Natural Contamination with Mycotoxins Produced by Fusarium graminearum and Fusarium poae in Malting Barley in Argentina
Abstract
:1. Introduction
2. Results
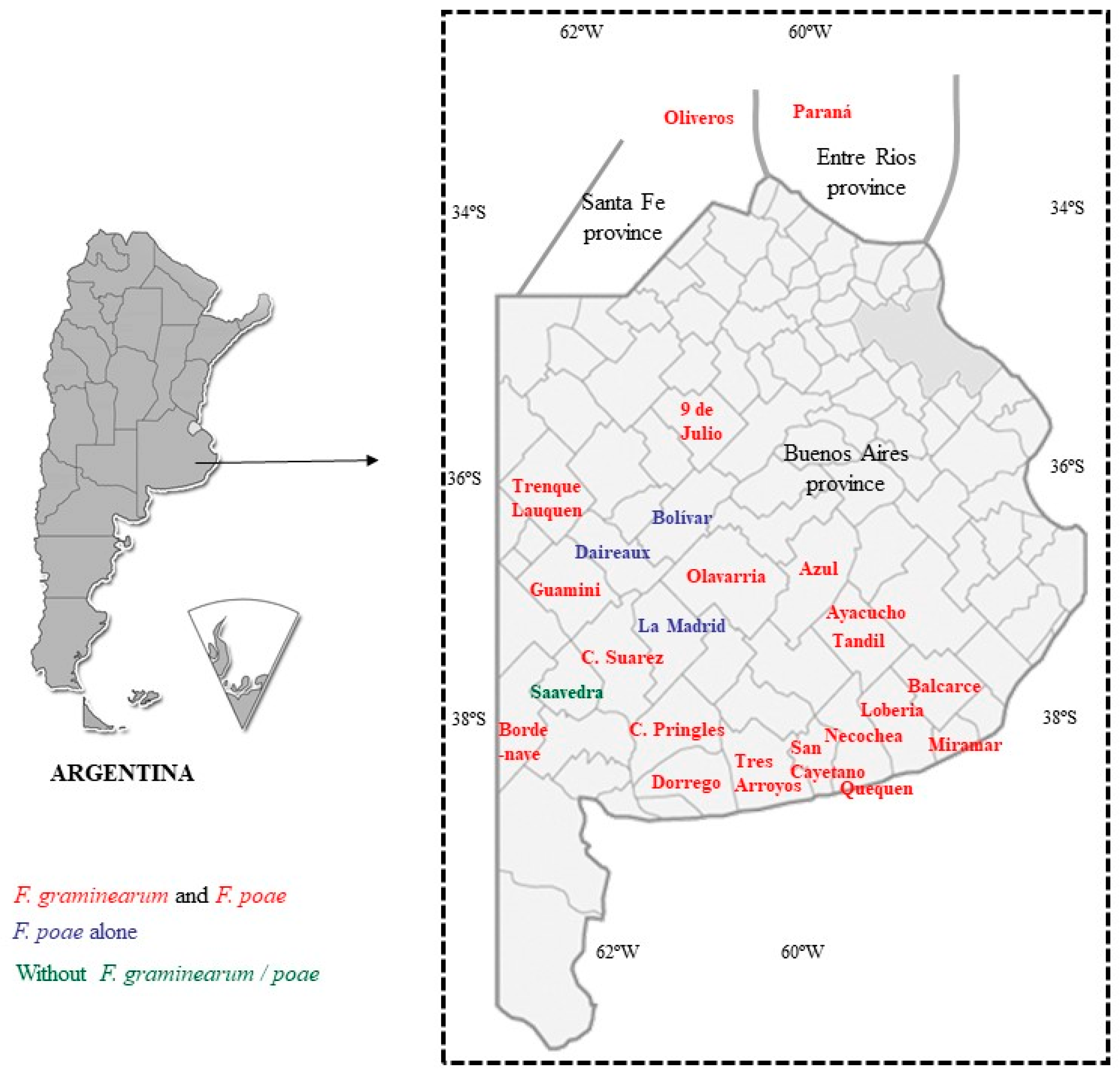
2.1. Fusarium-Species Identification
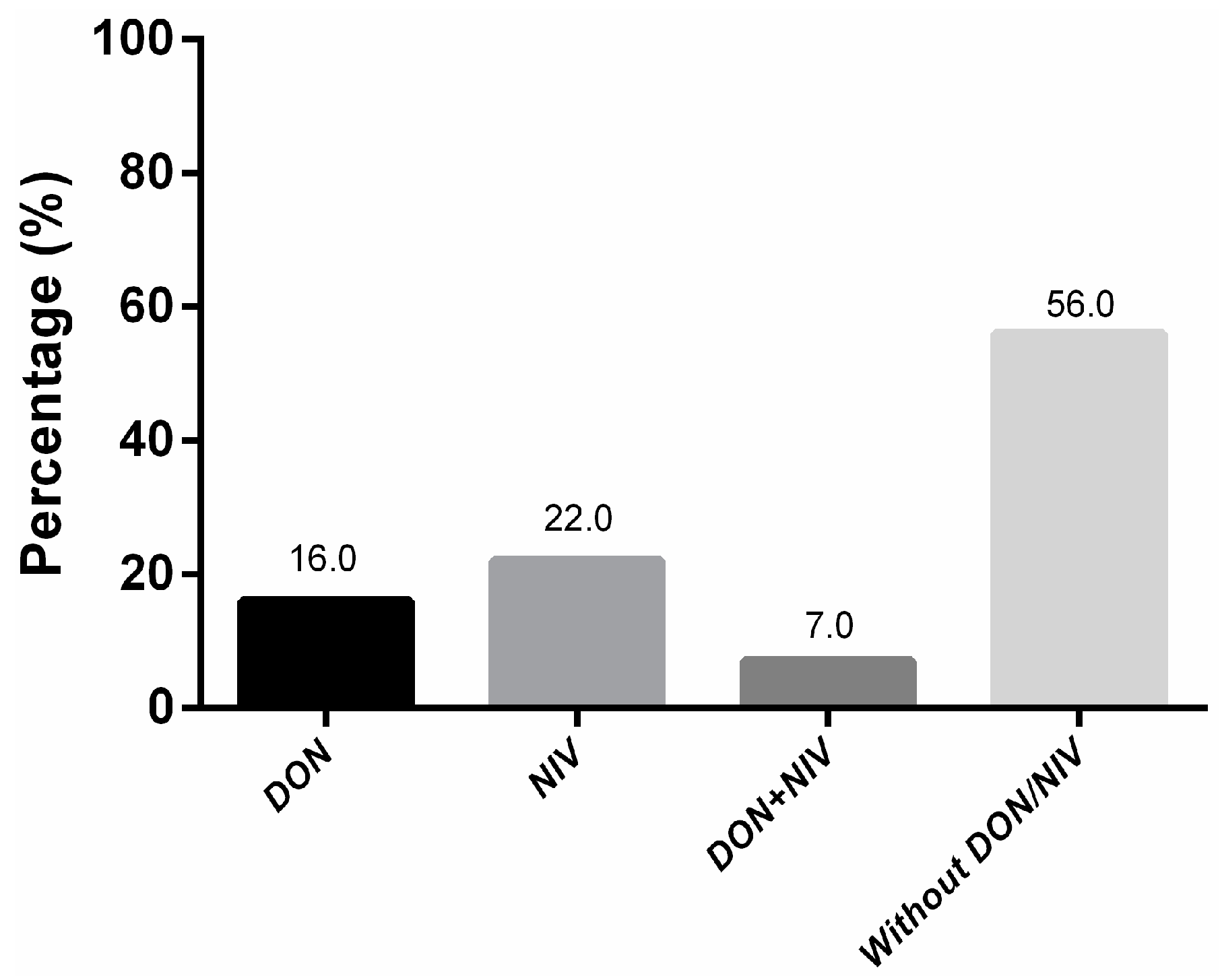
