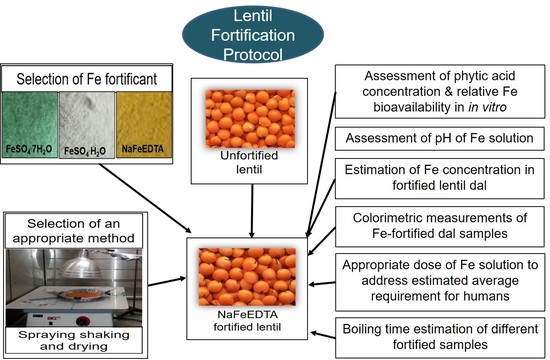
Iron Fortification of Lentil (Lens culinaris Medik.) to Address Iron Deficiency
Abstract
:1. Introduction
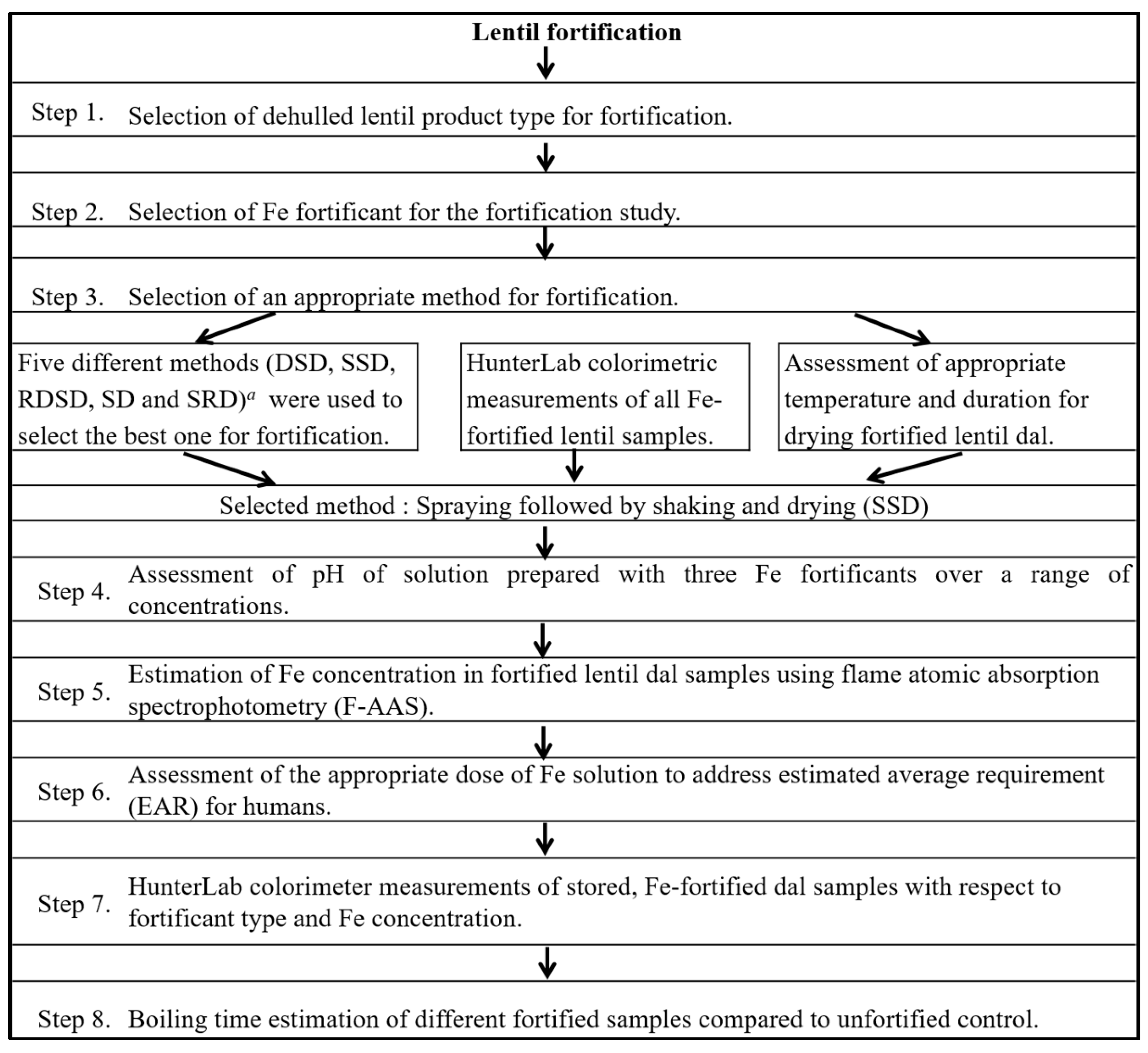
2. Materials and Methods
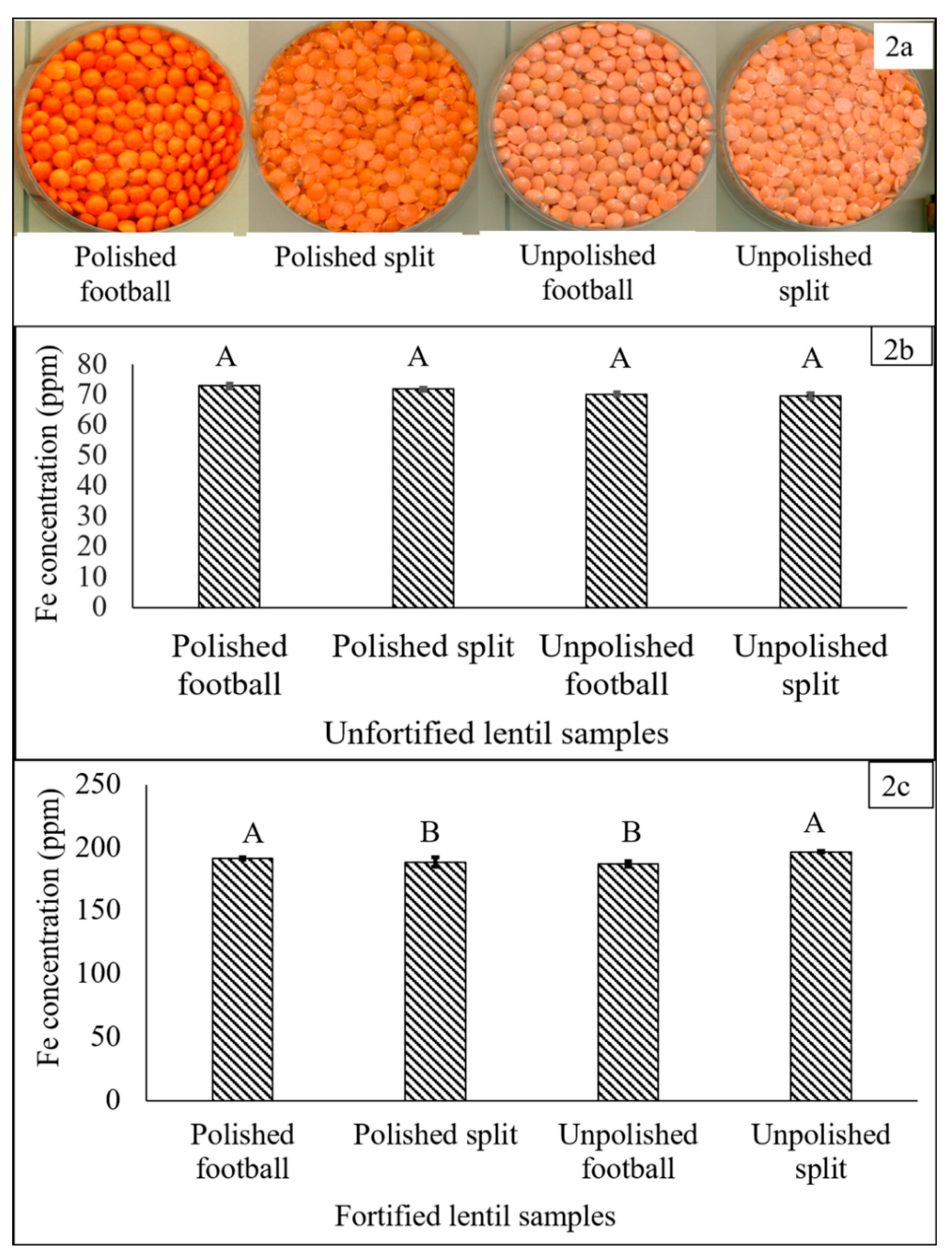
2.1. Selection of Lentil Genotype and Dehulled Lentil Product Type
2.2. Selection and Evaluation of the Most Suitable Fe Fortificant for Lentil
2.3. Selection of an Appropriate Method of Fortification
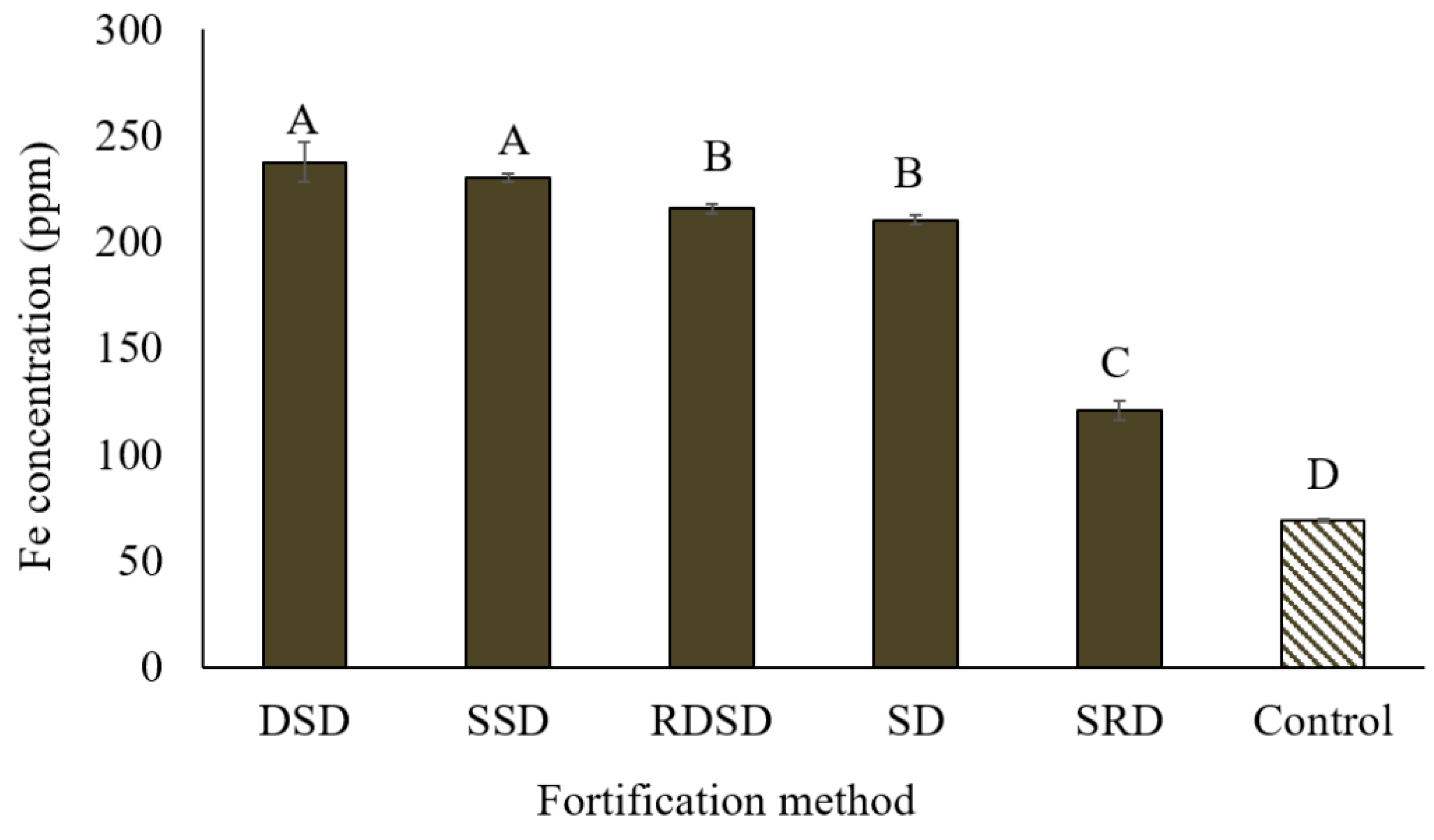
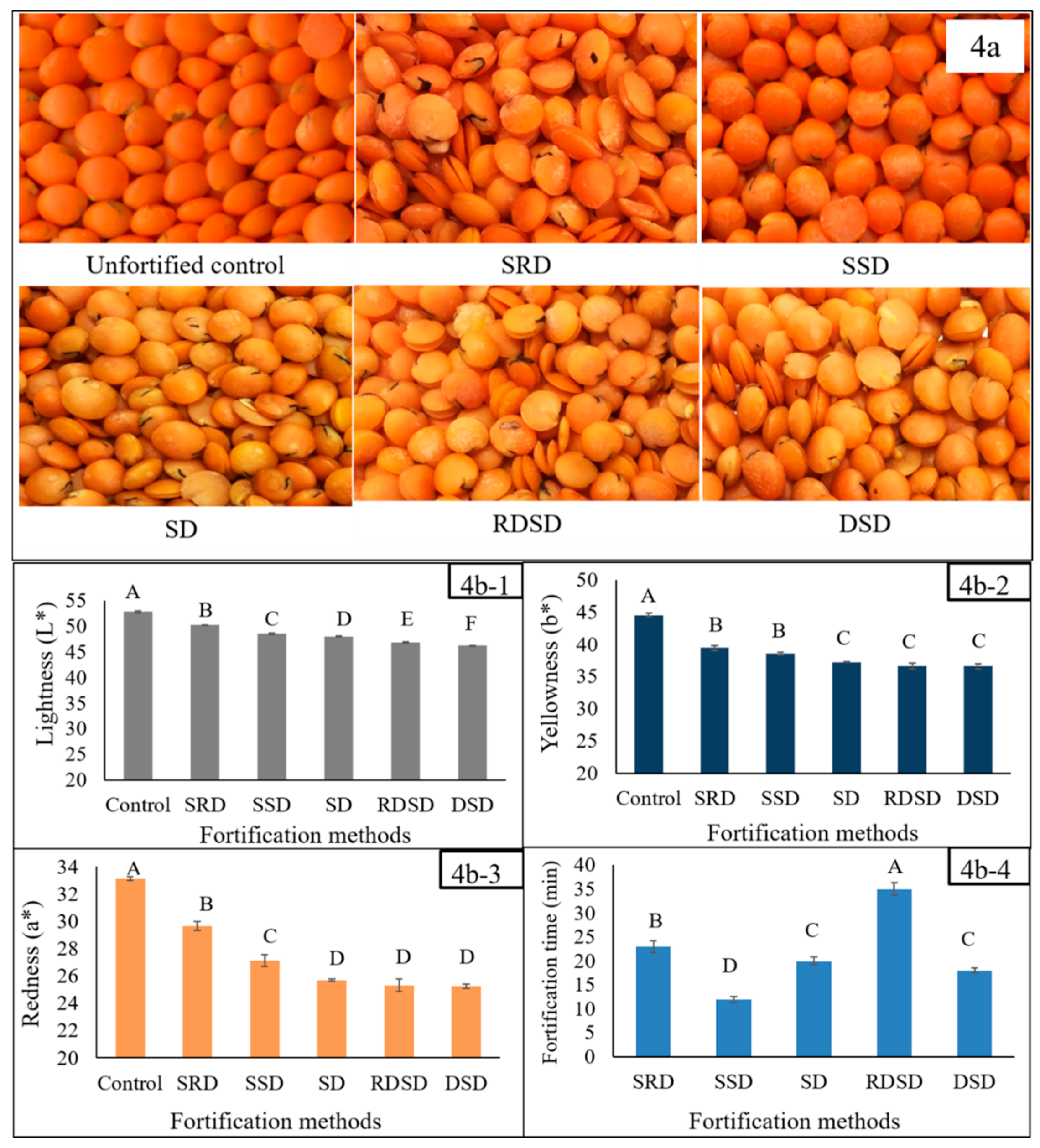
2.3.1. Techniques Used for Lentil Fortification
2.3.2. HunterLab Colorimetric Measurements of Fe-Fortified Lentil Samples
2.3.3. Assessment of Appropriate Temperature and Duration for Drying Fortified Lentil Dal
2.4. Estimation of Fe Concentration in Fortified Lentil Dal Samples by Flame Atomic Absorption Spectrophotometry (F-AAS)
2.5. Assessment of the Appropriate Dose of Fe Solution
2.6. HunterLab Colorimeter Measurements of Stored Fe-Fortified Dal Samples
2.7. Boiling Time Estimation of Fortified Lentil Dal Samples
