The Association between the Mediterranean Dietary Pattern and Cognitive Health: A Systematic Review
Abstract
:1. Introduction
2. Materials and Methods
2.1. Literature Search
2.2. Data Extraction
2.3. Quality Assessment
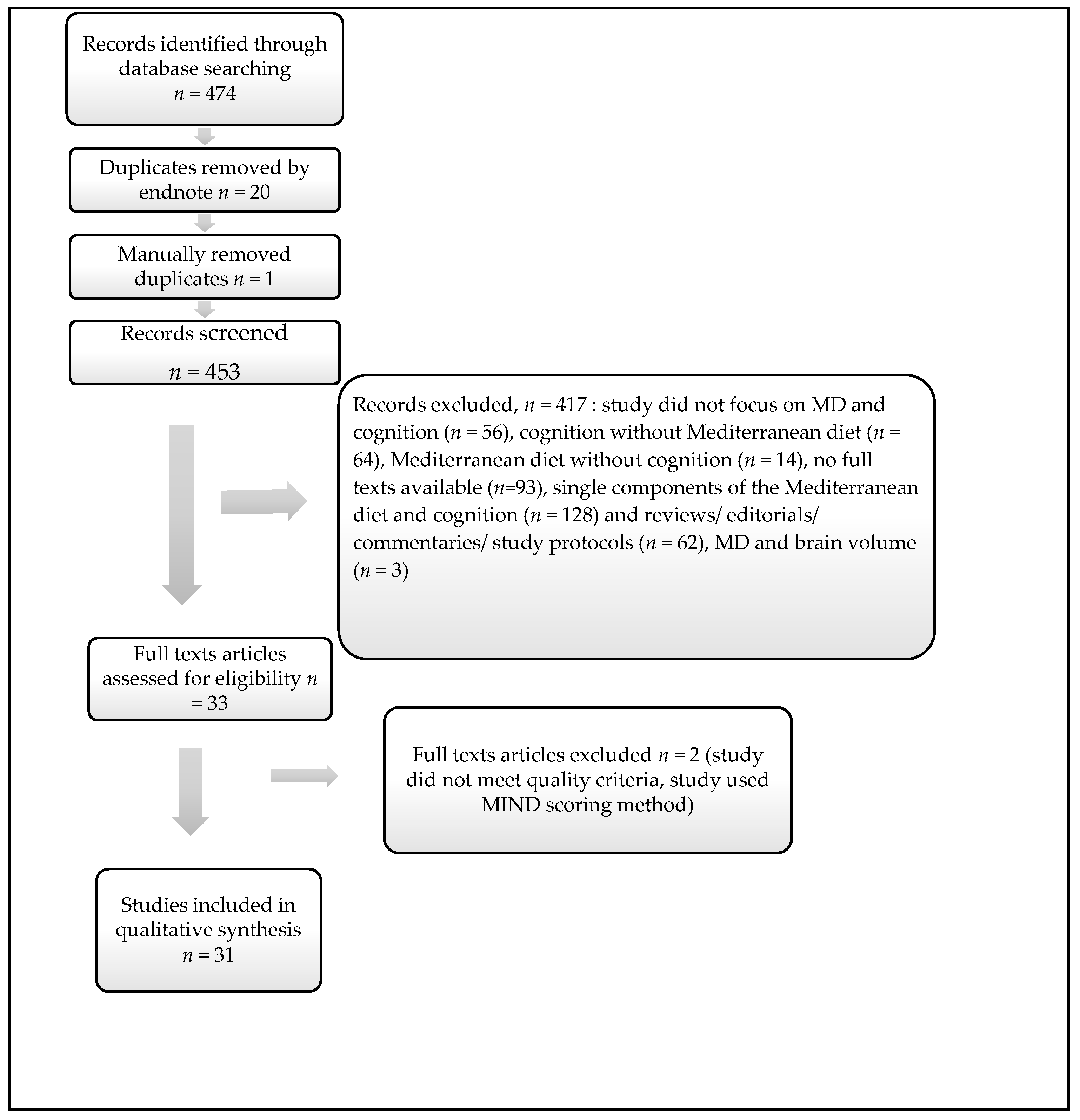
3. Results
3.1. Study Selection
3.2. MD and Cognitive Function
3.2.1. Cross Sectional Studies
3.2.2. Randomized Controlled Trials
3.2.3. Cohort Studies
3.3. MD and the Risk of Developing Alzheimer’s Disease
3.3.1. Studies That Detected Significance
3.3.2. Studies That Did Not Detect Significance
4. Discussion
4.1. Variations in Dietary Scores and Cognitive Tests
4.2. Variations in Age and Follow up Duration
4.3. Variations in Study Region and Gender
5. Strengths and Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
References
- World Health Organization. Dementia. Available online: http://www.who.int/mediacentre/factsheets/fs362/en/ (accessed on 28 June 2017).
- Arbias. Your Guide to Neuropsychological Assessment. Available online: https://www.google.ch/url?sa=t&rct=j&q=&esrc=s&source=web&cd=1&cad=rja&uact=8&ved=0ahUKEwj5wvWSud3UAhUlD8AKHSZRCRYQFggiMAA&url=https%3A%2F%2Fwww.covwarkpt.nhs.uk%2Fdownload.cfm%3Fdoc%3Ddocm93jijm4n1375.pdf%26ver%3D1678&usg=AFQjCNGQux8pe2y17Hu_4J4rjAHU6t0P4w (accessed on 27 June 2017).
- King, J.A.; Trinkler, I.; Hartley, T.; Vargha-Khadem, F.; Burgess, N. The hippocampal role in spatial memory and the familiarity-recollection distinction: A case study. Neuropsychology 2004, 18, 405–417. [Google Scholar] [CrossRef] [PubMed]
- Alzheimer’s Australia. Tests Used in Diagnosing Dementia. Available online: https://www.fightdementia.org.au/files/helpsheets/Helpsheet-DementiaQandA10-TestsUsedInDiagnosingDementia_english.pdf (accessed on 15 December 2016).
- Alzheimer’s Association. What Is Dementia? Available online: http://www.alz.org/what-is-dementia.asp (accessed on 15 December 2016).
- Alzheimer’s Society. Mediterranean Diet. Available online: https://www.alzheimers.org.uk/info/20010/risk_factors_and_prevention/149/mediterranean_diet (accessed on 15 December 2016).
