltage-Sensitive Dye (VSD) Imaging
2.4. Histology
3. Results
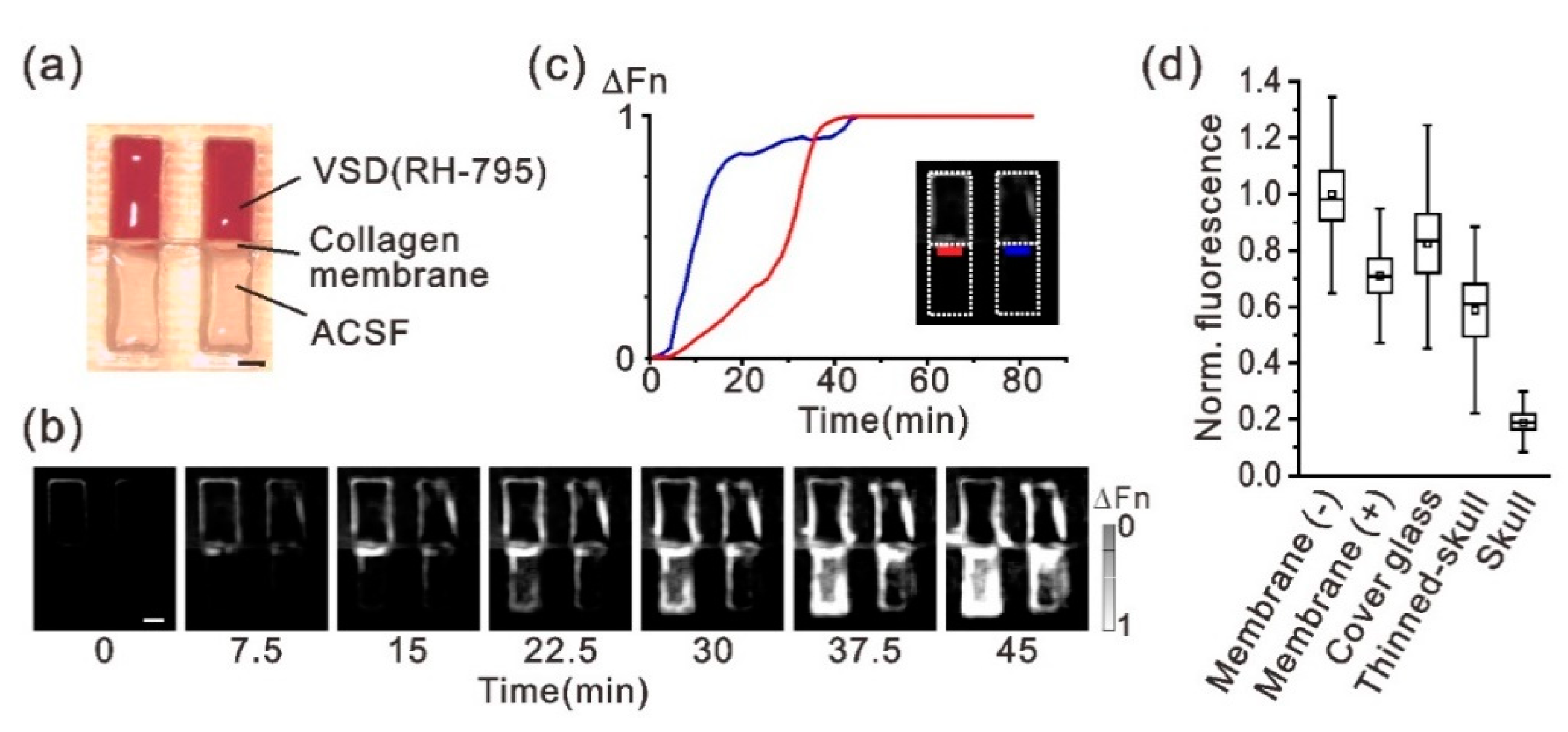
3.1. Chemical and Optical Properties of the Atelocollagen Membrane for VSD Imaging
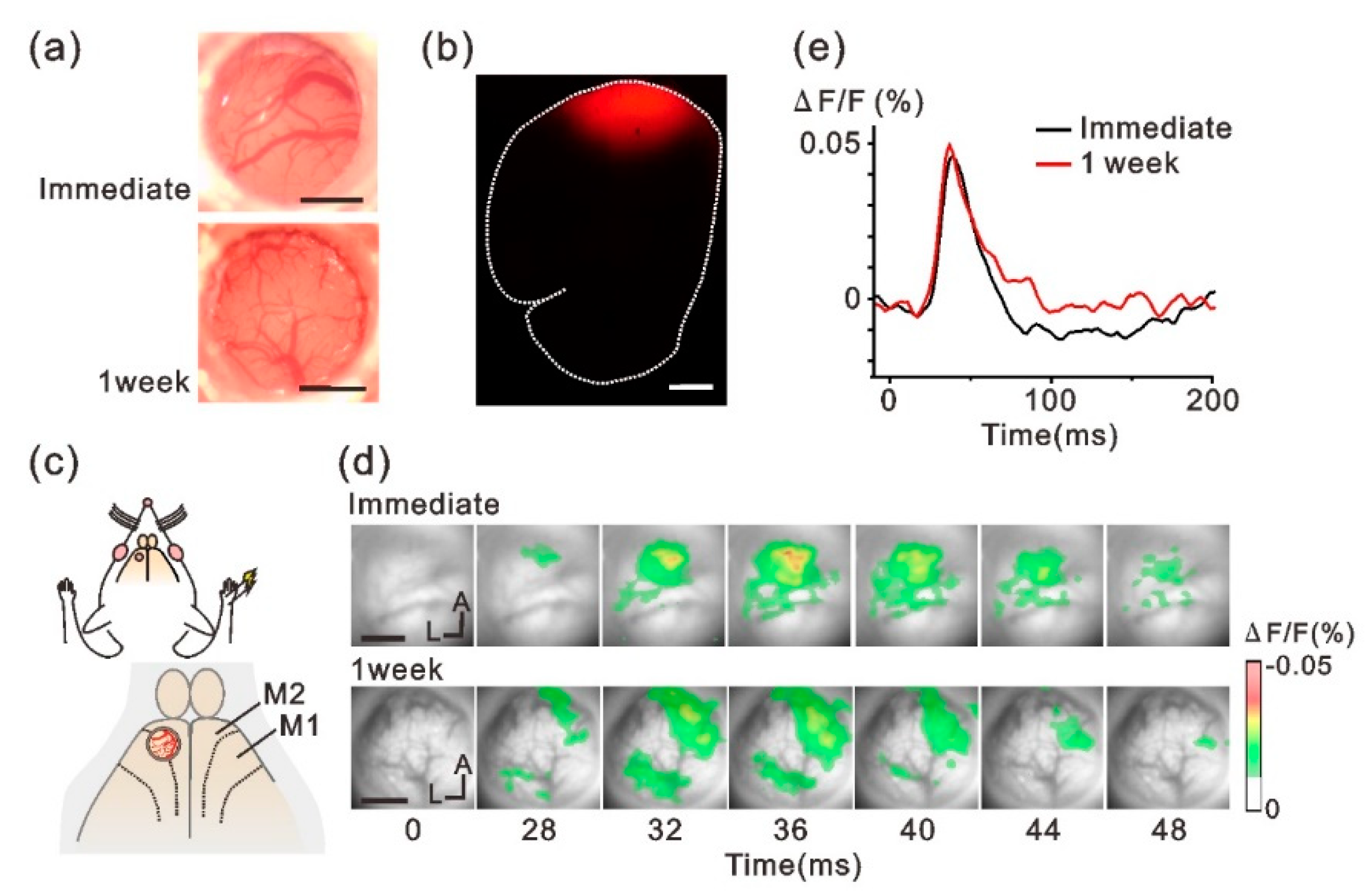
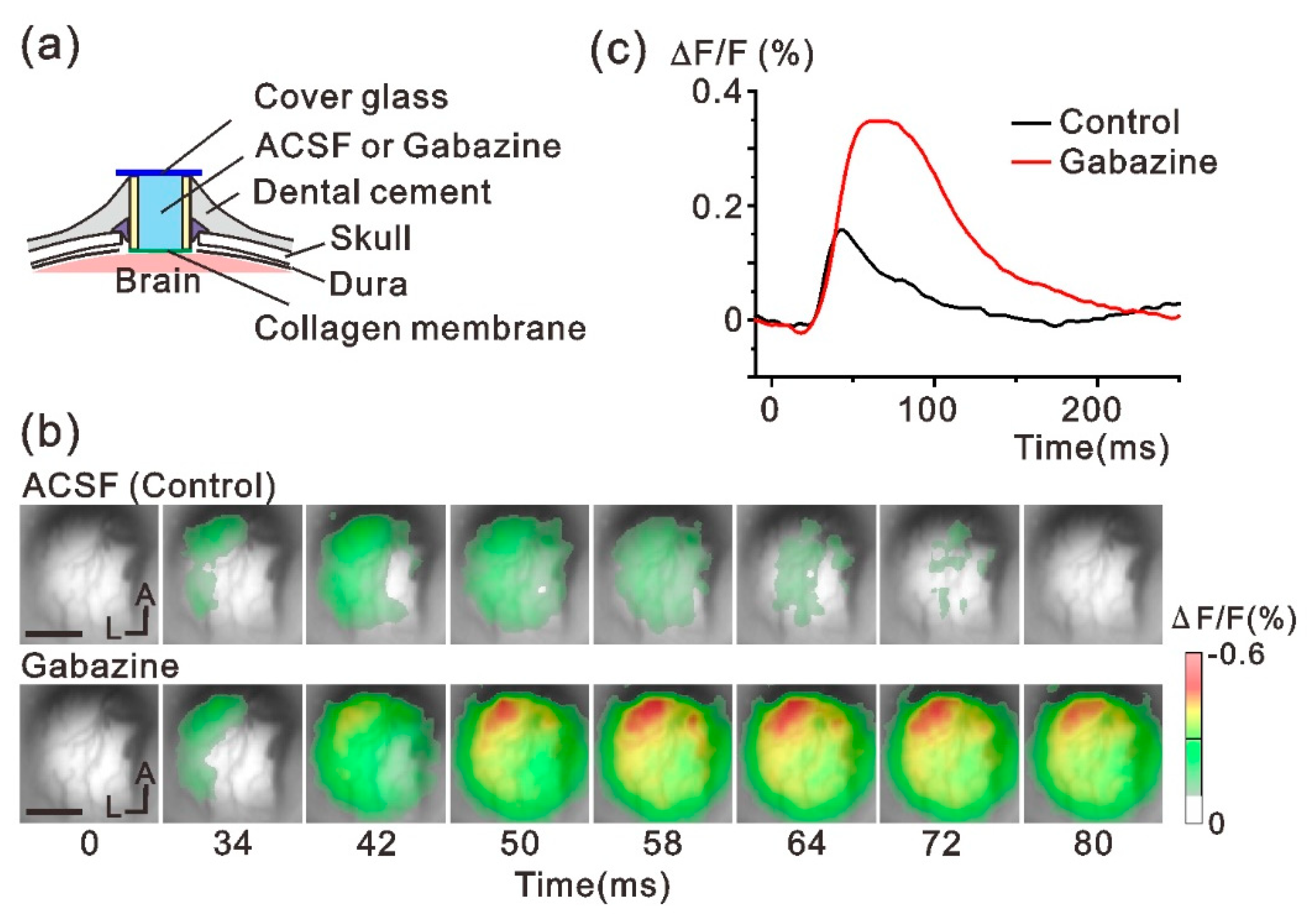
3.2. In Vivo VSD Imaging of the Sensory Response through the Implanted Window
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Choi, J.R.; Kim, S.M.; Ryu, R.H.; Kim, S.P.; Sohn, J.W. Implantable neural probes for brain-machine interfaces—Current developments and future prospects. Exp. Neurobiol. 2018, 27, 453–471. [Google Scholar] [CrossRef]
- Szostak, K.M.; Grand, L.; Constandinou, T.G. Neural interfaces for intracortical recording: Requirements, fabrication methods, and characteristics. Front. Neurosci. 2017, 11, 665. [Google Scholar] [CrossRef] [PubMed]
- Campbell, P.K.; Jones, K.E.; Huber, R.J.; Horch, K.W. A silicon-based, three-dimensional neural interface: Manufacturing processes for an intracortical electrode array. IEEE Trans. Biomed. Eng. 1991, 38, 758–768. [Google Scholar] [CrossRef] [PubMed]
- Maynard, E.M.; Nordhausen, C.T.; Normann, R.A. The Utah intracortical electrode array: A recording structure for potential brain-computer interfaces. Electroencephalogr. Clin. Neurophysiol. 1997, 102, 228–239. [Google Scholar] [CrossRef]
- Velliste, M.; Perel, S.; Spalding, M.C.; Whitford, A.S.; Schwartz, A.B. Cortical control of a prosthetic arm for self-feeding. Nature 2008, 453, 1098–1101. [Google Scholar] [CrossRef]
- Hochberg, L.R.; Serruya, M.D.; Friehs, G.M.; Mukand, J.A.; Saleh, M.; Caplan, A.H.; Branner, A.; Chen, D.; Penn, R.D.; Donoghue, J.P. Neuronal ensemble control of prosthetic devices by a human with tetraplegia. Nature 2006, 442, 164–171. [Google Scholar] [CrossRef]
