Fruits for Prevention and Treatment of Cardiovascular Diseases
Abstract
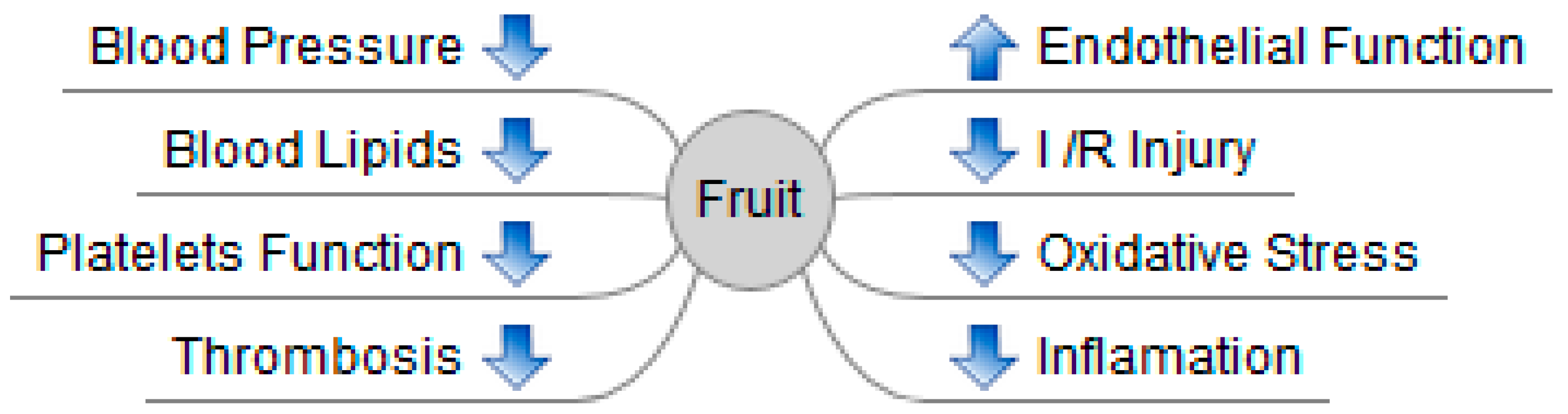
:1. Introduction
2. Epidemiological Studies
3. Experimental Studies
3.1. Grape
3.1.1. Protecting Endothelial Function
3.1.2. Decreasing Blood Lipids
3.1.3. Decreasing Blood Pressure
3.1.4. Suppressing Platelets Function
3.1.5. Alleviating Ischemia/Reperfusion Injury
3.1.6. Inhibiting Thrombosis
3.2. Blueberry
3.3. Pomegranate
3.4. Apple
3.5. Hawthorn
3.6. Avocado
3.7. Other Fruits
4. Clinical Trials
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- WHO. Cardiovascular Diseases (CVDs). Available online: http://www.who.int/cardiovascular_diseases/en/ (accessed on 13 March 2017).
- Rodríguez-Monforte, M.; Flores-Mateo, G.; Sánchez, E. Dietary patterns and CVD: A systematic review and meta-analysis of observational studies. Br. J. Nutr. 2015, 114, 1341–1359. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Marventano, S.; Yang, J.; Micek, A.; Pajak, A.; Scalfi, L.; Galvano, F.; Kales, S.N. A comprehensive meta-analysis on evidence of Mediterranean diet and cardiovascular disease: Are individual components equal? Crit. Rev. Food Sci. Nutr. 2017, 57, 3218–3232. [Google Scholar] [CrossRef] [PubMed]
- Du, H.D.; Li, L.M.; Bennett, D.; Guo, Y.; Key, T.J.; Bian, Z.; Sherliker, P.; Gao, H.Y.; Chen, Y.P.; Yang, L.; et al. Fresh fruit consumption and major cardiovascular disease in China. N. Engl. J. Med. 2016, 374, 1332–1343. [Google Scholar] [CrossRef] [PubMed]
- Lai, H.T.M.; Threapleton, D.E.; Day, A.J.; Williamson, G.; Cade, J.E.; Burley, V.J. Fruit intake and cardiovascular disease mortality in the UK Women’s Cohort Study. Eur. J. Epidemiol. 2015, 30, 1035–1048. [Google Scholar] [CrossRef] [PubMed]
- Hodgson, J.M.; Prince, R.L.; Woodman, R.J.; Bondonno, C.P.; Ivey, K.L.; Bondonno, N.; Rimm, E.B.; Ward, N.C.; Croft, K.D.; Lewis, J.R. Apple intake is inversely associated with all-cause and disease-specific mortality in elderly women. Br. J. Nutr. 2016, 115, 860–867. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Hayasaka, S.; Shibata, Y.; Ojima, T.; Saegusa, T.; Gotoh, T.; Ishikawa, S.; Nakamura, Y.; Kayaba, K. Frequency of citrus fruit intake is associated with the incidence of cardiovascular disease: The jichi medical school cohort study. J. Epidemiol. 2011, 21, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Ezzati, M.; Riboli, E. Behavioral and dietary risk factors for noncommunicable diseases. N. Engl. J. Med. 2013, 369, 954–964. [Google Scholar] [CrossRef] [PubMed]
- Quinones, M.; Guerrero, L.; Suarez, M.; Pons, Z.; Aleixandre, A.; Arola, L.; Muguerza, B. Low-molecular procyanidin rich grape seed extract exerts antihypertensive effect in males spontaneously hypertensive rats. Food Res. Int. 2013, 51, 587–595. [Google Scholar] [CrossRef]
- Xu, H.; Xu, H.E.; Ryan, D. A study of the comparative effects of hawthorn fruit compound and simvastatin on lowering blood lipid levels. Am. J. Chin. Med. 2009, 37, 903–908. [Google Scholar] [CrossRef] [PubMed]
- Lucas, E.A.; Li, W.J.; Peterson, S.K.; Brown, A.; Kuvibidila, S.; Perkins-Veazie, P.; Clarke, S.L.; Smith, B.J. Mango modulates body fat and plasma glucose and lipids in mice fed a high-fat diet. Br. J. Nutr. 2011, 106, 1495–1505. [Google Scholar] [CrossRef] [PubMed]
- Brasil, G.A.; Ronchi, S.N.; Do Nascimento, A.M.; de Lima, E.M.; Romao, W.; Da Costa, H.B.; Scherer, R.; Ventura, J.A.; Lenz, D.; Bissoli, N.S.; et al. Antihypertensive effect of carica papaya via a reduction in ACE activity and improved baroreflex. Planta Med. 2014, 80, 1580–1587. [Google Scholar] [CrossRef] [PubMed]
- Fu, L.; Xu, B.T.; Xu, X.R.; Gan, R.Y.; Zhang, Y.; Xia, E.Q.; Li, H.B. Antioxidant capacities and total phenolic contents of 62 fruits. Food Chem. 2011, 129, 345–350. [Google Scholar] [CrossRef]
- Zhang, Y.J.; Gan, R.Y.; Li, S.; Zhou, Y.; Li, A.N.; Xu, D.P.; Li, H.B. Antioxidant phytochemicals for the prevention and treatment of chronic diseases. Molecules 2015, 20, 21138–21156. [Google Scholar] [CrossRef] [PubMed]
- Song, F.L.; Gan, R.Y.; Zhang, Y.; Xiao, Q.; Kuang, L.; Li, H.B. Total phenolic contents and antioxidant capacities of selected chinese medicinal plants. Int. J. Mol. Sci. 2010, 11, 2362–2372. [Google Scholar] [CrossRef] [PubMed]
- Gan, R.Y.; Xu, X.R.; Song, F.L.; Kuang, L.; Li, H.B. Antioxidant activity and total phenolic content of medicinal plants associated with prevention and treatment of cardiovascular and cerebrovascular diseases. J. Med. Plants Res. 2010, 4, 2438–2444. [Google Scholar]
- Fu, L.; Xu, B.T.; Gan, R.Y.; Zhang, Y.; Xu, X.R.; Xia, E.Q.; Li, H.B. Total phenolic contents and antioxidant capacities of herbal and tea infusions. Int. J. Mol. Sci. 2011, 12, 2112–2124. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.J.; Deng, G.F.; Xu, X.R.; Wu, S.; Li, S.; Xia, E.Q.; Li, F.; Chen, F.; Ling, W.H.; Li, H.B. Antioxidant capacities, phenolic compounds and polysaccharide contents of 49 edible macro-fungi. Food Funct. 2012, 3, 1195–1205. [Google Scholar] [CrossRef] [PubMed]
- Deng, G.F.; Xu, X.R.; Zhang, Y.; Li, D.; Gan, R.Y.; Li, H.B. Phenolic compounds and bioactivities of pigmented rice. Crit. Rev. Food Sci. 2013, 53, 296–306. [Google Scholar] [CrossRef] [PubMed]
- Deng, G.F.; Lin, X.; Xu, X.R.; Gao, L.L.; Xie, J.F.; Li, H.B. Antioxidant capacities and total phenolic contents of 56 vegetables. J. Funct. Foods 2013, 5, 260–266. [Google Scholar] [CrossRef]
- Li, S.; Li, S.K.; Gan, R.Y.; Song, F.L.; Kuang, L.; Li, H.B. Antioxidant capacities and total phenolic contents of infusions from 223 medicinal plants. Ind. Crops Prod. 2013, 51, 289–298. [Google Scholar] [CrossRef]
- Li, A.N.; Li, S.; Li, H.B.; Xu, D.P.; Xu, X.R.; Chen, F. Total phenolic contents and antioxidant capacities of 51 edible and wild flowers. J. Funct. Foods 2014, 6, 319–330. [Google Scholar] [CrossRef]
- Xu, D.P.; Li, Y.; Meng, X.; Zhou, T.; Zhou, Y.; Zheng, J.; Zhang, J.J.; Li, H.B. Natural antioxidants in foods and medicinal plants: Extraction, assessment and resources. Int. J. Mol. Sci. 2017, 18, 96. [Google Scholar] [CrossRef] [PubMed]
- Fu, L.; Xu, B.T.; Xu, X.R.; Qin, X.S.; Gan, R.Y.; Li, H.B. Antioxidant capacities and total phenolic contents of 56 wild fruits from South China. Molecules 2010, 15, 8602–8617. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Scalbert, A.; Morand, C.; Remesy, C.; Jimenez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [PubMed]
- Ginter, E.; Simko, V. Plant polyphenols in prevention of heart disease. Bratisl. Med. J. 2012, 113, 476–480. [Google Scholar] [CrossRef]
- Li, A.N.; Li, S.; Zhang, Y.J.; Xu, X.R.; Chen, Y.M.; Li, H.B. Resources and biological activities of natural polyphenols. Nutrients 2014, 6, 6020–6047. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Zhang, X.; Gao, Y.T.; Li, H.; Yang, G.; Huang, J.; Zheng, W.; Xiang, Y.B.; Shu, X.O. Fruit and vegetable intake and risk of CHD: Results from prospective cohort studies of Chinese adults in Shanghai. Br. J. Nutr. 2014, 111, 353–362. [Google Scholar] [CrossRef] [PubMed]
- Gan, Y.; Tong, X.; Li, L.; Cao, S.; Yin, X.; Gao, C.; Herath, C.; Li, W.; Jin, Z.; Chen, Y.; et al. Consumption of fruit and vegetable and risk of coronary heart disease: A meta-analysis of prospective cohort studies. Int. J. Cardiol. 2015, 183, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Hansen, L.; Dragsted, L.O.; Olsen, A.; Christensen, J.; Tjonneland, A.; Schmidt, E.B.; Overvad, K. Fruit and vegetable intake and risk of acute coronary syndrome. Br. J. Nutr. 2010, 104, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Virtamo, J.; Wolk, A. Total and specific fruit and vegetable consumption and risk of stroke: A prospective study. Atherosclerosis 2013, 227, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Oude Griep, L.M.; Verschuren, W.M.M.; Kromhout, D.; Ocke, M.C.; Geleijnse, J.M. Colors of fruit and vegetables and 10-year incidence of stroke. Stroke 2011, 42, 3190–3195. [Google Scholar] [CrossRef] [PubMed]
- Oude Griep, L.M.; Verschuren, W.M.M.; Kromhout, D.; Ocke, M.C.; Geleijnse, J.M. Raw and processed fruit and vegetable consumption and 10-year stroke incidence in a population-based cohort study in the Netherlands. Eur. J. Clin. Nutr. 2011, 65, 791–799. [Google Scholar] [CrossRef] [PubMed]
- Cassidy, A.; Rimm, E.B.; O’Reilly, E.J.; Logroscino, G.; Kay, C.; Chiuve, S.E.; Rexrode, K.M. Dietary flavonoids and risk of stroke in women. Stroke 2012, 43, 946–975. [Google Scholar] [CrossRef] [PubMed]