2.8. Relative Fe Bioavailability and Phytic Acid Content of Fortified Lentils
3. Results and Discussion
3.1. Selection of Dehulled Lentil Product Type for Fortification
3.2. Selection and Evaluation of the Most Suitable Fe Fortificant for Lentil
3.3. Selection of Appropriate Methods for Fortification
3.3.1. Techniques Used for Lentil Fortification
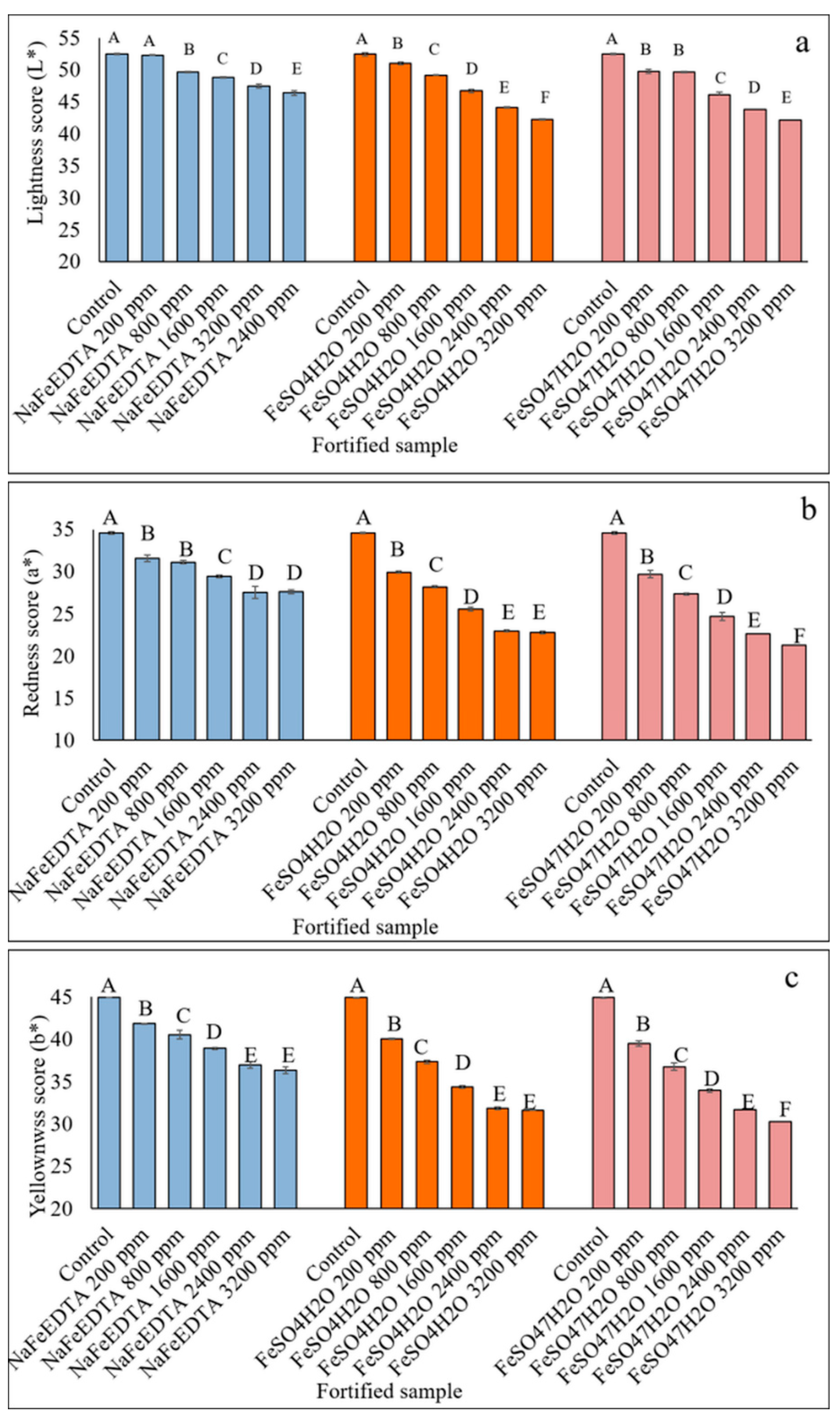
3.3.2. HunterLab Colorimetric Measurements of Fe-Fortified Lentil Samples
3.3.3. Assessment of Appropriate Temperature and Duration for Drying Fortified Lentil Dal
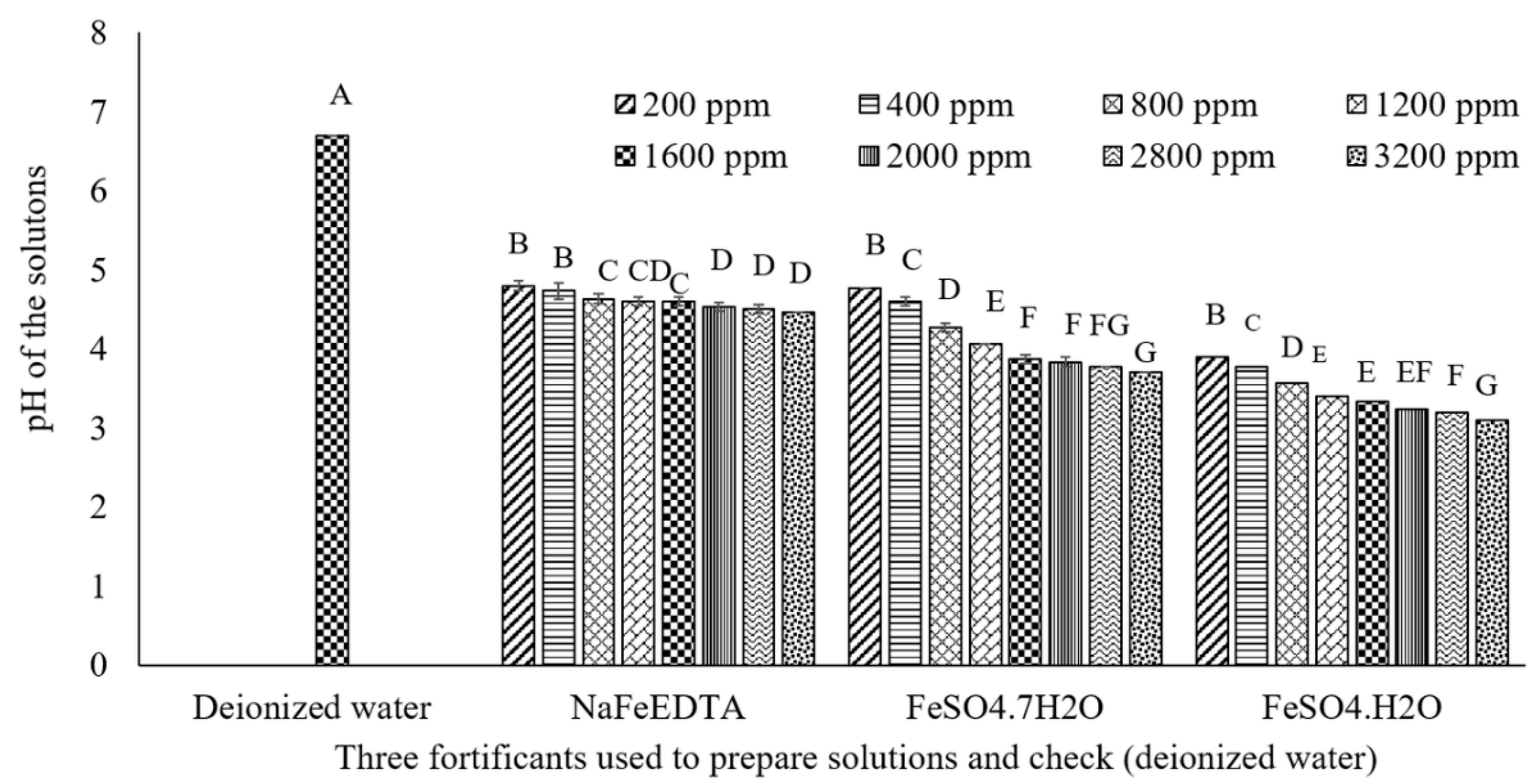
3.4. Assessment of the pH of Solutions Prepared with Three Fe Fortificants over a Range of Concentrations
3.5. Estimation of Fe Concentration in Fortified Lentil Dal Samples Using F-AAS
3.6. Assessment of the Appropriate Dose of Fe
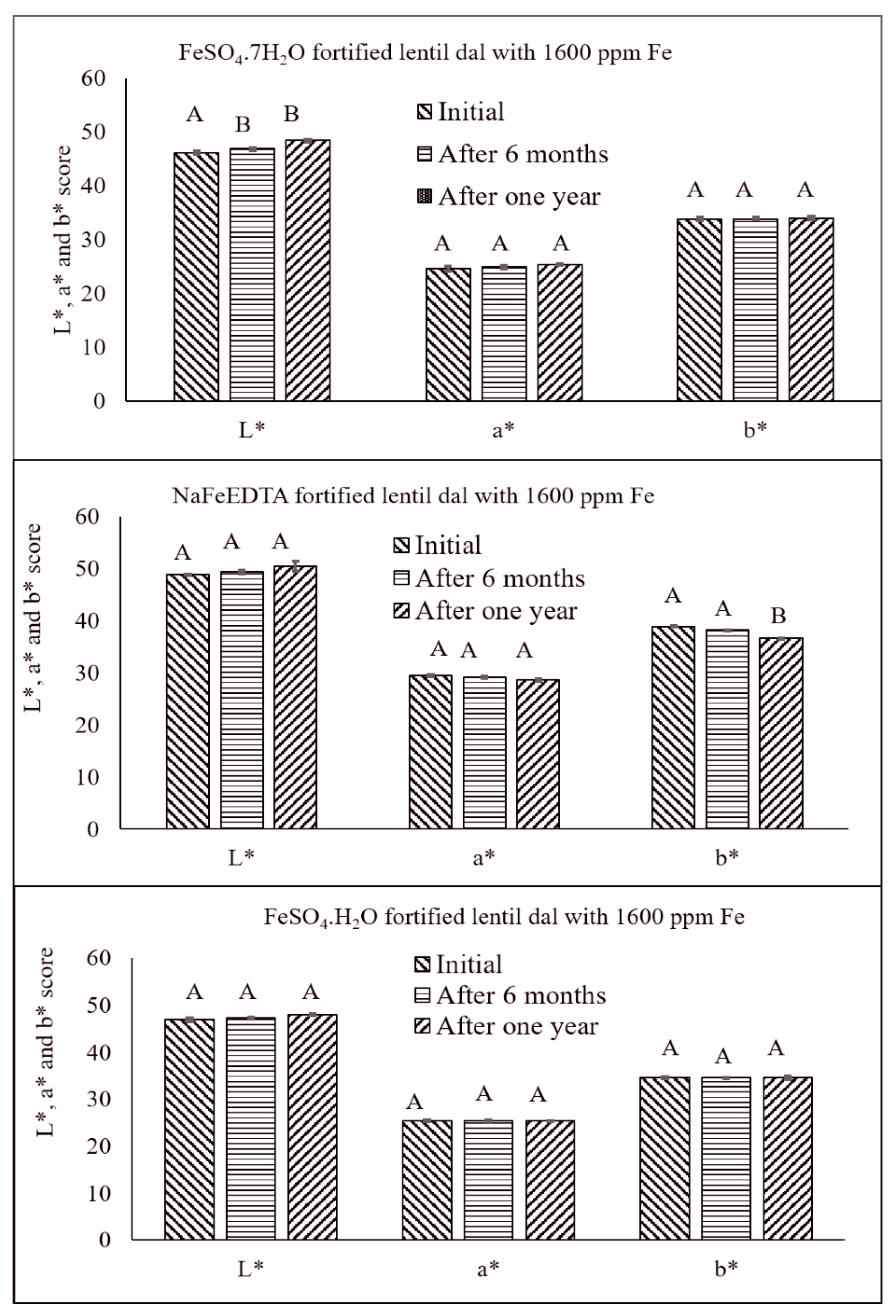
3.7. HunterLab Colorimeter Measurements of Stored Fe-Fortified Dal Samples
3.8. Boiling Time Estimation of Fortified Samples Compared to the Unfortified Control
