Aflatoxin B1 and Sterigmatocystin Binding Potential of Lactobacilli
Abstract
1. Introduction
2. Results
2.1. Analytical Determination of the Mycotoxins
2.2. Optimisation for Mycotoxin Binding Experiments
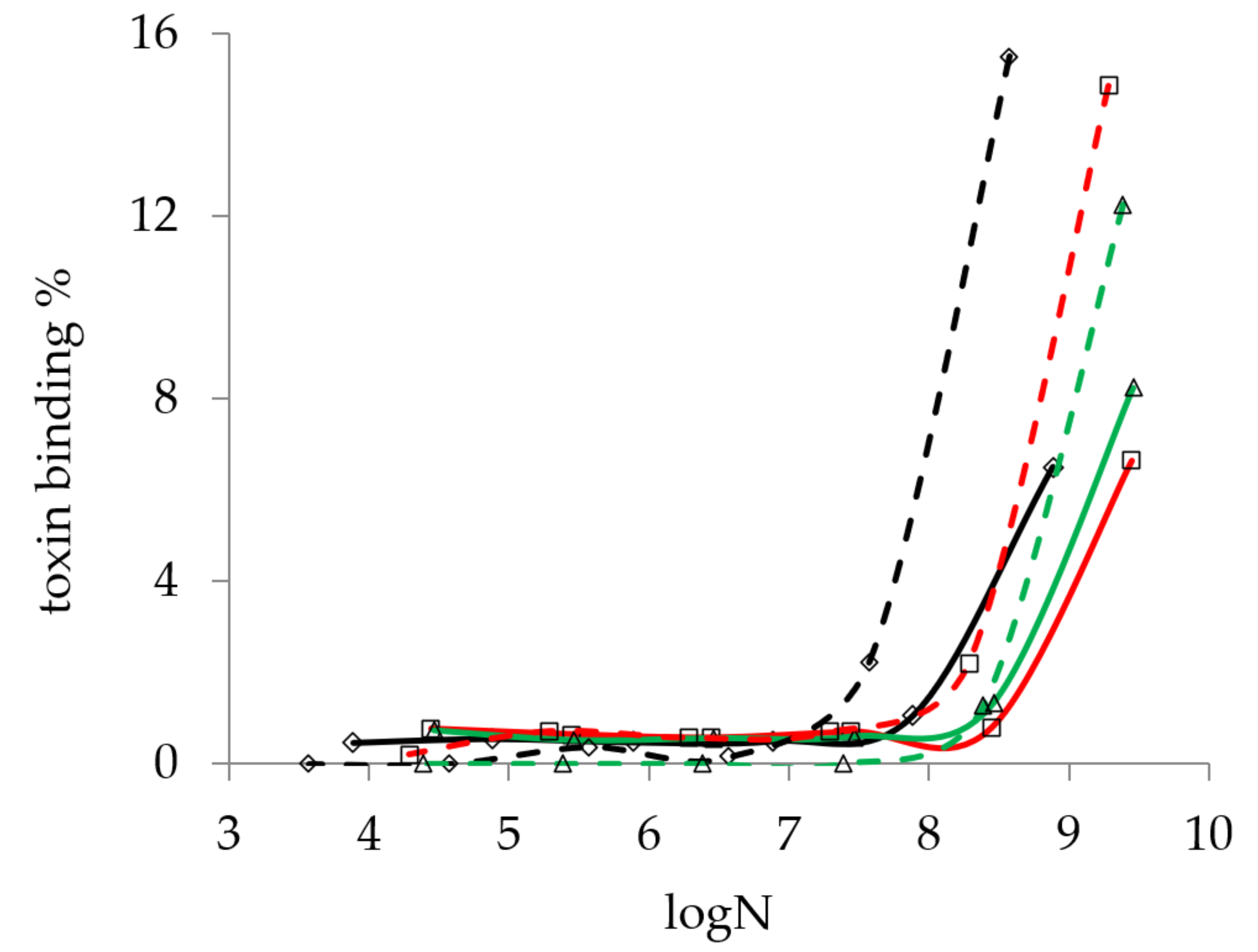
2.2.1. Study of the Effect of Bacterial Count on Mycotoxin Binding of Lactobacillus Strains
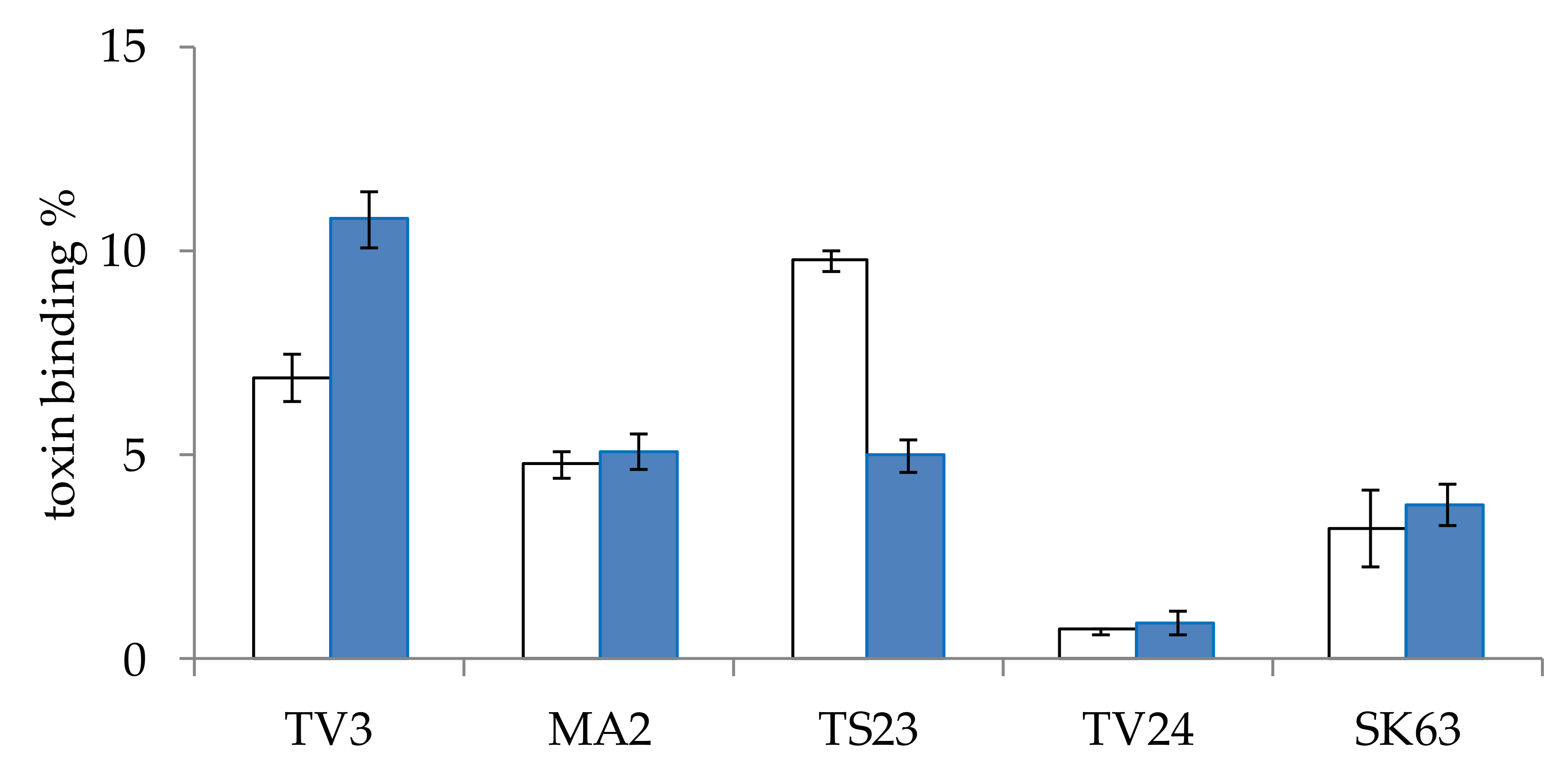
2.2.2. Study of the Effect of Incubation Time on Mycotoxin Binding of Lactobacillus Strains
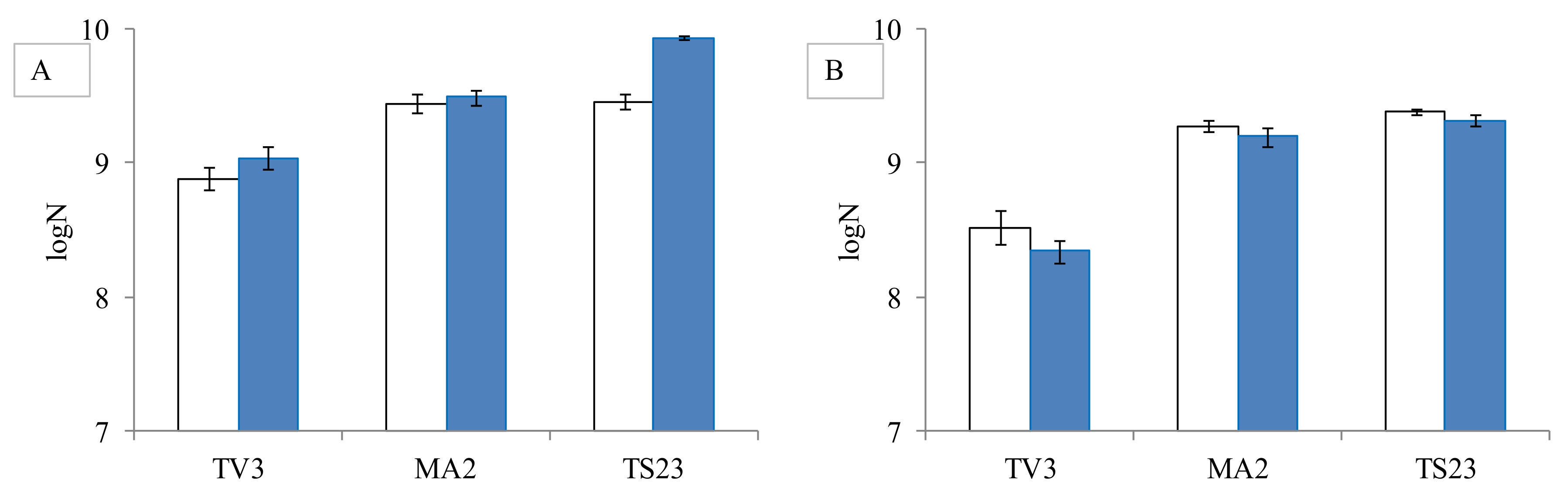
2.2.3. Study of the Effect of Mycotoxins on Lactobacillus Cell Count
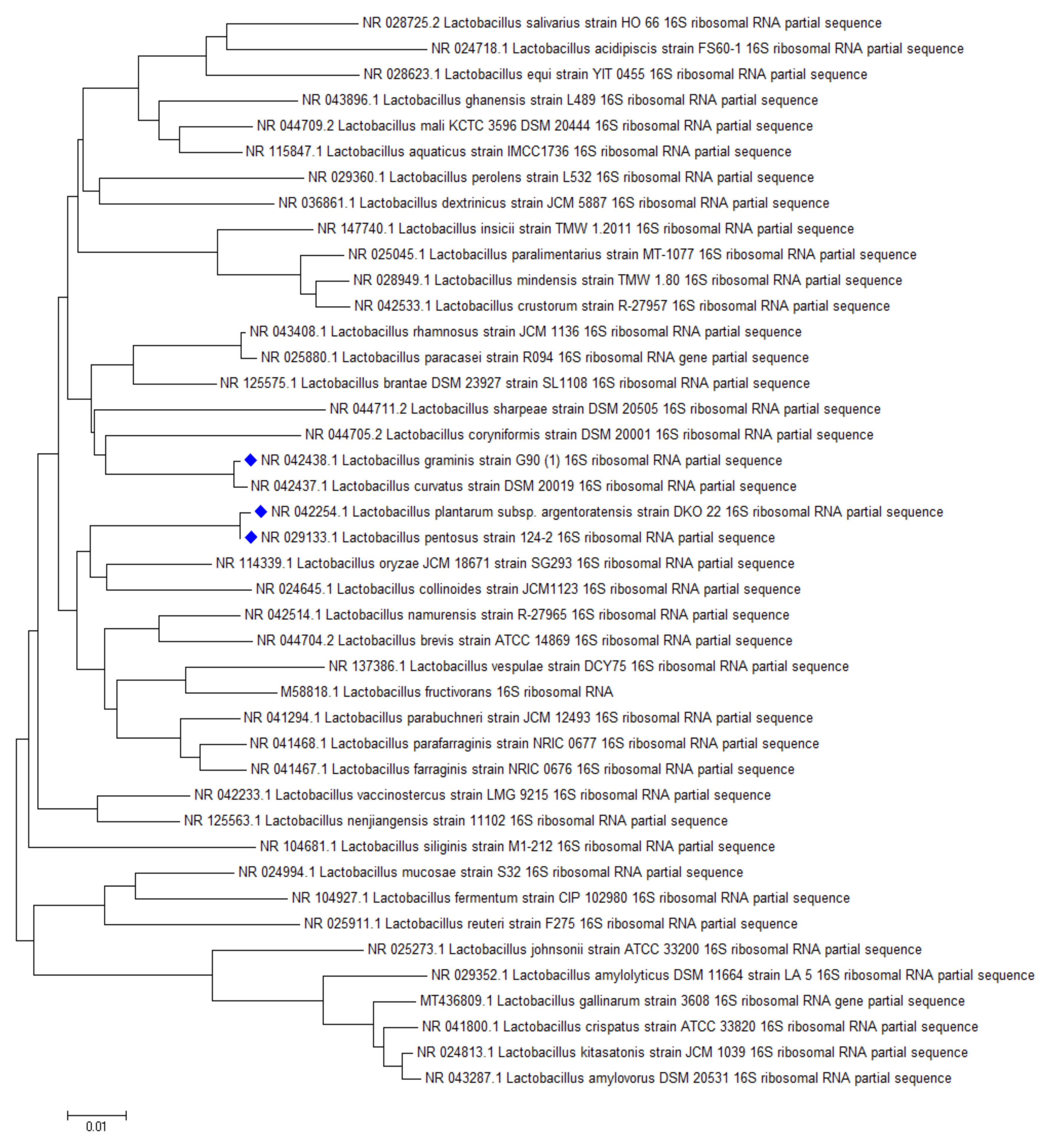
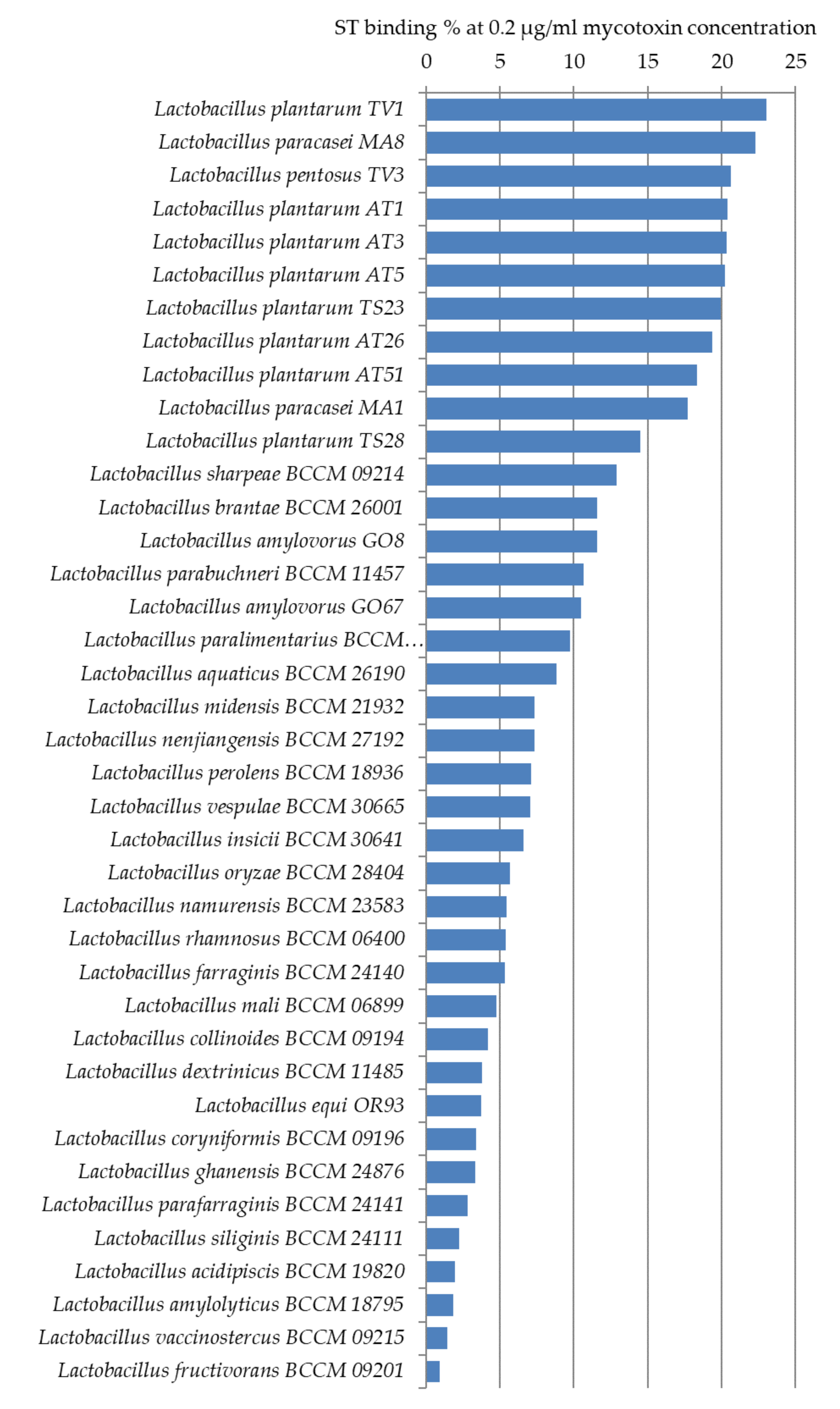
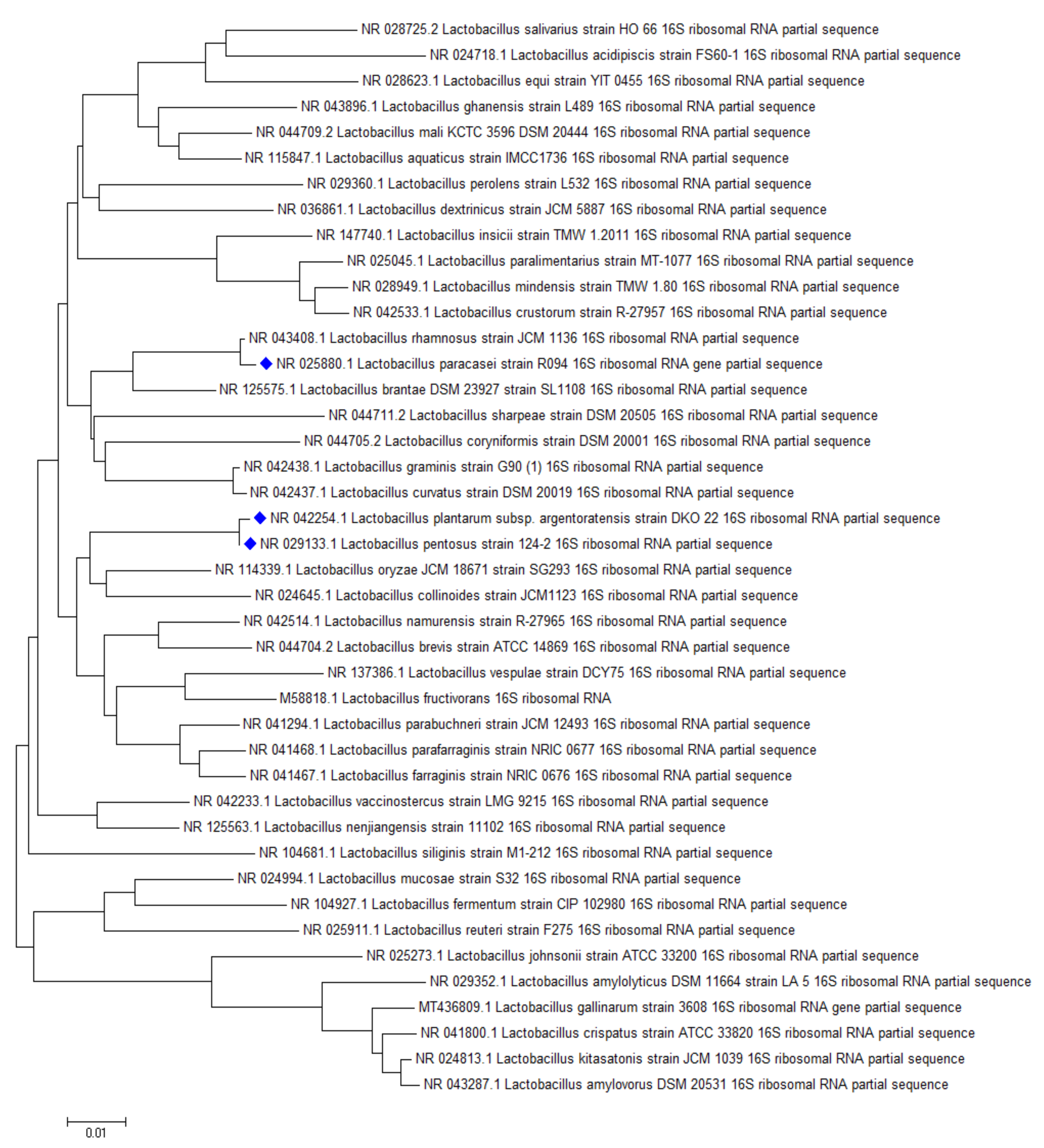
2.3. Screening Lactobacillus Strains for Mycotoxin Binding Capacities
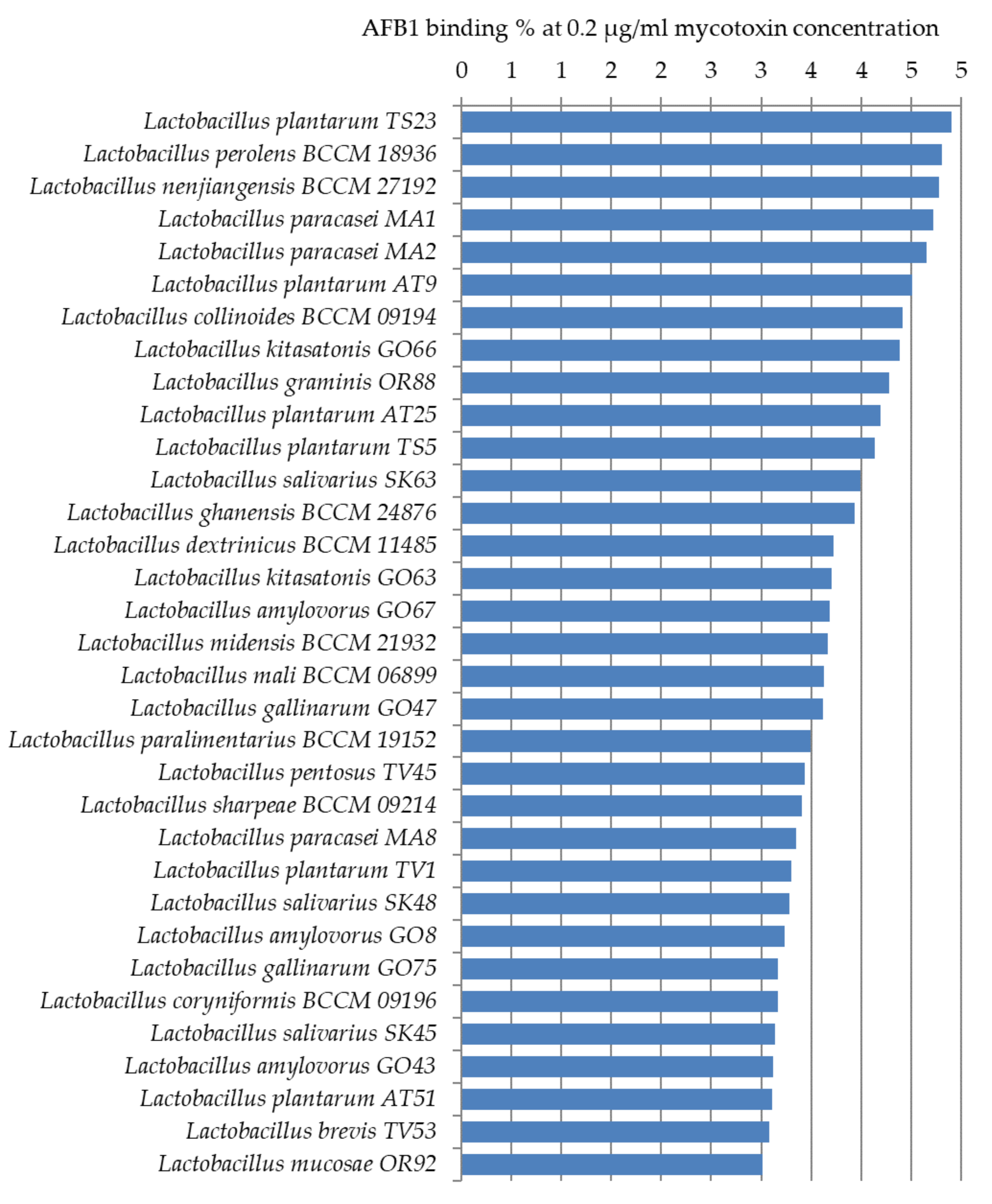
2.3.1. Aflatoxin B1 Binding Capacities of Lactobacilli
2.3.2. Sterigmatocystin Binding Capacities of Lactobacilli
3. Conclusions
4. Materials and Methods
4.1. Bacterial Strains
4.2. Mycotoxins
4.3. Mycotoxin Extraction and Analytical Determination
4.4. Optimisation for Mycotoxin Binding Experiments
4.4.1. Study of the Effect of Bacterial Count on Mycotoxin Binding of LAB Strains
4.4.2. Study of the Effect of Incubation Time on Mycotoxin Binding of Lactobacillus Strains
4.4.3. Study of the Effect of Mycotoxin on Lactobacillus Cell Count
4.5. Screening LAB Strains for Mycotoxin Binding Capacities
4.6. Statistical Analyses
Author Contributions
Funding
Conflicts of Interest
References