- Goetz, M.E.; Judd, S.E.; Hartman, T.J.; McClellan, W.; Anderson, A.; Vaccarino, V. Flavanone intake is inversely associated with risk of incident ischemic stroke in the REasons for geographic and racial differences in stroke (REGARDS) study. J. Nutr. 2016, 146, 2233–2243. [Google Scholar] [CrossRef] [PubMed]
- Collins, R.; Peto, R.; MacMahon, S.; Hebert, P.; Fiebach, N.H.; Eberlein, K.A.; Godwin, J.; Qizilbash, N.; Taylor, J.O.; Hennekens, C.H. Blood pressure, stroke, and coronary heart disease. Part 2, Short-term reductions in blood pressure: Overview of randomised drug trials in their epidemiological context. Lancet 1990, 335, 827–838. [Google Scholar] [CrossRef]
- Wang, L.; Manson, J.E.; Gaziano, J.M.; Buring, J.E.; Sesso, H.D. Fruit and vegetable intake and the risk of hypertension in middle-aged and older women. Am. J. Hypertens. 2012, 25, 180–189. [Google Scholar] [CrossRef] [PubMed]
- Tsubota-Utsugi, M.; Ohkubo, T.; Kikuya, M.; Metoki, H.; Kurimoto, A.; Suzuki, K.; Fukushima, N.; Hara, A.; Asayama, K.; Satoh, H.; et al. High fruit intake is associated with a lower risk of future hypertension determined by home blood pressure measurement: The Ohasama study. J. Hum. Hypertens. 2011, 25, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Borgi, L.; Muraki, I.; Satija, A.; Willett, W.C.; Rimm, E.B.; Forman, J.P. Fruit and vegetable consumption and the incidence of hypertension in three prospective cohort studies. Hypertension 2016, 67, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Song, H.J.; Paek, Y.J.; Choi, M.K.; Lee, H.J. Gender differences in the relationship between risk of hypertension and fruit intake. Prev. Med. 2014, 67, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Chan, H.T.; Yiu, K.H.; Wong, C.Y.; Li, S.W.; Tam, S.; Tse, H.F. Increased dietary fruit intake was associated with lower burden of carotid atherosclerosis in Chinese patients with type 2 diabetes mellitus. Diabetic Med. 2013, 30, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Zhang, Y.; Ling, W.; Feng, D.; Wei, X.; Yang, C.; Ma, J. Fruit consumption is associated with lower carotid intima-media thickness and C-reactive protein levels in patients with type 2 diabetes mellitus. J. Am. Diet Assoc. 2011, 111, 1536–1542. [Google Scholar] [CrossRef] [PubMed]
- Cassidy, A.; Bertoia, M.; Chiuve, S.; Flint, A.; Forman, J.; Rimm, E.B. Habitual intake of anthocyanins and flavanones and risk of cardiovascular disease in men. Am. J. Clin. Nutr. 2016, 104, 587–594. [Google Scholar] [CrossRef] [PubMed]
- Li, G.L.; Zhu, Y.N.; Zhang, Y.; Lang, J.; Chen, Y.M.; Ling, W.H. Estimated daily flavonoid and stilbene intake from fruits, vegetables, and nuts and associations with lipid profiles in chinese adults. J. Acad. Nutr. Diet. 2013, 113, 786–794. [Google Scholar] [CrossRef] [PubMed]
- Bendinelli, B.; Masala, G.; Saieva, C.; Salvini, S.; Calonico, C.; Sacerdote, C.; Agnoli, C.; Grioni, S.; Frasca, G.; Mattiello, A.; et al. Fruit, vegetables, and olive oil and risk of coronary heart disease in Italian women: The EPICOR study. Am. J. Clin. Nutr. 2011, 93, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Dauchet, L.; Montaye, M.; Ruidavets, J.; Arveiler, D.; Kee, F.; Bingham, A.; Ferrieres, J.; Haas, B.; Evans, A.; Ducimetiere, P.; et al. Association between the frequency of fruit and vegetable consumption and cardiovascular disease in male smokers and non-smokers. Eur. J. Clin. Nutr. 2010, 64, 578–586. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Vik, S.; Kolonel, L.N. Fruit and vegetable consumption, ethnicity and risk of fatal ischemic heart disease. J. Nutr. Health Aging 2014, 18, 573–578. [Google Scholar] [CrossRef] [PubMed]
- Sangita, S.; Vik, S.A.; Pakseresht, M.; Kolonel, L.N. Adherence to recommendations for fruit and vegetable intake, ethnicity and ischemic heart disease mortality. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 1247–1254. [Google Scholar] [CrossRef] [PubMed]
- Rautiainen, S.; Levitan, E.B.; Mittleman, M.A.; Wolk, A. Fruit and vegetable intake and rate of heart failure: A population-based prospective cohort of women. Eur. J. Heart Fail. 2015, 17, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Deanfield, J.; Donald, A.; Ferri, C.; Giannattasio, C.; Halcox, J.; Halligan, S.; Lerman, A.; Mancia, G.; Oliver, J.J.; Pessina, A.C.; et al. Endothelial function and dysfunction. Part I: Methodological issues for assessment in the different vascular beds: A statement by the working group on endothelin and endothelial factors of the European Society of Hypertension. J. Hypertens. 2005, 23, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Thandapilly, S.J.; LeMaistre, J.L.; Louis, X.L.; Anderson, C.M.; Netticadan, T.; Anderson, H.D. Vascular and cardiac effects of grape powder in the spontaneously hypertensive rat. Am. J. Hypertens. 2012, 25, 1070–1076. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Wang, J.; Gao, H.Q.; Wang, Q.Z.; Zhang, J.; Qiu, J. Beneficial effects of grape seed proanthocyanidin extract on arterial remodeling in spontaneously hypertensive rats via protecting against oxidative stress. Mol. Med. Rep. 2016, 14, 3711–3718. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.H.; Huang, L.L.; Yu, T.T.; Zhu, J.H.; Shen, B.; Zhang, Y.; Wang, H.Z.; Gao, S. Effects of oligomeric grape seed proanthocyanidins on heart, aorta, kidney in DOCA-salt mice: Role of oxidative stress. Phytother. Res. 2013, 27, 869–876. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Rodriguez, R.; Justo, M.L.; Claro, C.M.; Vila, E.; Parrado, J.; Herrera, M.D.; de Sotomayor, M.A. Endothelium-dependent vasodilator and antioxidant properties of a novel enzymatic extract of grape pomace from wine industrial waste. Food Chem. 2012, 135, 1044–1051. [Google Scholar] [CrossRef] [PubMed]
- Felice, F.; Zambito, Y.; Di Colo, G.; D’Onofrio, C.; Fausto, C.; Balbarini, A.; Di Stefano, R. Red grape skin and seeds polyphenols: Evidence of their protective effects on endothelial progenitor cells and improvement of their intestinal absorption. Eur. J. Pharm. Biopharm. 2012, 80, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Quintieri, A.M.; Baldino, N.; Filice, E.; Seta, L.; Vitetti, A.; Tota, B.; De Cindio, B.; Cerra, M.C.; Angelone, T. Malvidin, a red wine polyphenol, modulates mammalian myocardial and coronary performance and protects the heart against ischemia/reperfusion injury. J. Nutr. Biochem. 2013, 24, 1221–1231. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Liu, X.; Feng, H.; Zhao, S.; Gao, H. Grape seed proanthocyanidin extracts enhance endothelial nitric oxide synthase expression through 5′-AMP activated protein kinase/surtuin 1-krupple like factor 2 pathway and modulate blood pressure in ouabain induced hypertensive rats. Biol. Pharm. Bull. 2012, 35, 2192–2197. [Google Scholar] [CrossRef] [PubMed]
- Goutzourelas, N.; Stagos, D.; Housmekeridou, A.; Karapouliou, C.; Kerasioti, E.; Aligiannis, N.; Skaltsounis, A.L.; Spandidos, D.A.; Tsatsakis, A.M.; Kouretas, D. Grape pomace extract exerts antioxidant effects through an increase in GCS levels and GST activity in muscle and endothelial cells. Int. J. Mol. Med. 2015, 36, 433–441. [Google Scholar] [CrossRef] [PubMed]
- Luzak, B.; Kosiorek, A.; Syska, K.; Rozalski, M.; Bijak, M.; Podsedek, A.; Balcerczak, E.; Watala, C.; Golanski, J. Does grape seed extract potentiate the inhibition of platelet reactivity in the presence of endothelial cells? Adv. Med. Sci. 2014, 59, 178–182. [Google Scholar] [CrossRef] [PubMed]
- Razavi, S.M.; Gholamin, S.; Eskandari, A.; Mohsenian, N.; Ghorbanihaghjo, A.; Delazar, A.; Rashtchizadeh, N.; Keshtkar-Jahromi, M.; Argani, H. Red grape seed extract improves lipid profiles and decreases oxidized low-density lipoprotein in patients with mild hyperlipidemia. J. Med. Food 2013, 16, 255–258. [Google Scholar] [CrossRef] [PubMed]
- Leibowitz, A.; Faltin, Z.; Perl, A.; Eshdat, Y.; Hagay, Y.; Peleg, E.; Grossman, E. Red grape berry-cultured cells reduce blood pressure in rats with metabolic-like syndrome. Eur. J. Nutr. 2014, 53, 973–980. [Google Scholar] [CrossRef] [PubMed]
- Caimari, A.; Del Bas, J.M.; Crescenti, A.; Arola, L. Low doses of grape seed procyanidins reduce adiposity and improve the plasma lipid profile in hamsters. Int. J. Obes. 2013, 37, 576–583. [Google Scholar] [CrossRef] [PubMed]
- Eren, E.; Yilmaz, N.; Aydin, O. High density lipoprotein and it’s dysfunction. Open Biochem. J. 2012, 6, 78–93. [Google Scholar] [CrossRef] [PubMed]
- Zagayko, A.L.; Kravchenko, G.B.; Krasilnikova, O.A.; Ogai, Y.O. Grape polyphenols increase the activity of HDL enzymes in old and obese rats. Oxid. Med. Cell. Longev. 2013, 593761. [Google Scholar] [CrossRef] [PubMed]
