Consuming Almonds vs. Isoenergetic Baked Food Does Not Differentially Influence Postprandial Appetite or Neural Reward Responses to Visual Food Stimuli
Abstract
1. Introduction
2. Materials and Methods
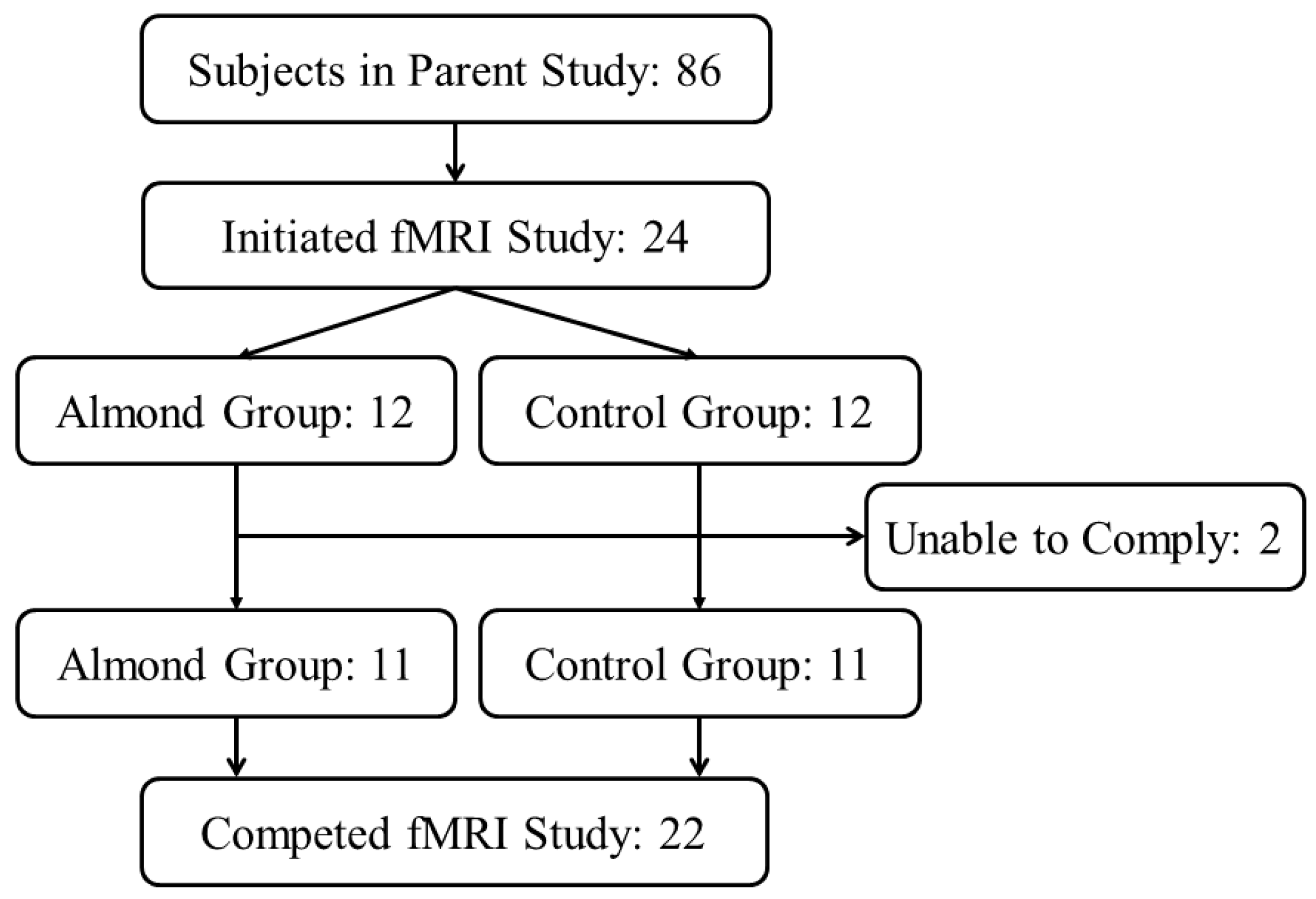
2.1. Subjects
2.2. Experimental Design of Parent Weight Loss Intervention