2.2. Trichothecene Screening
2.3. Statistical Analyses
3. Discussion
4. Materials and Methods
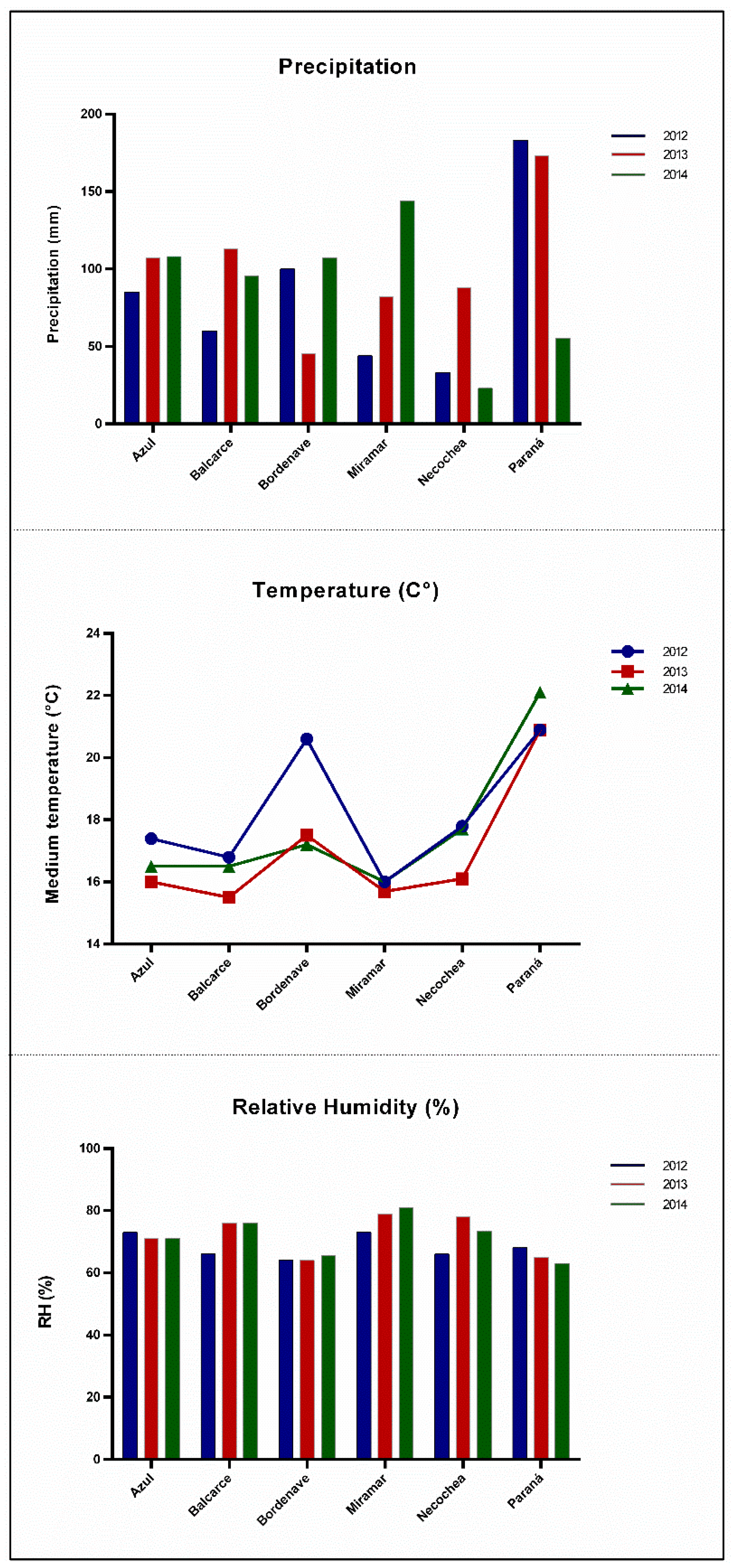
4.1. Barley Samples and Climatic Information
4.2. Fungal Isolation
4.3. Molecular Identification
4.4. Mycotoxin Analysis
4.5. Statistical Analyses
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Food and Agriculture Organization of the United Nations. Agricultura Mundial: Hacia los Años 2015/2030. 2017. Available online: http://www.fao.org/docrep/004/y3557s/y3557s08.htm (accessed on 24 February 2017).
