Combined Effect of Light and Temperature on the Production of Saxitoxins in Cylindrospermopsis raciborskii Strains
Abstract
:1. Introduction
2. Results
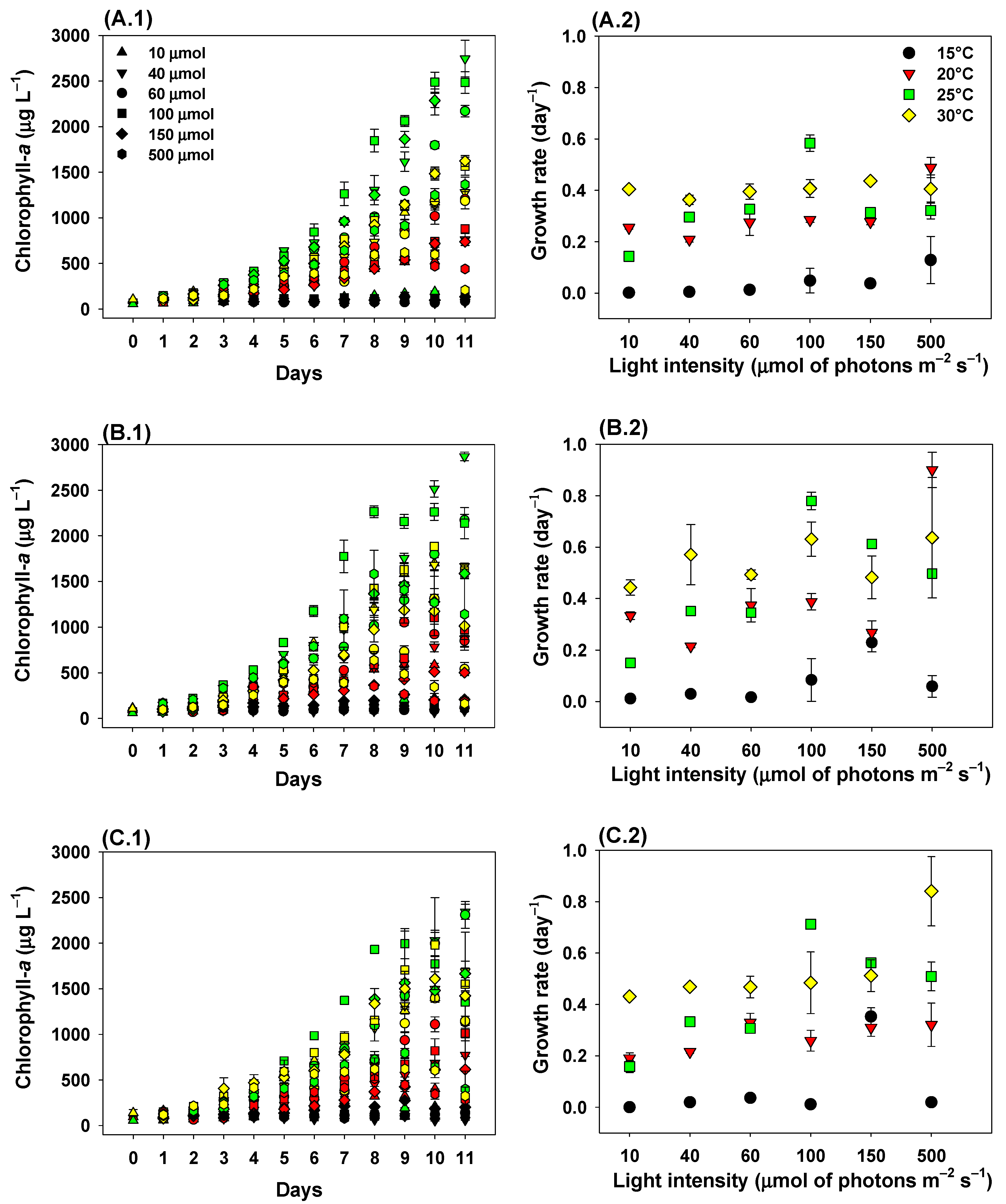
2.1. Growth Rates
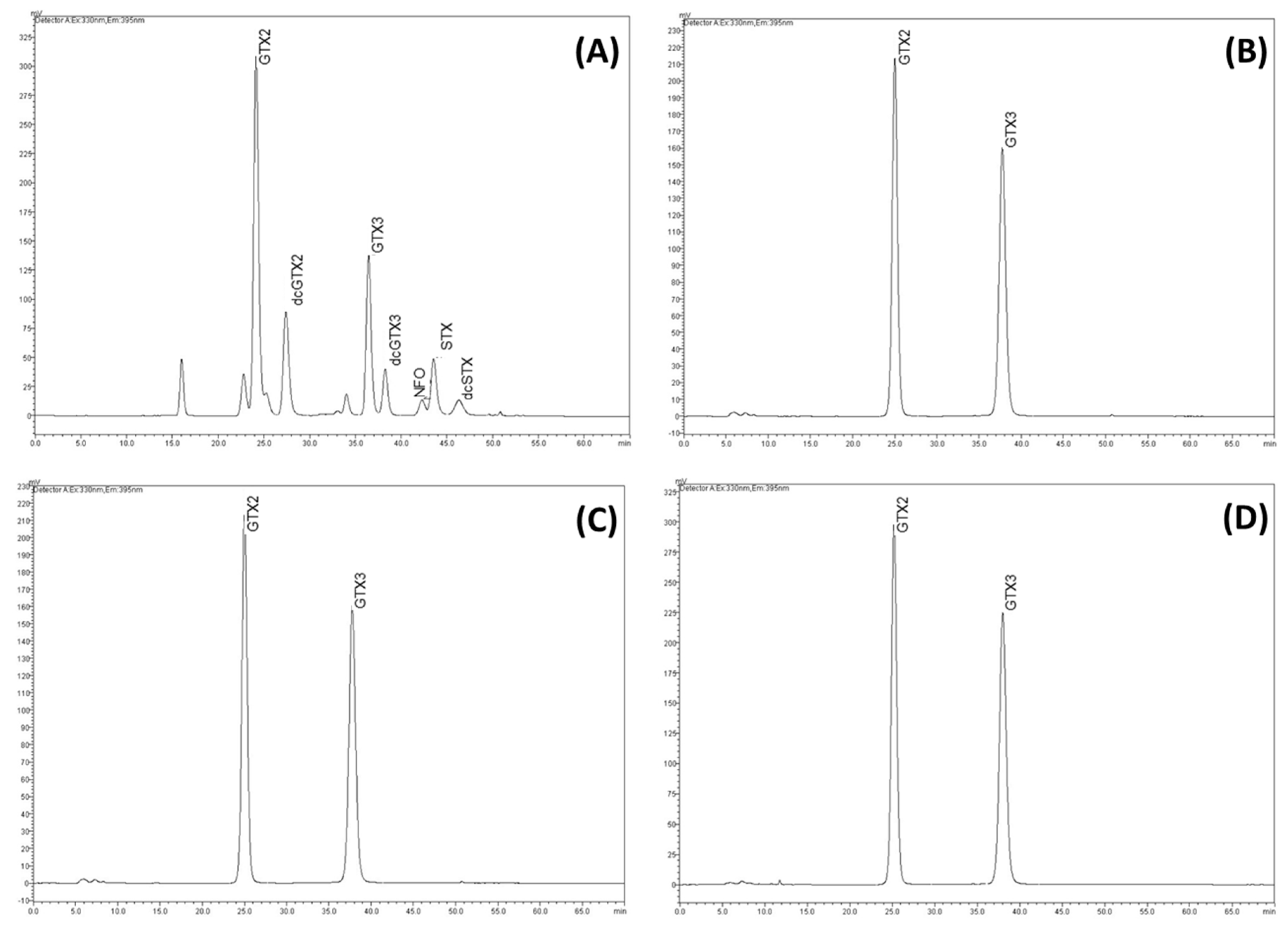
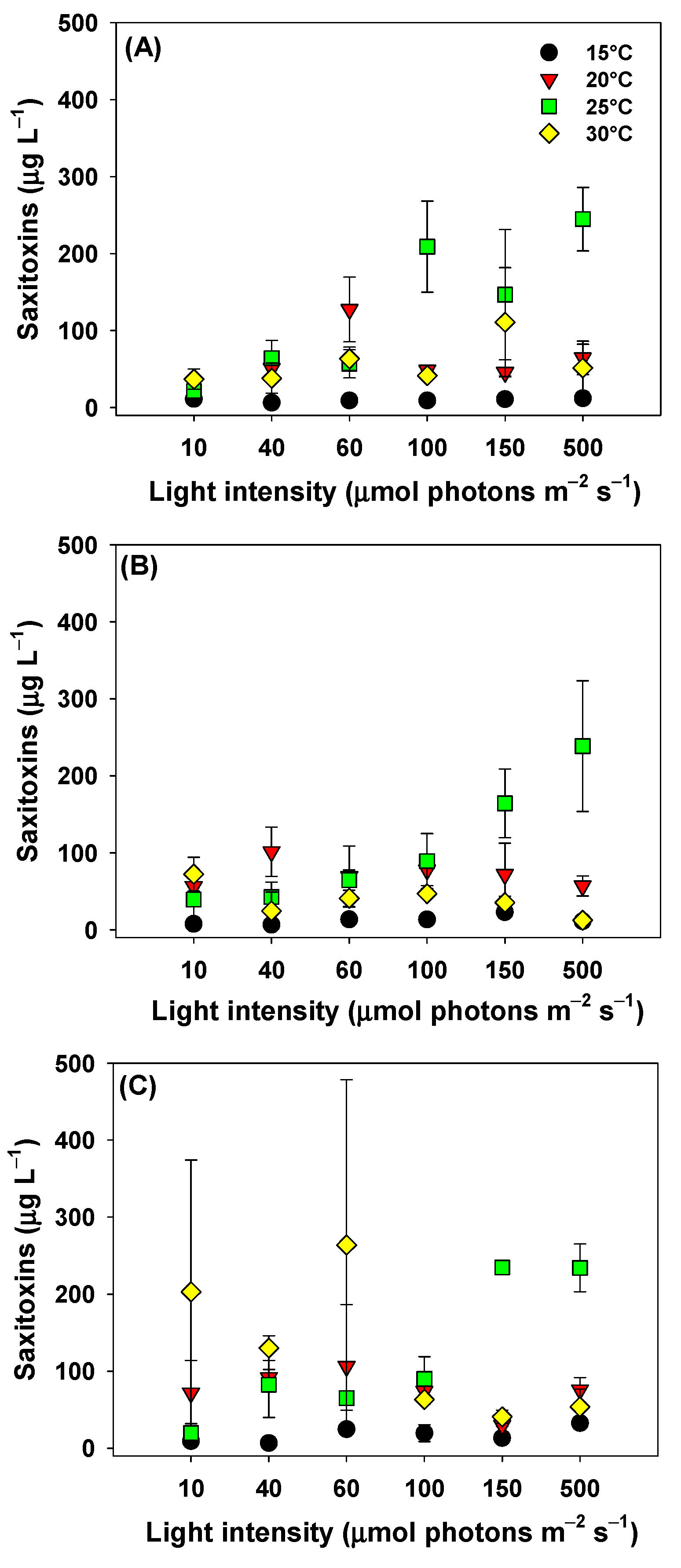
2.2. Saxitoxins (STXs) Concentrations
2.3. STXs Cellular Quota
3. Discussion
4. Conclusions
5. Material and Methods
Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Antunes, J.T.; Leão, P.N.; Vasconcelos, V.M. Cylindrospermopsis raciborskii: Review of the distrbution, phylogeography, and ecophysiology of global invasive species. Front. Microbiol. 2015, 6, 473. [Google Scholar] [CrossRef] [PubMed]
- Briand, J.F.; Leboulanger, C.; Humbert, J.F.; Bernard, C.; Dufour, P. Cylindrospermopsis raciborskii (Cyanobacteria) invasion at mid-latitudes: Selection, wide physiological tolerance, or global warming? J. Phycol. 2004, 40, 231–238. [Google Scholar] [CrossRef]
- Bittencourt-Oliveira, M.C.; Buch, B.; Hereman, T.C.; Arruda-Neto, J.D.T.; Moura, A.N.V.; Zocchi, S.S. Effects of light intensity and temperature on Cylindrospermopsis raciborskii (Cyanobacteria) with straight and coiled trichomes: Growth rate and morphology. Braz. J. Biol. 2012, 72, 343–351. [Google Scholar] [CrossRef]
- Bonilla, S.; Gonzáles-Piana, M.; Soares, M.C.S.; Huszar, V.L.M.; Becker, V.; Somma, A.; Marinho, M.M.; Kokociński, M.; Dokulil, M.; Antoniades, D.; et al. The success of the cyanobacterium Cylindrospermopsis raciborskii in freshwaters is enhanced by the combined effects of light intensity and temperature. J. Limnol. 2016, 75, 606–617. [Google Scholar] [CrossRef]
- Bonilla, S.; Aubriot, L.; Soares, M.C.S.; Gonzáles-Piana, M.; Fabre, A.; Huszar, V.L.M.; Lürling, M.; Antoniades, D.; Padisál, J.; Kruk, C. What drives the distribution of the Bloom-forming cyanobacteria Planktothrix agardhii and Cylindrospermopsis raciborskii? FEMS Microbiol. Ecol. 2012, 79, 594–607. [Google Scholar] [CrossRef]
