Mycotoxin Contamination in the EU Feed Supply Chain: A Focus on Cereal Byproducts
Abstract
:1. Introduction
2. Global Occurrence of Mycotoxins in Feed
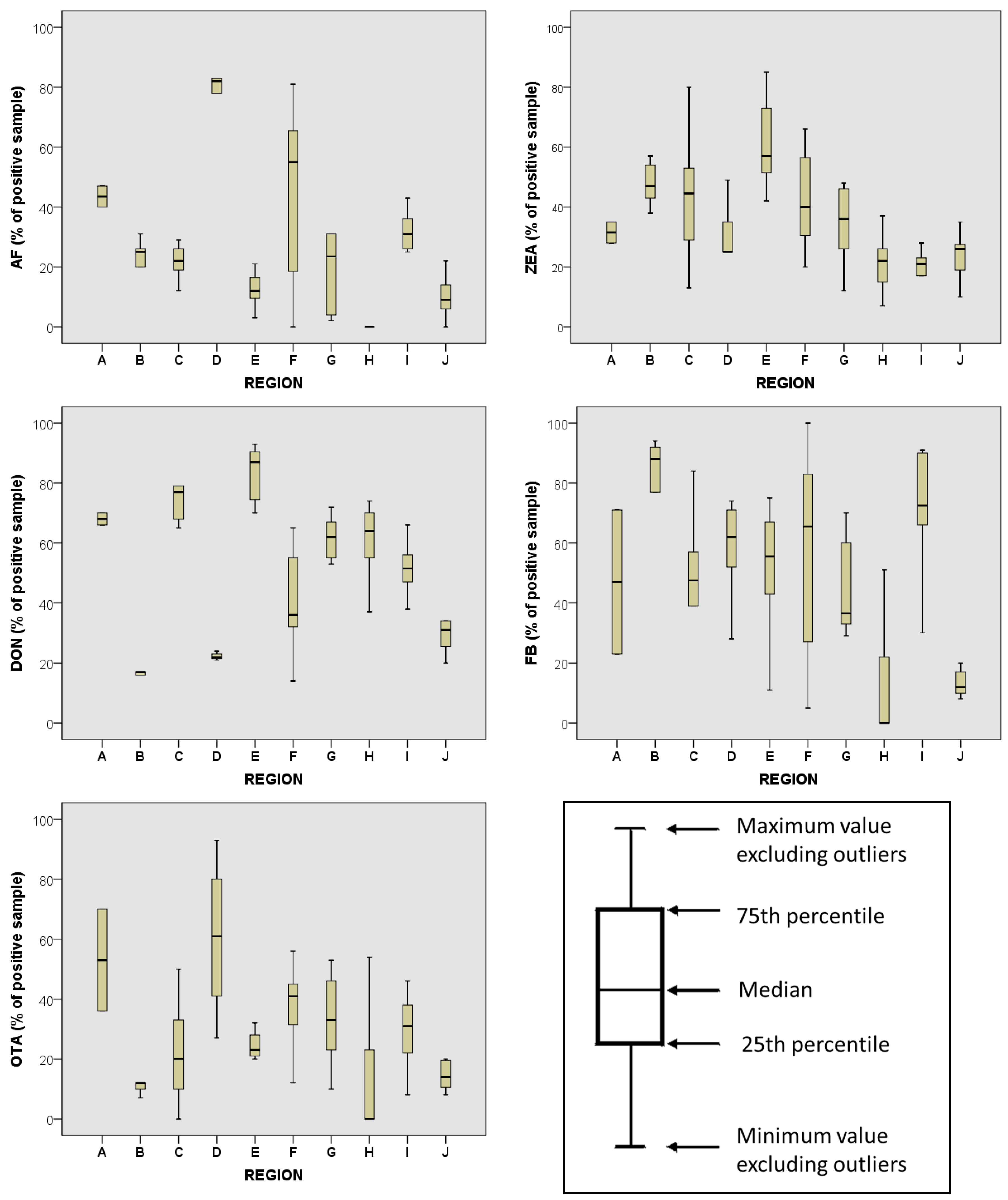
| Geographical Region | AFB1 | ZEA | DON | FB1 | OTA |
|---|---|---|---|---|---|
| North America | 8 | 271 | 1,947 | 902 | 1 |
| Central South America | 2–3 | 0–111 | 51–237 | 1030–3121 | 0–9 |
| Europe | 0–3 | 3–37 | 88–968 | 925–3052 | 0–9 |
| Asia | 8–90 | 32–219 | 61–691 | 380–797 | 1–15 |
| Oceania | 1 | 50 | 94 | 109 | 1 |
| Africa | 42 | 25 | 745 | 855 | 6 |
3. Mycotoxins in Feed: Economic and Legislative Context
| Mycotoxin | Cereal and Cereal Products | Maximum Levels, µg/kg |
|---|---|---|
| Aflatoxin B1 | All cereals and all products derived from cereals | 2.0 |
| Maize to be subjected to sorting or other physical treatment before human consumption or use as an ingredient in foodstuffs | 5.0 | |
| Aflatoxins, sum of B1, B2, G1 and G2 | All cereals and all products derived from cereals | 4.0 |
| Maize to be subjected to sorting or other physical treatment before human consumption or use as an ingredient in foodstuffs | 10.0 | |
| Deoxynivalenol | Unprocessed cereals other than durum wheat, oats and maize | 1250 |
| Unprocessed durum wheat and oats | 1750 | |
| Unprocessed maize, with the exception of unprocessed maize intended to be processed by wet milling | 1750 | |
| Cereals intended for direct human consumption, cereal flour, bran and germ as end product marketed for direct human consumption | 750 | |
| Zearalenone | Unprocessed cereals other than maize | 100 |
| Unprocessed maize with the exception of unprocessed maize intended to be processed by wet milling | 350 | |
| Cereals intended for direct human consumption, cereal flour, bran and germ as end product marketed for direct human consumption | 75 | |
| Maize intended for direct human consumption, maize-based snacks and maize-based breakfast cereals | 100 | |
| Ochratoxin A | Unprocessed cereals | 5.0 |
| All products derived from unprocessed cereals, including processed cereal products and cereals intended for direct human consumption | 3.0 | |
| Fumonisin B1 + B2 | Unprocessed maize, with the exception of unprocessed maize intended to be processed by wet milling | 4000 |
| Maize intended for direct human consumption, maize-based foods for direct human consumption | 1000 | |
| Sum T-2 and HT-2 toxin(*) | Unprocessed cereals | - |
| Barley and maize | 200 | |
| Oats | 1000 | |
| Wheat, rye and other cereals | 100 | |
| Sum T-2 and HT-2 toxin(*) | Cereals grains for direct human consumption | - |
| Oats | 200 | |
| Maize | 100 | |
| Other cereals | 50 |
| Mycotoxin | Cereal and Cereal Products | Maximum Levels, mg/kg |
|---|---|---|
| Aflatoxin B1(*) | All feed materials | 0.02 |
| Complete feedstuffs for cattle, sheep and goats with the exception of: | 0.02 | |
| Complete feedstuffs for dairy animals | 0.005 | |
