NaCl Inhibits Citrinin and Stimulates Monascus Pigments and Monacolin K Production
Abstract
:1. Introduction
2. Results
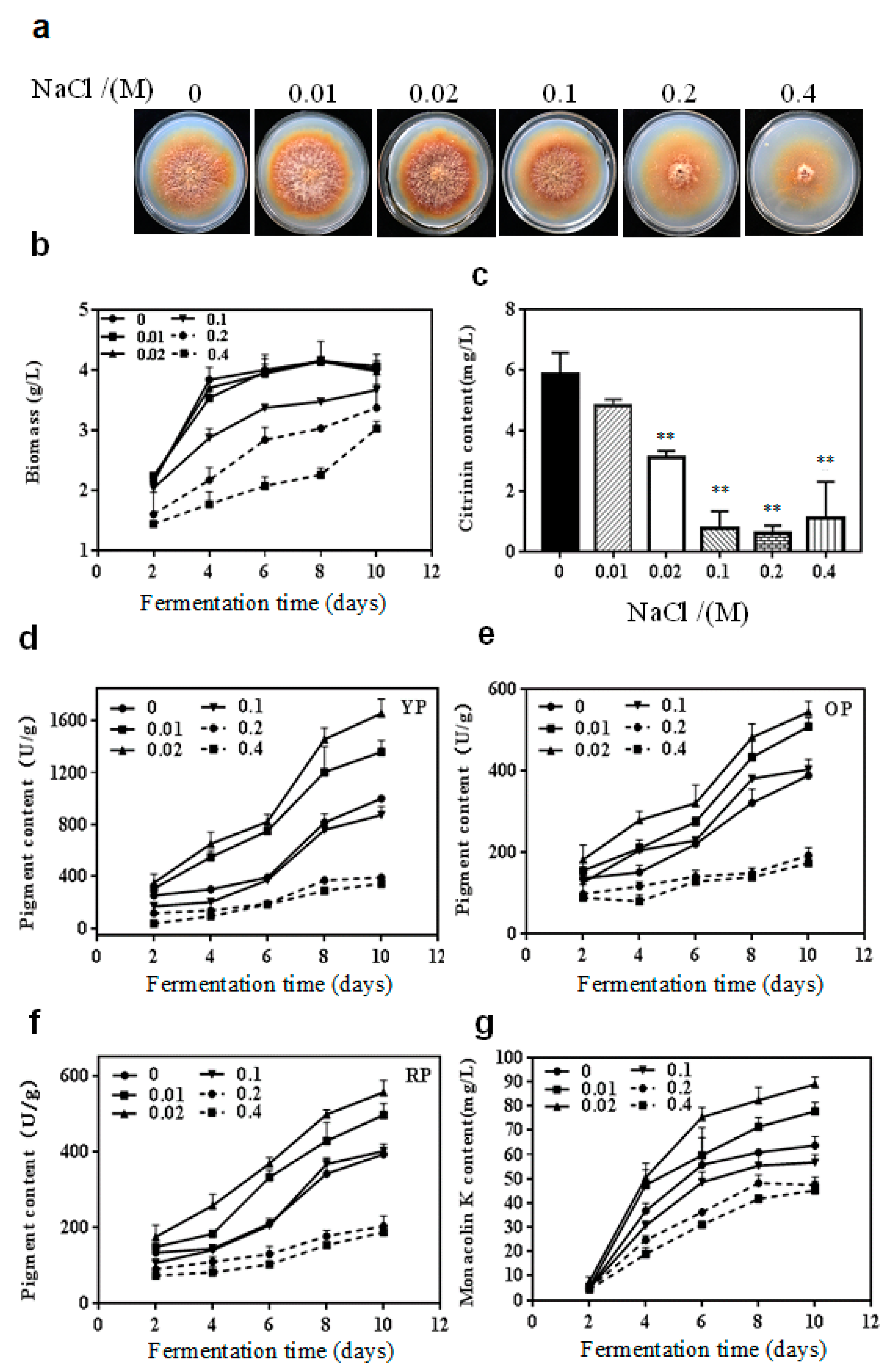
2.1. Effect of NaCl on the Growth and Secondary Metabolites of M. purpureus
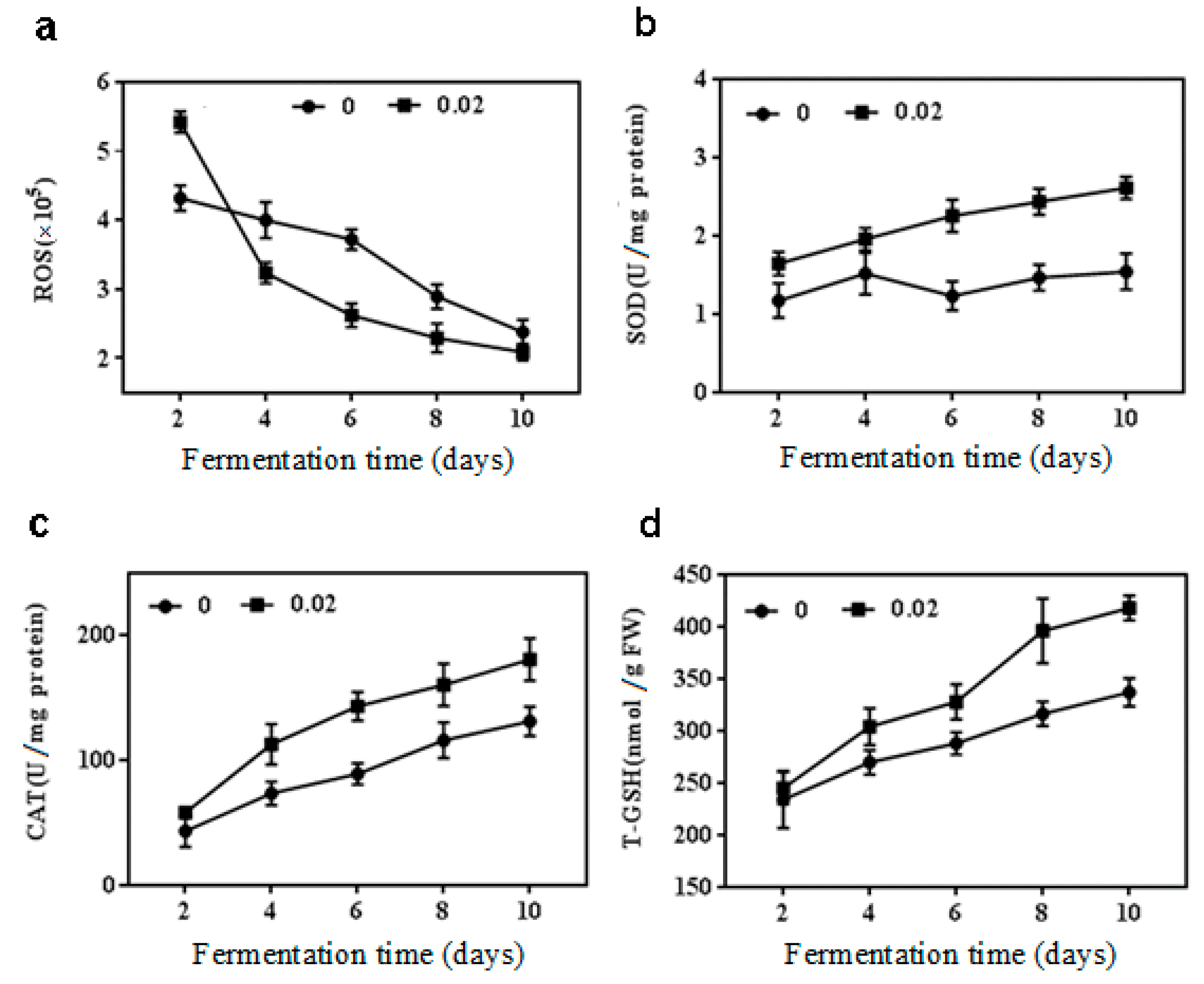
2.2. Effect of NaCl on Antioxidant Enzymes Activities, ROS and T-GSH Contents
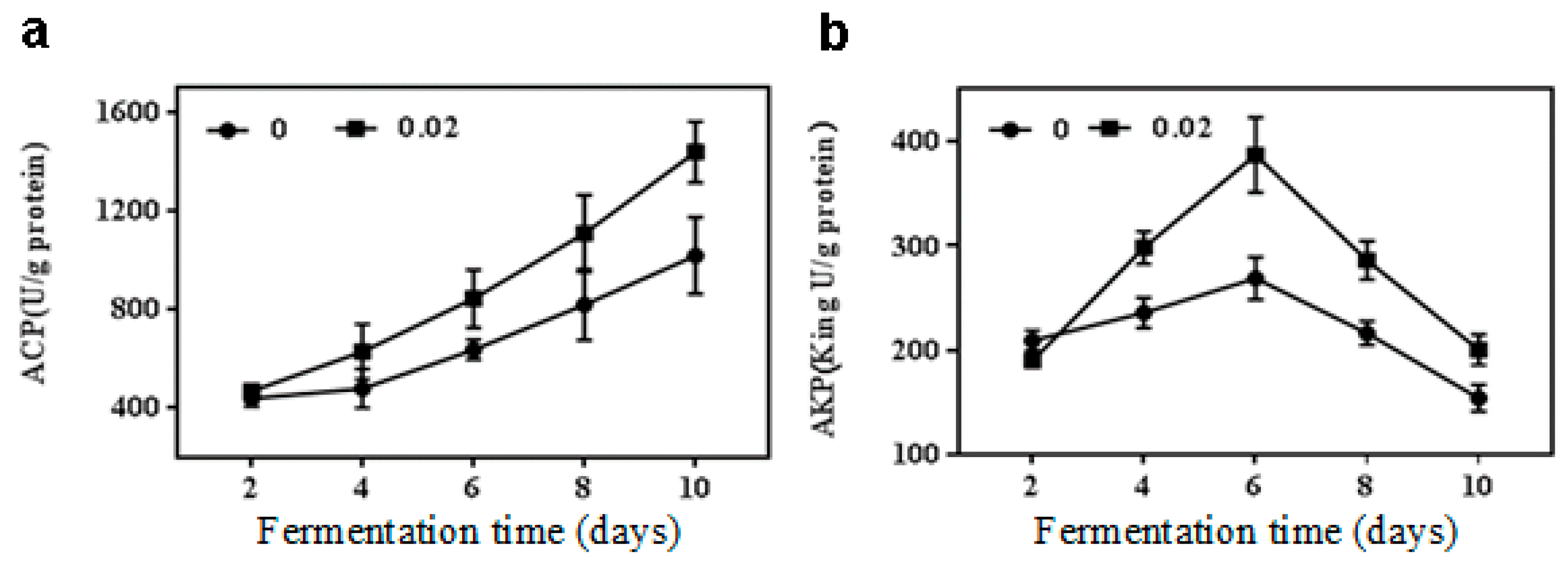
2.3. Effect of NaCl on Acid and Alkaline Phosphatase Activities
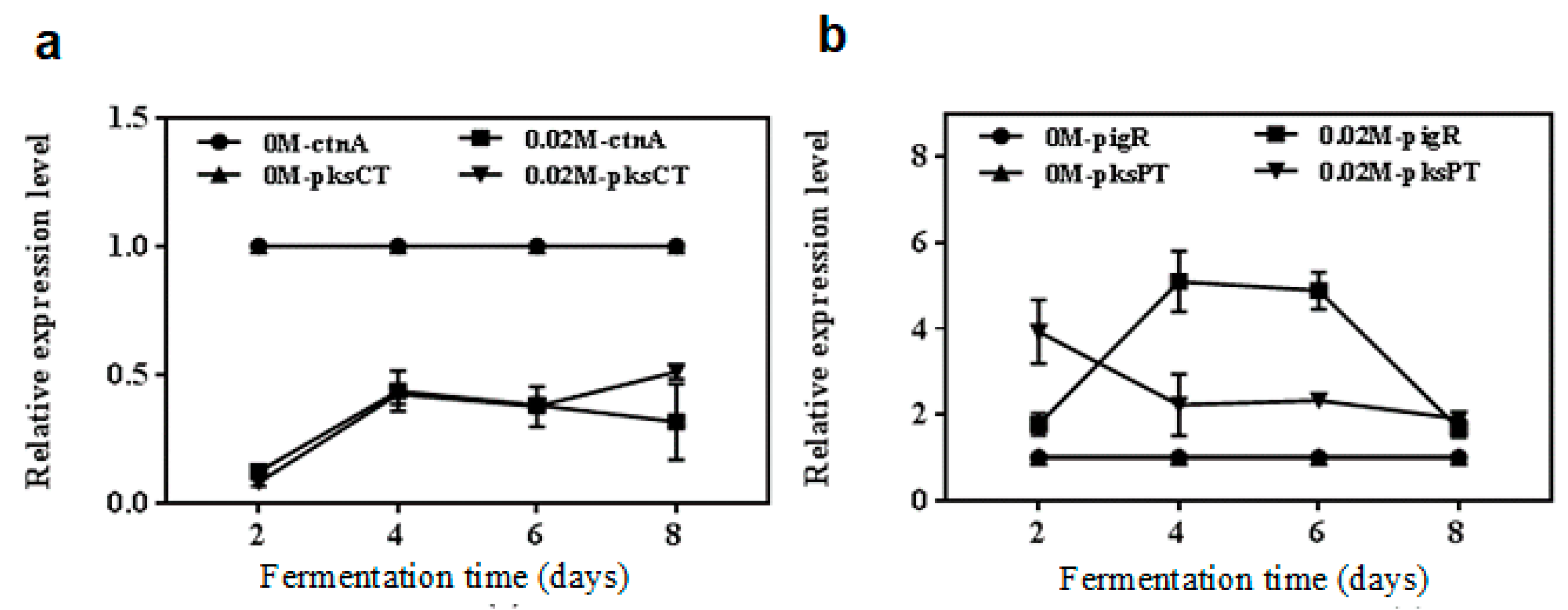
2.4. qRT-PCR Analyses of Genes Relative Levels
3. Discussion
4. Materials and Methods
4.1. Fungal Strain, Culture Media, and Growth Conditions
4.2. Biomass Determination
4.3. Citrinin, Monacolin K and MPs Determination
4.4. ROS Determination
4.5. SOD, CAT Activities and T-GSH Determination
4.6. Acid and Alkaline Phosphatase Activities Determination
4.7. Quantitative RT-PCR Analyses
4.8. Statistical Analyses
Author Contributions
Funding
Conflicts of Interest
References
- Feng, Y.; Shao, Y.; Chen, F. Monascus pigments. Appl. Microbiol. Biot. 2012, 96, 1421–1440. [Google Scholar] [CrossRef] [PubMed]
- Jůzlová, P.; Martínková, L.; Křen, V. Secondary metabolites of the fungus Monascus: A review. J. Ind. Microbiol. Biot. 1996, 16, 163–170. [Google Scholar] [CrossRef]
- Patakova, P. Monascus secondary metabolites: Production and biological activity. J. Ind. Microbiol. Biot. 2013, 40, 169–181. [Google Scholar] [CrossRef] [PubMed]
- Endo, A. Monacolin K, a new hypocholesterolemic agent that specifically inhibits 3-hydroxy-3-methylglutaryl coenzyme A reductase. J. Antibiot. 1980, 33, 334–336. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.C.; Wang, J.J.; Lin, T.T.; Pan, T.M. Production of the secondary metabolites γ-aminobutyric acid and monacolin K by Monascus. J. Ind. Microbiol. Biot. 2003, 30, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, T.; Kinoshita, H.; Ishihara, S.; Sakai, K.; Nagai, S.; Nihira, T. Polyketide Synthase Gene Responsible for Citrinin Biosynthesis in Monascus purpureus. Appl. Environ. Microbiol. 2005, 71, 3453–3457. [Google Scholar] [CrossRef]
- Pisareva, E.I.; Kujumdzieva, A.V. Influence of carbon and nitrogen sources on growth and pigment production by Monascus pilosus C1 strain. Biotechnol. Biotec. Eq. 2010, 24, 501–506. [Google Scholar] [CrossRef]
- Orozco, S.F.B.; Kilikian, B.V. Effect of pH on citrinin and red pigments production by Monascus purpureus CCT3802. World J. Microb. Biot. 2008, 24, 263–268. [Google Scholar] [CrossRef]
- Kang, B.; Zhang, X.; Wu, Z.; Wang, Z.; Park, S. Production of citrinin-free Monascus pigments by submerged culture at low pH. Enzyme Microb. Tech. 2014, 55, 50–57. [Google Scholar] [CrossRef]
- Chen, G.; Tang, R.; Tian, X.; Qin, P.; Wu, Z. Change of Monascus pigment metabolism and secretion in different extractive fermentation process. Bioproc. Biosyst. Eng. 2017, 40, 857–866. [Google Scholar] [CrossRef]
- Babitha, S.; Soccol, C.R.; Pandey, A. Effect of stress on growth, pigment production and morphology of Monascus sp. in solid cultures. J. Basic Microb. 2010, 47, 118–126. [Google Scholar] [CrossRef]
- Tallapragada, P. Comparative study of Monascus sanguineus and Monascus purpureus for red pigment production under stress condition. Int. Food Res. J. 2013, 20, 1235–1238. [Google Scholar]
