DI/LC–MS/MS-Based Metabolome Analysis of Plasma Reveals the Effects of Sequestering Agents on the Metabolic Status of Dairy Cows Challenged with Aflatoxin B1
Abstract
:1. Introduction
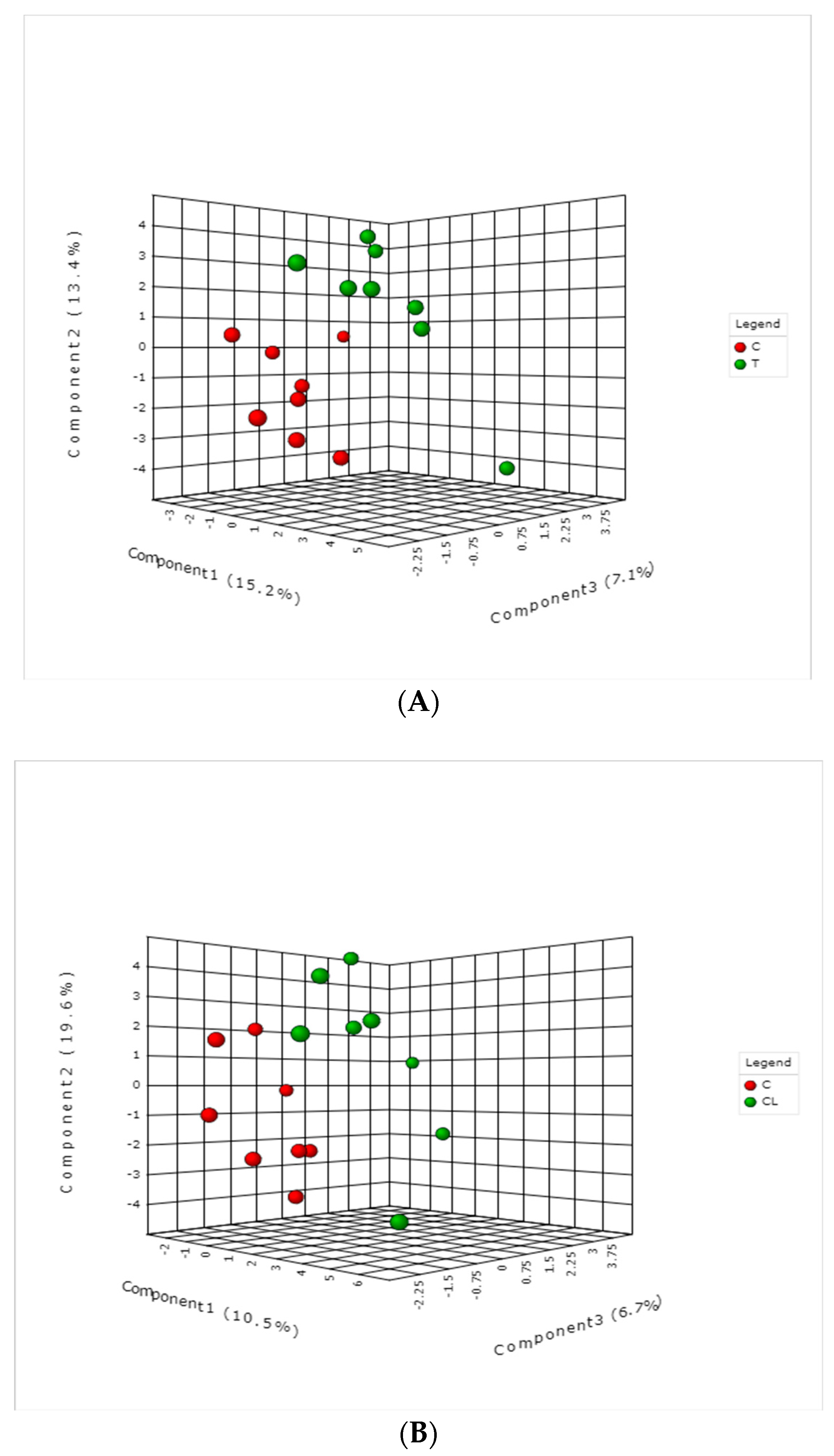
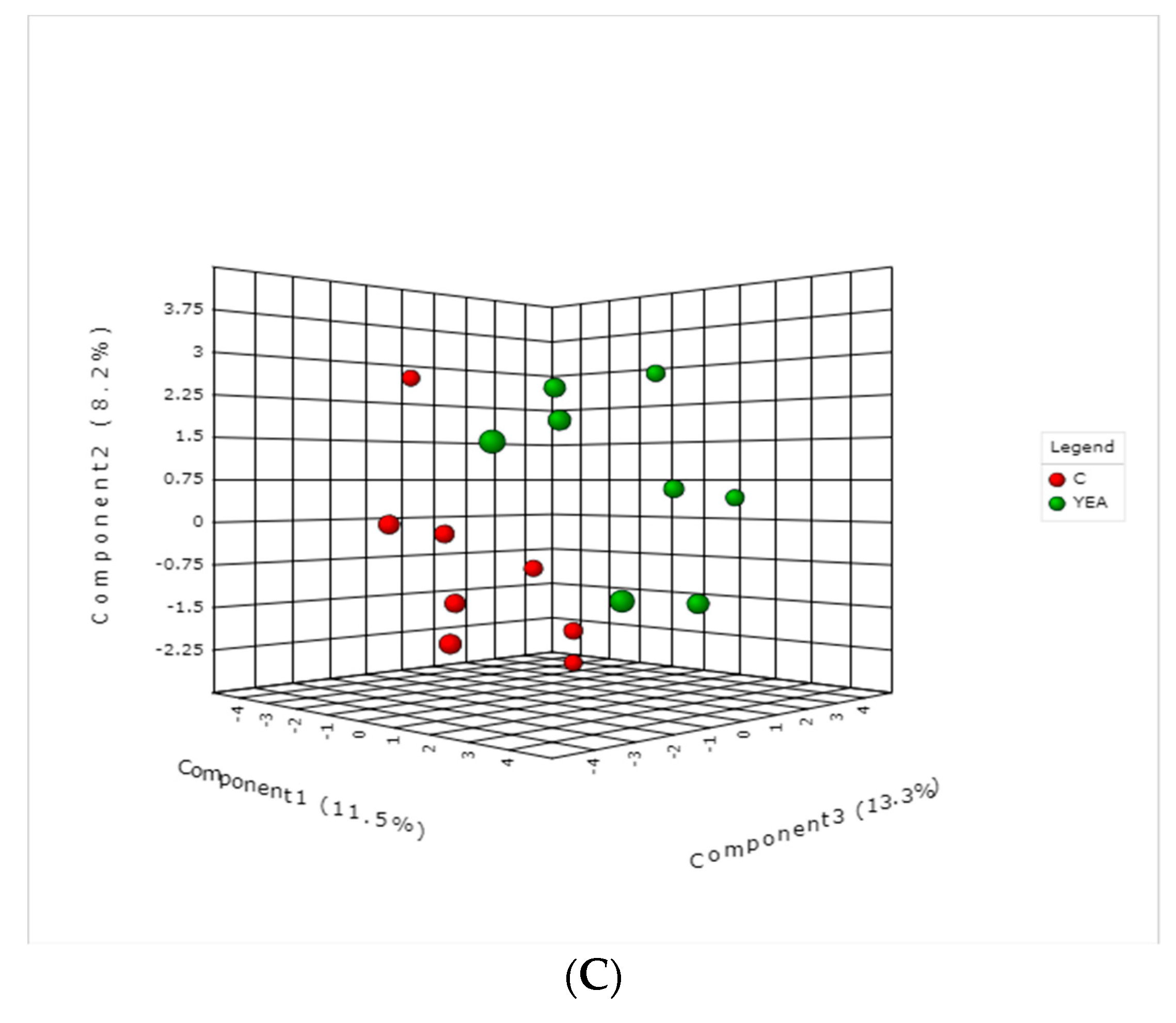
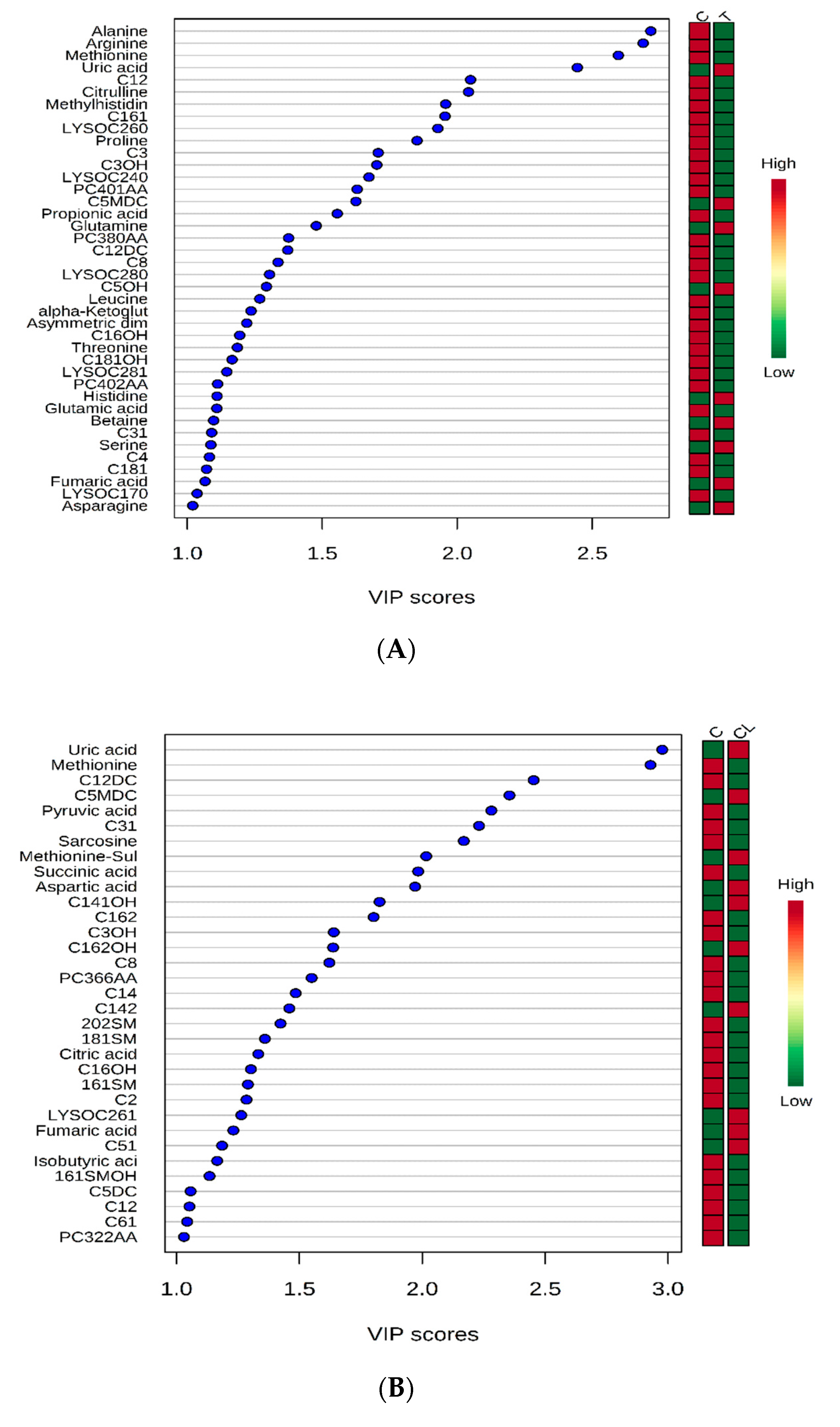
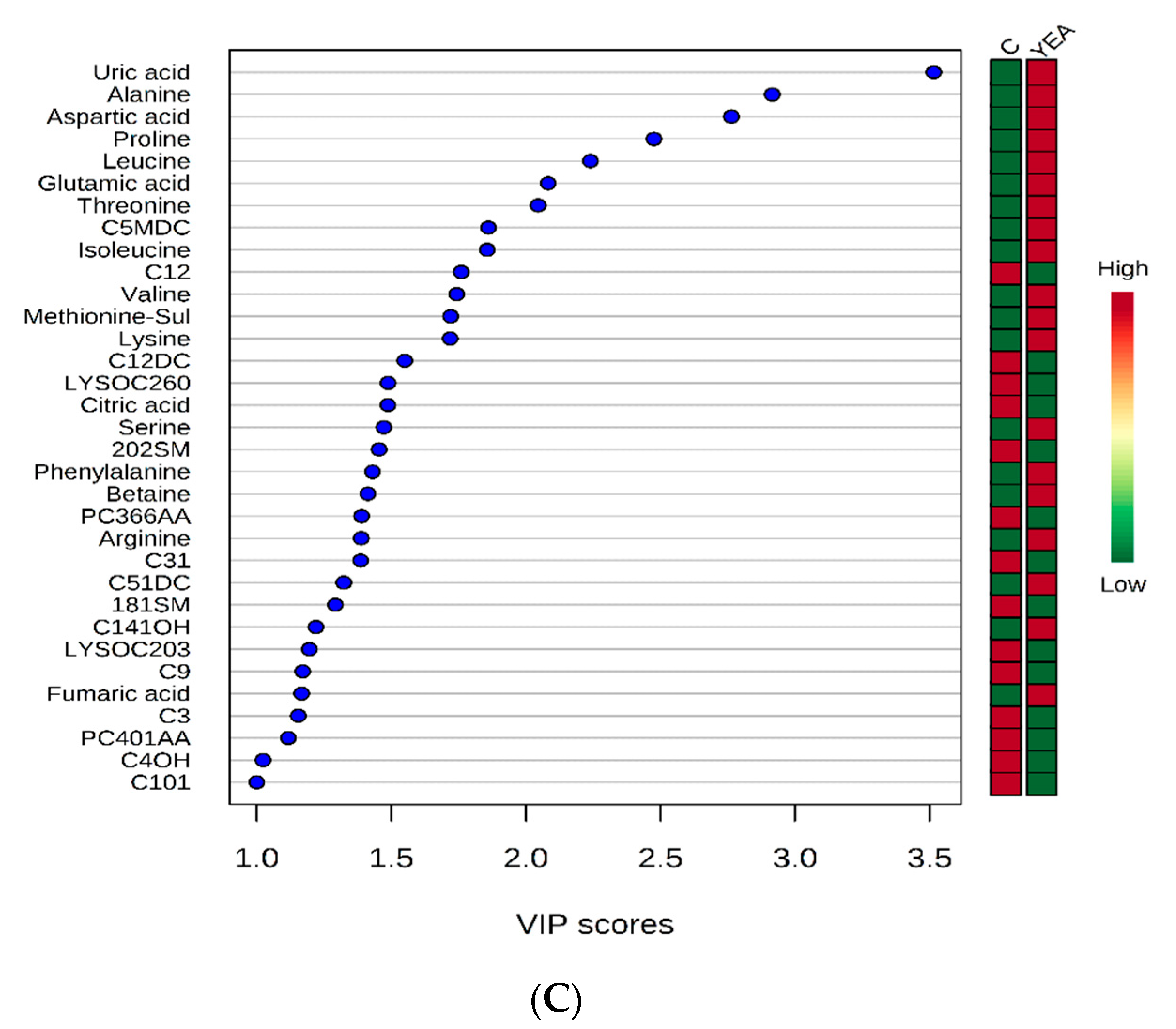
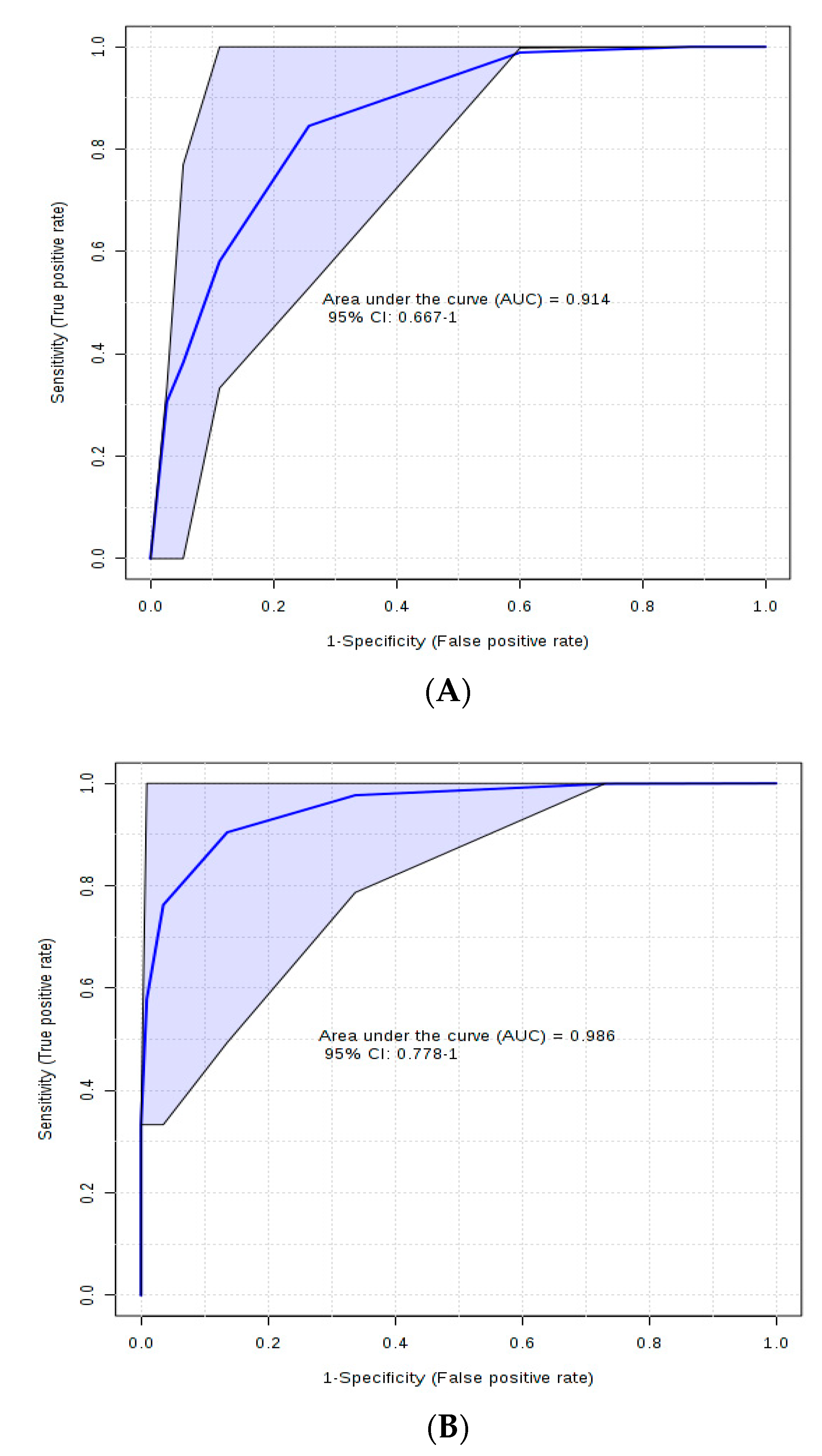
2. Results and Discussions
3. Materials and Methods
4. Data and Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Gallo, A.; Giuberti, G.; Frisvad, J.C.; Bertuzzi, T.; Nielsen, K.F. Review on mycotoxin issues in ruminants: Occurrence in forages, effects of mycotoxin ingestion on health status and animal performance and ractical strategies to counteract their negative effects. Toxins 2015, 7, 3057–3111. [Google Scholar] [CrossRef] [PubMed]
- Abdallah, M.F.; Girgin, G.; Baydar, T.; Krska, R.; Sulyok, M. Occurrence of multiple mycotoxins and other fungal metabolites in animal feed and maize samples from Egypt using LC-MS/MS. J. Sci. Food Agric. 2017, 97, 4419–4428. [Google Scholar] [CrossRef] [PubMed]
- Richard, E.; Heutte, N.; Bouchart, V.; Garon, D. Evaluation of fungal contamination and mycotoxin production in maize silage. Anim. Feed Sci. Technol. 2009, 148, 309–320. [Google Scholar] [CrossRef]
