Absorption Profile of (Poly)Phenolic Compounds after Consumption of Three Food Supplements Containing 36 Different Fruits, Vegetables, and Berries
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Test Capsules
2.3. Study Design
2.4. Chemicals
2.5. Plasma Extraction
2.6. UHPLC/MSn Analysis
3. Results
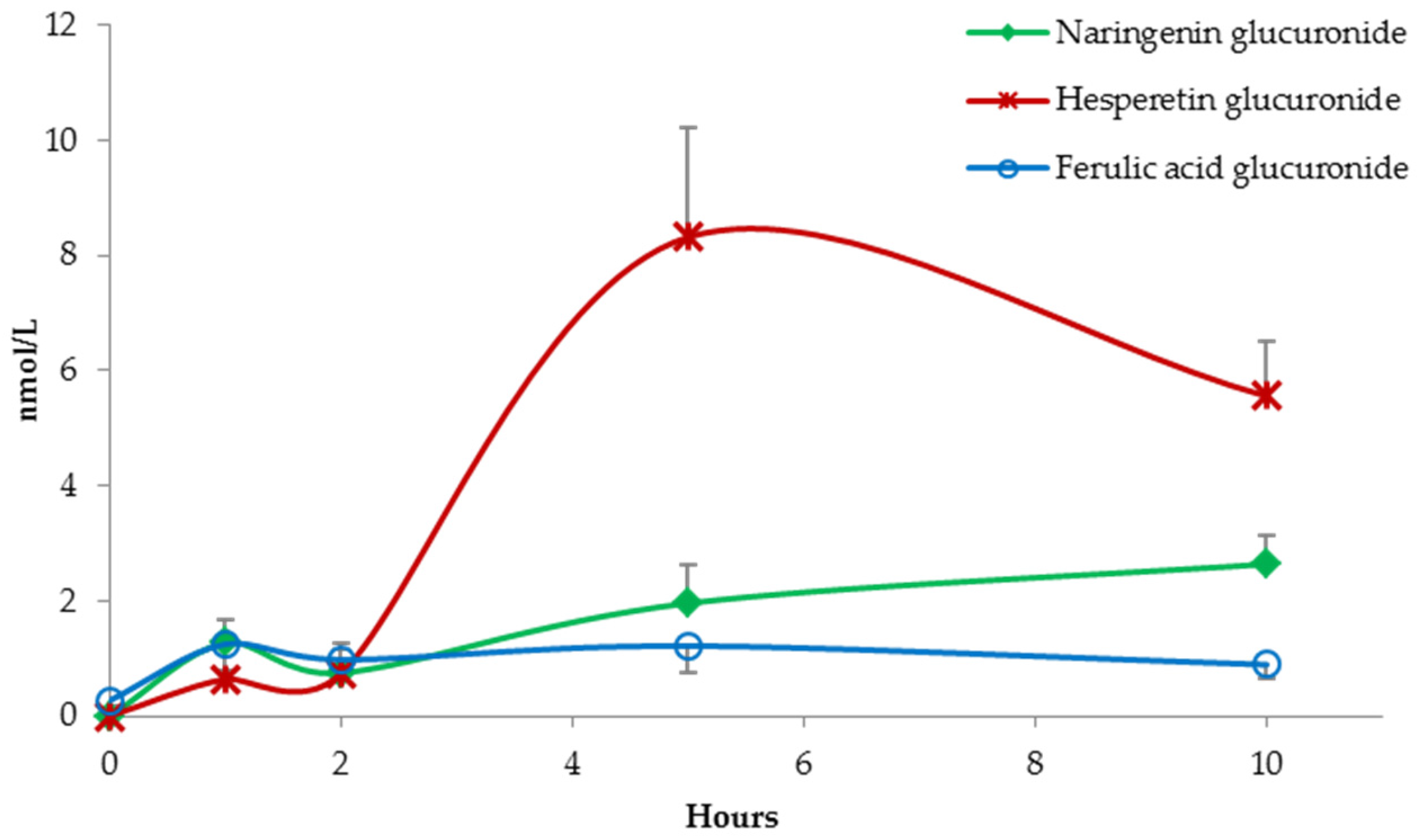
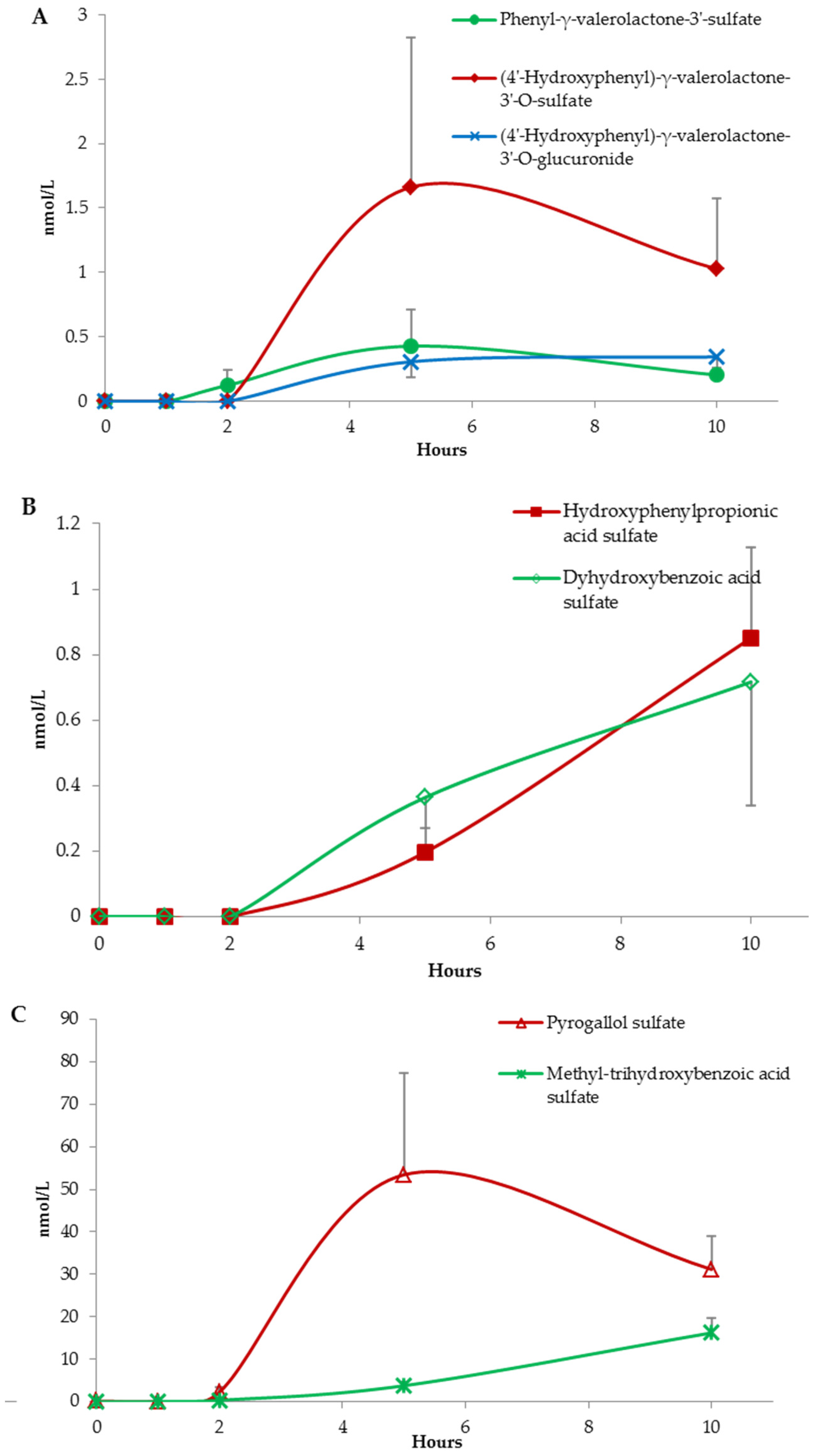
(Poly)Phenolic Compound Absorption
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Agudo, A. Measuring intake of fruit and vegetables. In Joint FAO/WHO Workshop on Fruit and Vegetables for Health, WHO/FAO, Kobe, Japan, 1–3 September 2004; World Health Organization: Geneva, Switzerland, 2005. [Google Scholar]
- World Health Organization. European Action Plan for Food and Nutrition Policy 2007–2012; World Health Organization: Copenhagen, Denmark, 2008. [Google Scholar]
