Biochemical Characterization of the SPATE Members EspPα and EspI
Abstract
:1. Introduction
2. Results and Discussion
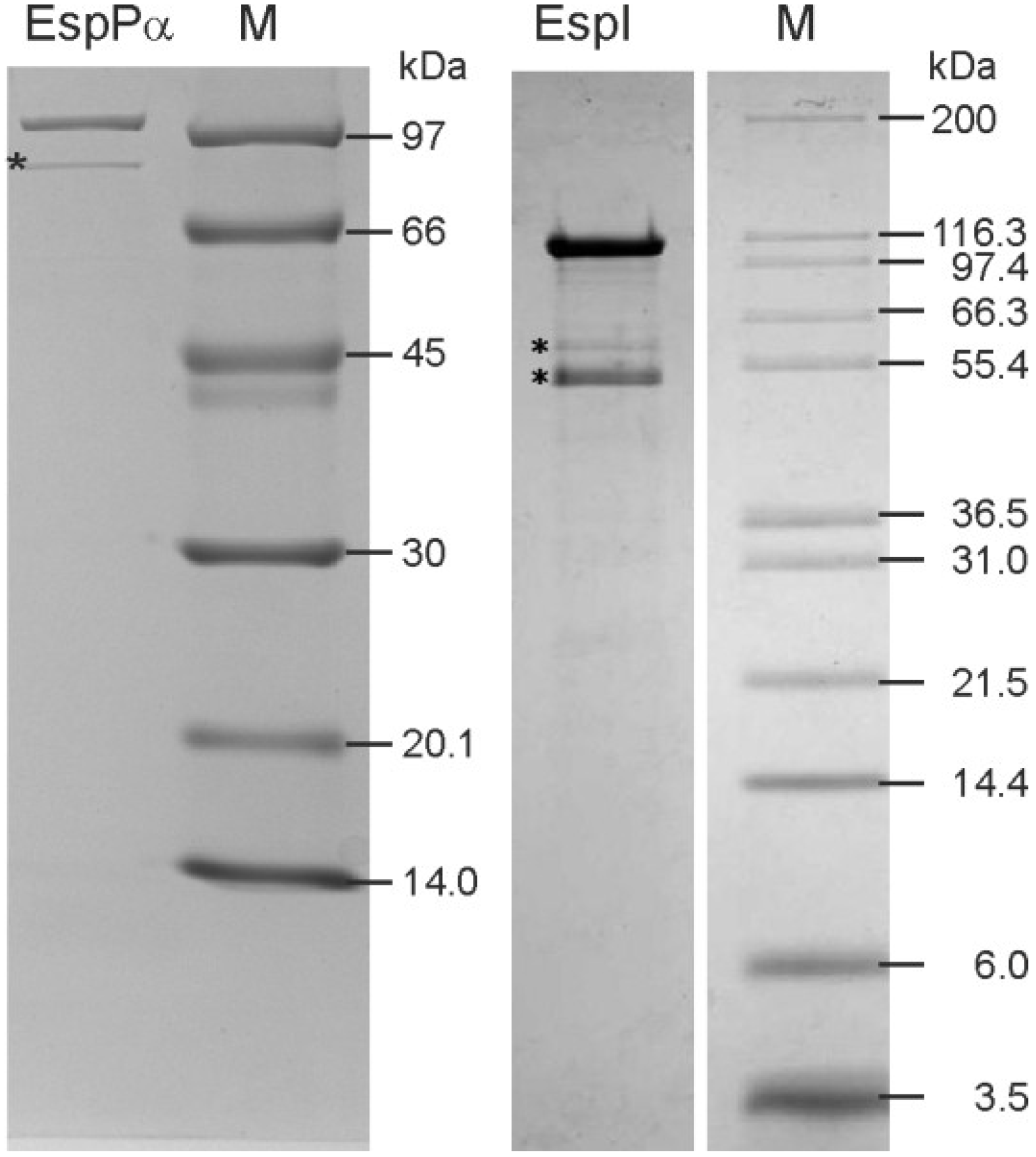
2.1. Purification of EspPα and EspI
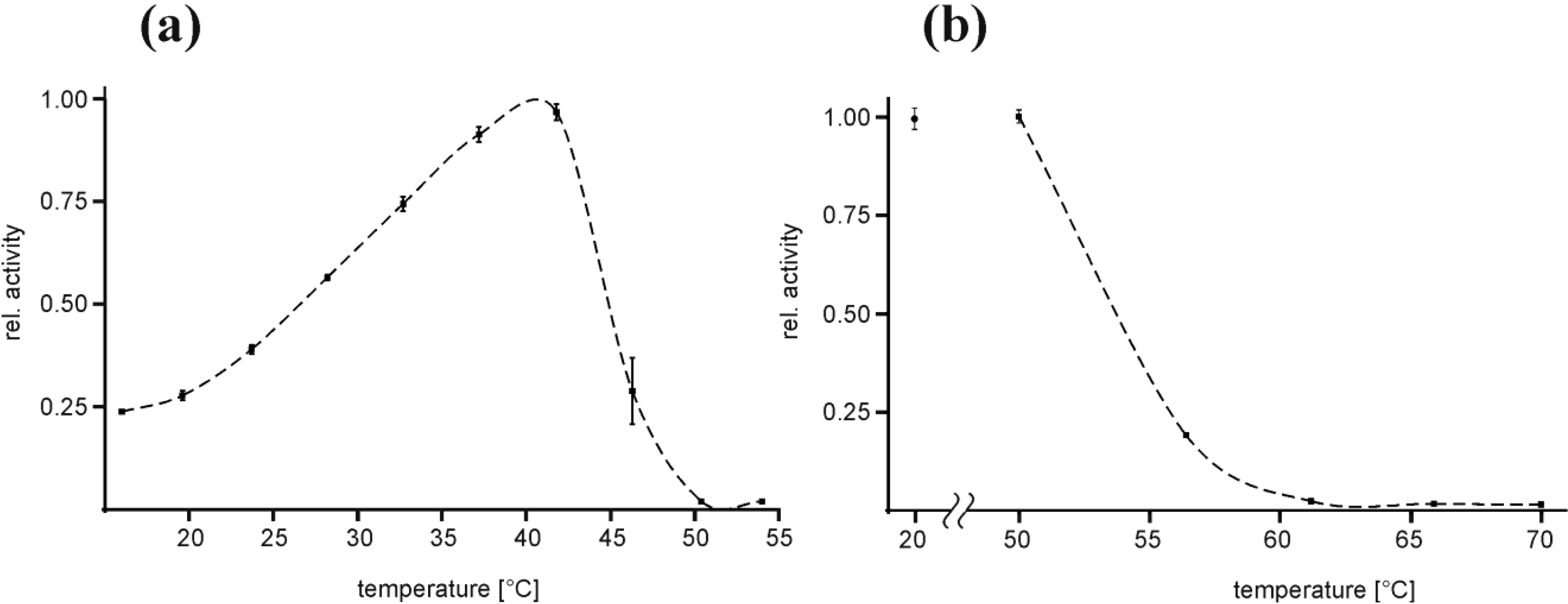
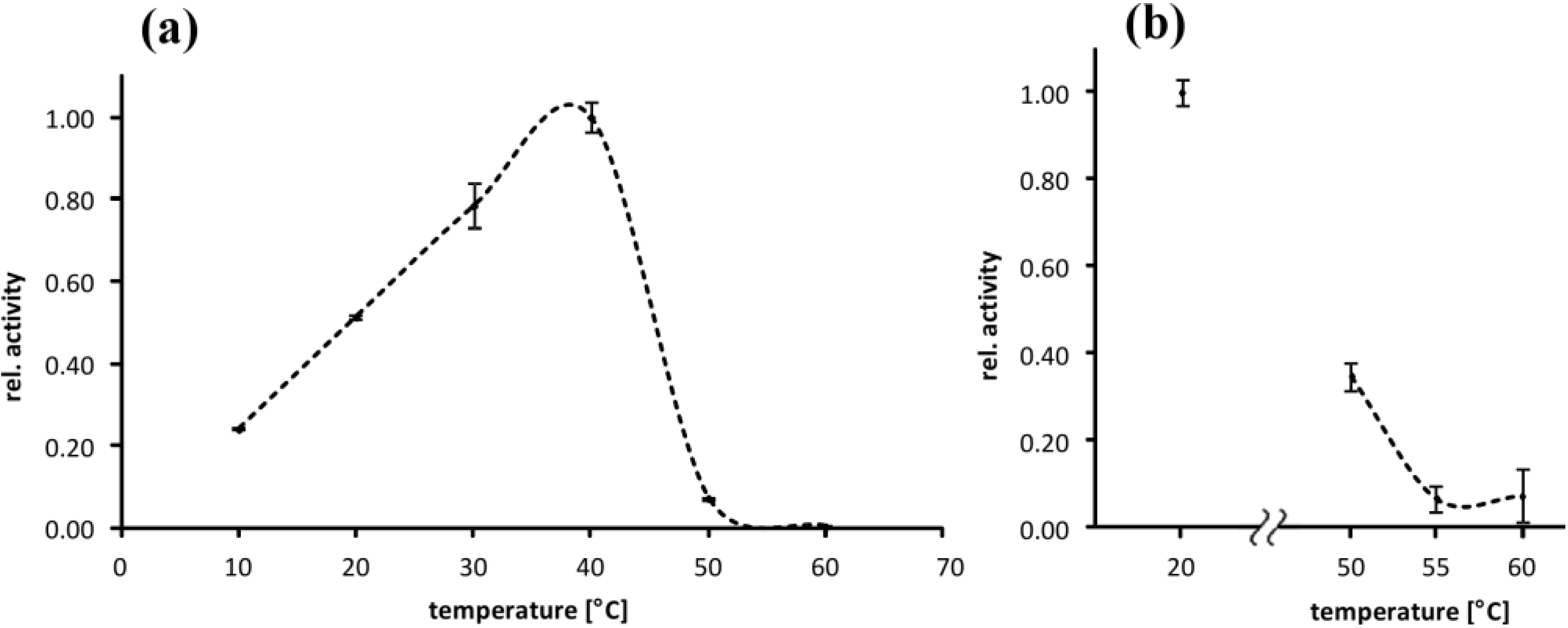
2.2. Temperature Optimum of EspPα and EspI
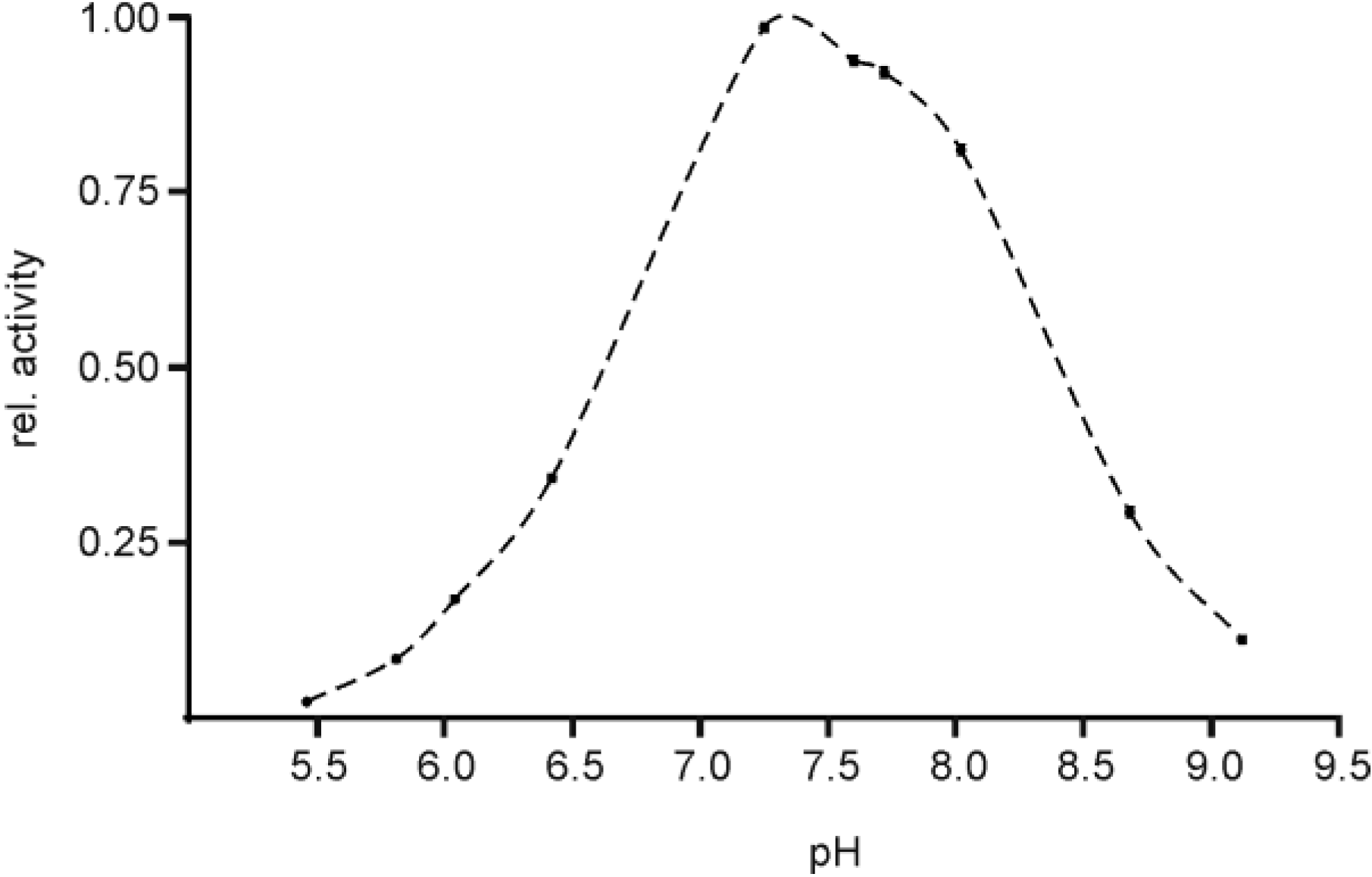
2.3. pH Optimum of EspPα and EspI
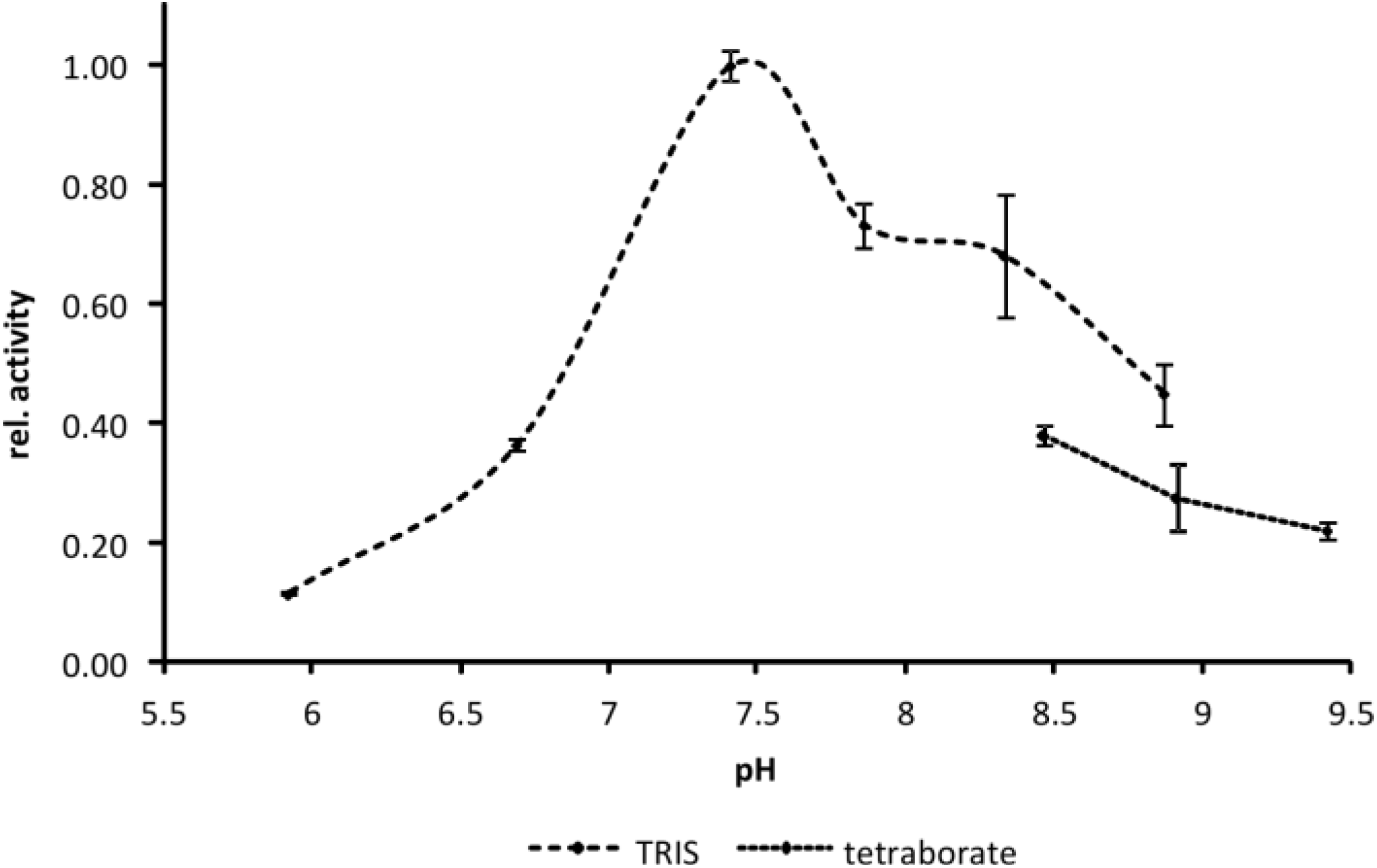
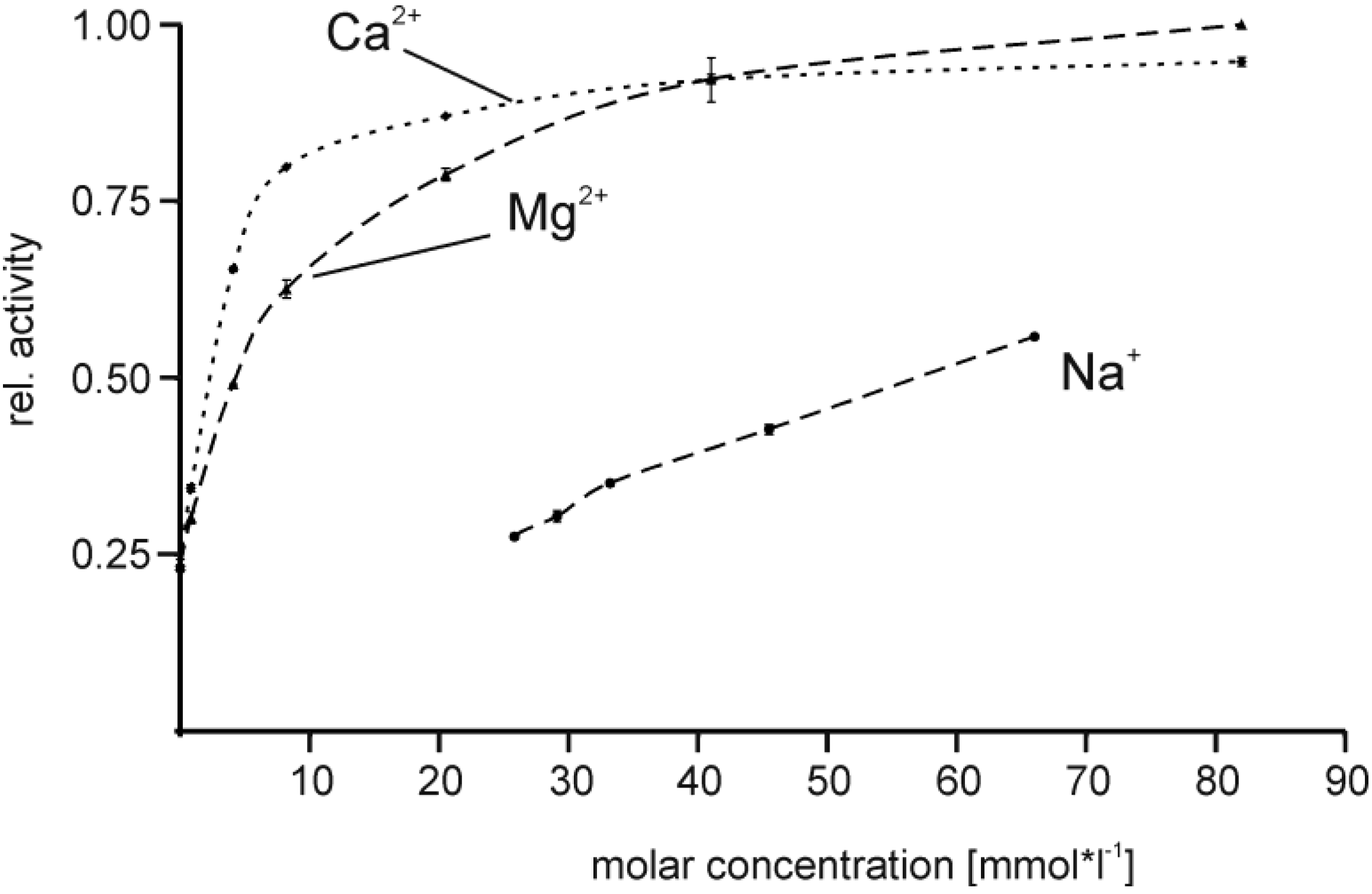
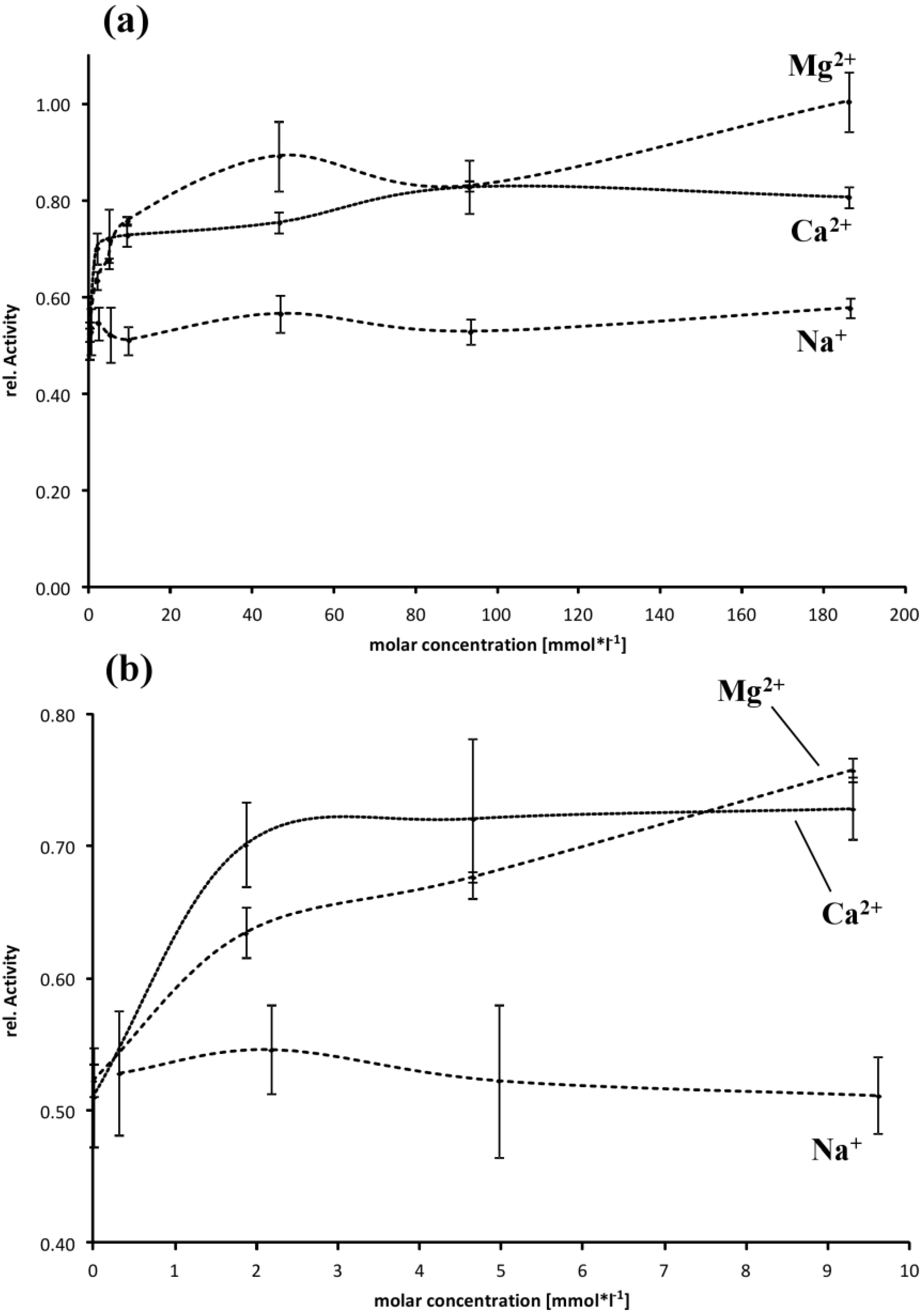
2.4. Effect of Buffer Composition
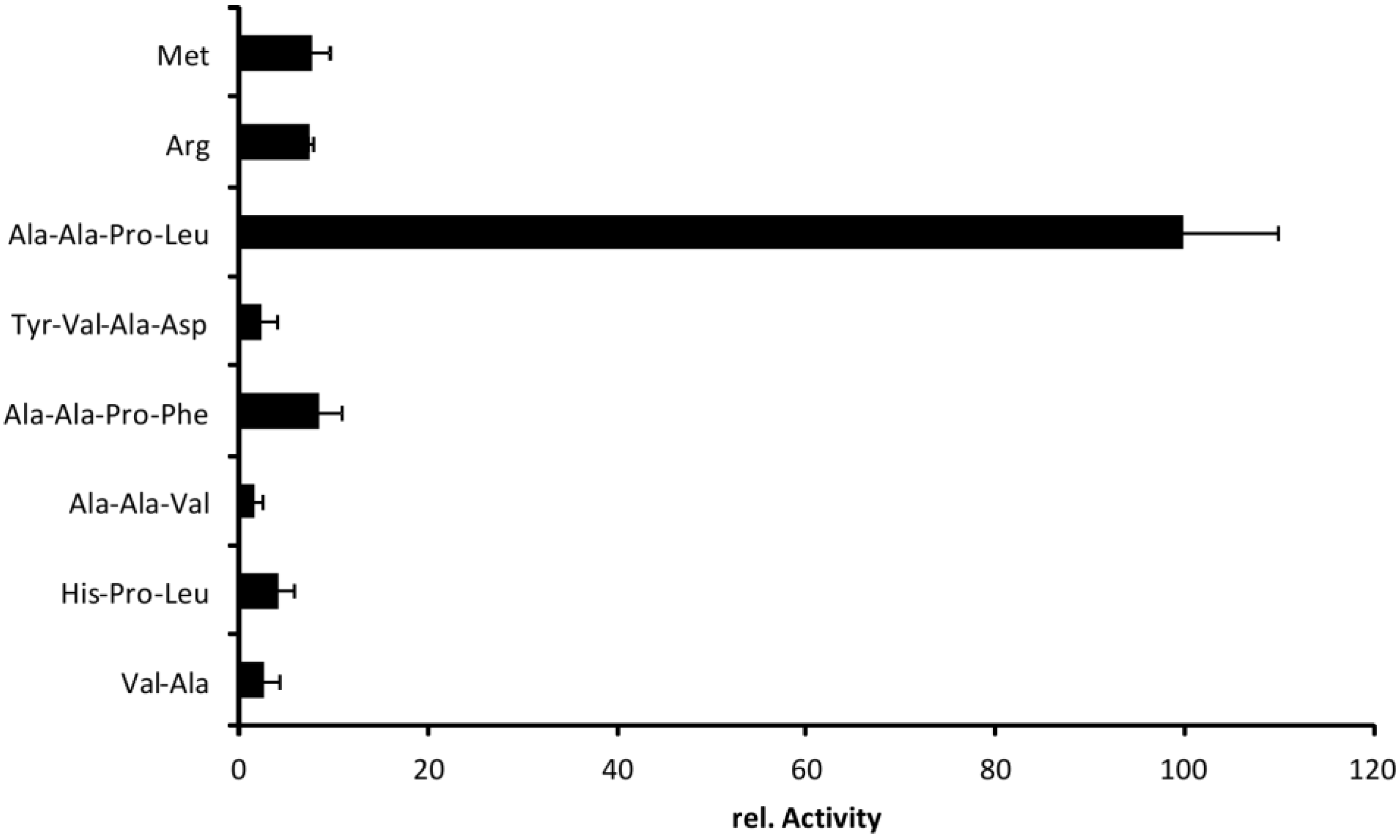
2.5. Cleavage Profile of EspI
3. Experimental Section
3.1. Purification of EspPα and EspI
3.2. Temperature Optimum of EspPα and EspI
3.3. pH Optimum of EspPα and EspI
3.4. Effect of Buffer Composition
3.5. Cleavage Profile of EspI
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Brunder, W.; Schmidt, H.; Karch, H. EspP, a novel extracellular serine protease of enterohaemorrhagic Escherichia coli O157:H7 cleaves human coagulation factor V. Mol. Microbiol. 1997, 24, 767–778. [Google Scholar] [CrossRef] [PubMed]