- Ogunade, I.M.; Arriola, K.G.; Jiang, Y.; Driver, J.P.; Staples, C.R.; Adesogan, A.T. Effects of 3 sequestering agents on milk aflatoxin M1 concentration and the performance and immune status of dairy cows fed diets artificially contaminated with aflatoxin B1. J. Dairy Sci. 2016, 99, 6263–6273. [Google Scholar] [CrossRef] [PubMed]
- Herzallah, S.M. Determination of aflatoxins in eggs, milk, meat and meat products using HPLC fluorescent and UV detectors. Food Chem. 2009, 114, 1141–1146. [Google Scholar] [CrossRef]
- Jiang, Y.; Ogunade, I.M.; Kim, D.H.; Li, X.; Pech-Cervantes, A.A.; Arriola, K.G.; Oliveira, A.S.; Driver, J.P.; Ferraretto, L.F.; Staples, C.R.; et al. Effect of sequestering agents based on a Saccharomyces cerevisiae fermentation product and clay on the health and performance of lactating dairy cows challenged with dietary aflatoxin B1. J. Dairy Sci. 2018, 101, 3008–3020. [Google Scholar] [CrossRef]
- Xiong, J.L.; Wang, Y.M.; Nennich, Y.M.; Li, Y.; Liu, J.X. Transfer of dietary aflatoxin B1 to milk aflatoxin M1 and effect of inclusion of adsorbent in the diet of dairy cows. J. Dairy Sci. 2015, 98, 2545–2554. [Google Scholar] [CrossRef]