- Dittmann, E.; Fewer, D.P.; Neilan, B.A. Cyanobacterial toxins: Biosynthetic routes and evolutionary roots. FEMS Microbiol. Rev. 2013, 37, 23–43. [Google Scholar] [CrossRef] [PubMed]
- Carneiro, R.L.; Santos, M.E.V.D.; Pacheco, A.B.F.; Azevedo, S.M.F.O. Effects of light intensity and light quality on growth and circadian rhythm of saxitoxins production in Cylindrospermopsis raciborskii (Cyanobacteria). J. Plankton Res. 2009, 31, 481–488. [Google Scholar] [CrossRef]
- Molica, R.; Onodera, H.; García, C.; Rivas, M.; Andrinolo, D.; Nascimento, S.; Meguro, H.; Oshima, Y.; Azevedo, S.; Lagos, N. Toxins in the freshwater cyanobacterium Cylindrospermopsis raciborskii, isolated from Tabocas reservoir in Caruaru, Pernambuco. Braz. Phycol. 2002, 41, 606–611. [Google Scholar] [CrossRef]
- Lagos, N.; Onodera, H.; Zagatto, P.A.; Andrinolo, D.; Azevedo, S.M.F.O.; Oshima, Y. The first evidence of paralytic shelfish toxins in the freshwater cyanobacterium Cylindrospermopsis raciborskii, isolated from Brazil. Toxicon 1999, 37, 1359–1373. [Google Scholar] [CrossRef]
- Carneiro, R.L.; Silva, A.P.R.; Magalhães, V.F.; Azevedo, S.M.F.O. Use of the cell quota and chlorophyll content for normalization of cylindropermopsin produced by two Cylindrospermopsis raciborskii strains grown under different light intensities. Ecotoxicol. Environ. Contam. 2013, 8, 93–100. [Google Scholar]
- Burns, J. Toxic cyanobacteria in Florida Waters. In Cyanobacterial Harmful Algal Blooms: State of the Science and Research Needs; Springer: New York, NY, USA, 2008; Volume 619, pp. 127–137. [Google Scholar]
- Yilmaz, M.; Philips, E.J. Toxicity and genetic diversity of Cylindrospermopsis raciborskii in Florida, USA. Lake Reserv. Manag. 2011, 27, 235–244. [Google Scholar] [CrossRef]
