Light Chain Diversity among the Botulinum Neurotoxins
Abstract
1. Background on Botulinum Neurotoxin
2. BoNT Structure and Function
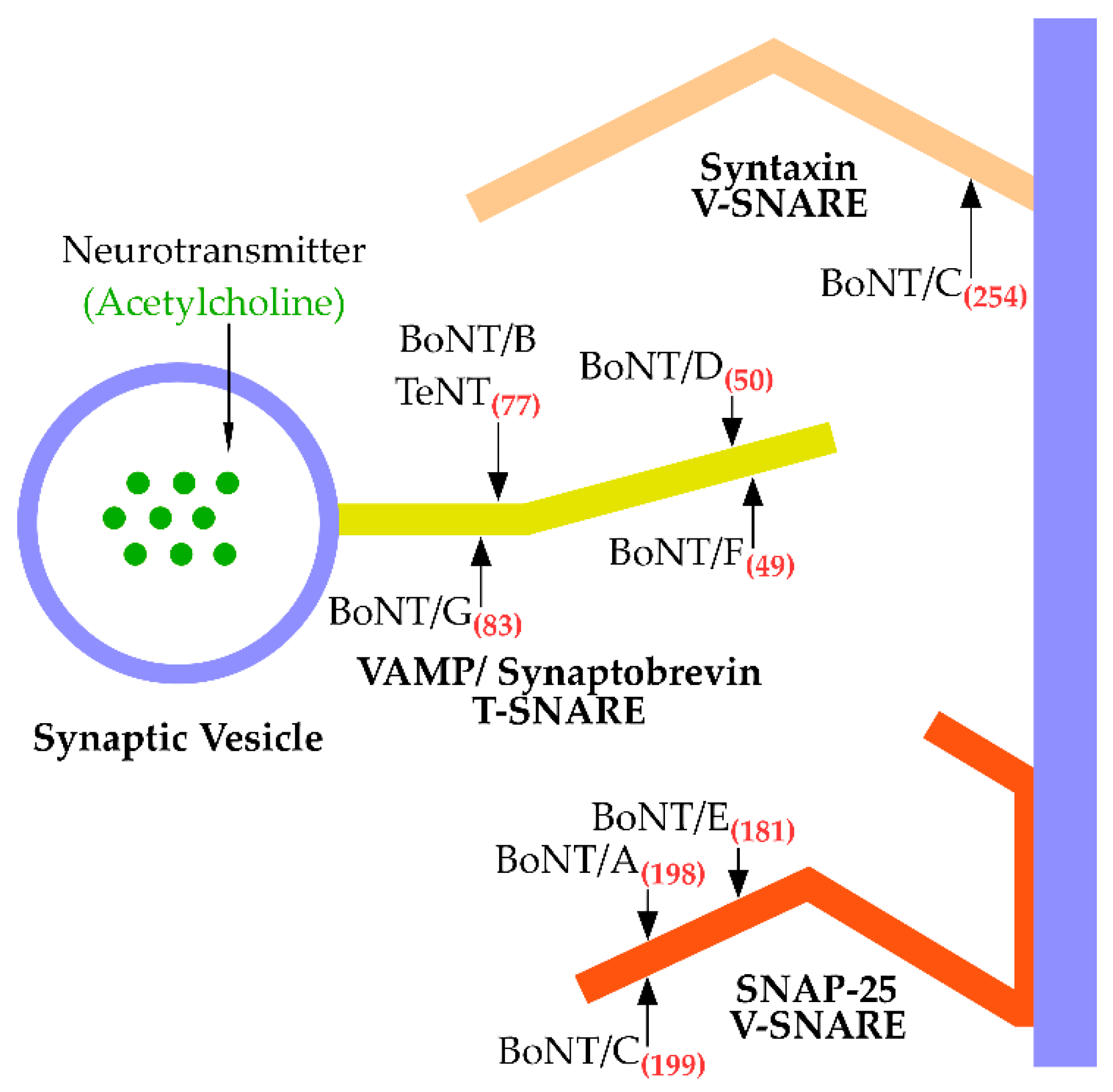
3. Cleavage of BoNT Substrates
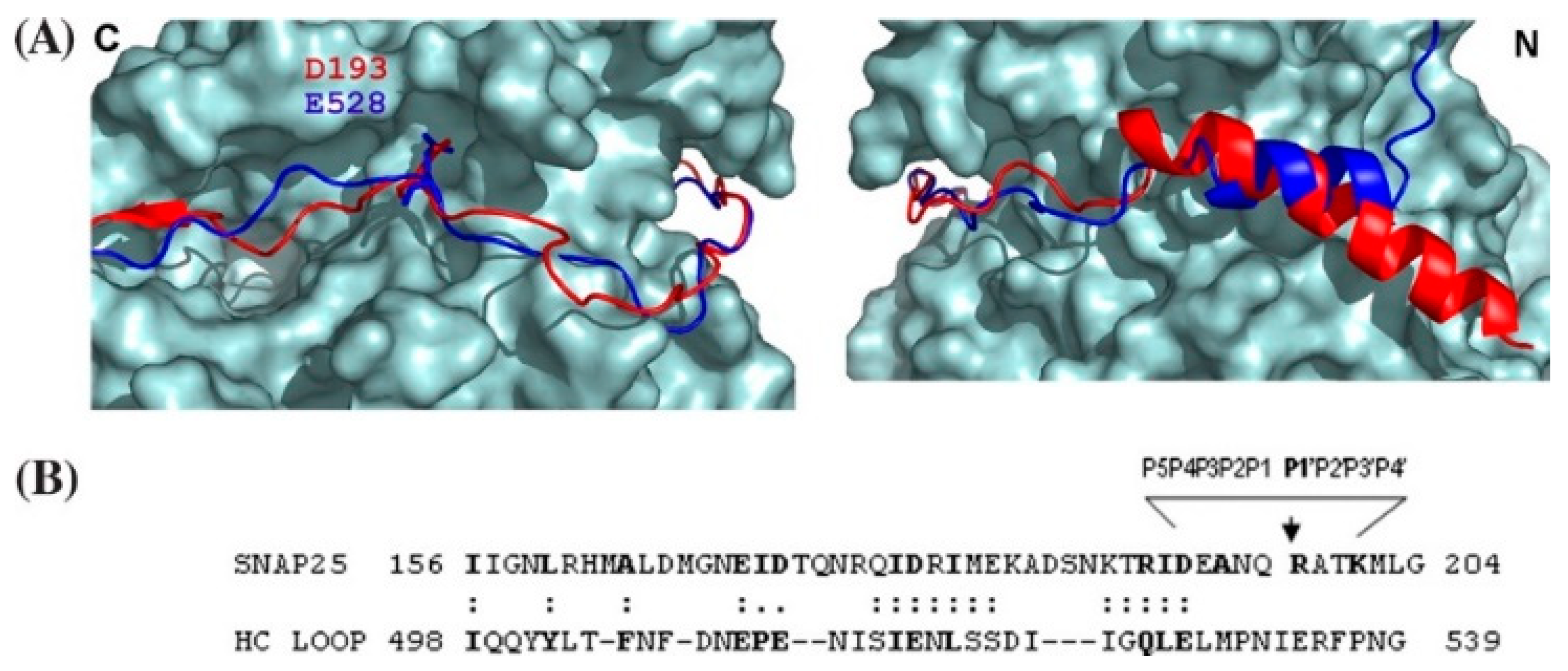
4. Exosite-Pocket Model for LC-Mediated SNARE Cleavage
5. BoNT Duration and Potency
6. Evading Host Degradation
7. BoNT Engineering
8. Conclusions
Conflicts of Interest
References
- Schiavo, G.; Matteoli, M.; Montecucco, C. Neurotoxins affecting neuroexocytosis. Physiol. Rev. 2000, 80, 717–766. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.A.; Bradshaw, M. Clostridium botulinum and its neurotoxins: A metabolic and cellular perspective. Toxicon 2001, 39, 1703–1722. [Google Scholar] [CrossRef]
- Scott, A.B. Botulinum toxin injection of eye muscles to correct strabismus. Trans. Am. Ophthalmol. Soc. 1981, 79, 734–770. [Google Scholar] [PubMed]
- Niemann, H.; Blasi, J.; Jahn, R. Clostridial neurotoxins: New tools for dissecting exocytosis. Trends Cell Biol. 1994, 4, 179–185. [Google Scholar] [CrossRef]
- Zhang, S.; Lebreton, F.; Mansfield, M.J.; Miyashita, S.I.; Zhang, J.; Schwartzman, J.A.; Tao, L.; Masuyer, G.; Martinez-Carranza, M.; Stenmark, P.; et al. Identification of a botulinum neurotoxin-like toxin in a commensal strain of Enterococcus faecium. Cell Host Microbe 2018, 23, 169.e6–176.e6. [Google Scholar] [CrossRef] [PubMed]
