CCD Based Detector for Detection of Abrin Toxin Activity
Abstract
1. Introduction
2. Results
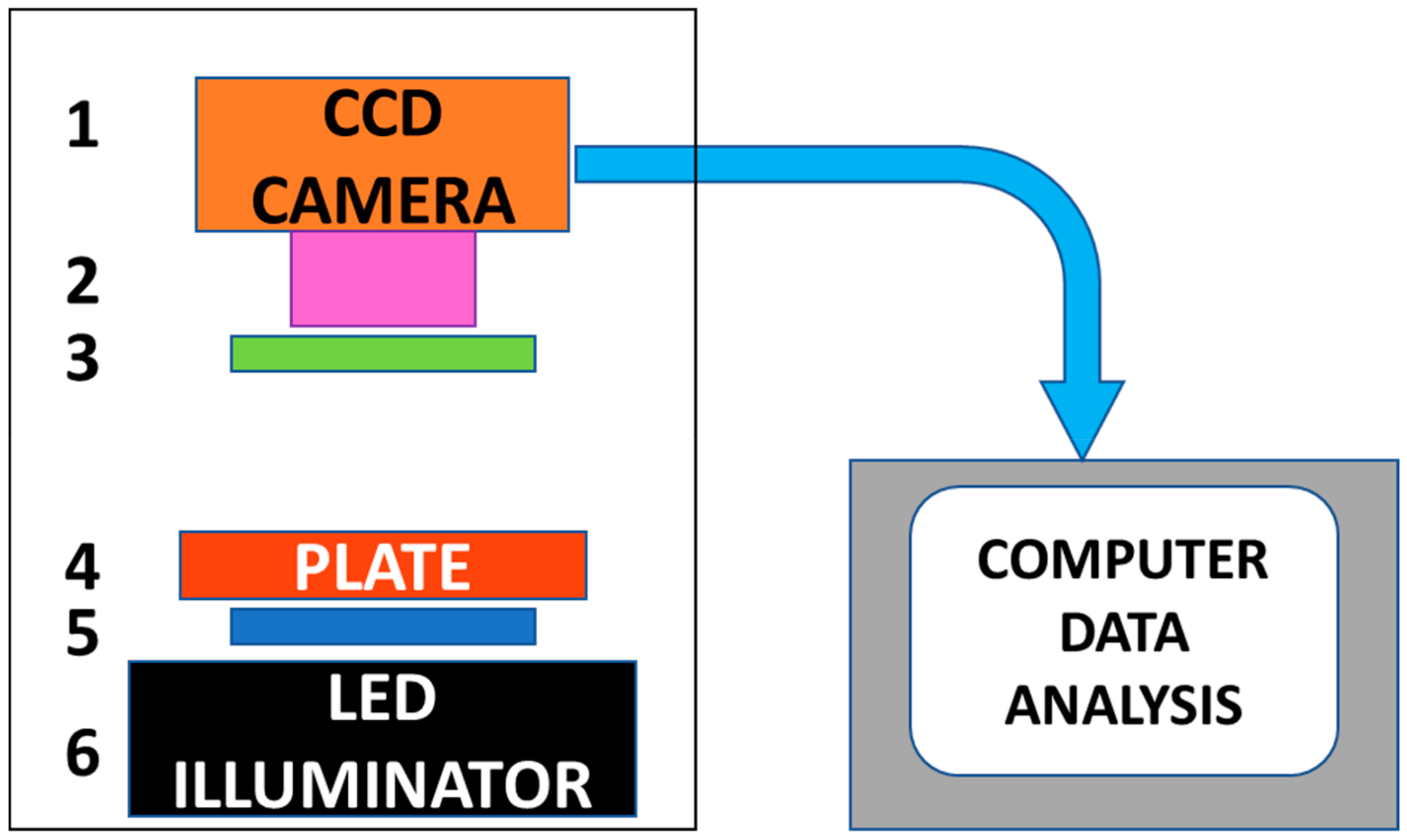
2.1. CCD Based Detector for Abrin Toxin Activity
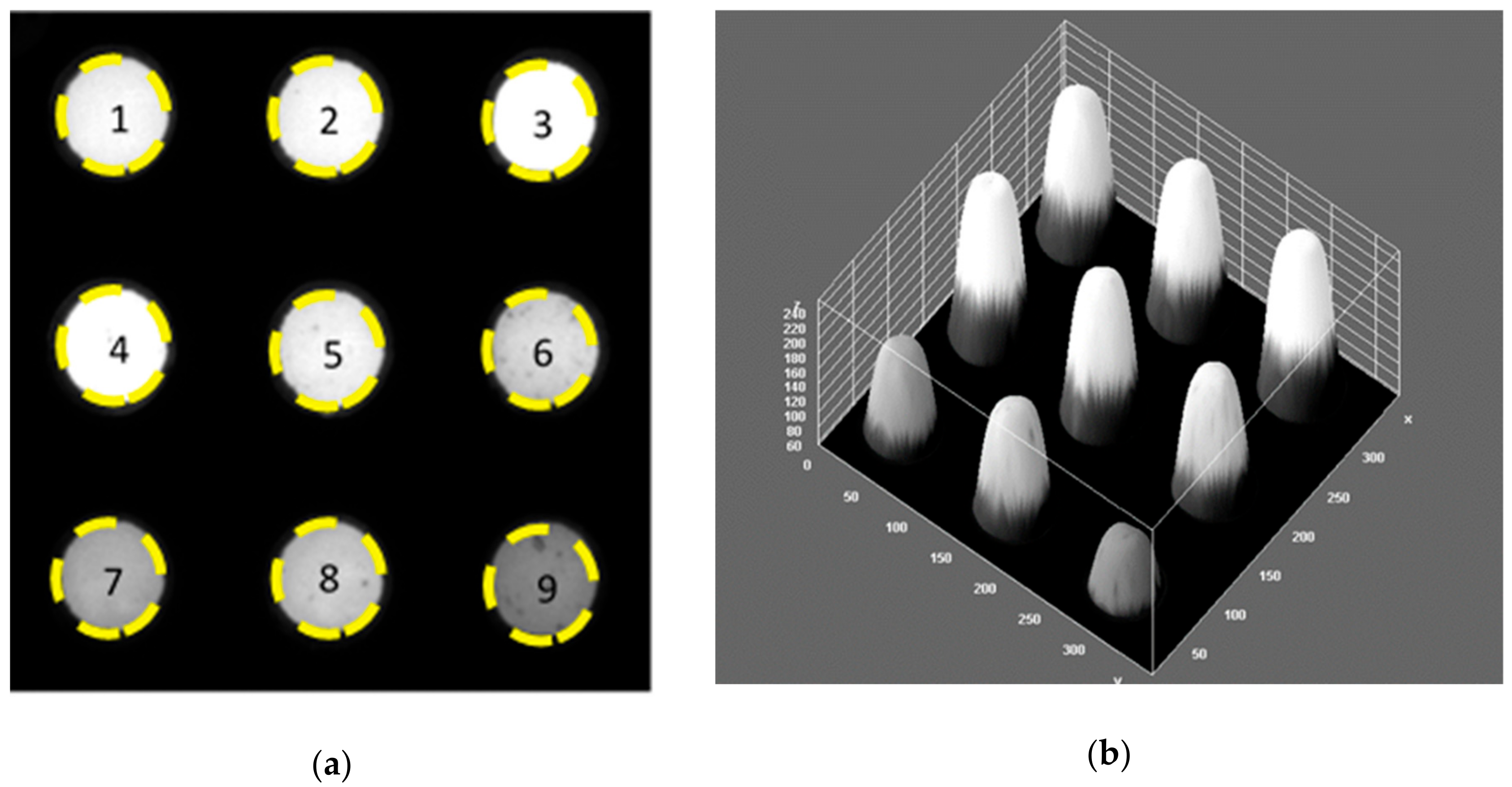
2.2. Miniature Assay Plates
2.3. Image Analysis
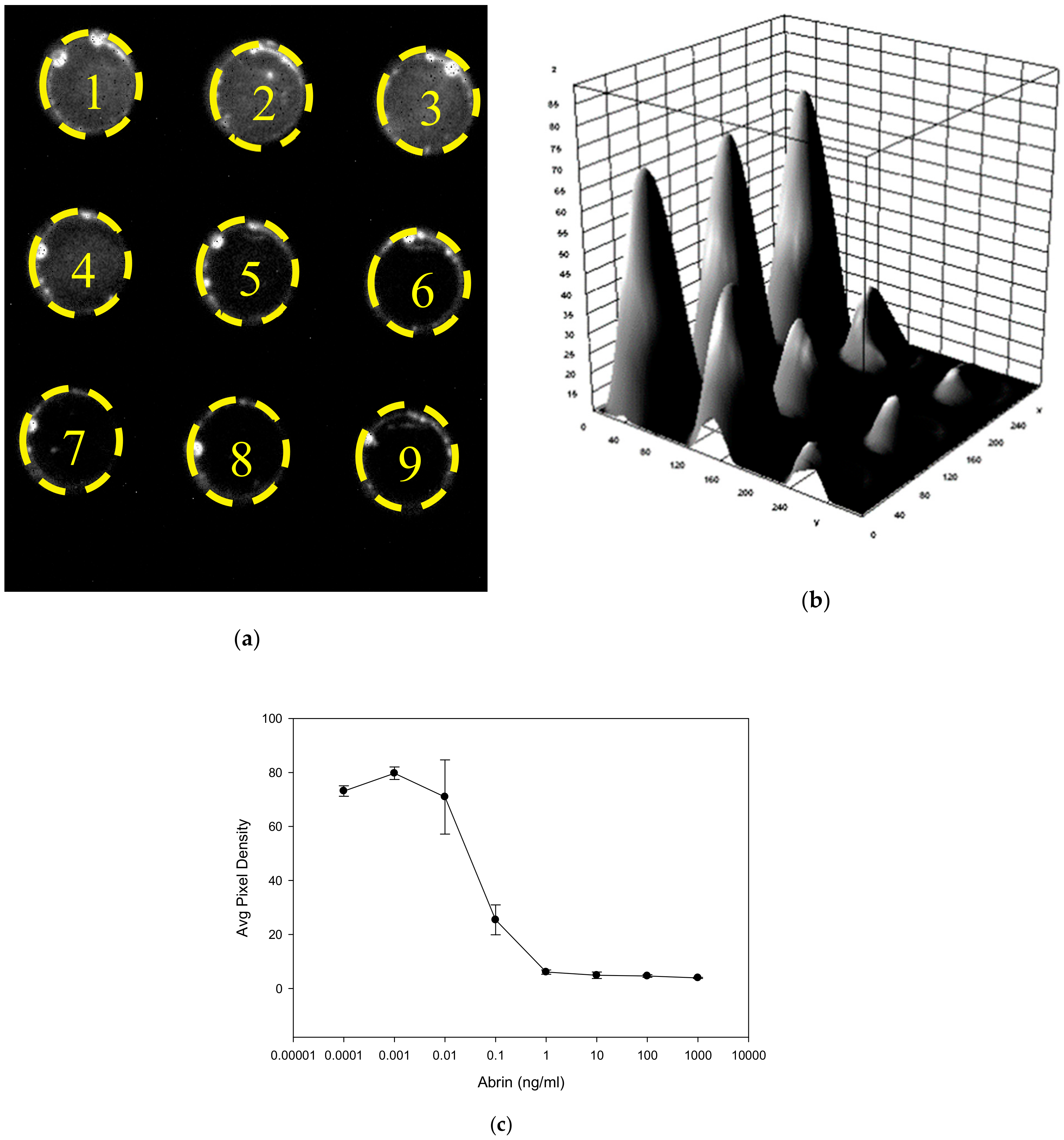
2.4. Cell Based in vitro Detection of Abrin
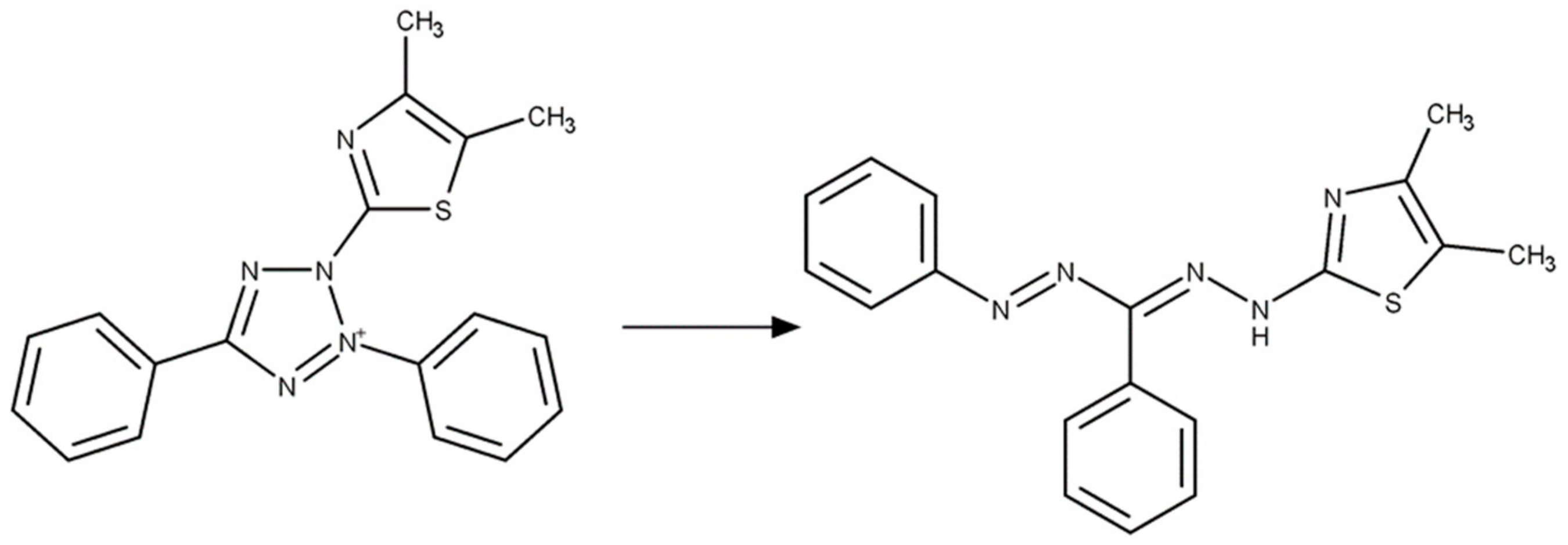
2.4.1. Colorimetric Measurement for Rapid Quantitative Detection of Abrin
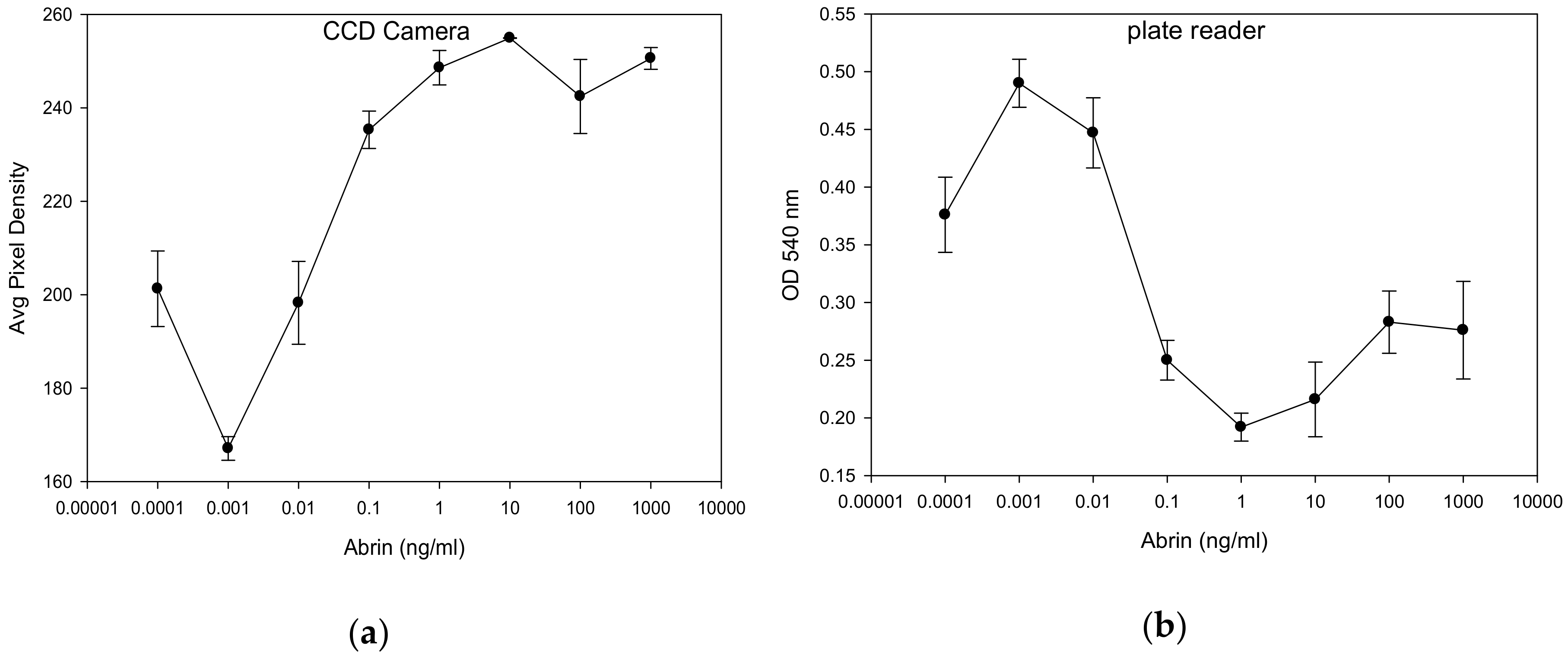
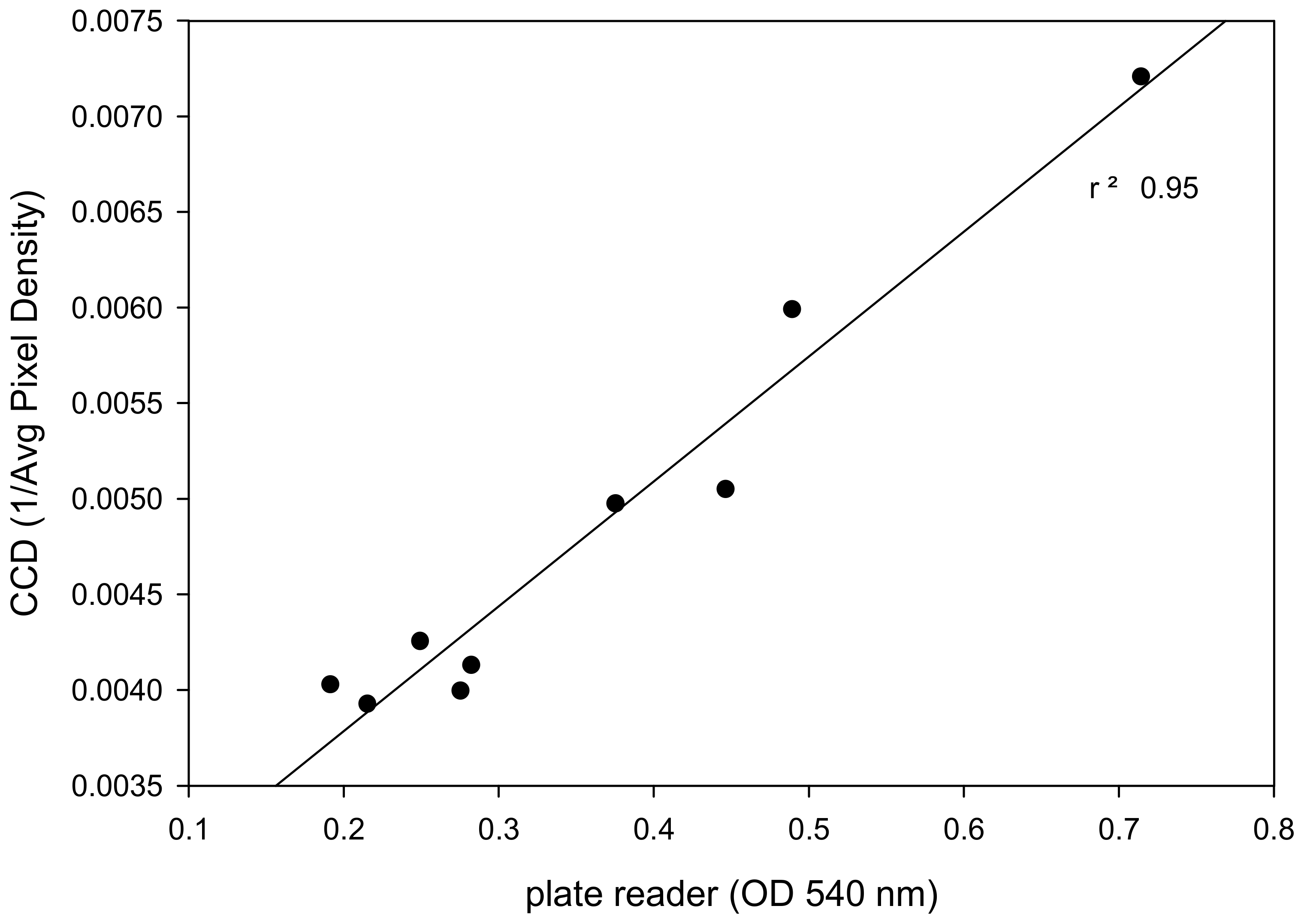
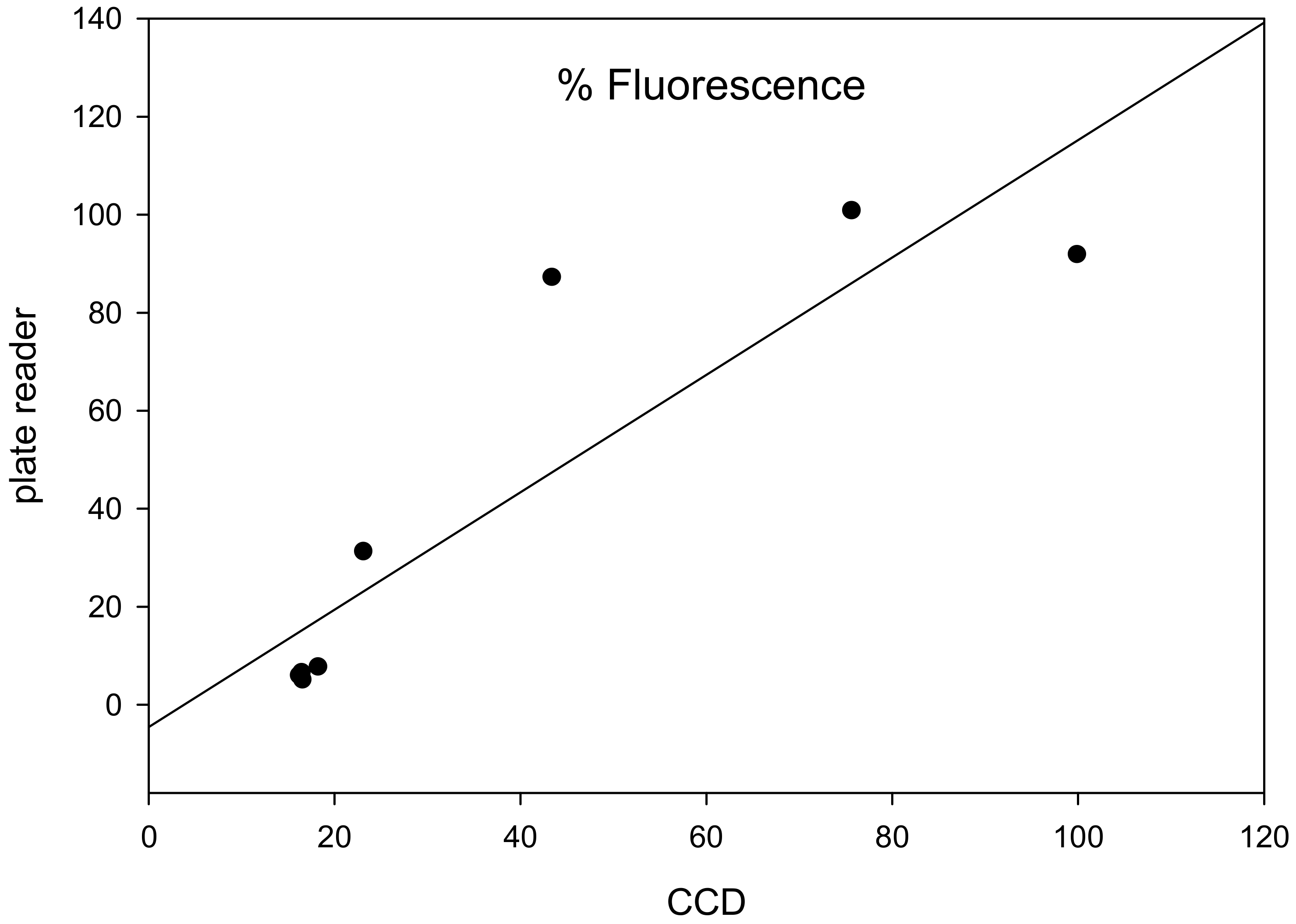