- Carneiro, R.; Pacheco, A.B.F.; Azevedo, S.M.F.O. Growth and saxitoxin production by Cylindrospermopsis raciborskii (Cyanobacteria) correlate with water hardness. Mar. Drugs 2013, 11, 2949–2963. [Google Scholar] [CrossRef]
- Costa, S.M.; Ferrão-Filho, A.S.; Azevedo, S.M.F.O. Effects of saxitoxin and non-saxitoxin-producing strains of the cyanobacterium Cylindrospermopsis raciborskii on the fitness of temperate and tropical cladocerans. Harmful Algae 2013, 28, 55–63. [Google Scholar] [CrossRef]
- Wiese, M.; D’agostino, P.M.; Mihali, T.K.; Moffitt, M.C.; Neilan, B.A. Neurotoxic alkaloids: Saxitoxin and its analogs. Mar. Drugs 2010, 8, 2185–2211. [Google Scholar] [CrossRef] [PubMed]
- Murray, S.A.; Mihali, T.K.; Neilan, B.A. Extraordinary conservation, gene loss, and positive selection in the evolution of an ancient neurotoxin. Mol. Biol. Evol. 2011, 28, 1173–1182. [Google Scholar] [CrossRef] [PubMed]
- Hackett, J.D.; Wisecaver, J.H.; Brosnahan, M.L.; Kulis, D.M.; Anderson, D.M.; Bhattacharya, D.; Plumley, F.G.; Erdner, D.L. Evolution of saxitoxin synthesis in cyanobacteria and dinoflagellates. Mol. Biol. Evol. 2013, 30, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Hoff-Risseti, C.; Dörr, F.A.; Schaker, P.D.C.; Pinto, E.; Wener, V.R.; Fiore, M.F. Cylindrospermopsin and saxitoxin synthetase genes in Cylindrospermopsis raciborskii strains from brazilian freshwater. PLoS ONE 2013, 8, e74238. [Google Scholar] [CrossRef] [PubMed]
- Boopathi, T.; Ki, J.S. Impact of environmental factors on the regulation of cyanotoxin production. Toxins 2014, 6, 1951–1978. [Google Scholar] [CrossRef]
- Brentano, D.M.; Giehl, E.L.; Petrucio, M.M. Abiotic variables affect STX concentration in a meso-oligotrophic subtropical coastal lake dominated by Cylindrospermopsis raciborskii (Cyanophyceae). Harmful Algae 2016, 56, 22–28. [Google Scholar] [CrossRef]
