Venom Down Under: Dynamic Evolution of Australian Elapid Snake Toxins
Abstract
:1. Introduction
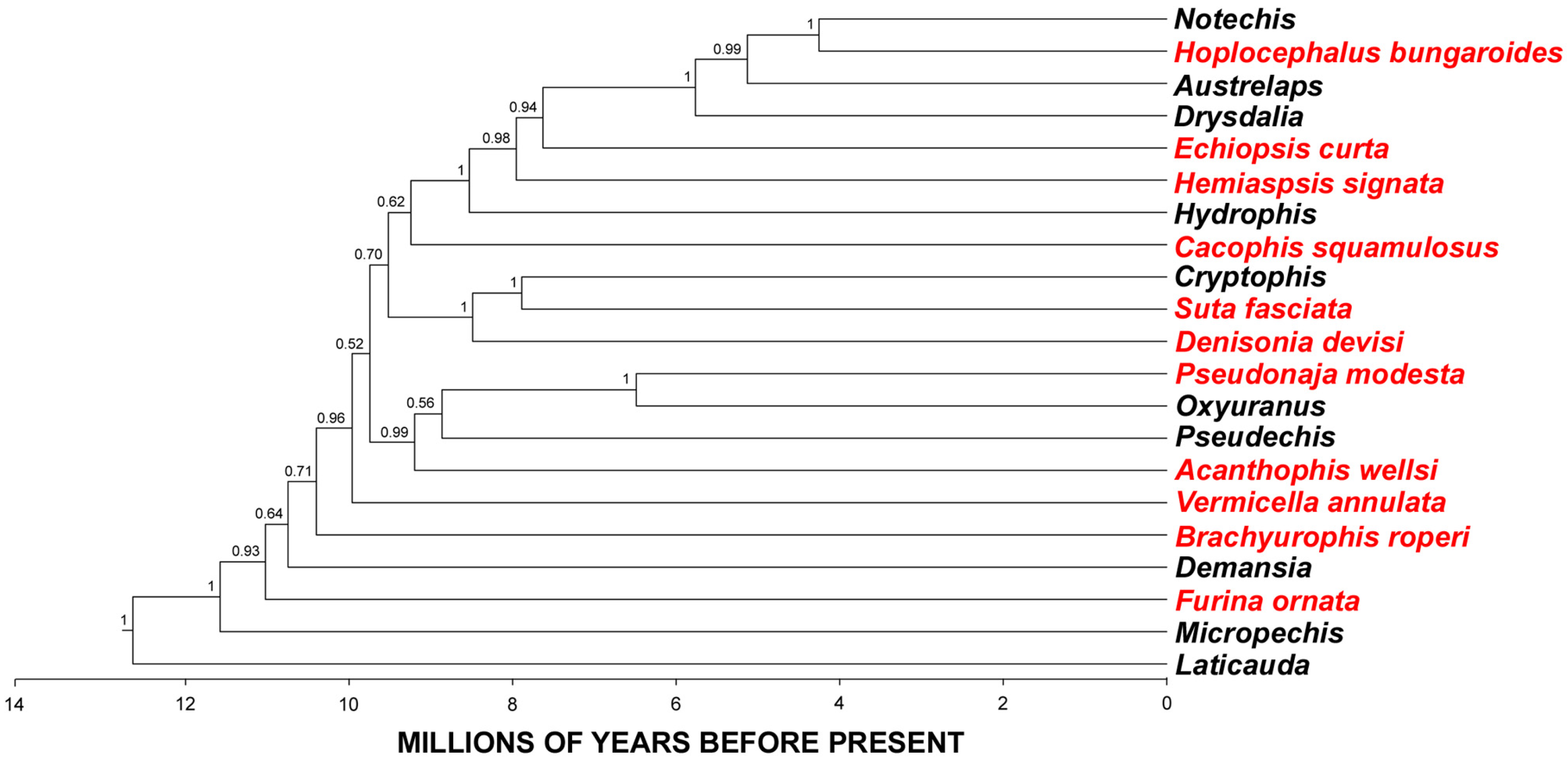
2. Results and Discussion
| Species/Toxin | CRiSP | fXaTx | fVaTx | Kunitz | Lectin | Natriuretic | PLA2 | SVMP | 3FTx | Waprin |
|---|---|---|---|---|---|---|---|---|---|---|
| Acanthophis wellsi | X | X | X | X | X | X | X | X | ||
| Brachyurophis roperi | X | X | X | X | X | X | ||||
| Cacophis squamulosus | X | X | X | X | X | X | X | |||
| Denisonia devisi | X | X | X | X | X | X | X | X | ||
| Echiopsis curta | X | X | X | X | X | X | X | |||
| Furina ornata | X | X | X | X | ||||||
| Hemiaspis signata | X | X | X | X | X | X | X | X | ||
| Hoplocephalus bungaroides | X | X | X | X | X | X | X | |||
| Pseudonaja modesta | X | X | X | X | X | X | X | X | ||
| Suta fasciata | X | X | X | X | X | X | X | X | X | |
| Vermicella annulata | X | X | X | X | X | X |
2.1. 3FTx
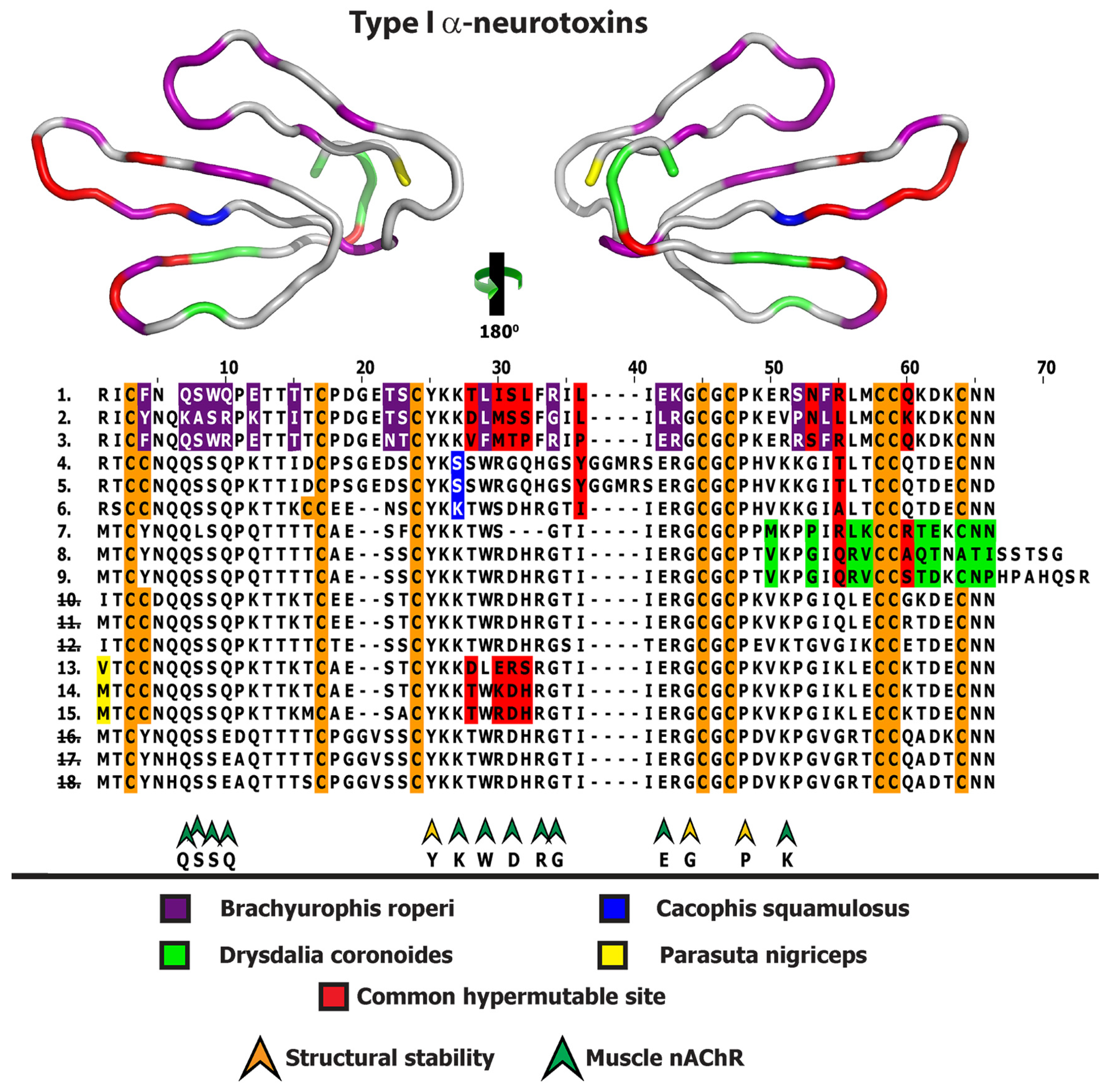
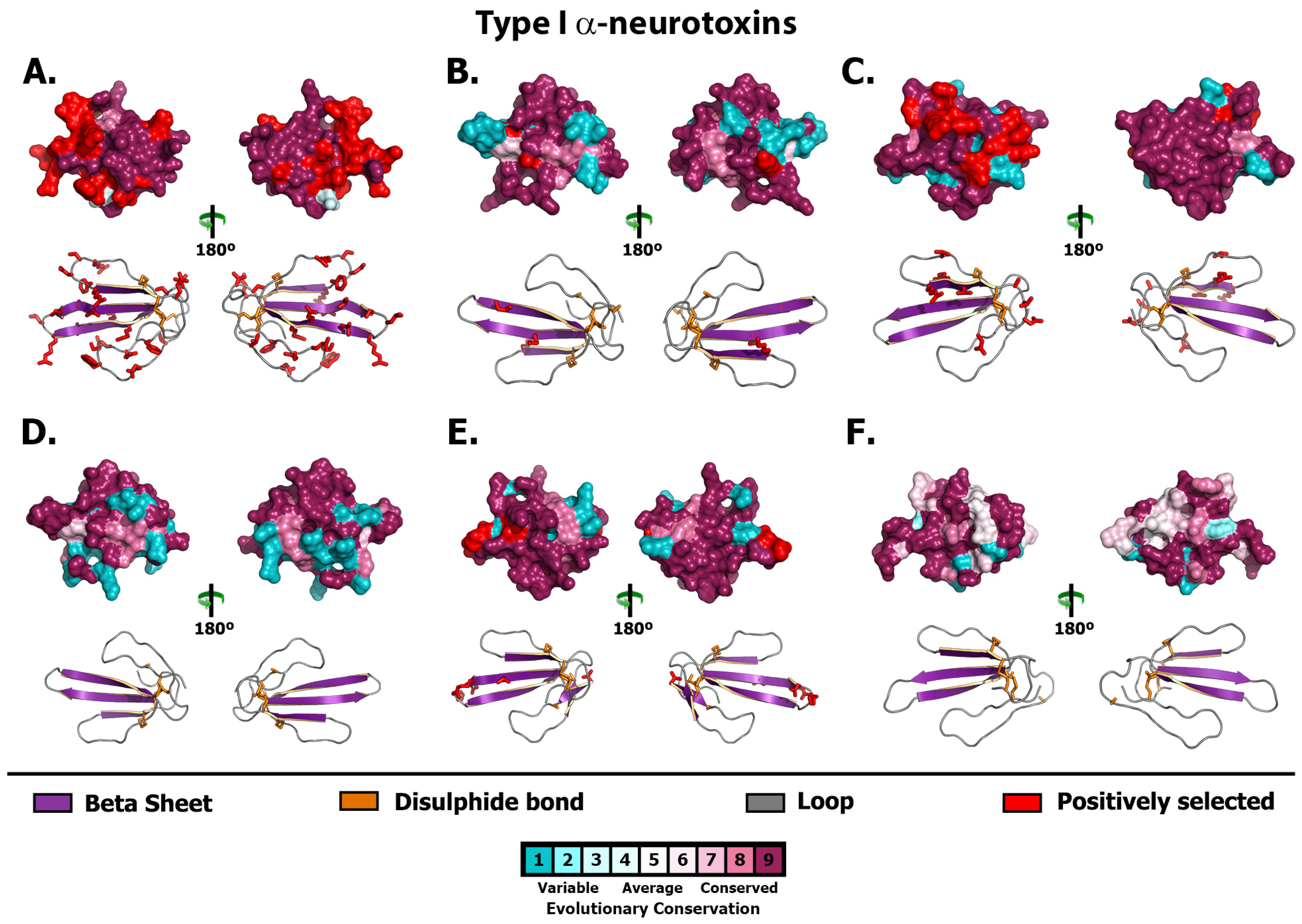
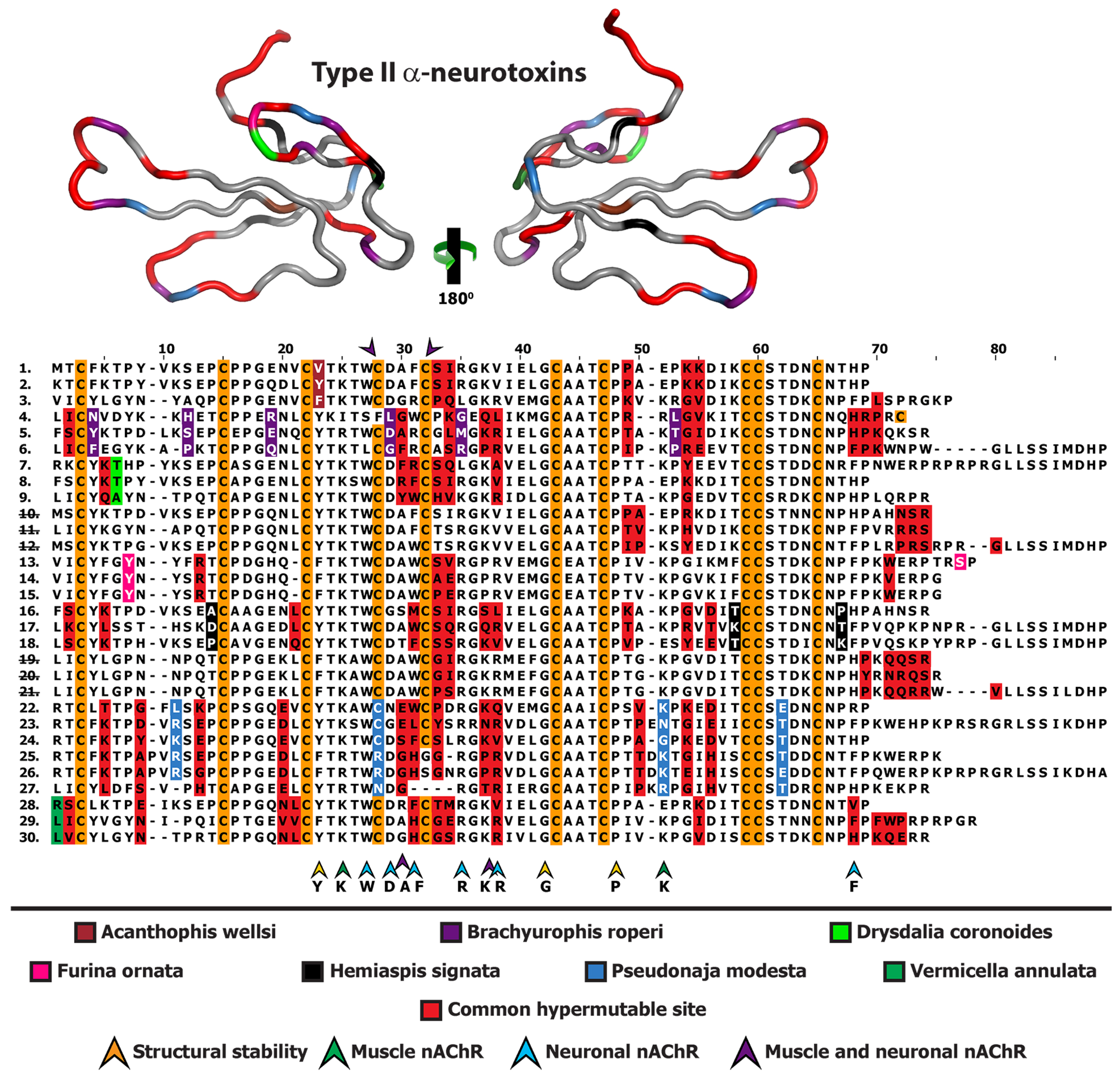

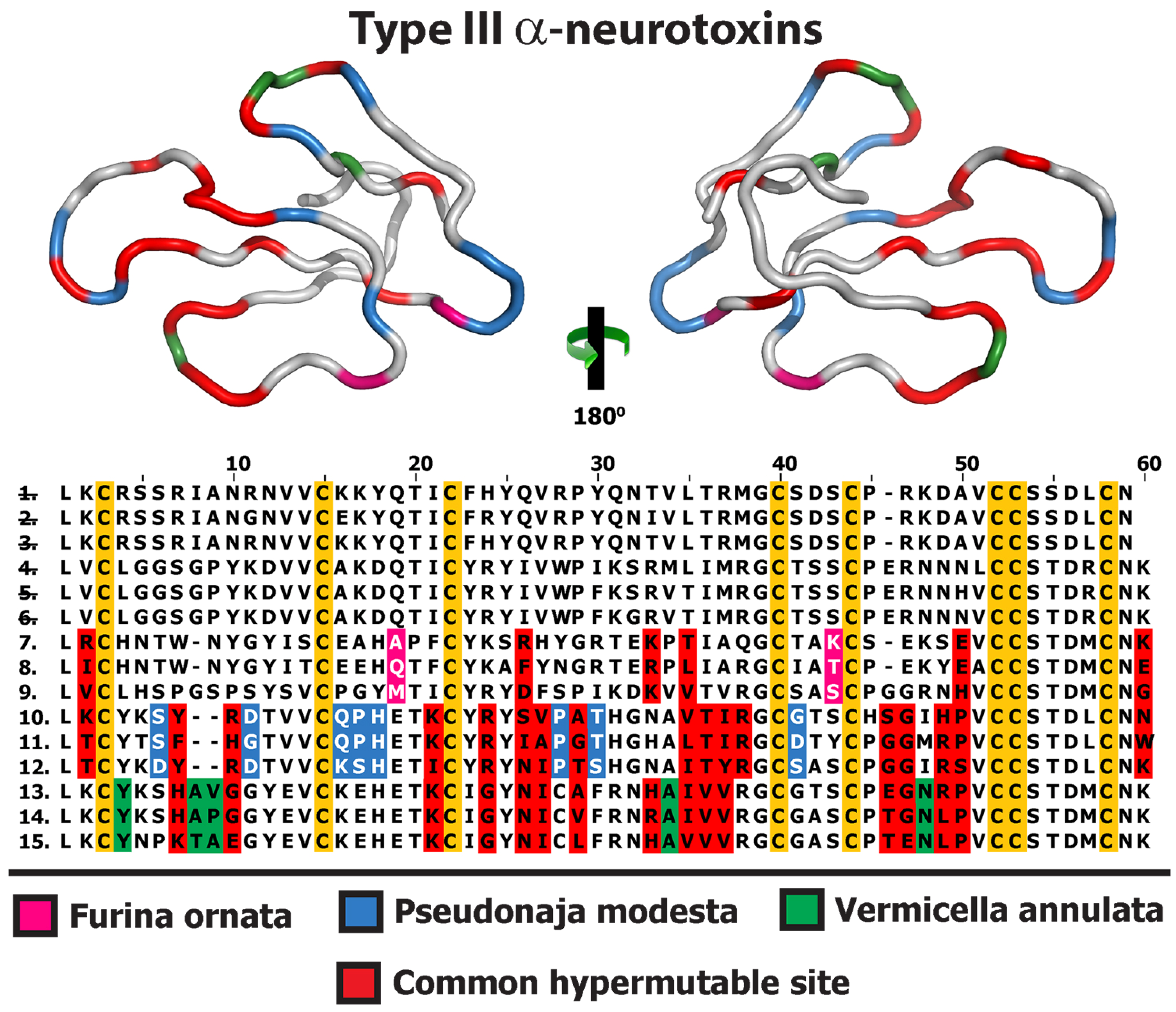
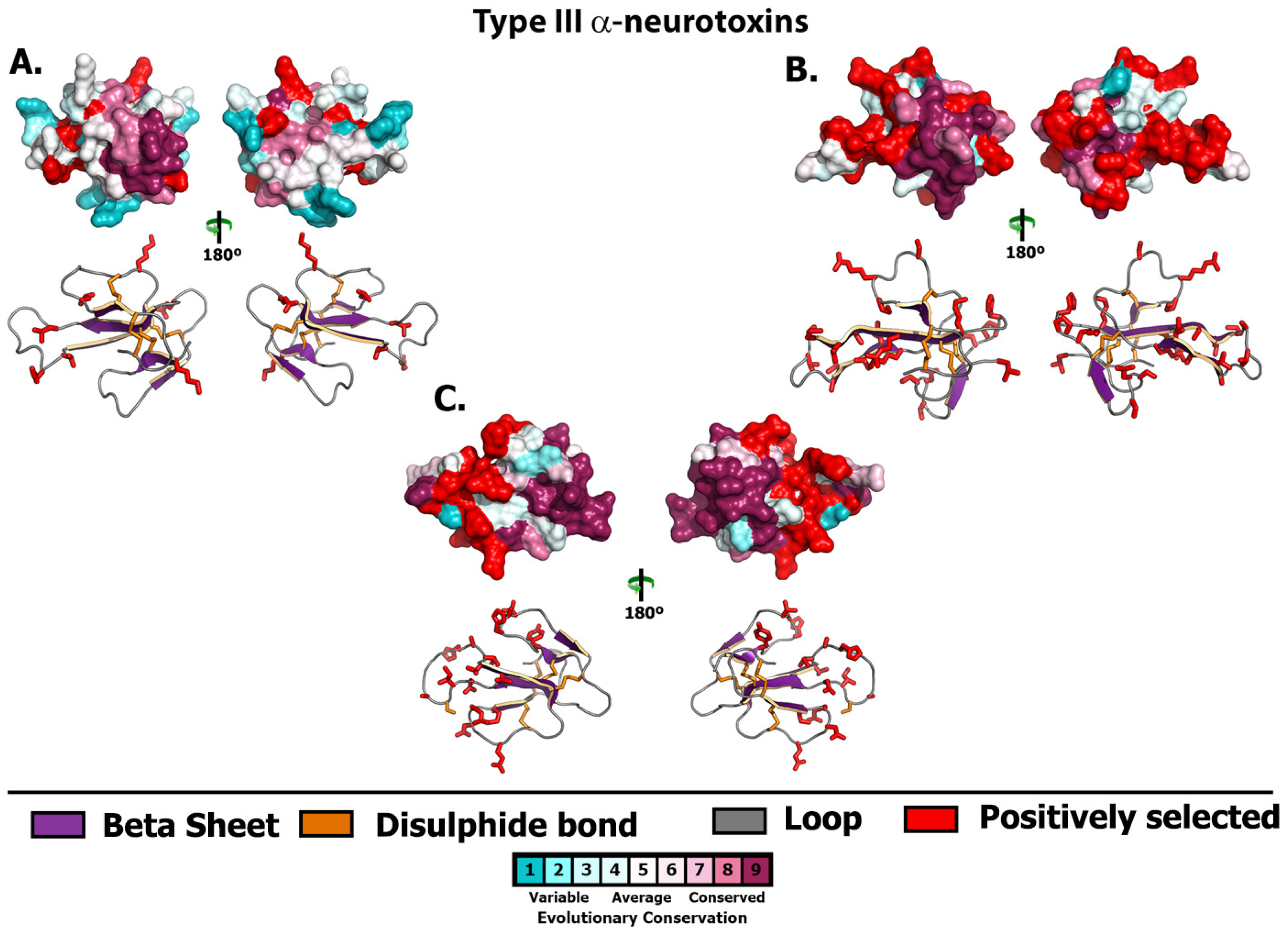
2.2. Lectin
2.3. Natriuretic Peptides
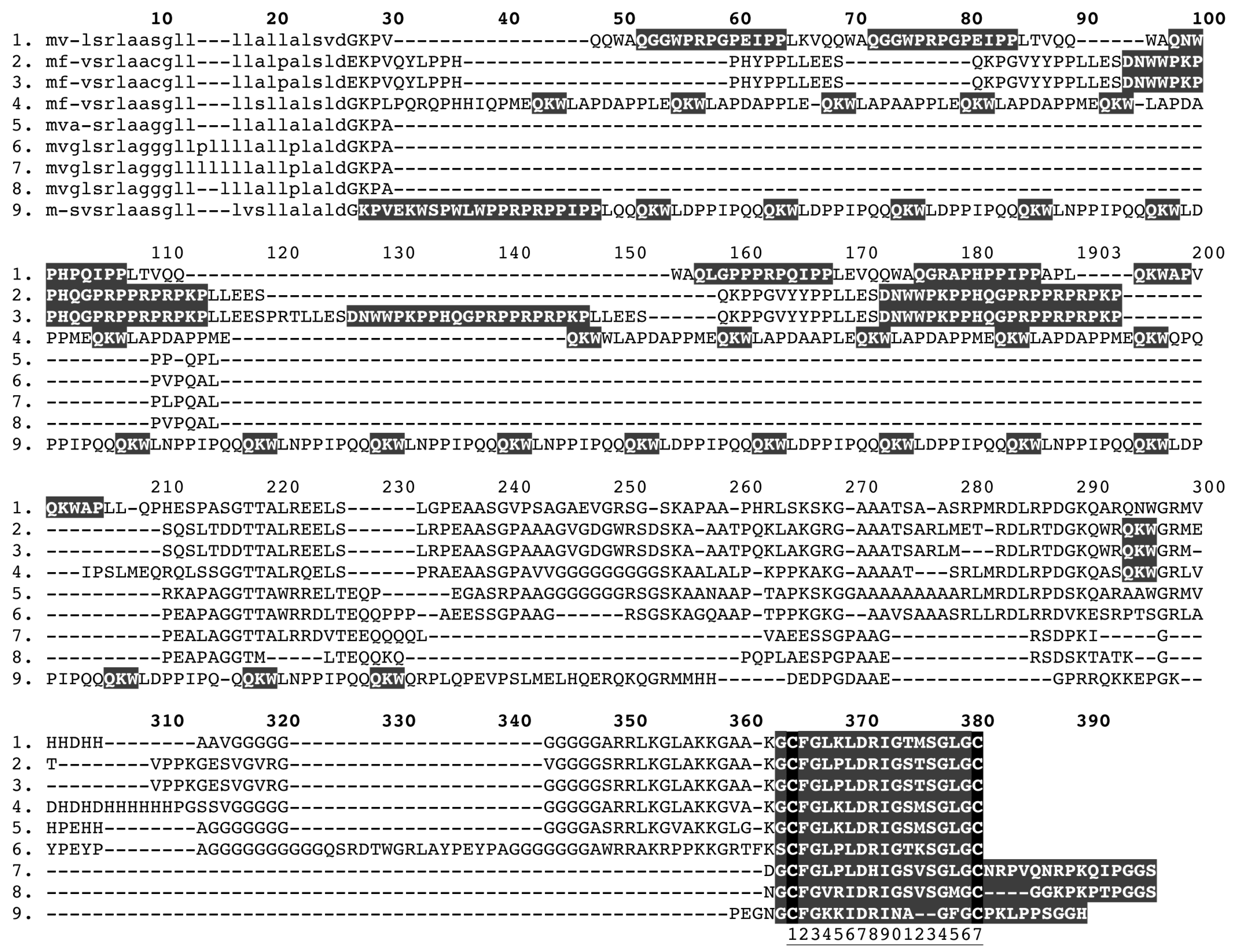
2.4. PLA2
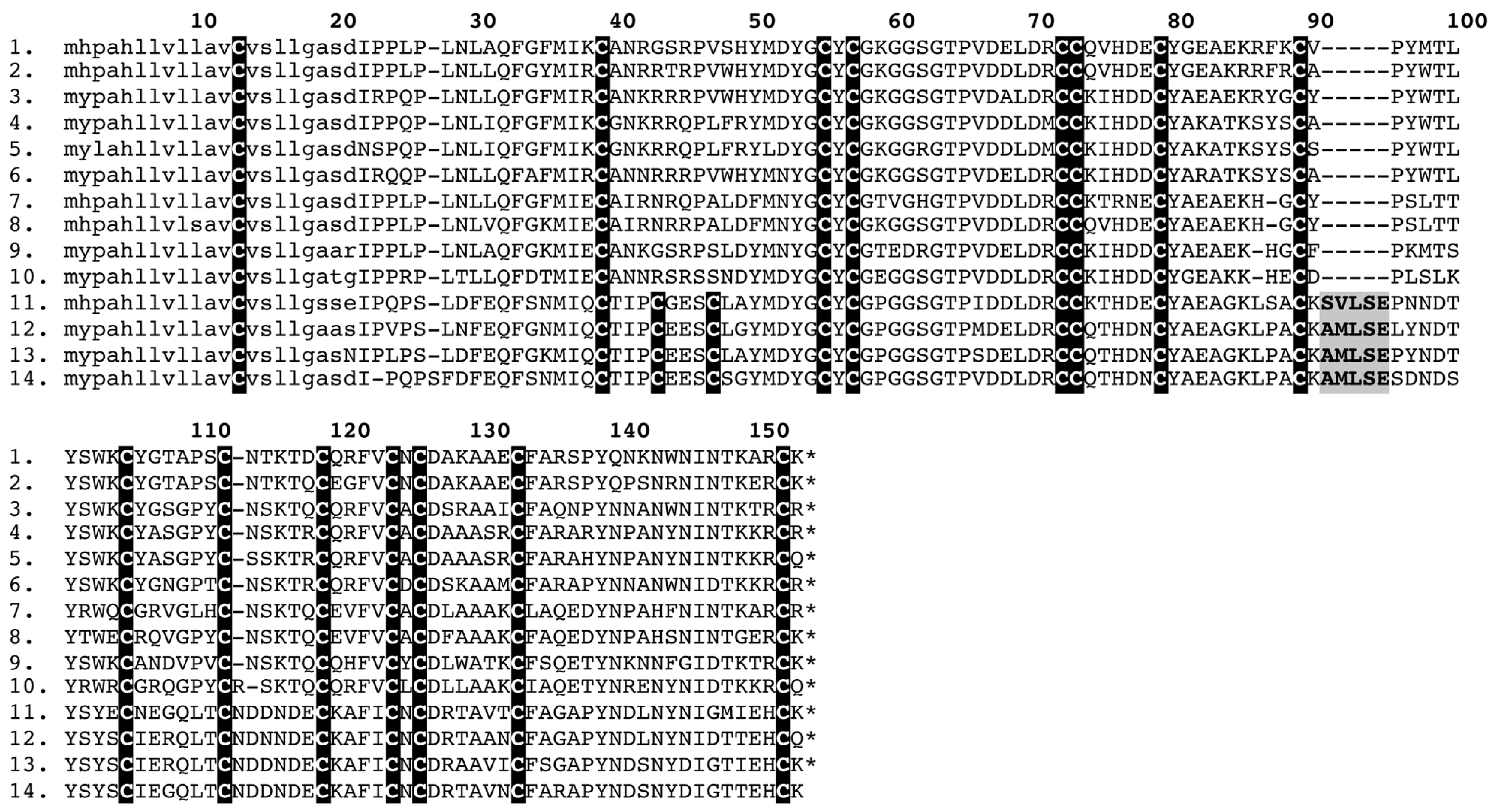
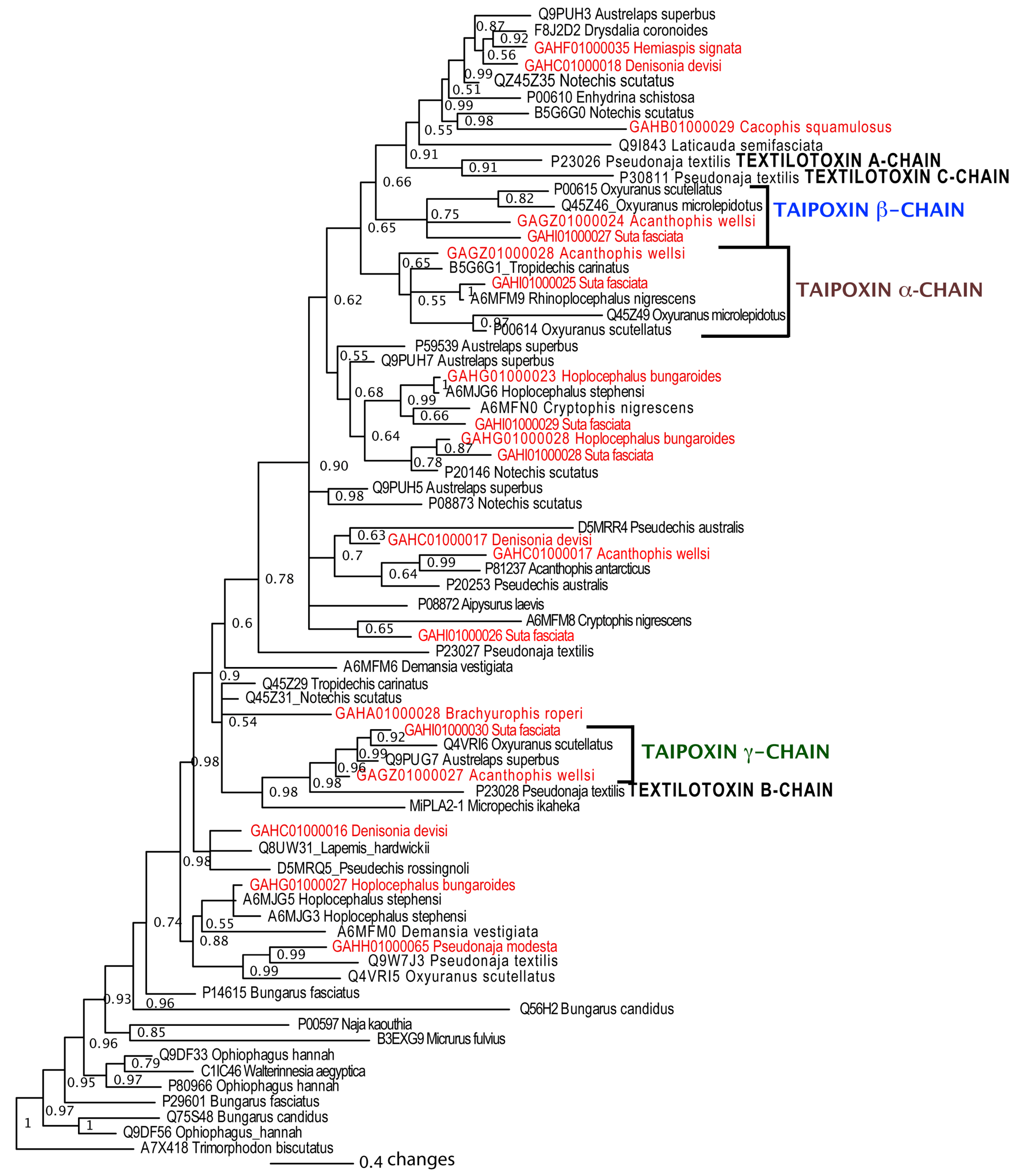
2.5. Kunitz and Waprin

2.6. Procoagulant Toxins and Clinical Implications
2.7. Influence of Prey Preference on Venom Gland Transcriptome
2.8. Brachyurophis roperi: A Unique, Oophagous Burrowing Elapid Snake
3. Experimental Section
3.1. Study Species
3.2. Transcriptome Sequencing
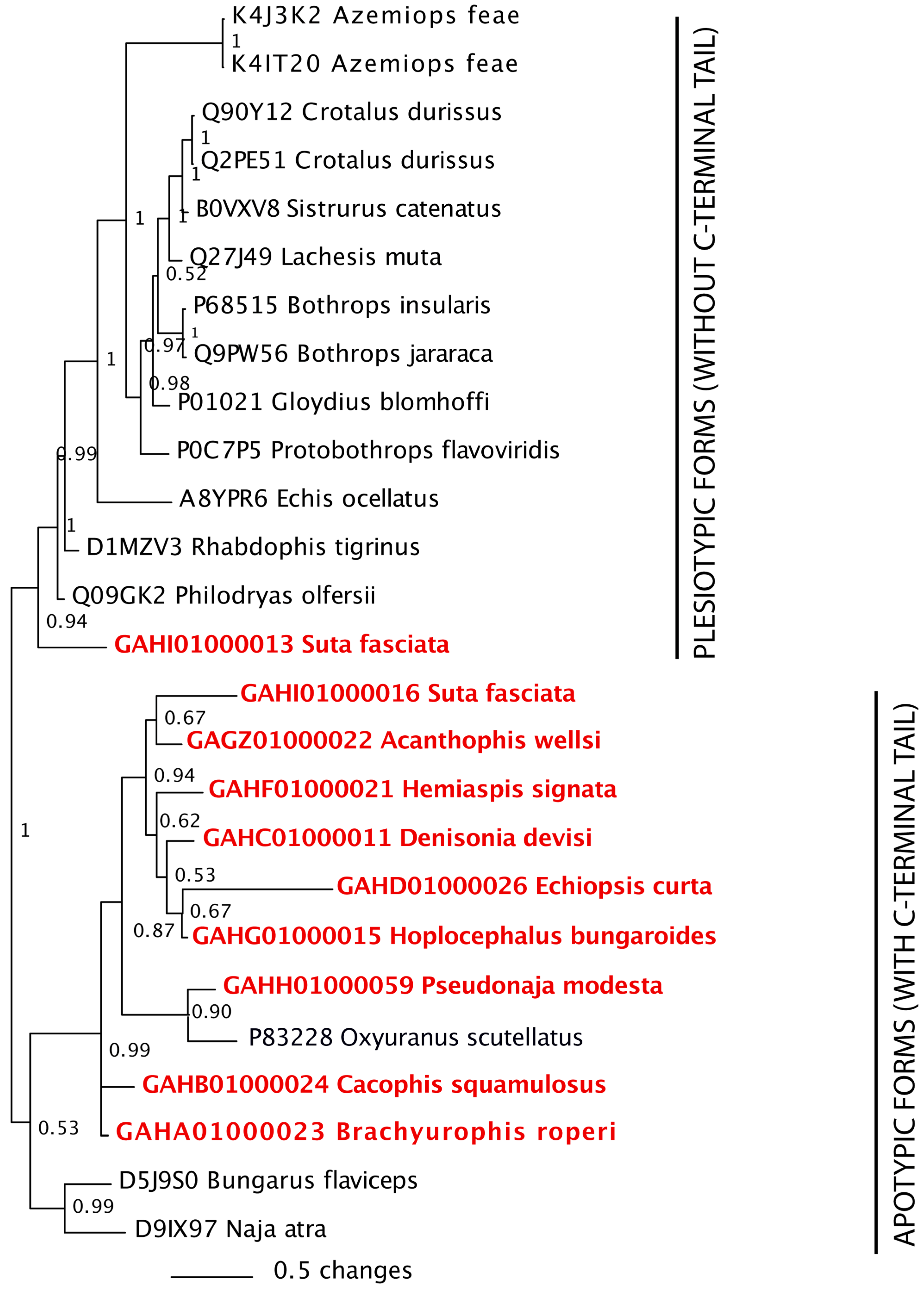
3.3. Phylogenetics
3.4. Selection Analyses
3.5. Structural Analyses
4. Conclusion
Acknowledgements
References
- Fry, B.G. From genome to "venome": molecular origin and evolution of the snake venom proteome inferred from phylogenetic analysis of toxin sequences and related body proteins. Genome Res. 2005, 15((3)), 403–420. [Google Scholar] [CrossRef]
- Nisani, Z.; Boskovic, D.S.; Dunbar, S.G.; Kelln, W.; Hayes, W.K. Investigating the chemical profile of regenerated scorpion (Parabuthus transvaalicus) venom in relation to metabolic cost and toxicity. Toxicon 2012, 60((3)), 315–323. [Google Scholar] [CrossRef]
- Fry, B.G.; Roelants, K.; Champagne, D.E.; Scheib, H.; Tyndall, J.D.; King, G.F.; Nevalainen, T.J.; Norman, J.A.; Lewis, R.J.; Norton, R.S.; Renjifo, C.; de la Vega, R.C. The toxicogenomic multiverse: convergent recruitment of proteins into animal venoms. Annu. Rev. Genomics Hum. Genet. 2009, 10, 483–511. [Google Scholar] [CrossRef]
- Casewell, N.R.; Wuster, W.; Vonk, F.J.; Harrison, R.A.; Fry, B.G. Complex cocktails: the evolutionary novelty of venoms. Trends Ecol. Evol. 2013, 28((4)), 219–229. [Google Scholar] [CrossRef]
- Earl, S.T.; Masci, P.P.; de Jersey, J.; Lavin, M.F.; Dixon, J. Drug development from Australian elapid snake venoms and the Venomics pipeline of candidates for haemostasis: Textilinin-1 (Q8008), Haempatch (Q8009) and CoVase (V0801). Toxicon 2012, 59((4)), 456–463. [Google Scholar] [CrossRef]
- Vetter, I.; Davis, J.L.; Rash, L.D.; Anangi, R.; Mobli, M.; Alewood, P.F.; Lewis, R.J.; King, G.F. Venomics: a new paradigm for natural products-based drug discovery. Amino Acids 2011, 40((1)), 15–28. [Google Scholar] [CrossRef]
- Vink, S.; Jin, A.H.; Poth, K.J.; Head, G.A.; Alewood, P.F. Natriuretic peptide drug leads from snake venom. Toxicon 2012, 59((4)), 434–445. [Google Scholar] [CrossRef]
- Vonk, F.J.; Admiraal, J.F.; Jackson, K.; Reshef, R.; de Bakker, M.A.; Vanderschoot, K.; van den Berge, I.; van Atten, M.; Burgerhout, E.; Beck, A.; Mirtschin, P.J.; Kochva, E.; Witte, F.; Fry, B.G.; Woods, A.E.; Richardson, M.K. Evolutionary origin and development of snake fangs. Nature 2008, 454((7204)), 630–633. [Google Scholar] [CrossRef]
