Trichothecene Genotypes of Fusarium graminearum Populations Isolated from Winter Wheat Crops in Serbia
Abstract
:1. Introduction
2. Results
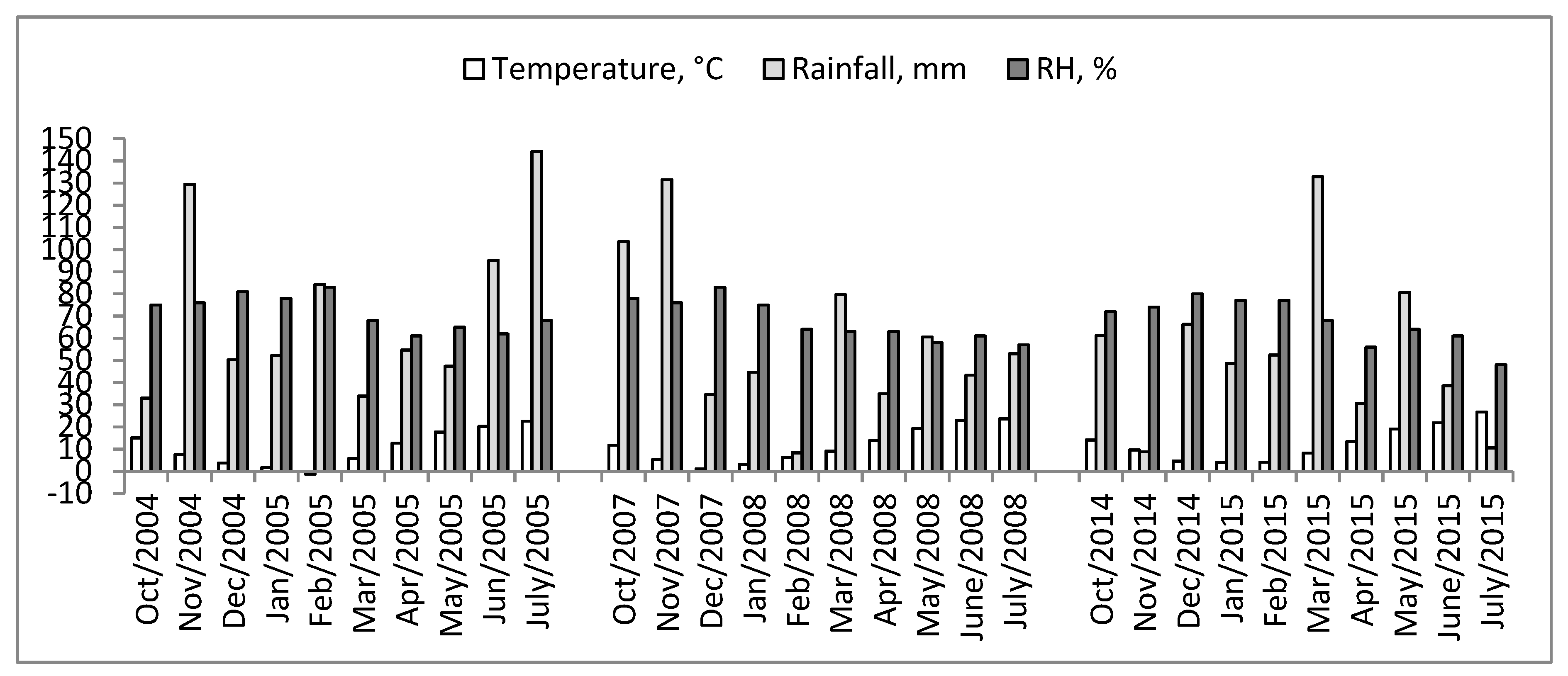
2.1. Climatic Conditions
2.2. Morphological and Molecular Identification of Species
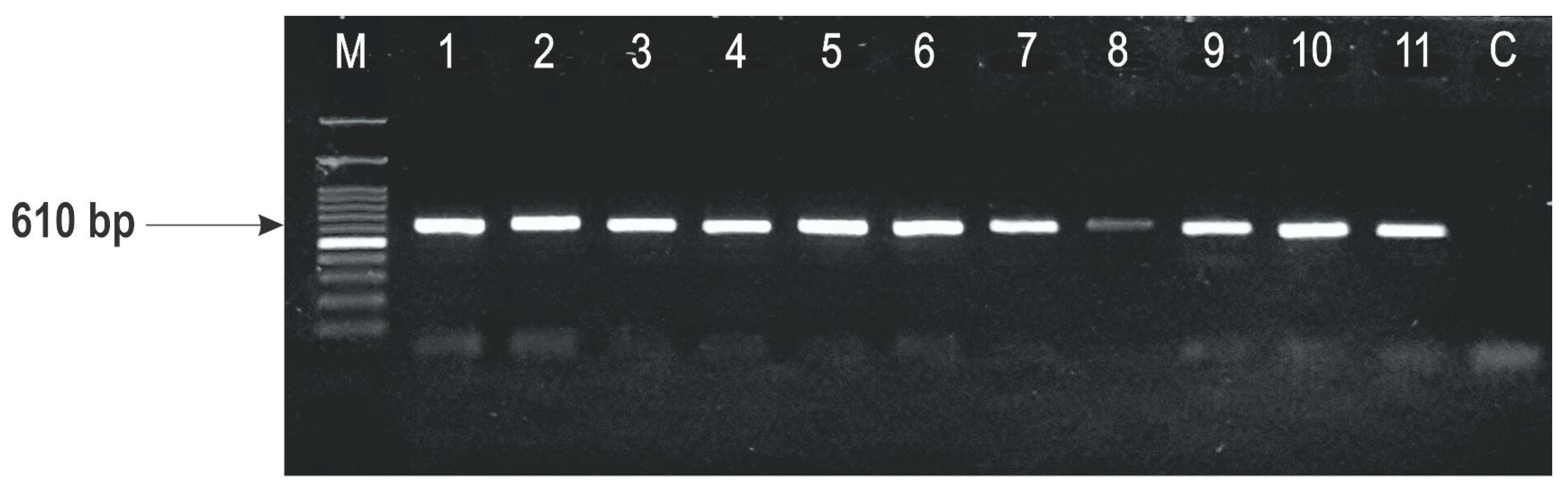
2.3. Identification of F. graminearum Genotypes
2.4. Deoxynivalenol Production
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Fungal Strains
5.2. DNA Extraction
5.3. Polymerase Chain Reaction
5.4. Deoxynivalenol Production
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Goswami, R.S.; Kistler, H.C. Heading for disaster: Fusarium graminearum on cereal crops. Mol. Plant Pathol. 2004, 5, 515–525. [Google Scholar] [CrossRef] [PubMed]
- Parry, D.W.; Jenkinson, P.; McLeod, L. Fusarium ear blight (scab) in small grain cereals—A review. Plant Pathol. 1995, 44, 207–238. [Google Scholar] [CrossRef]
- Lemmens, M.; Haim, K.; Lew, H.; Ruckenbauer, P. The effect of nitrogen fertilization on Fusarium head blight development and deoxynivalenol contamination in wheat. J. Phytopathol. 2004, 152, 1–8. [Google Scholar] [CrossRef]
- Stanković, S.Ž.; Lević, J.T.; Krnjaja, V.S.; Bočarov-Stančić, A.S.; Tančić, S.L.; Kovačević, T.M. Frequency of toxigenic Fusarium species and fusariotoxins in wheat grain in Serbia. Matica Srp. Proc. Nat. Sci. 2007, 113, 93–102. [Google Scholar] [CrossRef]
- Bottalico, A.; Perrone, G. Toxigenic Fusarium species and mycotoxins associated with head blight in small-grains cereals in Europe. Eur. J. Plant Pathol. 2002, 108, 611–624. [Google Scholar] [CrossRef]
- Lee, T.; Han, Y.K.; Kim, K.H.; Yun, S.H.; Lee, Y.W. Tri13 and Tri7 determine deoxynivalenol- and nivalenol-producing chemotypes of Gibberella zeae. Appl. Environ. Microbiol. 2002, 68, 2148–2154. [Google Scholar] [CrossRef] [PubMed]
- Wegulo, S.N. Factors influencing deoxynivalenol accumulation in small grain cereals. Toxins 2012, 4, 1157–1180. [Google Scholar] [CrossRef] [PubMed]
- Covarelli, L.; Beccari, G.; Prodi, A.; Generotti, S.; Etruschi, F.; Juan, C.; Ferrer, E.; Mañes, J. Fusarium species, chemotype characterisation and trichothecene contamination of durum and soft wheat in an area of central Italy. J. Sci. Food Agric. 2015, 95, 540–551. [Google Scholar] [CrossRef] [PubMed]
