Transfer of Deoxynivalenol (DON) through Placenta, Colostrum and Milk from Sows to Their Offspring during Late Gestation and Lactation
Abstract
1. Introduction
2. Results
2.1. DON Uptake in Sows
2.1.1. DON and DON Derivatives in Sow Feed
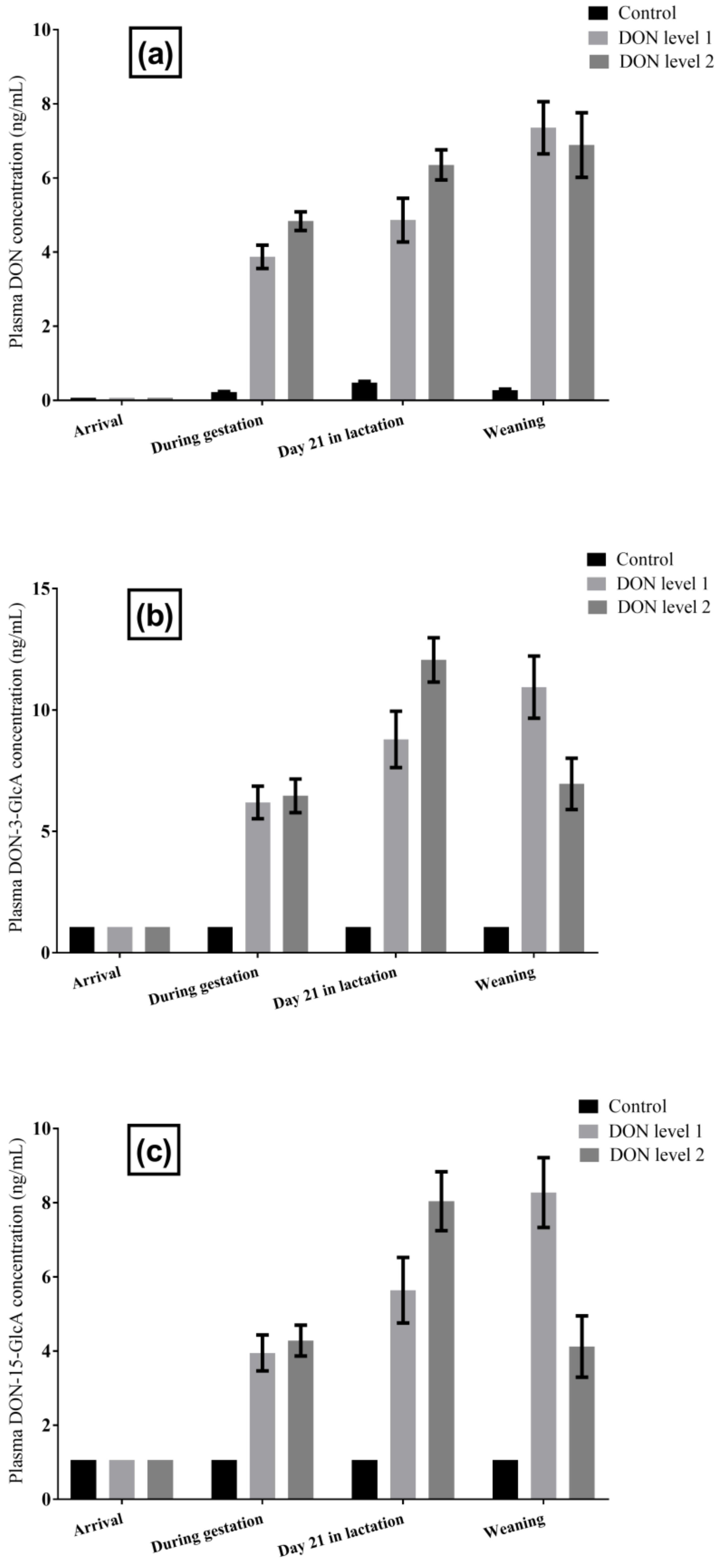
2.1.2. DON and DON Metabolites in Sow Plasma
2.2. DON Transfer from Sows to Piglets
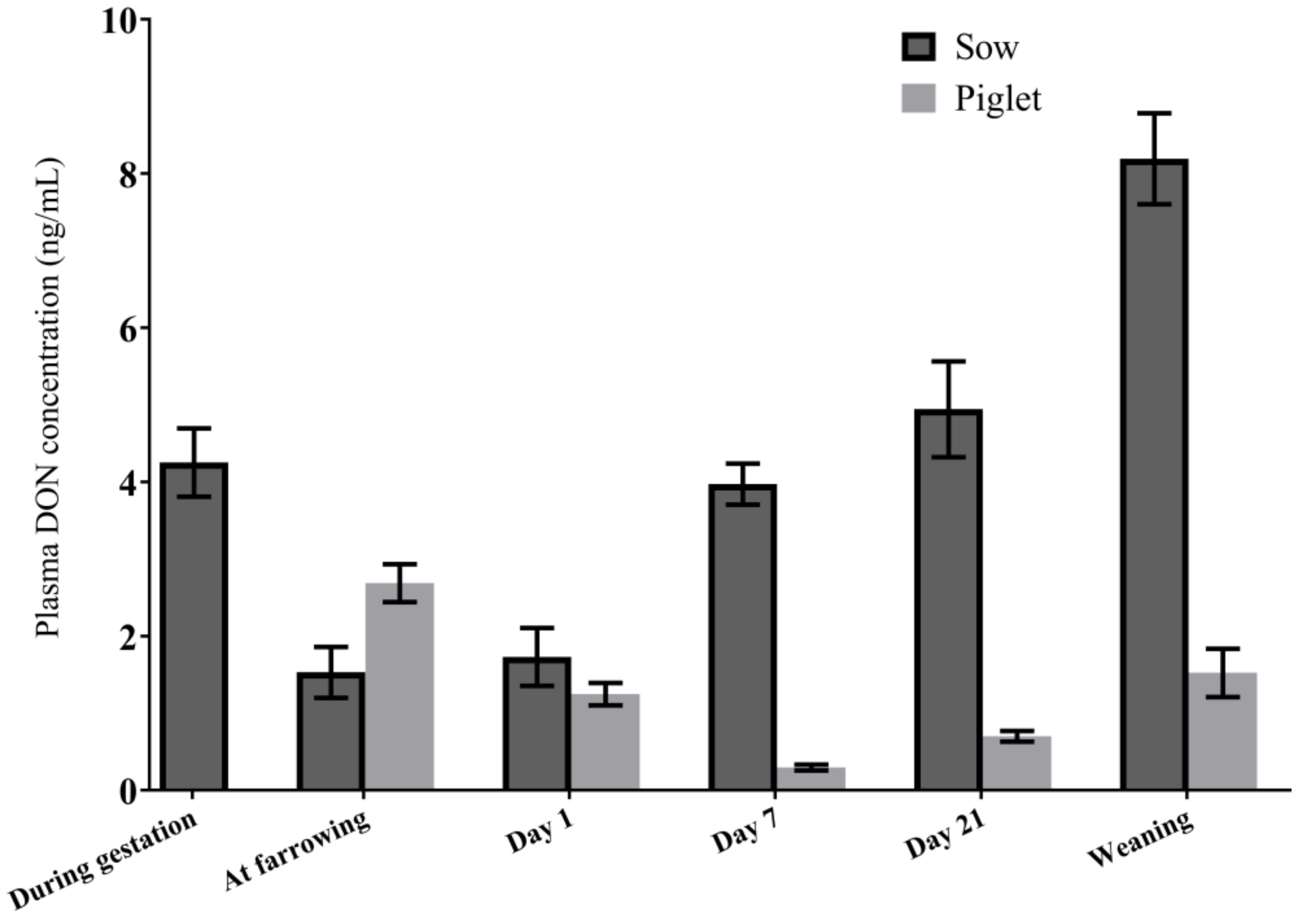
2.2.1. DON Plasma Concentrations in Sows in the Transfer Study
2.2.2. Piglet Survival and Growth Performance
2.2.3. DON and DON Metabolites in Piglet Plasma
2.2.4. Association between Plasma DON Concentrations in Sows and Piglets
2.2.5. DON and DON Metabolites in Sow Milk
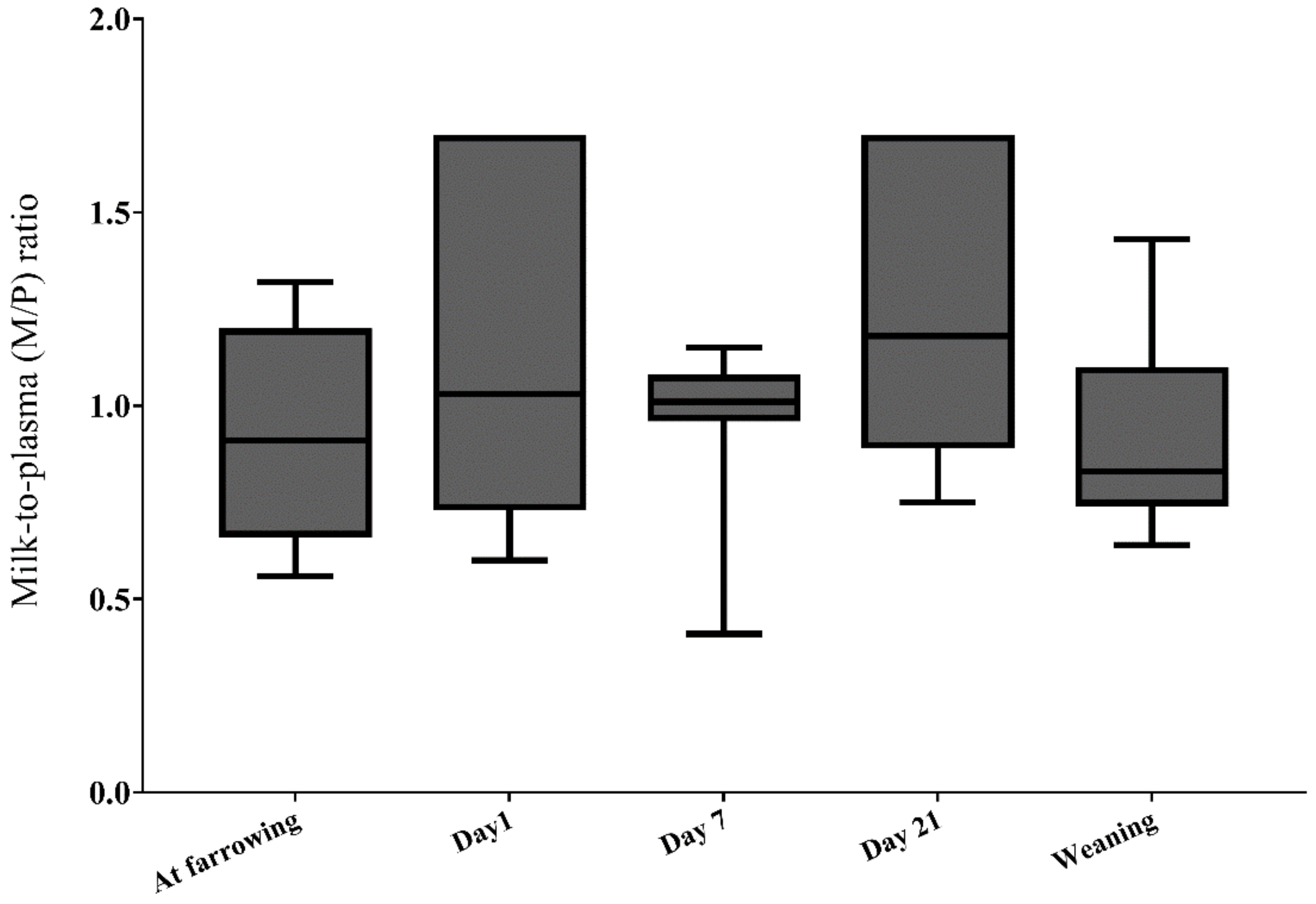
2.2.6. DON Transfer from Sow Plasma to Milk (Lactational Transfer)
3. Discussion
3.1. DON Uptake in Sows
3.2. Plasma Profiles of DON and DON-Glucuronides in Different Age Groups in Pigs
3.3. Growth Performance of Piglets of DON-Exposed Sows
3.4. DON Transfer from Sows to Unborn Piglets
3.5. DON Transfer from Sows to Suckling Piglets
4. Conclusions
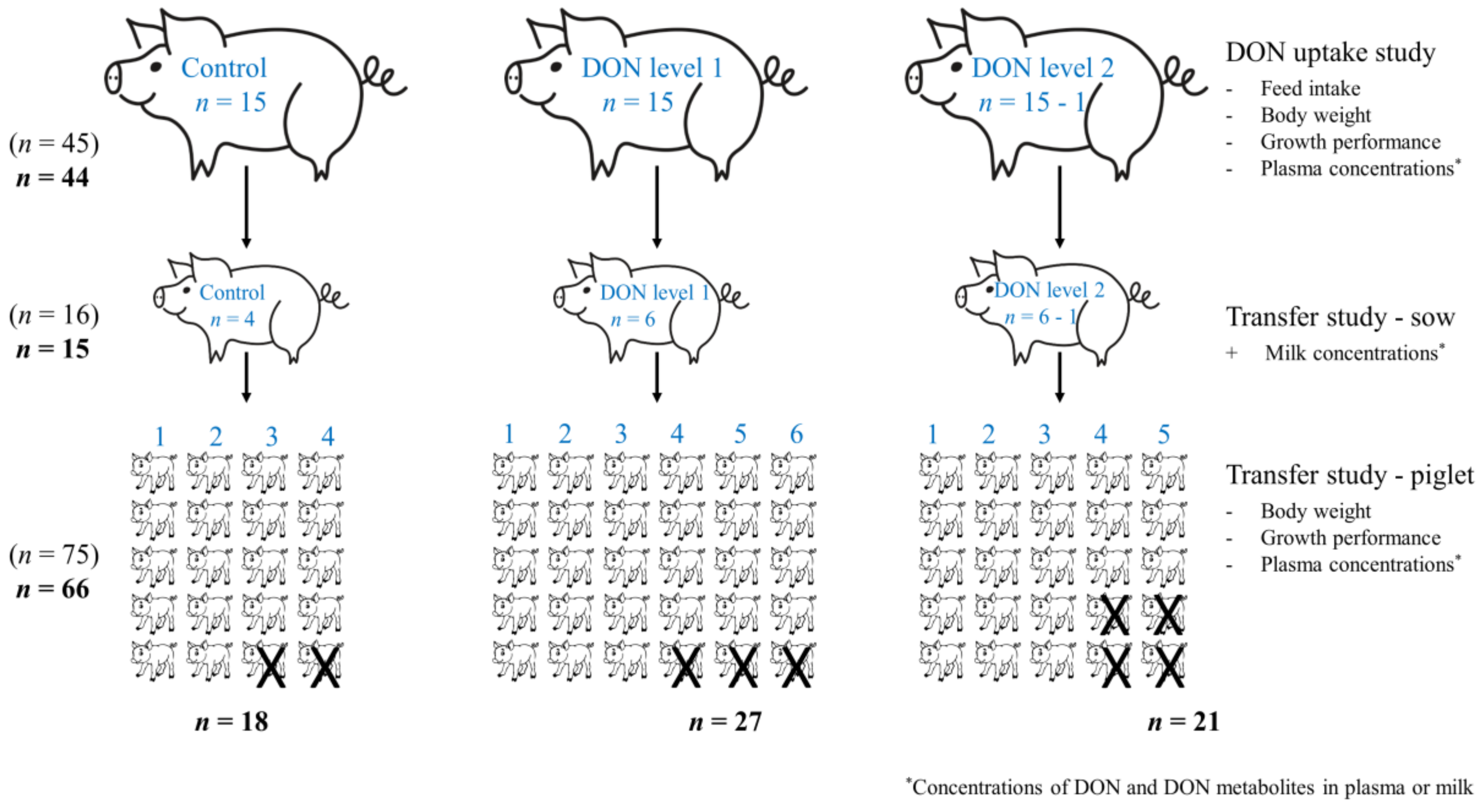
5. Materials and Methods
5.1. Animals, Housing, and Management
5.2. Origin and Preparation of the Naturally Contaminated Experimental Diets
5.3. Measuring of Feed Intake and Growth Performance of the Sows
5.4. Reagents for Chemical Analyses
5.5. Analysis of Mycotoxins in Experimental Diets
5.6. Blood and Milk Sampling for Analysis of Mycotoxins
5.6.1. DON Uptake Study
5.6.2. Transfer Study
5.7. Sample Preparation of Plasma for Analysis of Mycotoxins
5.8. Sample Preparation of Sow Milk for Analysis of DON, DOM-1 and DON-15-GlcA
5.9. Quantitative Analysis of Mycotoxins in Plasma and Milk
5.10. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pestka, J.J. Deoxynivalenol: Mechanisms of action, human exposure, and toxicological relevance. Arch. Toxicol. 2010, 84, 663–679. [Google Scholar] [CrossRef] [PubMed]
- Pestka, J.J. Deoxynivalenol: Toxicity, mechanisms and animal health risks. Anim. Feed Sci. Technol. 2007, 137, 283–298. [Google Scholar] [CrossRef]