- Dobolyi, C.; Sebők, F.; Varga, J.; Kocsubé, S.; Szigeti, G.; Baranyi, N.; Szécsi, Á.; Tóth, B.; Varga, M.; Kriszt, B.; et al. Occurrence of aflatoxin producingAspergillus flavusisolates in maize kernel in Hungary. Acta Aliment. 2013, 42, 451–459. [Google Scholar] [CrossRef]
- Alassane-Kpembi, I.; Schatzmayr, G.; Taranu, I.; Marin, D.; Puel, O.; Oswald, I.P. Mycotoxins co-contamination: Methodological aspects and biological relevance of combined toxicity studies. Crit. Rev. Food Sci. Nutr. 2017, 57, 3489–3507. [Google Scholar] [CrossRef] [PubMed]
- Jelinek, C.F.; E Pohland, A.; E Wood, G. Worldwide occurrence of mycotoxins in foods and feeds—An update. J. Assoc. Off. Anal. Chem. 1989, 72, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Pitt, J.I. Toxigenic fungi and mycotoxins. Br. Med. Bull. 2000, 56, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Raters, M.; Matissek, R. Thermal stability of aflatoxin B1 and ochratoxin A. Mycotoxin Res. 2008, 24, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Commission Regulation (EU) No 574/2011. Annex I to Directive 2002/32/EC of the European Parliament and of the Council as Regards Maximum Levels for Nitrite, Melamine, Ambrosia spp. and Carry-over of Certain Coccidiostats and Histomonostats and Consolidating Annexes I and II. 2011, L 159/7. Available online: https://eur-lex.europa.eu/eli/reg/2011/574/oj?locale=en (accessed on 1 October 2020).
- Sweeney, M.J.; Dobson, A.D. Molecular biology of mycotoxin biosynthesis. FEMS Microbiol. Lett. 1999, 175, 149–163. [Google Scholar] [CrossRef] [PubMed]