- Casali, S.P.; Santos, A.C.A.; Falco, P.B.; Calijuri, M.C. Influence of environmental variables on saxitoxin yields by Cylindrospermopsis raciborskii in a mesotrophic subtropical reservoir. J. Water Health 2017, 15, 509–518. [Google Scholar] [CrossRef]
- Castro, D.; Vera, D.; Lagos, N.; Garcia, C.; Vasquez, M. The effect of temperature on growth and production of paralytic shellfish poisoning toxins by the cyanobacterium Cylindrospermopsis raciborskii C10. Toxicon 2004, 44, 483–489. [Google Scholar] [CrossRef] [PubMed]
- Rangel, L.M.; Ger, K.A.; Silva, L.H.S.; Soares, M.C.S.; Faassen, E.J.; Lürling, M. Toxicity overrides morphology on Cylindrospermopsis raciborskii grazing resistance to the calanoid copepod Eudiaptomus gracilis. Microb. Ecol. 2016, 71, 835–844. [Google Scholar] [CrossRef] [PubMed]
- Xiao, M.; Willis, A.; Burford, M. Differences in cyanobacterial strain responses to light and temperature reflect species plasticity. Harmful Algae 2017, 62, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Padisák, J.; Reynolds, C.S. Selection of phytoplankton associations in Lake Balaton, Hungary, in response to eutrophication and restoration measures, with special reference to the cyanoprokaryotes. Hydrobiologia 1998, 384, 41–53. [Google Scholar] [CrossRef]
- Pierangelini, M.; Stojkovic, S.; Orr, P.T.; Beardall, J. Photosynthetic characteristics of two Cylindrospermopsis raciborskii strains differing in their toxicity. J. Phycol. 2014, 50, 292–302. [Google Scholar] [CrossRef] [PubMed]
- Soares, M.C.S.; Lürling, M.; Huszar, V.L.M. Growth and temperature-related phenotypic plasticity in the cyanobacterium Cylindrospermopsis raciborskii. Phycol. Res. 2013, 61, 61–67. [Google Scholar] [CrossRef]