- Sobrova, P.; Adam, V.; Vasatkova, A.; Beklova, M.; Zeman, L.; Kizek, R. Deoxynivalenol and its toxicity. Interdiscip. Toxicol. 2010, 3, 94–99. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Munkvold, G.P. Mycotoxins in ethanol co-products: Modeling economic impacts on the livestock industry and management strategies. J. Agric. Food Chem. 2008, 56, 3900–3911. [Google Scholar] [CrossRef] [PubMed]
- Faeste, C.K.; Ivanova, L.; Sayyari, A.; Hansen, U.; Sivertsen, T.; Uhlig, S. Prediction of deoxynivalenol toxicokinetics in humans by in vitro-to-in vivo extrapolation and allometric scaling of in vivo animal data. Arch. Toxicol. 2018, 2195–2216. [Google Scholar] [CrossRef] [PubMed]
- Pestka, J.J.; Clark, E.S.; Schwartz-Zimmermann, H.E.; Berthiller, F. Sex is a determinant for deoxynivalenol metabolism and elimination in the mouse. Toxins 2017, 9, 240. [Google Scholar] [CrossRef] [PubMed]
- Andretta, I.; Kipper, M.; Lehnen, C.R.; Hauschild, L.; Vale, M.M.; Lovatto, P.A. Meta-analytical study of productive and nutritional interactions of mycotoxins in growing pigs. Animal 2012, 6, 1476–1482. [Google Scholar] [CrossRef]
- Ghareeb, K.; Awad, W.A.; Bohm, J.; Zebeli, Q. Impacts of the feed contaminant deoxynivalenol on the intestine of monogastric animals: Poultry and swine. J. Appl. Toxicol. 2015, 35, 327–337. [Google Scholar] [CrossRef]
- Accensi, F.; Pinton, P.; Callu, P.; Abella-Bourges, N.; Guelfi, J.F.; Grosjean, F.; Oswald, I.P. Ingestion of low doses of deoxynivalenol does not affect hematological, biochemical, or immune responses of piglets. J. Anim. Sci. 2006, 84, 1935–1942. [Google Scholar] [CrossRef]
- Debouck, C.; Haubruge, E.; Bollaerts, P.; van Bignoot, D.; Brostaux, Y.; Werry, A.; Rooze, M. Skeletal deformities induced by the intraperitoneal administration of deoxynivalenol (vomitoxin) in mice. Int. Orthop. 2001, 25, 194–198. [Google Scholar] [CrossRef]
- Tiemann, U.; Brussow, K.P.; Kuchenmeister, U.; Jonas, L.; Pohland, R.; Reischauer, A.; Jager, K.; Dänicke, S. Changes in the spleen and liver of pregnant sows and full-term piglets after feeding diets naturally contaminated with deoxynivalenol and zearalenone. Vet. J. 2008, 176, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, J.K.; Vikstrom, A.C.; Turner, P.; Knudsen, L.E. Deoxynivalenol transport across the human placental barrier. Food Chem. Toxicol. 2011, 49, 2046–2052. [Google Scholar] [CrossRef]
