Worldwide Mycotoxins Exposure in Pig and Poultry Feed Formulations
Abstract
:1. Introduction
2. Pig and Poultry Diets
2.1. Nutrient Requirements and Feed Formulation
2.2. Energy Sources
2.3. Protein Sources
2.4. Alternative Ingredients
3. Mycotoxin Contamination
3.1. Mycotoxin Analysis
3.2. Factors Affecting Mycotoxin Contamination
3.3. Contamination of Raw Materials by Mycotoxins
3.3.1. Maize, Wheat and Soybean Meal
3.3.2. Other Raw Materials
4. Pig and Poultry Exposure
4.1. Impact of Age and Production
4.2. Impact of Feed Substitution
Conflicts of Interest
References
- Bennett, J.W.; Klich, M. Mycotoxins. Clin. Microbiol. Rev. 2003, 16, 497–516. [Google Scholar] [CrossRef] [PubMed]
- Commission Directive 2003/100/EC. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32003L0100&rid=2 (accessed on 11 August 2016).
- Commission Recommendation of 17 August 2006 on the Presence of Deoxynivalenol, Zearalenone, Ochratoxin A, T-2 and HT-2 and Fumonisins in Products Intended for Animal Feeding. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32006H0576&from=EN (accessed on 11 August 2016).
- Commission Recommendation of 27 March 2013 on the Presence of T-2 and HT-2 Toxin in Cereals and Cereal Products. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32013H0165&from=EN (accessed on 11 August 2016).
- FDA Mycotoxin Regulatory Guidance, August 2011. Available online: https://www.ngfa.org/wp-content/uploads/NGFAComplianceGuide-FDARegulatoryGuidanceforMycotoxins8-2011.pdf (accessed on 11 August 2016).
- Government of Canada, C.F.I.A. RG-8 Regulatory Guidance: Contaminants in Feed (formerly RG-1, Chapter 7). Available online: http://www.inspection.gc.ca/animals/feeds/regulatory-guidance/rg-8/eng/1347383943203/1347384015909?chap=1 (accessed on 11 August 2016).
- Feed Section- Mycotoxins in Feed Grains. Available online: http://www.oisc.purdue.edu/feed/mycotoxins.html (accessed on 11 August 2016).
- Nutrient Requirements of Swine: Eleventh Revised Edition; National Academies Press: Washington, DC, USA, 2012; Available online: https://www.nap.edu/read/13298/chapter/1 (accessed on 23 November 2016).
- Nutrient Requirements of Poultry, Ninth Revised Edition; National Academy Press: Washington, DC, USA, 1994; Available online: http://www.lamolina.edu.pe/facultad/zootecnia/biblioteca2012/NRC%20Poultry%201994[1].pdf (accessed on 23 November 2016).
- Applegate, T.J.; Angel, R. Nutrient requirements of poultry publication: History and need for an update. J. Appl. Poult. Res. 2014. [Google Scholar] [CrossRef]
- Kyntäjä, S.; Partanen, K.; Siljander-Rasi, H.; Jalava, T. Tables of Composition and Nutritional Values of Organically Produced Feed Materials for Pigs and Poultry. Available online: http://www.mtt.fi/mttraportti/pdf/mttraportti164.pdf.
- Ravindran, V. Poultry Feed Availability and Nutrition in Developing Countries: Main Ingredients Used in Poultry Feed Formulations. In Poultry Development Review; FAO: Rome, Italy, 2013; pp. 1–3. [Google Scholar]
- Lammers, P.J.; Stender, D.R.; Honeyman, M.S. Feed Budgets. Available online: http://www.ipic.iastate.edu/publications/840.FeedBudgets.pdf (accessed on 25 August 2016).
- Bryden, W.L.; Li, X. Amino acid digestibility and poultry feed formulation: Expression, limitations and application. Rev. Bras. Zootec. 2010, 39, 279–287. [Google Scholar] [CrossRef]
- Rostagno, H.S.; Pupa, J.M.R.; Pack, M. Diet Formulation for Broilers Based on Total versus Digestible Amino Acids. J. Appl. Poult. Res. 1995, 4, 293–299. [Google Scholar] [CrossRef]
- Xue, P.C.; Ragland, D.; Adeola, O. Determination of additivity of apparent and standardized ileal digestibility of amino acids in diets containing multiple protein sources fed to growing pigs. J. Anim. Sci. 2014, 92, 3937–3944. [Google Scholar] [CrossRef] [PubMed]
- Stein, H.H.; Sève, B.; Fuller, M.F.; Moughan, P.J.; de Lange, C.F.M. Committee on Terminology to Report AA Bioavailability and Digestibility Invited review: Amino acid bioavailability and digestibility in pig feed ingredients: Terminology and application. J. Anim. Sci. 2007, 85, 172–180. [Google Scholar] [CrossRef] [PubMed]
- Vieira, S.L.; Stefanello, C.; Sorbara, J.O.B. Formulating poultry diets based on their indigestible components. Poult. Sci. 2014, 93, 2411–2416. [Google Scholar] [CrossRef] [PubMed]
- Slominski, B.A. Recent advances in research on enzymes for poultry diets. Poult. Sci. 2011, 90, 2013–2023. [Google Scholar] [CrossRef] [PubMed]
- Bedford, M.R.; Schulze, H. Exogenous enzymes for pigs and poultry. Nutr. Res. Rev. 1998, 11, 91–114. [Google Scholar] [CrossRef] [PubMed]
- Swiatkiewicz, S.; Swiatkiewicz, M.; Arczewska-Wlosek, A.; Jozefiak, D. Efficacy of feed enzymes in pig and poultry diets containing distillers dried grains with solubles: A review. J. Anim. Physiol. Anim. Nutr. 2016, 100, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Nahm, K.H. Feed formulations to reduce N excretion and ammonia emission from poultry manure. Bioresour. Technol. 2007, 98, 2282–2300. [Google Scholar] [CrossRef] [PubMed]
- Bridges, T.C.; Turner, L.W.; Cromwell, G.L.; Pierce, J.L. Modeling the Effects of Diet Formulation on Nitrogen and Phosphorus Excretion in Swine Waste. Appl. Eng. Agric. 1995, 11, 731–739. [Google Scholar] [CrossRef]
- Murugesan, G.R.; Ledoux, D.R.; Naehrer, K.; Berthiller, F.; Applegate, T.J.; Grenier, B.; Phillips, T.D.; Schatzmayr, G. Prevalence and effects of mycotoxins on poultry health and performance, and recent development in mycotoxin counteracting strategies. Poult. Sci. 2015, 94, 1298–1315. [Google Scholar] [CrossRef] [PubMed]
- Devreese, M.; De Backer, P.; Croubels, S. Different methods to counteract mycotoxin production and its impact on animal health. Vlaams Diergeneeskd. Tijdschr. 2013, 82, 181–190. [Google Scholar]
- Arroyo, J.; Dubois, J.P.; Lavigne, F.; Brachet, M.; Fortun-Lamothe, L. Effects of replacing corn with sorghum on the performance of overfed mule ducks. Poult. Sci. 2016, 95, 1304–1311. [Google Scholar] [CrossRef] [PubMed]
- Arroyo, J.; Auvergne, A.; Dubois, J.P.; Lavigne, F.; Bijja, M.; Bannelier, C.; Manse, H.; Fortun-Lamothe, L. Effects of substituting yellow corn for sorghum in geese diets on magret and foie gras quality. Poult. Sci. 2013, 92, 2448–2456. [Google Scholar] [CrossRef] [PubMed]
- Ravindran, V. Main ingredients used in poultry feed formulations. Available online: http://www.fao.org/3/a-al705e.pdf (accessed on 23 November 2016).
- Agricultural Production Statistics by Country. Available online: http://www.indexmundi.com/agriculture/ (accessed on 11 August 2016).
