Assessment of Toxigenic Fusarium Species and Their Mycotoxins in Brewing Barley Grains
Abstract
1. Introduction
2. Results
2.1. Water Activity and Identification of Isolated Fungi
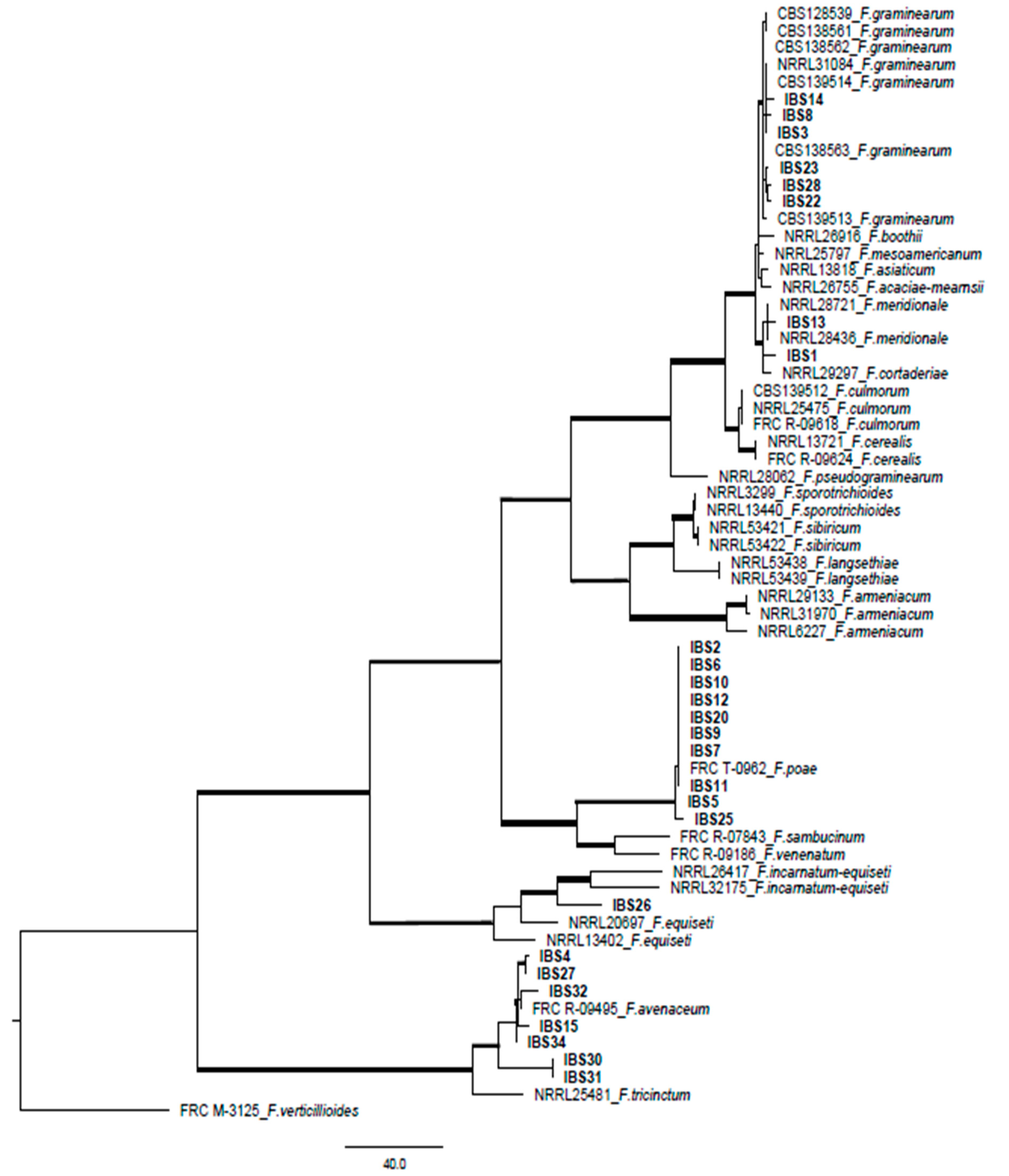
2.2. Phylogenetic Study
2.3. Mycotoxins Analysis
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Barley Samples
5.2. Water Activity
5.3. Mycobiota and Identification of Fungi
5.4. Identification of the Fusarium Species
5.5. Phylogenetic Analysis
5.6. Mycotoxin Analysis
5.6.1. Chemicals and Reagents
5.6.2. DON and ZEA Extraction
5.7. Production of DON and ZEA by the Strains
5.8. Chromatography Conditions
Method Validation
5.9. Data Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Desjardins, A.E. Fusarium Mycotoxins: Chemistry, Genetics and Biology. Plant Pathol. 2007, 56, 337. [Google Scholar] [CrossRef]
- Ma, L.-J.; Geiser, D.M.; Proctor, R.H.; Rooney, A.P.; O’Donnell, K.; Trail, F.; Gardiner, D.M.; Manners, J.M.; Kazan, K. Fusarium pathogenomics. Annu. Rev. Microbiol. 2013, 67, 399–416. [Google Scholar] [CrossRef]
- FAO. FAO Statistical Yearbook 2013: World Food and Agriculture; FAO: Rome, Italy, 2013; ISBN 9789251073964. [Google Scholar]
- WHO. International Programme on Chemical Safety (IPCS): Environmental Health Criteria 105 Selected Mycotoxins: Ochratoxins, Trichothecenes, Ergot. Trichothecenes; World Health Organization: Geneva, Switzerland, 1990; pp. 71–164. [Google Scholar]
- Pestka, J.J. Deoxynivalenol: Mechanisms of action, human exposure, and toxicological relevance. Arch. Toxicol. 2010, 84, 663–679. [Google Scholar] [CrossRef] [PubMed]
- Zinedine, A.; Soriano, J.M.; Moltó, J.C.; Mañes, J. Review on the toxicity, occurrence, metabolism, detoxification, regulations and intake of zearalenone: An oestrogenic mycotoxin. Food Chem. Toxicol. 2007, 45, 1–18. [Google Scholar] [CrossRef] [PubMed]
- International Agency for Research on Cancer. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; International Agency for Research on Cancer: Lyon CEDEX, France, 2002; Volume 96. [Google Scholar] [CrossRef]
- Pascari, X.; Ramos, A.J.; Marín, S.; Sanchís, V. Mycotoxins and beer. Impact of beer production process on mycotoxin contamination. A review. Food Res. Int. 2018, 103, 121–129. [Google Scholar] [CrossRef]
- Bullerman, L.B.; Bianchini, A. Stability of mycotoxins during food processing. Int. J. Food Microbiol. 2007, 119, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Piacentini, K.C.; Savi, G.D.; Olivo, G.; Scussel, V.M. Quality and occurrence of deoxynivalenol and fumonisins in craft beer. Food Control 2015, 50, 925–929. [Google Scholar] [CrossRef]