- Mose, T.; Mathiesen, L.; Karttunen, V.; Nielsen, J.K.S.; Sieppi, E.; Kummu, M.; Mørck, T.A.; Myöhänen, K.; Partanen, H.; Vähäkangas, K.; et al. Meta-analysis of data from human ex vivo placental perfusion studies on genotoxic and immunotoxic agents within the integrated european project newgeneris. Placenta 2012, 33, 433–439. [Google Scholar] [CrossRef] [PubMed]
- Goyarts, T.; Dänicke, S.; Brussow, K.P.; Valenta, H.; Ueberschar, K.H.; Tiemann, U. On the transfer of the fusarium toxins deoxynivalenol (DON) and zearalenone (ZON) from sows to their fetuses during days 35–70 of gestation. Toxicol. Lett. 2007, 171, 38–49. [Google Scholar] [CrossRef] [PubMed]
- Dänicke, S.; Brussow, K.P.; Goyarts, T.; Valenta, H.; Ueberschar, K.H.; Tiemann, U. On the transfer of the fusarium toxins deoxynivalenol (DON) and zearalenone (ZON) from the sow to the full-term piglet during the last third of gestation. Food Chem. Toxicol. 2007, 45, 1565–1574. [Google Scholar] [CrossRef]
- Fink-Gremmels, J. Mycotoxins in cattle feeds and carry-over to dairy milk: A review. Food Addit. Contam. Part A 2008, 25, 172–180. [Google Scholar] [CrossRef] [PubMed]
- Seeling, K.; Dänicke, S.; Valenta, H.; Van Egmond, H.P.; Schothorst, R.C.; Jekel, A.A.; Lebzien, P.; Schollenberger, M.; Razzazi-Fazeli, E.; Flachowsky, G. Effects of fusarium toxin-contaminated wheat and feed intake level on the biotransformation and carry-over of deoxynivalenol in dairy cows. Food Addit. Contam. 2006, 23, 1008–1020. [Google Scholar] [CrossRef] [PubMed]
- Sayyari, A.; Framstad, T.; Krogenæs, A.K.; Sivertsen, T. Effects of feeding naturally contaminated deoxynivalenol diets to sows during late gestation and lactation in a high-yield specific pathogen-free herd. Porcine Health Manag. 2018, 4, 26. [Google Scholar] [CrossRef] [PubMed]
- Fleishaker, J.C. Models and methods for predicting drug transfer into human milk. Adv. Drug. Deliv. Rev. 2003, 55, 643–652. [Google Scholar] [CrossRef]
- Theil, P.K. Transition feeding of sows. In The Gestating and Lactating Sow; Wageningen Academic Publishers: Wageningen, The Netherlands, 2014; pp. 147–172. [Google Scholar]
- Eissen, J.J.; Apeldoorn, E.J.; Kanis, E.; Verstegen, M.W.; de Greef, K.H. The importance of a high feed intake during lactation of primiparous sows nursing large litters. J. Anim. Sci. 2003, 81, 594–603. [Google Scholar] [CrossRef]