3.9. Iron Concentration, Relative Fe Bioavailability and Phytic Acid Concentration of Fortified Lentils
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Podder, R.; Banniza, S.; Vandenberg, A. Screening of wild and cultivated lentil germplasm for resistance to stemphylium blight. Plant Genet. Resour. 2013, 11, 26–35. [Google Scholar] [CrossRef]
- Thavarajah, D.; Thavarajah, P.; Wejesuriya, A.; Rutzke, M.; Glahn, R.P.; Combs, G.F.; Vandenberg, A. The potential of lentil (Lens culinaris L.) as a whole food for increased selenium, iron, and zinc intake: Preliminary results from a 3 year study. Euphytica 2011, 180, 123–128. [Google Scholar] [CrossRef]
- Thavarajah, D.; Thavarajah, P.; Sarker, A.; Vandenberg, A. Lentils (Lens culinaris Medikus Subspecies culinaris): A Whole Food for Increased Iron and Zinc Intake. J. Agric. Food Chem. 2009, 57, 5413–5419. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Daun, J.K. Effects of variety and crude protein content on nutrients and anti-nutrients in lentils (Lens culinaris). Food Chem. 2006, 95, 493–502. [Google Scholar] [CrossRef]
- Hoover, R.; Hughes, T.; Chung, H.J.; Liu, Q. Composition, molecular structure, properties, and modification of pulse starches: A review. Food Res. Int. 2010, 43, 399–413. [Google Scholar] [CrossRef]
- Huang, J.; Schols, H.A.; van Soest, J.J.G.; Jin, Z.; Sulmann, E.; Voragen, A.G.J. Physicochemical properties and amylopectin chain profiles of cowpea, chickpea and yellow pea starches. Food Chem. 2007, 101, 1338–1345. [Google Scholar] [CrossRef]
- DellaValle, D.M.; Glahn, R.P.; Shaff, J.E.; O′Brien, K.O. Iron Absorption from an Intrinsically Labeled Lentil Meal Is Low but Upregulated in Women with Poor Iron Status. J. Nutr. 2015, 145, 2253–2257. [Google Scholar] [CrossRef] [PubMed]
- Lynch, S.R.; Beard, J.L.; Dassenko, S.A.; Cook, J.D. Iron absorption from legumes in humans. Am. J. Clin. Nutr. 1984, 40, 42–47. [Google Scholar] [PubMed]