2.4.2. Relationship between Data Acquired by a Commercial Plate Reader and Low-cost CCD Detector
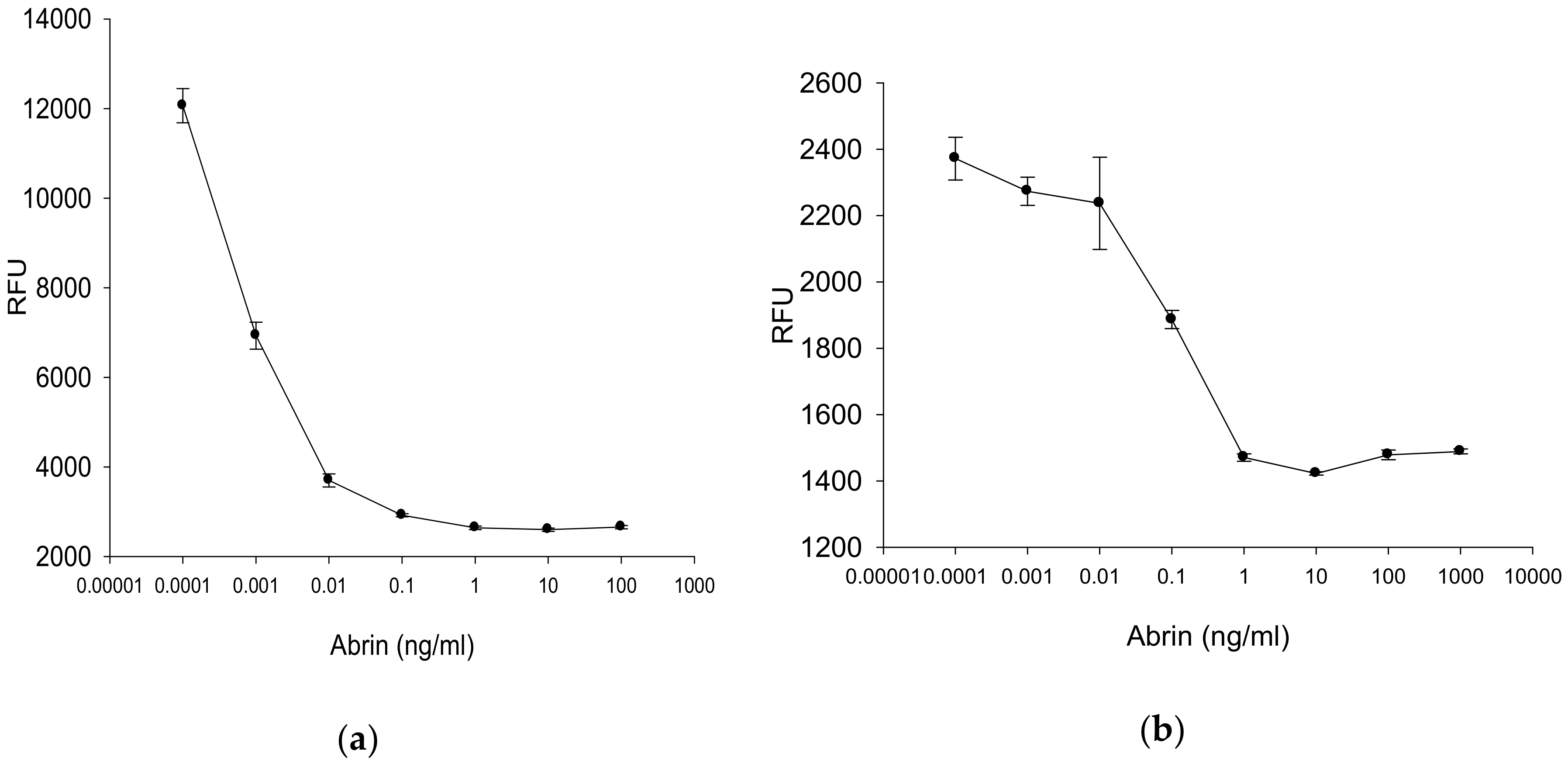
2.4.3. Fluorescence Measurement for Rapid Quantitative Detection of Abrin
2.4.4. Quantitative Fluorescence Digital Imaging Analysis of Abrin
2.4.5. Correlation between CCD Detector and Commercial Fluorometer Using Photomultipliers
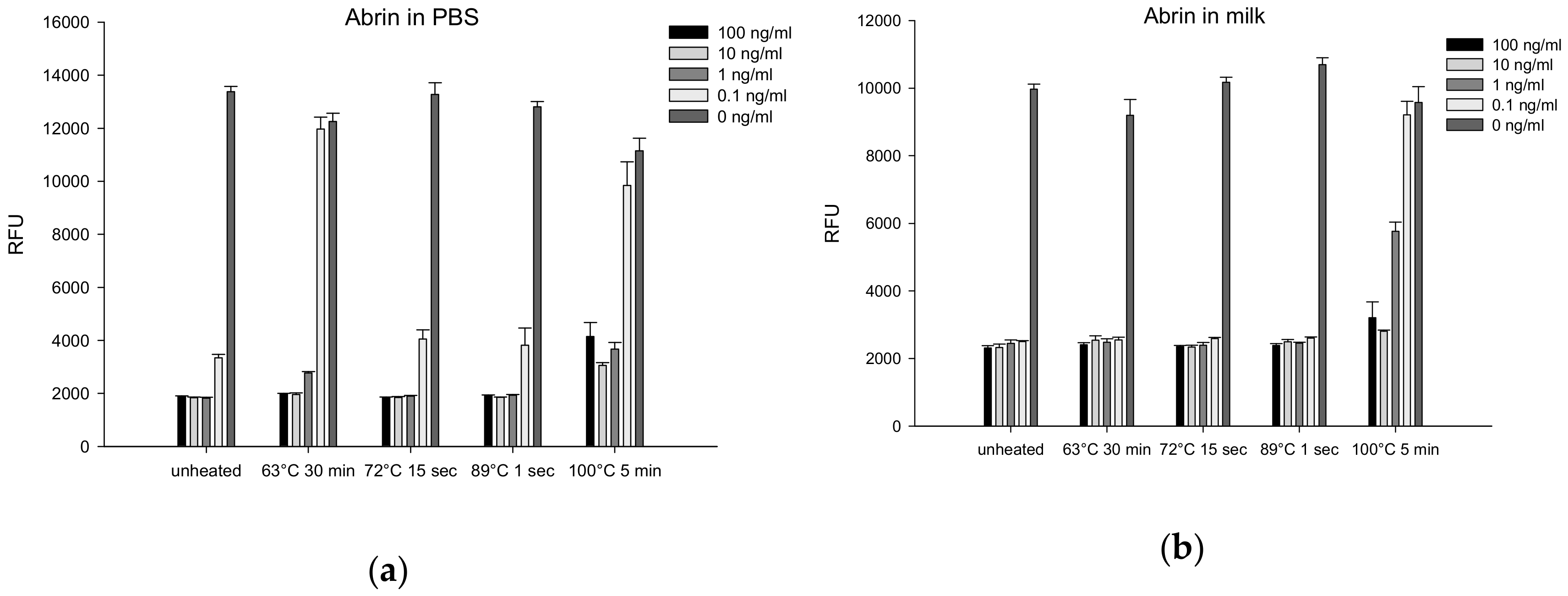
2.4.6. Thermal Inactivation of Abrin
3. Discussion
4. Materials and Methods
4.1. CCD Based Detector system
Computer Control and Data Analysis
Biological and Cell Culture Reagents
Colorimetric Measurement Reagents