- Malinowska, J.; Oleszek, W.; Stochmal, A.; Olas, B. The polyphenol-rich extracts from black chokeberry and grape seeds impair changes in the platelet adhesion and aggregation induced by a model of hyperhomocysteinemia. Eur. J. Nutr. 2013, 52, 1049–1057. [Google Scholar] [CrossRef] [PubMed]
- Safwen, K.; Selima, S.; Mohamed, E.; Ferid, L.; Pascal, C.; Mohamed, A.; Ezzedine, A.; Meherzia, M. Protective effect of grape seed and skin extract on cerebral ischemia in rat: Implication of transition metals. Int. J. Stroke 2015, 10, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Gao, H.; Qiu, J.; Lu, W.; Wei, X. The molecular mechanism of protective effects of grape seed proanthocyanidin extract on reperfusion arrhythmias in rats in vivo. Biol. Pharm. Bull. 2010, 33, 759–767. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Shi, H.; Wang, W.; Ke, Z.; Xu, P.; Zhong, Z.; Li, X.; Wang, S. Antithrombotic effect of grape seed proanthocyanidins extract in a rat model of deep vein thrombosis. J. Vasc. Surg. 2011, 53, 743–753. [Google Scholar] [CrossRef] [PubMed]
- Stroher, D.J.; Escobar, P.J.C.; Gullich, A.A.; Pilar, B.C.; Coelho, R.P.; Bruno, J.B.; Faoro, D.; Manfredini, V. 14 Days of supplementation with blueberry extract shows anti-atherogenic properties and improves oxidative parameters in hypercholesterolemic rats model. Int. J. Food Sci. Nutr. 2015, 66, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Mateos, A.; Ishisaka, A.; Mawatari, K.; Vidal-Diez, A.; Spencer, J.P.; Terao, J. Blueberry intervention improves vascular reactivity and lowers blood pressure in high-fat-, high-cholesterol-fed rats. Br. J. Nutr. 2013, 109, 1746–1754. [Google Scholar] [CrossRef] [PubMed]
- Xie, C.; Kang, J.; Chen, J.R.; Nagarajan, S.; Badger, T.M.; Wu, X. Phenolic acids are in vivo atheroprotective compounds appearing in the serum of rats after blueberry consumption. J. Agric. Food Chem. 2011, 59, 10381–10387. [Google Scholar] [CrossRef] [PubMed]
- Cao, K.; Xu, J.; Pu, W.J.; Dong, Z.Z.; Sun, L.; Zang, W.J.; Gao, F.; Zhang, Y.; Feng, Z.H.; Liu, J.K. Punicalagin, an active component in pomegranate, ameliorates cardiac mitochondrial impairment in obese rats via AMPK activation. Sci. Rep. 2015, 5, 14014. [Google Scholar] [CrossRef] [PubMed]
- Al-Jarallah, A.; Igdoura, F.; Zhang, Y.; Tenedero, C.B.; White, E.J.; MacDonald, M.E.; Igdoura, S.A.; Trigatti, B.L. The effect of pomegranate extract on coronary artery atherosclerosis in SR-BI/APOE double knockout mice. Atherosclerosis 2013, 228, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.Y.; Yan, C.H.; Frost, B.; Wang, X.; Hou, C.; Zeng, M.Q.; Gao, H.L.; Kang, Y.M.; Liu, J.K. Pomegranate extract decreases oxidative stress and alleviates mitochondrial impairment by activating AMPK-Nrf2 in hypothalamic paraventricular nucleus of spontaneously hypertensive rats. Sci. Rep. 2016, 6, 34246. [Google Scholar] [CrossRef] [PubMed]
- Hajipour, S.; Sarkaki, A.; Mohammad, S.; Mansouri, T.; Pilevarian, A.; RafieiRad, M. Motor and cognitive deficits due to permanent cerebral hypoperfusion/ischemia improve by pomegranate seed extract in rats. Pak. J. Biol. Sci. 2014, 17, 991–998. [Google Scholar] [PubMed]
- Serra, A.T.; Rocha, J.; Sepodes, B.; Matias, A.A.; Feliciano, R.P.; de Carvalho, A.; Bronze, M.R.; Duarte, C.M.M.; Figueira, M.E. Evaluation of cardiovascular protective effect of different apple varieties—Correlation of response with composition. Food Chem. 2012, 135, 2378–2386. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, J.; Donoso, W.; Sandoval, N.; Reyes, M.; Gonzalez, P.; Gajardo, M.; Morales, E.; Neira, A.; Razmilic, I.; Yuri, J.A.; et al. Apple peel supplemented diet reduces parameters of metabolic syndrome and atherogenic progression in ApoE−/− mice. Evid. Based Complement. Altern. 2015, 918384. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.J.; Luo, X.G.; Dong, Q.Q.; Mu, A.; Shi, G.L.; Wang, Q.T.; Chen, X.Y.; Zhou, H.; Zhang, T.C.; Pan, L.W. Ethanol extract of Zhongtian hawthorn lowers serum cholesterol in mice by inhibiting transcription of 3-hydroxy-3-methylglutaryl-CoA reductase via nuclear factor-kappa B signal pathway. Exp. Biol. Med. 2016, 241, 667–674. [Google Scholar] [CrossRef] [PubMed]
- Li, T.P.; Zhu, R.G.; Dong, Y.P.; Liu, Y.H.; Li, S.H.; Chen, G. Effects of pectin pentaoligosaccharide from hawthorn (Crataegus pinnatifida Bunge. Var. Major) on the activity and mRNA levels of enzymes involved in fatty acid oxidation in the liver of mice fed a high-fat diet. J. Agric. Food Chem. 2013, 61, 7599–7605. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.Y.; Zhang, L.; Geng, Y.; Geng, Y.H. Hawthorn fruit attenuates atherosclerosis by improving the hypolipidemic and antioxidant activities in apolipoprotein E-Deficient mice. J. Atheroscler. Thromb. 2014, 21, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Liang, R.; Wang, L.; Yan, R.; Hou, R.; Gao, S.; Yang, B. Effects of an aqueous extract of Crataegus pinnatifida Bge. Var. Major N.E.Br. fruit on experimental atherosclerosis in rats. J. Ethnopharmacol. 2013, 148, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Duarte, P.F.; Chaves, M.A.; Borges, C.D.; Mendonca, C. Avocado: Characteristics, health benefits and uses. Cienc. Rural 2016, 46, 747–754. [Google Scholar] [CrossRef]
- Villa-Rodriguez, J.A.; Molina-Corral, F.J.; Ayala-Zavala, J.F.; Olivas, G.I.; Gonzalez-Aguilar, G.A. Effect of maturity stage on the content of fatty acids and antioxidant activity of ‘Hass’ avocado. Food Res. Int. 2011, 44, 1231–1237. [Google Scholar] [CrossRef]
- Rodriguez-Sanchez, D.G.; Flores-Garcia, M.; Silva-Platas, C.; Rizzo, S.; Torre-Amione, G.; De la Pena-Diaz, A.; Hernandez-Brenes, C.; Garcia-Rivas, G. Isolation and chemical identification of lipid derivatives from avocado (Persea americana) pulp with antiplatelet and antithrombotic activities. Food Funct. 2015, 6, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Carvajal-Zarrabal, O.; Nolasco-Hipolito, C.; Aguilar-Uscanga, M.G.; Melo-Santiesteban, G.; Hayward-Jones, P.M.; Barradas-Dermitz, D.M. Avocado oil supplementation modifies cardiovascular risk profile markers in a rat model of sucrose-induced metabolic changes. Dis. Markers 2014, 2014, 386425. [Google Scholar] [CrossRef] [PubMed]
- Carvajal-Zarrabal, O.; Nolasco-Hipolito, C.; Aguilar-Uscanga, M.G.; Santiesteban, G.M.; Hayward-Jones, P.M.; Barradas-Dermitz, D.M. Effect of dietary intake of avocado oil and olive oil on biochemical markers of liver function in sucrose-fed rats. Biomed. Res. Int. 2014, 595479. [Google Scholar] [CrossRef] [PubMed]
- Dabas, D.; Shegog, R.M.; Ziegler, G.R.; Lambert, J.D. Avocado (Persea americana) seed as a source of bioactive phytochemicals. Curr. Pharm. Des. 2013, 19, 6133–6140. [Google Scholar] [CrossRef] [PubMed]
- Czompa, A.; Gyongyosi, A.; Czegledi, A.; Csepanyi, E.; Bak, I.; Haines, D.D.; Tosaki, A.; Lekli, I. Cardioprotection afforded by sour cherry seed kernel: The role of heme oxygenase-1. J. Cardiovasc. Pharmacol. 2014, 64, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; He, J.; Gao, Y.; Xing, Y.; Hou, J.; Tian, J. Preventive effect of total flavones of Choerospondias axillaries on ischemia/reperfusion-induced myocardial infarction-related MAPK signaling pathway. Cardiovasc. Toxicol. 2014, 14, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Zapata-Sudo, G.; Da, S.J.; Pereira, S.L.; Souza, P.J.; de Moura, R.S.; Sudo, R.T. Oral treatment with Euterpe oleracea Mart. (acai) extract improves cardiac dysfunction and exercise intolerance in rats subjected to myocardial infarction. BMC Complement. Altern. Med. 2014, 14, 227. [Google Scholar] [CrossRef] [PubMed]
- Mauray, A.; Felgines, C.; Morand, C.; Mazur, A.; Scalbert, A.; Milenkovic, D. Bilberry anthocyanin-rich extract alters expression of genes related to atherosclerosis development in aorta of apo E-deficient mice. Nutr. Metab. Cardiovasc. Dis. 2012, 22, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Ash, M.M.; Wolford, K.A.; Carden, T.J.; Hwang, K.T.; Carr, T.P. Unrefined and refined black raspberry seed oils significantly lower triglycerides and moderately affect cholesterol metabolism in male Syrian hamsters. J. Med. Food 2011, 14, 1032–1038. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Suo, Y.R.; Chen, D.L.; Tong, L. Protection against vascular endothelial dysfunction by polyphenols in sea buckthorn berries in rats with hyperlipidemia. Biosci. Trends 2016, 10, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, Y.; Hayashida, A.; Tsurushima, K.; Nagai, R.; Yoshitomi, M.; Daiguji, N.; Sakashita, N.; Takeya, M.; Tsukamoto, S.; Ikeda, T. Triterpenoids isolated from Zizyphus jujuba inhibit foam cell formation in macrophages. J. Agric. Food Chem. 2011, 59, 4544–4552. [Google Scholar] [CrossRef] [PubMed]
