Biomarker of Aflatoxin Ingestion: 1H NMR-Based Plasma Metabolomics of Dairy Cows Fed Aflatoxin B1 with or without Sequestering Agents
Abstract
1. Introduction
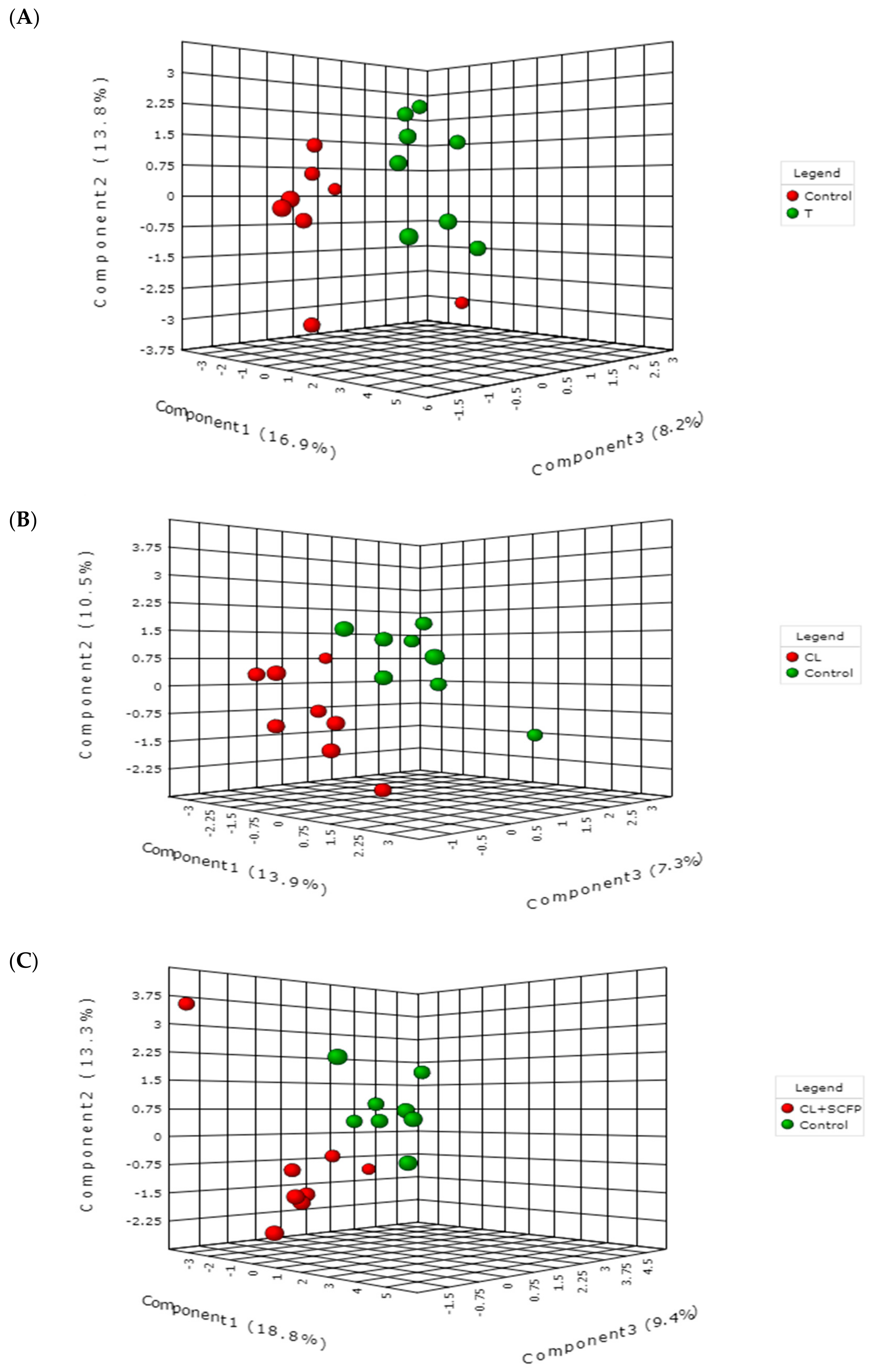
2. Results and Discussion
3. Materials and Methods
3.1. Animals, Housing, and Feeding
3.2. Blood Sample Collection
3.3. Sample Preparation and NMR Spectroscopy
3.4. Data and Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bennett, J.W.; Klich, M. Mycotoxins. Clin. Microbiol. Rev. 2003, 16, 497–516. [Google Scholar] [CrossRef] [PubMed]
- Squire, R.A. Ranking animal carcinogens: A proposed regulatory approach. Science 1981, 214, 877–880. [Google Scholar] [CrossRef] [PubMed]
- Upadhaya, S.D.; Park, M.A.; Ha, J.K. Mycotoxins and their biotransformation in the rumen: A review. Asian-Australas. J. Anim. Sci. 2010, 23, 1250–1260. [Google Scholar] [CrossRef]
- Fink-Gremmels, J. Mycotoxins in cattle feeds and carry-over to dairy milk: A review. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2008, 25, 172–180. [Google Scholar] [CrossRef] [PubMed]
- Kutz, R.E.; Sampson, J.D.; Pompeu, L.B.; Ledoux, D.R.; Spain, J.N.; Vazquez-Anon, M.; Rottinghaus, G.E. Efficacy of Solis, NovasilPlus, and MTB-100 to reduce aflatoxin M1 levels in milk of early to mid-lactation dairy cows fed aflatoxin B1. J. Dairy Sci. 2009, 92, 3959–3963. [Google Scholar] [CrossRef] [PubMed]
- Queiroz, O.C.M.; Han, J.H.; Staples, C.R.; Adesogan, A.T. Effect of adding a mycotoxin-sequestering agent on milk aflatoxin M1 concentration and the performance and immune response of dairy cattle fed an aflatoxin B1-contaminated diet. J. Dairy Sci. 2012, 95, 5901–5908. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Ogunade, I.M.; Kim, D.H.; Li, X.; Pech-Cervantes, A.A.; Arriola, K.G.; Oliveira, A.S.; Driver, J.P.; Ferraretto, L.F.; Staples, C.R.; et al. Effect of sequestering agents based on a Saccharomyces cerevisiae fermentation product and clay on the health and performance of lactating dairy cows challenged with dietary aflatoxin B1. J. Dairy Sci. 2018, 101, 3008–3020. [Google Scholar] [CrossRef] [PubMed]
- Dhanasekaran, D.; Shanmugapriya, S.; Thajuddin, N.; Panneerselvam, A. Aflatoxins and aflatoxicosis in human and animals. In Aflatoxins—Biochemistry and Molecular Biology; Guevara-Gonzalez, R.G., Ed.; InTech: Houston, TX, USA, 2011; pp. 221–254. [Google Scholar]
- Whitaker, T.B.; Slate, A.; Doko, B.; Maestroni, B.; Cannavan, A. Sampling Procedures to Detect Mycotoxins in Agricultural Commodities; Springer: New York, NY, USA, 2011. [Google Scholar]