2.3. Experimental Design of the fMRI Study
2.4. Body Mass and Composition
2.5. Appetite Assessments
2.6. fMRI Data Acquisition, Preprocessing, and Analysis
2.7. Statistical Analysis
3. Results
3.1. Participant Characteristics
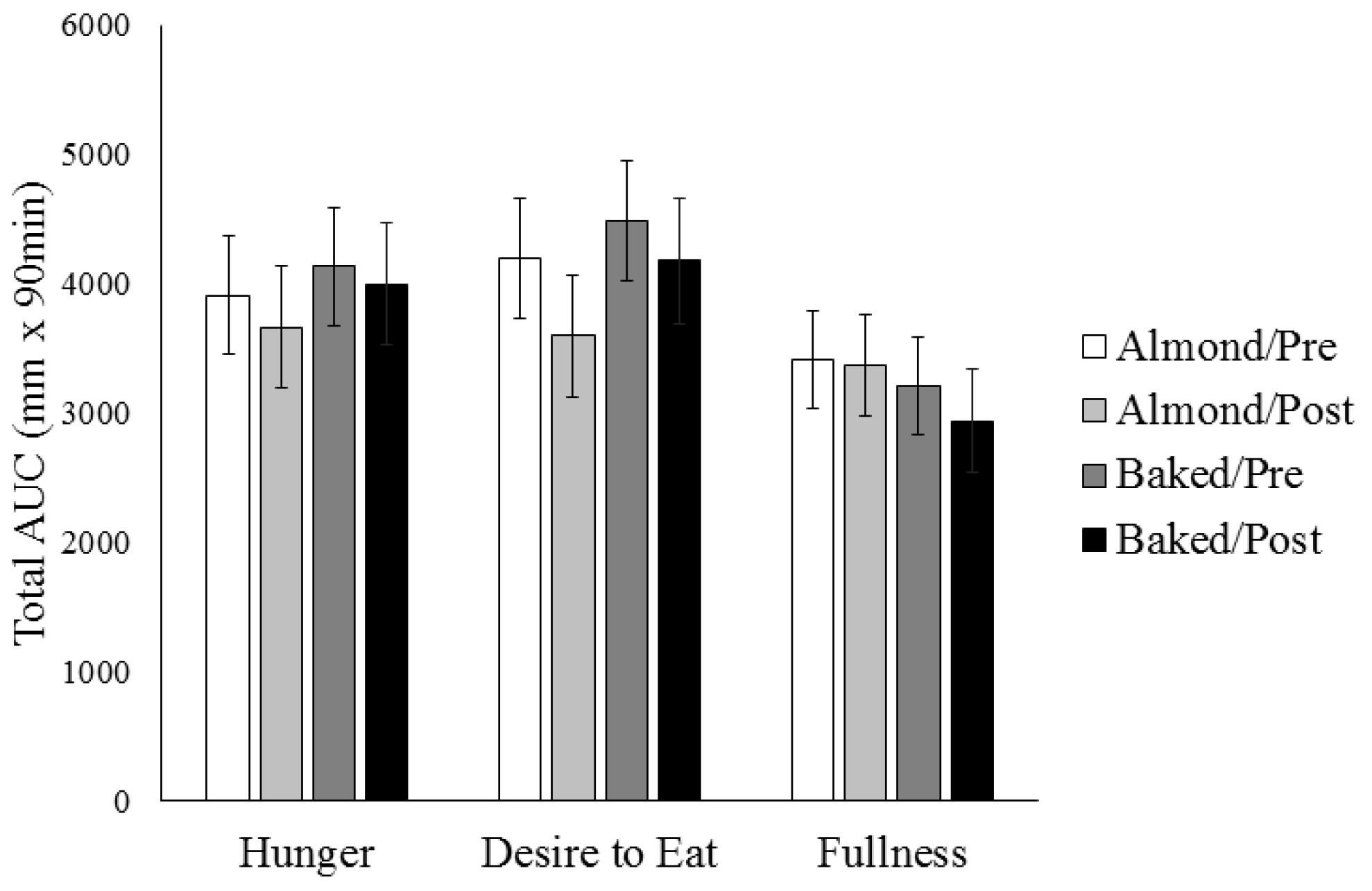
3.2. Appetite Ratings