- Samapundo, S.; Deschuyffeleer, N.; Laere, D.V.; Leyn, I.D.; Devlieghere, F. Effect of NaCl reduction and replacement on the growth of fungi important to the spoilage of bread. Food Microbiol. 2010, 27, 749–756. [Google Scholar] [CrossRef] [PubMed]
- Garg, N. Role of arbuscular mycorrhizal (AM) fungi on growth, cadmium uptake, osmolyte, and phytochelatin synthesis in Cajanus cajan (L.) Millsp. under NaCl and Cd stresses. J. Plant Growth Regul. 2012, 31, 292–308. [Google Scholar] [CrossRef]
- Elabyad, M.S.; Hindorf, H.; Rizk, M.A. Impact of salinity stress on soil-borne fungi of sugarbeet. I. Pathogenicity implications. Plant Soil. 1988, 110, 27–32. [Google Scholar] [CrossRef]
- Ochiai, N.; Tokai, T.; Nishiuchi, T.; Takahashi-Ando, N.; Fujimura, M.; Kimura, M. Involvement of the osmosensor histidine kinase and osmotic stress-activated protein kinases in the regulation of secondary metabolism in Fusarium graminearum. Biochem. Bioph. Res. Co. 2007, 363, 639–644. [Google Scholar] [CrossRef] [PubMed]
- Park, S.M.; Choi, E.S.; Kim, M.J.; Cha, B.J.; Yang, M.S.; Kim, D.H. Characterization of HOG1 homologue, CpMK1, from Cryphonectria parasitica and evidence for hypovirus-mediated perturbation of its phosphorylation in response to hypertonic stress. Mol. Microbiol. 2010, 51, 1267–1277. [Google Scholar] [CrossRef]
- Kawahara, Y.; Kikuchi, Y.; Kurahashi, O.; Kimura, E.; Nakamatsu, T.; Goto, S. Stress-resistant microorganism and method of producing fermentative product. U.S. Patent No. 6,338,956, 15 January 2002. [Google Scholar]
- Bajwa, M.S.; Choudhary, O.P.; Josan, A.S. Effect of continuous irrigation with sodic and saline-sodic waters on soil properties and crop yields under cotton-wheat rotation in northwestern India. Agr. Water Manage. 1992, 22, 345–356. [Google Scholar] [CrossRef]