- Willett, W.C.; Sacks, F.; Trichopoulou, A.; Drescher, G.; Ferro-Luzzi, A.; Helsing, E.; Trichopoulos, D. Mediterranean diet pyramid: A cultural model for healthy eating. Am. J. Clin. Nutr. 1995, 61, 1402S–1406S. [Google Scholar] [PubMed]
- Trichopoulou, A.; Kouris-Blazos, A.; Wahlqvist, M.L.; Gnardellis, C.; Lagiou, P.; Polychronopoulos, E.; Vassilakou, T.; Lipworth, L.; Trichopoulos, D. Diet and overall survival in elderly people. BMJ 1995, 311, 1457–1460. [Google Scholar] [CrossRef] [PubMed]
- Panagiotakos, D.B.; Pitsavos, C.; Arvaniti, F.; Stefanadis, C. Adherence to the mediterranean food pattern predicts the prevalence of hypertension, hypercholesterolemia, diabetes and obesity, among healthy adults; the accuracy of the Meddietscore. Prev. Med. 2007, 44, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Jones, D.P.; Goldberg, J.; Ziegler, T.R.; Bostick, R.M.; Wilson, P.W.; Manatunga, A.K.; Shallenberger, L.; Jones, L.; Vaccarino, V. Association between adherence to the Mediterranean diet and Oxidative stress. Am. J. Clin. Nutr. 2008, 88, 1364–1370. [Google Scholar] [PubMed]
- Gaskins, A.J.; Rovner, A.J.; Mumford, S.L.; Yeung, E.; Browne, R.W.; Trevisan, M.; Perkins, N.J.; Wactawski-Wende, J.; Schisterman, E.F.; Group, B.S. Adherence to a Mediterranean diet and plasma concentrations of lipid peroxidation in premenopausal women. Am. J. Clin. Nutr. 2010, 92, 1461–1467. [Google Scholar] [CrossRef] [PubMed]
- Wojtunik-Kulesza, K.A.; Oniszczuk, A.; Oniszczuk, T.; Waksmundzka-Hajnos, M. The influence of common free radicals and antioxidants on development of Alzheimer’s Disease. Biomed. Pharmacother. 2016, 78, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Giasson, B.I.; Ischiropoulos, H.; Lee, V.M.-Y.; Trojanowski, J.Q. The relationship between oxidative/nitrative stress and pathological inclusions in Alzheimer’s and Parkinson’s diseases 1, 2. Free Radic. Biol. Med. 2002, 32, 1264–1275. [Google Scholar] [CrossRef]
- Knight, A.; Bryan, J.; Murphy, K. Is the Mediterranean diet a feasible approach to preserving cognitive function and reducing risk of dementia for older adults in Western countries? New insights and future directions. Ageing Res. Rev. 2016, 25, 85–101. [Google Scholar] [CrossRef] [PubMed]
- Mori, T.A.; Beilin, L.J. Omega-3 fatty acids and inflammation. Curr. Atheroscler. Rep. 2004, 6, 461–467. [Google Scholar] [CrossRef] [PubMed]
- Esposito, K.; Nappo, F.; Giugliano, F.; Di Palo, C.; Ciotola, M.; Barbieri, M.; Paolisso, G.; Giugliano, D. Meal modulation of circulating interleukin 18 and adiponectin concentrations in healthy subjects and in patients with type 2 diabetes mellitus. Am. J. Clin. Nutr. 2003, 78, 1135–1140. [Google Scholar] [PubMed]
- Panagiotakos, D.; Kalogeropoulos, N.; Pitsavos, C.; Roussinou, G.; Palliou, K.; Chrysohoou, C.; Stefanadis, C. Validation of the meddietscore via the determination of plasma fatty acids. Int. J. Food Sci. Nutr. 2009, 60, 168–180. [Google Scholar] [CrossRef] [PubMed]
- Esposito, K.; Marfella, R.; Ciotola, M.; Di Palo, C.; Giugliano, F.; Giugliano, G.; D’armiento, M.; D’andrea, F.; Giugliano, D. Effect of a Mediterranean-style diet on endothelial dysfunction and markers of vascular inflammation in the metabolic syndrome: A randomized trial. JAMA 2004, 292, 1440–1446. [Google Scholar] [CrossRef] [PubMed]
- O’callaghan, J.P.; Sriram, K.; Miller, D.B. Defining “neuroinflammation”. Ann. N. Y. Acad. Sci. 2008, 1139, 318–330. [Google Scholar] [CrossRef] [PubMed]
- Heneka, M.T.; O’Banion, M.K. Inflammatory processes in Alzheimer’s disease. J. Neuroimmunol. 2007, 184, 69–91. [Google Scholar] [CrossRef] [PubMed]
- Roberts, R.O.; Knopman, D.S.; Geda, Y.E.; Cha, R.H.; Roger, V.L.; Petersen, R.C. Coronary heart disease is associated with non-amnestic mild cognitive impairment. Neurobiol. Aging 2010, 31, 1894–1902. [Google Scholar] [CrossRef] [PubMed]
- Qiu, C.; Winblad, B.; Marengoni, A.; Klarin, I.; Fastbom, J.; Fratiglioni, L. Heart failure and risk of dementia and Alzheimer disease: A population-based cohort study. Arch. Intern. Med. 2006, 166, 1003–1008. [Google Scholar] [CrossRef] [PubMed]
- Knecht, S.; Oelschläger, C.; Duning, T.; Lohmann, H.; Albers, J.; Stehling, C.; Heindel, W.; Breithardt, G.; Berger, K.; Ringelstein, E.B. Atrial fibrillation in stroke-free patients is associated with memory impairment and hippocampal atrophy. Eur. Heart J. 2008, 29, 2125–2132. [Google Scholar] [CrossRef] [PubMed]
- Justin, B.N.; Turek, M.; Hakim, A.M. Heart disease as a risk factor for dementia. Clin. Epidemiol. 2013, 5, 135–145. [Google Scholar] [PubMed]
- De Bruijn, R.F.; Ikram, M.A. Cardiovascular risk factors and future risk of Alzheimer’s disease. BMC Med. 2014, 12, 130. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Marventano, S.; Yang, J.; Micek, A.; Pajak, A.; Scalfi, L.; Galvano, F.; Kales, S.N. A comprehensive meta-analysis on evidence of Mediterranean diet and cardiovascular disease: Are individual components equal? Crit. Rev. Food Sci. Nutr. 2015, 57, 3218–3232. [Google Scholar] [CrossRef] [PubMed]
- Nordmann, A.J.; Suter-Zimmermann, K.; Bucher, H.C.; Shai, I.; Tuttle, K.R.; Estruch, R.; Briel, M. Meta-analysis comparing Mediterranean to low-fat diets for modification of cardiovascular risk factors. Am. J. Med. 2011, 124, 841–851. [Google Scholar] [CrossRef] [PubMed]