- Wilson, S.; Swan, G. A Complete Guide to Reptiles of Australia, 3rd ed.; Chatswood: New Holland, Australia, 2010. [Google Scholar]
- Sutherland, S.K.; Tibbals, J. Australian Animal Toxins: The Creatures, Their Toxins and Care of the Poisoned Patient, 2nd ed.; Oxford University Press: Melbourne, Australia, 2001. [Google Scholar]
- Pycroft, K.; Fry, B.G.; Isbister, G.K.; Kuruppu, S.; Lawrence, J.; Ian Smith, A.; Hodgson, W.C. Toxinology of venoms from five Australian lesser known elapid snakes. Basic Clin. Pharmacol. Toxicol. 2012, 111((4)), 268–274. [Google Scholar]
- Shine, R. Food-habits and reproductive-biology of Australian elapid snakes of the genus Denisonia. J. Herpetol. 1983, 17((2)), 171–175. [Google Scholar] [CrossRef]
- Birrell, G.W.; Earl, S.T.; Wallis, T.P.; Masci, P.P.; de Jersey, J.; Gorman, J.J.; Lavin, M.F. The diversity of bioactive proteins in Australian snake venoms. Mol. Cell. Proteomics 2007, 6((6)), 973–986. [Google Scholar] [CrossRef]
- Fry, B.G. Structure-function properties of venom components from Australian elapids. Toxicon 1999, 37((1)), 11–32. [Google Scholar] [CrossRef]
- Ching, A.T.; Rocha, M.M.; Paes Leme, A.F.; Pimenta, D.C.; de Fatima, D.F.M.; Serrano, S.M.; Ho, P.L.; Junqueira-de-Azevedo, I.L. Some aspects of the venom proteome of the Colubridae snake Philodryas olfersii revealed from a Duvernoy's (venom) gland transcriptome. FEBS Lett. 2006, 580((18)), 4417–4422. [Google Scholar] [CrossRef]
- Fry, B.G.; Roelants, K.; Norman, J.A. Tentacles of venom: toxic protein convergence in the Kingdom Animalia. J. Mol. Evol. 2009, 68((4)), 311–321. [Google Scholar]
- Fry, B.G.; Roelants, K.; Winter, K.; Hodgson, W.C.; Griesman, L.; Kwok, H.F.; Scanlon, D.; Karas, J.; Shaw, C.; Wong, L.; Norman, J.A. Novel venom proteins produced by differential domain-expression strategies in beaded lizards and gila monsters (genus Heloderma). Mol. Biol. Evol. 2010, 27((2)), 395–407. [Google Scholar] [CrossRef]
- Fry, B.G.; Scheib, H.; de L M Junqueira de Azevedo, I.; Silva, D.A.; Casewell, N.R. Novel transcripts in the maxillary venom glands of advanced snakes. Toxicon 2012, 59((7–8)), 696–708. [Google Scholar] [CrossRef]
- Fry, B.G.; Scheib, H.; van der Weerd, L.; Young, B.; McNaughtan, J.; Ramjan, S.F.; Vidal, N.; Poelmann, R.E.; Norman, J.A. Evolution of an arsenal: structural and functional diversification of the venom system in the advanced snakes (Caenophidia). Mol. Cell. Proteomics 2008, 7((2)), 215–246. [Google Scholar]
- Fry, B.G.; Undheim, E.A.; Ali, S.A.; Jackson, T.N.; Debono, J.; Scheib, H.; Ruder, T.; Morgenstern, D.; Cadwallader, L.; Whitehead, D.; Nabuurs, R.; van der Weerd, L.; Vidal, N.; Roelants, K.; Hendrikx, I.; Gonzalez, S.P.; Koludarov, I.; Jones, A.; King, G.F.; Antunes, A.; Sunagar, K. Squeezers and leaf-cutters: differential diversification and degeneration of the venom system in toxicoferan reptiles. Mol. Cell. Proteomics 2013, 12((7)), 1881–1899. [Google Scholar] [CrossRef]
- Fry, B.G.; Vidal, N.; Norman, J.A.; Vonk, F.J.; Scheib, H.; Ramjan, S.F.; Kuruppu, S.; Fung, K.; Hedges, S.B.; Richardson, M.K.; Hodgson, W.C.; Ignjatovic, V.; Summerhayes, R.; Kochva, E. Early evolution of the venom system in lizards and snakes. Nature 2006, 439((7076)), 584–588. [Google Scholar] [CrossRef]
- Fry, B.G.; Winter, K.; Norman, J.A.; Roelants, K.; Nabuurs, R.J.; van Osch, M.J.; Teeuwisse, W.M.; van der Weerd, L.; McNaughtan, J.E.; Kwok, H.F.; Scheib, H.; Greisman, L.; Kochva, E.; Miller, L.J.; Gao, F.; Karas, J.; Scanlon, D.; Lin, F.; Kuruppu, S.; Shaw, C.; Wong, L.; Hodgson, W.C. Functional and structural diversification of the Anguimorpha lizard venom system. Mol. Cell. Proteomics 2010, 9((11)), 2369–2390. [Google Scholar] [CrossRef]