- Wild, C.P.; Turner, P.C. The toxicology of aflatoxins as a basis for public health decisions. Mutagenesis 2002, 17, 471–481. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, J.K.; Lindon, J.C.; Holmes, E. Metabonomics: Understanding the metabolic responses of living systems to pathophysiological stimuli via multivariate statistical analysis of biological NMR spectroscopic data. Xenobiotica 1999, 29, 1181–1189. [Google Scholar] [CrossRef] [PubMed]
- Maher, A.D.; Rochfort, S.J. Applications of NMR in dairy research. Metabolites 2014, 4, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.K.; Choi, Y.H.; Verpoorte, R. NMR-based plant metabolomics: Where do we stand, where do we go? Trends Biotechnol. 2011, 29, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Tikunov, A.P.; Johnson, C.B.; Lee, H.; Stoskopf, M.K.; Macdonald, J.M. Metabolomic investigations of American oysters Using 1H-NMR spectroscopy. Mar. Drugs 2010, 8, 2578–2596. [Google Scholar] [CrossRef] [PubMed]
- Emwas, A.H. The strengths and weaknesses of NMR spectroscopy and mass spectrometry with particular focus on metabolomics research. Metabonomics Methods Protoc. 2015, 1277, 161–193. [Google Scholar] [CrossRef]
- Sun, L.W.; Zhang, H.Y.; Wu, L.; Shu, S.; Xia, C.; Xu, C.; Zheng, J.S. 1H-Nuclear magnetic resonance-based plasma metabolic profiling of dairy cows with clinical and subclinical ketosis. J. Dairy Sci. 2014, 97, 1552–1562. [Google Scholar] [CrossRef] [PubMed]
- Sporn, M.B.; Dingman, C.W.; Phelps, H.L.; Wogan, G.N. Aflatoxin B1: Binding to DNA in vitro and alteration of RNA metabolism in vivo. Science 1966, 151, 1539–1541. [Google Scholar] [CrossRef] [PubMed]
- Garvican, L.; Cajone, F.; Rees, K.R. The mechanism of action of aflatoxin B1 on protein synthesis; Observations on malignant, viral transformed and untransformed cells in culture. Chem.-Biol. Interact. 1973, 7, 39–50. [Google Scholar] [CrossRef]
- Doelman, J.; Kim, J.J.M.; Carson, M.; Metcalf, J.A.; Cant, J.P. Branched chain amino acid and lysine deficiencies exert different effects on mammary translational regulation. J. Dairy Sci. 2015, 98, 7846–7855. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Proud, C.G. The mTOR pathway in the control of protein synthesis. Physiology 2006, 21, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Jones, F.T.; Genter, M.B.; Hagler, W.M.; Hansen, J.A.; Mowrey, B.A.; Poore, M.H.; Whitlow, L.W. Understanding and Coping with Effects of Mycotoxins in Livestock Feed and Forage; North Carolina Cooperative Extension Service: Winston-Salem, NC, USA, 1994; pp. 1–14. [Google Scholar]