- Windels, C.E. Economic and social impacts of Fusarium head blight: Changing farms and rural communities in the Northern Great Plains. Phytopathology 2000, 90, 17–21. [Google Scholar] [CrossRef] [PubMed]
- Hooker, D.C.; Schaafsma, A.W.; Tamburic-Ilincic, L. Using weather variables pre- and postheading to predict deoxynivalenol content in winter wheat. Plant Dis. 2002, 86, 611–619. [Google Scholar] [CrossRef]
- De Wolf, E.D.; Madden, L.V.; Lipps, P.E. Risk assesment models for wheat Fusarium head blight epidemics based on within-season weather data. Phytopathology 2003, 93, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Scheider, N.; Guo, J.R.; Verreet, J.A.; Beyer, M. Assessing the intensity of Fusarium-damage in wheat: A comparison of selected disease parameters during disease development and the role of fungicides. J. Plant Dis. Protect. 2009, 116, 118–123. [Google Scholar] [CrossRef]
- Bai, G.; Shaner, G. Management and resistance in wheat and barley to Fusarium head blight. Annu. Rev. Phytopathol. 2004, 42, 135–161. [Google Scholar] [CrossRef] [PubMed]
- Miedaner, T.; Reinbrecht, C.; Lauber, U.; Schollenberger, M.; Geiger, H.H. Effects of genotype and genotype-environment interaction on deoxynivalenol accumulation and resistance to Fusarium head blight in rye, triticale, and wheat. Plant Breed. 2001, 120, 97–105. [Google Scholar] [CrossRef]
- Simpson, D.R.; Weston, G.E.; Turner, J.A.; Jennings, P.; Nicholson, P. Differential control of head blight pathogens of wheat by fungicides and consequences for mycotoxin contamination of grain. Eur. J. Plant Pathol. 2001, 107, 421–431. [Google Scholar] [CrossRef]
- Wegulo, S.N.; Baenziger, P.S.; Nopsa, J.H.; Bockus, W.W.; Hallen-Adams, H. Management of Fusarium head blight of wheat and barley. Crop Protect. 2015, 73, 100–107. [Google Scholar] [CrossRef]
- Righetti, L.; Galaverna, G.; Dall’Asta, C. Group detection of DON and its modified forms by an ELISA kit. Food Addit. Contam. Part A 2017, 2, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Maragos, C.M.; Plattner, R.D. Rapid fluorescence polarization immunoassay for the mycotoxin deoxynivalenol in wheat. J. Agric. Food Chem. 2002, 50, 1827–1832. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.L.; Cheng, Y.H. Identification and trichothecene genotypes of Fusarium graminearum species complex from wheat in Taiwan. J. Plant Dis. Protect. 2017, 58, 4. [Google Scholar] [CrossRef] [PubMed]