- Tektonidis, T.G.; Åkesson, A.; Gigante, B.; Wolk, A.; Larsson, S.C. A Mediterranean diet and risk of myocardial infarction, heart failure and stroke: A population-based cohort study. Atherosclerosis 2015, 243, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Gonzalez, M.A.; Bes-Rastrollo, M.; Serra-Majem, L.; Lairon, D.; Estruch, R.; Trichopoulou, A. Mediterranean food pattern and the primary prevention of chronic disease: Recent developments. Nutr. Rev. 2009, 67, S111–S116. [Google Scholar] [CrossRef] [PubMed]
- Solfrizzi, V.; Panza, F. Mediterranean diet and cognitive decline. A lesson from the whole-diet approach: What challenges lie ahead? J. Alzheimer’s Dis. 2014, 39, 283–286. [Google Scholar]
- Woodside, J.; Gallagher, N.; Neville, C.; McKinley, M. Mediterranean diet interventions to prevent cognitive decline—Opportunities and challenges. Eur. J. Clin. Nutr. 2014, 68, 1241–1244. [Google Scholar] [CrossRef] [PubMed]
- Academy of Nutrition and Dietetics. Evidence Analysis Manual: Steps in the Academy Evidence Analysis Process; Academy of Nutrition and Dietetics: Chicago, IL, USA, 2012; pp. 90–92. [Google Scholar]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, P. Preferred reporting items for systematic reviews and meta-analyses: The Prisma statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Chan, R.; Chan, D.; Woo, J. A cross sectional study to examine the association between dietary patterns and cognitive impairment in older Chinese people in Hong Kong. J. Nutr. Health Aging 2013, 17, 757–765. [Google Scholar] [CrossRef] [PubMed]
- Crichton, G.E.; Bryan, J.; Hodgson, J.M.; Murphy, K.J. Mediterranean diet adherence and self-reported psychological functioning in an Australian sample. Appetite 2013, 70, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Corley, J.; Starr, J.M.; McNeill, G.; Deary, I.J. Do dietary patterns influence cognitive function in old age? Int. Psychogeriatr. 2013, 25, 1393–1407. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.; Scott, T.; Gao, X.; Maras, J.E.; Bakun, P.J.; Tucker, K.L. Mediterranean diet, healthy eating index 2005, and cognitive function in middle-aged and older Puerto Rican adults. J. Acad. Nutr. Diet. 2013, 113, 276–281. [Google Scholar] [CrossRef] [PubMed]
- Katsiardanis, K.; Diamantaras, A.A.; Dessypris, N.; Michelakos, T.; Anastasiou, A.; Katsiardani, K.P.; Kanavidis, P.; Papadopoulos, F.C.; Stefanadis, C.; Panagiotakos, D.B.; et al. Cognitive impairment and dietary habits among elders: The Velestino study. J. Med. Food 2013, 16, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Zbeida, M.; Goldsmith, R.; Shimony, T.; Vardi, H.; Naggan, L.; Shahar, D.R. Mediterranean diet and functional indicators among older adults in non-Mediterranean and Mediterranean countries. J. Nutr. Health Aging 2014, 18, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Lapiscina, E.H.; Clavero, P.; Toledo, E.; Estruch, R.; Salas-Salvado, J.; San Julian, B.; Sanchez-Tainta, A.; Ros, E.; Valls-Pedret, C.; Martinez-Gonzalez, M.A. Mediterranean diet improves cognition: The predimed-navarra randomised trial. J. Neurol. Neurosurg. Psychiatry 2013, 84, 1318–1325. [Google Scholar] [CrossRef] [PubMed]
- Valls-Pedret, C.; Sala-Vila, A.; Serra-Mir, M.; Corella, D.; de la Torre, R.; Martinez-Gonzalez, M.A.; Martinez-Lapiscina, E.H.; Fito, M.; Perez-Heras, A.; Salas-Salvado, J.; et al. Mediterranean diet and age-related cognitive decline: A randomized clinical trial. JAMA Intern. Med. 2015, 175, 1094–1103. [Google Scholar] [CrossRef] [PubMed]
- Cherbuin, N.; Anstey, K.J. The mediterranean diet is not related to cognitive change in a large prospective investigation: The path through life study. Am. J. Geriatr. Psychiatry Off. J. Am. Assoc. Geriatr. Psychiatry 2012, 20, 635–639. [Google Scholar] [CrossRef] [PubMed]
- Haring, B.; Wu, C.; Mossavar-Rahmani, Y.; Snetselaar, L.; Brunner, R.; Wallace, R.B.; Neuhouser, M.L.; Wassertheil-Smoller, S. No association between dietary patterns and risk for cognitive decline in older women with 9-year follow-up: Data from the women’s health initiative memory study. J. Acad. Nutr. Diet. 2016, 116, 921–930.e1. [Google Scholar] [CrossRef] [PubMed]
- Samieri, C.; Okereke, O.I.; Devore, E.E.; Grodstein, F. Long-term adherence to the mediterranean diet is associated with overall cognitive status, but not cognitive decline, in women. J. Nutr. 2013, 143, 493–499. [Google Scholar] [CrossRef] [PubMed]
- Samieri, C.; Grodstein, F.; Rosner, B.A.; Kang, J.H.; Cook, N.R.; Manson, J.E.; Buring, J.E.; Willett, W.C.; Okereke, O.I. Mediterranean diet and cognitive function in older age. Epidemiology 2013, 24, 490–499. [Google Scholar] [CrossRef] [PubMed]
- Vercambre, M.N.; Grodstein, F.; Berr, C.; Kang, J.H. Mediterranean diet and cognitive decline in women with cardiovascular disease or risk factors. J. Acad. Nutr. Diet. 2012, 112, 816–823. [Google Scholar] [CrossRef] [PubMed]
- Gardener, S.L.; Rainey-Smith, S.R.; Barnes, M.B.; Sohrabi, H.R.; Weinborn, M.; Lim, Y.Y.; Harrington, K.; Taddei, K.; Gu, Y.; Rembach, A.; et al. Dietary patterns and cognitive decline in an Australian study of ageing. Mol. Psychiatry 2015, 20, 860–866. [Google Scholar] [CrossRef] [PubMed]
- Psaltopoulou, T.; Kyrozis, A.; Stathopoulos, P.; Trichopoulos, D.; Vassilopoulos, D.; Trichopoulou, A. Diet, physical activity and cognitive impairment among elders: The epic-greece cohort (European prospective investigation into cancer and nutrition). Public Health Nutr. 2008, 11, 1054–1062. [Google Scholar] [CrossRef] [PubMed]