- Bouvy, M.; Molica, R.; de Oliveira, S.; Marinho, M.M.; Beker, B. Dynamics of a toxic cyanobacterial bloom (Cylindrospermopsis raciborskii) in a shallow reservoir in the semi-arid region of northeast Brazil. Aquat. Microb. Ecol. 1999, 20, 285–297. [Google Scholar] [CrossRef]
- Soares, M.C.S.; Rocha, M.I.A.; Marinho, M.M.; Azevedo, S.M.F.O.; Branco, C.W.C.; Huszar, V.L.M. Changes in species composition during annual cyanobacterial dominance in a tropical reservoir: Physical factors, nutrients and grazing effects. Aquat. Microb. Ecol. 2009, 57, 137–149. [Google Scholar] [CrossRef]
- Reynolds, C.S. Ecology of Phytoplankton: Ecology, Biodiversity, and Conservation; Cambridge University Press: Cambridge, UK, 2006. [Google Scholar]
- Coles, J.F.; Jones, R.C. Effect of temperature on photosynthesis-light response and growth of four phytoplankton species isolated from a tidal freshwater river. J. Phycol. 2000, 36, 7–16. [Google Scholar] [CrossRef]
- Kellmann, R.; Neilan, B.A. Biochemical characterization paralytic shellfish toxin biosynthesis in vitro. J. Phycol. 2007, 43, 497–508. [Google Scholar] [CrossRef]
- IPCC (2014). Climate Change 2014: Synthesis Report. Contribution of Working Groups, I., II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Pachauri, R.K., Meyer, L.A., Eds.; IPCC: Geneva, Switzerland, 2014; 151p. [Google Scholar]
- Peng, G.; Martin, R.M.; Dearth, S.P.; Sun, X.; Boyer, G.L.; Campagna, S.R.; Lin, S.; Wilhelm, S.W. Seasonally relevant cool temperatures interact with n chemistry to increase microcystins produced in lab cultures of Microcystis aeruginosa NIES-843. Environ. Sci. Technol. 2018, 52, 4127–4136. [Google Scholar] [CrossRef] [PubMed]
- Lürling, M.; Beekman, W. Grazer-induced defenses in Scenedesmus (Chlorococcales; Chlorophyceae): Coenobium and spine formation. Phycologia 1999, 28, 368–376. [Google Scholar] [CrossRef]