- Liu, M.; Liang, Z.; Ma, H.; Huang, L.; Wang, M. Responses of rice (Oryza saliva L.) growth and yield to phosphogypsumamendment in saline-sodic soils of North-East China. J. Food Agric. Environ. 2010, 8, 827–833. [Google Scholar]
- Zou, Y.N.; Wu, Q.S. Efficiencies of five Arbuscular mycorrhizal fungi in alleviating salt stress of trifoliate orange. Int. J. Agric. Biol. 2011, 13, 991–995. [Google Scholar]
- Rawat, L.; Singh, Y.; Shukla, N.; Kumar, J. Salinity tolerant Trichoderma harzianum reinforces NaCl tolerance and reduces population dynamics of Fusarium oxysporum f.sp. ciceri in chickpea (Cicer arietinum L.) under salt stress conditions. Arch. Phytopathol. Plant Protect. 2013, 46, 1442–1467. [Google Scholar] [CrossRef]
- Kohler, J.H.J.A.; Caravaca, F.; Roldan, A. Induction of antioxidant enzymes is involved in the greater effectiveness of a PGPR versus AM fungi with respect to increasing the rolerance of lettuce to severe salt stress. Environ. Exp. Bot. 2009, 65, 245–252. [Google Scholar] [CrossRef]
- Zhou, X.; Ferraris, J.D.; Burg, M.B. Mitochondrial reactive oxygen species contribute to high NaCl-induced activation of the transcription factor TonEBP/OREBP. Am. J. Physiol. Renal Physiol. 2006, 290, F1169–F1176. [Google Scholar] [CrossRef] [PubMed]
- Miranda, R.U.; Gómez-Quiroz, L.E.; Mendoza, M.; Pérez-Sánchez, A.; Fierro, F.; Barrios-González, J. Reactive oxygen species regulate lovastatin biosynthesis in Aspergillus terreus during submerged and solid-state fermentations. Fungal Biol. 2014, 118, 979–989. [Google Scholar] [CrossRef] [PubMed]
- Xie, N.; Liu, Q.; Chen, F. Deletion of pigR gene in Monascus ruber leads to loss of pigment production. Biotechnol. Lett. 2013, 35, 1425–1432. [Google Scholar] [CrossRef] [PubMed]