- Toerien, C.A.; Trout, D.R.; Cant, J.P. Nutritional stimulation of milk protein yield of cows is associated with changes in phosphorylation of mammary eukaryotic initiation factor 2 and ribosomal s6 kinase1. J. Nutr. 2010, 140, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Appuhamy, J.A.; Knapp, J.R.; Becvar, O.; Escobar, J.; Hanigan, M.D. Effects of jugular-infused lysine, methionine, and branched-chain amino acids on milk protein synthesis in high-producing dairy cows. J. Dairy Sci. 2011, 94, 1952–1960. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, N.B.; Storm, A.; Raun, B.M.L.; Røjen, B.A.; Harmon, D.L. Metabolism of silage alcohols in lactating dairy cows. J. Dairy Sci. 2007, 90, 1364–1377. [Google Scholar] [CrossRef]
- Battacone, G.; Nudda, A.; Cannas, A.; Borlino, A.C.; Bomboi, G.; Pulina, G. Excretion of aflatoxin M1 in milk of dairy ewes treated with different doses of aflatoxin B1. J. Dairy Sci. 2003, 86, 2667–2675. [Google Scholar] [CrossRef]
- Reed, K.M.; Mendoza, K.M.; Abrahante, J.E.; Coulombe, R.A. Comparative Response of the Hepatic Transcriptomes of Domesticated and Wild Turkey to Aflatoxin B1. Toxins 2018, 10, 42. [Google Scholar] [CrossRef] [PubMed]
- Shang, L.; Zhu, G.; Su, H.; Chen, B.; Ye, X.; Chen, X.; Xiao, K.; Li, L.; Peng, M.; Peng, T. Identification of alcohol dehydrogenase as a potential prognostic marker in HBV-related hepatocellular carcinoma. Int. J. Clin. Exp. Med. 2017, 10, 4457–4472. [Google Scholar]
- Allen, R.H.; Stabler, S.P.; Lindenbaum, J. Serum betaine, N,N-dimethylglycine and N-methylglycine levels in patients with cobalamin and folate deficiency and related inborn errors of metabolism. Metabolism 1993, 42, 1448–1460. [Google Scholar] [CrossRef]
- Pinto, M.C.; Simao, F.; da Costa, F.L.; Rosa, D.V.; de Paiva, M.J.; Resende, R.R.; Romano-Silva, M.A.; Gomez, M.V.; Gomez, R.S. Sarcosine preconditioning induces ischemic tolerance against global cerebral ischemia. Neuroscience 2014, 271, 160–169. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Sun, L.; Shu, S.; Zhu, K.; Xu, C.; Wang, J.; Wang, H. Nuclear magnetic resonance-based serum metabolic profiling of dairy cows with footrot. J. Vet. Med. Sci. 2016, 78, 1421–1428. [Google Scholar] [CrossRef] [PubMed]
- Huber, K.; Dänicke, S.; Rehage, J.; Sauerwein, H.; Otto, W.; Rolle-Kampczyk, U.; von Bergen, M.M. Metabotypes with properly functioning mitochondria and anti-inflammation predict extended productive life span in dairy cows. Sci. Rep. 2016, 6, 24642. [Google Scholar] [CrossRef] [PubMed]
- Leicester, H.C.W.; Robinson, P.H.; Erasmus, L.J. Effects of two yeast based direct fed microbials on performance of high producing dairy cows. Anim. Feed Sci. Technol. 2016, 215, 58–72. [Google Scholar] [CrossRef]