- Goyarts, T.; Dänicke, S.; Valenta, H.; Ueberschar, K.H. Carry-over of fusarium toxins (deoxynivalenol and zearalenone) from naturally contaminated wheat to pigs. Food Addit. Contam. 2007, 24, 369–380. [Google Scholar] [CrossRef] [PubMed]
- Sayyari, A.; Fæste, C.K.; Hansen, U.; Uhlig, S.; Framstad, T.; Schatzmayr, D.; Sivertsen, T. Effects and biotransformation of the mycotoxin deoxynivalenol in growing pigs fed with naturally contaminated pelleted grains with and without the addition of coriobacteriaceum dsm 11798. Food Addit. Contam. Part A 2018, 35, 1394–1409. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Peng, M.; Zhang, T.; Tao, J.-S.; Cai, Z.-Z.; Zhang, Y. Quantification of conjugated metabolites of drugs in biological matrices after the hydrolysis with β-glucuronidase and sufatase: A review of bio-analytical methods. Biomed. Chromatogr. 2013, 27, 1280–1295. [Google Scholar] [CrossRef] [PubMed]
- Bracarense, A.P.F.L.; Basso, K.M.; Da Silva, E.O.; Payros, D.; Oswald, I.P. Deoxynivalenol in the liver and lymphoid organs of rats: Effects of dose and duration on immunohistological changes. World Mycotoxin J. 2016, 10, 89–96. [Google Scholar] [CrossRef]
- Pestka, J.J.; Amuzie, C.J. Tissue distribution and proinflammatory cytokine gene expression following acute oral exposure to deoxynivalenol: Comparison of weanling and adult mice. Food Chem. Toxicol. 2008, 46, 2826–2831. [Google Scholar] [CrossRef]
- Gutzwiller, A. Effects of deoxynivalenol (DON) in the lactation diet on the feed intake and fertility of sows. Mycotoxin Res. 2010, 26, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Llano, G.; Smith, T.K. The effects of feeding grains naturally contaminated with fusarium mycotoxins with and without a polymeric glucomannan adsorbent on lactation, serum chemistry, and reproductive performance after weaning of first-parity lactating sows. J. Anim. Sci. 2007, 85, 1412–1423. [Google Scholar] [CrossRef]
- Diaz-Llano, G.; Smith, T.K. Effects of feeding grains naturally contaminated with fusarium mycotoxins with and without a polymeric glucomannan mycotoxin adsorbent on reproductive performance and serum chemistry of pregnant gilts. J. Anim. Sci. 2006, 84, 2361–2366. [Google Scholar] [CrossRef]