- Fry, B.G.; Wroe, S.; Teeuwisse, W.; van Osch, M.J.; Moreno, K.; Ingle, J.; McHenry, C.; Ferrara, T.; Clausen, P.; Scheib, H.; Winter, K.L.; Greisman, L.; Roelants, K.; van der Weerd, L.; Clemente, C.J.; Giannakis, E.; Hodgson, W.C.; Luz, S.; Martelli, P.; Krishnasamy, K.; Kochva, E.; Kwok, H.F.; Scanlon, D.; Karas, J.; Citron, D.M.; Goldstein, E.J.; McNaughtan, J.E.; Norman, J.A. A central role for venom in predation by Varanus komodoensis (Komodo Dragon) and the extinct giant Varanus (Megalania) priscus. Proc. Natl. Acad. Sci. USA 2009, 106((22)), 8969–8974. [Google Scholar] [CrossRef]
- Junqueira-de-Azevedo, I.L.; Ching, A.T.; Carvalho, E.; Faria, F.; Nishiyama, M.Y., Jr.; Ho, P.L.; Diniz, M.R. Lachesis muta (Viperidae) cDNAs reveal diverging pit viper molecules and scaffolds typical of cobra (Elapidae) venoms: implications for snake toxin repertoire evolution. Genetics 2006, 173((2)), 877–889. [Google Scholar] [CrossRef]
- Terrat, Y.; Sunagar, K.; Fry, B.G.; Jackson, T.N.; Scheib, H.; Fourmy, R.; Verdenaud, M.; Blanchet, G.; Antunes, A.; Ducancel, F. Atractaspis aterrima Toxins: The First Insight into the Molecular Evolution of Venom in Side-Stabbers. Toxins 2013, 5((11)), 1948–1964. [Google Scholar] [CrossRef]
- Wagstaff, S.C.; Harrison, R.A. Venom gland EST analysis of the saw-scaled viper, Echis ocellatus, reveals novel alpha9beta1 integrin-binding motifs in venom metalloproteinases and a new group of putative toxins, renin-like aspartic proteas. Gene 2006, 377, 21–32. [Google Scholar]
- Fry, B.G.; Wuster, W.; Kini, R.M.; Brusic, V.; Khan, A.; Venkataraman, D.; Rooney, A.P. Molecular evolution and phylogeny of elapid snake venom three-finger toxins. J. Mol. Evol. 2003, 57((1)), 110–129. [Google Scholar] [CrossRef]
- St Pierre, L.; Fischer, H.; Adams, D.J.; Schenning, M.; Lavidis, N.; de Jersey, J.; Masci, P.P.; Lavin, M.F. Distinct activities of novel neurotoxins from Australian venomous snakes for nicotinic acetylcholine receptors. Cell. Mol. Life Sci. 2007, 64((21)), 2829–2840. [Google Scholar]
- Sunagar, K.; Jackson, T.N.; Undheim, E.A.; Ali, S.A.; Antunes, A.; Fry, B.G. Three-Fingered RAVERs: Rapid Accumulation of Variations in Exposed Residues of Snake Venom Toxins. Toxins 2013, 5((11)), 2172–2208. [Google Scholar] [CrossRef]
- Gong, N.; Armugam, A.; Jeyaseelan, K. Postsynaptic short-chain neurotoxins from Pseudonaja textilis. cDNA cloning, expression and protein characterization. Eur. J. Biochem. 1999, 265((3)), 982–989. [Google Scholar]
- Gong, N.; Armugam, A.; Jeyaseelan, K. Molecular cloning, characterization and evolution of the gene encoding a new group of short-chain alpha-neurotoxins in an Australian elapid, Pseudonaja textilis. FEBS Lett. 2000, 473((3)), 303–310. [Google Scholar] [CrossRef]
- Brust, A.; Sunagar, K.; Undheim, E.A.; Vetter, I.; Yang, D.C.; Casewell, N.R.; Jackson, T.N.; Koludarov, I.; Alewood, P.F.; Hodgson, W.C.; Lewis, R.J.; King, G.F.; Antunes, A.; Hendrikx, I.; Fry, B.G. Differential evolution and neofunctionalization of snake venom metalloprotease domains. Mol. Cell. Proteomics 2013, 12((3)), 651–663. [Google Scholar] [CrossRef]
- Koludarov, I.; Sunagar, K.; Undheim, E.A.; Jackson, T.N.; Ruder, T.; Whitehead, D.; Saucedo, A.C.; Mora, G.R.; Alagon, A.C.; King, G.; Antunes, A.; Fry, B.G. Structural and molecular diversification of the Anguimorpha lizard mandibular venom gland system in the arboreal species Abronia graminea. J. Mol. Evol. 2012, 75((5–6)), 168–183. [Google Scholar] [CrossRef]
- Kozminsky-Atias, A.; Zilberberg, N. Molding the business end of neurotoxins by diversifying evolution. FASEB J. 2012, 26((2)), 576–586. [Google Scholar] [CrossRef]
- Low, D.H.; Sunagar, K.; Undheim, E.A.; Ali, S.A.; Alagon, A.C.; Ruder, T.; Jackson, T.N.; Pineda Gonzalez, S.; King, G.F.; Jones, A.; Antunes, A.; Fry, B.G. Dracula's children: Molecular evolution of vampire bat venom. J. Proteomics 2013, 89, 95–111. [Google Scholar]