- Brunt, J.; Carter, A.T.; Stringer, S.C.; Peck, M.W. Identification of a novel botulinum neurotoxin gene cluster in Enterococcus. FEBS Lett. 2018, 592, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Zornetta, I.; Azarnia Tehran, D.; Arrigoni, G.; Anniballi, F.; Bano, L.; Leka, O.; Zanotti, G.; Binz, T.; Montecucco, C. The first non clostridial botulinum-like toxin cleaves VAMP within the juxtamembrane domain. Sci. Rep. 2016, 6, 30257. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Masuyer, G.; Zhang, J.; Shen, Y.; Lundin, D.; Henriksson, L.; Miyashita, S.I.; Martinez-Carranza, M.; Dong, M.; Stenmark, P. Identification and characterization of a novel botulinum neurotoxin. Nat. Commun. 2017, 8, 14130. [Google Scholar] [CrossRef] [PubMed]
- Montecucco, C.; Berica Rasotto, M.B. On Botulinum Neurotoxin Variability. mBio 2015, 6, e02131-14. [Google Scholar] [CrossRef] [PubMed]
- Mansfield, M.J.; Doxey, A.C. Genomic insights into the evolution and ecology of botulinum neurotoxins. Pathog. Dis. 2018, 76, fty040. [Google Scholar] [CrossRef] [PubMed]
- Division of Select Agents and Toxins: What Is a Select Agent? Available online: https://www.cdc.gov/phpr/dsat/what-is-select-agents.htm (accessed on 10 April 2018).