- Brockmeyer, J.; Spelten, S.; Kuczius, T.; Bielaszewska, M.; Karch, H. Structure and function relationship of the autotransport and proteolytic activity of EspP from Shiga toxin-producing Escherichia coli. PLoS One 2009, 4, e6100. [Google Scholar] [CrossRef] [PubMed]
- Brockmeyer, J.; Bielaszewska, M.; Fruth, A.; Bonn, M.L.; Mellmann, A.; Humpf, H.U.; Karch, H. Subtypes of the plasmid-encoded serine protease EspP in Shiga toxin-producing Escherichia coli: Distribution, secretion, and proteolytic activity. Appl. Environ. Microbiol. 2007, 73, 6351–6359. [Google Scholar] [CrossRef] [PubMed]
- Bielaszewska, M.; Stoewe, F.; Fruth, A.; Zhang, W.; Prager, R.; Brockmeyer, J.; Mellmann, A.; Karch, H.; Friedrich, A.W. Shiga toxin, cytolethal distending toxin, and hemolysin repertoires in clinical Escherichia coli O91 isolates. J. Clin. Microbiol. 2009, 47, 2061–2066. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.B.; Naim, A.; Orth, D.; Grif, K.; Mohsin, M.; Prager, R.; Dierich, M.P.; Wurzner, R. Serine protease espP subtype alpha, but not beta or gamma, of Shiga toxin-producing Escherichia coli is associated with highly pathogenic serogroups. Int. J. Med. Microbiol. 