- Cole, R.J.; Cox, R.H. Handbook of Toxic Fungal Metabolites; Academic Press: London, UK, 1981; pp. 67–93. [Google Scholar]
- Mol, J.G.J.; Pietri, A.; MacDonald, S.J.; Anagnostopoulos, C.; Spanjer, M. Survey on Sterigmatocystin in Food; Research report EFSA supporting publication 2015 EN-774; RIKILT-Wageningen UR: Wageningen, The Netherlands, 2015. [Google Scholar]
- Bokhari, F.M.; Aly, M.M. Evolution of traditional means of roasting and mycotoxins contaminated coffee beans in Saudi Arabia. Adv. Biol. Res. 2009, 3, 71–78. [Google Scholar]
- Zhao, Y.; Wang, Q.; Huang, J.; Ma, L.; Chen, Z.; Wang, F. Aflatoxin B1 and sterigmatocystin in wheat and wheat products from supermarkets in China. Food Addit. Contam. Part B 2018, 11, 9–14. [Google Scholar] [CrossRef]
- Nomura, M.; Aoyama, K.; Ishibashi, T. Sterigmatocystin and aflatoxin B1 contamination of corn, soybean meal, and formula feed in Japan. Mycotoxin Res. 2017, 34, 21–27. [Google Scholar] [CrossRef]
- Bianchini, A.; Bullerman, L.B. Biological control of molds and mycotoxins in foods. In Mycotoxin Prevention and Control in Agriculture; Appell, M., Kendra, D., Trucksess, M., Eds.; American Chemical Society: Washington, DC, USA, 2010; Volume 1031, pp. 1–16. [Google Scholar]
- Yiannikouris, A.; François, J.; Poughon, L.; Dussap, C.G.; Bertin, G.; Jeminet, G.; Jouany, J.-P. Adsorption of Zearalenone by beta-D-glucans in the Saccharomyces cerevisiae cell wall. J. Food Prot. 2004, 67, 1195–1200. [Google Scholar] [CrossRef]
- Guan, S.; Gong, M.; Yin, Y.; Huang, R.; Ruan, Z.; Zhou, T.; Xie, M. Occurrence of mycotoxins in feeds and feed ingredients in China. J. Food Agric. Environ. 2011, 9, 163–167. [Google Scholar]