- Selle, P.H.; Cadogan, D.J.; Li, X.; Bryden, W.L. Implications of sorghum in broiler chicken nutrition. Anim. Feed Sci. Technol. 2010, 156, 57–74. [Google Scholar] [CrossRef]
- Batonon-Alavo, D.I.; Umar Faruk, M.; Lescoat, P.; Weber, G.M.; Bastianelli, D. Inclusion of sorghum, millet and cottonseed meal in broiler diets: A meta-analysis of effects on performance. Animal 2015, 9, 1120–1130. [Google Scholar] [CrossRef] [PubMed]
- Tokach, M.; Goodband, B.; DeRouchey, J. Sorghum in Swine Production. Feeding Guide. Available online: https://www.grains.org/sites/default/files/technical-publications/pdfs/Sorghum_Swine_Feeding_Handbook.pdf (accessed on 26 August 2016).
- Kumaraveland, V.; Natarajan, A. Replacement of maize with pearl millet in broiler chicken diet-A review. 2016, 3, 2197–2204. [Google Scholar]
- Lawrence, B.V.; Adeola, O.; Rogler, J.C. Nutrient digestibility and growth performance of pigs fed pearl millet as a replacement for corn. J. Anim. Sci. 1995, 73, 2026–2032. [Google Scholar] [CrossRef] [PubMed]
- Murry, A.C.; Hatfield, E.E.; Larsen, L. Performance and digestibility of pearl millet in diets for nursery pigs. Prof. Anim. Sci. 1998, 14, 147–151. [Google Scholar] [CrossRef]
- Sullivan, T.W.; Gleaves, E.W. G77-386 Wheat in Poultry Rations. Available online: http://digitalcommons.unl.edu/cgi/viewcontent.cgi?article=2290&context=extensionhist (accessed on 26 August 2016).
- Summers, J. The Feeding of Whole Wheat to Broilers. Available online: http://www.poultryindustrycouncil.ca/pdfs/factsheets/fs_79.pdf (accessed on 28 August 2016).
- Jacob, J. Feeding Wheat to Poultry—EXtension. Available online: http://articles.extension.org/pages/68430/feeding-wheat-to-poultry (accessed on 26 August 2016).
- Stein, H.H.; Pahm, A.A.; Roth, J.A. Feeding Wheat to Pigs. Available online: http://nutrition.ansci.illinois.edu/sites/default/files/SwineFocus002.pdf (accessed on 19 November 2016).
- Sullivan, Z.; Honeyman, M.; Gibson, L.; Nelson, M. Feeding Small Grains to Swine. Available online: https://store.extension.iastate.edu/Product/Feeding-Small-Grains-to-Swine-PDF (accessed on 22 November 2016).
- Harrold, R.L. Feeding Barley to Swine. Available online: http://www.thepigsite.com/articles/585/feeding-barley-to-swine/ (accessed on 26 August 2016).
- Jacob, J.P.; Pescatore, A.J. Using barley in poultry diets—A review. J. Appl. Poult. Res. 2012, 21, 915–940. [Google Scholar] [CrossRef]
- Myer Oats in Swine Diets. Available online: http://www.thepigsite.com/articles/2571/oats-in-swine-diets/ (accessed on 26 August 2016).
- Jacob, J. Feeding Oats to Poultry—eXtension. Available online: http://articles.extension.org/pages/70159/including-oats-in-organic-poultry-diets (accessed on 26 August 2016).
- Cave, N.A.; Hamilton, R.M.G.; Burrows, V.D. Evaluation of Naked Oats (Avena nuda ) as a Feedingstuff for laying hens. Can. J. Anim. Sci. 1989, 69, 789–799. [Google Scholar] [CrossRef]
- Józefiak, D.; Rutkowski, A.; Jensen, B.B.; Engberg, R.M. The effect of beta-glucanase supplementation of barley- and oat-based diets on growth performance and fermentation in broiler chicken gastrointestinal tract. Br. Poult. Sci. 2006, 47, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Asyifah, M.N.; Abd-Aziz, S.; Phang, L.Y.; Azlian, M.N. Brown rice as a potential feedstuff for poultry. J. Appl. Poult. Res. 2012, 21, 103–110. [Google Scholar] [CrossRef]
- Vicente, B.; Valencia, D.G.; Pérez-Serrano, M.; Lázaro, R.; Mateos, G.G. The effects of feeding rice in substitution of corn and the degree of starch gelatinization of rice on the digestibility of dietary components and productive performance of young pigs. J. Anim. Sci. 2008, 86, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Hazzledine, M.; Pine, A.; Mackinson, I.; Ratcliffe, J.; Salmon, L.; Levels, R.; Staffs, W. Estimating Displacement Ratios of Wheat DDGS in Animal Feed Rations in Great Britain. Available online: http://www.theicct.org/sites/default/files/publications/ICCT_GB-DDGS-displacement_Nov2011.pdf (accessed on 23 November 2016).
- Stein, H.H.; Shurson, G.C. Board-invited review: The use and application of distillers dried grains with solubles in swine diets. J. Anim. Sci. 2009, 87, 1292–1303. [Google Scholar] [CrossRef] [PubMed]
- Baião, N.C.; Lara, L.J.C. Oil and fat in broiler nutrition. Rev. Bras. Ciênc. Avícola 2005, 7, 129–141. [Google Scholar] [CrossRef]
- Wu, G. Dietary requirements of synthesizable amino acids by animals: A paradigm shift in protein nutrition. J. Anim. Sci. Biotechnol. 2014, 5, 34. [Google Scholar] [CrossRef] [PubMed]
- Hulshof, T.G.; van der Poel, A.F.B.; Hendriks, W.H.; Bikker, P. Processing of soybean meal and 00-rapeseed meal reduces protein digestibility and pig growth performance but does not affect nitrogen solubilization along the small intestine. J. Anim. Sci. 2016, 94, 2403–2414. [Google Scholar] [CrossRef] [PubMed]
- Beski, S.S.M.; Swick, R.A.; Iji, P.A. Specialized protein products in broiler chicken nutrition: A review. Anim. Nutr. 2015, 1, 47–53. [Google Scholar] [CrossRef]
- Lomascolo, A.; Uzan-Boukhris, E.; Sigoillot, J.-C.; Fine, F. Rapeseed and sunflower meal: A review on biotechnology status and challenges. Appl. Microbiol. Biotechnol. 2012, 95, 1105–1114. [Google Scholar] [CrossRef] [PubMed]
- Kaminska, B.Z. Substitution of soyabean meal with “00” rapeseed meal or its high-protein fraction in the nutrition of hens laying brown-shelled eggs. J. Anim. Feed Sci. 2003, 12, 111–119. [Google Scholar]
- Kamin’ska, B.Z.; Brzóska, F.; Skraba, B. High-protein fraction of 00 type rapeseed meal in broiler nutrition. J. Anim. Feed Sci. 2000, 1, 123–136. [Google Scholar]
- Choi, H.B.; Jeong, J.H.; Kim, D.H.; Lee, Y.; Kwon, H.; Kim, Y.Y. Influence of Rapeseed Meal on Growth Performance, Blood Profiles, Nutrient Digestibility and Economic Benefit of Growing-finishing Pigs. Asian-Australas. J. Anim. Sci. 2015, 28, 1345–1353. [Google Scholar] [CrossRef] [PubMed]
- Mejicanos, G.; Sanjayan, N.; Kim, I.H.; Nyachoti, C.M. Recent advances in canola meal utilization in swine nutrition. J. Anim. Sci. Technol. 2016, 58, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Khajali, F.; Slominski, B.A. Factors that affect the nutritive value of canola meal for poultry. Poult. Sci. 2012, 91, 2564–2575. [Google Scholar] [CrossRef] [PubMed]
- Wickramasuriya, S.S.; Yi, Y.-J.; Yoo, J.; Kang, N.K.; Heo, J.M. A review of canola meal as an alternative feed ingredient for ducks. J. Anim. Sci. Technol. 2015, 57, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Jankowski, J.; Lecewicz, A.; Zdunczyk, Z.; Juskiewicz, J.; Slominski, B.A. The effect of partial replacement of soyabean meal with sunflower meal on ileal adaptation, nutrient utilisation and growth performance of young turkeys. Br. Poult. Sci. 2011, 52, 456–465. [Google Scholar] [CrossRef] [PubMed]