- Boeing, H.; Bechthold, A.; Bub, A.; Ellinger, S.; Haller, D.; Kroke, A.; Leschik-Bonnet, E.; Muller, M.J.; Oberritter, H.; Schulze, M.; et al. Critical review: Vegetables and fruit in the prevention of chronic diseases. Eur. J. Nutr. 2012, 51, 637–663. [Google Scholar] [CrossRef] [PubMed]
- Lock, K.; Pomerleau, J.; Causer, L.; Altmann, D.R.; McKee, M. The global burden of disease attributable to low consumption of fruit and vegetables: Implications for the global strategy on diet. Bull. WHO 2005, 83, 100–108. [Google Scholar] [PubMed]
- Leenders, M.; Sluijs, I.; Ros, M.M.; Boshuizen, H.C.; Siersema, P.D.; Ferrari, P.; Weikert, C.; Tjonneland, A.; Olsen, A.; Boutron-Ruault, M.C.; et al. Fruit and vegetable consumption and mortality: European prospective investigation into cancer and nutrition. Am. J. Epidemiol. 2013, 178, 590–602. [Google Scholar] [CrossRef] [PubMed]
- Boffetta, P.; Couto, E.; Wichmann, J.; Ferrari, P.; Trichopoulos, D.; Bueno-de-Mesquita, H.B.; van Duijnhoven, F.J.; Buchner, F.L.; Key, T.; Boeing, H.; et al. Fruit and vegetable intake and overall cancer risk in the European Prospective Investigation into Cancer and Nutrition (EPIC). J. Natl. Cancer Inst. 2010, 102, 529–537. [Google Scholar] [CrossRef] [PubMed]
- Bazzano, L.A.; Li, T.Y.; Joshipura, K.J.; Hu, F.B. Intake of fruit, vegetables, and fruit juices and risk of diabetes in women. Diabetes Care 2008, 31, 1311–1317. [Google Scholar] [CrossRef] [PubMed]
- Mursu, J.; Virtanen, J.K.; Tuomainen, T.P.; Nurmi, T.; Voutilainen, S. Intake of fruit, berries, and vegetables and risk of type 2 diabetes in Finnish men: The Kuopio Ischaemic Heart Disease Risk Factor Study. Am. J. Clin. Nutr. 2014, 99, 328–333. [Google Scholar] [CrossRef] [PubMed]
- Public Health England; Food Standards Agency. Results of the National Diet and Nutrition Survey (NDNS) Rolling Programme for 2008 and 2009 to 2011 and 2012; National Diet and Nutrition Survey (NDNS); Public Health England: London, UK, 2014.