- Chapot-Chartier, M.-P. Interactions of the cell-wall glycopolymers of lactic acid bacteria with their bacteriophages. Front. Microbiol. 2014, 5, 236. [Google Scholar] [CrossRef] [PubMed]
- Fochesato, A.; Cuello, D.; Poloni, V.; Galvagno, M.A.; Dogi, C.A.; Cavaglieri, L.R. Aflatoxin B1adsorption/desorption dynamics in the presence of Lactobacillus rhamnosus RC007 in a gastrointestinal tract-simulated model. J. Appl. Microbiol. 2018, 126, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Lahtinen, S.J.; Haskard, C.A.; Ouwehand, A.C.; Salminen, S.J.; Ahokas, J.T. Binding of aflatoxin B1 to cell wall components of Lactobacillus rhamnosus strain GG. Food Addit. Contam. 2004, 21, 158–164. [Google Scholar] [CrossRef]
- Chapot-Chartier, M.P.; Vinogradov, E.; Sadovskaya, I.; Andre, G.; Mistou, M.Y.; Trieu-Cuot, P.; Furlan, S.; Bidnenko, E.; Courtin, P.; Péchoux, C.; et al. The cell surface of Lactococcus lactis is covered by a protective polysaccharide pellicle. J. Biol. Chem. 2010, 285, 10464–10471. [Google Scholar] [CrossRef]
- Linares, D.M.; Fitzgerald, G.; Hill, C.; Stanton, C.; Ross, P. Production of vitamins, exopolysaccharides and bacteriocins by probiotic bacteria. In Probiotic Dairy Products, 2nd ed.; Tamime, A., Thomas, L., Eds.; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 2018; pp. 359–388. [Google Scholar]
- Tsai, Y.T.; Cheng, P.C.; Pan, T.M. The immunomodulatory effects of lactic acid bacteria for improving immune functions and benefits. Appl. Microbiol. Biotechnol. 2012, 96, 853–862. [Google Scholar] [CrossRef]