- Ji, L.; Cao, K.; Hu, T.; Wang, S. Determination of deoxynivalenol and nivalenol chemotypes of Fusarium graminearum isolates from China by PCR assay. J. Phytopathol. 2007, 155, 505–512. [Google Scholar] [CrossRef]
- Przemieniecki, S.W.; Kurowski, T.P.; Korzekwa, K. Chemotypes and geographic distribution of the Fusarium graminearum species complex. Environ. Biotechnol. 2014, 10, 45–59. [Google Scholar] [CrossRef]
- Krnjaja, V.; Tomić, Z.; Stanković, S.; Petrović, T.; Bijelić, Z.; Mandić, V.; Obradović, A. Fusarium infection and deoxynivalenol contamination in winter wheat. Biotechnol. Anim. Husb. 2015, 31, 123–131. [Google Scholar] [CrossRef]
- Krnjaja, V.; Lević, J.; Stanković, S. Pathogenic fungi on wheat grain in Serbia. J. Plant Pathol. 2008, 90, 84. [Google Scholar]
- Obradović, A.; Stanković, S.; Krnjaja, V.; Nikolić, A.; Ignjatović-Micić, D.; Stepanović, J.; Duduk, B. Trichothecene chemotype diversity of Fusarium graminearum isolated from wheat, maize and barley in Serbia. Genetika 2017, 49, 355–364. [Google Scholar] [CrossRef]
- Boutigny, A.L.; Ward, T.J.; Ballos, N.; Iancu, G.; Ioos, R. Diversity of the Fusarium graminearum species complex on French cereals. Eur. J. Plant Pathol. 2014, 138, 133–148. [Google Scholar] [CrossRef]
- Bozac, P.; Popescu, S.; Botau, D.; Boldura, O.M.; Pirvulescu, P. Molecular characterization for some new Fusarium isolates collected from the West Part of Romania. Rom. Biotechnol. Lett. 2016, 21, 11560–11568. [Google Scholar]
- Yörük, E.; Albayrak, G. Chemotyping of Fusarium graminearum and F. culmorum isolates from Turkey by PCR assay. Mycopathologia 2012, 173, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Szécsi, Á.; Bartók, T.; Varga, M.; Magya, D.; Mesterházy, Á. Determination of trichothecene chemotypes of Fusarium graminearum strains isolated in Hungary. J. Phytopathol. 2005, 153, 445–448. [Google Scholar] [CrossRef]
- Bilska, K.; Jurczak, S.; Kulik, T.; Ropelewska, E.; Olszewski, J.; Żelechowski, M.; Zapotoczny, P. Species composition and trichothecene genotype profiling of Fusarium field isolates recovered from wheat in Poland. Toxins 2018, 10, 325. [Google Scholar] [CrossRef] [PubMed]
- Prodi, A.; Tonti, S.; Nipoti, P.; Pancaldi, D.; Pisi, A. Identification of deoxynivalenol and nivalenol producing chemotypes of Fusarium graminearum isolates from durum wheat in a restricted area of Northern Italy. J. Plant Pathol. 2009, 91, 727–731. [Google Scholar]