- Li, S.; Zhou, X.; Huang, Y.; Zhu, L.; Zhang, S.; Zhao, Y.; Guo, J.; Chen, J.; Chen, R. Identification and characterization of the zinc-regulated transporters, iron-regulated transporter-like protein (ZIP) gene family in maize. BMC Plant Biol. 2013, 13, 114. [Google Scholar] [CrossRef] [PubMed]
- Bermejo, F.; Garcia-Lopez, S. A guide to diagnosis of iron deficiency and iron deficiency anemia in digestive diseases. World J. Gastroenterol. 2009, 15, 4638–4643. [Google Scholar] [CrossRef] [PubMed]
- Detzel, P.; Wieser, S. Food fortification for addressing iron deficiency in filipino children: Benefits and cost-effectiveness. Ann. Nutr. Metab. 2015, 66, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Boccio, J.R.; Iyengar, V. Iron deficiency: Causes, consequences, and strategies to overcome this nutritional problem. Biol. Trace Elem. Res. 2003, 94, 1–32. [Google Scholar] [CrossRef]
- Allen, L.; de Benoist, B.; Dary, O.; Hurrell, R. Guidelines on Food Fortification with Micronutrients. Available online: http://www.unscn.org/layout/modules/resources/files/fortification_eng.pdf (accessed on 21 April 2017).