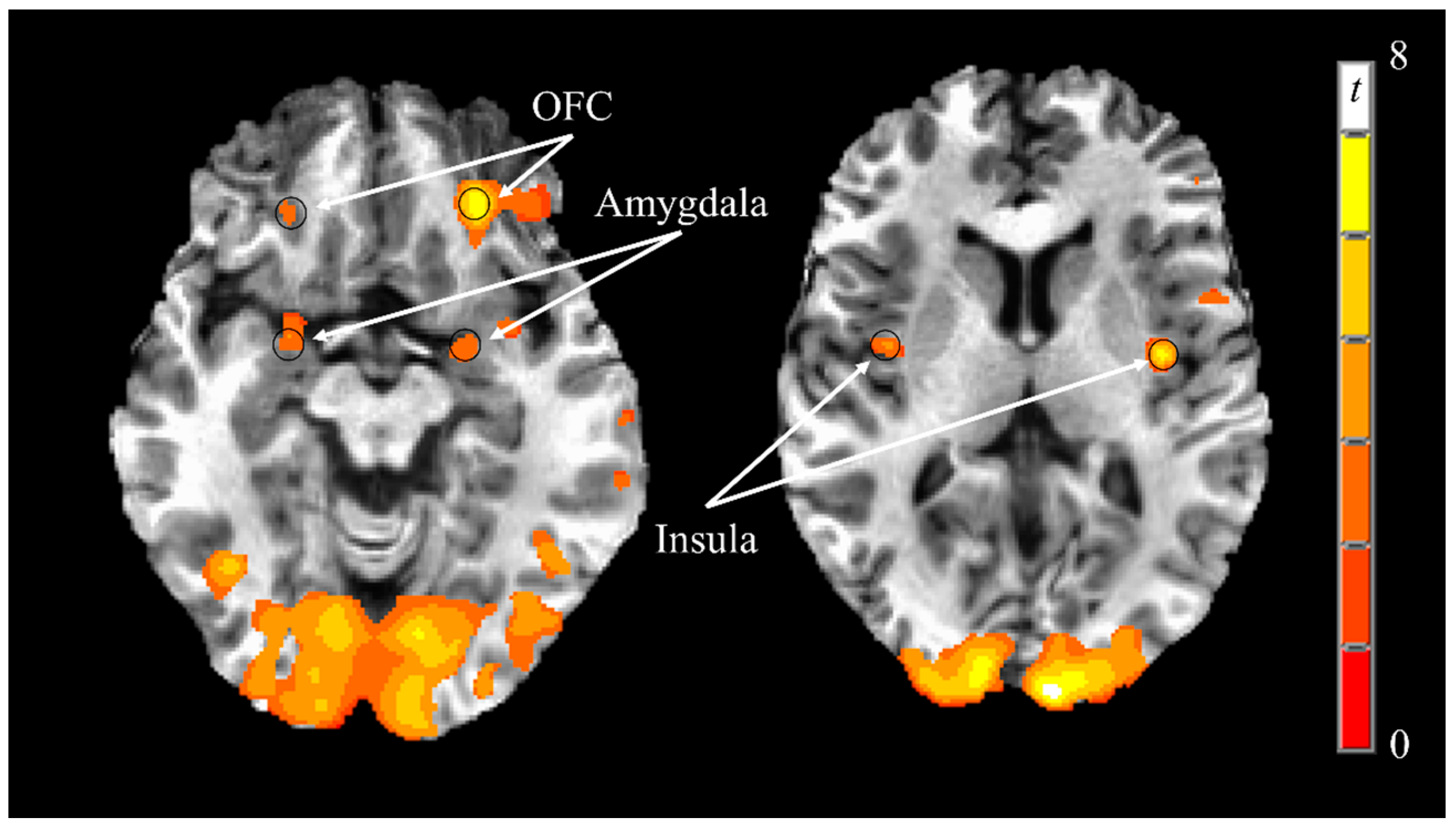
3.3. Neural Responses to Visual Food Stimuli
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dietary Guidelines for Americans 2015–2020, 8th ed.; U.S. Department of Health and Human Services and U.S. Department of Agriculture: Washington, DC, USA, 2015.