- Bioterrorism Agents/Diseases. Available online: https://emergency.cdc.gov/agent/agentlist-category.asp (accessed on 10 April 2018).
- Potential Bioterrorism Agents. Available online: https://www.bcm.edu/departments/molecular-virology-and-microbiology/emerging-infections-and-biodefense/potential-bioterrorism-agents (accessed on 10 April 2018).
- Select Agents and Toxins List. Available online: https://www.selectagents.gov/selectagentsandtoxinslist.html (accessed on 10 April 2018).
- Hambrosky, J.; Kroeger, A.; Wolf, S. Epidemiology and prevention of vaccine-preventable diseases. In Centers for Disease Control and Prevention; Public Health Foundation: Washington, DC, USA, 2015; Volume 13. [Google Scholar]
- Aoki, K.R.; Guyer, B. Botulinum toxin type A and other botulinum toxin serotypes: A comparative review of biochemical and pharmacological actions. Eur. J. Neurol. 2001, 8 (Suppl. 5), 21–29. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.R. Scientific and Therapeutic Aspects of Botulinum Toxin; Brin, M.F., Jankovic, J., Hallet, M., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2001; pp. 75–88. [Google Scholar]
- Singh, B.R.; DasGupta, B.R. Conformational changes associated with the nicking and activation of botulinum neurotoxin type E. Biophys. Chem. 1990, 38, 123–130. [Google Scholar] [CrossRef]
- Oguma, K.; Fujinaga, Y.; Inoue, K. Structure and function of Clostridium botulinum toxins. Microbiol. Immunol. 1995, 39, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Binz, T.; Niemann, H.; Singh, B.R. Probing the mechanistic role of glutamate residue in the zinc-binding motif of type a botulinum neurotoxin light chain. Biochemistry 2000, 39, 2399–2405. [Google Scholar] [CrossRef] [PubMed]
- Sathyamoorthy, V.; DasGupta, B.R. Separation, purification, partial characterization and comparison of the heavy and light chains of botulinum neurotoxin types A, B, and E. J. Biol. Chem. 1985, 260, 10461–10466. [Google Scholar] [PubMed]
- Binz, T.; Rummel, A. Cell entry strategy of clostridial neurotoxins. J. Neurochem. 2009, 109, 1584–1595. [Google Scholar] [CrossRef] [PubMed]
- Rummel, A. Double receptor anchorage of botulinum neurotoxins accounts for their exquisite neurospecificity. Curr. Top. Microbiol. Immunol. 2013, 364, 61–90. [Google Scholar] [PubMed]
- Hoch, D.H.; Romero-Mira, M.; Ehrlich, B.E.; Finkelstein, A.; DasGupta, B.R.; Simpson, L.L. Channels formed by botulinum, tetanus, and diphtheria toxins in planar lipid bilayers: Relevance to translocation of proteins across membranes. Proc. Natl. Acad. Sci. USA 1985, 82, 1692–1696. [Google Scholar] [CrossRef] [PubMed]
- Smart, O.S.; Breed, J.; Smith, G.R.; Sansom, M.S. A novel method for structure-based prediction of ion channel conductance properties. Biophys. J. 1997, 72, 1109–1126. [Google Scholar] [CrossRef]
- Falnes, P.O.; Olsnes, S. Cell-mediated reduction and incomplete membrane translocation of diphtheria toxin mutants with internal disulfides in the A fragment. J. Biol. Chem. 1995, 270, 20787–20793. [Google Scholar] [CrossRef] [PubMed]
- Koriazova, L.K.; Montal, M. Translocation of botulinum neurotoxin light chain protease through the heavy chain channel. Nat. Struct. Biol. 2003, 10, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Schiavo, G.; Rossetto, O.; Santucci, A.; DasGupta, B.R.; Montecucco, C. Botulinum neurotoxins are zinc proteins. J. Biol. Chem. 1992, 267, 23479–23483. [Google Scholar] [PubMed]
- Rawlings, N.; Barret, A. Introduction: Metallopeptidases and their clans. In Handbook of Proteolytic Enzyme; Barret, A., Rawlings, N., Woessner, J., Eds.; Academic Press: San Diego, CA, USA, 2004; Volume 2, pp. 231–268. [Google Scholar]
- Segelke, B.; Knapp, M.; Kadkhodayan, S.; Balhorn, R.; Rupp, B. Crystal structure of Clostridium botulinum neurotoxin protease in a product-bound state: Evidence for noncanonical zinc protease activity. Proc. Natl. Acad. Sci. USA 2004, 101, 6888–6893. [Google Scholar] [CrossRef] [PubMed]
- Sudhof, T.C.; Rothman, J.E. Membrane fusion: Grappling with SNARE and SM proteins. Science 2009, 323, 474–477. [Google Scholar] [CrossRef] [PubMed]