- Leclercq, C.; Arcella, D.; Piccinelli, R.; Sette, S.; Le Donne, C.; Turrini, A. The Italian National Food Consumption Survey INRAN-SCAI 2005–06: Main results in terms of food consumption. Public Health Nutr. 2009, 12, 2504–2532. [Google Scholar] [CrossRef] [PubMed]
- Niva, M. ‘All foods affect health’: Understandings of functional foods and healthy eating among health-oriented Finns. Appetite 2007, 48, 384–393. [Google Scholar] [CrossRef] [PubMed]
- Nile, S.H.; Park, S.W. Edible berries: Bioactive components and their effect on human health. Nutrition 2014, 30, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Murphy, M.M.; Barraj, L.M.; Herman, D.; Bi, X.; Cheatham, R.; Randolph, R.K. Phytonutrient intake by adults in the United States in relation to fruit and vegetable consumption. J. Acad. Nutr. Diet. 2012, 112, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.H. Dietary bioactive compounds and their health implications. J. Food Sci. 2013, 78 (Suppl. 1), A18–A25. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Stepaniak, U.; Topor-Madry, R.; Szafraniec, K.; Pajak, A. Estimated dietary intake and major food sources of polyphenols in the Polish arm of the HAPIEE study. Nutrition 2014, 30, 1398–1403. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Jiménez, J.; Fezeu, L.; Touvier, M.; Arnault, N.; Manach, C.; Hercberg, S.; Galan, P.; Scalbert, A. Dietary intake of 337 polyphenols in French adults. Am. J. Clin. Nutr. 2011, 93, 1220–1228. [Google Scholar] [CrossRef] [PubMed]
- Tresserra-Rimbau, A.; Medina-Remon, A.; Perez-Jimenez, J.; Martinez-Gonzalez, M.A.; Covas, M.I.; Corella, D.; Salas-Salvado, J.; Gomez-Gracia, E.; Lapetra, J.; Aros, F.; et al. Dietary intake and major food sources of polyphenols in a Spanish population at high cardiovascular risk: The PREDIMED study. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 953–959. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (poly)phenolics in human health: Structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxid. Redox Signal. 2013, 18, 1818–1892. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Mateos, A.; Vauzour, D.; Krueger, C.G.; Shanmuganayagam, D.; Reed, J.; Calani, L.; Mena, P.; del Rio, D.; Crozier, A. Bioavailability, bioactivity and impact on health of dietary flavonoids and related compounds: An update. Arch. Toxicol. 2014, 88, 1803–1853. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Stepaniak, U.; Micek, A.; Stefler, D.; Bobak, M.; Pajak, A. Dietary polyphenols are inversely associated with metabolic syndrome in Polish adults of the HAPIEE study. Eur. J. Nutr. 2016. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.J.; Zhan, J.; Liu, X.L.; Wang, Y.; Ji, J.; He, Q.Q. Dietary flavonoids intake and risk of type 2 diabetes: A meta-analysis of prospective cohort studies. Clin. Nutr. 2014, 33, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Godos, J.; Lamuela-Raventos, R.; Ray, S.; Micek, A.; Pajak, A.; Sciacca, S.; D’Orazio, N.; del Rio, D.; Galvano, F. A comprehensive meta-analysis on dietary flavonoid and lignan intake and cancer risk: Level of evidence and limitations. Mol. Nutr. Food Res. 2016. [Google Scholar] [CrossRef] [PubMed]
- Bresciani, L.; Calani, L.; Cossu, M.; Mena, P.; Sayegh, M.; Ray, S.; del Rio, D. (Poly)phenolic characterization of three food supplements containing 36 different fruits, vegetables and berries. PharmaNutrition 2015, 3, 11–19. [Google Scholar] [CrossRef]