- Talas, F.; Parzies, H.K.; Miedaner, T. Diversity in genetic structure and chemotype composition of Fusarium graminearum sensu stricto populations causing wheat head blight in individual fields in Germany. Eur. J. Plant Pathol. 2011, 131, 39–48. [Google Scholar] [CrossRef]
- Scoz, L.B.; Astolfi, P.; Reartes, D.S.; Schmale, D.G., III; Moraes, M.G.; Del Ponte, E.M. Trichothecene mycotoxin genotypes of Fusarium raminearum sensu stricto and Fusarium meridionale in wheat from southern Brazil. Plant Pathol. 2009, 58, 344–351. [Google Scholar] [CrossRef]
- Reynoso, M.M.; Ramirez, M.L.; Torres, A.M.; Chulze, S.N. Trichothecene genotypes and chemotypes in Fusarium graminearum strains isolated from wheat in Argentina. Int. J. Food Microbiol. 2011, 145, 444–448. [Google Scholar] [CrossRef] [PubMed]
- Boutigny, A.L.; Ward, T.J.; Van Coller, G.J.; Flett, B.; Lamprecht, S.C.; O’Donnell, K.; Viljoen, A. Analysis of the Fusarium graminearum species complex from wheat, barley and maize in South Africa provides evidence of species-specific differences in host preference. Fungal Genet. Biol. 2011, 48, 914–920. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.B.; Li, H.P.; Dang, F.J.; Qu, B.; Xu, Y.B.; Zhao, C.S.; Liao, Y.C. Determination of the trichothecene mycotoxin chemotypes and associated geographical distribution and phylogenetic species of the Fusarium graminearum clade from China. Mycol. Res. 2007, 111, 967–975. [Google Scholar] [CrossRef] [PubMed]
- Gale, L.R.; Ward, T.J.; Balmas, V.; Kistler, C. Population subdivision of Fusarium graminearum sensu stricto in the Upper Midwestern United States. Phytopathology 2007, 97, 1434–1439. [Google Scholar] [CrossRef] [PubMed]
- Gale, L.R.; Harrison, S.A.; Ward, T.J.; O’Donnell, K.; Milus, E.A.; Gale, S.W.; Kistler, H.C. Nivalenol-type populations of Fusarium graminearum and F. asiaticum are prevalent on wheat in southern Louisiana. Phytopathology 2011, 101, 124–134. [Google Scholar] [CrossRef] [PubMed]
- Van der Lee, T.; Zhang, H.; van Diepeningen, A.; Waalwijk, C. Biogeography of Fusarium graminearum species complex and chemotypes: A review. Food Addit. Contam. Part A 2015, 32, 453–460. [Google Scholar] [CrossRef] [PubMed]