- Ministerio de Agroindustria Argentina. Dirección de Estimaciones Agrícolas y Delegaciones. Estimaciones Agrícolas, Informe Semanal al 30 de Junio de 2016. 2016. Available online: http://www.agroindustria.gob.ar/sitio/areas/estimaciones/_archivos/estimaciones/160000_2016/000600_Junio/160630_Informe%20Semanal%20Estimaciones%20-%20al%2030-Jun-2016.pdf (accessed on 24 February 2017).
- Kaur, M.; Bowman, J.P.; Stewart, D.C.; Evans, D.E. The fungal community structure of barley malts from diverse geographical regions correlates with malt quality parameters. Int. J. Food Microbiol. 2015, 215, 71–78. [Google Scholar] [CrossRef] [PubMed]
- D’Mello, J.P.F.; Placinta, C.M.; Macdonald, A.M.C. Fusarium mycotoxins: A review of global implications for animal health, welfare and productivity. Anim. Feed Sci. Technol. 1999, 80, 183–205. [Google Scholar] [CrossRef]
- Schwarz, P.B. Impact of Fusarium head blight on malting and brewing quality of barley. In Fusarium Head Blight of Wheat and Barley; Leonard, K.J., Bushnell, W.R., Eds.; American Phytopathological Society Press: St. Paul, MN, USA, 2003; pp. 395–419. [Google Scholar]
- Desjardins, A.E. Fusarium Mycotoxins Chemistry, Genetics and Biology; American Phytopathological Society Press: St. Paul, MN, USA, 2006. [Google Scholar]
- Nicholson, P.; Chandler, E.; Draeger, R.C.; Gosman, N.E.; Dimpson, D.R.; Thomsett, M. Molecular tools to study epidemiology and toxicology of Fusarium head blight of cereals. Eur. J. Plant Pathol. 2003, 109, 691–703. [Google Scholar] [CrossRef]
- Beccari, G.; Prodi, A.; Tini, F.; Bonciarelli, U.; Onofri, A.; Oueslati, S.; Limayma, M.; Covarelli, L. Changes in the Fusarium head blight complex of malting barley in a three-year field experiment in Italy. Toxins 2017, 9, 120. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, I.; Friberg, H.; Kolseth, A.-K.; Steinberg, C.; Persson, P. Agricultural factors affecting Fusarium communities in wheat kernels. Int. J. Food Microbiol. 2017, 252, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Schöneberg, T.; Martinb, C.; Wettsteina, F.; Buchelia, T.D.; Mascherb, F.; Bertossab, M.; Musaa, T.; Kellerc, B.; Vogelgsang, S. Fusarium and mycotoxin spectra in Swiss barley are affected by various cropping techniques. Food Addit. Contam. Part A 2016, 10, 1608–1619. [Google Scholar] [CrossRef] [PubMed]
- Beccari, G.; Caproni, L.; Tini, F.; Uhlig, S.; Covarelli, L. Presence of Fusarium species and other toxigenic fungi in malting barley and multi-mycotoxin analysis by liquid chromatography-high-resolution mass spectrometry. J. Agric. Food Chem. 2016, 64, 4390–4399. [Google Scholar] [CrossRef] [PubMed]
- Hooker, D.C.; Schaafsma, A.W.; Tamburic-Ilincic, L. Using weather variables pre- and post-heading to predict deoxynivalenol content in winter wheat. Plant Dis. 2002, 86, 611–619. [Google Scholar] [CrossRef]
- Roer Hjelkrem, A.-G.; Torp, T.; Brodal, G.; Aamot, H.U.; Strand, E.; Nordskog, B.; Dill-Macky, R.; Edwards, S.G.; Hofgaard, I.S. DON content in oat grains in Norway related to weather conditions at different growth stages. Eur. J. Plant Pathol. 2017. [Google Scholar] [CrossRef]