- Schiavo, G.; Rossetto, O.; Catsicas, S.; Polverino de Laureto, P.; DasGupta, B.R.; Benfenati, F.; Montecucco, C. Identification of the nerve terminal targets of botulinum neurotoxin serotypes A, D, and E. J. Biol. Chem. 1993, 268, 23784–23787. [Google Scholar] [PubMed]
- Schiavo, G.; Santucci, A.; Dasgupta, B.R.; Mehta, P.P.; Jontes, J.; Benfenati, F.; Wilson, M.C.; Montecucco, C. Botulinum neurotoxins serotypes A and E cleave SNAP-25 at distinct COOH-terminal peptide bonds. FEBS Lett. 1993, 335, 99–103. [Google Scholar] [CrossRef]
- Blasi, J.; Chapman, E.R.; Yamasaki, S.; Binz, T.; Niemann, H.; Jahn, R. Botulinum neurotoxin C1 blocks neurotransmitter release by means of cleaving HPC-1/syntaxin. EMBO J 1993, 12, 4821–4828. [Google Scholar] [PubMed]
- Schiavo, G.; Benfenati, F.; Poulain, B.; Rossetto, O.; Polverino de Laureto, P.; DasGupta, B.R.; Montecucco, C. Tetanus and botulinum-B neurotoxins block neurotransmitter release by proteolytic cleavage of synaptobrevin. Nature 1992, 359, 832–835. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, S.; Hu, Y.; Binz, T.; Kalkuhl, A.; Kurazono, H.; Tamura, T.; Jahn, R.; Kandel, E.; Niemann, H. Synaptobrevin/vesicle-associated membrane protein (VAMP) of Aplysia californica: Structure and proteolysis by tetanus toxin and botulinal neurotoxins type D and F. Proc. Natl. Acad. Sci. USA 1994, 91, 4688–4692. [Google Scholar] [CrossRef] [PubMed]
- Schiavo, G.; Shone, C.C.; Rossetto, O.; Alexander, F.C.; Montecucco, C. Botulinum neurotoxin serotype F is a zinc endopeptidase specific for VAMP/synaptobrevin. J. Biol. Chem. 1993, 268, 11516–11519. [Google Scholar] [PubMed]
- Schiavo, G.; Malizio, C.; Trimble, W.S.; Polverino de Laureto, P.; Milan, G.; Sugiyama, H.; Johnson, E.A.; Montecucco, C. Botulinum G neurotoxin cleaves VAMP/synaptobrevin at a single Ala-Ala peptide bond. J. Biol. Chem. 1994, 269, 20213–20216. [Google Scholar] [PubMed]
- Hackett, G.; Moore, K.; Burgin, D.; Hornby, F.; Gray, B.; Elliott, M.; Mir, I.; Beard, M. Purification and characterization of recombinant botulinum neurotoxin serotype FA, also known as serotype H. Toxins 2018, 10, 195. [Google Scholar] [CrossRef] [PubMed]
- Dover, N.; Barash, J.R.; Hill, K.K.; Xie, G.; Arnon, S.S. Molecular characterization of a novel botulinum neurotoxin type H gene. J. Infect. Dis. 2014, 209, 192–202. [Google Scholar] [CrossRef] [PubMed]
- Masuyer, G.; Zhang, S.; Barkho, S.; Shen, Y.; Henriksson, L.; Kosenina, S.; Dong, M.; Stenmark, P. Structural characterisation of the catalytic domain of botulinum neurotoxin X—High activity and unique substrate specificity. Sci. Rep. 2018, 8, 4518. [Google Scholar] [CrossRef] [PubMed]
- Kiessling, V.; Ahmed, S.; Domanska, M.K.; Holt, M.G.; Jahn, R.; Tamm, L.K. Rapid fusion of synaptic vesicles with reconstituted target SNARE membranes. Biophys. J. 2013, 104, 1950–1958. [Google Scholar] [CrossRef] [PubMed]
- Mayorov, A.V.; Willis, B.; Di Mola, A.; Adler, D.; Borgia, J.; Jackson, O.; Wang, J.; Luo, Y.; Tang, L.; Knapp, R.J.; et al. Symptomatic relief of botulinum neurotoxin/a intoxication with aminopyridines: A new twist on an old molecule. ACS Chem. Biol. 2010, 5, 1183–1191. [Google Scholar] [CrossRef] [PubMed]
- Binz, T.; Blasi, J.; Yamasaki, S.; Baumeister, A.; Link, E.; Sudhof, T.C.; Jahn, R.; Niemann, H. Proteolysis of SNAP-25 by types E and A botulinal neurotoxins. J. Biol. Chem. 1994, 269, 1617–1620. [Google Scholar] [PubMed]
- Whitemarsh, R.C.; Tepp, W.H.; Johnson, E.A.; Pellett, S. Persistence of botulinum neurotoxin a subtypes 1–5 in primary rat spinal cord cells. PLoS ONE 2014, 9, e90252. [Google Scholar] [CrossRef] [PubMed]
- Schiavo, G.; Shone, C.C.; Bennett, M.K.; Scheller, R.H.; Montecucco, C. Botulinum neurotoxin type C cleaves a single Lys-Ala bond within the carboxyl-terminal region of syntaxins. J. Biol. Chem. 1995, 270, 10566–10570. [Google Scholar] [CrossRef] [PubMed]