- Wang, Q.; Zhang, Y.; Zheng, N.; Guo, L.; Song, X.; Zhao, S.; Wang, J. Biological system responses of dairy cows to aflatoxin B1 exposure revealed with metabolomic changes in multiple biofluids. Toxins 2019, 11, 77. [Google Scholar] [CrossRef]
- Peng, B.; Li, H.; Peng, X.X. Functional metabolomics: from biomarker discovery to metabolome reprogramming. Protein Cell 2015, 6, 628–637. [Google Scholar] [CrossRef]
- Patel, S.; Ahmed, S. Emerging field of metabolomics: big promise for cancer biomarker identification and drug discovery. J. Pharm. Biomed. Anal. 2015, 107, 63–74. [Google Scholar] [CrossRef]
- Gibbons, H.; O’Gorman, A.; Brennan, L. Metabolomics as a tool in nutritional research. Curr. Opin. Lipidol. 2015, 26, 30–34. [Google Scholar] [CrossRef] [PubMed]
- Ogunade, I.; Jiang, M.; Adeyemi, J.A.; Oliveira, A.; Vyas, D.; Adesogan, A.T. Biomarker of Aflatoxin Ingestion: 1H NMR-Based Plasma Metabolomics of Dairy Cows Fed Aflatoxin B1 with or without Sequestering Agents. Toxins 2018, 10, 545. [Google Scholar] [CrossRef] [PubMed]
- Veenstra, T.D. Metabolomics: the final frontier? Genome Med. 2012, 4, 40. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Dervishi, E.; Dunn, S.M.; Mandal, R.; Liu, P.; Han, B.; Wishart, D.S.; Ametaj, B.N. Metabotyping reveals distinct metabolic alterations in ketotic cows and identifies early predictive serum biomarkers for the risk of disease. Metabolomics 2017, 13, 43. [Google Scholar] [CrossRef]