- Das, S.K.; Biswas, A.; Neema, R.P.; Maity, B. Effect of soybean meal substitution by different concentrations of sunflower meal on egg quality traits of white and coloured dwarf dam lines. Br. Poult. Sci. 2010, 51, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Rama Rao, S.V.; Raju, M.V.L.N.; Panda, A.K.; Reddy, M.R. Sunflower seed meal as a substitute for soybean meal in commercial broiler chicken diets. Br. Poult. Sci. 2006, 47, 592–598. [Google Scholar] [CrossRef] [PubMed]
- Laudadio, V.; Ceci, E.; Lastella, N.M.B.; Tufarelli, V. Effect of feeding low-fiber fraction of air-classified sunflower (Helianthus annus L.) meal on laying hen productive performance and egg yolk cholesterol. Poult. Sci. 2014, 93, 2864–2869. [Google Scholar] [CrossRef] [PubMed]
- Brenes, A.; Centeno, C.; Viveros, A.; Arija, I. Effect of enzyme addition on the nutritive value of high oleic acid sunflower seeds in chicken diets. Poult. Sci. 2008, 87, 2300–2310. [Google Scholar] [CrossRef] [PubMed]
- Fafiolu, A.O.; Oduguwa, O.O.; Jegede, A.V.; Tukura, C.C.; Olarotimi, I.D.; Teniola, A.A.; Alabi, J.O. Assessment of enzyme supplementation on growth performance and apparent nutrient digestibility in diets containing undecorticated sunflower seed meal in layer chicks. Poult. Sci. 2015, 94, 1917–1922. [Google Scholar] [CrossRef] [PubMed]
- Bilal, M.; Mirza, M.A.; Kaleem, M.; Saeed, M.; Reyad-Ul-Ferdous, M.; Abd El-Hack, M.E. Significant effect of NSP-ase enzyme supplementation in sunflower meal-based diet on the growth and nutrient digestibility in broilers. J. Anim. Physiol. Anim. Nutr. 2016. [Google Scholar] [CrossRef] [PubMed]
- Nørgaard, J.V.; Fernández, J.A.; Jørgensen, H. Ileal digestibility of sunflower meal, pea, rapeseed cake, and lupine in pigs. J. Anim. Sci. 2012, 90, 203–205. [Google Scholar] [CrossRef] [PubMed]
- González-Vega, J.C.; Stein, H.H. Amino acid digestibility in canola, cottonseed, and sunflower products fed to finishing pigs. J. Anim. Sci. 2012, 90, 4391–4400. [Google Scholar] [CrossRef] [PubMed]
- Almeida, F.N.; Htoo, J.K.; Thomson, J.; Stein, H.H. Digestibility by growing pigs of amino acids in heat-damaged sunflower meal and cottonseed meal. J. Anim. Sci. 2014, 92, 585–593. [Google Scholar] [CrossRef] [PubMed]
- Adeola, O.; Kong, C. Energy value of distillers dried grains with solubles and oilseed meals for pigs. J. Anim. Sci. 2014, 92, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Nagalakshmi, D.; Rao, S.V.R.; Panda, A.K.; Sastry, V.R. Cottonseed meal in poultry diets: A review. J. Poult. Sci. 2007, 44, 119–134. [Google Scholar] [CrossRef]
- Henry, M.H.; Pesti, G.M.; Bakalli, R.; Lee, J.; Toledo, R.T.; Eitenmiller, R.R.; Phillips, R.D. The performance of broiler chicks fed diets containing extruded cottonseed meal supplemented with lysine. Poult. Sci. 2001, 80, 762–768. [Google Scholar] [CrossRef] [PubMed]
- Fombad, R.B.; Bryant, M.J. An evaluation of the use of cottonseed cake in the diet of growing pigs. Trop. Anim. Health Prod. 2004, 36, 295–305. [Google Scholar] [CrossRef] [PubMed]
- Fombad, R.B.; Bryant, M.J. Effects of cottonseed cake-based diets supplemented with blood meal, alone or with lysine, on the growth of pigs. Trop. Anim. Health Prod. 2004, 36, 385–395. [Google Scholar] [CrossRef] [PubMed]
- Alternative Ingredients to Be Used in Poultry Feed. Available online: http://www.allaboutfeed.net/New-Proteins/Articles/2013/4/Alternative-ingredients-to-be-used-in-poultry-feed-1242465W/ (accessed on 4 August 2016).
- Feed Resources for Smallholder Poultry in Nigeria. Available online: http://www.fao.org/docrep/V4440T/v4440T0a.htm (accessed on 4 August 2016).
- Carter, N.A.; Dewey, C.E.; Thomas, L.F.; Lukuyu, B.; Grace, D.; de Lange, C. Nutrient requirements and low-cost balanced diets, based on seasonally available local feedstuffs, for local pigs on smallholder farms in Western Kenya. Trop. Anim. Health Prod. 2016, 48, 337–347. [Google Scholar] [CrossRef] [PubMed]
- Krska, R.; Schubert-Ullrich, P.; Molinelli, A.; Sulyok, M.; MacDonald, S.; Crews, C. Mycotoxin analysis: An update. Food Addit. Contam. Part A 2008, 25, 152–163. [Google Scholar] [CrossRef] [PubMed]
- Anfossi, L.; Giovannoli, C.; Baggiani, C. Mycotoxin detection. Curr. Opin. Biotechnol. 2016, 37, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Turner, N.W.; Bramhmbhatt, H.; Szabo-Vezse, M.; Poma, A.; Coker, R.; Piletsky, S.A. Analytical Methods for determination of mycotoxins: An update (2009–2014). Anal. Chim. Acta 2015, 901, 12–33. [Google Scholar] [CrossRef] [PubMed]
- Whitaker, T.B. Sampling foods for mycotoxins. Food Addit. Contam. 2006, 23, 50–61. [Google Scholar] [CrossRef] [PubMed]
- Maragos, C.M.; Busman, M. Rapid and advanced tools for mycotoxin analysis: A review. Food Addit. Contam. Part A 2010, 27, 688–700. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Zhang, Z.; Hu, X.; Zhang, Q. Advanced hyphenated chromatographic-mass spectrometry in mycotoxin determination: Current status and prospects. Mass Spectrom. Rev. 2013, 32, 420–452. [Google Scholar] [CrossRef] [PubMed]
- Von Holst, C.; Stroka, J. Performance criteria for rapid screening methods to detect mycotoxins. World Mycotoxin J. 2014, 7, 439–447. [Google Scholar] [CrossRef]
- Whitaker, T.B. Standardisation of mycotoxin sampling procedures: An urgent necessity. Food Control 2003, 14, 233–237. [Google Scholar] [CrossRef]
- Streit, E.; Naehrer, K.; Rodrigues, I.; Schatzmayr, G. Mycotoxin occurrence in feed and feed raw materials worldwide: Long-term analysis with special focus on Europe and Asia. J. Sci. Food Agric. 2013, 93, 2892–2899. [Google Scholar] [CrossRef] [PubMed]
- Pinotti, L.; Ottoboni, M.; Giromini, C.; Dell’Orto, V.; Cheli, F. Mycotoxin Contamination in the EU Feed Supply Chain: A Focus on Cereal Byproducts. Toxins 2016, 8, 45. [Google Scholar] [CrossRef] [PubMed]
- Bryden, W.L. Mycotoxin contamination of the feed supply chain: Implications for animal productivity and feed security. Anim. Feed Sci. Technol. 2012, 173, 134–158. [Google Scholar] [CrossRef]
- Picot, A.; Hourcade-Marcolla, D.; Barreau, C.; Pinson-Gadais, L.; Caron, D.; Richard-Forget, F.; Lannou, C. Interactions between Fusarium verticillioides and Fusarium graminearum in maize ears and consequences for fungal development and mycotoxin accumulation. Plant Pathol. 2012, 61, 140–151. [Google Scholar] [CrossRef]
- Bruns, H.A. Controlling Aflatoxin and Fumonisin in Maize by Crop Management. J. Toxicol. Toxin Rev. 2003, 22, 153–173. [Google Scholar] [CrossRef]
- Cary, J.W.; Rajasekaran, K.; Brown, R.L.; Luo, M.; Chen, Z.-Y.; Bhatnagar, D. Developing Resistance to Aflatoxin in Maize and Cottonseed. Toxins 2011, 3, 678–696. [Google Scholar] [CrossRef] [PubMed]
- Magan, N.; Hope, R.; Cairns, V.; Aldred, D. Post-harvest fungal ecology: Impact of fungal growth and mycotoxin accumulation in stored grain. Eur. J. Plant Pathol. 2003, 109, 723–730. [Google Scholar] [CrossRef]
- Suleiman, R.A.; Rosentrater, K.A.; Bern, C.J. Effects of deterioration parameters on storage of maize. In Proceedings of the American Society of Agricultural and Biological Engineers, Kansas City, MO, USA, 21–24 July 2013; p. 1.