- Foran, P.; Lawrence, G.W.; Shone, C.C.; Foster, K.A.; Dolly, J.O. Botulinum neurotoxin C1 cleaves both syntaxin and SNAP-25 in intact and permeabilized chromaffin cells: Correlation with its blockade of catecholamine release. Biochemistry 1996, 35, 2630–2636. [Google Scholar] [CrossRef] [PubMed]
- Kalb, S.R.; Baudys, J.; Egan, C.; Smith, T.J.; Smith, L.A.; Pirkle, J.L.; Barr, J.R. Different substrate recognition requirements for cleavage of synaptobrevin-2 by Clostridium baratii and Clostridium botulinum type F neurotoxins. Appl. Environ. Microbiol. 2011, 77, 1301–1308. [Google Scholar] [CrossRef] [PubMed]
- Kalb, S.R.; Baudys, J.; Raphael, B.H.; Dykes, J.K.; Luquez, C.; Maslanka, S.E.; Barr, J.R. Functional characterization of botulinum neurotoxin serotype H as a hybrid of known serotypes F and A (BoNT F/A). Anal. Chem. 2015, 87, 3911–3917. [Google Scholar] [CrossRef] [PubMed]
- Kalb, S.R.; Baudys, J.; Webb, R.P.; Wright, P.; Smith, T.J.; Smith, L.A.; Fernandez, R.; Raphael, B.H.; Maslanka, S.E.; Pirkle, J.L.; et al. Discovery of a novel enzymatic cleavage site for botulinum neurotoxin F5. FEBS Lett. 2012, 586, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Pellett, S.; Tepp, W.H.; Lin, G.; Johnson, E.A. Substrate cleavage and duration of action of botulinum neurotoxin type FA (“H, HA”). Toxicon 2018, 147, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Lacy, D.B.; Tepp, W.; Cohen, A.C.; DasGupta, B.R.; Stevens, R.C. Crystal structure of botulinum neurotoxin type A and implications for toxicity. Nat. Struct. Biol. 1998, 5, 898–902. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Kim, J.J.; Barbieri, J.T. Mechanism of substrate recognition by botulinum neurotoxin serotype A. J. Biol. Chem. 2007, 282, 9621–9627. [Google Scholar] [CrossRef] [PubMed]
- Masuyer, G.; Davies, J.R.; Moore, K.; Chaddock, J.A.; Ravi Acharya, K. Structural analysis of Clostridium botulinum neurotoxin type D as a platform for the development of targeted secretion inhibitors. Sci. Rep. 2015, 5, 13397. [Google Scholar] [CrossRef] [PubMed]
- Brunger, A.T.; Rummel, A. Receptor and substrate interactions of clostridial neurotoxins. Toxicon 2009, 54, 550–560. [Google Scholar] [CrossRef] [PubMed]
- Cooper, G. A Molecular Approach, 2nd ed.; ASM Press: Washington, DC, USA; Sinauer Associates: Sunderland, MA, USA; Boston University: Boston, MA, USA, 2000. [Google Scholar]
- Kipreos, E.T. Ubiquitin-mediated pathways in c. elegans. WormBook 2005, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Cohen, G.M. Caspases: The executioners of apoptosis. Biochem. J. 1997, 326 Pt 1, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Belle, A.; Tanay, A.; Bitincka, L.; Shamir, R.; O’Shea, E.K. Quantification of protein half-lives in the budding yeast proteome. Proc. Natl. Acad. Sci. USA 2006, 103, 13004–13009. [Google Scholar] [CrossRef] [PubMed]
- Cambridge, S.B.; Gnad, F.; Nguyen, C.; Bermejo, J.L.; Kruger, M.; Mann, M. Systems-wide proteomic analysis in mammalian cells reveals conserved, functional protein turnover. J. Proteome Res. 2011, 10, 5275–5284. [Google Scholar] [CrossRef] [PubMed]
- Price, J.C.; Guan, S.; Burlingame, A.; Prusiner, S.B.; Ghaemmaghami, S. Analysis of proteome dynamics in the mouse brain. Proc. Natl. Acad. Sci. USA 2010, 107, 14508–14513. [Google Scholar] [CrossRef] [PubMed]
- Toyama, B.H.; Savas, J.N.; Park, S.K.; Harris, M.S.; Ingolia, N.T.; Yates, J.R., 3rd; Hetzer, M.W. Identification of long-lived proteins reveals exceptional stability of essential cellular structures. Cell 2013, 154, 971–982. [Google Scholar] [CrossRef] [PubMed]
- Heo, S.; Diering, G.H.; Na, C.H.; Nirujogi, R.S.; Bachman, J.L.; Pandey, A.; Huganir, R.L. Identification of long-lived synaptic proteins by proteomic analysis of synaptosome protein turnover. Proc. Natl. Acad. Sci. USA 2018, 115, E3827–E3836. [Google Scholar] [CrossRef] [PubMed]
- Pellett, S.; Bradshaw, M.; Tepp, W.H.; Pier, C.L.; Whitemarsh, R.C.M.; Chen, C.; Barbieri, J.T.; Johnson, E.A. The light chain defines the duration of action of botulinum toxin serotype A subtypes. MBio 2018, 9, e00089-18. [Google Scholar] [CrossRef] [PubMed]