4.2. Creation of Adenoviral Vectors that Express the Green Fluorescent Protein (GFP) Gene
4.3. Colorimetric Assay of Abrin Activity
4.4. Fluorometric Assay of Abrin Suppression of GFP
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Olsnes, S. The history of ricin, abrin and related toxins. Toxicon 2004, 44, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Stirpe, F. Ribosome-inactivating proteins. Toxicon 2004, 44, 371–383. [Google Scholar] [CrossRef] [PubMed]
- Stirpe, F.; Battelli, M.G. Ribosome-inactivating proteins: Progress and problems. Cell. Mol. Life Sci. 2006, 63, 1850–1866. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.K.; Hung, C.H.; Liaw, Y.C.; Lin, J.Y. Identification of amino acid residues of abrin-a A chain is essential for catalysis and reassociation with abrin-a B chain by site-directed mutagenesis. Protein Eng. 1997, 10, 827–833. [Google Scholar] [CrossRef] [PubMed]
- Owens, J.; Koester, C. Quantitation of abrine, an indole alkaloid marker of the toxic glycoproteins abrin, by liquid chromatography/tandem mass spectrometry when spiked into various beverages. J. Agric. Food Chem. 2008, 56, 11139–11143. [Google Scholar] [CrossRef] [PubMed]