| Complete feedstuffs for calves and lambs | 0.01 | |
| Complete feedstuffs for pigs and poultry (except young animals) | 0.02 | |
| Other complete feedstuffs | 0.01 | |
| Complementary feedstuffs for cattle, sheep and goats (except Complementary feedstuffs for dairy animals, calves and lambs) | 0.02 | |
| Complementary feedstuffs for pigs and poultry (except young animals) | - | |
| Other complementary feedstuffs | 0.02 | |
| Complete feedstuffs for cattle, sheep and goats with the exception of: | 0.005 | |
| Deoxynivalenol | Feed materials | - |
| Cereals and cereal products with the exception of maize byproducts | 8 | |
| Maize byproducts | 12 | |
| Complementary and complete feedstuffs with the exception of: | 5 | |
| Complementary and complete feedstuffs for pigs | 0.9 | |
| Complementary and complete feedstuffs for calves (<4 months), lambs and kids | 2 | |
| Zearalenone | Feed materials | - |
| Cereals and cereal products with the exception of maize byproducts | 2 | |
| Maize byproducts | 3 | |
| Complementary and complete feedstuffs | - | |
| Complementary and complete feedstuffs for piglets and gilts (young sows) | 0.1 | |
| Complementary and complete feedstuffs for sows and fattening pigs | 0.25 | |
| Complementary and complete feedstuffs for calves, dairy cattle, sheep (including lambs) and goats (including kids) | 0.5 | |
| Ochratoxin A | Feed materials | - |
| Cereals and cereal products | 0.25 | |
| Complementary and complete feedstuffs | - | |
| Complementary and complete feedstuffs for pigs | 0.05 | |
| Complementary and complete feedstuffs for poultry | 0.1 | |
| Fumonisin B1 + B2 | Feed materials | - |
| Maize and maize products | 60 | |
| Complementary and complete feedstuffs | - | |
| Pigs, horses (Equidae), rabbits and pet animals | 5 | |
| fish | 10 | |
| Poultry, calves (<4 months), lambs and kids | 20 | |
| adult ruminants (>4 months) and mink | 50 | |
| Sum T-2 and HT-2 toxin | Cereal products for feed and complementary feed | - |
| Oat milling products | 2000 | |
| Other cereal products | 500 |
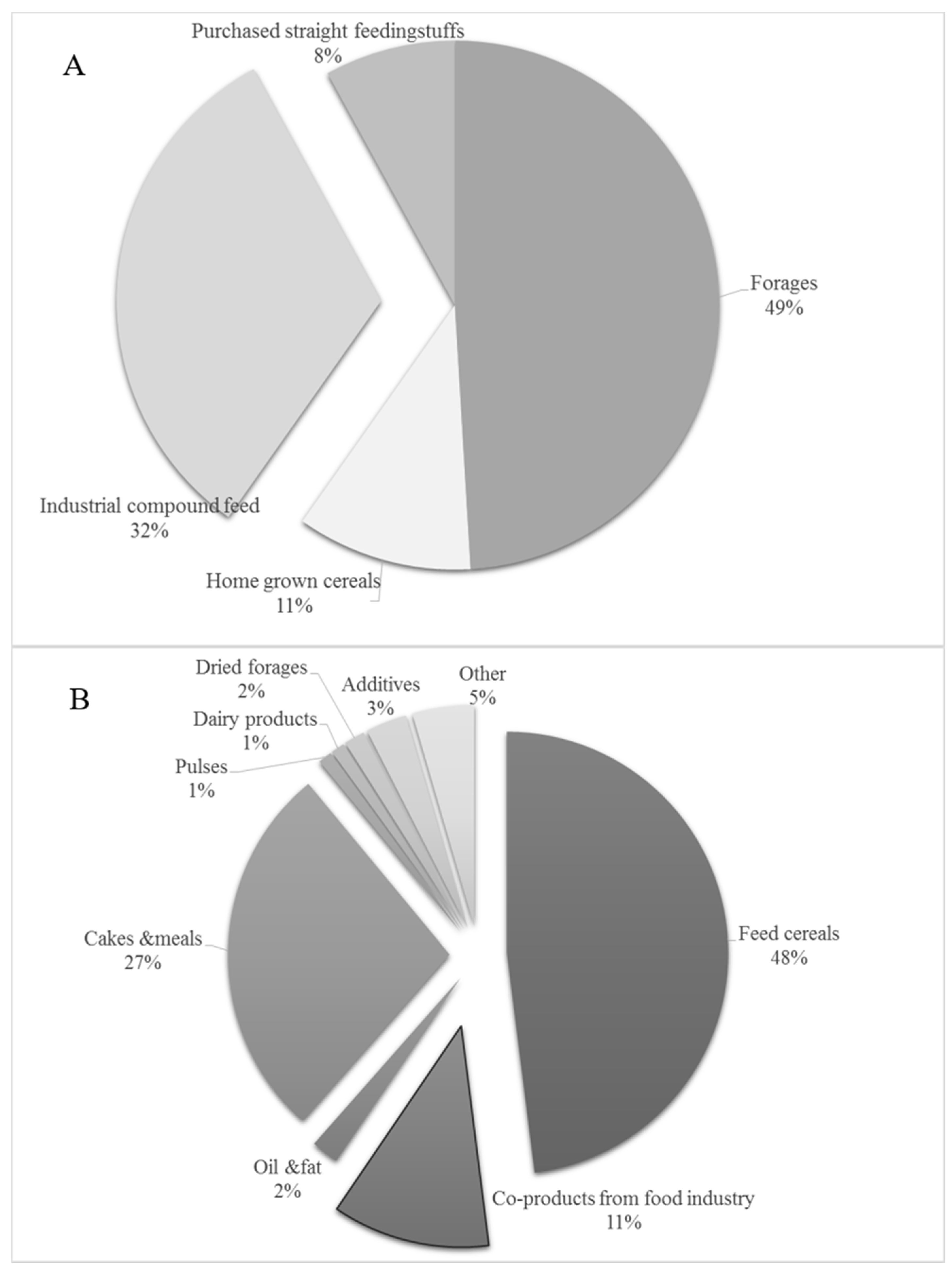
4. Cereals and Cereal Byproducts as Animal Feed
5. Fate of Mycotoxins during Cereal Processing
5.1. Cereal Milling
5.2. Byproduct from Bioethanol Production
5.3. Brewing
6. Industrial Use of Cereal Byproducts: Implication for Mycotoxins
| Level in the Industrial Process | Factor |
|---|---|
| Mycotoxin concentration in (original) cereal grains | Type and level of mycotoxin contamination Cereal processing technology Type of byproducts |
| Mycotoxin concentration in byproducts | Mycotoxin analysis of byproducts |
| Limits/practice in byproducts use |
7. Conclusions and Future Perspective
Conflicts of Interest
References
- FEFAC. The Compound Feed Industry in the EU Livestock Economy. Available online: http://www.fefac.eu/files/62107.pdf (accessed on 13 November 2015).