- Jakovac-Strajn, B.; Vengust, A.; Pestevsek, U. Effects of a deoxynivalenol-contaminated diet on the reproductive performance and immunoglobulin concentrations in pigs. Vet. Rec. 2009, 165, 713–718. [Google Scholar]
- Goyarts, T.; Dänicke, S.; Rothkotter, H.J.; Spilke, J.; Tiemann, U.; Schollenberger, M. On the effects of a chronic deoxynivalenol intoxication on performance, haematological and serum parameters of pigs when diets are offered either for ad libitum consumption or fed restrictively. J. Vet. Med. A Physiol. Pathol. Clin. Med. 2005, 52, 305–314. [Google Scholar] [CrossRef]
- Munoz, K.; Blaszkewicz, M.; Campos, V.; Vega, M.; Degen, G.H. Exposure of infants to ochratoxin A with breast milk. Arch. Toxicol. 2014, 88, 837–846. [Google Scholar] [PubMed]
- Devreese, M.; Croubels, S.; De Baere, S.; Gehring, R.; Antonissen, G. Comparative toxicokinetics and plasma protein binding of ochratoxin A in four avian species. J. Agric. Food Chem. 2018, 66, 2129–2135. [Google Scholar] [CrossRef] [PubMed]
- Malachova, A.; Sulyok, M.; Beltran, E.; Berthiller, F.; Krska, R. Optimization and validation of a quantitative liquid chromatography-tandem mass spectrometric method covering 295 bacterial and fungal metabolites including all regulated mycotoxins in four model food matrices. J. Chromatogr. A 2014, 1362, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Uhlig, S.; Ivanova, L.; Fæste, C.K. Enzyme-assisted synthesis and structural characterization of the 3-, 8-, and 15-glucuronides of deoxynivalenol. J. Agric. Food. Chem. 2013, 61, 2006–2012. [Google Scholar] [CrossRef] [PubMed]
- Ivanova, L.; Sahlstrøm, S.; Rud, I.; Uhlig, S.; Fæste, C.K.; Eriksen, G.S.; Divon, H.H. Effect of primary processing on the distribution of free and modified fusarium mycotoxins in naturally contaminated oats. World Mycotoxin. J. 2017, 10, 73–88. [Google Scholar] [CrossRef]
- Framstad, T.; Sjaastad, Ø.; Aass, R.A. Blodprøvetaking på gris (blood sampling in pigs). Nor. Vet. Tidsskr. 1988, 100, 265–272. [Google Scholar]