2009, 299, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, H.; Zhang, W.L.; Hemmrich, U.; Jelacic, S.; Brunder, W.; Tarr, P.I.; Dobrindt, U.; Hacker, J.; Karch, H. Identification and characterization of a novel genomic island integrated at selC in locus of enterocyte effacement-negative, Shiga toxin-producing Escherichia coli. Infect. Immun. 2001, 69, 6863–6873. [Google Scholar] [CrossRef] [PubMed]
- Orth, D.; Ehrlenbach, S.; Brockmeyer, J.; Khan, A.B.; Huber, G.; Karch, H.; Sarg, B.; Lindner, H.; Wurzner, R. EspP, a serine protease of enterohemorrhagic Escherichia coli, impairs complement activation by cleaving complement factors C3/C3b and C5. Infect. Immun. 2010, 78, 4294–4301. [Google Scholar] [CrossRef] [PubMed]
- Brockmeyer, J.; Aldick, T.; Soltwisch, J.; Zhang, W.; Tarr, P.I.; Weiss, A.; Dreisewerd, K.; Muthing, J.; Bielaszewska, M.; Karch, H.; et al. Enterohaemorrhagic Escherichia coli haemolysin is cleaved and inactivated by serine protease EspPalpha. Environ. Microbiol. 2011, 13, 1327–1341. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, L.F.; Irino, K.; Vaz, T.M.; Guth, B.E. Set of virulence genes and genetic relatedness of O113:H21 Escherichia coli strains isolated from the animal reservoir and human infections in Brazil. J. Med. Microbiol. 2010, 59, 634–640. [Google Scholar]