- Ono, M.; Yasuda, S.; Komatsu, H.; Fujiwara, Y.; Takeya, M.; Nohara, T. Triterpenoids from the fruits and leaves of the blackberry (Rubus allegheniensis) and their inhibitory activities on foam cell formation in human monocyte-derived macrophage. Nat. Prod. Res. 2014, 28, 2347–2350. [Google Scholar] [CrossRef] [PubMed]
- Konta, E.M.; Almeida, M.R.; Do, A.C.; Darin, J.D.; de Rosso, V.V.; Mercadante, A.Z.; Antunes, L.M.; Bianchi, M.L. Evaluation of the antihypertensive properties of yellow passion fruit pulp (Passiflora edulis Sims f. Flavicarpa Deg.) in spontaneously hypertensive rats. Phytother. Res. 2014, 28, 28–32. [Google Scholar] [CrossRef] [PubMed]
- Furuuchi, R.; Sakai, H.; Hirokawa, N.; Watanabe, Y.; Yokoyama, T.; Hirayama, M. Antihypertensive effect of boysenberry seed polyphenols on spontaneously hypertensive rats and identification of orally absorbable proanthocyanidins with vasorelaxant activity. Biosci. Biotechnol. Biochem. 2012, 76, 1694–1701. [Google Scholar] [CrossRef] [PubMed]
- De Souza, M.O.; Souza, E.S.L.; de Brito, M.C.; de Figueiredo, B.B.; Costa, D.C.; Silva, M.E.; Pedrosa, M.L. The hypocholesterolemic activity of acai (Euterpe oleracea Mart.) is mediated by the enhanced expression of the ATP-binding cassette, subfamily G transporters 5 and 8 and low-density lipoprotein receptor genes in the rat. Nutr. Res. 2012, 32, 976–984. [Google Scholar] [CrossRef] [PubMed]
- Pujari, R.R.; Vyawahare, N.S.; Kagathara, V.G. Evaluation of antioxidant and neuroprotective effect of date palm (Phoenix dactylifera L.) against bilateral common carotid artery occlusion in rats. Indian J. Exp. Biol. 2011, 49, 627–633. [Google Scholar] [PubMed]
- Varela, C.E.; Fromentin, E.; Roller, M.; Villarreal, F.; Ramirez-Sanchez, I. Effects of a natural extract of Aronia melanocarpa berry on endothelial cell nitric oxide production. J. Food Biochem. 2016, 40, 404–410. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.Z.; Le, K.; Li, W.D.; Ren, S.; Moghadasian, M.H.; Beta, T.; Shen, G.X. Effects of Saskatoon berry powder on monocyte adhesion to vascular wall of leptin receptor-deficient diabetic mice. J. Nutr. Biochem. 2014, 25, 851–857. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.Z.; Xie, X.P.; Le, K.; Li, W.D.; Moghadasian, M.H.; Beta, T.; Shen, G.X. Endoplasmic reticulum stress in diabetic mouse or glycated LDL-treated endothelial cells: Protective effect of Saskatoon berry powder and cyanidin glycans. J. Nutr. Biochem. 2015, 26, 1248–1253. [Google Scholar] [CrossRef] [PubMed]
- Torres-Urrutia, C.; Guzman, L.; Schmeda-Hirschmann, G.; Moore-Carrasco, R.; Alarcon, M.; Astudillo, L.; Gutierrez, M.; Carrasco, G.; Yuri, J.A.; Aranda, E.; et al. Antiplatelet, anticoagulant, and fibrinolytic activity in vitro of extracts from selected fruits and vegetables. Blood Coagul. Fibrinolysis 2011, 22, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Kono, R.; Okuno, Y.; Nakamura, M.; Inada, K.; Tokuda, A.; Yamashita, M.; Hidaka, R.; Utsunomiya, H. Peach (Prunus persica) extract inhibits angiotensin II-induced signal transduction in vascular smooth muscle cells. Food Chem. 2013, 139, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Isaak, C.K.; Petkau, J.C.; O, K.; Debnath, S.C.; Siow, Y.L. Manitoba Lingonberry (Vaccinium vitis-idaea) bioactivities in ischemia-reperfusion injury. J. Agric. Food Chem. 2015, 63, 5660–5669. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.G.; Kim, B.; Yang, Y.; Pham, T.X.; Park, Y.K.; Manatou, J.E.; Koo, S.I.; Chun, O.K.; Lee, J.Y. Berry anthocyanins suppress the expression and secretion of proinflammatory mediators in macrophages by inhibiting nuclear translocation of NF-kappa B independent of NRF2-mediated mechanism. J. Nutr. Biochem. 2014, 25, 404–411. [Google Scholar] [CrossRef] [PubMed]
- Rosenblat, M.; Volkova, N.; Borochov-Neori, H.; Judeinstein, S.; Aviram, M. Anti-atherogenic properties of date vs. Pomegranate polyphenols: The benefits of the combination. Food Funct. 2015, 6, 1496–1509. [Google Scholar] [CrossRef] [PubMed]
- Li, S.H.; Zhao, P.; Tian, H.B.; Chen, L.H.; Cui, L.Q. Effect of grape polyphenols on blood pressure: A meta-analysis of randomized controlled trials. PLoS ONE 2015, 10, 0137665. [Google Scholar] [CrossRef] [PubMed]
- Evans, M.; Wilson, D.; Guthrie, N. A randomized, double-blind, placebo-controlled, pilot study to evaluate the effect of whole grape extract on antioxidant status and lipid profile. J. Funct. Foods 2014, 7, 680–691. [Google Scholar] [CrossRef]
- Rahbar, A.R.; Mahmoudabadi, M.; Islam, M.S. Comparative effects of red and white grapes on oxidative markers and lipidemic parameters in adult hypercholesterolemic humans. Food Funct. 2015, 6, 1992–1998. [Google Scholar] [CrossRef] [PubMed]
- Yubero, N.; Sanz-Buenhombre, M.; Guadarrama, A.; Villanueva, S.; Carrion, J.M.; Larrarte, E.; Moro, C. LDL cholesterol-lowering effects of grape extract used as a dietary supplement on healthy volunteers. Int. J. Food Sci. Nutr. 2013, 64, 400–406. [Google Scholar] [CrossRef] [PubMed]
- Li, S.H.; Tian, H.B.; Zhao, H.J.; Chen, L.H.; Cui, L.Q. The acute effects of grape polyphenols supplementation on endothelial function in adults: Meta-Analyses of controlled trials. PLoS ONE 2013, 8, 0069818. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Suarez, J.M.; Giampieri, F.; Tulipani, S.; Casoli, T.; Di Stefano, G.; Gonzalez-Paramas, A.M.; Santos-Buelga, C.; Busco, F.; Quiles, J.L.; Cordero, M.D.; et al. One-month strawberry-rich anthocyanin supplementation ameliorates cardiovascular risk, oxidative stress markers and platelet activation in humans. J. Nutr. Biochem. 2014, 25, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Basu, A.; Betts, N.M.; Nguyen, A.; Newman, E.D.; Fu, D.; Lyons, T.J. Freeze-dried strawberries lower serum cholesterol and lipid peroxidation in adults with abdominal adiposity and elevated serum lipids. J. Nutr. 2014, 144, 830–837. [Google Scholar] [CrossRef] [PubMed]
- Basu, A.; Fu, D.X.; Wilkinson, M.; Simmons, B.; Wu, M.; Betts, N.M.; Du, M.; Lyons, T.J. Strawberries decrease atherosclerotic markers in subjects with metabolic syndrome. Nutr. Res. 2010, 30, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Ellis, C.L.; Edirisinghe, I.; Kappagoda, T.; Burton-Freeman, B. Attenuation of meal-induced inflammatory and thrombotic responses in overweight men and women after 6-week daily strawberry (Fragaria) intake-a randomized placebo-controlled trial. J. Atheroscler. Thromb. 2011, 18, 318–327. [Google Scholar] [CrossRef] [PubMed]
- Burton-Freeman, B.; Linares, A.; Hyson, D.; Kappagoda, T. Strawberry modulates LDL oxidation and postprandial lipemia in response to high-fat meal in overweight hyperlipidemic men and women. J. Am. Coll. Nutr. 2010, 29, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Udani, J.K.; Singh, B.B.; Singh, V.J.; Barrett, M.L. Effects of acai (Euterpe oleracea Mart.) berry preparation on metabolic parameters in a healthy overweight population: A pilot study. Nutr. J. 2011, 10, 45. [Google Scholar] [CrossRef] [PubMed]
- Kianbakht, S.; Abasi, B.; Dabaghian, F.H. Improved lipid profile in hyperlipidemic patients taking vaccinium arctostaphylos fruit hydroalcoholic extract: A randomized double-blind placebo-controlled clinical trial. Phytother. Res. 2014, 28, 432–436. [Google Scholar] [CrossRef] [PubMed]
- Larmo, P.S.; Kangas, A.J.; Soininen, P.; Lehtonen, H.M.; Suomela, J.P.; Yang, B.; Viikari, J.; Ala-Korpela, M.; Kallio, H.P. Effects of sea buckthorn and bilberry on serum metabolites differ according to baseline metabolic profiles in overweight women: A randomized crossover trial. Am. J. Clin. Nutr. 2013, 98, 941–951. [Google Scholar] [CrossRef] [PubMed]
- Lehtonen, H.M.; Suomela, J.P.; Tahvonen, R.; Yang, B.; Venojarvi, M.; Viikari, J.; Kallio, H. Different berries and berry fractions have various but slightly positive effects on the associated variables of metabolic diseases on overweight and obese women. Eur. J. Clin. Nutr. 2011, 65, 394–401. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Xia, M.; Ma, J.; Hao, Y.T.; Liu, J.; Mou, H.; Cao, L.; Ling, W.H. Anthocyanin supplementation improves serum LDL- and HDL-cholesterol concentrations associated with the inhibition of cholesteryl ester transfer protein in dyslipidemic subjects. Am. J. Clin. Nutr. 2009, 90, 485–492. [Google Scholar] [CrossRef] [PubMed]
- Chai, S.C.; Hooshmand, S.; Saadat, R.L.; Payton, M.E.; Brummel-Smith, K.; Arjmandi, B.H. Daily apple versus dried plum: Impact on cardiovascular disease risk factors in postmenopausal women. J. Acad. Nutr. Diet. 2012, 112, 1158–1168. [Google Scholar] [CrossRef] [PubMed]