- Pellett, S.; Yaksh, T.L.; Ramachandran, R. Current status and future directions of botulinum neurotoxins for targeting pain processing. Toxins 2015, 7, 4519–4563. [Google Scholar] [CrossRef] [PubMed]
- Foran, P.G.; Mohammed, N.; Lisk, G.O.; Nagwaney, S.; Lawrence, G.W.; Johnson, E.; Smith, L.; Aoki, K.R.; Dolly, J.O. Evaluation of the therapeutic usefulness of botulinum neurotoxin B, C1, E, and F compared with the long lasting type A. Basis for distinct durations of inhibition of exocytosis in central neurons. J. Biol. Chem. 2003, 278, 1363–1371. [Google Scholar] [CrossRef] [PubMed]
- Kull, S.; Schulz, K.M.; Weisemann, J.; Kirchner, S.; Schreiber, T.; Bollenbach, A.; Dabrowski, P.W.; Nitsche, A.; Kalb, S.R.; Dorner, M.B.; et al. Isolation and functional characterization of the novel Clostridium botulinum neurotoxin A8 subtype. PLoS ONE 2015, 10, e0116381. [Google Scholar] [CrossRef] [PubMed]
- Pellett, S.; Tepp, W.H.; Whitemarsh, R.C.; Bradshaw, M.; Johnson, E.A. In vivo onset and duration of action varies for botulinum neurotoxin A subtypes 1–5. Toxicon 2015, 107, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Whitemarsh, R.C.; Tepp, W.H.; Bradshaw, M.; Lin, G.; Pier, C.L.; Scherf, J.M.; Johnson, E.A.; Pellett, S. Characterization of botulinum neurotoxin a subtypes 1 through 5 by investigation of activities in mice, in neuronal cell cultures, and in vitro. Infect. Immun. 2013, 81, 3894–3902. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Salas, E.; Steward, L.E.; Ho, H.; Garay, P.E.; Sun, S.W.; Gilmore, M.A.; Ordas, J.V.; Wang, J.; Francis, J.; Aoki, K.R. Plasma membrane localization signals in the light chain of botulinum neurotoxin. Proc. Natl. Acad. Sci. USA 2004, 101, 3208–3213. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Barbieri, J.T. Association of botulinum neurotoxin serotype A light chain with plasma membrane-bound SNAP-25. J. Biol. Chem. 2011, 286, 15067–15072. [Google Scholar] [CrossRef] [PubMed]
- Kalb, S.R.; Baudys, J.; Rees, J.C.; Smith, T.J.; Smith, L.A.; Helma, C.H.; Hill, K.; Kull, S.; Kirchner, S.; Dorner, M.B.; et al. De novo subtype and strain identification of botulinum neurotoxin type B through toxin proteomics. Anal. Bioanal. Chem. 2012, 403, 215–226. [Google Scholar] [CrossRef] [PubMed]
- Eleopra, R.; Tugnoli, V.; Rossetto, O.; Montecucco, C.; De Grandis, D. Botulinum neurotoxin serotype C: A novel effective botulinum toxin therapy in human. Neurosci. Lett. 1997, 224, 91–94. [Google Scholar] [CrossRef]
- Sloop, R.R.; Cole, B.A.; Escutin, R.O. Human response to botulinum toxin injection: Type B compared with type A. Neurology 1997, 49, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Eleopra, R.; Tugnoli, V.; Rossetto, O.; De Grandis, D.; Montecucco, C. Different time courses of recovery after poisoning with botulinum neurotoxin serotypes A and E in humans. Neurosci. Lett. 1998, 256, 135–138. [Google Scholar] [CrossRef]
- Brin, M.F.; Lew, M.F.; Adler, C.H.; Comella, C.L.; Factor, S.A.; Jankovic, J.; O’Brien, C.; Murray, J.J.; Wallace, J.D.; Willmer-Hulme, A.; et al. Safety and efficacy of neurobloc (botulinum toxin type B) in type A-resistant cervical dystonia. Neurology 1999, 53, 1431–1438. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, K.; Kohda, T.; Shibata, Y.; Tsukamoto, K.; Arimitsu, H.; Hayashi, M.; Mukamoto, M.; Sasakawa, N.; Kozaki, S. Unique biological activity of botulinum D/C mosaic neurotoxin in murine species. Infect. Immun. 2012, 80, 2886–2893. [Google Scholar] [CrossRef] [PubMed]
- Pellett, S.; Tepp, W.H.; Scherf, J.M.; Pier, C.L.; Johnson, E.A. Activity of botulinum neurotoxin type D (strain 1873) in human neurons. Toxicon 2015, 101, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Peck, M.W.; Smith, T.J.; Anniballi, F.; Austin, J.W.; Bano, L.; Bradshaw, M.; Cuervo, P.; Cheng, L.W.; Derman, Y.; Dorner, B.G.; et al. Historical perspectives and guidelines for botulinum neurotoxin subtype nomenclature. Toxins 2017, 9, 38. [Google Scholar] [CrossRef] [PubMed]
- Kauffman, J.A.; Way, J.F., Jr.; Siegel, L.S.; Sellin, L.C. Comparison of the action of types a and f botulinum toxin at the rat neuromuscular junction. Toxicol. Appl. Pharmacol. 1985, 79, 211–217. [Google Scholar] [CrossRef]