- Bueno, D.J.; Casale, C.H.; Pizzolitto, R.P.; Salvano, M.A.; Oliver, G. Physical adsorption of aflatoxin B1 by lactic acid bacteria and Saccharomyces cerevisiae: A theoretical model. J. Food. Prot. 2007, 70, 2148–2154. [Google Scholar] [CrossRef]
- Wacoo, A.P.; Mukisa, I.M.; Meeme, R.; Byakika, S.; Wendiro, D.; Sybesma, W.; Kort, R. Probiotic enrichment and reduction of aflatoxins in a traditional African maize-based fermented food. Nutrients 2019, 11, 265. [Google Scholar] [CrossRef]
- Peltonen, K.; El-Nezami, H.; Haskard, C.; Ahokas, J.; Salminen, S. Aflatoxin B1 binding by dairy strains of lactic acid bacteria and bifidobacteria. J. Dairy Sci. 2001, 84, 2152–2156. [Google Scholar] [CrossRef]
- Alshannaq, A.F.; Yu, J.-H. A liquid chromatographic method for rapid and sensitive analysis of aflatoxins in laboratory fungal cultures. Toxins 2020, 12, 93. [Google Scholar] [CrossRef]
- Ma, Z.X.; Amaro, F.X.; Romero, J.J.; Pereira, O.G.; Jeong, K.C.; Adesogan, A.T. The capacity of silage inoculant bacteria to bind aflatoxin B1 in vitro and in artificially contaminated corn silage. J. Dairy Sci. 2017, 100, 7198–7210. [Google Scholar] [CrossRef] [PubMed]
- Kasmani, F.B.; Karimi Torshizi, M.A.; Allameh, A.A.; Shariatmadari, F. Aflatoxin detoxification potential of lactic acid bacteria isolated from Iranian poultry. Iran. J. Vet. Res. 2012, 13, 152–155. [Google Scholar]
- El-Nezami, H.; Kankaanpaa, P.; Salminen, S.; Ahokas, J. Ability of dairy strains of lactic acid bacteria to bind a common food carcinogen, aflatoxin B1. Food Chem. Toxicol. 1998, 36, 321–326. [Google Scholar] [CrossRef]