- Swanepoel, N.; Robinson, P.H.; Erasmus, L.J. Rumen microbial protein flow and plasma amino acid concentrations in early lactation multiparity Holstein cows fed commercial rations, and some relationships with dietary nutrients. Livest. Sci. 2016, 190, 58–69. [Google Scholar] [CrossRef]
- Erasmus, L.J.; Botha, P.M.; Kistner, A. Effect of yeast culture supplement on production, rumen fermentation, and duodenal nitrogen flow in dairy cows. J. Dairy Sci. 1992, 75, 3056–3065. [Google Scholar] [CrossRef]
- Robinson, P.H. Yeast Products for Growing and Lactating Ruminants: A Literature Summary of Impacts on Rumen Fermentation and Performance. 2010. Available online: https://animalscience.ucdavis.edu/sites/g/files/dgvnsk446/files/inline-files/Web200901.pdf (accessed on 16 October 2018).
- Jiang, Y.; Ogunade, I.M.; Arriola, K.G.; Qi, M.; Vyas, D.; Staples, C.R.; Adesogan, A.T. Effects of the dose and viability of Saccharomyces cerevisiae. 2. Ruminal fermentation, performance of lactating dairy cows, and correlations between ruminal bacteria abundance and performance measures. J. Dairy Sci. 2017, 100, 8102–8118. [Google Scholar] [CrossRef] [PubMed]
- Spring, P.; Wenk, C.; Connolly, A.; Kiers, A. A review of 733 published trials in Bio-Mos®, a mannan oligosaccharide, and Actigen®, a second generation mannose rich fraction, on farm and companion animals. J. Appl. Anim. Nutr. 2015, 3, 1–11. [Google Scholar] [CrossRef]
- Kelly, D.; Begbie, R.; King, T.P. Nutritional influences on interactions between bacteria and the small intestinal mucosa. Nutr. Res. Rev. 1994, 7, 233–257. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, J.T.; Bittar, C.M.; Ferreira, L.S. Evaluation of mannan-oligosaccharides offered in milk replacers or calf starters and their effect on performance and rumen development of dairy calves. R. Bras. Zootec. 2012, 41, 746–752. [Google Scholar] [CrossRef]
- Kuipers, S.; Aerts, P.C.; van Dijk, H. Differential microorganism-induced mannose-binding lectin activation. FEMS Immunol. Med. Microbiol. 2003, 36, 33–39. [Google Scholar] [CrossRef]
- Takahashi, K. Mannose-binding lectin and the balance between immune protection and complication. Expert Rev. Anti. Infect. Ther. 2011, 9, 1179–1190. [Google Scholar] [CrossRef] [PubMed]
- Duff, G.C.; Galyean, M.L. Board-invited review: Recent advances in management of highly stressed, newly received feedlot cattle. J. Anim. Sci. 2007, 85, 823–840. [Google Scholar] [CrossRef] [PubMed]