- Bes-Rastrollo, M.; Wedick, N.M.; Martinez-Gonzalez, M.A.; Li, T.Y.; Sampson, L.; Hu, F.B. Prospective study of nut consumption, long-term weight change, and obesity risk in women. Am. J. Clin. Nutr. 2009, 89, 1913–1919. [Google Scholar] [CrossRef] [PubMed]
- Bes-Rastrollo, M.; Sabaté, J.; Gómez-Gracia, E.; Alonso, A.; Martínez, J.A.; Martínez-González, M.A. Nut consumption and weight gain in a Mediterranean cohort: The SUN study. Obes. Silver Spring Md. 2007, 15, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Casas-Agustench, P.; Bulló, M.; Ros, E.; Basora, J.; Salas-Salvadó, J.; Nureta-PREDIMED investigators. Cross-sectional association of nut intake with adiposity in a Mediterranean population. Nutr. Metab. Cardiovasc. Dis. NMCD 2011, 21, 518–525. [Google Scholar] [CrossRef] [PubMed]
- Flores-Mateo, G.; Rojas-Rueda, D.; Basora, J.; Ros, E.; Salas-Salvado, J. Nut intake and adiposity: Meta-analysis of clinical trials. Am. J. Clin. Nutr. 2013, 97, 1346–1355. [Google Scholar] [CrossRef] [PubMed]
- Mattes, R.D.; Dreher, M.L. Nuts and healthy body weight maintenance mechanisms. Asia Pac. J. Clin. Nutr. 2010, 19, 137–141. [Google Scholar] [PubMed]
- Fraser, G.E.; Bennett, H.W.; Jaceldo, K.B.; Sabaté, J. Effect on body weight of a free 76 Kilojoule (320 calorie) daily supplement of almonds for six months. J. Am. Coll. Nutr. 2002, 21, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Hollis, J.; Mattes, R. Effect of chronic consumption of almonds on body weight in healthy humans. Br. J. Nutr. 2007, 98, 651–656. [Google Scholar] [CrossRef] [PubMed]
- Foster, G.D.; Shantz, K.L.; Vander Veur, S.S.; Oliver, T.L.; Lent, M.R.; Virus, A.; Szapary, P.O.; Rader, D.J.; Zemel, B.S.; Gilden-Tsai, A. A randomized trial of the effects of an almond-enriched, hypocaloric diet in the treatment of obesity. Am. J. Clin. Nutr. 2012, 96, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Wien, M.A.; Sabaté, J.M.; Iklé, D.N.; Cole, S.E.; Kandeel, F.R. Almonds vs. complex carbohydrates in a weight reduction program. Int. J. Obes. Relat. Metab. Disord. J. Int. Assoc. Study Obes. 2003, 27, 1365–1372. [Google Scholar] [CrossRef] [PubMed]
- Dhillon, J.; Tan, S.-Y.; Mattes, R.D. Almond Consumption during Energy Restriction Lowers Truncal Fat and Blood Pressure in Compliant Overweight or Obese Adults. J. Nutr. 2016, 146, 2513–2519. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.Y.; Mattes, R.D. Appetitive, dietary and health effects of almonds consumed with meals or as snacks: A randomized, controlled trial. Eur. J. Clin. Nutr. 2013, 67, 1205–1214. [Google Scholar] [CrossRef] [PubMed]
- Zaveri, S.; Drummond, S. The effect of including a conventional snack (cereal bar) and a nonconventional snack (almonds) on hunger, eating frequency, dietary intake and body weight. J. Hum. Nutr. Diet. Off. J. Br. Diet. Assoc. 2009, 22, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Hull, S.; Re, R.; Chambers, L.; Echaniz, A.; Wickham, M.S.J. A mid-morning snack of almonds generates satiety and appropriate adjustment of subsequent food intake in healthy women. Eur. J. Nutr. 2015, 54, 803–810. [Google Scholar] [CrossRef] [PubMed]
- Kirkmeyer, S.V.; Mattes, R.D. Effects of food attributes on hunger and food intake. Int. J. Obes. Relat. Metab. Disord. J. Int. Assoc. Study Obes. 2000, 24, 1167–1175. [Google Scholar] [CrossRef]