- Tenore, G.C.; Caruso, D.; Buonomo, G.; D’Urso, E.; D’Avino, M.; Campiglia, P.; Marinelli, L.; Novellino, E. Annurca (Malus pumila Miller cv. Annurca) apple as a functional food for the contribution to a healthy balance of plasma cholesterol levels: Results of a randomized clinical trial. J. Sci. Food Agric. 2017, 97, 2107–2115. [Google Scholar] [CrossRef] [PubMed]
- Ravn-Haren, G.; Dragsted, L.O.; Buch-Andersen, T.; Jensen, E.N.; Jensen, R.I.; Nemeth-Balogh, M.; Paulovicsova, B.; Bergstrom, A.; Wilcks, A.; Licht, T.R.; et al. Intake of whole apples or clear apple juice has contrasting effects on plasma lipids in healthy volunteers. Eur. J. Nutr. 2013, 52, 1875–1889. [Google Scholar] [CrossRef] [PubMed]
- Bondonno, C.P.; Yang, X.; Croft, K.D.; Considine, M.J.; Ward, N.C.; Rich, L.; Puddey, I.B.; Swinny, E.; Mubarak, A.; Hodgson, J.M. Flavonoid-rich apples and nitrate-rich spinach augment nitric oxide status and improve endothelial function in healthy men and women: A randomized controlled trial. Free Radic. Biol. Med. 2012, 52, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Hollands, W.J.; Hart, D.J.; Dainty, J.R.; Hasselwander, O.; Tiihonen, K.; Wood, R.; Kroon, P.A. Bioavailability of epicatechin and effects on nitric oxide metabolites of an apple flavanol-rich extract supplemented beverage compared to a whole apple puree: A randomized, placebo-controlled, crossover trial. Mol. Nutr. Food Res. 2013, 57, 1209–1217. [Google Scholar] [CrossRef] [PubMed]
- Stonehouse, W.; Gammon, C.S.; Beck, K.L.; Conlon, C.A.; von Hurst, P.R.; Kruger, R. Kiwifruit: Our daily prescription for health. Can. J. Physiol. Pharm. 2013, 91, 442–447. [Google Scholar] [CrossRef] [PubMed]
- Gammon, C.S.; Kruger, R.; Conlon, C.A.; von Hurst, P.R.; Jones, B.; Stonehouse, W. Inflammatory status modulates plasma lipid and inflammatory marker responses to kiwifruit consumption in hypercholesterolaemic men. Nutr. Metab. Cardiovas. 2014, 24, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Gammon, C.S.; Kruger, R.; Brown, S.J.; Conlon, C.A.; von Hurst, P.R.; Stonehouse, W. Daily kiwifruit consumption did not improve blood pressure and markers of cardiovascular function in men with hypercholesterolemia. Nutr. Res. 2014, 34, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Chang, W.H.; Liu, J.F. Effects of kiwifruit consumption on serum lipid profiles and antioxidative status in hyperlipidemic subjects. Int. J. Food Sci. Nutr. 2009, 60, 709–716. [Google Scholar] [CrossRef] [PubMed]
- Svendsen, M.; Tonstad, S.; Heggen, E.; Pedersen, T.R.; Seljeflot, I.; Bohn, S.K.; Bastani, N.E.; Blomhoff, R.; Holme, I.M.; Klemsdal, T.O. The effect of kiwifruit consumption on blood pressure in subjects with moderately elevated blood pressure: A randomized, controlled study. Blood Press. 2015, 24, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Karlsen, A.; Svendsen, M.; Seljeflot, I.; Laake, P.; Duttaroy, A.K.; Drevon, C.A.; Arnesen, H.; Tonstad, S.; Blomhoff, R. Kiwifruit decreases blood pressure and whole-blood platelet aggregation in male smokers. J. Hum. Hypertens. 2013, 27, 126–130. [Google Scholar] [CrossRef] [PubMed]
- Peou, S.; Milliard-Hasting, B.; Shah, S.A. Impact of avocado-enriched diets on plasma lipoproteins: A meta-analysis. J. Clin. Lipidol. 2016, 10, 161–171. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, D.J.A.; Srichaikul, K.; Kendall, C.W.C.; Sievenpiper, J.L.; Abdulnour, S.; Mirrahimi, A.; Meneses, C.; Nishi, S.; He, X.; Lee, S.; et al. The relation of low glycaemic index fruit consumption to glycaemic control and risk factors for coronary heart disease in type 2 diabetes. Diabetologia 2011, 54, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Terauchi, M.; Horiguchi, N.; Kajiyama, A.; Akiyoshi, M.; Owa, Y.; Kato, K.; Kubota, T. Effects of grape seed proanthocyanidin extract on menopausal symptoms, body composition, and cardiovascular parameters in middle-aged women: A randomized, double-blind, placebo-controlled pilot study. Menopause 2014, 21, 990–996. [Google Scholar] [CrossRef] [PubMed]
- Ras, R.T.; Zock, P.L.; Zebregs, Y.E.; Johnston, N.R.; Webb, D.J.; Draijer, R. Effect of polyphenol-rich grape seed extract on ambulatory blood pressure in subjects with pre- and stage I hypertension. Br. J. Nutr. 2013, 110, 2234–2241. [Google Scholar] [CrossRef] [PubMed]
- Tome-Carneiro, J.; Gonzalvez, M.; Larrosa, M.; Yanez-Gascon, M.J.; Garcia-Almagro, F.J.; Ruiz-Ros, J.A.; Garcia-Conesa, M.T.; Tomas-Barberan, F.A.; Espin, J.C. One-year consumption of a grape nutraceutical containing resveratrol improves the inflammatory and fibrinolytic status of patients in primary prevention of cardiovascular disease. Am. J. Cardiol. 2012, 110, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Tome-Carneiro, J.; Gonzalvez, M.; Larrosa, M.; Yanez-Gascon, M.J.; Garcia-Almagro, F.J.; Ruiz-Ros, J.A.; Tomas-Barberan, F.A.; Garcia-Conesa, M.T.; Espin, J.C. Grape resveratrol increases serum adiponectin and downregulates inflammatory genes in peripheral blood mononuclear cells: A triple-blind, placebo-controlled, one-year clinical trial in patients with stable coronary artery disease. Cardiovasc. Drug Ther. 2013, 27, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Basu, A.; Du, M.; Leyva, M.J.; Sanchez, K.; Betts, N.M.; Wu, M.; Aston, C.E.; Lyons, T.J. Blueberries decrease cardiovascular risk factors in obese men and women with metabolic syndrome. J. Nutr. 2010, 140, 1582–1587. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.A.; Figueroa, A.; Navaei, N.; Wong, A.; Kalfon, R.; Ormsbee, L.T.; Feresin, R.G.; Elam, M.L.; Hooshmand, S.; Payton, M.E.; et al. Daily blueberry consumption improves blood pressure and arterial stiffness in postmenopausal women with pre- and stage 1-hypertension: A randomized, double-blind, placebo-controlled clinical trial. J. Acad. Nutr. Diet. 2015, 115, 369–377. [Google Scholar] [CrossRef] [PubMed]
- McAnulty, L.S.; Collier, S.R.; Landram, M.J.; Whittaker, D.S.; Isaacs, S.E.; Klemka, J.M.; Cheek, S.L.; Arms, J.C.; McAnulty, S.R. Six weeks daily ingestion of whole blueberry powder increases natural killer cell counts and reduces arterial stiffness in sedentary males and females. Nutr. Res. 2014, 34, 577–584. [Google Scholar] [CrossRef] [PubMed]
- Riso, P.; Klimis-Zacas, D.; Del, B.C.; Martini, D.; Campolo, J.; Vendrame, S.; Moller, P.; Loft, S.; De Maria, R.; Porrini, M. Effect of a wild blueberry (Vaccinium angustifolium) drink intervention on markers of oxidative stress, inflammation and endothelial function in humans with cardiovascular risk factors. Eur. J. Nutr. 2013, 52, 949–961. [Google Scholar] [CrossRef] [PubMed]
- Moazen, S.; Amani, R.; Homayouni, R.A.; Shahbazian, H.; Ahmadi, K.; Taha, J.M. Effects of freeze-dried strawberry supplementation on metabolic biomarkers of atherosclerosis in subjects with type 2 diabetes: A randomized double-blind controlled trial. Ann. Nutr. Metab. 2013, 63, 256–264. [Google Scholar] [CrossRef] [PubMed]
- Alqurashi, R.M.; Galante, L.A.; Rowland, I.R.; Spencer, J.; Commane, D.M. Consumption of a flavonoid-rich acai meal is associated with acute improvements in vascular function and a reduction in total oxidative status in healthy overweight men. Am. J. Clin. Nutr. 2016, 104, 1227–1235. [Google Scholar] [CrossRef] [PubMed]
- Aghababaee, S.K.; Vafa, M.; Shidfar, F.; Tahavorgar, A.; Gohari, M.; Katebi, D.; Mohammadi, V. Effects of blackberry (Morus nigra L.) consumption on serum concentration of lipoproteins, apo A-I, apo B, and high-sensitivity-C-reactive protein and blood pressure in dyslipidemic patients. J. Res. Med. Sci. 2015, 20, 684–691. [Google Scholar] [PubMed]
- Zhao, S.; Bomser, J.; Joseph, E.L.; DiSilvestro, R.A. Intakes of apples or apple polyphenols decease plasma values for oxidized low-density lipoprotein/β2-glycoprotein I complex. J. Funct. Foods 2013, 5, 493–497. [Google Scholar] [CrossRef]
- Soriano-Maldonado, A.; Hidalgo, M.; Arteaga, P.; de Pascual-Teresa, S.; Nova, E. Effects of regular consumption of vitamin C-rich or polyphenol-rich apple juice on cardiometabolic markers in healthy adults: A randomized crossover trial. Eur. J. Nutr. 2014, 53, 1645–1657. [Google Scholar] [CrossRef] [PubMed]
- Auclair, S.; Chironi, G.; Milenkovic, D.; Hollman, P.C.H.; Renard, C.M.G.C.; Megnien, J.; Gariepy, J.; Paul, J.; Simon, A.; Scalbert, A. The regular consumption of a polyphenol-rich apple does not influence endothelial function: A randomised double-blind trial in hypercholesterolemic adults. Eur. J. Clin. Nutr. 2010, 64, 1158–1165. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Bordi, P.L.; Fleming, J.A.; Hill, A.M.; Kris-Etherton, P.M. Effect of a moderate fat diet with and without avocados on lipoprotein particle number, size and subclasses in overweight and obese adults: A randomized, controlled trial. J. Am. Heart Assoc. 2015, 4, 001355. [Google Scholar] [CrossRef] [PubMed]