- Piacentini, K.C.; Rocha, L.O.; Savi, G.D.; Carnielli-Queiroz, L.; Almeida, F.G.; Minella, E.; Corrêa, B. Occurrence of deoxynivalenol and zearalenone in brewing barley grains from Brazil. Mycotoxin Res. 2018. [Google Scholar] [CrossRef]
- Wegulo, S.N.; Baenziger, P.S.; Hernandez Nopsa, J.; Bockus, W.W.; Hallen-Adams, H. Management of Fusarium head blight of wheat and barley. Crop Prot. 2015, 73, 100–107. [Google Scholar] [CrossRef]
- Brasil Limites Máximos Tolerados (LMT) Para Micotoxinas; Agência Nacional Vigilância Sanitária: ANVISA, Brazil, 2017.
- European Commission. Commission Regulation (EC) No 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs. Off. J. Eur. Union 2006, L364, 5–24. [Google Scholar]
- Karlsson, I.; Friberg, H.; Kolseth, A.K.; Steinberg, C.; Persson, P. Agricultural factors affecting Fusarium communities in wheat kernels. Int. J. Food Microbiol. 2017, 252, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Hofer, K.; Geißinger, C.; König, C.; Gastl, M.; Hückelhoven, R.; Heß, M.; Coleman, A.D. Influence of Fusarium isolates on the expression of barley genes related to plant defense and malting quality. J. Cereal Sci. 2016, 69, 17–24. [Google Scholar] [CrossRef]
- Silva, J.J.; Viaro, H.P.; Ferranti, L.S.; Oliveira, A.L.M.; Ferreira, J.M.; Ruas, C.F.; Ono, E.Y.S.; Fungaro, M.H.P. Genetic structure of Fusarium verticillioides populations and occurrence of fumonisins in maize grown in Southern Brazil. Crop Prot. 2017, 99, 160–167. [Google Scholar] [CrossRef]
- Choi, S.; Jun, H.; Bang, J.; Chung, S.H.; Kim, Y.; Kim, B.S.; Kim, H.; Beuchat, L.R.; Ryu, J.H. Behaviour of Aspergillus flavus and Fusarium graminearum on rice as affected by degree of milling, temperature, and relative humidity during storage. Food Microbiol. 2015, 46, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Garcia, D.; Barros, G.; Chulze, S.; Ramos, A.J.; Sanchis, V.; Marín, S. Impact of cycling temperatures on Fusarium verticillioides and Fusarium graminearum growth and mycotoxins production in soybean. J. Sci. Food Agric. 2012, 92, 2952–2959. [Google Scholar] [CrossRef] [PubMed]
- Llorens, A.; Mateo, R.; Hinojo, M.J.; Valle-Algarra, F.M.; Jiménez, M. Influence of environmental factors on the biosynthesis of type B trichothecenes by isolates of Fusarium spp. from Spanish crops. Int. J. Food Microbiol. 2004, 94, 43–54. [Google Scholar] [CrossRef] [PubMed]
- Fleurat-Lessard, F. Integrated management of the risks of stored grain spoilage by seedborne fungi and contamination by storage mould mycotoxins—An update. J. Stored Prod. Res. 2017, 71, 22–40. [Google Scholar] [CrossRef]