- Rossetto, O.; Pirazzini, M.; Montecucco, C. Botulinum neurotoxins: Genetic, structural and mechanistic insights. Nat. Rev. Microbiol. 2014, 12, 535–549. [Google Scholar] [CrossRef] [PubMed]
- Lamanna, C.; Mc, E.O.; Eklund, H.W. The purification and crystallization of Clostridium botulinum type A toxin. Science 1946, 103, 613–614. [Google Scholar] [CrossRef] [PubMed]
- Duff, J.T.; Klerer, J.; Bibler, R.H.; Moore, D.E.; Gottfried, C.; Wright, G.G. Studies on immunity to toxins of Clostridium botulinum. II. Production and purification of type B toxin for toxoid. J. Bacteriol. 1957, 73, 597–601. [Google Scholar] [PubMed]
- Ohishi, I.; Iwasaki, M.; Sakaguchi, G. Purification and characterization of two components of botulinum C2 toxin. Infect. Immun. 1980, 30, 668–673. [Google Scholar] [PubMed]
- Cardella, M.A.; Duff, J.T.; Wingfield, B.H.; Gottfried, C. Studies on immunity to toxins of Clostridium botulinum. VI. Purification and detoxification of type D toxin and the immunological response to toxoid. J. Bacteriol. 1960, 79, 372–378. [Google Scholar] [PubMed]
- Gerwing, J.; Dolman, C.E.; Ko, A. Mechanism of tryptic activation of clostridium botulinum type E toxin. J. Bacteriol. 1965, 89, 1176–1179. [Google Scholar] [PubMed]
- Oishi, I.; Sakaguchi, G. Purification of Clostridium botuliunum type F progenitor toxin. Appl. Microbiol. 1974, 28, 923–928. [Google Scholar] [PubMed]
- Tepp, W.H.; Lin, G.; Johnson, E.A. Purification and characterization of a novel subtype A3 botulinum neurotoxin. Appl. Environ. Microbiol. 2012, 78, 3108–3113. [Google Scholar] [CrossRef] [PubMed]
- Syuto, B.; Kubo, S. Isolation and molecular size of clostridium botulinum type C toxin. Appl. Environ. Microbiol. 1977, 33, 400–405. [Google Scholar] [PubMed]
- Ciccarelli, A.S.; Whaley, D.N.; McCroskey, L.M.; Gimenez, D.F.; Dowell, V.R., Jr.; Hatheway, C.L. Cultural and physiological characteristics of Clostridium botulinum type G and the susceptibility of certain animals to its toxin. Appl. Environ. Microbiol. 1977, 34, 843–848. [Google Scholar] [PubMed]
- Tsai, Y.C.; Kotiya, A.; Kiris, E.; Yang, M.; Bavari, S.; Tessarollo, L.; Oyler, G.A.; Weissman, A.M. Deubiquitinating enzyme VCIP135 dictates the duration of botulinum neurotoxin type A intoxication. Proc. Natl. Acad. Sci. USA 2017, 114, E5158–E5166. [Google Scholar] [CrossRef] [PubMed]
- Tsai, Y.C.; Maditz, R.; Kuo, C.L.; Fishman, P.S.; Shoemaker, C.B.; Oyler, G.A.; Weissman, A.M. Targeting botulinum neurotoxin persistence by the ubiquitin-proteasome system. Proc. Natl. Acad. Sci. USA 2010, 107, 16554–16559. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Curran, J.E.; Liao, Z.; Gordon, R.K. The biological activity of ubiquitinated BoNT/B light chain in vitro and in human SHSY-5Y neuronal cells. J. Cell. Biochem. 2009, 108, 660–667. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, Z.; Liu, D.X.; Pagano, M.; Ravid, K. Ubiquitin-dependent degradation of cyclin B is accelerated in polyploid megakaryocytes. J. Biol. Chem. 1998, 273, 1387–1392. [Google Scholar] [CrossRef] [PubMed]
- Sadoul, K.; Berger, A.; Niemann, H.; Weller, U.; Roche, P.A.; Klip, A.; Trimble, W.S.; Regazzi, R.; Catsicas, S.; Halban, P.A. SNAP-23 is not cleaved by botulinum neurotoxin E and can replace SNAP-25 in the process of insulin secretion. J. Biol. Chem. 1997, 272, 33023–33027. [Google Scholar] [CrossRef] [PubMed]
- Vaidyanathan, V.V.; Yoshino, K.; Jahnz, M.; Dorries, C.; Bade, S.; Nauenburg, S.; Niemann, H.; Binz, T. Proteolysis of SNAP-25 isoforms by botulinum neurotoxin types A, C, and E: Domains and amino acid residues controlling the formation of enzyme-substrate complexes and cleavage. J. Neurochem. 1999, 72, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Jahn, R.; Scheller, R.H. SNAREs—Engines for membrane fusion. Nat. Rev. Mol. Cell Biol. 2006, 7, 631–643. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Barbieri, J.T. Engineering botulinum neurotoxin to extend therapeutic intervention. Proc. Natl. Acad. Sci. USA 2009, 106, 9180–9184. [Google Scholar] [CrossRef] [PubMed]