- Toszeghy, M.; Phillips, N.; Reeves, H.; Wu, G.; Teale, C.; Coldham, N.; Randall, L. Molecular and phenotypic characterisation of Extended Spectrum beta-lactamase CTX-M Escherichia coli from farm animals in Great Britain. Res. Vet. Sci. 2012, 93, 1142–1150. [Google Scholar] [CrossRef] [PubMed]
- Abreu, A.G.; Bueris, V.; Porangaba, T.M.; Sircili, M.P.; Navarro-Garcia, F.; Elias, W.P. Autotransporter protein-encoding genes of diarrheagenic Escherichia coli are found in both typical and atypical enteropathogenic E. coli. Appl. Environ. Microbiol. 2012. [Google Scholar] [CrossRef]
- Dutta, P.R.; Cappello, R.; Navarro-Garcia, F.; Nataro, J.P. Functional Comparison of Serine Protease Autotransporters of Enterobacteriaceae. Infect. Immun. 2002, 70, 7105–7113. [Google Scholar] [CrossRef] [PubMed]
- Puttamreddy, S.; Cornick, N.A.; Minion, F.C. Genome-wide transposon mutagenesis reveals a role for pO157 genes in biofilm development in Escherichia coli O157:H7 EDL933. Infect. Immun. 2010, 78, 2377–2384. [Google Scholar] [CrossRef] [PubMed]