- Halton, T.L.; Hu, F.B. The effects of high protein diets on thermogenesis, satiety and weight loss: A critical review. J. Am. Coll. Nutr. 2004, 23, 373–385. [Google Scholar] [CrossRef] [PubMed]
- Leidy, H.J.; Clifton, P.M.; Astrup, A.; Wycherley, T.P.; Westerterp-Plantenga, M.S.; Luscombe-Marsh, N.D.; Woods, S.C.; Mattes, R.D. The role of protein in weight loss and maintenance. Am. J. Clin. Nutr. 2015. [Google Scholar] [CrossRef] [PubMed]
- Wanders, A.J.; van den Borne, J.J.; de Graaf, C.; Hulshof, T.; Jonathan, M.C.; Kristensen, M.; Mars, M.; Schols, H.A.; Feskens, E.J. Effects of dietary fibre on subjective appetite, energy intake and body weight: A systematic review of randomized controlled trials. Obes. Rev. 2011, 12, 724–739. [Google Scholar] [CrossRef] [PubMed]
- Tieken, S.M.; Leidy, H.J.; Stull, A.J.; Mattes, R.D.; Schuster, R.A.; Campbell, W.W. Effects of solid versus liquid meal-replacement products of similar energy content on hunger, satiety, and appetite-regulating hormones in older adults. Horm. Metab. Res. Horm. Stoffwechselforschung Horm. Metab. 2007, 39, 389–394. [Google Scholar] [CrossRef] [PubMed]
- DiMeglio, D.P.; Mattes, R.D. Liquid versus solid carbohydrate: Effects on food intake and body weight. Int. J. Obes. Relat. Metab. Disord. J. Int. Assoc. Study Obes. 2000, 24, 794–800. [Google Scholar] [CrossRef]
- Mori, A.M.; Considine, R.V.; Mattes, R.D. Acute and second-meal effects of almond form in impaired glucose tolerant adults: A randomized crossover trial. Nutr. Metab. 2011, 8, 6. [Google Scholar] [CrossRef] [PubMed]
- Berthoud, H.R. Homeostatic and non-homeostatic pathways involved in the control of food intake and energy balance. Obes. Silver Spring Md. 2006, 14 (Suppl. 5), 197S–200S. [Google Scholar] [CrossRef] [PubMed]
- Tang, D.W.; Fellows, L.K.; Small, D.M.; Dagher, A. Food and drug cues activate similar brain regions: A meta-analysis of functional MRI studies. Physiol. Behav. 2012, 106, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Pursey, K.M.; Stanwell, P.; Callister, R.J.; Brain, K.; Collins, C.E.; Burrows, T.L. Neural responses to visual food cues according to weight status: A systematic review of functional magnetic resonance imaging studies. Front. Nutr. 2014, 1, 7. [Google Scholar] [CrossRef] [PubMed]
- Burger, K.S.; Berner, L.A. A functional neuroimaging review of obesity, appetitive hormones and ingestive behavior. Physiol. Behav. 2014, 136, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Leidy, H.J.; Lepping, R.J.; Savage, C.R.; Harris, C.T. Neural responses to visual food stimuli after a normal vs. higher protein breakfast in breakfast-skipping teens: A pilot fMRI study. Obes. Silver Spring Md. 2011, 19, 2019–2025. [Google Scholar] [CrossRef] [PubMed]
- Sayer, R.D.; Amankwaah, A.F.; Tamer, G.G.; Chen, N.; Wright, A.J.; Tregellas, J.R.; Cornier, M.-A.; Kareken, D.A.; Talavage, T.M.; McCrory, M.A.; et al. Effects of Dietary Protein and Fiber at Breakfast on Appetite, ad Libitum Energy Intake at Lunch, and Neural Responses to Visual Food Stimuli in Overweight Adults. Nutrients 2016, 8. [Google Scholar] [CrossRef] [PubMed]
- Random.org. Available online: http://www.random.org (accessed on 20 July 2017).