- Pinotti, L.; Dell’Orto, V. Feed safety in the feed supply chain. Biotechnol. Agron. Soc. Environ. 2011, 15, 9–14. [Google Scholar]
- Bryden, W.L. Mycotoxin contamination of the feed supply chain: Implications for animal productivity and feed security. Anim. Feed Sci. Technol. 2012, 173, 134–158. [Google Scholar] [CrossRef]
- Hussein, H.S.; Brasel, J.M. Toxicity, metabolism, and impact of mycotoxins on human and animals. Toxicology 2001, 167, 101–134. [Google Scholar] [CrossRef]
- Wild, C.P.; Gong, Y.Y. Mycotoxins and human disease: A largely ignored global health issue. Carcinogenesis 2010, 31, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Wu, F. Measuring the economic impacts of Fusarium toxins in animal feeds. Anim. Feed Sci. Technol. 2007, 137, 363–374. [Google Scholar] [CrossRef]
- Wu, F. Global impacts of aflatoxin in maize: Trade and human health. World Mycotoxin J. 2015, 8, 137–142. [Google Scholar] [CrossRef]
- Pulina, G.; Battacone, G.; Brambilla, G.; Cheli, F.; Danieli, P.P.; Masoero, F.; Pietri, A.; Ronchi, B. An update on the safety of foods of animal origin and feeds. Ital. J. Anim. Sci. 2014, 13, 845–856. [Google Scholar] [CrossRef]
- Binder, E.M.; Tan, L.M.; Chin, L.J.; Handl, J.; Richard, J. Worldwide occurrence of mycotoxins in commodities, feeds and feed ingredients. Anim. Feed Sci. Technol. 2007, 137, 265–282. [Google Scholar] [CrossRef]
- Marin, S.; Ramos, A.J.; Cano-Sancho, G.; Sanchis, V. Mycotoxins: Occurrence, toxicology, and exposure assessment. Food Chem. Toxicol. 2013, 60, 218–237. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, I.; Naehrer, K. A three-year survey on the worldwide occurrence of mycotoxins in feedstuffs and feed. Toxins 2012, 4, 663–675. [Google Scholar] [CrossRef] [PubMed]
- Streit, E.; Schatzmayr, G.; Tassis, P.; Tzika, E.; Marin, D.; Taranu, I.; Tabuc, C.; Nicolau, A.; Aprodu, I.; Puel, O.; et al. Current Situation of Mycotoxin Contamination and Co-occurrence in Animal Feed—Focus on Europe. Toxins 2012, 4, 788–809. [Google Scholar] [CrossRef] [PubMed]
- Streit, E.; Naehrer, K.; Rodrigues, I.; Schatzmayr, G. Mycotoxin occurrence in feed and feed raw materials worldwide: Long-term analysis with special focus on Europe and Asia. J. Sci. Food Agric. 2013, 93, 2892–2899. [Google Scholar] [CrossRef] [PubMed]
- FEFAC. Annual Report 2014–2015. Available online: http://www.fefac.eu/files/60917.pdf (accessed on 13 November 2015).
- Cheli, F.; Pinotti, L.; Rossi, L.; Dell’Orto, V. Effect of milling procedures on mycotoxin distribution in wheat fractions: A review. LWT-Food Sci. Technol. 2013, 54, 307–314. [Google Scholar] [CrossRef]
- Zhang, Y.; Caupert, J. Survey of Mycotoxins in U.S. Distiller’s Dried Grains with Solubles from 2009 to 2011. J. Agric. Food Chem. 2012, 60, 539–543. [Google Scholar] [CrossRef] [PubMed]
- Lancova, K.; Hajslova, J.; Poustka, J.; Krplova, A.; Zachariasova, M.; Dostalek, P.; Sachambula, L. Transfer of Fusarium mycotoxins and ‘masked’ deoxynivalenol (deoxynivalenol-3-glucoside) from field barley through malt to beer. Food Addit. Contam. A 2008, 25, 732–744. [Google Scholar] [CrossRef] [PubMed]
- Cheli, F.; Campagnoli, A.; Dell’Orto, V. Fungal populations and mycotoxins in silages: From occurrence to analysis. Anim. Feed Sci. Technol. 2013, 183, 1–16. [Google Scholar] [CrossRef]
- Driehuis, F.; Spanjer, M.C.; Scholten, J.M.; Te Giffel, M.C. Occurrence of mycotoxins in feedstuffs of dairy cows and estimation of total dietary intakes. J. Dairy Sci. 2008, 91, 4261–4271. [Google Scholar] [CrossRef] [PubMed]
- Alonso, V.A.; Pereyra, C.M.; Keller, L.A.M.; Dalcero, A.M.; Rosa, C.A.R.; Chiacchiera, S.M.; Cavaglieri, L.R. Fungi and mycotoxins in silage: An overview. J. Appl. Mirobiol. 2013, 115, 637–643. [Google Scholar] [CrossRef] [PubMed]
- Nichea, M.J.; Palacios, S.A.; Chiacchiera, S.M.; Sulyok, M.; Krska, R.; Chulze, S.N.; Torres, A.M.; Ramirez, M.L. Presence of multiple mycotoxins and other fungal metabolites in native grasses from a wetland ecosystem in Argentina intended for grazing cattle. Toxins 2015, 7, 3309–3329. [Google Scholar] [CrossRef] [PubMed]
- CAST. Mycotoxins: Risks in Plant, Animal, and Human Systems; Task Force Report 2003, No. 139; Council for Agricultural Science and Technology: Ames, IA, USA, 2003. [Google Scholar]
- Borutova, R.; Acosta Aragon, Y.; Nährer, K.; Berthiller, F. Co-occurrence and statistical correlations between mycotoxins in feedstuffs collected in the Asia-Oceania in 2010. Anim. Feed Sci. Technol. 2012, 178, 190–197. [Google Scholar] [CrossRef]
- Schatzmayr, G.; Streit, E. Global occurrence of mycotoxins in the food and feed chain: Facts and figures. World Mycotoxin J. 2013, 6, 213–222. [Google Scholar] [CrossRef]
- Marquardt, R.R.; Madhyastha, S. Mycotoxins in feed and animal products. In Book of Abstract, Proceedings of the 1st World Conference on Innovative Animal Nutrition and Feeding, Budapest, Hungary, 15–17 October 2015; Akadémiai Kiadó: Budapest, Hungary, 2015; pp. 58–63. [Google Scholar]
- Jestoi, M. Emerging Fusarium mycotoxins fusaproliferin, beauvericin, enniatins, and moniliformin—A review. Crit. Rev. Food Sci. Nutr. 2008, 48, 21–49. [Google Scholar] [CrossRef] [PubMed]
- Mortensen, A.; Granby, K.; Eriksen, F.D.; Cederberg, T.L.; Friis-Wandall, S.; Simonsen, Y.; Broesbøl-Jensen, B.; Bonnichsen, R. Levels and risk assessment of chemical contaminants in byproducts for animal feed in Denmark. J. Environ. Sci. Health Part B 2014, 49, 797–810. [Google Scholar] [CrossRef] [PubMed]
- Øverland, M.; Krogdahl, A.; Shurson, G.; Skrede, A.; Denstadli, V. Evaluation of distiller’s dried grains with solubles (DDGS) and high protein distiller’s dried grains (HPDDG) in diets for rainbow trout (Oncorhynchus mykiss). Aquaculture 2013, 416–417, 201–208. [Google Scholar] [CrossRef]