- Garvican, L.; Cajone, F.; Rees, K.R. The mechanism of action of aflatoxin B1 on protein synthesis; Observations on malignant, viral transformed and untransformed cells in culture. Chem. Biol. Interact. 1973, 7, 39–50. [Google Scholar] [CrossRef]
- Pate, R.T.; Paulus Compart, D.M.; Cardoso, F.C. Aluminosilicate clay improves production responses and reduces inflammation during an aflatoxin challenge in lactating Holstein cows. J. Dairy Sci. 2018, 101, 11421–11434. [Google Scholar] [CrossRef]
- Doelman, J.; Kim, J.J.M.; Carson, M.; Metcalf, J.A.; Cant, J.P. Branched chain amino acid and lysine deficiencies exert different effects on mammary translational regulation. J. Dairy Sci. 2015, 98, 7846–7855. [Google Scholar] [CrossRef]
- Powell, J.D.; Pollizzi, K.N.; Heikamp, E.B.; Horton, M.R. Regulation of immune responses by mTOR. Ann. Rev. Immunol. 2012, 30, 39. [Google Scholar] [CrossRef]
- Zhao, F.; Wu, T.; Zhang, H.; Loor, J.J.; Wang, M.; Peng, A.; Wang, H. Jugular infusion of arginine has a positive effect on antioxidant mechanisms in lactating dairy cows challenged intravenously with lipopolysaccharide. J. Anim. Sci. 2018, 96, 3850–3855. [Google Scholar] [CrossRef]