- Ritchie, R.J. Consistent sets of spectrophotometric chlorophyll equations for acetone, methanol and ethanol solvents. Photosynth. Res. 2006, 89, 27–41. [Google Scholar] [CrossRef]
- Briand, L.E.; Guillard, R.R.L.; Murphy, L.S. A method for the rapid and precise determination of acclimated phytoplankton reproduction rates. J. Plankton Res. 1981, 3, 193–201. [Google Scholar] [CrossRef]
- Diener, M.; Erler, K.; Hiller, S.; Christian, B.; Luckas, B. Determination of Paralytic Shellfish Poisoning (PSP) toxins in dietary supplements by application of a new HPLC/FD method. Eur. Food Res. Technol. 2006, 224, 147–151. [Google Scholar] [CrossRef]
- Diener, M.; Erler, K.; Christian, B.; Luckas, B. Application of a new zwitterionic hydrophillic interaction chromatography column for determination of paralytic shellfish poisoning toxins. J. Sep. Sci. 2007, 30, 1821–1826. [Google Scholar] [CrossRef] [PubMed]
| Source of Variation | DF | F | P |
|---|---|---|---|
| CYLCAM-01 | |||
| Light | 5 | 15.93 | <0.001 |
| Temperature | 3 | 270.40 | <0.001 |
| Light × Temperature | 15 | 5.18 | <0.001 |
| Residual | 48 | ||
| Total | 71 | ||
| CYLCAM-02 | |||
| Light | 5 | 22.84 | <0.001 |
| Temperature | 3 | 116.17 | <0.001 |
| Light × Temperature | 15 | 11.18 | <0.001 |
| Residual | 48 | ||
| Total | 71 | ||
| CYLCAM-03 | |||
| Light | 5 | 29.68 | <0.001 |
| Temperature | 3 | 195.63 | <0.001 |
| Light × Temperature | 15 | 13.72 | <0.001 |
| Residual | 48 | ||
| Total | 71 |
| Source of Variation | DF | F | P |
|---|---|---|---|
| CYLCAM-01 | |||
| Light | 5 | 5.583 | <0.001 |
| Temperature | 3 | 27.787 | <0.001 |
| Light × Temperature | 15 | 5.292 | <0.001 |
| Residual | 48 | ||
| Total | 71 | ||
| CYLCAM-02 | |||
| Light | 5 | 2.546 | <0.041 |
| Temperature | 3 | 24.874 | <0.001 |
| Light × Temperature | 15 | 5.162 | <0.001 |
| Residual | 46 | ||