- Gill, D.M. Bacterial toxins: A table of lethal amounts. Microbiol. Rev. 1982, 46, 86–94. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Patfield, S.; Cheng, L.W.; Stanker, L.H.; Rasooly, R.; McKeon, T.A.; Zhang, Y.; Brandon, D.L. Detection of Abrin Holotoxin Using Novel Monoclonal Antibodies. Toxins 2017, 9, 386. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Nie, C.; Wang, J.; Wang, J.; Kang, L.; Zhou, Y.; Wang, J.L. Colloidal gold-based immunochromatographic test strip for rapid detection of abrin in food samples. J. Food Prot. 2012, 75, 112–117. [Google Scholar] [CrossRef] [PubMed]
- Garber, E.A.; Venkateswaran, K.V.; O’Brien, T.W. Simultaneous multiplex detection and confirmation of the proteinaceous toxins abrin, ricin, botulinum toxins, and Staphylococcus enterotoxins A, B, and C in food. J. Agric. Food Chem. 2010, 58, 6600–6607. [Google Scholar] [CrossRef] [PubMed]
- Garber, E.A.; Walker, J.L.; O’Brien, T.W. Detection of abrin in food using enzyme-linked immunosorbent assay and electrochemiluminescence technologies. J. Food Prot. 2008, 71, 1868–1874. [Google Scholar] [CrossRef] [PubMed]
- Felder, E.; Mossbrugger, I.; Lange, M.; Wolfel, R. Simultaneous detection of ricin and abrin DNA by real-time PCR (qPCR). Toxins 2012, 4, 633–642. [Google Scholar] [CrossRef] [PubMed]