- Pitt, J.I.; Hocking, A.D. Fungi and Food Spoilage; Springer: Berlin/Heidelberg, Germany, 2009; ISBN 9780387922065. [Google Scholar]
- Savi, G.D.; Piacentini, K.C.; Rocha, L.O.; Carnielli-Queiroz, L.; Furtado, B.G.; Scussel, R.; Zanoni, E.T.; Machado-de-Ávila, R.A.; Corrêa, B.; Angioletto, E. Incidence of toxigenic fungi and zearalenone in rice grains from Brazil. Int. J. Food Microbiol. 2018, 270. [Google Scholar] [CrossRef]
- Stakheev, A.A.; Khairulina, D.R.; Zavriev, S.K. Four-locus phylogeny of Fusarium avenaceum and related species and their species-specific identification based on partial phosphate permease gene sequences. Int. J. Food Microbiol. 2016, 225, 27–37. [Google Scholar] [CrossRef]
- Beccari, G.; Caproni, L.; Tini, F.; Uhlig, S.; Covarelli, L. Presence of Fusarium species and other toxigenic fungi in malting barley and multi-mycotoxin analysis by liquid chromatography-high-resolution mass spectrometry. J. Agric. Food Chem. 2016, 64, 4390–4399. [Google Scholar] [CrossRef]
- Hietaniemi, V.; Rämö, S.; Yli-Mattila, T.; Jestoi, M.; Peltonen, S.; Kartio, M.; Sieviläinen, E.; Koivisto, T.; Parikka, P. Updated survey of Fusarium species and toxins in Finnish cereal grains. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2016, 33, 831–848. [Google Scholar] [CrossRef] [PubMed]
- Astolfi, P.; dos Santos, J.; Schneider, L.; Gomes, L.B.; Silva, C.N.; Tessmann, D.J.; Del Ponte, E.M. Molecular survey of trichothecene genotypes of Fusarium graminearum species complex from barley in Southern Brazil. Int. J. Food Microbiol. 2011, 148, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Ibáñez-Vea, M.; González-Peñas, E.; Lizarraga, E.; López de Cerain, A. Co-occurrence of mycotoxins in Spanish barley: A statistical overview. Food Control 2012, 28, 295–298. [Google Scholar] [CrossRef]
- Juan, C.; Ritieni, A.; Mañes, J. Occurrence of Fusarium mycotoxins in Italian cereal and cereal products from organic farming. Food Chem. 2013, 141, 1747–1755. [Google Scholar] [CrossRef] [PubMed]
- Juan, C.; Berrada, H.; Maes, J.; Oueslati, S. Multi-mycotoxin determination in barley and derived products from Tunisia and estimation of their dietary intake. Food Chem. Toxicol. 2017, 103, 148–156. [Google Scholar] [CrossRef] [PubMed]
- 401/2006/EC Commision Regulation (EC) No 401/2006 of 23 February 2006 laying down the methods of sampling and analysis for the official control of the levels of mycotoxins in foodstuffs. Off. J. Eur. Union 2006, L70, 12–34.