- Wan, Y.; Zhang, J.; Han, H.; Li, L.; Liu, Y.; Gao, M. Citrinin-producing capacity of Monascus purpureus in response to low-frequency magnetic fields. Process Biochem. 2017, 53, 25–29. [Google Scholar] [CrossRef]
- Evelin, H.; Kapoor, R.; Giri, B. Arbuscular mycorrhizal fungi in alleviation of salt stress: A review. Ann. Bot-London. 2009, 104, 1263–1280. [Google Scholar] [CrossRef]
- Bardgett, R.D.; Saggar, S. Effects of heavy metal contamination on the short-term decomposition of labelled [14C] glucose in a pasture soil. Soil Biol. Biochem. 1994, 26, 727–733. [Google Scholar] [CrossRef]
- Wang, Y.; Lu, Z.; Sun, K.; Zhu, W. Effects of high salt stress on secondary metabolite production in the marine-derived fungus Spicaria elegans. Mar. Drugs. 2011, 9, 535–542. [Google Scholar] [CrossRef]
- Hajjaj, H.; Klaebe, A.; Loret, M.O.; Goma, G.; Blanc, P.J.; Francois, J. Biosynthetic pathway of citrinin in the filamentous fungus Monascus ruber as revealed by 13C nuclear magnetic resonance. Appl. Environ. Microbiol. 1999, 65, 311–314. [Google Scholar]
- Manzoni, M.; Rollini, M. Biosynthesis and biotechnological production of statins by filamentous fungi and application of these cholesterol-lowering drugs. Appl. Microbiol. Biotechnol. 2002, 58, 555–564. [Google Scholar] [PubMed]
- Fountain, J.C.; Prasad, B.; Nayak, S.N.; Liming, Y.; Pandey, M.K.; Vinay, K. Responses of Aspergillus flavus to oxidative stress are related to fungal development regulator, antioxidant enzyme, and secondary metabolite biosynthetic gene expression. Front. Microbiol. 2016, 7, 2048. [Google Scholar] [CrossRef] [PubMed]