- Collinger, J.L.; Wodlinger, B.; Downey, J.E.; Wang, W.; Tyler-Kabara, E.C.; Weber, D.J.; McMorland, A.J.; Velliste, M.; Boninger, M.L.; Schwartz, A.B. High-performance neuroprosthetic control by an individual with tetraplegia. Lancet 2013, 381, 557–564. [Google Scholar] [CrossRef]
- Murayama, M.; Larkum, M.E. In vivo dendritic calcium imaging with a fiberoptic periscope system. Nat. Protoc. 2009, 4, 1551–1559. [Google Scholar] [CrossRef]
- Ghosh, K.K.; Burns, L.D.; Cocker, E.D.; Nimmerjahn, A.; Ziv, Y.; Gamal, A.E.; Schnitzer, M.J. Miniaturized integration of a fluorescence microscope. Nat. Methods 2011, 8, 871–878. [Google Scholar] [CrossRef]
- Kunori, N.; Takashima, I. Voltage-sensitive dye imaging of primary motor cortex activity produced by ventral tegmental area stimulation. J. Neurosci. 2014, 34, 8894–8903. [Google Scholar] [CrossRef]
- Frost, W.N.; Wang, J.; Brandon, C.J.; Moore-Kochlacs, C.; Sejnowski, T.J.; Hill, E.S. Use of fast-responding voltage-sensitive dyes for large-scale recording of neuronal spiking activity with single-cell resolution. In Membrane Potential Imaging in the Nervous System; Canepari, M., Zecevic, D., Eds.; Springer: New York, NY, USA, 2010. [Google Scholar]
- Chen, Y.; Palmer, C.R.; Seidemann, E. The relationship between voltage-sensitive dye imaging signals and spiking activity of neural populations in primate V1. J. Neurophysiol. 2012, 107, 3281–3295. [Google Scholar] [CrossRef] [PubMed]