- Streit, E.; Schwab, C.; Sulyok, M.; Naehrer, K.; Krska, R.; Schatzmayr, G. Multi-mycotoxin screening reveals the occurrence of 139 different secondary metabolites in feed and feed ingredients. Toxins 2013, 5, 504–523. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.E.; Kim, B.H.; Lee, C. Occurrence of Fusarium mycotoxin beauvericin in animal feeds in Korea. Anim. Feed Sci. Technol. 2010, 157, 190–194. [Google Scholar] [CrossRef]
- Cotty, P.J.; Jaime-Garcia, R. Effect of climate on aflatoxin producing fungi and aflatoxin contamination. Int. J. Food Microbiol. 2007, 119, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Munkvold, G.P. Cultural and genetic approaches to managing mycotoxins in maize. Annu. Rev. Phytopathol. 2003, 41, 99–116. [Google Scholar] [CrossRef] [PubMed]
- Teller, R.S.; Schmidt, R.J.; Whitlow, L.W.; Kung, L., Jr. Effect of physical damage to ears of corn before harvest and treatment with various additives on the concentration of mycotoxins, silage fermentation, and aerobic stability of corn silage. J. Dairy Sci. 2012, 95, 1428–1436. [Google Scholar] [CrossRef] [PubMed]
- Reyneri, A. The role of climatic condition on micotoxin production in cereal. Vet. Res. Comm. 2006, 30, 87–92. [Google Scholar] [CrossRef]
- Paterson, R.R.M.; Lima, N. How will climate change affect mycotoxins in food? Food Res. Int. 2010, 43, 1902–1914. [Google Scholar] [CrossRef]
- European Commission. Commission Directive 2003/100/EC of 31 October 2003 amending Annex I to Directive 2002/32/EC of the European Parliament and of the Council on undesirable substances in animal feed. Off. J. L 2003, 285, 33–37. [Google Scholar]
- Mngadi, P.T.; Govinden, R.; Odhav, B. Co-occurring mycotoxins in animal feeds. Afr. J. Biotechnol. 2008, 7, 2239–2243. [Google Scholar]
- Alassane-Kpembi, I.; Kolf-Clauw, M.; Gauthier, T.; Abrami, R.; Abiola, F.A.; Oswald, I.P.; Puel, O. New insights into mycotoxin mixtures: The toxicity of low doses of Type B trichothecenes on intestinal epithelial cells is synergistic. Toxicol. Appl. Pharmacol. 2013, 272, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Cheli, F.; Giromini, C.; Baldi, A. Mycotoxin mechanisms of action and health effects: “In vitro” or “in vivo” tests, that is the question. World Mycotoxin J. 2015, 8, 573–589. [Google Scholar] [CrossRef]
- Grenier, B.; Oswald, I. Mycotoxin co-contamination of food and feed: Meta-analysis of publications describing toxicological interactions. World Mycotoxin J. 2014, 3, 285–313. [Google Scholar] [CrossRef]
- Pedrosa, K.; Borutova, R. Synergistic effects of mycotoxins discussed. Feedstuffs 2011, 83, 1–3. [Google Scholar]
- Speijers, G.J.A.; Speijers, M.H.M. Combined toxic effects of mycotoxins. Toxicol. Lett. 2004, 153, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Tavares, A.M.; Alvito, P.; Loureiro, S.; Louro, H.; Silva, M.J. Multi-mycotoxin determination in baby foods and in vitro combined cytotoxic effects of aflatoxin M1 and ochratoxin A. World Mycotoxin J. 2013, 6, 375–388. [Google Scholar] [CrossRef]
- EFSA. Scientific Opinion on the risks for human and animal health related to the presence of modified forms of certain mycotoxins in food and feed. EFSA J. 2014, 12, 3916–4023. [Google Scholar]
- Berthiller, F.; Crews, C.; Dall’Asta, C.; De Saeger, S.; Haesaert, G.; Karlovsky, P.; Oswald, I.P.; Seefelder, W.; Speijers, G.; Stroka, J. Masked mycotoxins: A review. Mol. Nutr. Food Res. 2013, 57, 165–186. [Google Scholar] [CrossRef] [PubMed]
- Berthiller, F.; Dall’Asta, C.; Schuhmacher, R.; Lemmens, M.; Adam, G.; Krska, R. Masked mycotoxins: Determination of a deoxynivalenol glucoside in artificially and naturally contaminated wheat by liquid chromatography-tandem mass spectrometry. J. Agric. Food Chem. 2005, 53, 3421–3425. [Google Scholar] [CrossRef] [PubMed]
- Berthiller, F.; Dall’Asta, C.; Corradini, R.; Marchelli, R.; Sulyok, M.; Krska, R.; Adam, G.; Schuhmacher, R. Occurrence of deoxynivalenol and its 3-β-d-glucoside in wheat and maize. Food Addit. Contam. A 2009, 26, 507–511. [Google Scholar] [CrossRef] [PubMed]
- Kostelanska, M.; Dzuman, Z.; Malachova, A.; Capouchova, I.; Prokinova, E.; Skerikova, A.; Hajslova, J. Effects of milling and baking technologies on levels of deoxynivalenol and its masked form deoxynivalenol-3-glucoside. J. Agric. Food Chem. 2011, 59, 9303–9312. [Google Scholar] [CrossRef] [PubMed]
- Sasanya, J.J.; Hall, C.; Wolf-Hall, C. Analysis of deoxynivalenol, masked deoxynivalenol, and Fusarium graminearum pigment in wheat samples, using liquid chromatography-UV-mass spectrometry. J. Food Prot. 2008, 71, 1205–1213. [Google Scholar] [PubMed]
- Dall’Erta, A.; Cirlini, M.; Dall’Asta, M.; del Rio, D.; Galaverna, G.; Dall’Asta, C. Masked mycotoxins are efficiently hydrolyzed by human colonic microbiota releasing their aglycones. Chem. Res. Toxicol. 2013, 26, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Nagl, V.; Woechtl, B.; Schwartz-Zimmermann, H.E.; Hennig-Pauka, I.; Moll, W.D.; Adam, G.; Berthiller, F. Metabolism of the masked mycotoxin deoxynivalenol-3-glucoside in pigs. Toxicol. Lett. 2014, 229, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Nagl, V.; Schwartz, H.; Krska, R.; Moll, W.D.; Knasmüller, S.; Ritzmann, M.; Adam, G.; Berthiller, F. Metabolism of the masked mycotoxin deoxynivalenol-3-glucoside in rats. Toxicol. Lett. 2012, 3, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Pierron, A.; Mimoun, S.; Murate, L.; Loiseau, N.; Lippi, Y.; Bracarense, A.P.F.L.; Liaubet, L.; Schatmayr, G.; Berthiller, F.; Moll, W.D.; et al. Intestinal toxicity of the masked mycotoxin Deoxynivalenol-3-β-d-glucoside. Arch. Toxicol. 2015. [Google Scholar] [CrossRef] [PubMed]
- FAO—Food and Agriculture Organization. Worldwide regulations for mycotoxins in food and feed in 2003. In FAO Food and Nutrition Paper 81; Food and Agriculture Organization of the United Nations: Rome, Italy, 2004; Available online: http://www.fao.org/docrep/007/y5499e/y5499e00.htm (accessed on 13 November 2015).