- Bolechová, M.; Benešová, K.; Běláková, S.; Čáslavský, J.; Pospíchalová, M.; Mikulíková, R. Determination of seventeen mycotoxins in barley and malt in the Czech Republic. Food Control 2015, 47, 108–113. [Google Scholar] [CrossRef]
- Běláková, S.; Benešová, K.; Čáslavský, J.; Svoboda, Z.; Mikulíková, R. The occurrence of the selected fusarium mycotoxins in czech malting barley. Food Control 2014, 37, 93–98. [Google Scholar] [CrossRef]
- Piacentini, K.C.; Savi, G.D.; Pereira, M.E.V.; Scussel, V.M. Fungi and the natural occurrence of deoxynivalenol and fumonisins in malting barley (Hordeum vulgare L.). Food Chem. 2015, 187. [Google Scholar] [CrossRef]
- Poreda, A.; Czarnik, A.; Zdaniewicz, M.; Jakubowski, M.; Antkiewicz, P. Corn grist adjunct—Application and influence on the brewing process and beer quality. J. Inst. Brew. 2014, 120, 77–81. [Google Scholar] [CrossRef]
- Piacentini, K.C.; Rocha, L.O.; Fontes, L.C.; Carnielli, L.; Reis, T.A.; Corrêa, B. Mycotoxin analysis of industrial beers from Brazil: The influence of fumonisin B1and deoxynivalenol in beer quality. Food Chem. 2017, 218. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, M.S.; Rocha, A.; Sulyok, M.; Krska, R.; Mallmann, C.A. Natural mycotoxin contamination of maize (Zea mays L.) in the South region of Brazil. Food Control 2016, 1–6. [Google Scholar] [CrossRef]
- Zhang, H.; Dong, M.; Yang, Q.; Apaliya, M.T.; Li, J.; Zhang, X. Biodegradation of zearalenone by Saccharomyces cerevisiae: Possible involvement of ZEN responsive proteins of the yeast. J. Proteom. 2016, 143, 416–423. [Google Scholar] [CrossRef] [PubMed]
- Gareis, M.; Bauer, J.; Thiem, J.; Plank, G.; Grabley, S.; Gedek, B. Cleavage of Zearalenone-Glycoside, a “Masked” Mycotoxin, during Digestion in Swine. J. Vet. Med. Ser. B 1990, 37, 236–240. [Google Scholar] [CrossRef]
- Efsa Scientific Opinion on the risks for animal and public health related to the presence of Alternaria toxins in feed and food. EFSA J. 2011, 9, 97. [CrossRef]
- Wu, L.; Qiu, L.; Zhang, H.; Sun, J.; Hu, X.; Wang, B. Optimization for the production of deoxynivalenol and zearalenone by Fusarium graminearum using response surface methodology. Toxins (Basel) 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- AOAC. Official Methods of Analysis of AOAC International; AOAC: Rockville, MD, USA, 2005; ISBN 0935584544. [Google Scholar]
- Silva, N.D.; Junqueira, V.C.; Silveira, N.F.; Taniwaki, M.; Santos, R.F.; Gomes, R.A. Manual de Métodos de Análise Microbiológica de Alimentos e Água, 4th ed.; Português: Sao Paulo, Barzil, 2010. [Google Scholar]
- Degola, F.; Berni, E.; Dall’Asta, C.; Spotti, E.; Marchelli, R.; Ferrero, I.; Restivo, F.M. A multiplex RT-PCR approach to detect aflatoxigenic strains of Aspergillus flavus. J. Appl. Microbiol. 2007, 203, 409–427. [Google Scholar] [CrossRef] [PubMed]
- Geiser, D.M.; Jiménez-Gasco, M.D.M.; Kang, S.; Makalowska, I.; Veeraraghavan, N.; Ward, T.J.; Zhang, N.; Kuldau, G.A.; O’Donnell, K. FUSARIUM-ID v. 1.0: A DNA sequence database for identifying Fusarium. Eur. J. Plant Pathol. 2004, 110, 473–479. [Google Scholar] [CrossRef]
- O’Donnell, K.; Ward, T.J.; Geiser, D.M.; Kistler, H.C.; Aoki, T. Genealogical concordance between the mating type locus and seven other nuclear genes supports formal recognition of nine phylogenetically distinct species within the Fusarium graminearum clade. Fungal Genet. Biol. 2004, 41, 600–623. [Google Scholar] [CrossRef]
- Swofford, D.L. PAUP* phylogenetic analysis using parsimony (*and other methods). Version 4.0b10. Sinauer Assoc. 2002. [Google Scholar] [CrossRef]
- Swofford, D.L. Phylogenetic Analysis Using Parsimony. Options 2002, 42, 294–307. [Google Scholar] [CrossRef]