- Streit, E.; Schatzmayr, G.; Tassis, P.; Tzika, E.; Marin, D.; Taranu, I.; Tabuc, C.; Nicolau, A.; Aprodu, I.; Puel, O.; et al. Current Situation of Mycotoxin Contamination and Co-occurrence in Animal Feed—Focus on Europe. Toxins 2012, 4, 788–809. [Google Scholar] [CrossRef] [PubMed]
- Van der Fels-Klerx, H.J.; Asselt, E.D.; Madsen, M.S.; Olesen, J.E. Impact of Climate Change Effects on Contamination of Cereal Grains with Deoxynivalenol. PLoS ONE 2013, 8, e73602. [Google Scholar]
- Medina, A.; Rodriguez, A.; Magan, N. Effect of climate change on Aspergillus flavus and aflatoxin B1 production. Food Microbiol. 2014, 5, 348. [Google Scholar] [CrossRef] [PubMed]
- Binder, E.M.; Tan, L.M.; Chin, L.J.; Handl, J.; Richard, J. Worldwide occurrence of mycotoxins in commodities, feeds and feed ingredients. Anim. Feed Sci. Technol. 2007, 137, 265–282. [Google Scholar] [CrossRef]
- Rodrigues, I.; Naehrer, K. A Three-Year Survey on the Worldwide Occurrence of Mycotoxins in Feedstuffs and Feed. Toxins 2012, 4, 663–675. [Google Scholar] [CrossRef] [PubMed]
- Borutova, R.; Aragon, Y.A.; Nährer, K.; Berthiller, F. Co-occurrence and statistical correlations between mycotoxins in feedstuffs collected in the Asia-Oceania in 2010. Anim. Feed Sci. Technol. 2012, 178, 190–197. [Google Scholar] [CrossRef]
- Marin, S.; Ramos, A.J.; Cano-Sancho, G.; Sanchis, V. Mycotoxins: Occurrence, toxicology, and exposure assessment. Food Chem. Toxicol. 2013, 60, 218–237. [Google Scholar] [CrossRef] [PubMed]
- Schatzmayr, G.; Streit, E. Global occurrence of mycotoxins in the food and feed chain: Facts and figures. World Mycotoxin J. 2013, 6, 213–222. [Google Scholar] [CrossRef]
- Darwish, W.S.; Ikenaka, Y.; Nakayama, S.M.M.; Ishizuka, M. An Overview on Mycotoxin Contamination of Foods in Africa. J. Vet. Med. Sci. 2014, 76, 789–797. [Google Scholar] [CrossRef] [PubMed]
- Yassin, M.A.; El-Samawaty, A.-R.; Bahkali, A.; Moslem, M.; Abd-Elsalam, K.A.; Hyde, K.D. Mycotoxin-producing fungi occurring in sorghum grains from Saudi Arabia. Fungal Divers. 2010, 44, 45–52. [Google Scholar] [CrossRef]
- Hussaini, A.M.; Timothy, A.G.; Olufunmilayo, H.A.; Ezekiel, A.S.; Godwin, H.O. Fungi and Some Mycotoxins Found in Mouldy Sorghum in Niger State, Nigeria. World J. Agric. Sci. 2009, 5, 5–17. [Google Scholar]
- McMillian, W.W.; Wilson, D.M.; Mirocha, C.J.; Widstrom, N.W. Mycotoxin contamination in grain sorghum from fields in Georgia and Mississippi. Cereal Chem. 1983, 60, 226–227. [Google Scholar]
- Kumar, V.; Basu, M.S.; Rajendran, T.P. Mycotoxin research and mycoflora in some commercially important agricultural commodities. Crop Prot. 2008, 27, 891–905. [Google Scholar] [CrossRef]
- Makun, H.A.; Gbodi, T.A.; Tijani, A.S.; Abai, A.; Kadiri, G.U. Toxicologic screening of fungi isolated from millet (pennisetum spp) during the rainy and dry harmattan seasons in Niger state, Nigeria. Afr. J. Biotechnol. 2007, 6, 034–040. [Google Scholar]
- Penugonda, S.; Girisham, S.; Reddy, S.M. Elaboration of mycotoxins by seed-borne fungi of finger millet (Eleusine coracana L.). Int. J. Biotechnol. Mol. Biol. Res. 2010, 1, 58–60. [Google Scholar]
- Wilson, J.P.; Casper, H.H.; Wilson, D.M. Effect of delayed harvest on contamination of pearl millet grain with mycotoxin-producing fungi and mycotoxins. Mycopathologia 1995, 132, 27–30. [Google Scholar] [CrossRef]
- Campbell, H.; Choo, T.M.; Vigier, B.; Underhill, L. Mycotoxins in barley and oat samples from eastern Canada. Rev. Can. Phytotech. 2000, 80, 978–980. [Google Scholar] [CrossRef]
- Bolechová, M.; Benešová, K.; Běláková, S.; Čáslavský, J.; Pospíchalová, M.; Mikulíková, R. Determination of seventeen mycotoxins in barley and malt in the Czech Republic. Food Control 2015, 47, 108–113. [Google Scholar] [CrossRef]
- Vanova, M.; Hajslova, J.; Havlová, P.; Matusinsky, P.; Lancová, K.; Spitzerová, D. Effect of spring barley protection on the production of Fusarium spp. mycotoxins in grain and malt using fungicides in field trials. Plant Soil Environ. 2004, 50, 447–455. [Google Scholar]
- Nakajima, T.; Yoshida, M.; Tomimura, K. Effect of lodging on the level of mycotoxins in wheat, barley, and rice infected with the Fusarium graminearum species complex. J. Gen. Plant Pathol. 2008, 74, 289. [Google Scholar] [CrossRef]
- Edwards, S.G. Fusarium mycotoxin content of UK organic and conventional barley. Food Addit. Contam. Part A 2009, 26, 1185–1190. [Google Scholar] [CrossRef]
- Stenglein, S.A.; Dinolfo, M.I.; Barros, G.; Bongiorno, F.; Chulze, S.N.; Moreno, M.V. Fusarium poae Pathogenicity and Mycotoxin Accumulation on Selected Wheat and Barley Genotypes at a Single Location in Argentina. Plant Dis. 2014, 98, 1733–1738. [Google Scholar] [CrossRef]
- Abramson, D.; Hulasare, R.; White, N.D.G.; Jayas, D.S.; Marquardt, R.R. Mycotoxin formation in hulless barley during granary storage at 15 and 19% moisture content 11 Contribution number 1724 of the Agriculture and Agri-Food Canada, Cereal Research Center. J. Stored Prod. Res. 1999, 35, 297–305. [Google Scholar] [CrossRef]
- Horkỳ, P. Effect of Storage Time of Barley on the Occurrence of Mycotoxins after Artificial Inoculation (Fusarium culmorum). Research In Pig Breeding 2014, 8, 8–10. [Google Scholar]
- Tima, H.; Brückner, A.; Mohácsi-Farkas, C.; Kiskó, G. Fusarium mycotoxins in cereals harvested from Hungarian fields. Food Addit. Contam. Part B Surveill. 2016, 9, 127–131. [Google Scholar] [CrossRef] [PubMed]
- Nathanail, A.V.; Syvähuoko, J.; Malachová, A.; Jestoi, M.; Varga, E.; Michlmayr, H.; Adam, G.; Sieviläinen, E.; Berthiller, F.; Peltonen, K. Simultaneous determination of major type A and B trichothecenes, zearalenone and certain modified metabolites in Finnish cereal grains with a novel liquid chromatography-tandem mass spectrometric method. Anal. Bioanal. Chem. 2015, 407, 4745–4755. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Madden, L.V.; Edwards, S.G. Modeling the effects of environmental conditions on HT2 and T2 toxin accumulation in field oat grains. Phytopathology 2014, 104, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Sago, Y.; Zheng, Y.; Nakagawa, H.; Kushiro, M. Mycotoxins in rice. Int. J. Food Microbiol. 2007, 119, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Ferre, F.S. Worldwide occurrence of mycotoxins in rice. Food Control 2016, 62, 291–298. [Google Scholar] [CrossRef]