- Dow, C.A.; Going, S.B.; Chow, H.; Patil, B.S.; Thomson, C.A. The effects of daily consumption of grapefruit on body weight, lipids, and blood pressure in healthy, overweight adults. Metabolism 2012, 61, 1026–1035. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, A.; Wong, A.; Hooshmand, S.; Sanchez-Gonzalez, M.A. Effects of watermelon supplementation on arterial stiffness and wave reflection amplitude in postmenopausal women. Menopause 2013, 20, 573–577. [Google Scholar] [CrossRef] [PubMed]
| Subject | Study Type | Dose | Disease | Risk Estimates (95%CI) | References |
|---|---|---|---|---|---|
| 512,891 Chinese adults (age: 30–79 years) | cohort study | daily vs. never/rarely fresh fruits | cardiovascular death | 0.60 (0.54–0.67) | [4] |
| incident major coronary events | 0.66 (0.58–0.75) | ||||
| ischemic stroke | 0.75 (0.72–0.79) | ||||
| hemorrhagic stroke | 0.64 (0.56–0.74) | ||||
| 30,458 UK Women (age: 35–69 years) | cohort study | per 80 g/day total fruits | CVD | 0.94 (0.89–1.00) | [5] |
| CHD | 0.93 (0.85–1.01) | ||||
| per 80 g/day fresh fruits | CVD | 0.92 (0.85–1.00) | |||
| CHD | 0.89 (0.79–1.00) | ||||
| 1456 women (age: >70 years) | cohort study | per 129 g/day total fruits | CVD | NA (p < 0.05) | [6] |
| 10,623 Japanese (4147 men, 6476 women) | cohort study | near-daily vs. infrequent citrus fruits | CVD | Men: 0.57 (0.33–1.01) Women: 0.51 (0.29–0.88) | [7] |
| 67,211 women in Shanghai, China (age: 40–70 years) | cohort study | 449 vs. 83 g/day fruits | CHD | 0.62 (0.37, 1.03) | [28] |
| 23 cohort studies of 937,665 participants and 18,047 patients with CHD | meta-analysis | the highest vs. the lowest of total fruits | CHD | 0.86 (0.82–0.91) | [29] |
| per 300 g/day fruits | 0.84 (0.75–0.93) | ||||
| 25,065 men in Denmark (age: 50–64 years) | cohort study | per 25 g/day apples | ACS | 0.97 (0.94, 0.99) | [30] |
| 74,961 Swedish adults (34,670 women, 40,291 men; age: 45–83 years) | cohort study | 3.1 vs. 0.4 servings/day total fruits | total stroke | 0.87 (0.78–0.97) | [31] |
| 1.0 vs. 0.1 servings/day apples/pears | 0.89 (0.80–0.98) | ||||
| 20,069 adults in the Netherlands (age: 20–65 years) | cohort study | >120 vs. ≤120 g/day raw fruits | hemorrhagic stroke | 0.53 (0.28–1.01) | [33] |
| per 25 g/day white fruits (usual apples and pears) | stroke | 0.91 (0.85–0.97) | [32] | ||
| 69,622 women from the Nurses’ Health Study | cohort study | the fifth vs. the lowest quintile of citrus fruits/juices | ischemic stroke | 0.90 (0.77–1.05) | [34] |
| the fifth vs. the lowest quintile of flavanone | 0.81 (0.66–0.99) | ||||
| 20,024 participants without stroke history | cohort study | the highest vs. the lowest quintile of citrus fruits/juices | ischemic stroke | 0.69 (0.53–0.91) | [35] |
| the highest vs. the lowest quintile of flavonoid | 0.72 (0.55–0.95) | ||||
| 28,082 US women (age: ≥39 years) | cohort study | ≥3 vs. <0.5 servings/day total fruits | hypertension | 0.89 (0.81–0.96) | [37] |
| 745 residents from Ohasama, Japan without hypertension at baseline (age: ≥35 years) | cohort study | the highest vs. the lowest quartile of fruits | hypertension | 0.40 (0.21–0.74) | [38] |
| 3 large longitudinal cohort studies of 187,453 subjects | cohort study | ≥4 vs. ≤4 servings/week of total whole fruits | hypertension | 0.92 (0.87–0.97) | [39] |
| 9791 subjects in Korea (3819 men, 5972 women ) | case-control study | the fifth vs. the lowest quintile of fruits | hypertension | 0.73 (0.61–0.88) | [40] |
| 255 Chinese patients with type 2 diabetes (137 men, 118 women) | cross-sectional study | 92.6 ± 39.7 vs. 14.5 ± 8.6 g/day fruits | carotid IMT (0.97 ± 0.02 vs. 1.08 ± 0.03 mm) prevalence of carotid plaque (1.18 vs. 11.76%) | NA (p = 0.046) NA (p = 0.022) | [41] |
| 407 patients with type 2 diabetes (172 men, 235 women) | cross-sectional study | 101.3 ± 28.5 vs. 79.6 ± 24.2g/day fruits | carotid IMT | 0.92 (0.67–0.95) | [42] |
| hs-CRP | 0.69 (0.53–0.89) | ||||
| 43,880 healthy men who had no prior diagnosed CVDs or cancer | cohort study | higher anthocyanin intake | MI | 0.87 (0.75–1.00) | [43] |
| higher flavanone intake | ischemic stroke | 0.78 (0.62–0.97) | |||
| 1393 Chinese adults | cross-sectional study | higher anthocyanins intake | HDL-C | NA (p = 0.001) (women) | [44] |
| higher total flavonoid intake | TG | NA (p = 0.020) (women) | |||
| TG/HDL-C ratios | NA (p = 0.040) (women) | ||||
| higher flavonol intake | TG | NA (p = 0.035) (women) | |||
| TG/HDL-C ratios | NA (p = 0.045) (women) | ||||
| 29,689 Italian women | cohort study | the highest vs. the lowest quartile of fruits | CHD | no significant association | [45] |
| 8060 men aged 50–59 years in France and Northern Ireland | cohort study | ≥1.29 vs. ≤0.57 times/day fruits | ACS | no significant association | [46] |
| 164,617 men and women from five ethnic groups | cohort study | >4.9 vs. <1.5 servings/day fruits | ischemic heart disease | no significant association | [47,48] |
| 34,319 Swedish women aged 49–83 years | cohort study | ≥2.6 vs. ≤0.8 servings/day total fruits | heart failure | no significant association | [49] |
| Fruit | Subject | Study Type | Dose | Main Effects | References |
|---|---|---|---|---|---|
| Grape | |||||
| freeze-dried grape powder | SHR and Wistar-Kyoto (WKY) rats | in vivo | 600 mg/day | BP↓, arterial relaxation↑, vascular compliance↑, cardiac hypertrophy↓ | [51] |
| GSPE | SHR | in vivo | 250 mg/kg/day | arterial remodeling↓, ET-1↓, NO↑, SOD↑, CAT↑, MDA↓ | [52] |
| oligomeric grape seed proanthocyanidins (GSPs) | mice treated with DOCA-salt to induce cardiovascular remodeling | in vivo | NA | heart weight/body weight ratio↓, kidney weight/body weight atio↓, cross-sectional area of cardiomyocytes↓, collagen deposition in heart↓, histopathology injury↓, NO↑, SOD↑, MDA↓ | [53] |
| isolated thoracic aorta ring | in vitro | endothelial-dependent aorta ring relaxation↑ | |||
| GP-EE | rat aorta and small mesenteric artery (SMA) segments | in vitro | 0.3 and 10 μM | endothelium- and NO-dependent vasodilatation↑, phenylephrine(Phe)-induced response in aortic rings↓, O2−↓, contraction elicited by ET-1↓ | [54] |
| red grape skin and seeds polyphenols | human endothelial progenitor cells (EPC) | in vitro | 5, 50 and 150 µg/mL | EPC viability and function↑, endothelial dysfunction↓, hyperglycemia effect↓, ROS production↓ | [55] |
| GSPE | ouabain induced hypertensive rats model | in vivo | 250 mg/kg/day | BP↓, aortic NO production↑ | [57] |
| HUVECs | in vitro | 10 µg/mL | eNOS expression↑ | ||
| GPE | endothelial (EA. hy926) cells | in vitro | 0.068 and 0.250 µg/mL | GCS levels↑, GST activity↑, antioxidant activity↑ | [58] |
| GSE | HUVECs | in vitro | 1 μg/mL | platelet reactivity↓ | [59] |
| red grape berry powder | rats with metabolic syndrome | in vivo | 200, 400 and 800 mg/kg/day | BP↓, plasma TG↓, insulin↓ | [61] |
| HUVECs | in vitro | 20–1400 μg/mL | ET-1↓ | ||
| 0.011, 0.058, 0.29, 1.46 and 3.66 mg/mL | eNOS level↑ | ||||
| grape seed procyanidin extract | hamster | in vivo | 25 mg/kg/day | body weight gain↓, adiposity index↓, weight of white adipose tissue depots↓, plasma phospholipids↓, plasma FFA↓, mesenteric lipid and triglyceride accumulation↓ | [62] |
| grape polyphenols from Vitis vinifera grapes | 24-month-old obese rats | in vivo | 90 mg/kg/day | plasma HDL PON activity↑, LCAT activity↑, CETP activity↓ | [64] |
| grape seed procyanidin extract | SHR | in vivo | 375 mg/kg | SBP↓, DBP↓, GSH activity↑ | [9] |
| GSE or black chokeberry (Aronia melanocarpa) extract | human platelets incubated with Hcy (100 µM) or HTL (1 µM) | in vitro | 2.5, 5, 10 µg/mL | platelet adhesion to collagen and fibrinogen↓, platelet aggregation↓, O2•− production in platelet↓ | [65] |
| malvidin-rich red grape skin extract | isolated and Langendorff perfused rat heart | in vitro | 1–1000 ng/mL | I/R damages↓, coronary dilation↑, active PI3K/NO/cGMP/PKG pathway, intracellular cGMP↑, eNOS, PI3K-AKT, ERK1/2, and GSK-3 β phosphorylation↑ | [56] |
| GSSE | a rat model of global ischemia | in vivo | 2.5 g/kg | brain damage size and histology↓, oxidative stress↓, transition metals associated enzyme activities↑ | [66] |
| GSPE | isolated rat hearts | in vitro | NA | RA↓, Na+/K+-ATPase activity↑, Na+/K+-ATPase α1 subunit↑, free radical↓ | [67] |