- Van Egmond, H.P.; Schothorst, R.C.; Jonker, M.A. Regulations relating to mycotoxins in food. Perspectives in a global and European context. Review. Anal. Bioanal. Chem. 2007, 389, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Cheli, F.; Gallo, R.; Battaglia, D.; Dell’Orto, V. EU legislation on feed related issues: An update. Ital. J. Anim. Sci. 2013, 12, 295–312. [Google Scholar] [CrossRef]
- Cheli, F.; Battaglia, D.; Gallo, R.; Dell’Orto, V. EU legislation on cereal safety: An update with a focus on mycotoxins. Food Control 2014, 37, 315–325. [Google Scholar] [CrossRef]
- European Commission. Commission Recommendation 2006/576/EC of 17 August 2006 on the presence of deoxynivalenol, zearalenone, ochratoxin A, T-2 and HT-2 and fumonisins in products intended for animal feeding. Off. J. L 2006, 229, 7–9. [Google Scholar]
- EFSA Scientific Opinion. Scientific Opinion on the risks to human and animal health related to the presence of beauvericin and enniatins in food and feed. EFSA J. 2014, 12, 174. [Google Scholar]
- Commission Regulation (EU) 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs. Off. J. L 2006, 364, 5–24.
- Commission Recommendation No 2013/165/EU of 27 March 2013 on the presence of T-2 and HT-2 toxin in cereals and cereal products. Off. J. L 2013, 91, 12–15.
- Rapid Alert System for Food and Feed (RASFF) Annual Report 2013. Available online: http://ec.europa.eu/food/safety/rasff/docs/rasff_annual_report_2013.pdf (accessed on 13 November 2015).
- Rapid Alert System for Food and Feed (RASFF) Preliminary Annual Report 2014. Available online: http://ec.europa.eu/food/safety/rasff/docs/rasff_preliminary_annual_report_2014_en.pdf (accessed on 13 November 2015).
- Food and Agriculture Organization of the United Nations (FAO). Cereal Supply and Demand Brief. In FAO Cereal Supply and Demand Situation. 2015. Available online: http://www.fao.org/worldfoodsituation/csdb/en/ (accessed on 13 November 2015).
- Best, P. World Feed Panorama: Expensive grain slows industry expansion. Feed Int. 2011, 32, 10–12. [Google Scholar]
- COCERAL (Comité du Commerce des Céréales, Aliments du Bétail, Oléagineux, huile d’olive, huiles et graisses et Agrofournitures). EU-28 Grain Crops Forecasts. December 2014. Available online: http://www.coceral.com/data/1418633076Final_Coceral%20grain%20estimate_2014_December.pdf (accessed on 13 November 2015).
- Pinotti, L.; Krogdahl, A.; Givens, I.; Knight, C.; Baldi, A.; Baeten, V.; van Raamsdonk, L.; Woodgate, S.; Perez Marin, D.; Luten, J. The role of animal nutrition in designing optimal foods of animal origin as reviewed by the COST Action Feed for Health (FA0802). BASE 2014, 18, 1–9. [Google Scholar]
- Siuda, R.; Grabowski, A.; Lenc, L.; Ralcewicz, M.; Spychaj-Fabisiak, E. Influence of the degree of fusariosis on technological traits of wheat grain. Int. J. Food Sci. Technol. 2010, 45, 2596–2604. [Google Scholar] [CrossRef]
- Bullerman, L.B.; Bianchini, A. Stability of mycotoxins during food processing. Int. J. Food Microbiol. 2007, 119, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Kabak, B. The fate of mycotoxins during thermal food processing. J. Sci. Food Agric. 2009, 89, 549–554. [Google Scholar] [CrossRef]
- Kushiro, M. Effects of milling and cooking processes on the deoxynivalenol content in wheat. Int. J. Mol. Sci. 2008, 9, 2127–2145. [Google Scholar] [CrossRef] [PubMed]
- Scudamore, K.A. Fate of Fusarium mycotoxins in the cereal industry: Recent UK studies. World Mycotoxin J. 2008, 1, 315–323. [Google Scholar] [CrossRef]
- Scudamore, K.A.; Patel, S. The fate of deoxynivalenol and fumonisins in wheat and maize during commercial breakfast cereal production. World Mycotoxin J. 2008, 1, 437–448. [Google Scholar] [CrossRef]
- Visconti, A.; Pascale, M. An overview on Fusarium mycotoxins in the durum wheat pasta production chain. Cereal Chem. 2010, 87, 21–27. [Google Scholar] [CrossRef]
- Kaushik, G. Effect of Processing on Mycotoxin Content in Grains. Crit. Rev. Food Sci. Nutr. 2015, 55, 1672–1683. [Google Scholar] [CrossRef] [PubMed]
- Hemery, Y.; Rouau, X.; Lullien-Pellerin, V.; Barron, C.; Abecassis, J. Dry processes to develop wheat fractions and products with enhanced nutritional quality. J. Cereal Sci. 2007, 46, 327–347. [Google Scholar] [CrossRef]
- Cheli, F.; Campagnoli, A.; Ventura, V.; Brera, C.; Berdini, C.; Palmaccio, E.; Dell’Orto, V. Effect of industrial processing on the distributions of deoxynivalenol, cadmium and lead in durum wheat milling fractions. LWT-Food Sci. Technol. 2010, 43, 1050–1057. [Google Scholar] [CrossRef]
- Edwards, S.G.; Dickin, E.T.; MacDonald, S.; Buttler, D.; Hazel, C.M.; Patel, S.; Scudamore, K. Distribution of Fusarium mycotoxins in UK wheat mill fractions. Food Addit. Contam. Part A 2011, 28, 1694–1704. [Google Scholar]
- Lancova, K.; Hajslova, J.; Kostelanska, M.; Kohoutkova, J.; Nedelnik, J.; Moravcova, H.; Vanova, M. Fate of trichothecene mycotoxins during the processing: Milling and baking. Food Addit. Contam. Part A 2008, 25, 650–659. [Google Scholar] [CrossRef] [PubMed]
- Neuhof, T.; Koch, M.; Rasenko, T.; Nehls, I. Occurrence of zearalenone in wheat kernels infected with Fusarium culmorum. World Mycotoxin J. 2008, 1, 429–435. [Google Scholar] [CrossRef]
- Pascale, M.; Haidukowski, M.; Lattanzio, V.M.T.; Silvestri, M.; Ranieri, R.; Visconti, A. Distribution of T-2 and HT-2 toxins in milling fractions of durum wheat. J. Food Protect. 2011, 74, 1700–1707. [Google Scholar] [CrossRef] [PubMed]
- Hazel, C.M.; Patel, S. Influence of processing on trichothecene levels. Toxicol. Lett. 2004, 153, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Dexter, J.E.; Wood, P.J. Recent applications of debranning of wheat before milling. Trends Food Sci. Technol. 1996, 7, 35–41. [Google Scholar] [CrossRef]