- Jayaraman, P.; Kalyanasundaram, I. Natural occurrence of aflatoxins and toxigenic fungi in rice bran oil and de-oiled bran. Indian J. Sci. Technol. 2009, 2, 35–37. [Google Scholar]
- Ma, F.; Chen, R.; Li, P.; Zhang, Q.; Zhang, W.; Hu, X. Preparation of an immunoaffinity column with amino-silica gel Microparticles and its application in sample cleanup for aflatoxin detection in Agri-products. Molecules 2013, 18, 2222–2235. [Google Scholar] [CrossRef] [PubMed]
- Banu, N.; Muthumary, J. Aflatoxin B1 contamination in sunflower oil collected from sunflower oil refinery situated in Karnataka. Health (N. Y.) 2010, 2, 973–987. [Google Scholar] [CrossRef]
- Idris, Y.M.A.; Mariod, A.A.; Elnour, I.A.; Mohamed, A.A. Determination of aflatoxin levels in Sudanese edible oils. Food Chem. Toxicol. 2010, 48, 2539–2541. [Google Scholar] [CrossRef] [PubMed]
- Van Duijn, G. Fate of contaminants during the refining process of vegetable oils and fats: A calculation model. Eur. J. Lipid Sci. Technol. 2016, 118, 353–360. [Google Scholar] [CrossRef]
- Elshafie, S.Z.B.; ElMubarak, A.; El-Nagerabi, S.A.F.; Elshafie, A.E. Aflatoxin B1 Contamination of Traditionally Processed Peanuts Butter for Human Consumption in Sudan. Mycopathologia 2010, 171, 435–439. [Google Scholar] [CrossRef] [PubMed]
- Qian, M.; Zhang, H.; Wu, L.; Jin, N.; Wang, J.; Jiang, K. Simultaneous determination of zearalenone and its derivatives in edible vegetable oil by gel permeation chromatography and gas chromatography-triple quadrupole mass spectrometry. Food Chem. 2015, 166, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Mariod, A.A.; Idris, Y.M.A. Aflatoxin B1 levels in groundnut and sunflower oils in different Sudanese states. Food Addit. Contam. Part B Surveill. 2015, 8, 266–270. [Google Scholar] [PubMed]
- Sahay, S.S.; Prasad, T. The occurrence of aflatoxins in mustard and mustard products. Food Addit. Contam. 1990, 7, 509–513. [Google Scholar] [CrossRef] [PubMed]
- Nawaz, S.; Scudamore, K.A.; Rainbird, S.C. Mycotoxins in ingredients of animal feeding stuffs: I. Determination of Alternaria mycotoxins in oilseed rape meal and sunflower seed meal. Food Addit. Contam. 1997, 14, 249–262. [Google Scholar] [CrossRef] [PubMed]
- Pozzi, C.R.; Braghini, R.; Arcaro, J.R.P.; Zorzete, P.; Israel, A.L.M.; Pozar, I.O.; Denucci, S.; Corrêa, B. Mycoflora and occurrence of alternariol and alternariol monomethyl ether in Brazilian sunflower from sowing to harvest. J. Agric. Food Chem. 2005, 53, 5824–5828. [Google Scholar] [CrossRef] [PubMed]
- Lanier, C.; Heutte, N.; Richard, E.; Bouchart, V.; Lebailly, P.; Garon, D. Mycoflora and mycotoxin production in oilseed cakes during farm storage. J. Agric. Food Chem. 2009, 57, 1640–1645. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarti, D.K.; Ghosal, S. Mycotoxins produced by Fusarium oxysporum in the seeds of Brassica campestris during storage. Mycopathologia 1987, 97, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Jaime-Garcia, R.; Cotty, P.J. Aflatoxin contamination of commercial cottonseed in South Texas. Phytopathology 2003, 93, 1190–1200. [Google Scholar] [CrossRef] [PubMed]
- Lillehoj, E.B.; Wall, J.H.; Bowers, E.J. Preharvest aflatoxin contamination: Effect of moisture and substrate variation in developing cottonseed and corn kernels. Appl. Environ. Microbiol. 1987, 53, 584–586. [Google Scholar] [PubMed]
- Cotty, P.J. Effect of harvest date on aflatoxin contamination of cottonseed. Plant Dis. 1991, 75, 312–314. [Google Scholar] [CrossRef]
- Bock, C.H.; Cotty, P.J. The relationship of gin date to aflatoxin contamination of cottonseed in Arizona. Plant Dis. 1999, 83, 279–285. [Google Scholar] [CrossRef]
- Mazen, M.B.; el-Kady, I.A.; Saber, S.M. Survey of the mycoflora and mycotoxins of cotton seeds and cotton seed products in Egypt. Mycopathologia 1990, 110, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Scudamore, K.A.; Hetmanski, M.T.; Chan, H.K.; Collins, S. Occurrence of mycotoxins in raw ingredients used for animal feeding stuffs in the United Kingdom in 1992. Food Addit. Contam. 1997, 14, 157–173. [Google Scholar] [CrossRef] [PubMed]
- Céspedes, A.E.; Diaz, G.J. Analysis of aflatoxins in poultry and pig feeds and feedstuffs used in Colombia. J. AOAC Int. 1997, 80, 1215–1219. [Google Scholar] [PubMed]
- Buser, M.D.; Abbas, H.K. Effects of Extrusion Temperature and Dwell Time on Aflatoxin Levels in Cottonseed. J. Agric. Food Chem. 2002, 50, 2556–2559. [Google Scholar] [CrossRef] [PubMed]
- Cotty, P.J.; Howell, D.R.; Bock, C.; Tellez, A. Aflatoxin contamination of commercially grown transgenic BT cottonseed. In Proceedings of the Beltwide Cotton Production Research Conference, National Cotton Council of America, Memphis, TN, USA; 1997; pp. 108–110. [Google Scholar]
- Bedre, R.; Rajasekaran, K.; Mangu, V.R.; Sanchez Timm, L.E.; Bhatnagar, D.; Baisakh, N. Genome-Wide Transcriptome Analysis of Cotton (Gossypium hirsutum L.) Identifies Candidate Gene Signatures in Response to Aflatoxin Producing Fungus Aspergillus flavus. PLoS ONE 2015, 10, e0138025. [Google Scholar] [CrossRef] [PubMed]
- Guerre, P. Fusariotoxins in Avian Species: Toxicokinetics, Metabolism and Persistence in Tissues. Toxins 2015, 7, 2289–2305. [Google Scholar] [CrossRef] [PubMed]
- AFSSA. Évaluation des Risques liés à la Présence de Mycotoxines Dans les Chaînes Alimentaires Humaine et Animale; AFSSA: Paris, France, 2009; pp. 1–308. (In French) [Google Scholar]
- Holden, P.; Ewan, R.; Jurgens, M.; Stahly, T.; Zimmerman, D. Life Cycle Swine Nutrition. Available online: http://www.ipic.iastate.edu/LCSN/LCSNmetric.pdf (accessed on 30 August 2016).