- Sikorra, S.; Litschko, C.; Muller, C.; Thiel, N.; Galli, T.; Eichner, T.; Binz, T. Identification and characterization of botulinum neurotoxin a substrate binding pockets and their re-engineering for human SNAP-23. J. Mol. Biol. 2016, 428, 372–384. [Google Scholar] [CrossRef] [PubMed]
- Nugent, M.; Wang, J.; Lawrence, G.; Zurawski, T.; Geoghegan, J.A.; Dolly, J.O. Conjugate of an igg binding domain with botulinum neurotoxin a lacking the acceptor moiety targets its snare protease into TrkA-expressing cells when coupled to anti-TrkA IgG or FC-betaNGF. Bioconjug. Chem. 2017, 28, 1684–1692. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Meng, J.; Wang, J.; Hearty, S.; Dolly, J.O.; O’Kennedy, R. Targeted delivery of a snare protease to sensory neurons using a single chain antibody (SCFV) against the extracellular domain of P2X(3) inhibits the release of a pain mediator. Biochem. J. 2014, 462, 247–256. [Google Scholar] [CrossRef] [PubMed]
- Vazquez-Cintron, E.J.; Beske, P.H.; Tenezaca, L.; Tran, B.Q.; Oyler, J.M.; Glotfelty, E.J.; Angeles, C.A.; Syngkon, A.; Mukherjee, J.; Kalb, S.R.; et al. Engineering botulinum neurotoxin C1 as a molecular vehicle for intra-neuronal drug delivery. Sci. Rep. 2017, 7, 42923. [Google Scholar] [CrossRef] [PubMed]
- Collins, M.D.; East, A.K. Phylogeny and taxonomy of the food-borne pathogen Clostridium botulinum and its neurotoxins. J. Appl. Microbiol. 1998, 84, 5–17. [Google Scholar] [CrossRef] [PubMed]
- Montecucco, C.; Schiavo, G. Structure and function of tetanus and botulinum neurotoxins. Q. Rev. Biophys. 1995, 28, 423–472. [Google Scholar] [CrossRef] [PubMed]
- Ito, H.; Sagane, Y.; Miyata, K.; Inui, K.; Matsuo, T.; Horiuchi, R.; Ikeda, T.; Suzuki, T.; Hasegawa, K.; Kouguchi, H.; et al. HA-33 facilitates transport of the serotype D botulinum toxin across a rat intestinal epithelial cell monolayer. FEMS Immunol. Med. Microbiol. 2011, 61, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Gu, S.; Rumpel, S.; Zhou, J.; Strotmeier, J.; Bigalke, H.; Perry, K.; Shoemaker, C.B.; Rummel, A.; Jin, R. Botulinum neurotoxin is shielded by NTNHA in an interlocked complex. Science 2012, 335, 977–981. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). Notice of CDC’s discontinuation of investigational pentavalent (ABCDE) botulinum toxoid vaccine for workers at risk for occupational exposure to botulinum toxins. MMWR Morb. Mortal. Wkly. Rep. 2011, 60, 1454–1455. [Google Scholar]
- Webb, R.P.; Smith, T.J.; Smith, L.A.; Wright, P.M.; Guernieri, R.L.; Brown, J.L.; Skerry, J.C. Recombinant botulinum neurotoxin hc subunit (BoNT Hc) and catalytically inactive Clostridium botulinum holoproteins (ciBoNT HPs) as vaccine candidates for the prevention of botulism. Toxins 2017, 9, 269. [Google Scholar] [CrossRef] [PubMed]
- Przedpelski, A.; Tepp, W.H.; Zuverink, M.; Johnson, E.A.; Pellet, S.; Barbieri, J.T. Enhancing toxin-based vaccines against botulism. Vaccine 2018, 36, 827–832. [Google Scholar] [CrossRef] [PubMed]
| Serotype 1 | Toxicity | Duration |
|---|---|---|
| /A | 1.2 ng [82] | <9 months [45] |
| /B | 1.2 ng [83] | 2–4 months [73,74,75,76] |
| /C1 | 1.1 ng [89] | 4–6 months [73] |
| /D | 0.4 ng [85] | ~3 weeks [78] |
| /E | 1.1 ng [86] | 2–3 weeks [45,79] |
| /F | 2.5 ng [87] | <A1 [80] |
| /G | 120 LD50/kg 2 [90] | n.d. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gardner, A.P.; Barbieri, J.T. Light Chain Diversity among the Botulinum Neurotoxins. Toxins 2018, 10, 268. https://doi.org/10.3390/toxins10070268
Gardner AP, Barbieri JT. Light Chain Diversity among the Botulinum Neurotoxins. Toxins. 2018; 10(7):268. https://doi.org/10.3390/toxins10070268
Chicago/Turabian StyleGardner, Alexander P., and Joseph T. Barbieri. 2018. "Light Chain Diversity among the Botulinum Neurotoxins" Toxins 10, no. 7: 268. https://doi.org/10.3390/toxins10070268
APA StyleGardner, A. P., & Barbieri, J. T. (2018). Light Chain Diversity among the Botulinum Neurotoxins. Toxins, 10(7), 268. https://doi.org/10.3390/toxins10070268