- Ruder, T.; Sunagar, K.; Undheim, E.A.; Ali, S.A.; Wai, T.C.; Low, D.H.; Jackson, T.N.; King, G.F.; Antunes, A.; Fry, B.G. Molecular phylogeny and evolution of the proteins encoded by coleoid (cuttlefish, octopus, and squid) posterior venom glands. J. Mol. Evol. 2013, 76((4)), 192–204. [Google Scholar] [CrossRef]
- Sunagar, K.; Johnson, W.E.; O'Brien, S.J.; Vasconcelos, V.; Antunes, A. Evolution of CRISPs associated with toxicoferan-reptilian venom and mammalian reproduction. Mol. Biol. Evol. 2012, 29((7)), 1807–1822. [Google Scholar] [CrossRef]
- Sunagar, K.; Fry, B.G.; Jackson, T.N.W.; Casewell, N.R.; Undheim, E.A.B.; Vidal, N.; Ali, S.A.; King, G.F.; Vasudevan, K.; Vasconcelos, V.; Antunes, A. Molecular Evolution of Vertebrate Neurotrophins: Co-Option of the Highly Conserved Nerve Growth Factor Gene into the Advanced Snake Venom Arsenal. PLoS One 2013, 8((11)), e81827. [Google Scholar]
- Tian, C.; Yuan, Y.; Zhu, S. Positively selected sites of scorpion depressant toxins: possible roles in toxin functional divergence. Toxicon 2008, 51((4)), 555–562. [Google Scholar] [CrossRef]
- Zhu, S.; Bosmans, F.; Tytgat, J. Adaptive evolution of scorpion sodium channel toxins. J. Mol. Evol. 2004, 58((2)), 145–153. [Google Scholar] [CrossRef]
- Xu, Q.; Wu, X.F.; Xia, Q.C.; Wang, K.Y. Cloning of a galactose-binding lectin from the venom of Trimeresurus stejnegeri. Biochem. J. 1999, 341((Pt. 3)), 733–737. [Google Scholar]
- Earl, S.T.; Robson, J.; Trabi, M.; de Jersey, J.; Masci, P.P.; Lavin, M.F. Characterisation of a mannose-binding C-type lectin from Oxyuranus scutellatus snake venom. Biochimie 2011, 93((3)), 519–527. [Google Scholar]
- Fry, B.G.; Wickramaratana, J.C.; Lemme, S.; Beuve, A.; Garbers, D.; Hodgson, W.C.; Alewood, P. Novel natriuretic peptides from the venom of the inland taipan (Oxyuranus microlepidotus): isolation, chemical and biological characterisation. Biochem. Biophys. Res. Commun. 2005, 327((4)), 1011–1015. [Google Scholar]
- Schweitz, H.; Vigne, P.; Moinier, D.; Frelin, C.; Lazdunski, M. A new member of the natriuretic peptide family is present in the venom of the green mamba (Dendroaspis angusticeps). J. Biol. Chem. 1992, 267((20)), 13928–13932. [Google Scholar]
- Camargo, A.C.; Ianzer, D.; Guerreiro, J.R.; Serrano, S.M. Bradykinin-potentiating peptides: beyond captopril. Toxicon 2012, 59((4)), 516–523. [Google Scholar] [CrossRef]
- Vonk, F.J.; Jackson, K.; Doley, R.; Madaras, F.; Mirtschin, P.J.; Vidal, N. Snake venom: From fieldwork to the clinic: Recent insights into snake biology, together with new technology allowing high-throughput screening of venom, bring new hope for drug discovery. Bioessays 2011, 33((4)), 269–279. [Google Scholar] [CrossRef]
- St Pierre, L.; Flight, S.; Masci, P.P.; Hanchard, K.J.; Lewis, R.J.; Alewood, P.F.; de Jersey, J.; Lavin, M.F. Cloning and characterisation of natriuretic peptides from the venom glands of Australian elapids. Biochimie 2006, 88((12)), 1923–1931. [Google Scholar]
- St Pierre, L.; Woods, R.; Earl, S.; Masci, P.P.; Lavin, M.F. Identification and analysis of venom gland-specific genes from the coastal taipan (Oxyuranus scutellatus) and related species. Cell. Mol. Life Sci. 2005, 62((22)), 2679–2693. [Google Scholar] [CrossRef]
- Akashi, Y.J.; Springer, J.; Lainscak, M.; Anker, S.D. Atrial natriuretic peptide and related peptides. Clin. Chem. Lab. Med. 2007, 45((10)), 1259–1267. [Google Scholar]
- Amininasab, M.; Elmi, M.M.; Endlich, N.; Endlich, K.; Parekh, N.; Naderi-Manesh, H.; Schaller, J.; Mostafavi, H.; Sattler, M.; Sarbolouki, M.N.; Muhle-Goll, C. Functional and structural characterization of a novel member of the natriuretic family of peptides from the venom of Pseudocerastes persicus. FEBS Lett. 2004, 557((1–3)), 104–108. [Google Scholar] [CrossRef]