- Qin, B.; Adair, L.S.; Plassman, B.L.; Batis, C.; Edwards, L.J.; Popkin, B.M.; Mendez, M.A. Dietary patterns and cognitive decline among Chinese older adults. Epidemiology 2015, 26, 758–768. [Google Scholar] [CrossRef] [PubMed]
- Feart, C.; Samieri, C.; Rondeau, V.; Amieva, H.; Portet, F.; Dartigues, J.F.; Scarmeas, N.; Barberger-Gateau, P. Adherence to a mediterranean diet, cognitive decline, and risk of dementia. JAMA 2009, 302, 638–648. [Google Scholar] [CrossRef] [PubMed]
- Kesse-Guyot, E.; Andreeva, V.A.; Lassale, C.; Ferry, M.; Jeandel, C.; Hercberg, S.; Galan, P. Mediterranean diet and cognitive function: A french study. Am. J. Clin. Nutr. 2013, 97, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Galbete, C.; Toledo, E.; Toledo, J.B.; Bes-Rastrollo, M.; Buil-Cosiales, P.; Marti, A.; Guillen-Grima, F.; Martinez-Gonzalez, M.A. Mediterranean diet and cognitive function: The sun project. J. Nutr. Health Aging 2015, 19, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Koyama, A.; Houston, D.K.; Simonsick, E.M.; Lee, J.S.; Ayonayon, H.N.; Shahar, D.R.; Rosano, C.; Satterfield, S.; Yaffe, K. Association between the mediterranean diet and cognitive decline in a Biracial population. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2015, 70, 354–359. [Google Scholar] [CrossRef] [PubMed]
- Tangney, C.C.; Kwasny, M.J.; Li, H.; Wilson, R.S.; Evans, D.A.; Morris, M.C. Adherence to a mediterranean-type dietary pattern and cognitive decline in a community population. Am. J. Clin. Nutr. 2011, 93, 601–607. [Google Scholar] [CrossRef] [PubMed]
- Tangney, C.C.; Li, H.; Wang, Y.; Barnes, L.; Schneider, J.A.; Bennett, D.A.; Morris, M.C. Relation of dash- and mediterranean-like dietary patterns to cognitive decline in older persons. Neurology 2014, 83, 1410–1416. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Kyrozis, A.; Rossi, M.; Katsoulis, M.; Trichopoulos, D.; La Vecchia, C.; Lagiou, P. Mediterranean diet and cognitive decline over time in an elderly Mediterranean population. Eur. J. Nutr. 2015, 54, 1311–1321. [Google Scholar] [CrossRef] [PubMed]
- Wengreen, H.; Munger, R.G.; Cutler, A.; Quach, A.; Bowles, A.; Corcoran, C.; Tschanz, J.T.; Norton, M.C.; Welsh-Bohmer, K.A. Prospective study of dietary approaches to stop hypertension- and Mediterranean-style dietary patterns and age-related cognitive change: The cache county study on memory, health and aging. Am. J. Clin. Nutr. 2013, 98, 1263–1271. [Google Scholar] [CrossRef] [PubMed]
- Tsivgoulis, G.; Judd, S.; Letter, A.J.; Alexandrov, A.V.; Howard, G.; Nahab, F.; Unverzagt, F.W.; Moy, C.; Howard, V.J.; Kissela, B.; et al. Adherence to a Mediterranean diet and risk of incident cognitive impairment. Neurology 2013, 80, 1684–1692. [Google Scholar] [CrossRef] [PubMed]
- Gardener, S.; Gu, Y.; Rainey-Smith, S.R.; Keogh, J.B.; Clifton, P.M.; Mathieson, S.L.; Taddei, K.; Mondal, A.; Ward, V.K.; Scarmeas, N.; et al. Adherence to a Mediterranean diet and Alzheimer’s disease risk in an Australian population. Transl. Psychiatry 2012, 2, e164. [Google Scholar] [CrossRef] [PubMed]
- Scarmeas, N.; Stern, Y.; Mayeux, R.; Manly, J.J.; Schupf, N.; Luchsinger, J.A. Mediterranean diet and mild cognitive impairment. Arch. Neurol. 2009, 66, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Olsson, E.; Karlstrom, B.; Kilander, L.; Byberg, L.; Cederholm, T.; Sjogren, P. Dietary patterns and cognitive dysfunction in a 12-year follow-up study of 70 year old men. J. Alzheimer’s Dis. JAD 2015, 43, 109–119. [Google Scholar] [PubMed]
- Scarmeas, N.; Luchsinger, J.A.; Mayeux, R.; Stern, Y. Mediterranean diet and Alzheimer disease mortality. Neurology 2007, 69, 1084–1093. [Google Scholar] [CrossRef] [PubMed]
- Morris, M.C.; Tangney, C.C.; Wang, Y.; Sacks, F.M.; Bennett, D.A.; Aggarwal, N.T. Mind diet associated with reduced incidence of Alzheimer’s disease. Alzheimer’s Dement. J. Alzheimer’s Assoc. 2015, 11, 1007–1014. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Luchsinger, J.A.; Stern, Y.; Scarmeas, N. Mediterranean diet, inflammatory and metabolic biomarkers, and risk of Alzheimer’s disease. J. Alzheimer’s Dis. JAD 2010, 22, 483–492. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Costacou, T.; Bamia, C.; Trichopoulos, D. Adherence to a mediterranean diet and survival in a Greek population. N. Engl. J. Med. 2003, 348, 2599–2608. [Google Scholar] [CrossRef] [PubMed]
- Milà-Villarroel, R.; Bach-Faig, A.; Puig, J.; Puchal, A.; Farran, A.; Serra-Majem, L.; Carrasco, J.L. Comparison and evaluation of the reliability of indexes of adherence to the Mediterranean diet. Public Health Nutr. 2011, 14, 2338–2345. [Google Scholar] [CrossRef] [PubMed]
- Simmons, B.B.; Hartmann, B.; Dejoseph, D. Evaluation of suspected dementia. Ratio 2011, 1, 9. [Google Scholar]
- Velayudhan, L.; Ryu, S.-H.; Raczek, M.; Philpot, M.; Lindesay, J.; Critchfield, M.; Livingston, G. Review of brief cognitive tests for patients with suspected dementia. Int. Psychogeriatr. 2014, 26, 1247–1262. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, A.J.; Malladi, S. Screening and case finding tools for the detection of dementia. Part i: Evidence-based meta-analysis of Multidomain tests. Am. J. Geriatr. Psychiatry 2010, 18, 759–782. [Google Scholar] [CrossRef] [PubMed]
- Zucker, I.; Beery, A.K. Males still dominate animal studies. Nature 2010, 465, 690. [Google Scholar] [CrossRef] [PubMed]
- Bach-Faig, A.; Berry, E.M.; Lairon, D.; Reguant, J.; Trichopoulou, A.; Dernini, S.; Medina, F.X.; Battino, M.; Belahsen, R.; Miranda, G. Mediterranean diet pyramid today. Science and cultural updates. Public Health Nutr. 2011, 14, 2274–2284. [Google Scholar] [CrossRef] [PubMed]