- Queiroz, O.C.M.; Han, J.H.; Staples, C.R.; Adesogan, A.T. Effect of adding a mycotoxin-sequestering agent on milk aflatoxin M1 concentration and the performance and immune response of dairy cattle fed an aflatoxin B1-contaminated diet. J. Dairy Sci. 2012, 95, 5901–5908. [Google Scholar] [CrossRef]
- Chaytor, A.C.; See, M.T.; Hansen, J.A.; de Souza, A.L.; Middleton, T.F.; Kim, S.W. Effects of chronic exposure of diets with reduced concentrations of aflatoxin and deoxynivalenol on growth and immune status of pigs. J. Anim. Sci. 2011, 89, 124–135. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, C.K. Glucose balance in cattle. Florida Ruminant Nutrition Symposium. Available online: http://dairy.ifas. ufl.edu/rns/2005/Reynold Accessed online:s.pdf (accessed on 14 November 2005).
- Pietri, A.; Bertuzzi, T.; Pallaroni, L.; Piva, G. Occurrence of mycotoxins and ergosterol in maize harvested over 5 years in Northern Italy. Food Addit. Contam. 2004, 21, 479–487. [Google Scholar] [CrossRef] [PubMed]
- Pantaya, D.; Morgavi, D.P.; Silberberg, M.; Chaucheyras-Durand, F.; Martin, C.; Wiryawan, K.G.; Boudra, H. Bioavailability of aflatoxin B1 and ochratoxin A, but not fumonisin B1 or deoxynivalenol, is increased in starch-induced low ruminal pH in nonlactating dairy cows. J. Dairy Sci. 2016, 99, 9759–9767. [Google Scholar] [PubMed]