- Fry, B.G.; Wuster, W. Assembling an arsenal: origin and evolution of the snake venom proteome inferred from phylogenetic analysis of toxin sequences. Mol. Biol. Evol. 2004, 21((5)), 870–883. [Google Scholar]
- Chen, H.H.; Lainchbury, J.G.; Burnett, J.C., Jr. Natriuretic peptide receptors and neutral endopeptidase in mediating the renal actions of a new therapeutic synthetic natriuretic peptide dendroaspis natriuretic peptide. J. Am. Coll. Cardiol. 2002, 40((6)), 1186–1191. [Google Scholar] [CrossRef]
- Da Silva, S.L.; Dias-Junior, C.A.; Baldasso, P.A.; Damico, D.C.; Carvalho, B.M.; Garanto, A.; Acosta, G.; Oliveira, E.; Albericio, F.; Soares, A.M.; Marangoni, S.; Resende, R.R. Vascular effects and electrolyte homeostasis of the natriuretic peptide isolated from Crotalus oreganus abyssus (North American Grand Canyon rattlesnake) venom. Peptides 2012, 36((2)), 206–212. [Google Scholar] [CrossRef]
- Quinton, L.; Gilles, N.; Smargiasso, N.; Kiehne, A.; De Pauw, E. An unusual family of glycosylated peptides isolated from Dendroaspis angusticeps venom and characterized by combination of collision induced and electron transfer dissociation. J. Am. Soc. Mass Spectrom. 2011, 22((11)), 1891–1897. [Google Scholar] [CrossRef]
- Utkin, Y.N.; Weise, C.; Kasheverov, I.E.; Andreeva, T.V.; Kryukova, E.V.; Zhmak, M.N.; Starkov, V.G.; Hoang, N.A.; Bertrand, D.; Ramerstorfer, J.; Sieghart, W.; Thompson, A.J.; Lummis, S.C.; Tsetlin, V.I. Azemiopsin from Azemiops feae viper venom, a novel polypeptide ligand of nicotinic acetylcholine receptor. J. Biol. Chem. 2012, 287((32)), 27079–27086. [Google Scholar]
- Kuruppu, S.; Fry, B.G.; Hodgson, W.C. Presynaptic neuromuscular activity of venom from the brown-headed snake (Glyphodon tristis). Toxicon 2005, 45((3)), 383–388. [Google Scholar] [CrossRef]
- Fry, B.G.; Wuster, W.; Ryan Ramjan, S.F.; Jackson, T.; Martelli, P.; Kini, R.M. Analysis of Colubroidea snake venoms by liquid chromatography with mass spectrometry: evolutionary and toxinological implications. Rapid Commun. Mass Spectrom. 2003, 17((18)), 2047–2062. [Google Scholar] [CrossRef]
- Kuruppu, S.; Robinson, S.; Hodgson, W.C.; Fry, B.G. The in vitro neurotoxic and myotoxic effects of the venom from the Suta genus (curl snakes) of elapid snakes. Basic Clin. Pharmacol. Toxicol. 2007, 101((6)), 407–410. [Google Scholar] [CrossRef]
- Fohlman, J.; Lind, P.; Eaker, D. Taipoxin, an extremely potent presynaptic snake venom neurotoxin. Elucidation of the primary structure of the acidic carbohydrate-containing taipoxin-subunit, a prophospholipase homolog. FEBS Lett. 1977, 84((2)), 367–371. [Google Scholar]
- Blacklow, B.; Escoubas, P.; Nicholson, G.M. Characterisation of the heterotrimeric presynaptic phospholipase A(2) neurotoxin complex from the venom of the common death adder (Acanthophis antarcticus). Biochem. Pharmacol. 2010, 80((2)), 277–287. [Google Scholar] [CrossRef]
- Blacklow, B.; Konstantakopoulos, N.; Hodgson, W.C.; Nicholson, G.M. Presence of presynaptic neurotoxin complexes in the venoms of Australo-Papuan death adders (Acanthophis spp.). Toxicon 2010, 55((6)), 1171–1180. [Google Scholar] [CrossRef]
- Chaisakul, J.; Konstantakopoulos, N.; Smith, A.I.; Hodgson, W.C. Isolation and characterisation of P-EPTX-Ap1a and P-EPTX-Ar1a: pre-synaptic neurotoxins from the venom of the northern (Acanthophis praelongus) and Irian Jayan (Acanthophis rugosus) death adders. Biochem. Pharmacol. 2010, 80((6)), 895–902. [Google Scholar]
- Chaisakul, J.; Parkington, H.C.; Isbister, G.K.; Konstantakopoulos, N.; Hodgson, W.C. Differential myotoxic and cytotoxic activities of pre-synaptic neurotoxins from Papuan taipan (Oxyuranus scutellatus) and Irian Jayan death adder (Acanthophis rugosus) venoms. Basic Clin. Pharmacol. Toxicol. 2013, 112((5)), 325–334. [Google Scholar] [CrossRef]