- Northrop-Clewes, C.A. Food fortification. In Nutrition in Infancy; Humana Press: Totowa, NJ, USA, 2013; pp. 359–381. [Google Scholar]
- Dary, O. The importance and limitations of food fortification for the management of nutritional anemias. In The Guidebook Nutritional Anemia; Badham, J., Micheal, B., Zimmermann, K.K., Eds.; Sight and Life Press: Basel, Switzerland, 2007; pp. 315–336. ISBN 3-906412-35-0. [Google Scholar]
- Nilson, A.; Piza, J. Food fortification: A tool for fighting hidden hunger. Food Nutr. Bull. 1998, 19, 49–60. [Google Scholar] [CrossRef]
- Darnton-Hill, I.; Nalubola, R. Fortification strategies to meet micronutrient needs: Sucesses and failures. Proc. Nutr. Soc. 2002, 61, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Dwyer, J.T.; Wiemer, K.L.; Dary, O.; Keen, C.L.; King, J.C.; Miller, K.B.; Philbert, M.A.; Tarasuk, V.; Taylor, C.L.; Gaine, P.C.; et al. Fortification and Health: Challenges and Opportunities. Adv. Nutr. Int. Rev. J. 2015, 6, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Pachón, H.; Spohrer, R.; Mei, Z.; Serdula, M.K. Evidence of the effectiveness of flour fortification programs on iron status and anemia: A systematic review. Nutr. Rev. 2015, 73, 780–795. [Google Scholar] [CrossRef] [PubMed]
- Martı́nez-Navarrete, N.; Camacho, M.; Martı́nez-Lahuerta, J.; Martı́nez-Monzó, J.; Fito, P. Iron deficiency and iron fortified foods—A review. Food Res. Int. 2002, 35, 225–231. [Google Scholar] [CrossRef]
- DellaValle, D.M.; Thavarajah, D.; Thavarajah, P.; Vandenberg, A.; Glahn, R.P. Lentil (Lens culinaris L.) as a candidate crop for iron biofortification: Is there genetic potential for iron bioavailability? Field Crop. Res. 2013, 144, 119–125. [Google Scholar] [CrossRef]
- Wrolstad, R.E.; Smith, D.E. Color analysis. In Food Analysis; Nielson, S.S., Ed.; Springer International Publishing: Cham, Switzerland, 2010; pp. 545–555. ISBN 978-1-4419-1477-4. [Google Scholar]
- Diapari, M.; Sindhu, A.; Bett, K.; Deokar, A.; Warkentin, T.D.; Tar′an, B.; Francki, M. Genetic diversity and association mapping of iron and zinc concentrations in chickpea ( Cicer arietinum L.). Genome 2014, 57, 459–468. [Google Scholar] [CrossRef] [PubMed]
- Kohinoor, H.; Siddiqua, A.; Akhtar, S.; Hossain, M.G.; Podder, R.; Hossain, M.A. Nutrition and Easy Cooking of Pulses; Bangladesh Agricultural Research Institute Press: Gazipur, Bangladesh; Print Valley Printing Press: Gazipur, Bangladesh, 2010. [Google Scholar]
- DellaValle, D.M.; Glahn, R.P. Differences in relative iron bioavailability in traditional Bangladeshi meal plans. Food Nutr. Bull. 2014, 35, 431–439. [Google Scholar] [CrossRef] [PubMed]
- Glahn, R. Use of Caco-2 cells in defining nutrient bioavailability: Application to iron bioavailability of foods. In Designing Functional Foods: Measuring and Controlling Food Structure Breakdown and Nutrient Absorption; McClements, D.J., Eric, A.D., Eds.; Woodhead Publishing Limited Press: Cambridge, UK; CRC Press: Boca Raton, FL, USA, 2009; pp. 340–361. ISBN 9781845696603. [Google Scholar]
- Mckie, V.A.; Mccleary, B.V. A novel and rapid colorimetric method for measuring total phosphorus and phytic acid in foods and animal feeds. J. AOAC Int. 2016, 99, 738–743. [Google Scholar] [CrossRef] [PubMed]
- Erskine, W. Origin, phylogeny, domestication and spread. In The Lentil: Botany, Production and Uses; Erskine, W., Muehlbauer, J.F., Sarker, A., Sharma, B., Eds.; CAB International: Oxfordshire, UK, 2009; pp. 13–33. ISBN 9781845934873. [Google Scholar]
- Mellican, R.I.; Li, J.; Mehansho, H.; Nielsen, S.S. The role of iron and the factors affecting off-color development of polyphenols. J. Agric. Food Chem. 2003, 51, 2304–2316. [Google Scholar] [CrossRef] [PubMed]
- Hurrell, R.F. Preventing iron deficiency through food fortification. Nutr. Rev. 1997, 55, 210–222. [Google Scholar] [CrossRef] [PubMed]
- Hurrell, R.F.; Cook, J.D. Strategies for iron fortification of foods. Trends Food Sci. Technol. 1990, 1, 56–61. [Google Scholar] [CrossRef]
- Hurrell, R.F.; Reddy, M.B.; Burri, J.; Cook, J.D. An evaluation of EDTA compounds for iron fortification of cereal-based foods. Br. J. Nutr. 2000, 84, 903–910. [Google Scholar] [PubMed]
- Van Thuy, P.; Berger, J.; Davidsson, L.; Khan, N.C.; Lam, N.T.; Cook, J.D.; Hurrell, R.F.; Khoi, H.H. Regular consumption of NaFeEDTA-fortified fish sauce improves iron status and reduces the prevalence of anemia in anemic Vietnamese women. Am. J. Clin. Nutr. 2003, 78, 284–290. [Google Scholar]
- Amalrajan, V.; Thankachan, P.; Selvam, S.; Kurpad, A. Effect of wheat flour fortified with sodium iron EDTA on urinary zinc excretion in school-aged children. Food Nutr. Bull. 2012, 33, 177–179. [Google Scholar] [CrossRef] [PubMed]
- Davidsson, L.; Kastenmayer, P.; Hurrell, R.F. Sodium iron EDTA [NaFe(III)EDTA] as a food fortificant: The effect on the absorption and retention of zinc and calcium in women. Am. J. Clin. Nutr. 1994, 60, 231–237. [Google Scholar] [PubMed]
- Davidsson, L.; Almgren, A.; Hurrell, R.F. Sodium iron EDTA [NaFe(III)EDTA] as a food fortificant does not influence absorption and urinary excretion of manganese in healthy adults. J. Nutr. 1998, 128, 1139–1143. [Google Scholar] [PubMed]
- Longfils, P.; Monchy, D.; Weinheimer, H.; Chavasit, V.; Nakanishi, Y.; Schümann, K.A. Comparative intervention trial on fish sauce fortified with NaFe-EDTA and FeSO4+ citrate in iron deficiency anemic school children in Kampot, Cambodia. Asia Pac. J. Clin. Nutr. 2008, 17, 250–257. [Google Scholar] [PubMed]
- Prentice, A.M.; Mendoza, Y.A.; Pereira, D.; Cerami, C.; Wegmuller, R.; Constable, A.; Spieldenner, J. Dietary strategies for improving iron status: Balancing safety and efficacy. Nutr. Rev. 2017, 75, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Eichler, K.; Wieser, S.; Rüthemann, I.; Brügger, U. Effects of micronutrient fortified milk and cereal food for infants and children: A systematic review. BMC Public Health 2012, 12, 506. [Google Scholar] [CrossRef] [PubMed]
- FAO; WHO. Evaluation of Certain Food Additives and Contaminants. Available online: http://apps.who.int/iris/bitstream/10665/43870/1/9789241209472_eng.pdf (accessed on 12 June 2017).