- Hansbauer, E.M.; Worbs, S.; Volland, H.; Simon, S.; Junot, C.; Fenaille, F.; Dorner, B.G.; Becher, F. Rapid Detection of Abrin Toxin andIts Isoforms in Complex Matrices by ImmunoExtraction and Quantitative High Resolution Targeted Mass Spectrometry. Anal. Chem. 2017, 89, 11719–11727. [Google Scholar] [CrossRef] [PubMed]
- Tam, C.C.; Henderson, T.D., 2nd; Stanker, L.H.; Cheng, L.W. Influence of Food Matrices on the Stability and Bioavailability of Abrin. Toxins 2018, 10, 502. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rasooly, R.; Do, P.; Hernlem, B. CCD Based Detector for Detection of Abrin Toxin Activity. Toxins 2020, 12, 120. https://doi.org/10.3390/toxins12020120
Rasooly R, Do P, Hernlem B. CCD Based Detector for Detection of Abrin Toxin Activity. Toxins. 2020; 12(2):120. https://doi.org/10.3390/toxins12020120
Chicago/Turabian StyleRasooly, Reuven, Paula Do, and Bradley Hernlem. 2020. "CCD Based Detector for Detection of Abrin Toxin Activity" Toxins 12, no. 2: 120. https://doi.org/10.3390/toxins12020120
APA StyleRasooly, R., Do, P., & Hernlem, B. (2020). CCD Based Detector for Detection of Abrin Toxin Activity. Toxins, 12(2), 120. https://doi.org/10.3390/toxins12020120