- Sofi, F.; Valecchi, D.; Bacci, D.; Abbate, R.; Gensini, G.F.; Casini, A.; Macchi, C. Physical activity and risk of cognitive decline: A meta-analysis of prospective studies. J. Intern. Med. 2011, 269, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Petersson, S.D.; Philippou, E. Mediterranean diet, cognitive function, and dementia: A systematic review of the evidence. Adv. Nutr. Int. Rev. J. 2016, 7, 889–904. [Google Scholar] [CrossRef] [PubMed]
- Panza, F.; Solfrizzi, V.; Colacicco, A.; D’introno, A.; Capurso, C.; Torres, F.; Del Parigi, A.; Capurso, S.; Capurso, A. Mediterranean diet and cognitive decline. Public Health Nutr. 2004, 7, 959–963. [Google Scholar] [CrossRef] [PubMed]
- Sofi, F.; Cesari, F.; Abbate, R.; Gensini, G.; Casini, A. Adherence to Mediterranean diet and health status: Meta-analysis-art. No. A1344. Br. Med. J. 2008, 337, NIL_44–NIL_50. [Google Scholar] [CrossRef] [PubMed]
- Féart, C.; Samieri, C.; Barberger-Gateau, P. Mediterranean diet and cognitive function in older adults. Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 14–18. [Google Scholar] [CrossRef] [PubMed]
- Féart, C.; Samieri, C.; Allès, B.; Barberger-Gateau, P. Potential benefits of adherence to the Mediterranean diet on cognitive health. Proc. Nutr. Soc. 2013, 72, 140–152. [Google Scholar] [CrossRef] [PubMed]
- Lourida, I.; Soni, M.; Thompson-Coon, J.; Purandare, N.; Lang, I.A.; Ukoumunne, O.C.; Llewellyn, D.J. Mediterranean diet, cognitive function, and dementia: A systematic review. Epidemiology 2013, 24, 479–489. [Google Scholar] [CrossRef] [PubMed]
- Opie, R.S.; Ralston, R.A.; Walker, K.Z. Adherence to a Mediterranean-style diet can slow the rate of cognitive decline and decrease the risk of dementia: A systematic review. Nutr. Diet. 2013, 70, 206–217. [Google Scholar] [CrossRef]
- Van de Rest, O.; Berendsen, A.A.; Haveman-Nies, A.; de Groot, L.C. Dietary patterns, cognitive decline, and dementia: A systematic review. Adv. Nutr. Int. Rev. J. 2015, 6, 154–168. [Google Scholar] [CrossRef] [PubMed]
- Hardman, R.J.; Kennedy, G.; Macpherson, H.; Scholey, A.B.; Pipingas, A. Adherence to a Mediterranean-style diet and effects on cognition in adults: A qualitative evaluation and systematic review of longitudinal and prospective trials. Front. Nutr. 2016, 3, 22. [Google Scholar] [CrossRef] [PubMed]
- Tangney, C.C. Dash and Mediterranean-type dietary patterns to maintain cognitive health. Curr. Nutr. Rep. 2014, 3, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.; Parsaik, A.K.; Mielke, M.M.; Erwin, P.J.; Knopman, D.S.; Petersen, R.C.; Roberts, R.O. Association of Mediterranean diet with mild cognitive impairment and Alzheimer’s disease: A systematic review and meta-analysis. J. Alzheimer’s Dis. 2014, 39, 271–282. [Google Scholar]
- Cao, L.; Tan, L.; Wang, H.-F.; Jiang, T.; Zhu, X.-C.; Lu, H.; Tan, M.-S.; Yu, J.-T. Dietary patterns and risk of dementia: A systematic review and meta-analysis of cohort studies. Mol. Neurobiol. 2016, 53, 6144–6154. [Google Scholar] [CrossRef] [PubMed]
- Shim, J.-S.; Oh, K.; Kim, H.C. Dietary assessment methods in epidemiologic studies. Epidemiol. Health 2014, 36, e2014009. [Google Scholar] [CrossRef] [PubMed]
- Galasso, R.; Panico, S.; Celentano, E.; Del Pezzo, M. Relative validity of multiple telephone versus face-to-face 24-h dietary recalls. Ann. Epidemiol. 1994, 4, 332–336. [Google Scholar] [CrossRef]
- Prince, M.J. World Alzheimer Report 2015: The Global Impact of Dementia: An Analysis of Prevalence, Incidence, Cost and Trends; Alzheimer’s Disease International (ADI): London, UK, 2015. [Google Scholar]
- Roman, B.; Carta, L.; Martinez-Gonzalez, M.; Serra-Majem, L. Effectiveness of the Mediterranean diet in the elderly. Clin. Interv. Aging 2008, 3, 97–109. [Google Scholar] [PubMed]
- Castro-Quezada, I.; Román-Viñas, B.; Serra-Majem, L. The Mediterranean diet and nutritional adequacy: A review. Nutrients 2014, 6, 231–248. [Google Scholar] [CrossRef] [PubMed]
- Dangour, A.D.; Whitehouse, P.J.; Rafferty, K.; Mitchell, S.A.; Smith, L.; Hawkesworth, S.; Vellas, B. B-vitamins and fatty acids in the prevention and treatment of Alzheimer’s disease and dementia: A systematic review. J. Alzheimer’s Dis. 2010, 22, 205–224. [Google Scholar] [CrossRef] [PubMed]
- Grant, W.B. Does vitamin d reduce the risk of dementia? J. Alzheimer’s Dis. 2009, 17, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Mohajeri, M.H.; Troesch, B.; Weber, P. Inadequate supply of Vitamins and Dha in the elderly: Implications for brain aging and Alzheimer-type dementia. Nutrition 2015, 31, 261–275. [Google Scholar] [CrossRef] [PubMed]
- Morris, M. The role of nutrition in Alzheimer’s disease: Epidemiological evidence. Eur. J. Neurol. 2009, 16, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Nachum-Biala, Y.; Troen, A.M. B-vitamins for Neuroprotection: Narrowing the evidence gap. Biofactors 2012, 38, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Redman, K.; Ruffman, T.; Fitzgerald, P.; Skeaff, S. Iodine deficiency and the brain: Effects and mechanisms. Crit. Rev. Food Sci. Nutr. 2016, 56, 2695–2713. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, I.H. Effects of folate and vitamin b12 on cognitive function in adults and the elderly. Food Nutr. Bull. 2008, 29, S132–S142. [Google Scholar] [CrossRef] [PubMed]