- Huang, L.; Cuicui Duan, C.; Zhao, Y.; Gao, L.; Niu, C.; Xu, J.; Li, S. Reduction of aflatoxin B1 toxicity by Lactobacillus plantarum C88: A potential probiotic strain isolated from Chinese traditional fermented food “tofu”. PLoS ONE 2017, 12, e0170109. [Google Scholar] [CrossRef]
- Belkacem-Hanfi, N.; Fhoula, I.; Semmar, N.; Guesmi, A.; Perraud-Gaime, I.; Ouzari, H.-I.; Boudabous, A.; Roussos, S. Lactic acid bacteria against post-harvest moulds and ochratoxin A isolated from stored wheat. Biol. Control. 2014, 76, 52–59. [Google Scholar] [CrossRef]
- Apás, A.L.; González, S.N.; Arena, M.E. Potential of goat probiotic to bind mutagens. Anaerobe 2014, 28, 8–12. [Google Scholar] [CrossRef]
- Liew, W.-P.-P.; Nurul-Adilah, Z.; Than, L.T.L.; Mohd-Redzwan, S. The binding efficiency and interaction of Lactobacillus casei Shirota toward aflatoxin B1. Front. Microbiol. 2018, 9, 1503. [Google Scholar] [CrossRef]
- Hernandez-Mendoza, A.; Garcia, H.; Steele, J. Screening of Lactobacillus casei strains for their ability to bind aflatoxin B1. Food Chem. Toxicol. 2009, 47, 1064–1068. [Google Scholar] [CrossRef]
- Oluwafemi, F.; Da-Silva, F.A. Removal of aflatoxins by viable and heat-killed Lactobacillus species isolated from fermented maize. J. Appl. Biosci. 2009, 16, 871–876. [Google Scholar]
- Jakšić, D.; Klarić, M. Šegvić; Crnolatac, I.; Vujičić, N. Šijaković; Smrečki, V.; Górecki, M.; Pescitelli, G.; Piantanida, I. Unique Aggregation of Sterigmatocystin in Water Yields Strong and Specific Circular Dichroism Response Allowing Highly Sensitive and Selective Monitoring of Bio-Relevant Interactions. Mar. Drugs 2019, 17, 629. [Google Scholar]