- Lamprecht, M.; Obermayer, G.; Steinbauer, K.; Cvirn, G.; Hofmann, L.; Ledinski, G.; Greilberger, J.F.; Hallstroem, S. Supplementation with a juice powder concentrate and exercise decrease oxidation and inflammation, and improve the microcirculation in obese women: Randomised controlled trial data. Br. J. Nutr. 2013, 110, 1685–1695. [Google Scholar] [CrossRef] [PubMed]
- De Spirt, S.; Sies, H.; Tronnier, H.; Heinrich, U. An encapsulated fruit and vegetable juice concentrate increases skin microcirculation in healthy women. Skin Pharmacol. Physiol. 2012, 25, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Roll, S.; Nocon, M.; Willich, S.N. Reduction of common cold symptoms by encapsulated juice powder concentrate of fruits and vegetables: A randomised, double-blind, placebo-controlled trial. Br. J. Nutr. 2011, 105, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Nantz, M.P.; Rowe, C.A.; Nieves, C., Jr.; Percival, S.S. Immunity and antioxidant capacity in humans is enhanced by consumption of a dried, encapsulated fruit and vegetable juice concentrate. J. Nutr. 2006, 136, 2606–2610. [Google Scholar] [PubMed]
- Plotnick, G.D.; Corretti, M.C.; Vogel, R.A.; Hesslink, R., Jr.; Wise, J.A. Effect of supplemental phytonutrients on impairment of the flow-mediated brachial artery vasoactivity after a single high-fat meal. J. Am. Coll. Cardiol. 2003, 41, 1744–1749. [Google Scholar] [CrossRef]
- Ottaviani, J.I.; Momma, T.Y.; Heiss, C.; Kwik-Uribe, C.; Schroeter, H.; Keen, C.L. The stereochemical configuration of flavanols influences the level and metabolism of flavanols in humans and their biological activity in vivo. Free Radic. Biol. Med. 2011, 50, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Pereira-Caro, G.; Borges, G.; van der Hooft, J.; Clifford, M.N.; Del Rio, D.; Lean, M.E.; Roberts, S.A.; Kellerhals, M.B.; Crozier, A. Orange juice (poly)phenols are highly bioavailable in humans. Am. J. Clin. Nutr. 2014, 100, 1378–1384. [Google Scholar] [CrossRef] [PubMed]
- Curti, C.; Brindani, N.; Battistini, L.; Sartori, A.; Pelosi, G.; Mena, P.; Brighenti, F.; Zanardi, F.; del Rio, D. Catalytic, Enantioselective Vinylogous Mukaiyama Aldol Reaction of Furan-Based Dienoxy Silanes: A Chemodivergent Approach to γ-Valerolactone Flavan-3-ol Metabolites and δ-Lactone Analogues. Adv. Synth. Catal. 2015. [Google Scholar] [CrossRef]
- Liu, Y.; Lien, I.F.; Ruttgaizer, S.; Dove, P.; Taylor, S.D. Synthesis and protection of aryl sulfates using the 2,2,2-trichloroethyl moiety. Org. Lett. 2004, 6, 209–212. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Jagdmann, G.E., Jr.; Van Zandt, M.; Sheeler, R.; Beckett, P.; Schroeter, H. Chemical synthesis and characterization of epicatechin glucuronides and sulfates: Bioanalytical standards for epicatechin metabolite identification. J. Nat. Prod. 2013, 76, 157–169. [Google Scholar] [CrossRef] [PubMed]
- Urpi-Sarda, M.; Monagas, M.; Khan, N.; Llorach, R.; Lamuela-Raventos, R.M.; Jauregui, O.; Estruch, R.; Izquierdo-Pulido, M.; Andres-Lacueva, C. Targeted metabolic profiling of phenolics in urine and plasma after regular consumption of cocoa by liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2009, 1216, 7258–7267. [Google Scholar] [CrossRef] [PubMed]