- Aureli, G.; D’Egidio, M.G. Efficacy of debranning on lowering of deoxynivalenol (DON) level in manufacturing processes of durum wheat. Tecnica Molit. 2007, 58, 729–733. [Google Scholar]
- Rios, G.; Pinson-Gadais, L.; Abecassis, J.; Zakhia-Rozis, N.; Lullien-Pellerin, V. Assessment of dehulling efficiency to reduce deoxynivalenol and Fusarium level in durum wheat grains. J. Cereal Sci. 2009, 49, 387–392. [Google Scholar] [CrossRef]
- Sovrani, V.; Blandino, M.; Scarpino, V.; Reyneri, A.; Coïsson, J.D.; Travaglia, F.; Locatelli, M.; Bordiga, M.; Montella, R.; Arlorio, M. Bioactive compound content, antioxidant activity, deoxynivalenol and heavy metal contamination of pearled wheat fractions. Food Chem. 2012, 135, 39–46. [Google Scholar] [CrossRef]
- Thammawong, M.; Okadome, H.; Shiina, T.; Nakagawa, H.; Nagashima, H.; Nakajima, T.; Kushiro, M. Distinct distribution of deoxynivalenol, nivalenol, and ergosterol in Fusarium-infected Japanese soft red winter wheat milling fractions. Mycopathologia 2011, 172, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Tibola, C.S.; Fernandes, J.M.C.; Guarienti, E.M.; Nicolau, M. Distribution of Fusarium mycotoxins in wheat milling process. Food Control 2015, 53, 91–95. [Google Scholar] [CrossRef]
- Thammawong, M.; Okabe, M.; Kawasaki, T.; Nakagawa, H.; Nagashima, H.; Okadome, H.; Nakajima, T.; Kushiro, M. Distribution of deoxynivalenol and nivalenol in milling fractions from Fusarium-infected Japanese wheat cultivars. J. Food Prot. 2010, 73, 1817–1823. [Google Scholar] [PubMed]
- Schwake-Anduschus, C.; Proske, M.; Sciurba, E.; Muenzing, K.; Koch, M.; Maul, R. Distribution of deoxynivalenol, zearalenone, and their respective modified analogues in milling fractions of naturally contaminated wheat grains. World Mycotoxin J. 2015, 8, 433–443. [Google Scholar] [CrossRef]
- Vaclavikova, M.; Malachova, A.; Veprikova, Z.; Dzuman, Z.; Zachariasova, M.; Hajslova, J. ‘Emerging’ mycotoxins in cereals processing chains: Changes of enniatins during beer and bread making. Food Chem. 2013, 136, 750–757. [Google Scholar] [CrossRef] [PubMed]
- Alexander, R.J. Corn dry milling: Processes, products, and applications. In Corn Chemistry and Technology; Watson, S.A., Ramstad, P.E., Eds.; American Association of Cereal Chemists: St. Paul, MN, USA, 1987; pp. 351–376. [Google Scholar]
- Brera, C.; Catano, C.; de Santis, B.; Debegnach, F.; de Giacomo, M.; Pannunzi, E.; Miraglia, M. Effect of Industrial Processing on the Distribution of Aflatoxins and Zearalenone in Corn-Milling Fractions. J. Agric. Food Chem. 2006, 54, 5014–5019. [Google Scholar] [CrossRef] [PubMed]
- Fandohan, R.; Ahouansou, P.; Houssou, K.; Hell, H.; Marasas, W.F.O.; Wingfield, M.J. Impact of mechanical shelling and dehulling on Fusarium infection and fumonisin contamination in maize. Food Addit. Contam. 2006, 23, 415–421. [Google Scholar]
- Pietri, M.; Zanetti, A.; Bertuzzi, T. Distribution of aflatoxins and fumonisins in dry-milled maize fractions. Food Addit. Contam. Part A 2009, 26, 372–380. [Google Scholar] [CrossRef] [PubMed]
- Brera, C.; Debegnach, F.; Grossi, S.; Miraglia, M. Effect of processing on the distribution of fumonisins B1 in corn milling fractions derived from an industrial plant. J. Food Prot. 2004, 67, 1261–1266. [Google Scholar] [PubMed]
- Castells, M.; Marín, S.; Sanchis, V.; Ramos, A.J. Distribution of fumonisins and aflatoxins in corn fractions during industrial cornflake processing. Int. J. Food Microbiol. 2008, 123, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Park, D.L. Effect of processing on aflatoxin. Adv. Exp. Med. Biol. 2002, 504, 173–179. [Google Scholar] [PubMed]
- Scudamore, K.A.; Banks, J.; MacDonald, S.J. Fate of ochratoxin A in the processing of whole wheat grains during milling and bread production. Food Addit. Contam. 2003, 20, 1153–1163. [Google Scholar] [CrossRef] [PubMed]
- Schollenberger, M.; Müller, H.M.; Rüfle, M.; Suchy, S.; Drochner, W. Redistribution of 16 Fusarium Toxins during Commercial Dry Milling of Maize. Cereal Chem. 2008, 85, 557–560. [Google Scholar] [CrossRef]
- Vanara, F.; Reyneri, A.; Blandino, M. Fate of fumonisin B1 in the processing of whole maize kernels during dry-milling. Food Control 2009, 20, 235–238. [Google Scholar] [CrossRef]
- Burger, H.-M.; Shephard, G.S.; Louw, W.; Rheeder, J.P.; Gelderblom, W.C.A. The mycotoxin distribution in maize milling fractions under experimental conditions. Int. J. Food Microbiol. 2013, 165, 57–64. [Google Scholar]
- Bothast, R.J.; Schlicher, M.A. Biotechnological processes for conversion of corn into ethanol. Appl. Microbiol. Biotechnol. 2005, 67, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Burton, E.J.; Scholey, D.V.; Williams, P.E.V. Use of cereal crops for food and fuel—Characterization of a novel bioethanol coproduct for use in meat poultry diets. Food Energy Secur. 2013, 2, 197–206. [Google Scholar] [CrossRef]
- Schingoethe, D.J.; Kalscheur, K.F.; Hippen, A.R.; Garcia, A.D. The use of distillers products in dairy cattle diets. J. Dairy Sci. 2009, 92, 5802–5813. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Munkvold, G.P. Mycotoxins in Ethanol Co-products: Modeling Economic Impacts on the Livestock Industry and Management Strategies. J. Agric. Food Chem. 2008, 56, 3900–3911. [Google Scholar] [CrossRef] [PubMed]
- OECD-FAO Agricultural Outlook 2011–2020. OECD Publishing. 2011. Available online: http://www.oecd-ilibrary.org/agriculture-and-food/oecd-fao-agricultural-outlook-2011_agr_outlook-2011-en (accessed on 13 November 2015).