- Shtoyerman, E.; Arieli, A.; Slovin, H.; Vanzetta, I.; Grinvald, A. Long-term optical imaging and spectroscopy reveal mechanisms underlying the intrinsic signal and stability of cortical maps in V1 of behaving monkeys. J. Neurosci. 2000, 20, 8111–8121. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.M.; Heider, B.; Williams, G.V.; Healy, F.L.; Ramsden, B.M.; Roe, A.W. A chamber and artificial dura method for long-term optical imaging in the monkey. J. Neurosci. Methods 2002, 113, 41–49. [Google Scholar] [CrossRef]
- Lu, H.D.; Chen, G.; Tanigawa, H.; Roe, A.W. A motion direction map in macaque V2. Neuron 2010, 68, 1002–1013. [Google Scholar] [CrossRef] [PubMed]
- Arieli, A.; Grinvald, A.; Slovin, H. Dural substitute for long-term imaging of cortical activity in behaving monkeys and its clinical implications. J. Neurosci. Methods 2002, 114, 119–133. [Google Scholar] [CrossRef]
- Slovin, H.; Arieli, A.; Hildesheim, R.; Grinvald, A. Long-term voltage-sensitive dye imaging reveals cortical dynamics in behaving monkeys. J. Neurophysiol. 2002, 88, 3421–3438. [Google Scholar] [CrossRef]
- Imaizumi, F.; Asahina, I.; Moriyama, T.; Ishii, M.; Omura, K. Cultured mucosal cell sheet with a double layer of keratinocytes and fibroblasts on a collagen membrane. Tissue Eng. 2004, 10, 657–664. [Google Scholar] [CrossRef]
- Nagamine, K.; Hirata, T.; Okamoto, K.; Abe, Y.; Kaji, H.; Nishizawa, M. Portable micropatterns of neuronal cells supported by thin hydrogel films. ACS Biomater. Sci. Eng. 2015, 1, 329–334. [Google Scholar] [CrossRef]
- Kunori, N.; Takashima, I. A transparent epidural electrode array for use in conjunction with optical imaging. J. Neurosci. Methods 2015, 251, 130–137. [Google Scholar] [CrossRef]