- Kern, S.M.; Bennett, R.N.; Mellon, F.A.; Kroon, P.A.; Garcia-Conesa, M.T. Absorption of hydroxycinnamates in humans after high-bran cereal consumption. J. Agric. Food Chem. 2003, 51, 6050–6055. [Google Scholar] [CrossRef] [PubMed]
- Bresciani, L.; Scazzina, F.; Leonardi, R.; Dall’Aglio, E.; Newell, M.; Dall’Asta, M.; Melegari, C.; Ray, S.; Brighenti, F.; del Rio, D. Bioavailability and metabolism of phenolic compounds from wholegrain wheat and aleurone-rich wheat bread. Mol. Nutr. Food Res. 2016, 60, 2343–2354. [Google Scholar] [CrossRef] [PubMed]
- Roowi, S.; Mullen, W.; Edwards, C.A.; Crozier, A. Yoghurt impacts on the excretion of phenolic acids derived from colonic breakdown of orange juice flavanones in humans. Mol. Nutr. Food Res. 2009, 53 (Suppl. 1), S68–S75. [Google Scholar] [CrossRef] [PubMed]
- Mulder, T.P.; Rietveld, A.G.; van Amelsvoort, J.M. Consumption of both black tea and green tea results in an increase in the excretion of hippuric acid into urine. Am. J. Clin. Nutr. 2005, 81 (Suppl. 1), 256S–260S. [Google Scholar] [PubMed]
- Del Rio, D.; Stalmach, A.; Calani, L.; Crozier, A. Bioavailability of coffee chlorogenic acids and green tea flavan-3-ols. Nutrients 2010, 2, 820–833. [Google Scholar] [CrossRef] [PubMed]
- Ozdal, T.; Sela, D.A.; Xiao, J.; Boyacioglu, D.; Chen, F.; Capanoglu, E. The Reciprocal Interactions between Polyphenols and Gut Microbiota and Effects on Bioaccessibility. Nutrients 2016, 8, 78. [Google Scholar] [CrossRef] [PubMed]
- Feliciano, R.P.; Boeres, A.; Massacessi, L.; Istas, G.; Ventura, M.R.; Nunes Dos Santos, C.; Heiss, C.; Rodriguez-Mateos, A. Identification and quantification of novel cranberry-derived plasma and urinary (poly)phenols. Arch. Biochem. Biophys. 2016, 599, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Ou, K.; Sarnoski, P.; Schneider, K.R.; Song, K.; Khoo, C.; Gu, L. Microbial catabolism of procyanidins by human gut microbiota. Mol. Nutr. Food Res. 2014, 58, 2196–2205. [Google Scholar] [CrossRef] [PubMed]
- Dall'Asta, M.; Calani, L.; Tedeschi, M.; Jechiu, L.; Brighenti, F.; del Rio, D. Identification of microbial metabolites derived from in vitro fecal fermentation of different polyphenolic food sources. Nutrition 2012, 28, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Mullen, W.; Edwards, C.A.; Crozier, A. Absorption, excretion and metabolite profiling of methyl-, glucuronyl-, glucosyl- and sulpho-conjugates of quercetin in human plasma and urine after ingestion of onions. Br. J. Nutr. 2006, 96, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Meyer, H.; Bolarinwa, A.; Wolfram, G.; Linseisen, J. Bioavailability of apigenin from apiin-rich parsley in humans. Ann. Nutr. Metab. 2006, 50, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Tang, D.; Chen, K.; Huang, L.; Li, J. Pharmacokinetic properties and drug interactions of apigenin, a natural flavone. Expert Opin. Drug Metab. Toxicol. 2017, 13, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.; Bai, Y.; Peng, W.; Su, W. Identification of Naringin Metabolites in Human Urine and Feces. Eur. J. Drug Metab. Pharmacokinet. 2016. [Google Scholar] [CrossRef] [PubMed]
- Pereira-Caro, G.; Ludwig, I.A.; Polyviou, T.; Malkova, D.; Garcia, A.; Moreno-Rojas, J.M.; Crozier, A. Identification of Plasma and Urinary Metabolites and Catabolites Derived from Orange Juice (Poly)phenols: Analysis by High-Performance Liquid Chromatography-High-Resolution Mass Spectrometry. J. Agric. Food Chem. 2016, 64, 5724–5735. [Google Scholar] [CrossRef] [PubMed]