- Stubbs, R.J.; Hughes, D.A.; Johnstone, A.M.; Rowley, E.; Reid, C.; Elia, M.; Stratton, R.; Delargy, H.; King, N.; Blundell, J.E. The use of visual analogue scales to assess motivation to eat in human subjects: A review of their reliability and validity with an evaluation of new hand-held computerized systems for temporal tracking of appetite ratings. Br. J. Nutr. 2000, 84, 405–415. [Google Scholar] [CrossRef] [PubMed]
- Marsh-Richard, D.M.; Hatzis, E.S.; Mathias, C.W.; Venditti, N.; Dougherty, D.M. Adaptive Visual Analog Scales (AVAS): A modifiable software program for the creation, administration, and scoring of visual analog scales. Behav. Res. Methods 2009, 41, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Drew Sayer, R.; Tamer, G.G.; Chen, N.; Tregellas, J.R.; Cornier, M.-A.; Kareken, D.A.; Talavage, T.M.; McCrory, M.A.; Campbell, W.W. Reproducibility assessment of brain responses to visual food stimuli in adults with overweight and obesity. Obes. Silver Spring Md. 2016, 24, 2057–2063. [Google Scholar] [CrossRef] [PubMed]
- Peirce, J.W. Generating Stimuli for Neuroscience Using PsychoPy. Front. Neuroinform. 2008, 2, 10. [Google Scholar] [CrossRef] [PubMed]
- Burger, K.S.; Cornier, M.A.; Ingebrigtsen, J.; Johnson, S.L. Assessing food appeal and desire to eat: The effects of portion size & energy density. Int. J. Behav. Nutr. Phys. Act. 2011, 8, 101. [Google Scholar] [CrossRef] [PubMed]
- Cornier, M.A. The effects of overfeeding and propensity to weight gain on the neuronal responses to visual food cues. Physiol. Behav. 2009, 97, 525–530. [Google Scholar] [CrossRef] [PubMed]
- Cornier, M.A.; Melanson, E.L.; Salzberg, A.K.; Bechtell, J.L.; Tregellas, J.R. The effects of exercise on the neuronal response to food cues. Physiol. Behav. 2012, 105, 1028–1034. [Google Scholar] [CrossRef] [PubMed]
- Cornier, M.A.; Von Kaenel, S.S.; Bessesen, D.H.; Tregellas, J.R. Effects of overfeeding on the neuronal response to visual food cues. Am. J. Clin. Nutr. 2007, 86, 965–971. [Google Scholar] [PubMed]
- Cornier, M.A.; Salzberg, A.K.; Endly, D.C.; Bessesen, D.H.; Rojas, D.C.; Tregellas, J.R. The effects of overfeeding on the neuronal response to visual food cues in thin and reduced-obese individuals. PLoS ONE 2009, 4, e6310. [Google Scholar] [CrossRef] [PubMed]
- Cox, R.W. AFNI: Software for analysis and visualization of functional magnetic resonance neuroimages. Comput. Biomed. Res. 1996, 29, 162–173. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.Y.; Dhillon, J.; Mattes, R.D. A review of the effects of nuts on appetite, food intake, metabolism, and body weight. Am. J. Clin. Nutr. 2014, 100 (Suppl. 1), 412S–422S. [Google Scholar] [CrossRef] [PubMed]
- Blundell, J.; de Graaf, C.; Hulshof, T.; Jebb, S.; Livingstone, B.; Lluch, A.; Mela, D.; Salah, S.; Schuring, E.; van der Knaap, H.; et al. Appetite control: Methodological aspects of the evaluation of foods. Obes. Rev. 2010, 11, 251–270. [Google Scholar] [CrossRef] [PubMed]