- Ward, T.J.; Clear, R.M.; Rooney, A.P.; O’Donnell, K.; Gaba, D.; Patrick, S.; Starkey, D.E.; Gilbert, J.; Geiser, D.M.; Nowicki, T.W. An adaptive evolutionary shift in Fusarium head blight pathogen populations is driving the rapid spread of more toxigenic Fusarium graminearum in North America. Fungal Genet. Biol. 2008, 45, 473–484. [Google Scholar] [CrossRef] [PubMed]
- Beyer, M.; Pogoda, F.; Pallez, M.; Lazic, J.; Hoffmann, L.; Pasquali, M. Evidence for a reversible drought induced shift in the species composition of mycotoxin producing Fusarium head blight pathogens isolated from symptomatic wheat heads. Int. J. Food Microbiol. 2014, 182–183, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Stanković, S.; Tančić, S.; Lević, J.; Krnjaja, V. Production of deoxynivalenol by Fusarium graminearum and Fusarium culmorum isolated from wheat kernels in Serbia. Cereal Res. Commun. 2008, 36, 395–396. [Google Scholar]
- Schaarschmidt, S.; Fauhl-Hassek, C. The fate of mycotoxins during the processing of wheat for human consumption. Compr. Rev. Food Sci. Food Saf. 2018, 17, 556–593. [Google Scholar] [CrossRef]
- Tibola, C.S.; Fernandes, J.M.C.; Guarienti, E.M.; Nicolau, M. Distribution of Fusarium mycotoxins in wheat milling process. Food Control 2015, 53, 91–95. [Google Scholar] [CrossRef]
- Prodi, A.; Purahong, W.; Tonti, S.; Salomoni, D.; Nipoti, P.; Covarelli, L.; Pisi, A. Difference in chemotype composition of Fusarium graminearum populations isolated from durum wheat in adjacent areas separated by the Apennines in Northern-Central Italy. Plant Pathol. J. 2011, 27, 354–359. [Google Scholar] [CrossRef]
- Audenaert, K.; Van Broeck, R.; Bekaert, B.; De Witte, F.; Heremans, B.; Messens, K.; Höfte, M.; Haesaert, G. Fusarium head blight (FHB) in Flanders: Population diversity, inter-species associations and DON contamination in commercial winter wheat varieties. Eur. J. Plant Pathol. 2009, 125, 445–458. [Google Scholar] [CrossRef]
- Alvarez, C.L.; Azcarate, M.P.; Pinto, V.F. Toxigenic potential of Fusarium graminearum sensu stricto isolates from wheat in Argentina. Int. J. Food Microbiol. 2009, 135, 131–135. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, P.; Simpson, D.R.; Weston, G.; Rezanoor, H.N.; Lees, A.K.; Parry, D.W.; Joyce, D. Detection and quantification of Fusarium culmorum and Fusarium graminearum in cereals using PCR assays. Physiol. Mol. Plant Pathol. 1998, 53, 17–37. [Google Scholar] [CrossRef]