- Aschoff, J.K.; Riedl, K.M.; Cooperstone, J.L.; Hogel, J.; Bosy-Westphal, A.; Schwartz, S.J.; Carle, R.; Schweiggert, R.M. Urinary excretion of Citrus flavanones and their major catabolites after consumption of fresh oranges and pasteurized orange juice: A randomized cross-over study. Mol. Nutr. Food Res. 2016, 60, 2602–2610. [Google Scholar] [CrossRef] [PubMed]
- Brett, G.M.; Hollands, W.; Needs, P.W.; Teucher, B.; Dainty, J.R.; Davis, B.D.; Brodbelt, J.S.; Kroon, P.A. Absorption, metabolism and excretion of flavanones from single portions of orange fruit and juice and effects of anthropometric variables and contraceptive pill use on flavanone excretion. Br. J. Nutr. 2009, 101, 664–675. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, D.; Calani, L.; Cordero, C.; Salvatore, S.; Pellegrini, N.; Brighenti, F. Bioavailability and catabolism of green tea flavan-3-ols in humans. Nutrition 2010, 26, 1110–1116. [Google Scholar] [CrossRef] [PubMed]
- Van der Hooft, J.J.; de Vos, R.C.; Mihaleva, V.; Bino, R.J.; Ridder, L.; de Roo, N.; Jacobs, D.M.; van Duynhoven, J.P.; Vervoort, J. Structural elucidation and quantification of phenolic conjugates present in human urine after tea intake. Anal. Chem. 2012, 84, 7263–7271. [Google Scholar] [CrossRef] [PubMed]
- Ottaviani, J.I.; Kwik-Uribe, C.; Keen, C.L.; Schroeter, H. Intake of dietary procyanidins does not contribute to the pool of circulating flavanols in humans. Am. J. Clin. Nutr. 2012, 95, 851–858. [Google Scholar] [CrossRef] [PubMed]
- Calani, L.; Del Rio, D.; Luisa Callegari, M.; Morelli, L.; Brighenti, F. Updated bioavailability and 48 h excretion profile of flavan-3-ols from green tea in humans. Int. J. Food Sci. Nutr. 2012, 63, 513–521. [Google Scholar] [CrossRef] [PubMed]
- Wiese, S.; Esatbeyoglu, T.; Winterhalter, P.; Kruse, H.P.; Winkler, S.; Bub, A.; Kulling, S.E. Comparative biokinetics and metabolism of pure monomeric, dimeric, and polymeric flavan-3-ols: A randomized cross-over study in humans. Mol. Nutr. Food Res. 2015, 59, 610–621. [Google Scholar] [CrossRef] [PubMed]
- Urpi-Sarda, M.; Monagas, M.; Khan, N.; Lamuela-Raventos, R.M.; Santos-Buelga, C.; Sacanella, E.; Castell, M.; Permanyer, J.; Andres-Lacueva, C. Epicatechin, procyanidins, and phenolic microbial metabolites after cocoa intake in humans and rats. Anal. Bioanal. Chem. 2009, 394, 1545–1556. [Google Scholar] [CrossRef] [PubMed]
- Appeldoorn, M.M.; Vincken, J.P.; Aura, A.M.; Hollman, P.C.; Gruppen, H. Procyanidin dimers are metabolized by human microbiota with 2-(3,4-dihydroxyphenyl)acetic acid and 5-(3,4-dihydroxyphenyl)-gamma-valerolactone as the major metabolites. J. Agric. Food Chem. 2009, 57, 1084–1092. [Google Scholar] [CrossRef] [PubMed]
- Bartolome, B.; Monagas, M.; Garrido, I.; Gomez-Cordoves, C.; Martin-Alvarez, P.J.; Lebron-Aguilar, R.; Urpi-Sarda, M.; Llorach, R.; Andres-Lacueva, C. Almond (Prunus dulcis (Mill.) D.A. Webb) polyphenols: From chemical characterization to targeted analysis of phenolic metabolites in humans. Arch. Biochem. Biophys. 2010, 501, 124–133. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wang, Y.; Li, D.; Ho, C.T.; Li, J.; Wan, X. The absorption, distribution, metabolism and excretion of procyanidins. Food Funct. 2016. [Google Scholar] [CrossRef] [PubMed]