- Xia, J.; Wishart, D.S. Using metaboAnalyst 3.0 for comprehensive metabolomics data analysis. Curr. Prot. Bioinform. 2016, 55, 10–14. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Chang, G.; Zhang, K.; Xu, L.; Jin, D.; Bilal, M.S.; Shen, X. Rumen-derived lipopolysaccharide provoked inflammatory injury in the liver of dairy cows fed a high-concentrate diet. Oncotarget 2017, 8, 46769–46780. [Google Scholar] [CrossRef]
- Erasmus, L.J.; Botha, P.M.; Kistner, A. Effect of yeast culture supplement on production, rumen fermentation, and duodenal nitrogen flow in dairy cows. J. Dairy Sci. 1992, 75, 3056–3065. [Google Scholar] [CrossRef]
- Bach, A.; Iglesias, C.; Devant, M. Daily rumen pH pattern of loose-housed dairy cattle as affected by feeding pattern and live yeast supplementation. Anim. Feed Sci. Technol. 2007, 136, 146–153. [Google Scholar] [CrossRef]
- Callaway, E.S.; Martin, S.A. Effects of a Saccharomyces cerevisiae culture on ruminal bacteria that utilize lactate and digest cellulose. J. Dairy Sci. 1997, 80, 2035–2044. [Google Scholar] [CrossRef]
- NRC. Nutrient Requirements of Dairy Cattle, 7th ed.; National Academies Press: Washington, DC, USA, 2001. [Google Scholar]