- Li, X.; Michlmayr, H.; Schweiger, W.; Malachova, A.; Shin, S.; Huang, Y.; Dong, Y.; Wiesenberg, G.; McCormick, S.; Lemmens, M.; et al. A barley UDP-glucosyltransferase inactivates nivalenol and provides Fusarium head blight resistance in transgenic wheat. J. Exp. Bot. 2017. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Shin, S.; Heinen, S.; Dill-Macky, R.; Berthiller, F.; Nersesian, N.; Clemente, T.; McCormick, S.; Muehlbauer, G.J. Transgenic wheat expressing a barley UDP-glucosyltransferase detoxifies deoxynivalenol and provides high levels of resistance to Fusarium graminearum. Mol. Plant-Microbe Interact. 2015, 28, 1237–1246. [Google Scholar] [CrossRef] [PubMed]
- Osborne, L.E.; Stein, J.M. Epidemiology of Fusarium head blight on small-grain cereals. Int. J. Food Microbiol. 2007, 119, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Stenglein, S.A.; Dinolfo, M.I.; Barros, G.; Bongiorno, F.; Chulze, S.; Moreno, M.V. Fusarium poae pathogenicity and mycotoxin accumulation on selected wheat and barley genotypes at a single location in Argentina. Plant Dis. 2014, 98, 1733–1738. [Google Scholar] [CrossRef]
- Yli-Mattila, T.; Paavanen-Huhtala, S.; Jestoi, M.; Parikka, P.; Hietaniemi, V.; Gagkaeva, T.; Sarlin, T.; Haikara, A.; Laaksonen, S.; Rizzo, A. Real-time PCR detection and quantification of Fusarium poae, F. graminearum, F. sporotrichioides and F. langsethiae in cereal grains in Finland and Russia. Arch. Phytopathol. Plant Protect. 2008, 41, 243–260. [Google Scholar] [CrossRef]
- Thrane, U.; Adler, A.; Clasen, P.-E.; Galvano, F.; Langseth, W.; Lew, H.; Logrieco, A.; Nielsen, K.F.; Ritieni, A. Diversity in metabolite production by Fusarium langsethiae, Fusarium poae and Fusarium sporotrichioides. Int. J. Food Microbiol. 2004, 95, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, L.K.; Cookb, D.J.; Edward, S.G.; Ray, R.V. The prevalence and impact of Fusarium head blight pathogens and mycotoxins on malting barley quality in UK. Int. J. Food Microbiol. 2014, 179, 38–49. [Google Scholar] [CrossRef] [PubMed]
- Lindblad, M.; Gidlund, A.; Sulyok, M.; Börjesson, T.; Krska, R.; Olsen, M.; Fredlund, E. Deoxynivalenol and other selected Fusarium toxins in Swedish wheat—Occurrence and correlation to specific Fusarium species. Int. J. Food Microbiol. 2013, 167, 284–291. [Google Scholar] [CrossRef] [PubMed]
- Martinez, M.; Castañares, E.; Dinolfo, M.I.; Pacheco, W.G.; Moreno, M.V.; Stenglein, S.A. Presencia de Fusarium graminearum en muestras de trigo destinado al consumo humano. Rev. Argent. Microbiol. 2014, 46, 41–44. [Google Scholar] [CrossRef]
- Fredlund, E.; Gidlund, A.; Sulyok, M.; Börjesson, T.; Krska, R.; Olsen, M.; Lindblad, M. Deoxynivalenol and other selected Fusarium toxins in Swedish oats—Occurrence and correlation to specific Fusarium species. Int. J. Food Microbiol. 2013, 167, 276–283. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Commission Regulation (EC) no. 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuff. Off. J. Eur. Union 2006, L364, 5–24. [Google Scholar]
- Agência Nacional de Vigilância Sanitària. RDC no. 138. Dispõe sobre limites máximos tolerados (LMT) para micotoxinas. Diário Oficial da União, 9 de fevreiro de 2017. [Google Scholar]