- Xicohtencatl-Cortes, J.; Saldana, Z.; Deng, W.; Castaneda, E.; Freer, E.; Tarr, P.I.; Finlay, B.B.; Puente, J.L.; Giron, J.A. Bacterial macroscopic rope-like fibers with cytopathic and adhesive properties. J. Biol. Chem. 2010, 285, 32336–32342. [Google Scholar]
- Christen, P.; Jaussi, R. Biochemie, 1st ed.; Springer: Berlin/Heidelberg, Germany, 2005; Volume 1. [Google Scholar]
- Schechter, I.; Berger, A. On the size of the active site in proteases. I. Papain. Biochem. Biophys. Res. Commun. 1967, 27, 157–162. [Google Scholar]
- Dautin, N. Serine Protease Autotransporters of Enterobacteriaceae (SPATEs): Biogenesis and Function. Toxins 2010, 2, 1179–1206. [Google Scholar] [CrossRef] [PubMed]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef] [PubMed]
- Schagger, H.; von Jagow, G. Tricine-sodium dodecyl sulfate-polyacrylamide gel electrophoresis for the separation of proteins in the range from 1 to 100 kDa. Anal. Biochem. 1987, 166, 368–379. [Google Scholar] [CrossRef] [PubMed]
- Helland, R.; Larsen, A.N.; Smalas, A.O.; Willassen, N.P. The 1.8 A crystal structure of a proteinase K-like enzyme from a psychrotroph Serratia species. FEBS J. 2006, 273, 61–71. [Google Scholar]