- Waalwijk, C.; Kastelein, P.; Vries, I.; Kerenyi, Z.; van der Lee, T.; Hesselink, T.; Kohl, J.; Kema, G. Major changes in Fusarium spp. in wheat in the Netherlands. Eur. J. Plant Pathol. 2003, 109, 743–754. [Google Scholar] [CrossRef]
- Ward, T.J.; Bielawski, J.P.; Kistler, H.C.; Sullivan, E.; O’Donnell, K. Ancestral polymorphism and adaptive evolution in the trichothecene mycotoxin gene cluster of phytopathogenic Fusarium. Proc. Natl. Acad. Sci. USA 2002, 99, 9278–9283. [Google Scholar] [CrossRef] [PubMed]
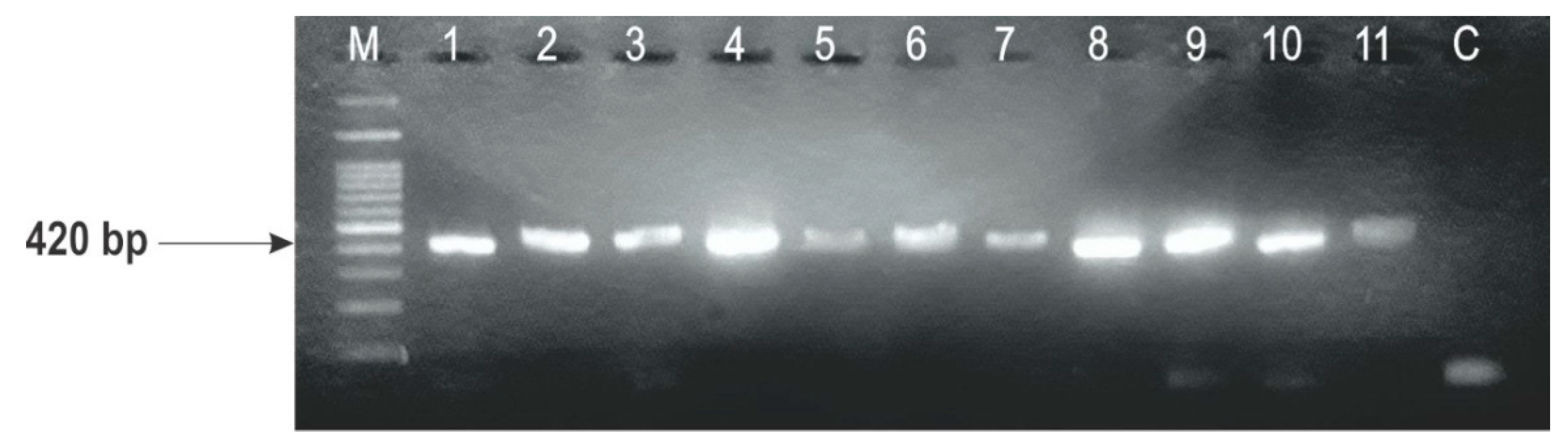
| Strain | Wheat Variety (Host) | Year of Isolation | F. graminearum Genotypes | |||
|---|---|---|---|---|---|---|
| TRI13 Primers | TRI3 Primers | |||||
| DON | NIV | 3-ADON | 15-ADON | |||
| IZS 146 | Pobeda | 2005 | + | − | − | + |
| IZS 147 | Pobeda | 2005 | + | − | − | + |
| IZS 148 | Pobeda | 2005 | + | − | − | + |
| IZS 149 | Pobeda | 2005 | + | − | − | + |
| IZS 150 | Pobeda | 2005 | + | − | − | + |
| IZS 151 | Pobeda | 2005 | + | − | − | + |
| IZS 152 | Pobeda | 2005 | + | − | − | + |
| IZS 153 | Pobeda | 2005 | + | − | − | + |
| IZS 243 | Pobeda | 2008 | + | − | − | + |
| IZS 244 | Pobeda | 2008 | + | − | − | + |
| IZS 250 | Pobeda | 2008 | + | − | − | + |
| IZS 251 | Pobeda | 2008 | + | − | − | + |
| IZS 252 | Pobeda | 2008 | + | − | − | + |
| IZS 254 | Pobeda | 2008 | + | − | − | + |
| IZS 255 | Pobeda | 2008 | + | − | − | + |
| IZS 256 | Pobeda | 2008 | + | − | − | + |
| IZS 257 | Pobeda | 2008 | + | − | − | + |
| IZS 258 | Pobeda | 2008 | + | − | − | + |
| IZS 362 | Simonida | 2015 | + | − | − | + |
| IZS 363 | Simonida | 2015 | + | − | − | + |
| IZS 364 | Simonida | 2015 | + | − | − | + |
| IZS 365 | Simonida | 2015 | + | − | − | + |
| IZS 366 | Simonida | 2015 | + | − | − | + |
| IZS 367 | NS40S | 2015 | + | − | − | + |
| IZS 368 | NS40S | 2015 | + | − | − | + |
| IZS 369 | NS40S | 2015 | + | − | − | + |
| IZS 370 | NS40S | 2015 | + | − | − | + |
| IZS 371 | NS40S | 2015 | + | − | − | + |
| IZS 372 | Simonida | 2015 | + | − | − | + |
| IZS 373 | NS40S | 2015 | + | − | − | + |
| IZS 374 | NS40S | 2015 | + | − | − | + |
| IZS 375 | NS40S | 2015 | + | − | − | + |
| IZS 376 | NS40S | 2015 | + | − | − | + |
| IZS 377 | Simonida | 2015 | + | − | − | + |
| IZS 378 | NS40S | 2015 | + | − | − | + |
| IZS 379 | NS40S | 2015 | + | − | − | + |
| IZS 380 | NS40S | 2015 | + | − | − | + |