- Tardieu, D.; Bailly, J.D.; Benard, G.; Tran, T.S.; Guerre, P. Toxicity of maize containing known levels of fumonisin B1 during force-feeding of ducks. Poult. Sci. 2004, 83, 1287–1293. [Google Scholar] [CrossRef] [PubMed]
- Marquardt, R.R.; Boros, D.; Guenter, W.; Crow, G. The nutritive value of barley, rye, wheat and corn for young chicks as affected by use of a Trichoderma reesei enzyme preparation. Anim. Feed Sci. Technol. 1994, 45, 368–378. [Google Scholar] [CrossRef]
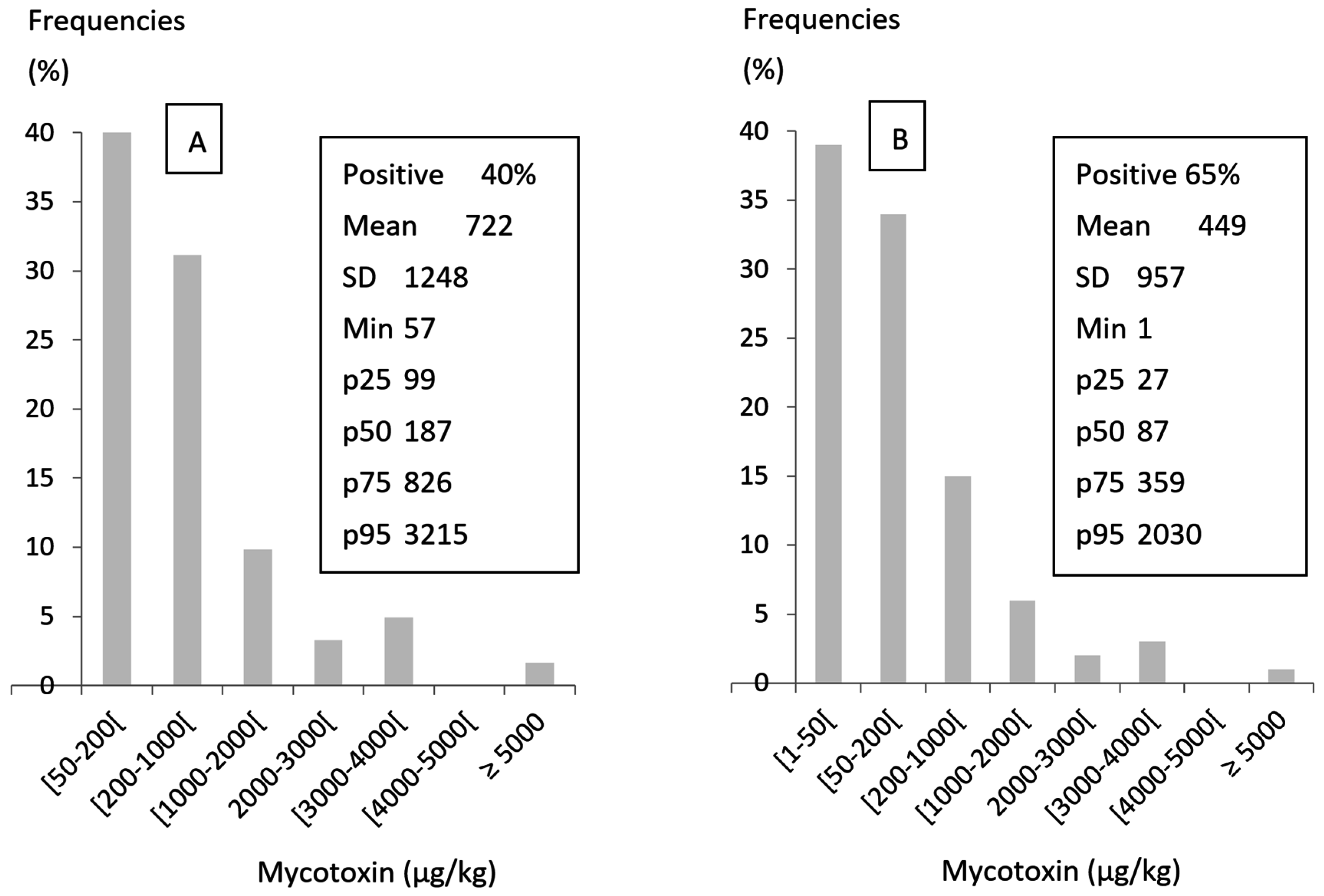
| Mycotoxin | Feed Materials | Maximum Levels, µg/kg |
|---|---|---|
| a Aflatoxin B1 | All feed materials | 20 |
| Complete feedstuffs for pigs and poultry (except young animals) | 20 | |
| Other complete feedstuffs | 10 | |
| Complementary feedstuffs for pigs and poultry (except young animals) | 20 | |
| Other complementary feedstuffs | 5 | |
| b Deoxynivalenol | Feed materials | - |
| Cereals and cereal products with the exception of maize by-products | 8000 | |
| Maize by-products | 12,000 | |
| Complementary and complete feedstuffs with the exception of: | 5000 | |
| Complementary and complete feedstuffs for pigs | 900 | |
| b Zearalenone | Feed materials | - |
| Cereals and cereal products with the exception of maize by-products | 2000 | |
| Maize byproducts | 3000 | |
| Complementary and complete feedstuffs | - | |
| Complementary and complete feedstuffs for piglets and gilts (young sows) | 100 | |
| Complementary and complete feedstuffs for sows and fattening pigs | 250 | |
| b Ochratoxin A | Feed materials | - |
| Cereals and cereal products | 250 | |
| Complementary and complete feedstuffs | - | |
| Complementary and complete feedstuffs for pigs | 50 | |
| Complementary and complete feedstuffs for poultry | 100 | |
| b Fumonisin B1 + B2 | Feed materials | - |
| Maize and maize products | 60,000 | |
| Complementary and complete feedstuffs for: | - | |
| Pigs | 5000 | |
| Poultry | 20,000 | |
| b Sum T-2 and HT-2 toxin | Cereal products for feed and compound feed | - |
| Oat milling products (husks) | 2000 | |
| Other cereal products | 500 | |
| Compound feed | 250 |
| Mycotoxin | Intended Use/Class of Animal | Grain, Grain by-Product, Feed or Other Products | Maximum Levels, µg/kg |
|---|---|---|---|
| Aflatoxin B1 | Immature animals | Maize, peanut products, and other animal feeds and ingredients, excluding cottonseed meal | 20 |
| Animals not listed above, or unknown use | Maize, peanut products, cottonseed, and other animal feeds and ingredients | 20 | |
| Breeding swine and mature poultry | Maize and peanut products | 100 | |
| Finishing swine 100 pounds or greater in weight | Maize and peanut products | 200 | |
| Swine or poultry, regardless of age or breeding status | Cottonseed meal | 300 | |
| Vomitoxin * | Swine | Grain and grain by-products not to exceed 20% of diet | 5000 |
| Complete diet | 1000 | ||
| Chickens | Grain and grain by-products not to exceed 20% of diet | 10,000 | |
| Complete diet | 5000 | ||
| Fumonisin ** | Swine | Maize and maize by-products not to exceed 50% of the diet | 20,000 |
| Complete diet | 10,000 | ||
| Poultry being raised for slaughter and hens laying eggs for human consumption | Maize and maize by-products not to exceed 50% of the diet | 100,000 | |
| Complete diet | 50,000 | ||
| All other species or classes of livestock | Maize and maize by-products not to exceed 50% of the diet | 10,000 | |
| Complete diet | 10,000 |
| Raw Material | % in Diet | Mycotoxins n |
|---|---|---|
| Maize | 0–60 a | High levels of Afla, OTA, DON, FB, ZEA contamination possible. Marked geographic variations. |
| Sorghum | 0–60 b | Few data available. Same mycotoxins as maize but at lower rates. |
| Millet | 0–60 | Few data available. Could be relatively resistant to fungal infection compared with other cereals. |
| Wheat | 0–30 c | Contamination by OTA, DON, FB, ZEA possible, with marked geographic variations. Usually weakly contaminated by Afla. |
| Barley | 0–30 d | Same mycotoxins as wheat, with marked variations depending on the breed. |
| Oats | 0–90 e | Same mycotoxins as wheat, marked variations. T2 and HT2 can be high. |
| Rice | 0–30 | Same mycotoxins as wheat with lower concentrations of mycotoxins except Afla and OTA, which can be high. |