| Total | 69 | ||
| CYLCAM-03 | |||
| Light | 5 | 0.726 | 0.607 |
| Temperature | 3 | 7.644 | <0.001 |
| Light × Temperature | 15 | 2.798 | 0.004 |
| Residual | 47 | ||
| Total | 70 |
| Source of Variation | DF | F | P |
|---|---|---|---|
| CYLCAM-01 | |||
| Light | 5 | 1.42 | 0.232 |
| Temperature | 3 | 18.03 | <0.001 |
| Light × Temperature | 15 | 3.58 | <0.001 |
| Residual | 48 | ||
| Total | 71 | ||
| CYLCAM-02 | |||
| Light | 5 | 8.70 | <0.001 |
| Temperature | 3 | 19.87 | <0.001 |
| Light × Temperature | 15 | 6.87 | <0.001 |
| Residual | 48 | ||
| Total | 71 | ||
| CYLCAM-03 | |||
| Light | 5 | 1.83 | 0.123 |
| Temperature | 3 | 2.35 | 0.083 |
| Light × Temperature | 15 | 1.82 | 0.058 |
| Residual | 48 | ||
| Total | 71 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mesquita, M.C.B.; Lürling, M.; Dorr, F.; Pinto, E.; Marinho, M.M. Combined Effect of Light and Temperature on the Production of Saxitoxins in Cylindrospermopsis raciborskii Strains. Toxins 2019, 11, 38. https://doi.org/10.3390/toxins11010038
Mesquita MCB, Lürling M, Dorr F, Pinto E, Marinho MM. Combined Effect of Light and Temperature on the Production of Saxitoxins in Cylindrospermopsis raciborskii Strains. Toxins. 2019; 11(1):38. https://doi.org/10.3390/toxins11010038
Chicago/Turabian StyleMesquita, Marcella C. B., Miquel Lürling, Fabiane Dorr, Ernani Pinto, and Marcelo M. Marinho. 2019. "Combined Effect of Light and Temperature on the Production of Saxitoxins in Cylindrospermopsis raciborskii Strains" Toxins 11, no. 1: 38. https://doi.org/10.3390/toxins11010038
APA StyleMesquita, M. C. B., Lürling, M., Dorr, F., Pinto, E., & Marinho, M. M. (2019). Combined Effect of Light and Temperature on the Production of Saxitoxins in Cylindrospermopsis raciborskii Strains. Toxins, 11(1), 38. https://doi.org/10.3390/toxins11010038