- Walsh, B.H.; Broadhurst, D.I.; Mandal, R.; Wishart, D.S.; Boylan, G.B.; Kenny, L.C.; Murray, D.M. The metabolomic Profile of umbilical cord blood in neonatal hypoxic ischaemic encephalopathy. PLoS ONE 2012, 7, e50520. [Google Scholar] [CrossRef]
- Hailemariam, D.; Zhang, G.; Mandal, R.; Wishart, D.S.; Ametaj, B.N. Identification of serum metabolites associated with the risk of metritis in transition dairy cows. Can. J. Anim. Sci. 2018, 98, 525–537. [Google Scholar] [CrossRef]
| Item | Treatment 2 | SEM | p-Value | |||
|---|---|---|---|---|---|---|
| Control | T | CL | YEA | |||
| Alanine | 337 b | 284 c | 334 b | 462 a | 18.1 | 0.01 |
| Valine | 331 y | 312 y | 325 y | 387 x | 14.5 | 0.06 |
| Proline | 143 b | 127 b | 137 b | 194 a | 7.47 | 0.01 |
| Threonine | 152 b | 136 b | 140 b | 172 a | 7.82 | 0.01 |
| Leucine | 281 b | 227 b | 284 b | 362 a | 14.2 | 0.01 |
| Isoleucine | 129 b | 131 b | 128 b | 148 a | 6.30 | 0.03 |
| Aspartic acid | 23.5 y | 26.9 xy | 29.5 xy | 43.5 x | 5.62 | 0.09 |
| Glutamic acid | 236 b | 219 b | 206 b | 305 a | 9.95 | 0.01 |
| Arginine | 128 b | 96 c | 131 b | 159 a | 10.4 | 0.02 |
| Phenylalanine | 68.4 b | 71.1 b | 69.4 b | 83.1 a | 4.37 | 0.05 |
| Citrulline | 73.6 xy | 62.6 y | 81.0 x | 71.6 xy | 4.81 | 0.07 |