- Despot, D.J.; Kocsubé, S.; Bencsik, O.; Kecskeméti, A.; Szekeres, A.; Vágvölgyi, C.; Varga, J.; Klarić, M. Šegvić Species diversity and cytotoxic potency of airborne sterigmatocystin-producing Aspergilli from the section Versicolores. Sci. Total. Environ. 2016, 562, 296–304. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Fan, J.; Huang, X.; Jin, Q.; Zhu, G. Determination of Sterigmatocystin in Infant Cereals from Hangzhou, China. J. AOAC Int. 2016, 99, 1273–1278. [Google Scholar] [CrossRef] [PubMed]

| Species | Strains |
|---|---|
| Lactobacillus amylovorus | GO5, GO8, GO43, GO45, GO67 |
| L. brevis | AT70, TV23, TV50, TV53 |
| L. crispatus | GO48 |
| L. crustorum | TV19 |
| L. curvatus | TS4 |
| L. equi | OR7, OR25, OR86, OR93 |
| L. fermentum | SK64 |
| L. gallinarum | GO47, GO75, GO78 |
| L. graminis | OR12, OR81, OR88, TV24, TV35 |
| L. johnsonii | GO76 |
| L. kitasatonis | GO6, GO13, GO16, GO17, GO63, GO66, GO73, GO95, GO98 |
| L. mucosae | OR2, OR13, OR17, OR23, OR28, OR48, OR63, OR66, OR80, OR92 |
| L. paracasei | MA1, MA2, MA4, MA8, MA99 |
| L. pentosus | TV3, TV45 |
| L. plantarum | AT1, AT3, AT5, AT6, AT9, AT25, AT26, AT27, AT51, TS5, TS16, TS23, TS62, TV1 |
| L. reuterii | VO12, VO26 |
| L. salivarius | SK6, SK12, SK17, SK20, SK29, SK41, SK42, SK45, SK46, SK48, SK63, VO20 |
| Strains: |
|---|
| Lactobacillus farraginis BCCM 24140 |
| L. acidipiscis BCCM 19820 |