- Uhlig, S.; Stanic, A.; Hussain, F.; Miles, C.O. Selectivity of commercial immunoaffinity columns for modified forms of the mycotoxin 4-deoxynivalenol (DON). J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2017, 1061–1062, 322–326. [Google Scholar] [CrossRef]
- Croghan, C.W.; Egeghy, P.P. Methods of dealing with values below the limit of detection using SAS. In Proceedings of the Southeastern SAS User Group, St. Petersburg, FL, USA, 22–24 Sepember 2003. [Google Scholar]
- Kwak, S.K.; Kim, J.H. Statistical data preparation: Management of missing values and outliers. Korean J. Anesthesiol. 2017, 70, 407–411. [Google Scholar] [CrossRef]
| Diets | Parameters | Late Gestation | Lactation |
|---|---|---|---|
| Mean ± SD a | Mean ± SD | ||
| Control (n = 15) | Body Weight b | 290 ± 40 | 251 ± 42 |
| ADFI c | 3.8 ± 0.2 | 6.6 ± 0.7 | |
| DON intake d | <0.8 | <1.3 | |
| DON dose e | <2.6 | <5.4 | |
| DON level 1 (n = 15) | Body Weight | 301 ± 34 | 258 ± 40 |
| ADFI | 3.4 ± 0.3 | 6.5 ± 1.2 | |
| DON intake | 4.7 ± 0.4 | 9.1 ± 1.7 | |
| DON dose | 15.7 ± 1.5 | 35.4 ± 3.3 | |
| DON level 2 (n = 14) | Body Weight | 303 ± 25 | 255 ± 34 |
| ADFI | 3.7 ± 0.3 | 6.1 ± 0.4 | |
| DON intake | 6.2 ± 0.5 | 10.3 ± 0.9 | |
| DON dose | 20.6 ± 1.9 | 40.9 ± 5.0 |
| Sampling Point | 10 days after Arrival | At Farrowing | Day 1 | Day 7 | Day 21 | Weaning | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diets | Con | Level 1 | Level 2 | Con | Level 1 | Level 2 | Con | Level 1 | Level 2 | Con | Level 1 | Level 2 | Con | Level 1 | Level 2 | Con | Level 1 | Level 2 |
| Sow plasma (ng/mL) | ||||||||||||||||||
| Mean ± SD | 0.22 ± 0.04 | 3.71 ± 1.81 | 4.90 ± 0.57 | 0.10 ± 0.04 | 1.79 ± 1.12 | 1.13 ± 0.90 | <LOD | 2.65 ± 0.70 | 0.81 ± 0.76 | 0.24 ± 0.05 | 4.22 ± 1.14 | 3.67 ± 0.37 | 0.46 ± 0.05 | 4.31 ± 2.65 | 5.70 ± 0.69 | 0.30 ± 0.11 | 9.06 ± 1.57 | 7.15 ± 1.99 |
| Median | 0.22 | 3.98 | 4.79 | 0.09 | 1.77 | 0.96 | <LOD | 2.88 | 0.53 | 0.23 | 3.96 | 3.56 | 0.46 | 4.52 | 6.02 | 0.27 | 9.42 | 7.27 |
| Range | 0.17–0.25 | 0.48–5.52 | 4.36–5.66 | <LOD–0.16 | 0.68–3.71 | 0.23–2.38 | <LOD | 1.67–3.50 | <LOD–1.69 | 0.18–0.30 | 3.03–6.06 | 3.20–4.16 | 0.41–0.53 | 0.52–7.82 | 4.80–6.44 | 0.20–0.45 | 7.2–11.4 | 5.0–10.1 |
| Piglet plasma (ng/mL) | ||||||||||||||||||