- Hayma, J. The Storage of Tropical Agricultural Products; Van Otterloo-Butler, S., Ed.; Agromisa: Wageningen, The Netherlands, 2003; ISBN 9077073604. [Google Scholar]
- McVicar, R.; McCall, P.; Brenzil, C.; Hartley, S.; Panchuk, K.; Mooleki, P. Lentils in Saskatchewan. Available online: http://publications.gov.sk.ca/documents/20/86381-Lentils in Saskatchewan.pdf (accessed on 12 June 2017).
- Oghbaei, M.; Prakash, J. Effect of primary processing of cereals and legumes on its nutritional quality: A comprehensive review. Cogent Food Agric. 2016, 2. [Google Scholar] [CrossRef]
- Mehansho, H. Iron fortification technology development: New approaches. J. Nutr. 2006, 136, 1059–1063. [Google Scholar] [PubMed]
- García-Casal, M.N.; Layrisse, M. The effect of change in pH on the solubility of iron bis-glycinate chelate and other iron compounds. Arch. Latinoam. Nutr. 2001, 51, 35–36. [Google Scholar] [PubMed]
- Hurrell, R.; Egli, I. Iron bioavailability and dietary reference values. Am. J. Clin. Nutr. 2010, 91, 1461S–1467S. [Google Scholar] [CrossRef] [PubMed]
- Marie Minihane, A.; Rimbach, G. Iron absorption and the iron binding and anti-oxidant properties of phytic acid. Int. J. Food Sci. Technol. 2002, 37, 741–748. [Google Scholar] [CrossRef]
- Zhang, B.; Deng, Z.; Tang, Y.; Chen, P.; Liu, R.; Ramdath, D.D.; Liu, Q.; Hernandez, M.; Tsao, R. Fatty acid, carotenoid and tocopherol compositions of 20 Canadian lentil cultivars and synergistic contribution to antioxidant activities. Food Chem. 2014, 161, 296–304. [Google Scholar] [CrossRef] [PubMed]
| Fe Concentration in Fortificant Solution (ppm) | Fe Concentration in Fortified Lentil Dal | ||
|---|---|---|---|
| FeSO4·7H2O | NaFeEDTA | FeSO4·H2O | |
| Control | 69.0 ± 0.9 a | 69.0 ± 0.9 a | 65.6 ± 0.8 a |
| 100 | 76.0 ± 1.9 a | 83.7 ± 2.5 a | 71.8 ± 0.7 b |
| 400 | 132.5 ± 3.2 b | 113.2 ± 4.2 b | 108.6 ± 1.1 c |
| 800 | 147.9 ± 4.7 c | 182.9 ± 5.8 c | 151.4 ± 2.8 d |
| 1200 | 157.8 ± 4.3 c | 185.3 ± 5.6 c | 185.0 ± 6.6 e |
| 1600 | 203.6 ± 3.9 d | 205.3 ± 2.8 d | 207.5 ± 3.9 f |
| 2000 | 217.5 ± 8.2 d | 274.7 ± 5.6 e | 261.8 ± 3.9 g |
| 2400 | 246.6 ± 9.3 e | 309.7 ± 10.0 f | 322.3 ± 3.7 h |
| 2800 | 286.7 ± 6.0 f | 346.7 ± 5.2 g | 363.5 ± 6.2 i |
| 3200 | 349.0 ± 1.8 g | 326 ± 3.1 h | 381.7 ± 3.6 j |
| Fe Concentration (ppm) | Lightness (L*) | Redness (a*) | Yellowness (b*) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Initial | After 6 Months | After One Year | Initial | After 6 Months | After One Year | Initial | After 6 Months | After One Year | |
| FeSO4·7H2O fortified samples | |||||||||
| Control | 50.6 ± 0.4 a | 50.8 ± 0.2 a | 51.0 ± 0.2 a | 31.5 ± 0.2 a | 31.3 ± 0.2 a | 30.6 ± 0.6 a | 41.6 ± 1.0 a | 41.2 ± 1.0 a | 40.3 ± 1.0 a |
| 200 | 49.9 ± 0.6 ab | 50.6 ± 0.6 a | 52.0 ± 0.5 b | 29.7 ± 0.8 b | 29.4 ± 0.8 b | 28.8 ± 0.8 b | 40.5 ± 0.1 b | 38.9 ± 0.1 b | 37.9 ± 0.1 b |
| 800 | 49.6 ± 0.2 b | 50.3 ± 0.1 a | 51.5 ± 0.0 b | 27.4 ± 0.3 c | 26.8 ± 0.2 c | 25.8 ± 0.3 ac | 37.8 ± 0.3 c | 36.4 ± 0.3 c | 34.6 ± 0.3 c |
| 1600 | 46.2 ± 0.5 c | 46.9 ± 0.5 b | 48.5 ± 0.4 c | 24.6 ± 0.7 d | 24.9 ± 0.6 d | 25.5 ± 1.2 c | 36.4 ± 0.1 d | 33.9 ± 0.1 d | 34.0 ± 0.1 c |
| 2400 | 43.9 ± 0.2 d | 44.5 ± 0.1 c | 45.8 ± 0.2 c | 22.6 ± 0.2 e | 22.2 ± 0.1 e | 21.3 ± 0.2 d | 32.0 ± 0.3 e | 31.2 ± 0.1 e | 30.0 ± 0.4 d |
| 3200 | 42.1 ±0.6 e | 42.7 ± 0.6 d | 43.9 ± 0.6 d | 21.3 ± 0.8 f | 34.4 ± 0.9 f | 20.3 ± 1.2 d | 30.0 ± 0.2 f | 29.7 ± 0.7 f | 28.6 ± 0.3 e |