- Benmoussa-Dahmen, I.; Chtourou, H.; Rezgui, F.; Sayadi, S.; Dhouib, A. Salinity stress increases lipid, secondary metabolites and enzyme activity in Amphora subtropica and Dunaliella sp. for biodiesel production. Bioresource Technol. 2016, 218, 816–825. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Liu, Q.; Zhang, Y.; Cui, Q.; Liang, Y. Effect of acid phosphatase produced by Trichoderma asperellum Q1 on growth of Arabidopsis under salt stress. J. Integr. Agr. 2017, 16, 1341–1346. [Google Scholar] [CrossRef]
- Guimarães, L.H.S.; Júnior, A.B.; Jorge, J.A.; Terenzi, H.F.; Polizeli, M.L.T.M. Purification and biochemical characterization of a mycelial alkaline phosphatase without DNAase activity produced by Aspergillus caespitosus. Folia Microbiol. 2007, 52, 231–236. [Google Scholar] [CrossRef]
- Strausfeld, U.; Labbé, J.C.; Fesquet, D.; Cavadore, J.C.; Picard, A.; Sadhu, K.; Russell, P.; Dorée, M. Dephosphorylation and activation of a p34cdc2/cyclin B complex in vitro by human CDC25 protein. Nature 1991, 351, 242–245. [Google Scholar] [CrossRef] [PubMed]
- Millar, J.B.; Russell, P.; Dixon, J.E.; Guan, K.L. Negative regulation of mitosis by two functionally overlapping PTPase in fission yeast. EMBO J. 1992, 11, 4943–4952. [Google Scholar] [CrossRef]
- Pugh, D.; Cawson, R.A. The cytochemical localization of acid hydrolases in four common fungi. Cell. Mol. Biol. 1977, 22, 125–132. [Google Scholar]
- Reyes, F.; Villanueva, P.; Alfonso, C. Comparative study of acid and alkaline phosphatase during the autolysis of filamentous fungi. Lett. Appl. Microbiol. 2010, 10, 175–177. [Google Scholar] [CrossRef]