- Yusufov, M.; Weyandt, L.L.; Piryatinsky, I. Alzheimer’s disease and diet: A systematic review. Int. J. Neurosci. 2017, 127, 161–175. [Google Scholar] [CrossRef] [PubMed]
- Germani, A.; Vitiello, V.; Giusti, A.M.; Pinto, A.; Donini, L.M.; del Balzo, V. Environmental and economic sustainability of the mediterranean diet. Int. J. Food Sci. Nutr. 2014, 65, 1008–1012. [Google Scholar] [CrossRef] [PubMed]
- Prince, M.; Albanese, E.; Guerchet, M.; Prina, M. Nutrition and Dementia: A Review of Available Research; Alzheimer’s Disease International: London, UK, 2014. [Google Scholar]
| Authors/Year/Country | Study Quality Score | Study Design | Participants (n; Age Mean, Mean ± SD, or Range) | Control Group | Intervention | Follow Up Period | Dietary Measure | Cognitive Outcome Measure (s) |
|---|---|---|---|---|---|---|---|---|
| Studies linking MD to cognitive function | ||||||||
| Chan et al., 2013 [34] China | 0 | Cross-sectional | 3670 men: 72 ± 4.8 years women: 71.6 ± 4.8 years | 0–9 MD score | CSI-D | |||
| Crichton et al., 2013 [35] Australia | 0 | Cross-sectional | 1183 50.6 ± 5.8 years | 0–9 MD score | CFQ and MFQ | |||
| Corley et al., 2013 [36] Scotland | 0 | Cross-sectional | 882 69.5 ± 0.8 years | 0–9 MD score | MMSE NART WTAR | |||
| Ye et al., 2013 [37] USA | + | Cross-sectional | 1269 57.3 ± 7.6 years | 0–9 MD score | MMSE | |||
| Katsiardanis et al., 2013 [38] Greece | + | Cross-sectional | 557 65+ years | 0–55 MD score | MMSE | |||
| Zbeida et al., 2014 [39] USA | 0 | Cross-sectional | 4577 NHANES 71.19 ± 7.78 years MABAT ZAHAV 74.9 ± 6.25 years | 0–9 MD score | Wechsler adult intelligence scale | |||
| Martínez-Lapiscina et al., 2013 [40] Spain | + | Randomized controlled trial | 522, high CVD risk age 74.6 ± 5.7 years | advised to reduce all types of fat | MD+ EVOO (1 L/week) or MD + 30 g/day of raw, unprocessed mixed nuts | 6.5 years | MMSE CDT | |
| Valls-Pedret et al., 2015 [41] Spain | + | Randomized controlled trial | 447 high CVD risk mean 66.8 years | advised to reduce dietary fat | MD + EVOO (1 L/week), or MD + mixed nuts (30 g/day) | 4.1 years Range (1.0–8.8) | MMSE RAVLT, Animals Semantic Fluency, Wechsler Adult Intelligence Scale, Wechsler Memory Scale, CTT | |
| Cherbuin et al., 2012 [42] Australia | + | Cohort | 1528 62.54 ± 1.52 years | 4 years | 0–9 MD score | International Consensus Criteria, CDR | ||
| Haring et al., 2016 [43] USA | + | Cohort | 6425 women 65–79 years | 9.11 years | 0–9 MD score | Consortium to Establish a Registry for Alzheimer’s Disease battery of neuropsychologic tests 3MS | ||
| Samieri et al., 2013 [44] USA | + | Cohort | 16,058 women 74.3 ± 2.3 years | 13 years | 0–9 MD score | TICS EBMT Delayed recall of the TICS 10-word list category fluency; digit span-backward | ||
| Samieri et al., 2013 [45] USA | 0 | Cohort | 6174 women 72 ± 4.1 years | 5 years | 0–9 MD score | TICS EBMT; Delayed recall of the TICS ten-word list Category fluency | ||
| Vercambre et al., 2012 [46] USA | 0 | Cohort | 2504 women with prevalent vascular disease or more than 3 coronary risk factors 71.9 ± 3.9 years low MD score 72.5 ± 4.3 years middle MD score 72.6 ± 4.0 years High MD score | 5.4 years (range 4.1–6.1) | 0–9 and 0–55 MD score | TICS 10-word list East Boston Memory Category fluency | ||
| Gardener et al., 2015 [47] Australia | 0 | Cohort | 527 69.3 ± 6.4 years | 3 years | 0–9 MD score | Battery assessed six cognitive domains (verbal memory, visual memory, executive function, language, attention and visuospatial functioning | ||
| Psaltopoulou et al., 2008 [48] Greece | 0 | Cohort | 743 >65 years | Median 8 years, range (6–13) | 0–9 MD score | MMSE | ||
| Qin et al., 2015 [49] China | + | Cohort | 1650 55+ years | 5.3 years | 0–9 MD score | Immediate and delayed recall of a 10-word list; counting backward and serial 7’s | ||
| Feart et al., 2009 [50] France | 0 | Cohort | 1410 75.9 (range, 67.7–94.9) years | 4.1 years | 0–9 MD score | BVRT FCSRT MMSE IST | ||
| Kesse-Guyot et al., 2013 [51] France | 0 | Cohort | 3083 52.0 ± 4.6 years | 13 years | 0–9 MD score 0–100 MSDPS | RI- 48 (Rappel indice’ (cued recall)-48 items) | ||
| Galbete et al., 2015 [52] Spain | 0 | Cohort | 823 62 ± 6 years | 8 years | 0–9 MD score | TICS-m | ||
| Koyama et al., 2015 [53] USA | + | Cohort | 2326 74.6 ± 2.9 years | 7.9 ± 0.1 years | 0–55 MD score | 3MS | ||
| Tangney et al., 2011 [54] USA | 0 | Cohort | 3790 75.4 ± 6.2 years | 7.6 years | 0–55 MD score | East Boston tests of immediate and delayed recall MMSE Symbol Digit Modalities Test | ||
| Tangney et al., 2014 [55] USA | 0 | Cohort | 826 81.5 ± 7.1 years | 4.1 years | 0–55 MD score | 19 cognitive tests | ||
| Trichopoulou et al., 2015 [56] Greece | 0 | Cohort | 401 mean = 74 years | 6.6 years | 0–9 MD score | MMSE | ||
| Wengreen et al., 2013 [57] USA | + | Cohort | 3831 73.8; 10.2 74.1; 10.2 74.4; 10.0 74.0; 9.7 74.2; 9.7 per 5 MD quintiles | 10.6 years | 0–9 MD score | 3MS | ||
| Tsivgoulis et al., 2013 [58] USA | + | Cohort | 17,478 64.4 ± 9.1 years | 4.0 ± 1.5 years | 0–9 MD score | SIS | ||
| Studies linking MD to AD development and mortality | ||||||||
| Gardener et al., 2012 [59] Australia | 0 | Cross-sectional | 970 71.72 ± 7.86 years | 0–9 MD score | MMSE Logical Memory II California Verbal Learning Test II Delis-Kaplan Executive Function System Verbal Fluency | |||
| Scarmeas et al., 2009 [60] USA | + | Cohort | 1393 76.9 ± 6.5 years | 4.3 ± 2.7 years | 0–9 MD score | Alzheimer’s incidence rate CDR | ||