| Cereal by-products | 0–30 f | Physicochemical properties of mycotoxins, location in the grain, and process used influence partitioning. |
| Oil or fat | <5 | Usually low in refined oils. |
| Soybean meal | 0–40 g | Low level contamination by Afla, OTA, DON, FB, ZEA. Marked geographic variations. |
| Rapeseed and canola meals | 0–20 h | Indirect evidence for the presence of Afla, ZEA, DON, alternariol, tenuazonic acid and gliotoxin at levels near that of soybean meal. Complementary data required. |
| Sunflower meal | 0–40 i | |
| Cottonseed meal | 0–40 j | High level of Afla observed in some parts of the world. |
| Animal protein | <5 k | Very low contamination. |
| Alternative ingredients | 0–80 l | Sparse data, Afla the most frequently reported. Complementary data required. |
| Other ingredients | <1–15 m | Not contaminated |
| Mycotoxin | Raw Material | North America | South America | Central Europe | Southern Europe |
| Afla b | Maize | 10.1–62–920 (375; 26%) | 1.8–4.9–273 (809; 25%) | 1.5–1.8–3 (16; 31%) | 4.0–12–44 (42; 36%) |
| Wheat | 4.1–6.6–9.0 (15; 20%) | 2.6–2.6–3.0 (40; 3%) | 1.6–2–2 (13; 31%) | 1.6–1.8–6 (14; 43%) | |
| Soybean meal | 2.0–2.0–2.0 (74; 1%) | 1.0–1.0–1.0A (60; 8%) | 1.2–1.2–1.3 (8; 38%) | 1.8–2.7–21 (23; 22%) | |
| OTA c | Maize | 2.3–3.1–18 (126; 10%) | 71–275–355 (147; 12%) | 2.4–2.5–3 (21; 10%) | 9.3–29–46 (31; 29%) |
| Wheat | 0.8–0.8–0.8 (2; 50%) | 33–39–43 (11; 45%) | 3.8–5.4–331 (22; 23%) | 0.7–0.7–1.0 (13; 8%) | |
| Soybean meal | 4.6–5.2–6 (18; 17%) | 1.0–10–10 (51; 10%) | 21–21–21 (3; 33%) | 1.2–1.3–1.3 (22; 18%) | |
| DON d | Maize | 565–931–24,900 (390; 79%) | 172–241–939 (322; 17%) | 716–1576–26,121 (535; 72%) | 523–705–3851 (59; 47%) |
| Wheat | 600–976–7000 (25; 76%) | 906–1006–2520 (17; 53%) | 514–960–49,000 (436; 55%) | 716–1864–3505 (24; 38%) | |
| Soybean meal | 187–734–5500 (45; 18%) | 197–292–428 (55; 29%) | 450–494–741 (43; 21%) | 339–428–908 (25; 24%) | |
| FB e | Maize | 490–1161–22,900 (466; 39%) | 2008–3890–53,700 (807; 92%) | 684–4504–7680 (30; 60%) | 1407–3266–11,050 (48; 90%) |
| Wheat | (7; 0%) | 1407–1561–1715 (40; 5%) | 246–348–450 (9; 33%) | 151–538–925 (10; 30%) | |
| Soybean meal | (46; 0%) | 274–295–315 (60; 5%) | (2; 0%) | 95–511–5088 (21; 29%) | |
| ZEA f | Maize | 86–168–4787 (395; 29%) | 87–222–1800 (321; 43%) | 79–155–849 (379; 39%) | 166–276–1546 (52; 21%) |
| Wheat | 275–394–513 (16; 13%) | 73–232–393 (32; 47%) | 65–123–336 (256; 12%) | (17; 0%) | |
| Soybean meal | 51–142–144 (50; 10%) | 81–130–807 (53; 34%) | 36–46–56 (31; 6%) | (23; 0%) | |
| Mycotoxin | Raw Material | North Asia | South-East Asia | South Asia | Oceania |
| Afla a | Maize | 7.0–36–4687 (447; 32%) | 97.0–206–2601 (330; 71%) | 96.0–312–2230 (108; 82%) | 3.0–4.0–5.0 (11; 18%) |
| Wheat | 3.3–3.6–20 (76; 7%) | 1.0–1.0–1.0 (40; 3%) | ND | 2.0–5.0–30 (109; 5%) | |
| Soybean meal | 2.8–2.9–3 (36; 6%) | 1.0–3.3–74 (109; 22%) | 2.0–3.5–7 (16; 63%) | 1.0–1.0–1.0 (3; 67%) | |
| OTA | Maize | 1.4–4.1–19 (420; 10%) | 3.0–6.3–80 (218; 12%) | 7.4–15–400 (107; 27%) | 1.2–1.2–1.2 (11; 9%) |
| Wheat | 1.0–2.0–7 (67; 22%) | 3.9–5.7–30 (40; 30%) | ND | 1.6–3.7–4 (108; 8%) | |
| Soybean meal | 1.6–4.4–19 (33; 24%) | 2.4–5.6–21 (105; 16%) | 14–23–46 (16; 56%) | 3.1–3.1–3.1 (3; 33%) | |
| DON | Maize | 640–1444–15,073 (477; 92%) | 182–352–4805 (218; 45%) | 190–349–1150 (106; 22%) | 179–214–249 (11; 27%) |
| Wheat | 426–1279–5331 (75; 87%) | 199–1483–41,439 (40; 65%) | ND | 719–5870–49,307 (109; 48%) | |
| Soybean meal | 107–202–314 (37; 38%) | 228–285–973 (105; 18%) | 249–251–259 (16; 31%) | 150–150–150 (3; 33%) | |
| FB | Maize | 1519–3594–23,499 (443; 75%) | 1033–1720–19,289 (326; 83%) | 541–796–6196 (108; 74%) | 2344–4023–5438 (11; 64%) |
| Wheat | 298–471–874 (73; 11%) | 172–232–292 (40; 5%) | ND | 196–216–1196 (109; 12%) | |
| Soybean meal | 316–319–321 (35; 6%) | 228–285–973 (109; 4%) | (16; 0%) | (3; 0%) | |
| ZEA | Maize | 176–435–7446 (470; 67%) | 97–206–2601 (319; 20%) | 79–174–1099 (108; 9%) | 626–939–1251 (11; 27%) |
| Wheat | 48–82–465 (72; 42%) | 53–217–6641 (40; 40%) | ND | 180–351–23,278 (115; 28%) | |
| Soybean meal | 31–42–398 (34; 35%) | 38–53–70 (105; 15%) | (16; 0%) | 150–150–150 (3; 33 %) |
| Ingredient (g/kg) | Growing Pig (kg) | Sow | ||