- Fry, B.G.; Wickramaratna, J.C.; Hodgson, W.C.; Alewood, P.F.; Kini, R.M.; Ho, H.; Wuster, W. Electrospray liquid chromatography/mass spectrometry fingerprinting of Acanthophis (death adder) venoms: taxonomic and toxinological implications. Rapid Commun. Mass Spectrom. 2002, 16((6)), 600–608. [Google Scholar]
- Doley, R.; Tram, N.N.; Reza, M.A.; Kini, R.M. Unusual accelerated rate of deletions and insertions in toxin genes in the venom glands of the pygmy copperhead (Austrelaps labialis) from Kangaroo island. BMC Evol. Biol. 2008, 8, 70. [Google Scholar] [CrossRef]
- Marlor, C.W.; Delaria, K.A.; Davis, G.; Muller, D.K.; Greve, J.M.; Tamburini, P.P. Identification and cloning of human placental bikunin, a novel serine protease inhibitor containing two Kunitz domains. J. Biol. Chem. 1997, 272((18)), 12202–12208. [Google Scholar]
- Doley, R.; Pahari, S.; Reza, M.A.; Mackessy, S.P.; Kini, R.M. The Gene Structure and Evolution of ku-wap-fusin (Kunitz Waprin Fusion Protein), a Novel Evolutionary Intermediate of the Kunitz Serine Protease Inhibitors and Waprins from Sistrurus catenatus (Massasauga Rattlesnake) Venom Glands. Open Evol.J. 2010, 4, 31–41. [Google Scholar]
- St Pierre, L.; Earl, S.T.; Filippovich, I.; Sorokina, N.; Masci, P.P.; De Jersey, J.; Lavin, M.F. Common evolution of waprin and kunitz-like toxin families in Australian venomous snakes. Cell. Mol. Life Sci. 2008, 65((24)), 4039–4054. [Google Scholar] [CrossRef]
- Francischetti, I.M.; My-Pham, V.; Harrison, J.; Garfield, M.K.; Ribeiro, J.M. Bitis. gabonica (Gaboon viper) snake venom gland: toward a catalog for the full-length transcripts (cDNA) and proteins. Gene 2004, 337, 55–69. [Google Scholar]
- Nair, D.G.; Fry, B.G.; Alewood, P.; Kumar, P.P.; Kini, R.M. Antimicrobial activity of omwaprin, a new member of the waprin family of snake venom proteins. Biochem. J. 2007, 402((1)), 93–104. [Google Scholar] [CrossRef]
- Isbister, G.K.; Dawson, A.H.; Whyte, I.M. Two cases of bites by the black-bellied swamp snake (Hemiaspis signata). Toxicon 2002, 40((3)), 317–319. [Google Scholar] [CrossRef]
- Isbister, G.K.; White, J.; Currie, B.J.; O'Leary, M.A.; Brown, S.G. Clinical effects and treatment of envenoming by Hoplocephalus spp. snakes in Australia: Australian Snakebite Project (ASP-12). Toxicon 2011, 58((8)), 634–640. [Google Scholar] [CrossRef]
- White, J.; Williams, V.; Passehl, J.H. The five-ringed brown snake, Pseudonaja modesta (Gunther): report of a bite and comments on its venom. Med. J. Aust. 1987, 147((11–12)), 603–605. [Google Scholar]
- Reza, M.A.; Minh Le, T.N.; Swarup, S.; Manjunatha Kini, R. Molecular evolution caught in action: gene duplication and evolution of molecular isoforms of prothrombin activators in Pseudonaja textilis (brown snake). J. Thromb. Haemost. 2006, 4((6)), 1346–1353. [Google Scholar]
- Sanders, K.L.; Lee, M.S.; Leys, R.; Foster, R.; Keogh, J.S. Molecular phylogeny and divergence dates for Australasian elapids and sea snakes (Hydrophiinae): evidence from seven genes for rapid evolutionary radiations. J. Evol. Biol. 2008, 21((3)), 682–695. [Google Scholar]
- Isbister, G.K.; Scorgie, F.E.; O'Leary, M.A.; Seldon, M.; Brown, S.G.; Lincz, L.F. Factor deficiencies in venom-induced consumption coagulopathy resulting from Australian elapid envenomation: Australian Snakebite Project (ASP-10). J. Thromb. Haemost. 2010, 8((11)), 2504–2513. [Google Scholar] [CrossRef]
- Fry, B.G.; Casewell, N.R.; Wuster, W.; Vidal, N.; Young, B.; Jackson, T.N. The structural and functional diversification of the Toxicofera reptile venom system. Toxicon 2012, 60((4)), 434–448. [Google Scholar]
- Filippovich, I.; Sorokina, N.; St Pierre, L.; Flight, S.; de Jersey, J.; Perry, N.; Masci, P.P.; Lavin, M.F. Cloning and functional expression of venom prothrombin activator protease from Pseudonaja textilis with whole blood procoagulant activity. Br. J. Haematol. 2005, 131((2)), 237–246. [Google Scholar]