| L. fructivorans BCCM 09201 |
| L. oryzae BCCM 28404 |
| L. vaccinostercus BCCM 09215 |
| L. siliginis BCCM 24111 |
| L. parafarraginis BCCM 24141 |
| L. amylolyticus BCCM 18795 |
| L. namurensis BCCM 23583 |
| L. aquaticus BCCM 26190 |
| L. vespulae BCCM 30665 |
| L. coryniformis BCCM 09196 |
| L. sharpeae BCCM 09214 |
| L. paralimentarius BCCM 19152 |
| L. mali BCCM 06899 |
| L. midensis BCCM 21932 |
| L. dextrinicus BCCM 11485 |
| L. ghanensis BCCM 24876 |
| L. collinoides BCCM 09194 |
| L. nenjiangensis BCCM 27192 |
| L. perolens BCCM 18936 |
| L. rhamnosus BCCM 06400 |
| L. brantae BCCM 26001 |
| L. insicii BCCM 30641 |
| L. parabuchneri BCCM 11457 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kosztik, J.; Mörtl, M.; Székács, A.; Kukolya, J.; Bata-Vidács, I. Aflatoxin B1 and Sterigmatocystin Binding Potential of Lactobacilli. Toxins 2020, 12, 756. https://doi.org/10.3390/toxins12120756
Kosztik J, Mörtl M, Székács A, Kukolya J, Bata-Vidács I. Aflatoxin B1 and Sterigmatocystin Binding Potential of Lactobacilli. Toxins. 2020; 12(12):756. https://doi.org/10.3390/toxins12120756
Chicago/Turabian StyleKosztik, Judit, Mária Mörtl, András Székács, József Kukolya, and Ildikó Bata-Vidács. 2020. "Aflatoxin B1 and Sterigmatocystin Binding Potential of Lactobacilli" Toxins 12, no. 12: 756. https://doi.org/10.3390/toxins12120756
APA StyleKosztik, J., Mörtl, M., Székács, A., Kukolya, J., & Bata-Vidács, I. (2020). Aflatoxin B1 and Sterigmatocystin Binding Potential of Lactobacilli. Toxins, 12(12), 756. https://doi.org/10.3390/toxins12120756