- Liu, K. Chemical Composition of Distillers Grains, a Review. J. Agric. Food Chem. 2011, 59, 1508–1526. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Caupert, J.; Imerman, P.M.; Richard, J.L.; Shurson, G.C. The Occurrence and Concentration of Mycotoxins in U.S. Distillers Dried Grains with Solubles. J. Agric. Food Chem. 2009, 57, 9828–9837. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, I.; Chin, L.J. A comprehensive survey on the occurrence of mycotoxins in maize dried distillers’grain and solubles sourced worldwide. World Mycotoxin J. 2012, 5, 83–88. [Google Scholar] [CrossRef]
- Khatibi, P.A.; McMaster, N.J.; Musser, R.; Schmale, D.G., III. Survey of Mycotoxins in Corn Distillers’ Dried Grains with Solubles from Seventy-Eight Ethanol Plants in Twelve States in the U.S. in 2011. Toxins 2014, 6, 1155–1168. [Google Scholar] [CrossRef] [PubMed]
- Bowers, E.L.; Munkvold, G.P. Fumonisins in Conventional and Transgenic, Insect-Resistant Maize Intended for Fuel Ethanol Production: Implications for Fermentation Efficiency and DDGS Co-Product Quality. Toxins 2014, 6, 2804–2825. [Google Scholar] [CrossRef] [PubMed]
- Hanschmann, G.; Krieg, D. The fate of fusarium toxins in the course of the synthesis of bioethanol from contaminated grain. Mycotoxin Res. 2006, 22, 174–177. [Google Scholar] [CrossRef] [PubMed]
- Schaafsma, A.W.; Limay-Rios, V.; Paul, D.E.; Miller, J.D. Mycotoxins in fuel ethanol co-products derived from maize: A mass balance for deoxynivalenol. J. Sci. Food Agric. 2009, 89, 1574–1580. [Google Scholar] [CrossRef]
- Bartkiene, E.; Bakutis, B.; Baliukoniene, V.; Juodeikiene, G.; Basinskiene, L.; Vidmantiene, D. Effect of xylanlytic enzymes on the efficiency of bioethanol production from Fusarium contaminated grains. Vet. Zootech. 2010, 49, 17–25. [Google Scholar]
- Klosowski, G.; Mikulski, D.; Grajewski, J.; Blajet-Kosicka, A. The influence of raw material contamination with mycotoxins on alcoholic fermentation indicators. Bioresour. Technol. 2010, 101, 3147–3152. [Google Scholar] [CrossRef] [PubMed]
- Murthy, G.S.; Townsend, D.E.; Meerdink, G.L.; Bargren, G.L.; Tumbleson, M.E.; Singh, V. Effect of aflatoxin B1 on the dry grind ethanol process. Cereal Chem. 2005, 82, 302–304. [Google Scholar] [CrossRef]
- Prettl, Z.S.; Lepossa, A.; Tóth, É.; Kelemen-Horváth, I.; Németh, Á.Sz.; Nagyprettl, E. Effects and changes of zearalenone and fumonisin contamination in corn-based bioethanol process. Hung. J. Ind. Chem. 2011, 39, 427–431. [Google Scholar]
- Siegel, D.; Babuscio, T. Mycotoxin management in the European cereal trading sector. Food Control 2011, 22, 1145–1153. [Google Scholar] [CrossRef]
- Sosa, M.A.; Chovau, S.; Van der Bruggen, B.; Espinosa, J. Ethanol Production from Corn Contaminated with Fumonisins: A Preliminary Economic Analysis Including Novel Processing Alternatives. Ind. Eng. Chem. Res. 2013, 52, 7504–7513. [Google Scholar] [CrossRef]
- Beer Production Worldwide from 1998 to 2014. Available online: http://www.statista.com/statistics/270275/worldwide-beer-production/ (accessed on 13 November 2015).
- The Brewers Association. Available online: https://brewersassociation.org (accessed on 13 November 2015).
- The Brewers of Europe. Available online: http://www.brewersofeurope.org/site/index.php (accessed on 13 November 2015).
- Mussatto, S.I.; Dragone, G.; Roberto, I.C. Brewers’ spent grain: Generation, characteristics and potential applications. J. Cereal Sci. 2006, 43, 1–14. [Google Scholar] [CrossRef]
- Schwarz, P.B.; Schwarz, J.G.; Zhou, A.; Prom, L.K.; Steffenson, B.J. Effect of Fusarium graminearum and F-poae infection on barley and malt quality. Monats Brws. 2001, 54, 55–63. [Google Scholar]
- Foroud, N.A.; Eudes, F. Review—Trichothecenes in cereal grains. Int. J. Mol. Sci. 2009, 10, 147–173. [Google Scholar] [CrossRef]
- Maenetje, P.W.; Dutton, M.F. The incidence of fungi and mycotoxins in South African barley and barley products. J Environ. Sci. B 2007, 42, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Malachova, A.; Cerkal, R.; Ehrenbergerova, J.; Dzuman, Z.; Vaculova, K.; Hajslova, J. Fusarium mycotoxins in various barley cultivars and their transfer into malt. J. Sci. Food Agric. 2010, 90, 2495–2505. [Google Scholar] [CrossRef] [PubMed]
- Lattanzio, B.C.; Terzi, V.; Ghizzoni, R.; McCormick, S.P.; Pascale, M. Study of the natural occurrence of T-2 and HT-2 toxins and their glucosyl derivatives from field barley to malt by high-resolution Orbitrap mass spectrometry. Food Addit. Contam. A 2015. [Google Scholar] [CrossRef] [PubMed]
- Varga, E.; Malachova, A.; Schwartz, H.; Krska, R.; Berthiller, F. Survey of deoxynivalenol and its conjugates deoxynivalenol-3-glucoside and 3-acetyldeoxynivalenol in 374 beer samples. Food Addit. Contam. A 2013, 30, 137–146. [Google Scholar] [CrossRef] [PubMed]
- Terzi, V.; Tumino, G.; Stanca, A.M.; Morcia, C. Reducing the incidence of cereal head infection and mycotoxins in small grain cereal species. J. Cereal Sci. 2013, 59, 284–293. [Google Scholar] [CrossRef]
- Kostelanska, M.; Hajslova, J.; Zachariasova, M.; Malachova, A.; Kalachova, K.; Poustka, J.; Fiala, J.; Scott, P.M.; Berthiller, F.; Krska, R. Occurrence of Deoxynivalenol and Its Major Conjugate, Deoxynivalenol-3-Glucoside, in Beer and Some Brewing Intermediates. J. Agric. Food Chem. 2009, 57, 3187–3194. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, P.B.; Casper, H.H.; Beattie, S. Fate and Development of Naturally Occurring Fusarium Mycotoxins During Malting and Brewing. J. Am. Soc. Brew. Chem. 1995, 53, 121–126. [Google Scholar]
- Ioos, R.; Belhadj, A.; Menez, M.; Faure, A. The effect of fungicides on Fusarium spp. and Microdochium nivale and their associated trichothecene mycotoxins in French naturally-infected cereals grains. Crop Prot. 2005, 24, 894–902. [Google Scholar] [CrossRef]