- Huesgen, P.F.; Scholz, P.; Adamska, I. The serine protease HhoA from Synechocystis sp. strain PCC 6803: Substrate specificity and formation of a hexameric complex are regulated by the PDZ domain. J. Bacteriol. 2007, 189, 6611–6618. [Google Scholar]
- Patel, A.K.; Singh, V.K.; Jagannadham, M.V. Carnein, a serine protease from noxious plant weed Ipomoea carnea (morning glory). J. Agric. Food Chem. 2007, 55, 5809–5818. [Google Scholar] [CrossRef] [PubMed]
- Weiss, A.; Brockmeyer, J. Prevalence, biogenesis, and functionality of the serine protease autotransporter EspP. Toxins 2013, 5, 25–48. [Google Scholar] [CrossRef]
- Ieva, R.; Bernstein, H.D. Interaction of an autotransporter passenger domain with BamA during its translocation across the bacterial outer membrane. Proc. Natl. Acad. Sci. USA 2009, 106, 19120–19125. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Perez, F.; Henderson, I.R.; Leyton, D.L.; Rossiter, A.E.; Zhang, Y.; Nataro, J.P. Roles of periplasmic chaperone proteins in the biogenesis of serine protease autotransporters of Enterobacteriaceae. J. Bacteriol. 2009, 191, 6571–6583. [Google Scholar]
- Ruiz-Perez, F.; Henderson, I.R.; Nataro, J.P. Interaction of FkpA, a peptidyl-prolyl cis/trans isomerase with EspP autotransporter protein. Gut Microbes 2010, 1, 339–344. [Google Scholar]