| GSPE | a rat model of deep vein thrombosis (DVT) | in vivo | 400 mg/kg/day | thrombus length and weight↓, protecte endothelium integrity, IL-6, IL-8 and TNF-α↓ | [68] |
| Blueberry | |||||
| blueberry extract (Vaccinium ashei Reade) | hypercholesterolemic rat | in vivo | 25, 50 mg/kg | aortic lesions↓, oxidative damage to lipids and proteins↓, TC↓, LDL-C↓, TG↓, activity of CAT, SOD and GSH-Px↑ | [69] |
| freeze-dried blueberry powder | rats fed a high-fat/cholesterol diet | in vivo | 2% (w/w) | SBP↓, aorta relaxation↑, endothelial dysfunction↓ | [70] |
| 7 phenolic acids of freeze-dried blueberry | murine macrophage cell line RAW 264.7 | in vitro | NA | TNF-α and IL-6 mRNA expression and protein levels↓, MAPK, JNK, p38, and Erk1/2 phosphorylation↓, mRNA expression and protein levels of scavenger receptor CD36↓, foam cell formation↓, expression and protein levels of ABCA1↑ | [71] |
| Pomegranate | |||||
| PE | SR-BI/apoE double KO mice | in vivo | 307.5 µL/L in water | aortic sinus and coronary artery atherosclerosis↓, oxidative stress and inflammation in the vessel wall↓ | [73] |
| PE containing 40% punicalagin | SHR | in vivo | 150 mg/kg/day | BP↓, cardiac hypertrophy↓, oxidative stress↓, antioxidant defense system↑, paraventricular nucleus inflammation↓, mitochondrial superoxide anion levels↓, mitochondrial function↑ | [74] |
| PE containing 40% punicalagin | heart of a high-fat diet-induced obesity rat model | in vivo | 150 mg/kg/day | mitochondrial biogenesis↑, oxidative stress↓, phase II enzymes↑, cardiac metabolic disorders↓ | [72] |
| pomegranate seed extract | CHI rat model | in vivo | 100, 200, 400, 800 mg/kg/day | motor and cognitive coordination↑ | [75] |
| Apple | |||||
| Bravo de Esmolfe apple | male Wistar rats fed a cholesterol-enriched diet (+2% cholesterol) | in vivo | 20% (w/w) = 5g/rat/day (~2–3 apples/person/day) for 30 days | serum TG↓, TC↓, LDL-C↓, oxLDL↓ | [76] |
| Fuji apple peel Granny Smith apple peel | CF-1 mice with MS apoE−/− mice | in vivo | 20% (w/w) for 43 days 20% (w/w) for 10 weeks | glycaemia↓, TC↓, HDL-C↓, LDL-C↓, ureic nitrogen↓, TG↓, insulin↓, ADMA↓ atherogenic progression↓, cholesterol accumulation area↓ | [77] |
| Hawthorn | |||||
| HFC | apoE−/− atherosclerotic mice with high blood lipid levels fed with a high cholesterol diet | in vivo | 0.5 mL/day | TG↓, LDL-C/TC ratio↓ | [10] |
| HPPS | the liver of high fat diet induced hyperlipidemic mice | in vivo | 150 mg/kg | weight gain↓, TG↓, lipid excretion in feces↑, mRNAs and activities of acyl-CoA oxidase, carnitine palmitoyltransferase I, 3-ketoacyl-CoA thiolase, and 2,4-dienoyl-CoA reductase↑, gene and protein expressions of PPAR-α↑ | [79] |
| freeze dried hawthorn fruit (Crataegus pinnatifida) | apoE−/− mice | in vivo | 1% (w/w) | atherosclerotic lesions↓, TC↓, TG↓, T-AOC values↑, SOD and GSH-Px activities↑, hepatic FAS and SREBP-1c mRNA levels↓, hepatic SOD1, SOD2, Gpx3 mRNA levels↑ | [80] |
| sugar-free aqueous extract of hawthorn fruit (Crataegus pinnatifida var. Major) | high fat diet fed rats | in vivo | 72 and 288 mg/kg/day | TC, TG and LDL-C↓, HDL-C↑, CRP, IL-1β, IL-8 and IL-18↓, ET, 6-keto-PGF1α and TXB2↑, pathological changes in the arteries↓, IMT↓ | [81] |
| Avocado | |||||
| avocado pulp (Persea americana) extract | male adult CD 1 mice | in vivo | 25 mg/kg | thrombus formation↓ | [84] |
| platelet | in vitro | 10 µL | platelet aggregation↓ | ||
| avocado oil | rats ingested with sucrose | in vivo | 7.5% (w/w) | TG↓, VLDL↓, LDL↓, hs-CRP↓ | [85] |
| Others | |||||
| freeze-dried mango pulp | male C57BL/6J mice fed a high-fat diet | in vivo | 1% or 10% (w/w) | epididymal fat mass↓, percentage of body fat↓, improve glucose tolerance, insulin resistance↓ | [11] |
| methanolic extract of papaya (Carica papaya) | SHR | in vivo | 100 mg/kg (twice a day) | BP↓, angiotensin converting enzyme(ACE) activity↓, cardiac hypertrophy↓, improve baroreflex sensitivity | [12] |
| sour cherry seed kernel extract | hearts from Sprague-Dawley rats | in vitro | 30 mg/kg/day | post ischemic cardiac functions↑, infarct size↓, heme oxygenase-1 (HO-1)↑, Bcl-2↑ | [88] |
| total flavonoids of Guangzao (Choerospondias axillaris) | I/R male Sprague-Dawley rats | in vivo | 75, 150 and 300 mg/kg/day | cardiac function↑, heart pathologic lesion↓, CAT↑, GSH-Px↑, SOD↑, MDA↓, TUNEL-positive nuclear staining↓, Bcl-2-associated X protein (Bax)↓, caspase-3↓, Bcl-2↑, p38 MAPK activity↓, JNK activity↓ | [89] |
| hydroalcoholic extract of acai (Euterpe oleracea Mart.) seeds | male Wistar rats subjected to myocardial infarction | in vivo | 100 mg/kg/day | prevent the development of exercise intolerance, cardiac hypertrophy, fibrosis, and dysfunction | [90] |
| acai pulp | female Fischer rat of dietary-induced hypercholesterolemia | in vivo | 2% (w/w) | TC↓, LDL-C↓, atherogenic index↓, HDL-C↑, cholesterol excretion in feces↑, expression of the LDL-R, ABCG5, and ABCG8 genes↑ | [98] |
| bilberry (Vaccinium myrtillus L.) anthocyanin-rich extract | apoE−/− mice | in vivo | 0.02% (w/w) | improve hypercholesterolemia against atherosclerosis | [91] |
| unrefined black raspberry seed oils | male Syrian hamsters fed high-cholesterol (0.12%), high-fat (9%) diets | in vivo | NA | plasma and liver TG↓, hypertriglyceridemia↓ | [92] |
| polyphenols from sea buckthorn berry | rats with hyperlipidemia | in vivo | 7–28 mg/kg | serum lipids↓, TNF-α↓, IL-6↓, antioxidant enzymes activity↑, eNOS, ICAM-1, and LOX-1 mRNA expression and proteins in aortas↓ | [93] |
| Jujube (Zizyphus jujuba) fructus and semen extract | human macrophages | in vitro | NA | the foam cell formation induced by acetylated LDL↓, prevent atherosclerosis | [94] |
| methanol extract of blackberry (Rubus allegheniensis Port.) | human monocyte-derived macrophages induced by acetylated LDL | in vitro | 50 μM | foam cell formation↓ | [95] |
| yellow passion fruit pulp | SHR | in vivo | 5, 6 or 8 g/kg/day | SBP↓, GSH↑, thiobarbituric acid-reactive substances (TBARS)↓ | [96] |
| proanthocyanidins in boysenberry seed extract | SHR | in vivo | 100 and 200 mg/kg | SBP↓ | [97] |
| rat aorta rings | in vitro | vasorelaxant activity↑ | |||
| methanolic extract of date palm (Phoenix dactylifera L.) | cerebral ischemia rats | in vivo | 100, 300 mg/kg | SOD↑, CAT↑, GSH↑, glutathione reductase↑, lipid peroxidation↓, oxidative stress↓, neuronal damage↓ | [99] |
| black chokeberry (Aronia melanocarpa) extract | bovine coronary artery endothelial cells | in vitro | 0.1 g/mL | NO↑, eNOS phosphorylation↑ | [100] |
| saskatoon berry powder | leptin receptor-deficient diabetic mice | in vivo | 5% (w/w) | monocyte adhesion to aorta↓, inflammatory, fibrinolytic or stress regulators in aorta or heart apex↓ | [101] |
| saskatoon berry powder | leptin receptor-deficient diabetic mice | in vivo | 5% (w/w) | endoplasmic reticulum stress (ERS)↓, unfolded protein response (UPR)↓ | [102] |
| glycated LDL-treated HUVECs | in vitro | ||||
| 19 fruits widely consumed in central Chile | NA | in vitro | 1 mg/mL | anticoagulant activities: grape, raspberry fibrinolytic activity: raspberry | [103] |
| peach (Prunus persica) pulp ethylacetate extract | cultured vascular smooth muscle cells (VSMCs) | in vitro | 50, 100, or 200 µg/mL | Angiotensin II (Ang II) induced intracellular Ca2+ elevation↓, generation of ROS↓ | [104] |
| methanolic extract of Lingonberry (Vaccinium vitis-idaea L.) | H9c2 rat myoblasts simulated IR | in vitro | 5 and 10 µM | apoptosis↓, markers of nuclei condensation , caspase-3 activation, and MAPK signaling↓ | [105] |
| blueberry anthocyanin fraction (BBA), blackberry anthocyanin fraction (BKA), and blackcurrant anthocyanin fraction (BCA) | RAW 264.7 macrophages treated by LPS bone marrow-derived macrophages from Nrf2+/+ mice treated by LPS | in vitro | 0–20 μg/mL | IL-1 β mRNA levels↓, NF-κB p65 translocation to the nucleus↓ cellular ROS levels↓, IL-1β mRNA levels↓ | [106] |
| pomegranate juice, together with date fruit and date seeds extract | apoE−/− mice | in vivo | 0.5 µM gallic acid equivalents (GAE)/day | TC↓, TG↓, PON1 activity↑, mouse peritoneal macrophage (MPM) oxidative stress↓, MPM cholesterol content↓, and MPM LDL uptake↓, aortas lipid peroxide content↓, aortas PON lactonase activity↑ | [107] |
| Subject | Component | Treatment | Duration | Outcome | References |
|---|---|---|---|---|---|
| 152 patients with type 2 diabetes | low glycaemic index fruit | −3.1 to 2.7 servings/day | 6 months | HbA1c1↓, SBP↓, CHD risk↓ | [135] |
| 52 patients with mild hyperlipidemia | red grape seed extract (RGSE) | 200 mg/day | 8 weeks | TC↓, LDL-C↓, Ox-LDL↓ | [60] |