| NaFeEDTA fortified samples | |||||||||
| Control | 50.5 ± 0.4 a | 50.8 ± 0.2 a | 50.8 ± 0.2 a | 31.5 ± 0.2 a | 31.3 ± 0.3 a | 30.6 ± 0.6 a | 41.6 ± 0.3 a | 41.2 ± 0.1 a | 40.3 ± 0.7 a |
| 200 | 50.4 ± 0.1 a | 51.0 ± 0.2 a | 51.0 ± 0.2 a | 31.6 ± 0.7 a | 31.1 ± 0.8 a | 30.3 ± 0.8 a | 41.9 ± 0.1 a | 41.5 ± 0.1 a | 40.6 ± 0.3 a |
| 800 | 50.1 ± 0.2 a | 50.6 ± 0.6 a | 50.6 ± 0.6 b | 31.1 ± 0.3 a | 30.5 ± 0.2 a | 29.0 ± 0.5 b | 40.6 ± 0.9 b | 39.3 ± 0.4 a | 36.9 ± 0.8 b |
| 1600 | 48.8 ± 0.1 b | 52.0 ± 0.5 b | 52.0 ± 0.5 b | 29.4 ± 0.3 b | 29.1 ± 0.2 b | 28.6 ± 0.4 b | 38.9 ± 0.2 c | 38.2 ± 0.2 b | 36.6 ± 0.5 b |
| 2400 | 47.5 ± 0.2 c | 50.3 ± 0.1 c | 50.3 ± 0.1 c | 27.5 ± 1.3 c | 27.0 ± 1.2 c | 26.1 ± 1.1 c | 36.3 ± 0.7 d | 35.8 ± 0.6 c | 34.6 ± 0.6 c |
| 3200 | 46.4 ± 0.5 d | 51.5 ± 0.0 d | 51.5 ± 0.0 c | 27.8 ± 0.4 c | 27.4 ± 0.4 c | 26.5 ± 0.4 c | 36.9 ± 0.7 d | 36.4 ± 0.8 c | 35.2 ± 0.9 c |
| FeSO4·H2O fortified samples | |||||||||
| Control | 50.5 ± 0.4 a | 50.5 ± 0.4 a | 50.8 ± 0.2 a | 51.2 ± 0.3 a | 31.5 ± 0.2 a | 31.3 ± 0.2 a | 30.6 ± 0.6 a | 41.6 ± 0.3 a | 41.2 ± 0.1 a |
| 200 | 51.1 ± 0.5 a | 51.1 ± 0.5 a | 51.3 ± 0.3 b | 51.7 ± 0.3 b | 30.0 ± 0.7 b | 29.9 ± 0.7 a | 29.8 ± 0.7 b | 39.9 ± 0.1 b | 39.6 ± 0.1 b |
| 800 | 49.3±0.7 b | 49.7 ± 0.7 b | 50.4 ± 0.5 b | 27.9 ± 0.3 c | 27.6 ± 0.4 c | 27.1 ± 0.4 a | 37.3 ± 0.9 c | 36.9 ± 0.4 c | 36.5 ± 0.8 c |
| 1600 | 46.9 ± 0.7 c | 47.3 ± 0.4 c | 48.1 ± 0.2 c | 25.4 ± 0.3 d | 25.4 ± 0.3 d | 25.4 ± 0.4 c | 34.6 ± 0.2 d | 34.6 ± 0.2 d | 34.6 ± 0.5 d |
| 2400 | 44.4 ± 0.6 d | 44.7 ± 0.4 d | 45.4 ± 0.4 d | 23.3 ± 0.7 e | 22.8 ± 0.7 e | 21.9 ± 0.9 d | 32.2 ± 0.7 e | 31.9 ± 0.6 e | 30.2 ± 0.6 e |
| 3200 | 42.6 ± 0.3 e | 42.6 ± 0.3 e | 42.7 ± 0.5 e | 22.7 ± 0.7 e | 22.1 ± 0.7 e | 21.1 ± 0.8 d | 31.5 ± 0.7 e | 30.9 ± 0.8 f | 29.8 ± 0.9 f |
| Cooked Lentil Sample | Fe Concentration (ppm) a | Ferritin Formation (ng Ferritin (mg Protein)−1) a | Relative Fe Bioavailability (% Control Lentil) a | Phytic Acid (mg g−1) a |
|---|---|---|---|---|
| Unfortified dehulled lentil | 68.7 ± 0.3 a | 12.7 ± 1.0 a | 68.3 ± 14.8 a | 8.0 ± 0.1 a |
| NaFeEDTA fortified (1600 ppm Fe) | 230.8 ± 8.5 b | 17.4 ± 2.7 b | 100.5 ± 7.5 b | 8.0 ± 0.2 a |
| FeSO4·H2O fortified (1600 ppm Fe) | 220.5 ± 2.1 c | 17.6 ± 2.2 b | 104.9 ± 16.7 b | 7.2 ± 0.1 c |
| FeSO4·7H2O fortified (1600 ppm Fe) | 238.5 ± 4.7 b | 21.2 ± 1.9 b | 103.4 ± 10.4 b | 7.4 ± 0.1 b |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Podder, R.; Tar’an, B.; Tyler, R.T.; Henry, C.J.; DellaValle, D.M.; Vandenberg, A. Iron Fortification of Lentil (Lens culinaris Medik.) to Address Iron Deficiency. Nutrients 2017, 9, 863. https://doi.org/10.3390/nu9080863
Podder R, Tar’an B, Tyler RT, Henry CJ, DellaValle DM, Vandenberg A. Iron Fortification of Lentil (Lens culinaris Medik.) to Address Iron Deficiency. Nutrients. 2017; 9(8):863. https://doi.org/10.3390/nu9080863
Chicago/Turabian StylePodder, Rajib, Bunyamin Tar’an, Robert T. Tyler, Carol J. Henry, Diane M. DellaValle, and Albert Vandenberg. 2017. "Iron Fortification of Lentil (Lens culinaris Medik.) to Address Iron Deficiency" Nutrients 9, no. 8: 863. https://doi.org/10.3390/nu9080863
APA StylePodder, R., Tar’an, B., Tyler, R. T., Henry, C. J., DellaValle, D. M., & Vandenberg, A. (2017). Iron Fortification of Lentil (Lens culinaris Medik.) to Address Iron Deficiency. Nutrients, 9(8), 863. https://doi.org/10.3390/nu9080863