- Ieva, R.; Skillman, K.M.; Bernstein, H.D. Incorporation of a polypeptide segment into the beta-domain pore during the assembly of a bacterial autotransporter. Mol. Microbiol. 2008, 67, 188–201. [Google Scholar] [CrossRef] [PubMed]
- Jong, W.S.; ten Hagen-Jongman, C.M.; den Blaauwen, T.; Slotboom, D.J.; Tame, J.R.; Wickstrom, D.; de Gier, J.W.; Otto, B.R.; Luirink, J. Limited tolerance towards folded elements during secretion of the autotransporter Hbp. Mol. Microbiol. 2007, 63, 1524–1536. [Google Scholar] [CrossRef] [PubMed]
- Jelkmann, W. Blut und Immunabwehr. In Physiologie des Menschen, 30 bearbeitete und aktualisierte Auflage ed.; Springer: Berlin/Heidelberg, Germany, 2007; Volume 30, pp. 523–549. [Google Scholar]
- Siezen, R.J.; Leunissen, J.A. Subtilases: The superfamily of subtilisin-like serine proteases. Protein Sci. 1997, 6, 501–523. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, K.; Yoon, Y.H.; Kurihara, A.; Unzai, S.; Luirink, J.; Park, S.Y.; Tame, J.R. Role of domains within the autotransporter Hbp/Tsh. Acta Crystallogr. D 2010, 66, 1295–1300. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Mian, H.S.; Sandercock, L.E.; Chirgadze, N.Y.; Pai, E.F. Crystal structure of the passenger domain of the Escherichia coli autotransporter EspP. J. Mol. Biol. 2011, 413, 985–1000. [Google Scholar] [CrossRef] [PubMed]
- Liang, M.P. WebFEATURE: An interactive web tool for identifying and visualizing functional sites on macromolecular structures. Nucleic Acids Res. 2003, 31, 3324–3327. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Weiss, A.; Kortemeier, D.; Brockmeyer, J. Biochemical Characterization of the SPATE Members EspPα and EspI. Toxins 2014, 6, 2719-2731. https://doi.org/10.3390/toxins6092719
Weiss A, Kortemeier D, Brockmeyer J. Biochemical Characterization of the SPATE Members EspPα and EspI. Toxins. 2014; 6(9):2719-2731. https://doi.org/10.3390/toxins6092719
Chicago/Turabian StyleWeiss, André, David Kortemeier, and Jens Brockmeyer. 2014. "Biochemical Characterization of the SPATE Members EspPα and EspI" Toxins 6, no. 9: 2719-2731. https://doi.org/10.3390/toxins6092719
APA StyleWeiss, A., Kortemeier, D., & Brockmeyer, J. (2014). Biochemical Characterization of the SPATE Members EspPα and EspI. Toxins, 6(9), 2719-2731. https://doi.org/10.3390/toxins6092719