- Pan, D.; Bonsignore, F.; Rivas, F.; Perera, G.; Bettucci, L. Deoxynivalenol in barley samples from Uruguay. Int. J. Food Microbiol. 2007, 114, 149–152. [Google Scholar] [CrossRef] [PubMed]
- Piacentini, K.C.; Savi, G.D.; Pereira, M.E.V.; Scussel, V.M. Fungi and the natural occurrence of deoxynivalenol and fumonisins in maltinh barley (Hordeum vulgare L.). Food Chem. 2015, 187, 204–209. [Google Scholar] [CrossRef] [PubMed]
- González, H.H.L.; Moltó, G.A.; Pacin, A.; Resnik, S.L.; Zelaya, M.J.; Masana, M.; Martínez, E.J. Trichothecenes and mycoflora in wheat harvested in nine locations in Buenos Aires province, Argentina. Mycopathologia 2008, 165, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Castañares, E.; Ramirez Albuquerque, D.; Dinolfo, M.I.; Fernandez Pinto, V.; Patriarca, A.; Stenglein, S.A. Trichothecene genotypes and production profiles of Fusarium graminearum isolates obtained from barley cultivated in Argentina. Int. J. Food Microbiol. 2014, 179, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Castañares, E.; Dinolfo, M.I.; Del Ponte, E.M.; Pan, D.; Stenglein, S.A. Species composition and genetic structure of Fusarium graminearum species complex populations affecting the main barley growing regions of South America. Plant Pathol. 2016, 65, 930–939. [Google Scholar] [CrossRef]
- Oliveira, P.M.; Mauch, A.; Jacob, F.; Waters, D.M.; Arendt, E.K. Fundamental study on the influence of Fusarium infection on quality and ultrastructure of barley malt. Int. J. Food Microbiol. 2012, 156, 32–43. [Google Scholar] [CrossRef] [PubMed]
- Sarlin, T.; Laitila, A.; Pekkarinen, A.; Haikara, A. Effects of three Fusarium species on the quality of barley and malt. Am. Soc. Brew. Chem. 2005, 63, 43–49. [Google Scholar]
- Bourdages, J.V.; Marchand, S.; Rioux, S.; Belzile, F.J. Diversity and prevalence of Fusarium species from Quebec barley fields. Can. J. Plant Pathol. 2006, 28, 419–425. [Google Scholar] [CrossRef]
- Dinolfo, M.I.; Stenglein, S.A. Fusarium poae and mycotoxins: potential risk for consumers. Bol. Soc. Argent. Bot. 2014, 49, 5–20. [Google Scholar]
- Xu, M.N.; Nicholson, P.; Thomset, M.A.; Simpson, D.; Cooke, B.M.; Doohan, F.M.; Brennan, J.; Monaghan, S.; Moretti, A.; Mule, G.; et al. Relationship between the fungal complex causing Fusarium head blight of wheat and environmental conditions. Phytopathology 2008, 98, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, L.K.; Jensen, J.D.; Nielsen, G.C.; Jensen, J.E.; Spliid, N.H.; Thomsen, I.K.; Justesen, A.F.; Collinge, D.B.; Jørgensen, L.N. Fusarium head blight of cereals in Denmark: species complex and related mycotoxins. Phytopathology 2011, 101, 960–969. [Google Scholar] [CrossRef] [PubMed]
- Leslie, J.F.; Summerell, B.A. The Fusarium Laboratory Manual; Blackwell Publishing: Ames, IA, USA, 2006. [Google Scholar]
- Stenglein, S.A.; Balatti, P.A. Genetic diversity of Phaeoisariopsis griseola in Argentina as revealed by virulence and molecular markers. Physiol. Mol. Plant Pathol. 2006, 68, 158–167. [Google Scholar] [CrossRef]
- Nicholson, P.; Simpson, D.R.; Weston, G.; Rezanoor, H.N.; Lees, A.K.; Parry, D.W.; Joyce, D. Detection and quantification of Fusarium culmorum and Fusarium graminearum in cereals using PCR assays. Physiol. Mol. Plant Pathol. 1998, 53, 17–37. [Google Scholar] [CrossRef]