| 24 pre-hypertensive, overweight, and/or pre-diabetic subjects | whole grape extract (WGE) | 350 mg/day | 6 weeks | SOD↓, 8-isoprostane↓, Ox-LDL↓, TC/HDL-C ratios↓, HDL-C ↑ | [109] |
| 69 patients with hyperlipidemia | Condori red grapes or Shahroodi white grapes | 500 g/day | 8 weeks | thiobarbituric acid reactive substances (TBARS)↓, total antioxidant capacity (TAC)↑, TC↓, LDL-C↓ | [110] |
| 60 healthy volunteers | polyphenol-rich grape extract supplementation | 700 mg/day | 56 days | TC↓, LDL-C↑, TAC↑, vitamin E↑ | [111] |
| 96 women aged 40–60 years who had at least one menopausal symptom | grape seed extract tablets | =100 or 200 mg proanthocyanidin/day | 4 weeks | SBP↓, DBP↓ | [136] |
| 70 untreated subjects with pre- and stage I hypertension (SBP: 120–159 mmHg) | grape seed extract (GSE) rich in low-molecular-weight polyphenolic compounds | 300 mg/day | 8 weeks | BP values were modestly, but not significantly, affected | [137] |
| 75 patients at high risk of CVD (with diabetes or hypercholesterolemia plus ≥1 other CV risk factor) and undergoing primary prevention of CVDs | resveratrol-rich grape supplementation | 350 mg/day = 8 mg resveratrol for the first 6 months and a double dose for the next 6 months | 12 months | hs-CRP↓, TNF-á↓, plasminogen activator inhibitor type 1 (PAI-1)↓, IL-6/IL-10 ratio↓, IL-10↑ | [138] |
| 75 stable patients with CHD treated according to currently accepted guidelines for secondary prevention of CVDs | resveratrol-rich grape supplementation | 350 mg/day = 8 mg resveratrol for the first 6 months and a double dose for the next 6 months | 12 months | serum adiponectin↑, PAI-1↓, inflammatory genes in peripheral blood mononuclear cells (PBMCs) | [139] |
| 48 participants with MS (4 men, 44 women; BMI: 37.8 ± 2.3 kg/m2; age: 50.0 ± 3.0 years) | freeze-dried blueberry | 50 g(~350 g fresh)/day | 8 weeks | SBP↓, DBP↓, Ox-LDL↓, MDA↓, serum hydroxynonenal↓ | [140] |
| 58 postmenopausal women with pre-andstage 1-hypertension | freeze-dried blueberry powder | 22 g/day | 8 weeks | SBP↓, DBP↓, brachial-ankle pulse wave velocity↓, NO↑ | [141] |
| 25 sedentary men and postmenopausal women (age: 18–50 years) | whole blueberry powder | ~250 g berries/day | 6 weeks | natural killer(NK) cells↑, augmentation index (AIx)↓, aortic systolic pressures (ASPs)↓, diastolic pressures↓ | [142] |
| 18 male volunteers (age: 47.8 ± 9.7 years; BMI: 24.8 ± 2.6 kg/m2) | freeze-dried wild blueberries (Vaccinium angustifolium) powder | 25 g = 375 mg anthocyanins | 6 weeks | endogenously oxidized DNA bases↓, H2O2-induced DNA damage↓ | [143] |
| 23 healthy subjects (11 men, 12 women; age: 27 ± 3.2 years; weight: 63.5 ± 12.7 kg; BMI: 21.74 ± 2.5 kg/m2) | strawberries | 500 g/day | 1 month | TC↓, LDL-C↓, TG↓, MDA↓, urinary 8-OHdG↓, isoprostanes↓, TAC↑, spontaneous and oxidative hemolysis↓, activated platelets↓ | [113] |
| 60 volunteers (5 men, 55 women; age: 49 ± 10 years; BMI: 36 ± 5 kg/m2) | freeze-dried strawberries (FDS) | 25 or 50 g/day | 12 weeks | TC↓, LDL-C↓, MDA↓ | [114] |
| 27 subjects with MS (2 men, 25 women; age: 47.0 ± 3.0 years; BMI: 37.5 ± 2.15 kg/m2) | FDS | 50g (~3 cups fresh)/day | 8 weeks | TC↓, LDL-C↓, small LDL particles↓, vascular cell adhesion molecule-1(VCAM-1)↓ | [115] |
| 36 subjects with type 2 diabetes (13 men, 23 women; age: 51.57 ± 10 years; BMI: 27.90 ± 3.7 kg/m2 ) | FDS | 50 g (~500 g fresh)/day | 6 weeks | CRP↓, MDA↓, HbA1c↓, TAC↑ | [144] |
| 24 overweight and obese subjects (10 men, 14 women; age: 50.9 ± 15 years; ,BMI: 29.2 ± 2.3 kg/m2) consumed high carbohydrate/fat meal | strawberry (Fragaria) beverage | =10 g FDS (~100 g fresh)/day | 6 weeks | TG↓, Ox-LDL↓, PAI-1↓, IL-1 β↓ | [116,117] |
| 10 overweight adults (BMI: 25–30 kg/m2) | acai pulp (Euterpe oleracea Mart.) | 100 g twice/day | 1 month | fasting glucose↓, postprandial plasma glucose↓, insulin↓, TC↓, LDL-C↓, TC/HDL-C ratio↓ | [118] |
| 23 healthy male volunteers (age: 30–65 years; BMI: 25–30 kg/m2) | acai-based smoothie | =694 mg total phenolics | 1 d | flow-mediated dilatation (FMD)↑ | [145] |
| 72 dyslipidemic patients | blackberry (Morus nigra L.) juice with pulp | 300 mL/day | 8 weeks | apo A-I↑, HDL↑, apo B↓, hs-CRP↓, SBP↓ | [146] |
| 40 hyperlipidemic patients (age: 20–60 years) | Caucasian whortleberry (Vaccinium arctostaphylos L.) fruit hydroalcoholic extract | 350 mg/8 h | 2 months | TC↓, TG↓, LDL-C↓, HDL-C↑ | [119] |
| 80 overweight and obese female volunteers (BMI: 29.6 ± 2.1 kg/m2) | sea buckthorn berries (SB) sea buckthorn oil (SBo) SB phenolic extract (SBe) bilberries (BB) | ~100 g/day fresh berries | 33–35 days | SB: TG and VLDL↓, waist circumference↓; SBo: total lipoprotein, intermediate-density lipoprotein (IDL), LDL and LDL-C↓, vascular cell adhesion molecule (VCAM)↓; SBe: VLDL fractions and serum TG↑, intercellular adhesion molecule (ICAM)↓; BB: improve serum lipids and lipoproteins, waist circumference↓,body weight↓, VCAM↓ | [120,121] |
| 120 dyslipidemic subjects (age: 40–65 years) | berry-derived anthocyanin | 160 mg twice/day | 12 weeks | LDL-C↓, HDL-C↑, cellular cholesterol efflux to serum↑, mass and activity of plasma CETP ↓ | [122] |
| 160 postmenopausal women | dried apple | 75 g/day | 1 year | TC↓, LDL-C↓, lipid hydroperoxide↓, CRP↓ | [123] |
| 50 mildly hypercholesterolaemic healthy subjects (28 men, 22 women) | Annurca apple (Malus pumila Miller cv. Annurca) | 2/day | 4 months | TC↓, LDL-C↓, HDL-C↑ | [124] |
| 23 healthy volunteers | whole apples | 550 g/day | 4 weeks | whole apple, pomace and cloudy juice lowered serum TC and LDL-C | [125] |
| apple pomace | 22 g/day | ||||
| clear apple juices | 500 mL/day | ||||
| cloudy apple juices | 500 mL/day | ||||
| 51 healthy adults (age: 40–60 years) | apple | 1/day | 4 weeks | Ox-LDL/β2-glycoprotein I complex (Ox-LDL-β2 GPI) | [147] |
| 20 subjects (age: 21–29 years) | apple juice | two glasses (2 × 250 mL/day) | 4 weeks | plasma antioxidant activity (FRAP)↑, insulin↑, HOMA↑, total GSH↓ | [148] |
| 30 healthy subjects (6 men, 24 women; age: 47.3 ± 13.6 years) | flavonoid-rich apple | 120 g flesh + 80 g skin twice/day | 1 d | NO status↑, endothelial function↑, FMD↑, pulse pressure↓, SBP↓ | [126] |
| 14 subjects (age: 45–70 years) | drink containing epicatechin from an apple extract | =140 mg epicatechin/day | 1 d | NO metabolites | [127] |
| 30 hypercholesterolemic volunteers | polyphenol-rich apple | 40 g = 1.43 polyphenols/day | 4 weeks | did not improve vascular function | [149] |
| 85 hypercholesterolemic men consumed a healthy diet | green kiwifruit | 2/day | 8 weeks | plasma HDL-C↑, TC/HDL-C ratio↓, hs-CRP↓, IL-6↓ | [129] |
| did not improve BP and markers of cardiovascular function | [130] | ||||
| 43 subjects who had hyperlipidemia in Taiwan (13 men, 30 women) | kiwifruit | 2/day | 8 weeks | HDL-C↑, LDL-C/HDL-C ratio↓, TC/HDL-C ratio↓, vitamin C↑, vitamin E↑, LDL oxidation↓, MDA↓, 4-hydroxy-2-nonenal↓ | [131] |
| 118 subjects with moderately elevated BP or stage 1 hypertension (SBP: 130–159 mmHg, DBP: 85–99 mmHg) | kiwifruit | 3/day | 8 weeks | 24-h ambulatory BP↓ | [132] |
| 102 male smokers (age: 44–74 years) | kiwifruit | 3/day | 8 weeks | SBP↓, DBP↓, platelet aggregation↓, ACE activity↓ | [133] |
| 45 overweight or obese participants with baseline LDL-C in the 25–90% | fresh Hass avocado | 1(~36 g)/day | 5 weeks | LDL-C↓, LDL-particle number↓, small dense LDL-C↓, LDL-C/HDL-C ratio↓ | [150] |
| 74 overweight adults | fresh Rio-Red grapefruit | 0.5 with each meal (3x)/day | 6 weeks | waist circumference↓, SBP↓, TC↓, LDL↓ | [151] |
| 12 obese postmenopausal women (age: 57 ± 1 years; BMI: 38.1 ± 2.1 kg/m2; SBP: 153 ± 4 mmHg) | L-citrulline-rich watermelon supplementation | =6 g L-citrulline/day | 6 weeks | arterial stiffness↓, aortic SBP↓, pressure wave reflection amplitude↓ | [152] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, C.-N.; Meng, X.; Li, Y.; Li, S.; Liu, Q.; Tang, G.-Y.; Li, H.-B. Fruits for Prevention and Treatment of Cardiovascular Diseases. Nutrients 2017, 9, 598. https://doi.org/10.3390/nu9060598
Zhao C-N, Meng X, Li Y, Li S, Liu Q, Tang G-Y, Li H-B. Fruits for Prevention and Treatment of Cardiovascular Diseases. Nutrients. 2017; 9(6):598. https://doi.org/10.3390/nu9060598
Chicago/Turabian StyleZhao, Cai-Ning, Xiao Meng, Ya Li, Sha Li, Qing Liu, Guo-Yi Tang, and Hua-Bin Li. 2017. "Fruits for Prevention and Treatment of Cardiovascular Diseases" Nutrients 9, no. 6: 598. https://doi.org/10.3390/nu9060598
APA StyleZhao, C.-N., Meng, X., Li, Y., Li, S., Liu, Q., Tang, G.-Y., & Li, H.-B. (2017). Fruits for Prevention and Treatment of Cardiovascular Diseases. Nutrients, 9(6), 598. https://doi.org/10.3390/nu9060598