- Burdick Sanchez, N.C.; Young, T.R.; Carroll, J.A.; Corley, J.R.; Rathmann, R.J.; Johnson, B.J. Yeast cell wall supplementation alters the metabolic responses of crossbred heifers to an endotoxin challenge. Innate Immun. 2014, 20, 104–112. [Google Scholar] [CrossRef] [PubMed]
- NRC. Nutrient Requirements of Dairy Cattle, 7th rev. ed.; National Academies Press: Washington, DC, USA, 2001. [Google Scholar]
- Psychogios, N.; Hau, D.D.; Peng, J.; Guo, A.C.; Mandal, R.; Bouatra, S.; Sinelnikov, I.; Krishnamurthy, R.; Eisner, R.; Gautam, B.; et al. The human serum metabolome. PLoS ONE. 2011, 6, e16957. [Google Scholar] [CrossRef] [PubMed]
- Saude, E.J.; Slupksy, C.M.; Sykes, B.D. Optimization of NMR analysis of biological fluids for quantitative accuracy. Metabolomics 2006, 2, 113–123. [Google Scholar] [CrossRef]
- Ravanbakhsh, S.; Liu, P.; Bjorndahl, T.C.; Mandal, R.; Grant, J.R.; Wilson, M.; Eisner, R.; Sinelnikov, I.; Hu, X.; Luchinat, C.; et al. Accurate, fully-automated NMR spectral profiling for metabolomics. PLoS ONE 2015, 10, e0124219. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Wishart, D.S. Using metaboAnalyst 3.0 for comprehensive metabolomics data analysis. Curr. Prot. Bioinform. 2016, 55. [Google Scholar] [CrossRef] [PubMed]
- Graham, S.F.; Kumar, P.K.; Bjorndahl, T.; Han, B.; Yilmaz, A.; Sherman, E.; Bahado-Singh, R.O.; Wishart, D.; Mann, D.; Green, B.D. Metabolic signatures of Huntington’s disease (HD): 1H NMR analysis of the polar metabolome in post-mortem human brain. Biochim. Biophys. Acta 2016, 1862, 1675–1684. [Google Scholar] [CrossRef] [PubMed]
- Gromski, P.S.; Muhamadali, H.; Ellis, D.I.; Xu, Y.; Correa, E.; Turner, M.L.; Goodacre, R. A tutorial review: Metabolomics and partial least squares-discriminant analysis—A marriage of convenience or a shotgun wedding. Anal. Chim. Acta 2015, 879, 10–23. [Google Scholar] [CrossRef] [PubMed]
- Liang, Q.; Liu, H.; Wang, C.; Li, B. Phenotypic characterization analysis of human hepatocarcinoma by urine metabolomics approach. Sci. Rep. 2016, 6, 19763. [Google Scholar] [CrossRef] [PubMed]
- Kalantari, S.; Nafar, M.; Samavat, S.; Parvin, M. NMR-based metabolomics exploring urinary biomarkers correlated with proteinuria in focal segmental glomerulosclerosis: A pilot study: Urine metabolomics based on NMR. Magn. Reson. Chem. 2017, 54, 821–826. [Google Scholar] [CrossRef] [PubMed]
| Item | Treatment 2 | SEM | Control vs. T | Control vs. CL | Control vs. CL + SCFP | |||
|---|---|---|---|---|---|---|---|---|
| Control | T | CL | CL + SCFP | |||||
| l-Glutamic acid | 295 | 271 | 274 | 374 | 17.2 | 0.35 | 0.41 | 0.01 |