- O’Donnell, K.; Rooney, A.P.; Proctor, R.H.; Brown, D.W.; McCormick, S.P.; Ward, T.J.; Frandsen, R.J.N.; Lysøe, E.; Rehner, S.A.; Aoki, T.; et al. Phylogenetic analyses of RPB1 and RPB2 support a middle Cretaceous origin for a clade comprising all agriculturally and medically important fusaria. Fungal Genet. Biol. 2013, 52, 20–31. [Google Scholar] [CrossRef] [PubMed]
- Al-Taher, F.; Cappozzo, J.; Zweigenbaum, J.; Lee, H.J.; Jackson, L.; Ryu, D. Detection and quantitation of mycotoxins in infant cereals in the U.S. market by LC-MS/MS using a stable isotope dilution assay. Food Control 2017, 72, 27–35. [Google Scholar] [CrossRef]
- Savi, G.D.; Vitorino, V.; Bortoluzzi, A.J.; Scussel, V.M. Effect of zinc compounds on Fusarium verticillioides growth, hyphae alterations, conidia, and fumonisin production. J. Sci. Food Agric. 2013, 93, 3395–3402. [Google Scholar] [CrossRef]
- Savi, G.D.; Bortoluzzi, A.J.; Scussel, V.M. Antifungal properties of Zinc-compounds against toxigenic fungi and mycotoxin. Int. J. Food Sci. Technol. 2013, 48, 1834–1840. [Google Scholar] [CrossRef]
- Njumbe Ediage, E.; Van Poucke, C.; De Saeger, S. A multi-analyte LC-MS/MS method for the analysis of 23 mycotoxins in different sorghum varieties: The forgotten sample matrix. Food Chem. 2015, 177, 397–404. [Google Scholar] [CrossRef] [PubMed]
- EC Commission regulation (EC) 401/2006 of 23 february 2006 laying down the methods of sampling and analysis for the official control of the levels of mycotoxins in foodstuffs. Off. J. Eur. Communities 2000, L269, 1–15.
| Species Complex * | Frequency % | Fusarium Species | % Samples Contaminated |
|---|---|---|---|
| FSAMSC | 56.25 | F. graminearum | 23.4 |
| F. poae | 17.2 | ||
| F. meridionale | 1.6 | ||
| FTSC | 31.25 | F. avenaceum | 23.4 |
| FFSC | 8.33 | F. proliferatum | 3.1 |
| F. verticillioides | 3.1 | ||
| FIESC | 2 | F. incarnatum-equiseti | 1.6 |
| FOSC | 2 | F. oxysporum | 1.6 |
| Number Samples | Deoxynivalenol | Zearalenone | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Positive Samples/% * | Range of Positive Samples (µg/kg) | Mean ± SD (µg/kg) | Median (µg/kg) | Positive Samples/% * | Range of Positive Samples (µg/kg) | Mean ± SD (µg/kg) | Median (µg/kg) | ||
| Barley grains | 64 | 58/90.6 | 45.95–1155.21 | 147.65 ± 167.16 | 98.68 | 56/87.5 | 82.41–423.71 | 123.24 ± 45.29 | 119.26 |
| Mycotoxin | Retention Time (min) | Precursor ion (m/z) | Product ion (m/z) * | CE (V) | TubeLens |
|---|---|---|---|---|---|
| DON | 2.19 | 297 [M + H] | 203Q | 17 | 71 |
| 175C | 18 | 71 | |||
| 91C | 39 | 71 | |||
| ZEA | 6.55 | 319 [M + H] | 283Q | 11 | 79 |
| 187C | 25 | 79 | |||
| 185C | 20 | 79 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piacentini, K.C.; Rocha, L.O.; Savi, G.D.; Carnielli-Queiroz, L.; De Carvalho Fontes, L.; Correa, B. Assessment of Toxigenic Fusarium Species and Their Mycotoxins in Brewing Barley Grains. Toxins 2019, 11, 31. https://doi.org/10.3390/toxins11010031
Piacentini KC, Rocha LO, Savi GD, Carnielli-Queiroz L, De Carvalho Fontes L, Correa B. Assessment of Toxigenic Fusarium Species and Their Mycotoxins in Brewing Barley Grains. Toxins. 2019; 11(1):31. https://doi.org/10.3390/toxins11010031
Chicago/Turabian StylePiacentini, Karim C., Liliana O. Rocha, Geovana D. Savi, Lorena Carnielli-Queiroz, Livia De Carvalho Fontes, and Benedito Correa. 2019. "Assessment of Toxigenic Fusarium Species and Their Mycotoxins in Brewing Barley Grains" Toxins 11, no. 1: 31. https://doi.org/10.3390/toxins11010031
APA StylePiacentini, K. C., Rocha, L. O., Savi, G. D., Carnielli-Queiroz, L., De Carvalho Fontes, L., & Correa, B. (2019). Assessment of Toxigenic Fusarium Species and Their Mycotoxins in Brewing Barley Grains. Toxins, 11(1), 31. https://doi.org/10.3390/toxins11010031