| Olsson et al., 2015 [61] Sweden | + | Cohort | 1038 men 71 ± 0.6 years | 12 years | 0–8 MD score | Alzheimer’s incidence rate NINCDS-ADRDA DSM-IV criteria MMSE | ||
| Scarmeas et al., 2007 [62] USA | + | Cohort | 192 82.9 ± 7.7 years | 4.4 ± 3.6 years | 0-9 MD score | Mortality rate | ||
| Morris et al., 2015 [63] USA | 0 | Cohort | 923 58–98 years | Average 4.5 years | 0–55 MD score | Alzheimer’s incidence rate | ||
| Gu et al., 2010 [64] USA | 0 | Cohort | 1219 76.7 ± 6.4 years | 3.8 ± 1.3 years | 0–9 MD score | DSM III R NINCDS-ADRDA | ||
| Study | Results | Limitations |
|---|---|---|
| Chan et al., 2013 [34] | MD was not associated with cognitive function | Cross-sectional study (cannot establish causality); cognitive status was self-reported; participants were highly educated |
| Crichton et al., 2013 [35] | MD was not associated with cognitive function | Cross-sectional study (cannot establish causality; cognitive status was self-reported; inclusion exclusion criteria not clearly stated |
| Corley et al., 2013 [36] | MD was associated with improved cognitive function; however, the association was no longer significant after adjusting for childhood IQ and socio-economic status | Self-selecting sample; inclusion exclusion criteria not clearly stated; response rate not stated |
| Ye et al., 2013 [37] | MD was associated with improved MMSE scores (OR of cognitive impairment = 0.87, 95% CI (0.80–0.94) | Cross-sectional study (cannot establish causality) |
| Katsiardanis et al., 2013 [38] | MD was associated with improved MMSE scores in male; OR of cognitive impairment = 0.88, 95% CI (0.80–0.98); however, no significant association was observed among females | Cross-sectional study (cannot establish causality) |
| Zbeida et al., 2014 [39] | MD was associated with improved cognitive function in both NHANES and MABAT-ZAHAV cohorts, p < 0.001 and 0.008 respectively | Cross-sectional study (cannot establish causality); used a 24 h recall which does not represent usual intake; inclusion exclusion criteria not clearly stated; sources of funding not mentioned |
| Martínez-Lapiscina et al., 2013 [40] | MD + EVOO and MD + Nuts diet were significantly associated with better cognitive function. MD + EVOO group’s mean global cognitive function scores differences from the control group (+0.62, 95% CI (0.18–1.05), p = 0.005 for MMSE, and +0.51, 95% CI (0.20–0.82), p = 0.001 for CDT). MD + Nuts group’s differences from the control group +0.57, 95% CI (0.11–1.03), p = 0.015 for MMSE and +0.33, 95% CI (0.003–0.67), p = 0.048 for CDT) | Participants had CVD risk factors which may improve effect size seen; intervention was MD plus EVOO or nuts; no baseline data |
| Valls-Pedret et al., 2015 [41] | MD + EVOO was significantly associated with RAVLT and color trail test, p value = 0.04 and 0.045 respectively. MD + EVOO was not associated with other cognitive tests measured MD + Nuts was significantly associated with better composite memory, and MD + EVOO was was significantly associated with better global cognition | Participants had CVD risk factors; intervention was MD plus EVOO or nuts; funding may have caused a conflict of interest |
| Cherbuin et al., 2012 [42] | MD was not associated with cognitive function | No limitations identified |
| Haring et al., 2016 [43] | MD was not associated with cognitive function | Female participants only; baseline dietary data only |
| Samieri et al., 2013 [44] | MD was associated with cognitive function. Cross-sectional analysis showed that higher MD was associated with better TICS scores, global cognition and verbal memory, p = 0.004, 0.002 and <0.001 respectively. No effect of MD on cognitive function over time | Female only; highly educated; telephone assessment; unclear reliability |
| Samieri et al., 2013 [45] | MD was not associated with cognitive function | Female only; highly educated; telephone assessment |
| Vercambre et al., 2012 [46] | MD was not associated with cognitive function | Female participants only; participants had CVD risk factors; cognitive function assessed via telephone; baseline dietary data only |
| Gardener et al., 2015 [47] | MD was associated with executive cognitive function only among APOE allele carrier; change in cognitive function = 8.6%, p < 0.01 | Cohort medians for food intakes were used instead of traditional medians |
| Psaltopoulou et al., 2008 [48] | MD was not associated with cognitive function | No baseline data; exclusion criteria not clearly stated |
| Qin et al., 2015 [49] | MD was associated with slower cognitive decline, β = 0.042, 95% CI (0.002–0.081) | 24 h recall which does not represent usual intake |
| Feart et al., 2009 [50] | MD was associated with better cognition, MMSE errors, β = −0.006, 95% CI (−0.01, −0.0003) per 1-unit increase in MD score
MD was not associated with other cognitive tests (IST, BVRT, or FCSRT) and the risk of developing dementia | Selection bias, participants with missing data were significantly different than those with available data |
| Galbete et al., 2015 [51] | MD was associated with better cognition, higher MD scores had lower rates of cognitive decline, p = 0.011 | Participants were highly educated and did not represent general public |
| Kesse-Guyot et al., 2013 [52] | MD was associated with lower phonemic fluency score, p = 0.048 and a lower backward digit span score, p = 0.03 | No baseline data; low response rate |
| Koyama et al., 2015 [53] | MD was associated with better cognition among African-American participants, high MD was associated with better 3MS scores, difference = 0.22, 95% CI (0.05–0.39), p = 0.01 | African-Americans are at a higher risk of CVD |