|---|---|---|---|---|
| 10–30 | 80–160 | Gestation | Lactation | |
| Maize | 358 | 766 | 863 | 688 |
| Soybean meal | 460 | 210 | 100 | 275 |
| Dried whey | 150 | 0 | 0 | 0 |
| Other ingredients a | 32 | 24 | 37 | 37 |
| World area | Production | Afla b | OTA c | DON d | FB e | ZEA f | |
|---|---|---|---|---|---|---|---|
| North America | Pig a | 10–30 | 4.5–23.1–330 | 2.9–3.5–9.2 | 288–671–11,444 | 175–416–8198 | 54.2–126–1780 |
| 80–160 | 8.1–47.9–705 | 2.7–3.5–15 | 472–867–20,228 | 375–889–17,541 | 76.6–159–3697 | ||
| Sow | Gestation | 8.9–53.7–794 | 2.4–3.2–16.1 | 506–877–22,039 | 423–1002–19,763 | 79.3–159–4146 | |
| Lactation | 7.5–43.2–634 | 2.8–3.6–14 | 440–842–18,644 | 337–799–15,755 | 73.2–155–3333 | ||
| South America | Pig a | 10–30 | 1.1–2.2–98.2 | 25.9–90.9–341 | 152–221–533 | 845–1528–19,370 | 68.4–139–1016 |
| 80–160 | 1.6–4–209 | 54.6–187–721 | 173–246–809 | 1596–3042–41,200 | 83.7–197–1548 | ||
| Sow | Gestation | 1.7–4.3–236 | 61.4–209–811 | 168–237–853 | 1760–3387–46,375 | 83.2–205–1634 | |
| Lactation | 1.5–3.6–188 | 49.1–169–649 | 172–246–764 | 1457–2757–37,032 | 82.1–189–1460 | ||
| Southern Europe | Pig a | 10–30 | 2.3–5.5–25.4 | 3.9–11–17.1 | 343–449–1796 | 547–1404–6296 | 59.4–98.8–554 |
| 80–160 | 3.4–9.8–38.1 | 7.4–22.5–35.5 | 472–630–3140 | 1098–2609–9533 | 127–211–1184 | ||
| Sow | Gestation | 3.6–10.6–40.1 | 8.1–25.2–39.8 | 485–651–3414 | 994–2388–10,045 | 143–238–1334 | |
| Lactation | 3.2–9–36 | 6.7–20.3–32 | 453–603–2899 | 994–2388–9002 | 114–190–1064 | ||
| Northern Asia | Pig a | 10–30 | 3.8–14.2–1679 | 1.2–3.5–15.5 | 278–610–5541 | 689–1433–8560 | 77.3–175–2849 |
| 80–160 | 6–28.2–3591 | 1.4–4.1–18.5 | 513–1149–11,612 | 1230–2820–18,068 | 141–342–5787 | ||
| Sow | Gestation | 6.3–31.4–4045 | 1.4–4–18.3 | 563–1266–13,039 | 1343–3134–20,312 | 155–380–6466 | |
| Lactation | 5.6–25.6–3226 | 1.4–4–18.3 | 470–1049–10,457 | 1132–2560–16,256 | 130–311–5232 | ||
| South-East Asia | Pig a | 10–30 | 35.2–75.3–965 | 2.2–4.8–38.3 | 170–257–2168 | 475–747–7353 | 52.2–98.1–963 |
| 80–160 | 74.5–159–2008 | 2.8–6–65.7 | 187–330–3885 | 839–1377–14,980 | 82.3–169–2007 | ||
| Sow | Gestation | 83.8–178–2252 | 2.8–6–71.1 | 180–332–4244 | 914–1513–16,744 | 87.5–183–2252 | |
| Lactation | 67–178–1810 | 2.7–5.9–60.8 | 188–321–3573 | 773–1262–13,538 | 77.2–156–1809 | ||
| Ingredient (g/kg) | Maize/Soybean | Wheat/Soybean |
|---|---|---|
| Maize | 628.3 | 0 |
| Wheat | 0 | 676.5 |
| Soybean meal | 300 | 200 |
| Other energy source a | 20 | 70 |
| Other protein source b | 10 | 10 |
| Other ingredients c | 41.7 | 43.5 |
| World Area | Feed a | Afla b | OTA | DON | FB | ZEA |
|---|---|---|---|---|---|---|
| North America | C + S | 6.9–39.6–579 | 2.8–3.5–13.1 | 411–805–17,295 | 308–729–14,388 | 69–148–3051 |
| W + S | 3.2–4.9–6.5 | 1.5–1.6–1.7 | 443–807–5836 | 0–0–0 | 196–288–376 | |
| South America | C + S | 1.4–3.4–172 | 44.9–154–593 | 167–239–718 | 1344–2533–33,834 | 79–178–1373 |
| W + S | 2–2–2.2 | 22.5–28.4–31.1 | 652–739–1790 | 1007–1115–1223 | 66–183–427 | |
| Southern Europe | C + S | 3.1–8.3–33.9 | 6.2–18.6–29.3 | 430–571–2692 | 913–2205–8469 | 104–173–971 |
| W + S | 1.4–1.8–8.3 | 0.7–0.7–0.9 | 552–1347–2553 | 121–466–1643 | 0–0–0 | |
| Northern Asia | C + S | 5.2–23.5–2946 | 1.4–3.9–17.6 | 434–968–9565 | 1049–2354–14,861 | 120–286–4798 |
| W + S | 2.8–3–14.1 | 1–2.2–8.5 | 310–906–3669 | 265–382–655 | 39–64–394 | |
| South-East Asia | C + S | 61.2–130–1656 | 2.6–5.6–56.6 | 183–307–3311 | 717–1166–12,411 | 72–145–1655 |
| W + S | 0.9–1.3–15.5 | 3.1–5–24–5 | 180–1060–28,228 | 162–214–392 | 43–157–4507 |
© 2016 by the author; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guerre, P. Worldwide Mycotoxins Exposure in Pig and Poultry Feed Formulations. Toxins 2016, 8, 350. https://doi.org/10.3390/toxins8120350
Guerre P. Worldwide Mycotoxins Exposure in Pig and Poultry Feed Formulations. Toxins. 2016; 8(12):350. https://doi.org/10.3390/toxins8120350
Chicago/Turabian StyleGuerre, Philippe. 2016. "Worldwide Mycotoxins Exposure in Pig and Poultry Feed Formulations" Toxins 8, no. 12: 350. https://doi.org/10.3390/toxins8120350
APA StyleGuerre, P. (2016). Worldwide Mycotoxins Exposure in Pig and Poultry Feed Formulations. Toxins, 8(12), 350. https://doi.org/10.3390/toxins8120350