- Bos, M.H.; Boltz, M.; St Pierre, L.; Masci, P.P.; de Jersey, J.; Lavin, M.F.; Camire, R.M. Venom factor V from the common brown snake escapes hemostatic regulation through procoagulant adaptations. Blood 2009, 114((3)). [Google Scholar]
- Shine, R. Constraints, allometry, and adaptation-food-habits and reproductive-biology of australian brownsnakes (Pseudonaja, Elapidae). Herpetologica 1989, 45((2)), 195–207. [Google Scholar]
- Kulkeaw, K.; Chaicumpa, W.; Sakolvaree, Y.; Tongtawe, P.; Tapchaisri, P. Proteome and immunome of the venom of the Thai cobra, Naja kaouthia. Toxicon 2007, 49((7)), 1026–1041. [Google Scholar] [CrossRef]
- Daltry, J.C.; Wuster, W.; Thorpe, R.S. Diet and snake venom evolution. Nature 1996, 379((6565)), 537–540. [Google Scholar] [CrossRef]
- Gibbs, H.L.; Sanz, L.; Sovic, M.G.; Calvete, J.J. Phylogeny-based comparative analysis of venom proteome variation in a clade of rattlesnakes (Sistrurus sp.). PLoS One 2013, 8((6)), e67220. [Google Scholar]
- Pawlak, J.; Mackessy, S.P.; Fry, B.G.; Bhatia, M.; Mourier, G.; Fruchart-Gaillard, C.; Servent, D.; Menez, R.; Stura, E.; Menez, A.; Kini, R.M. Denmotoxin, a three-finger toxin from the colubrid snake Boiga dendrophila (Mangrove Catsnake) with bird-specific activity. J. Biol. Chem. 2006, 281((39)), 29030–29041. [Google Scholar] [CrossRef]
- Shine, R. Reproduction, feeding and growth in the australian burrowing snake Vermicella annulata. J. Herpetol. 1980, 14((1)), 71–77. [Google Scholar] [CrossRef]
- Durban, J.; Perez, A.; Sanz, L.; Gomez, A.; Bonilla, F.; Rodriguez, S.; Chacon, D.; Sasa, M.; Angulo, Y.; Gutierrez, J.M.; Calvete, J.J. Integrated "omics" profiling indicates that miRNAs are modulators of the ontogenetic venom composition shift in the Central American rattlesnake, Crotalus simus simus. BMC Genomics 2013, 14, 234. [Google Scholar] [CrossRef]
- Li, M.; Fry, B.G.; Kini, R.M. Eggs-only diet: its implications for the toxin profile changes and ecology of the marbled sea snake (Aipysurus eydouxii). J. Mol. Evol. 2005, 60((1)), 81–89. [Google Scholar] [CrossRef]
- Li, M.; Fry, B.G.; Kini, R.M. Putting the brakes on snake venom evolution: the unique molecular evolutionary patterns of Aipysurus eydouxii (Marbled sea snake) phospholipase A2 toxins. Mol. Biol. Evol. 2005, 22((4)), 934–941. [Google Scholar] [CrossRef]
- Scanlon, J.D.; Shine, R. Dentition and diet in snakes - adaptations to oophagy in the Australian elapid genus Simoselaps. J. Zool. 1988, 216, 519–528. [Google Scholar] [CrossRef]
- Shine, R. Comparative ecology of 3 australian snake species of the genus Cacophis (Serpentes, Elapidae). Copeia 1980, 4, 831–838. [Google Scholar] [CrossRef]
- Shine, R. Ecology of Australian elapid snakes of the genera Furina and Glyphodon. J. Herpetol. 1981, 15((2)), 219–224. [Google Scholar] [CrossRef]
- Shine, R. Arboreality in snakes: ecology of the Australian elapid genus Hoplocephalus. Copeia 1983, 1983((1)), 198–205. [Google Scholar] [CrossRef]
- Shine, R. Reproductive-biology and food-habits of the Australian elapid snakes of the genus Cryptophis. J. Herpetol. 1984, 18((1)), 33–39. [Google Scholar]
- Shine, R. Food-habits and reproductive-biology of australian snakes of the genus Hemiaspis (Elapidae). J. Herpetol. 1987, 21((1)), 71–74. [Google Scholar]
- Shine, R. Food-habits and reproductive-biology of small australian snakes of the genera Unechis and Suta (elapidae). J. Herpetol. 1988, 22((3)), 307–315. [Google Scholar]
- Goldman, N.; Yang, Z. A codon-based model of nucleotide substitution for protein-coding DNA sequences. Mol. Biol. Evol. 1994, 11((5)), 725–736. [Google Scholar]
- Yang, Z. Likelihood ratio tests for detecting positive selection and application to primate lysozyme evolution. Mol. Biol. Evol. 1998, 15((5)), 568–573. [Google Scholar]