- Gianinazzi-Pearson, V.; Gianinazzi, S. Enzymatic studies on the metabolism of vesicular-arbuscular mycorrhiza II. Soluble alkaline phosphatase specific to mycorrhizal infection in onion roots. Physiol. Plant Pathol. 1978, 12, 45–48. [Google Scholar] [CrossRef]
| Name | Sequences(5’→3’) | Descriptions |
|---|---|---|
| ctnA-S | AACCATGGAGGCATTGGACC | For qRT-PCR analysis of ctnA |
| ctnA-A | CCTTGTCGGTCACACCGAAT | |
| pksCT-S | TTCTGACACGACCATCAGCC | For qRT-PCR analysis of pksCT |
| pksCT-A | ACGACGACGAGTGTCAGTTC | |
| pksPT-S | GGCAACCTTCAGTCCGCTAT | For qRT-PCR analysis of pksPT |
| pksPT-A | GATCAGTGCGATGCCATGTG | |
| pigR-S | ACTCTGGAAAGCTGCTTCGG | For qRT-PCR analysis of pigR |
| pigR-A | GGACGTTCTGGATGGCGTAT | |
| GAPDH-S | CAAGCTCACTGGCATGTCTATG | For qRT-PCR analysis of GAPDH |
| GAPDH-A | AAGTTCGAGTTGAGGGCGATA |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhen, Z.; Xiong, X.; Liu, Y.; Zhang, J.; Wang, S.; Li, L.; Gao, M. NaCl Inhibits Citrinin and Stimulates Monascus Pigments and Monacolin K Production. Toxins 2019, 11, 118. https://doi.org/10.3390/toxins11020118
Zhen Z, Xiong X, Liu Y, Zhang J, Wang S, Li L, Gao M. NaCl Inhibits Citrinin and Stimulates Monascus Pigments and Monacolin K Production. Toxins. 2019; 11(2):118. https://doi.org/10.3390/toxins11020118
Chicago/Turabian StyleZhen, Zhixin, Xiaoqian Xiong, Yingbao Liu, Jialan Zhang, Shaojin Wang, Li Li, and Mengxiang Gao. 2019. "NaCl Inhibits Citrinin and Stimulates Monascus Pigments and Monacolin K Production" Toxins 11, no. 2: 118. https://doi.org/10.3390/toxins11020118
APA StyleZhen, Z., Xiong, X., Liu, Y., Zhang, J., Wang, S., Li, L., & Gao, M. (2019). NaCl Inhibits Citrinin and Stimulates Monascus Pigments and Monacolin K Production. Toxins, 11(2), 118. https://doi.org/10.3390/toxins11020118