| Mean ± SD | 0.47 ± 0.28 | 2.97 ± 0.85 | 2.35 ± 0.65 | 0.14 ± 0.12 | 0.94 ± 1.11 | 1.62 ± 0.20 | <LOD | 0.26 ± 0.13 | 0.32 ± 0.18 | 0.12 ± 0.11 | 0.80 ± 0.43 | 0.58 ± 0.16 | 0.11 ± 0.09 | 1.69 ± 1.39 | 1.20 ± 1.41 | |||
| Median | 0.41 | 2.69 | 2.26 | <LOD | 0.73 | 1.61 | <LOD | 0.25 | 0.32 | <LOD | 0.71 | 0.51 | <LOD | 1.05 | 0.61 | |||
| Range | <LOD–1.61 | 1.74–4.74 | 1.34–3.43 | <LOD–0.37 | 0.49–6.66 | 1.20–2.08 | <LOD | <LOD–0.57 | <LOD–0.79 | <LOD–0.38 | 0.45–2.70 | 0.38–0.94 | <LOD–0.41 | 0.32–5.27 | 0.20–6.33 | |||
| Piglet/sow plasma ratio b | ||||||||||||||||||
| Mean ± SD | – | 2.05 ± 0.87 | 3.64 ± 3.25 | – | 0.30 ± 0.08 | 6.78 ± 8.41 | – | 0.06 ± 0.03 | 0.09 ± 0.03 | – | 0.20 ± 0.09 | 0.11 ± 0.04 | – | 0.21 ± 0.17 | 0.20 ± 0.19 | |||
| Parameters | n | Diets | ||
|---|---|---|---|---|
| Control a | DON Level 1 a | DON Level 2 a | ||
| Piglet body weight (kg): | ||||
| 48 ± 12 h after parturition | 71 | 2.2 ± 0.4 | 1.8 ± 0.3 | 1.8 ± 0.4 |
| Day 7 | 68 | 3.1 ± 0.8 | 2.7 ± 0.5 | 2.9 ± 0.8 |
| Day 21 | 67 | 7.4 ± 1.8 | 7.1 ± 1.2 | 7.2 ± 1.6 |
| Weaning | 66 | 11.9 ± 2.4 | 10.7 ± 1.6 | 11.0 ± 2.1 |
| Average weaning age (days) | 35 | 33 | 33 | |
| Total average daily gain (g/day) | 297 ± 67 | 290 ± 48 | 292 ± 59 | |
| (n = 18) | (n = 27) | (n = 21) | ||
| Sampling Point | 10 Days after Arrival | At Farrowing | Day 1 | Day 7 | Day 21 | Weaning |
|---|---|---|---|---|---|---|
| Sow plasma (ng/mL) | ||||||
| Mean ± SD | 4.25 ± 1.47 | 1.53 ± 1.04 | 1.73 ± 1.19 | 3.97 ± 0.88 | 4.94 ± 2.06 | 8.19 ± 1.96 |
| Median | 4.61 | 1.24 | 1.68 | 3.73 | 5.56 | 7.80 |
| Range | 0.48–5.66 | 0.23–3.71 | <LOD–3.50 | 3.03–6.06 | 0.52–7.82 | 5.0–11.4 |
| Piglet plasma (ng/mL) | ||||||
| Mean ± SD | 2.69 ± 0.81 | 1.25 ± 0.89 | 0.29 ± 0.16 | 0.70 ± 0.35 | 1.48 ± 1.41 | |
| Median | 2.55 | 1.11 | 0.30 | 0.64 | 0.86 | |
| Range | 1.34–4.74 | 0.49–6.66 | <LOD–0.79 | 0.38–2.70 | 0.20–6.33 | |
| Piglet/sow plasma ratio b | ||||||
| Mean ± SD | 2.14 ± 0.80 | 2.30 ± 2.81 | 0.08 ± 0.03 | 0.16 ± 0.08 | 0.20 ± 0.17 | |
| Sampling Point | At Farrowing | Day 1 | Day 7 | Day 21 | Weaning | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diets | Con | Level 1 | Level 2 | Con | Level 1 | Level 2 | Con | Level 1 | Level 2 | Con | Level 1 | Level 2 | Con | Level 1 | Level 2 |
| DON (ng/mL) | |||||||||||||||