- Takashima, I.; Ichikawa, M.; Iijima, T. High-speed CCD imaging system for monitoring neural activity in vivo and in vitro, using a voltage-sensitive dye. J. Neurosci. Methods 1999, 91, 147–159. [Google Scholar] [CrossRef]
- Paxinos, G.; Watson, C. The Rat Brain in Stereotaxic Coordinates, 7th ed.; Academic Press: San Diego, CA, USA, 2013. [Google Scholar]
- Kunori, N.; Takashima, I. High-order motor cortex in rats receives somatosensory inputs from the primary motor cortex via cortico-cortical pathways. Eur. J. Neurosci. 2016, 44, 2925–2934. [Google Scholar] [CrossRef] [PubMed]
- Treger, J.S.; Priest, M.F.; Iezzi, R.; Bezanilla, F. Real-time imaging of electrical signals with an infrared FDA-approved dye. Biophys. J. 2014, 107, L09–L12. [Google Scholar] [CrossRef] [PubMed]
- Takashima, I.; Kajiwara, R.; Iijima, T. Voltage-sensitive dye versus intrinsic signal optical imaging: Comparison of optically determined functional maps from rat barrel cortex. Neuroreport 2001, 12, 2889–2894. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, Y.; Kajiwara, R.; Takashima, I. Optical imaging of rat prefrontal neuronal activity evoked by stimulation of the ventral tegmental area. Neuroreport 2009, 20, 875–880. [Google Scholar] [CrossRef] [PubMed]
- Grinvald, A.; Hildesheim, R. VSDI: A new era in functional imaging of cortical dynamics. Nat. Rev. Neurosci. 2004, 5, 874–885. [Google Scholar] [CrossRef]
- Omer, D.B.; Fekete, T.; Ulchin, Y.; Hildesheim, R.; Grinvald, A. Dynamic patterns of spontaneous ongoing activity in the visual cortex of anesthetized and awake monkeys are different. Cereb. Cortex 2019, 29, 1291–1304. [Google Scholar] [CrossRef]