| Tangney et al., 2011 [54] | MD was associated with better cognition, MD was associated with slower rates of cognitive decline β = 0.0014 per 1-point increase, p = 0.0004 | 24 h recall which does not represent usual intake |
| Tangney et al., 2014 [55] | MD was associated with better cognition. In linear analysis, MD was associated with better Global cognition, Episodic memory and Semantic memory, β = 0.002, 0.003, 0.003 and
p = 0.01, 0.02 and 0.02 respectively In the categorical analysis, MD was associated with better working memory, β = 0.033, p = 0.01 | Sample does not represent the general public |
| Trichopoulou et al., 2015 [56] | MD was associated with better cognition. Low MMSE scores were associated with low adherence to the MD; 20% of participants with low MMSE adhered well to the MD (MD score 6–9), as compared to a 41% in the high MMSE group. OR comparing high to low MD adherence was 0.46, 95 % CI (0.25–0.87) and 0.34, 95 % CI (0.13–0.89) for mild versus no decline and substantial versus no decline respectively. For highest MD scores OR = 0.46, 95 % CI (0.25–0.87) and 0.34, 95 % CI (0.13–0.89) for mild and substantial cognitive decline respectively | High rates of withdrawals |
| Wengreen et al., 2013 [57] | MD was associated with better cognition. Participants with the highest MD scores scored 1.4 times higher on the 3MS cognitive score, p = 0.0014 | No limitations identified |
| Tsivgoulis et al., 2013 [58] | MD was associated with better cognition only among non-diabetics. Higher MD scores were associated with lower risk of incident cognitive impairment, OR = 0.81, 95% CI (0.70–0.94) | Dietary intake was only assessed at baseline |
| Gardener et al., 2012 [59] | MD was associated with better cognition. Participants with AD and mild cognitive impairment had lower MD scores, p < 0.001 and <0.05 respectively. Each 1-unit increase was associated with 13–19% and 19–36% lower risk of being in the MCI and AD group respectively. MD was also linearly correlated with MMSE scores, p = 0.014. | Some under-reporting in FFQ; did not collect participant’s country of origin; inclusion exclusion criteria not clearly stated |
| Scarmeas et al., 2009 [60] | MD was associated with lower risk of developing AD. Participants within the middle and highest tertile of MD had a 45%, 95% CI (0.34–0.90) and 48%, 95% CI (0.53–0.95) of developing AD. | No limitations detected |
| Olsson et al., 2015 [61] | MD was not associated with cognitive function | Single 3-day food records with no follow-up data; male only |
| Scarmeas et al., 2007 [62] | MD was associated with lower mortality rates among AD patients; participants with highest adherence to MD had a mortality HR = 0.27, 95% CI (0.10–0.69) | Urban setting |
| Morris et al., 2015 [63] | MD was associated with lower risk of developing AD. Participants in the highest MD tertile had a HR of developing AD = 0.46, 95% CI (0.29, 0.74) | Participants were volunteers who are usually more health aware |
| Gu et al., 2010 [64] | MD was associated with lower risk of developing AD. Participants in the highest MD tertile had a 34% less risk of developing AD, p = 0.04 | Characteristics of participants that loss follow-up were different than those who remained; inclusion criteria was not clear |
| Cohort | Follow Up Time (Years) | Cohort | Follow Up Time (Years) |
|---|---|---|---|
| Studies linking MD to cognitive function | |||
| Studies that detected significance | Studies that didn’t detected significance | ||
| Tsivgoulis et al., 2013 [48] | 4 | Gardener et al., 2015 [47] | 3 |
| Tangney et al., 2014 [55] | 4.1 | Cherbuin et al., 2012 [42] | 4 |
| Feart et al., 2009 [50] | 4.1 | Samieri et al., 2013 [45] | 5 |
| Qin et al., 2015 [49] | 5.3 | Vercambre et al., 2012 [46] | 5.4 |
| Trichopoulou et al., 2015 [56] | 6.6 | Psaltopoulou et al., 2008 [48] | 8 |
| Tangney et al., 2011 [54] | 7.6 | Haring et al., 2016 [43] | 9.1 |
| Koyama et al., 2015 [53] | 7.9 | Samieri et al., 2013 [44] | 13 |
| Galbete et al., 2015 [52] | 8 | ||
| Wengreen et al., 2013 [57] | 10.6 | ||
| Kesse-Guyot et al., 2013 [51] | 13 | ||
| Studies linking MD to AD development and mortality | |||
| Studies that detected significance | Studies that didn’t detected significance | ||
| Gu et al., 2010 [64] | 3.8 | Olsson et al., 2015 [61] | 12 |
| Scarmeas et al., 2009 [60] | 4.3 | ||
| Scarmeas et al., 2007 [62] | 4.4 | ||
| Morris et al., 2015 [63] | 4.5 | ||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aridi, Y.S.; Walker, J.L.; Wright, O.R.L. The Association between the Mediterranean Dietary Pattern and Cognitive Health: A Systematic Review. Nutrients 2017, 9, 674. https://doi.org/10.3390/nu9070674
Aridi YS, Walker JL, Wright ORL. The Association between the Mediterranean Dietary Pattern and Cognitive Health: A Systematic Review. Nutrients. 2017; 9(7):674. https://doi.org/10.3390/nu9070674
Chicago/Turabian StyleAridi, Yasmine S., Jacqueline L. Walker, and Olivia R. L. Wright. 2017. "The Association between the Mediterranean Dietary Pattern and Cognitive Health: A Systematic Review" Nutrients 9, no. 7: 674. https://doi.org/10.3390/nu9070674
APA StyleAridi, Y. S., Walker, J. L., & Wright, O. R. L. (2017). The Association between the Mediterranean Dietary Pattern and Cognitive Health: A Systematic Review. Nutrients, 9(7), 674. https://doi.org/10.3390/nu9070674