- Laitila, A.; Alakomi, H.L.; Raaska, L.; Mattila-Sandholm, T.; Haikara, A. Antifungal activities of two Lactobacillus plantarum strains against Fusarium moulds in vitro and in malting of barley. J. Appl. Microbiol. 2002, 93, 566–576. [Google Scholar] [CrossRef] [PubMed]
- Rouse, S.; van Sinderen, D. Bioprotective Potential of Lactic Acid Bacteria in Malting and Brewing. J. Food Prot. 2008, 71, 1724–1733. [Google Scholar] [PubMed]
- Cavaglieri, L.R.; Keller, K.M.; Pereyra, C.M.; González Pereyra, M.L.; Alonso, V.A.; Rojo, F.G.; Dalcero, A.M.; Rosa, C.A.R. Fungi and natural incidence of selected mycotoxins in barley rootlets. J. Stored Prod. Res. 2009, 45, 147–150. [Google Scholar] [CrossRef]
- González Pereyra, M.L.; Rosa, C.A.R.; Dalcero, A.M.; Cavaglieri, L.R. Mycobiota and mycotoxins in malted barley and brewer’s spent grain from Argentinean breweries. Lett. Appl. Microbiol. 2011, 53, 649–655. [Google Scholar] [CrossRef] [PubMed]
- Cheli, F.; Campagnoli, A.; Pinotti, L.; Fusi, E.; Dell’Orto, V. Sampling feed for mycotoxins: Acquiring knowledge from food. It. J. Anim. Sci. 2009, 8, 5–22. [Google Scholar] [CrossRef]
- European Commission. Commission Regulation (EC) No 401/2006 of 23 February 2006 laying down the methods of sampling and analysis for the official control of the levels of mycotoxins in foodstuffs. Off. J. L 2006, 70, 12–34. [Google Scholar]
- Berthiller, F.; Burdaspal, P.A.; Crews, C.; Iha, M.H.; Krska, R.; Lattanzio, W.M.T.; MacDonald, S.; Malone, R.J.; Maragos, C.; Solfrizzo, M.; et al. Developments in mycotoxin analysis: An update for 2012–2013. World Mycotoxin J. 2014, 7, 3–33. [Google Scholar] [CrossRef]
- Logrieco, A.; Arrigan, D.W.M.; Brengel-Pesce, K.; Siciliano, P.; Tothill, I. DNA arrays, electronic noses and tongues, biosensors and receptors for rapid detection of toxigenic fungi and mycotoxins: A review. Food Addit. Contam. 2005, 22, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Krska, R.; Schubert-Ullrich, P.; Molinelli, A.; Sulyok, M.; MacDonald, S.; Crews, C. Mycotoxin analysis: An update. Food Addit. Contam. Part A Chem. Anal. Control Expo Risk Assess. 2008, 25, 152–163. [Google Scholar] [CrossRef] [PubMed]
- Rahmani, A.; Jinap, S.; Soleimany, F. Qualitative and Quantitative Analysis of Mycotoxins. Compr. Rev. Food Sci. Food Saf. 2009, 8, 202–251. [Google Scholar] [CrossRef]
- Shephard, G.S.; Berthiller, F.; Burdaspal, P.A.; Crews, C.; Jonker, M.A.; Krska, R.; Lattanzio, V.M.T.; MacDonald, S.; Malone, R.J.; Maragos, C.; et al. Developments in mycotoxin analysis: An update for 2011–2012. World Mycotoxin J. 2013, 6, 3–30. [Google Scholar] [CrossRef]
- Turner, N.W.; Subrahmanyam, S.; Piletsky, S.A. Analytical methods for determination of mycotoxins: A review. Anal. Chim. Acta 2009, 632, 168–180. [Google Scholar] [CrossRef] [PubMed]
- Busman, M.; Poling, S.M.; Maragos, C.M. Observation of T-2 Toxin and HT-2 Toxin glucosides from Fusarium sporotrichioides by liquid chromatography coupled to tandemmass spectrometry (LC-MS/MS). Toxins 2011, 3, 1554–1568. [Google Scholar] [CrossRef] [PubMed]
- Maragos, C.M.; Busman, M. Rapid and advanced tools for mycotoxin analysis: A review. Food Addit. Contam. A 2010, 27, 688–700. [Google Scholar] [CrossRef] [PubMed]
- Campagnoli, A.; Dell’Orto, V. Potential Application of Electronic Olfaction Systems in Feedstuffs Analysis and Animal Nutrition. Sensors 2013, 13, 14611–14632. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.Z.; Goto, T. Near- and mid-infrared spectroscopy as efficient tools for detection of fungal and mycotoxin contamination in agricultural commodities. World Mycotoxin J. 2014, 7, 507–515. [Google Scholar] [CrossRef]
- Lippolis, V.; Maragos, C. Fluorescence polarisation immunoassays for rapid, accurate and sensitive determination of mycotoxins. World Mycotoxin J. 2014, 7, 479–490. [Google Scholar] [CrossRef]
- Meneely, J.P.; Elliott, C.T. Rapid surface plasmon resonance immunoassays for the determination of mycotoxins in cereals and cereal-based food products. World Mycotoxin J. 2014, 7, 491–505. [Google Scholar] [CrossRef]
- Reichel, M.; Staiger, S.; Biselli, S. Analysis of Fusarium toxins in grain via dust: A promising field of application for rapid test systems. World Mycotoxin J. 2014, 7, 465–477. [Google Scholar] [CrossRef]
- Sanders, M.; Landschoot, S.; Audenaert, K.; Haesaert, G.; Eeckhout, M.; de Saeger, S. Deoxynivalenol content in wheat dust versus wheat grain: A comparative study. World Mycotoxin J. 2014, 7, 285–290. [Google Scholar] [CrossRef]
- Tittlemier, S.A.; Sobering, D.; Bowler, K.; Zirdum, T.; Gaba, D.; Chan, J.M.; Roscoe, M.; Blagden, R.; Campbell, L. By-products of grain cleaning: An opportunity for rapid sampling and screening of wheat for mycotoxins. World Mycotoxin J. 2015, 8, 45–53. [Google Scholar] [CrossRef]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pinotti, L.; Ottoboni, M.; Giromini, C.; Dell’Orto, V.; Cheli, F. Mycotoxin Contamination in the EU Feed Supply Chain: A Focus on Cereal Byproducts. Toxins 2016, 8, 45. https://doi.org/10.3390/toxins8020045
Pinotti L, Ottoboni M, Giromini C, Dell’Orto V, Cheli F. Mycotoxin Contamination in the EU Feed Supply Chain: A Focus on Cereal Byproducts. Toxins. 2016; 8(2):45. https://doi.org/10.3390/toxins8020045
Chicago/Turabian StylePinotti, Luciano, Matteo Ottoboni, Carlotta Giromini, Vittorio Dell’Orto, and Federica Cheli. 2016. "Mycotoxin Contamination in the EU Feed Supply Chain: A Focus on Cereal Byproducts" Toxins 8, no. 2: 45. https://doi.org/10.3390/toxins8020045
APA StylePinotti, L., Ottoboni, M., Giromini, C., Dell’Orto, V., & Cheli, F. (2016). Mycotoxin Contamination in the EU Feed Supply Chain: A Focus on Cereal Byproducts. Toxins, 8(2), 45. https://doi.org/10.3390/toxins8020045