- Ferezou, I.; Bolea, S.; Petersen, C.C. Visualizing the cortical representation of whisker touch: Voltage-sensitive dye imaging in freely moving mice. Neuron 2006, 50, 617–629. [Google Scholar] [CrossRef] [PubMed]
- Nassi, J.J.; Cetin, A.H.; Roe, A.W.; Callaway, E.M.; Deisseroth, K.; Reynolds, J.H. A precise and minimally invasive approach to optogenetics in the awake primate. Proc. SPIE 2013, 8586, 8586A. [Google Scholar]
- Ruiz, O.; Lustig, B.R.; Nassi, J.J.; Cetin, A.; Reynolds, J.H.; Albright, T.D.; Callaway, E.M.; Stoner, G.R.; Roe, A.W. Optogenetics through windows on the brain in the nonhuman primate. J. Neurophysiol. 2013, 110, 1455–1467. [Google Scholar] [CrossRef]
- Ucar, B.; Humpel, C. Collagen for brain repair: Therapeutic perspectives. Neural Regen. Res. 2018, 13, 595–598. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kunori, N.; Takashima, I. An Implantable Cranial Window Using a Collagen Membrane for Chronic Voltage-Sensitive Dye Imaging. Micromachines 2019, 10, 789. https://doi.org/10.3390/mi10110789
Kunori N, Takashima I. An Implantable Cranial Window Using a Collagen Membrane for Chronic Voltage-Sensitive Dye Imaging. Micromachines. 2019; 10(11):789. https://doi.org/10.3390/mi10110789
Chicago/Turabian StyleKunori, Nobuo, and Ichiro Takashima. 2019. "An Implantable Cranial Window Using a Collagen Membrane for Chronic Voltage-Sensitive Dye Imaging" Micromachines 10, no. 11: 789. https://doi.org/10.3390/mi10110789
APA StyleKunori, N., & Takashima, I. (2019). An Implantable Cranial Window Using a Collagen Membrane for Chronic Voltage-Sensitive Dye Imaging. Micromachines, 10(11), 789. https://doi.org/10.3390/mi10110789