- Bryant, M.; Truesdale, K.P.; Dye, L. Modest changes in dietary intake across the menstrual cycle: Implications for food intake research. Br. J. Nutr. 2006, 96, 888–894. [Google Scholar] [CrossRef] [PubMed]
- Van Vugt, D.A. Brain imaging studies of appetite in the context of obesity and the menstrual cycle. Hum. Reprod. Update 2010, 16, 276–292. [Google Scholar] [CrossRef] [PubMed]
| Nutrient | Almonds | Bake Food Product |
|---|---|---|
| Total Energy (kcal) | 174 | 174 |
| Total Mass (g) | 28 | 40 |
| Energy Density (kcal/g) | 6.2 | 4.4 |
| Carbohydrates (g) 1 | 6 | 6 |
| Protein (g) | 6 | 5 |
| Fat (g) | 14 | 14 |
| Fiber (g) | 3 | 3 |
| Variable | Almond Group (n = 11) | Control Group (n = 11) | Combined (n = 22) | p Value for Difference 2 |
|---|---|---|---|---|
| Female/Male | 9/2 | 10/1 | 19/3 | - |
| Age (years) | 33 ± 4 | 37 ± 4 | 35 ± 3 | 0.48 |
| Body Mass (kg) | 82.8 ± 4.1 | 80.8 ± 4.1 | 81.8 ± 2.8 | 0.73 |
| BMI (kg/m2) | 29.9 ± 1.2 | 30.0 ± 1.6 | 30.0 ± 1.0 | 0.96 |
| % Body Fat | 40.8 ± 25.8 | 38.9 ± 2.2 | 39.9 ± 1.6 | 0.55 |
| Brain Region | X2 | Y | Z | t Statistic | p Value |
|---|---|---|---|---|---|
| Insula (L) | −38 | −7 | 6 | 6.04 | <0.0001 |
| Insula (R) | 39 | −4 | 4 | 4.47 | 0.0002 |
| Amygdala (L) | −23 | 0 | −17 | 3.26 | 0.0036 |
| Amygdala (R) | 24 | 0 | −18 | 3.66 | 0.0014 |
| Orbitofrontal Cortex (L) | −25 | 35 | −18 | 4.18 | 0.0004 |
| Orbitofrontal Cortex (R) | 23 | 33 | −20 | 3.77 | 0.0010 |
| Brain Region | Food | Time | Food × Time |
|---|---|---|---|
| Insula (L) | 0.45 | 0.11 | 0.41 |
| Insula (R) | 0.72 | 0.08 | 0.08 |
| Amygdala (L) | 0.90 | 0.49 | 0.46 |
| Amygdala (R) | 0.89 | 0.31 | 0.29 |
| Orbitofrontal Cortex (L) | 0.81 | 0.12 | 0.83 |
| Orbitofrontal Cortex (R) | 0.36 | 0.08 | 0.33 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sayer, R.D.; Dhillon, J.; Tamer, G.G.; Cornier, M.-A.; Chen, N.; Wright, A.J.; Campbell, W.W.; Mattes, R.D. Consuming Almonds vs. Isoenergetic Baked Food Does Not Differentially Influence Postprandial Appetite or Neural Reward Responses to Visual Food Stimuli. Nutrients 2017, 9, 807. https://doi.org/10.3390/nu9080807
Sayer RD, Dhillon J, Tamer GG, Cornier M-A, Chen N, Wright AJ, Campbell WW, Mattes RD. Consuming Almonds vs. Isoenergetic Baked Food Does Not Differentially Influence Postprandial Appetite or Neural Reward Responses to Visual Food Stimuli. Nutrients. 2017; 9(8):807. https://doi.org/10.3390/nu9080807
Chicago/Turabian StyleSayer, R. Drew, Jaapna Dhillon, Gregory G. Tamer, Marc-Andre Cornier, Ningning Chen, Amy J. Wright, Wayne W. Campbell, and Richard D. Mattes. 2017. "Consuming Almonds vs. Isoenergetic Baked Food Does Not Differentially Influence Postprandial Appetite or Neural Reward Responses to Visual Food Stimuli" Nutrients 9, no. 8: 807. https://doi.org/10.3390/nu9080807
APA StyleSayer, R. D., Dhillon, J., Tamer, G. G., Cornier, M.-A., Chen, N., Wright, A. J., Campbell, W. W., & Mattes, R. D. (2017). Consuming Almonds vs. Isoenergetic Baked Food Does Not Differentially Influence Postprandial Appetite or Neural Reward Responses to Visual Food Stimuli. Nutrients, 9(8), 807. https://doi.org/10.3390/nu9080807