| l-Alanine | 395 | 310 | 370 | 590 | 23.1 | 0.01 | 0.38 | 0.01 |
| l-Leucine | 297 | 229 | 297 | 387 | 14.7 | 0.01 | 0.99 | 0.01 |
| l-Valine | 382 | 347 | 375 | 468 | 19.6 | 0.06 | 0.78 | 0.01 |
| l-Proline | 153 | 133 | 154 | 205 | 9.67 | 0.15 | 0.91 | 0.01 |
| l-Threonine | 143 | 126 | 133 | 188 | 10.2 | 0.27 | 0.51 | 0.01 |
| l-Isoleucine | 135 | 131 | 137 | 172 | 9.09 | 0.71 | 0.92 | 0.01 |
| l-Serine | 148 | 160 | 147 | 179 | 10.5 | 0.36 | 0.94 | 0.03 |
| l-Arginine | 128 | 96 | 131 | 159 | 12.7 | 0.07 | 0.86 | 0.09 |
| l-Phenylalanine | 72.1 | 71.1 | 68.4 | 85.5 | 4.46 | 0.89 | 0.57 | 0.05 |
| d-mannose | 68.8 | 66.9 | 66.6 | 78.8 | 2.87 | 0.65 | 0.61 | 0.02 |
| l-Aspartic acid | 23.5 | 26.9 | 29.5 | 43.5 | 4.78 | 0.62 | 0.39 | 0.01 |
| Acetic acid | 1348 | 973 | 1258 | 1340 | 103 | 0.02 | 0.54 | 0.95 |
| Sarcosine | 1.83 | 1.74 | 1.50 | 1.95 | 0.12 | 0.59 | 0.07 | 0.45 |
| Ethanol | 6.81 | 19.9 | 6.38 | 5.19 | 9.29 | 0.01 | 0.85 | 0.26 |
| l-Lysine | 165 | 180 | 171 | 208 | 14.4 | 0.50 | 0.81 | 0.05 |
| Item | AUC 1 | p-Value 2 | FC 3 | Sensitivity | Specificity |
|---|---|---|---|---|---|
| Acetic acid | 0.91 | 0.01 | 0.72 | 1.0 | 0.8 |
| l-Arginine | 0.89 | 0.04 | 0.75 | 0.9 | 0.9 |
| Ethanol | 0.88 | 0.02 | 3.56 | 0.9 | 0.9 |
| l-Alanine | 0.86 | 0.01 | 0.78 | 0.9 | 0.9 |
| l-Methylhistidine | 0.86 | 0.01 | 0.88 | 0.8 | 0.8 |
| l-Proline | 0.81 | 0.04 | 0.89 | 0.8 | 0.8 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ogunade, I.; Jiang, Y.; Adeyemi, J.; Oliveira, A.; Vyas, D.; Adesogan, A. Biomarker of Aflatoxin Ingestion: 1H NMR-Based Plasma Metabolomics of Dairy Cows Fed Aflatoxin B1 with or without Sequestering Agents. Toxins 2018, 10, 545. https://doi.org/10.3390/toxins10120545
Ogunade I, Jiang Y, Adeyemi J, Oliveira A, Vyas D, Adesogan A. Biomarker of Aflatoxin Ingestion: 1H NMR-Based Plasma Metabolomics of Dairy Cows Fed Aflatoxin B1 with or without Sequestering Agents. Toxins. 2018; 10(12):545. https://doi.org/10.3390/toxins10120545
Chicago/Turabian StyleOgunade, Ibukun, Yun Jiang, James Adeyemi, Andre Oliveira, Diwakar Vyas, and Adegbola Adesogan. 2018. "Biomarker of Aflatoxin Ingestion: 1H NMR-Based Plasma Metabolomics of Dairy Cows Fed Aflatoxin B1 with or without Sequestering Agents" Toxins 10, no. 12: 545. https://doi.org/10.3390/toxins10120545
APA StyleOgunade, I., Jiang, Y., Adeyemi, J., Oliveira, A., Vyas, D., & Adesogan, A. (2018). Biomarker of Aflatoxin Ingestion: 1H NMR-Based Plasma Metabolomics of Dairy Cows Fed Aflatoxin B1 with or without Sequestering Agents. Toxins, 10(12), 545. https://doi.org/10.3390/toxins10120545