- Yang, Z. PAML 4: phylogenetic analysis by maximum likelihood. Mol. Biol. Evol. 2007, 24((8)), 1586–1591. [Google Scholar]
- Nielsen, R.; Yang, Z. Likelihood models for detecting positively selected amino acid sites and applications to the HIV-1 envelope gene. Genetics 1998, 148((3)), 929–936. [Google Scholar]
- Yang, Z.; Wong, W.S.; Nielsen, R. Bayes empirical bayes inference of amino acid sites under positive selection. Mol. Biol. Evol. 2005, 22((4)), 1107–1118. [Google Scholar]
- Pond, S.L.; Frost, S.D.; Muse, S.V. HyPhy: hypothesis testing using phylogenies. Bioinformatics 2005, 21((5)), 676–679. [Google Scholar]
- Murrell, B.; Moola, S.; Mabona, A.; Weighill, T.; Sheward, D.; Kosakovsky Pond, S.L.; Scheffler, K. FUBAR: a fast, unconstrained bayesian approximation for inferring selection. Mol. Biol. Evol. 2013, 30((5)), 1196–1205. [Google Scholar]
- Murrell, B.; Wertheim, J.O.; Moola, S.; Weighill, T.; Scheffler, K.; Kosakovsky Pond, S.L. Detecting individual sites subject to episodic diversifying selection. PLoS Genet. 2012, 8((7)), e1002764. [Google Scholar] [CrossRef]
- Pond, S.L.; Scheffler, K.; Gravenor, M.B.; Poon, A.F.; Frost, S.D. Evolutionary fingerprinting of genes. Mol. Biol. Evol. 2010, 27((3)), 520–536. [Google Scholar]
- Kosakovsky Pond, S.L.; Murrell, B.; Fourment, M.; Frost, S.D.; Delport, W.; Scheffler, K. A random effects branch-site model for detecting episodic diversifying selection. Mol. Biol. Evol. 2011, 28((11)), 3033–3043. [Google Scholar]
- Guindon, S.; Dufayard, J.F.; Lefort, V.; Anisimova, M.; Hordijk, W.; Gascuel, O. New algorithms and methods to estimate maximum-likelihood phylogenies: assessing the performance of PhyML 3.0. Syst. Biol. 2010, 59((3)), 307–321. [Google Scholar]
- Kelley, L.A.; Sternberg, M.J. Protein structure prediction on the Web: a case study using the Phyre server. Nat. Protoc. 2009, 4((3)), 363–371. [Google Scholar] [CrossRef]
- DeLano, W.L. The PyMOL Molecular Graphics System; version 1.5; Schrödinger, LLC: Camberley, UK, 2002. [Google Scholar]
- Armon, A.; Graur, D.; Ben-Tal, N. ConSurf: an algorithmic tool for the identification of functional regions in proteins by surface mapping of phylogenetic information. J. Mol. Biol. 2001, 307((1)), 447–463. [Google Scholar] [CrossRef]
- Steuten, J.; Winkel, K.; Carroll, T.; Williamson, N.A.; Ignjatovic, V.; Fung, K.; Purcell, A.W.; Fry, B.G. The molecular basis of cross-reactivity in the Australian Snake Venom Detection Kit (SVDK). Toxicon 2007, 50((8)), 1041–1052. [Google Scholar] [CrossRef]
Supplementary Files
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Jackson, T.N.W.; Sunagar, K.; Undheim, E.A.B.; Koludarov, I.; Chan, A.H.C.; Sanders, K.; Ali, S.A.; Hendrikx, I.; Dunstan, N.; Fry, B.G. Venom Down Under: Dynamic Evolution of Australian Elapid Snake Toxins. Toxins 2013, 5, 2621-2655. https://doi.org/10.3390/toxins5122621
Jackson TNW, Sunagar K, Undheim EAB, Koludarov I, Chan AHC, Sanders K, Ali SA, Hendrikx I, Dunstan N, Fry BG. Venom Down Under: Dynamic Evolution of Australian Elapid Snake Toxins. Toxins. 2013; 5(12):2621-2655. https://doi.org/10.3390/toxins5122621
Chicago/Turabian StyleJackson, Timothy N. W., Kartik Sunagar, Eivind A. B. Undheim, Ivan Koludarov, Angelo H. C. Chan, Kate Sanders, Syed A. Ali, Iwan Hendrikx, Nathan Dunstan, and Bryan G. Fry. 2013. "Venom Down Under: Dynamic Evolution of Australian Elapid Snake Toxins" Toxins 5, no. 12: 2621-2655. https://doi.org/10.3390/toxins5122621
APA StyleJackson, T. N. W., Sunagar, K., Undheim, E. A. B., Koludarov, I., Chan, A. H. C., Sanders, K., Ali, S. A., Hendrikx, I., Dunstan, N., & Fry, B. G. (2013). Venom Down Under: Dynamic Evolution of Australian Elapid Snake Toxins. Toxins, 5(12), 2621-2655. https://doi.org/10.3390/toxins5122621