| F. graminearum Strains | Wheat Variety | Year of Isolation | DON (µg kg−1) |
|---|---|---|---|
| IZS 146 | Pobeda | 2005 | >25,000 |
| IZS 147 | Pobeda | 2005 | >25,000 |
| IZS 148 | Pobeda | 2005 | >25,000 |
| IZS 149 | Pobeda | 2005 | 23,830 |
| IZS 150 | Pobeda | 2005 | 23,610 |
| IZS 151 | Pobeda | 2005 | 20,020 |
| IZS 243 | Pobeda | 2008 | >25,000 |
| IZS 244 | Pobeda | 2008 | 2061 |
| IZS 250 | Pobeda | 2008 | 21,460 |
| IZS 251 | Pobeda | 2008 | 20,260 |
| IZS 252 | Pobeda | 2008 | >25,000 |
| IZS 254 | Pobeda | 2008 | 6130 |
| IZS 362 | Simonida | 2015 | 9250 |
| IZS 363 | Simonida | 2015 | 3089 |
| IZS 364 | Simonida | 2015 | >25,000 |
| IZS 367 | NS40S | 2015 | 7500 |
| IZS 368 | NS40S | 2015 | 7420 |
| IZS 369 | NS40S | 2015 | 21,050 |
| Control | - | - | <40 |
| Gene | Primer | Sequence (5′–3′) | Size (bp) | |
|---|---|---|---|---|
| FG16 | FG16F/FG16R | CTCCGGATATGTTGCGTCAA GGTAGGTATCCGACATGGCAA | 400–500 | |
| TRI13 | TRI13F/TRI13R | TACGTGAAACATTGTTGGC GGTGTCCCAGGATCTGCG | 234 or 415 | |
| TRI3 | common | 3CON | TGGCAAAGACTGGTTCAC | - |
| specific | 3NA | GTGCACAGAATATACGAGC | TRI NIV 840 | |
| 3D15A | ACTGACCCAAGCTGCCATC | 15-ADON 610 | ||
| 3D3A | CGCATTGGCTAACACATG | 3-ADON 243 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krnjaja, V.; Stanković, S.; Obradović, A.; Petrović, T.; Mandić, V.; Bijelić, Z.; Božić, M. Trichothecene Genotypes of Fusarium graminearum Populations Isolated from Winter Wheat Crops in Serbia. Toxins 2018, 10, 460. https://doi.org/10.3390/toxins10110460
Krnjaja V, Stanković S, Obradović A, Petrović T, Mandić V, Bijelić Z, Božić M. Trichothecene Genotypes of Fusarium graminearum Populations Isolated from Winter Wheat Crops in Serbia. Toxins. 2018; 10(11):460. https://doi.org/10.3390/toxins10110460
Chicago/Turabian StyleKrnjaja, Vesna, Slavica Stanković, Ana Obradović, Tanja Petrović, Violeta Mandić, Zorica Bijelić, and Manja Božić. 2018. "Trichothecene Genotypes of Fusarium graminearum Populations Isolated from Winter Wheat Crops in Serbia" Toxins 10, no. 11: 460. https://doi.org/10.3390/toxins10110460
APA StyleKrnjaja, V., Stanković, S., Obradović, A., Petrović, T., Mandić, V., Bijelić, Z., & Božić, M. (2018). Trichothecene Genotypes of Fusarium graminearum Populations Isolated from Winter Wheat Crops in Serbia. Toxins, 10(11), 460. https://doi.org/10.3390/toxins10110460