| Sarcosine | 1.83 xy | 1.74 xy | 1.50 y | 1.95 x | 0.12 | 0.06 |
| Lysine | 159 b | 173 ab | 170 ab | 194 a | 9.05 | 0.05 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ogunade, I.; Jiang, Y.; Pech Cervantes, A. DI/LC–MS/MS-Based Metabolome Analysis of Plasma Reveals the Effects of Sequestering Agents on the Metabolic Status of Dairy Cows Challenged with Aflatoxin B1. Toxins 2019, 11, 693. https://doi.org/10.3390/toxins11120693
Ogunade I, Jiang Y, Pech Cervantes A. DI/LC–MS/MS-Based Metabolome Analysis of Plasma Reveals the Effects of Sequestering Agents on the Metabolic Status of Dairy Cows Challenged with Aflatoxin B1. Toxins. 2019; 11(12):693. https://doi.org/10.3390/toxins11120693
Chicago/Turabian StyleOgunade, Ibukun, Yun Jiang, and Andres Pech Cervantes. 2019. "DI/LC–MS/MS-Based Metabolome Analysis of Plasma Reveals the Effects of Sequestering Agents on the Metabolic Status of Dairy Cows Challenged with Aflatoxin B1" Toxins 11, no. 12: 693. https://doi.org/10.3390/toxins11120693
APA StyleOgunade, I., Jiang, Y., & Pech Cervantes, A. (2019). DI/LC–MS/MS-Based Metabolome Analysis of Plasma Reveals the Effects of Sequestering Agents on the Metabolic Status of Dairy Cows Challenged with Aflatoxin B1. Toxins, 11(12), 693. https://doi.org/10.3390/toxins11120693