| Mean ± SD | 0.42 ± 0.22 | 1.35 ± 0.52 | 1.20 ± 0.39 | 0.29 ± 0.09 | 2.14 ± 0.30 | 1.43 ± 1.49 | 0.48 ± 0.05 | 3.72 ± 1.98 | 4.14 ± 1.19 | 0.50 ± 0.00 | 5.67 ± 1.72 | 5.75 ± 1.52 | 0.73 ± 0.17 | 7.49 ± 1.99 | 7.03 ± 2.79 |
| Range | 0.25–0.75 | 0.82–2.29 | 0.81–1.65 | 0.23–0.43 | 1.68–2.44 | 0.46–4.06 | 0.43–0.53 | 1.33–5.97 | 3.08–6.20 | 0.50–0.50 | 2.97–7.09 | 4.61–8.29 | 0.57–0.97 | 5.4–10.5 | 4.1–10.4 |
| DOM-1 (ng/mL) | |||||||||||||||
| Mean ± SD | <LOD | <LOD | 0.15 ± 0.02 | <LOD | <LOD | <LOD | <LOD | <LOD | <LOD | <LOD | <LOD | 0.18 ± 0.05 | <LOD | 0.17 ± 0.04 | 0.22 ± 0.06 |
| Range | – | – | <LOD–0.19 | – | – | – | – | – | – | – | – | <LOD–0.24 | – | <LOD–0.23 | <LOD–0.30 |
| DON-15-GlcA (ng/mL) | |||||||||||||||
| Mean ± SD | <LOD | 3.18 ± 1.42 | 4.39 ± 0.77 | <LOD | 2.42 ± 1.57 | 2.17 ± 0.86 | <LOD | 1.89 ± 1.12 | 3.52 ± 3.95 | <LOD | 2.62 ± 1.04 | 7.00 ± 4.55 | 0.82 ± 0.27 | 6.37 ± 4.65 | 10.8 ± 1.9 |
| Range | – | 1.86–5.02 | 3.49–5.14 | – | 0.66–4.37 | 1.20–3.08 | – | <LOD–3.51 | 1.20–10.5 | – | 1.75–4.51 | 2.3–13.2 | 0.57–1.06 | 2.6–13.1 | 8.6–13.6 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sayyari, A.; Uhlig, S.; Fæste, C.K.; Framstad, T.; Sivertsen, T. Transfer of Deoxynivalenol (DON) through Placenta, Colostrum and Milk from Sows to Their Offspring during Late Gestation and Lactation. Toxins 2018, 10, 517. https://doi.org/10.3390/toxins10120517
Sayyari A, Uhlig S, Fæste CK, Framstad T, Sivertsen T. Transfer of Deoxynivalenol (DON) through Placenta, Colostrum and Milk from Sows to Their Offspring during Late Gestation and Lactation. Toxins. 2018; 10(12):517. https://doi.org/10.3390/toxins10120517
Chicago/Turabian StyleSayyari, Amin, Silvio Uhlig, Christiane Kruse Fæste, Tore Framstad, and Tore Sivertsen. 2018. "Transfer of Deoxynivalenol (DON) through Placenta, Colostrum and Milk from Sows to Their Offspring during Late Gestation and Lactation" Toxins 10, no. 12: 517. https://doi.org/10.3390/toxins10120517
APA StyleSayyari, A., Uhlig, S., Fæste, C. K., Framstad, T., & Sivertsen, T. (2018). Transfer of Deoxynivalenol (DON) through Placenta, Colostrum and Milk from Sows to Their Offspring during Late Gestation and Lactation. Toxins, 10(12), 517. https://doi.org/10.3390/toxins10120517