- Parry, D.W.; Nicholson, P. Development of a PCR assay to detect Fusarium poae in wheat. Plant Pathol. 1996, 45, 383–391. [Google Scholar] [CrossRef]
- Chandler, E.; Simpson, D.; Thomsett, M.; Nicholson, P. Development of a PCR assays to Tri7 and Tri13 trichothecene biosynthetic genes, and characterisation of chemotypes of Fusarium graminearum, Fusarium culmorum and Fusarium cerealis. Physiol. Mol. Plant Pathol. 2003, 62, 355–367. [Google Scholar] [CrossRef]
- Dinolfo, M.I.; Barros, G.G.; Stenglein, S.A. Development of a PCR assay to detect the potential production of nivalenol in Fusarium poae. FEMS Microbiol. Lett. 2012, 332, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Ward, T.J.; Clear, R.M.; Rooney, A.P.; O’Donnell, K.; Gaba, D.; Patrick, S.; Starkey, D.E.; Gilbert, J.; Geiser, D.M.; Nowicki, T.W. An adaptive evolutionary shift in Fusarium head blight pathogen populations is driving the rapid spread of more toxigenic Fusarium graminearum in North America. Fungal Genet. Biol. 2008, 45, 473–484. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Health and Human Services, FDA, CDER, CVM. Guidance for Industry, Bioanalytical Method Validation; U.S. Food and Drug Administration: Maryland, MD, USA, 2001.
- European Commission Decision. Implementing Council Directive Concerning the performance of analytical methods and the interpretation of the results. Off. J. Eur. Commun. 2012, L221, 23–33. [Google Scholar]
- Di Rienzo, J.A.; Casanoves, F.; Balzarini, M.; Gonzalez, L.; Cuadroda, M.; Robledo, C. InfoStat Versión 2016; Grupo InfoStat, FCA, Universidad Nacional de Córdoba: Córdoba, Argentina, 2008. [Google Scholar]

| Year | 2012 | 2013 | 2014 |
|---|---|---|---|
| % with DON | 9 | 13 | 47 |
| % with NIV | 27 | 18 | 47 |
| % without mycotoxins | 64 | 69 | 6 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nogueira, M.S.; Decundo, J.; Martinez, M.; Dieguez, S.N.; Moreyra, F.; Moreno, M.V.; Stenglein, S.A. Natural Contamination with Mycotoxins Produced by Fusarium graminearum and Fusarium poae in Malting Barley in Argentina. Toxins 2018, 10, 78. https://doi.org/10.3390/toxins10020078
Nogueira MS, Decundo J, Martinez M, Dieguez SN, Moreyra F, Moreno MV, Stenglein SA. Natural Contamination with Mycotoxins Produced by Fusarium graminearum and Fusarium poae in Malting Barley in Argentina. Toxins. 2018; 10(2):78. https://doi.org/10.3390/toxins10020078
Chicago/Turabian StyleNogueira, María Soledad, Julieta Decundo, Mauro Martinez, Susana Nelly Dieguez, Federico Moreyra, Maria Virginia Moreno, and Sebastian Alberto Stenglein. 2018. "Natural Contamination with Mycotoxins Produced by Fusarium graminearum and Fusarium poae in Malting Barley in Argentina" Toxins 10, no. 2: 78. https://doi.org/10.3390/toxins10020078
APA StyleNogueira, M. S., Decundo, J., Martinez, M., Dieguez, S. N., Moreyra, F., Moreno, M. V., & Stenglein, S. A. (2018). Natural Contamination with Mycotoxins Produced by Fusarium graminearum and Fusarium poae in Malting Barley in Argentina. Toxins, 10(2), 78. https://doi.org/10.3390/toxins10020078