- Pan, P.; Skaer, C.W.; Stirdivant, S.M.; Young, M.R.; Stoner, G.D.; Lechner, J.F.; Huang, Y.W.; Wang, L.S. Beneficial Regulation of Metabolic Profiles by Black Raspberries in Human Colorectal Cancer Patients. Cancer Prev. Res. (Phila.) 2015, 8, 743–750. [Google Scholar] [CrossRef] [PubMed]
- Serra, A.; Macià, A.; Romero, M.-P.; Reguant, J.; Ortega, N.; Motilva, M.-J. Metabolic pathways of the colonic metabolism of flavonoids (flavonols, flavones and flavanones) and phenolic acids. Food Chem. 2012, 130, 383–393. [Google Scholar] [CrossRef]
- Ludwig, I.A.; Mena, P.; Calani, L.; Borges, G.; Pereira-Caro, G.; Bresciani, L.; del Rio, D.; Lean, M.E.; Crozier, A. New insights into the bioavailability of red raspberry anthocyanins and ellagitannins. Free Radic. Biol. Med. 2015, 89, 758–769. [Google Scholar] [CrossRef] [PubMed]
- De Ferrars, R.M.; Cassidy, A.; Curtis, P.; Kay, C.D. Phenolic metabolites of anthocyanins following a dietary intervention study in post-menopausal women. Mol. Nutr. Food Res. 2014, 58, 490–502. [Google Scholar] [CrossRef] [PubMed]
- de Ferrars, R.M.; Czank, C.; Zhang, Q.; Botting, N.P.; Kroon, P.A.; Cassidy, A.; Kay, C.D. The pharmacokinetics of anthocyanins and their metabolites in humans. Br. J. Pharmacol. 2014, 171, 3268–3282. [Google Scholar] [CrossRef] [PubMed]
- Stalmach, A.; Mullen, W.; Barron, D.; Uchida, K.; Yokota, T.; Cavin, C.; Steiling, H.; Williamson, G.; Crozier, A. Metabolite profiling of hydroxycinnamate derivatives in plasma and urine after the ingestion of coffee by humans: Identification of biomarkers of coffee consumption. Drug Metab. Dispos. 2009, 37, 1749–1758. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Barrio, R.; Edwards, C.A.; Crozier, A. Colonic catabolism of ellagitannins, ellagic acid, and raspberry anthocyanins: In vivo and in vitro studies. Drug Metab. Dispos. 2011, 39, 1680–1688. [Google Scholar] [CrossRef] [PubMed]
- Selma, M.V.; Espin, J.C.; Tomas-Barberan, F.A. Interaction between phenolics and gut microbiota: Role in human health. J. Agric. Food Chem. 2009, 57, 6485–6501. [Google Scholar] [CrossRef] [PubMed]
- Margalef, M.; Pons, Z.; Bravo, F.I.; Muguerza, B.; Arola-Arnal, A. Plasma kinetics and microbial biotransformation of grape seed flavanols in rats. J. Funct. Foods 2015, 12, 478–488. [Google Scholar] [CrossRef]
- Pimpao, R.C.; Dew, T.; Figueira, M.E.; McDougall, G.J.; Stewart, D.; Ferreira, R.B.; Santos, C.N.; Williamson, G. Urinary metabolite profiling identifies novel colonic metabolites and conjugates of phenolics in healthy volunteers. Mol. Nutr. Food Res. 2014, 58, 1414–1425. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Mateos, A.; Cifuentes-Gomez, T.; Gonzalez-Salvador, I.; Ottaviani, J.I.; Schroeter, H.; Kelm, M.; Heiss, C.; Spencer, J.P. Influence of age on the absorption, metabolism, and excretion of cocoa flavanols in healthy subjects. Mol. Nutr. Food Res. 2015, 59, 1504–1512. [Google Scholar] [CrossRef] [PubMed]
- Mennen, L.I.; Sapinho, D.; Ito, H.; Galan, P.; Hercberg, S.; Scalbert, A. Urinary excretion of 13 dietary flavonoids and phenolic acids in free-living healthy subjects - variability and possible use as biomarkers of polyphenol intake. Eur. J. Clin. Nutr. 2008, 62, 519–525. [Google Scholar] [CrossRef] [PubMed]
- Tomas-Barberan, F.A.; Selma, M.V.; Espin, J.C. Interactions of gut microbiota with dietary polyphenols and consequences to human health. Curr. Opin. Clin. Nutr. Metab. Care 2016, 19, 471–476. [Google Scholar] [CrossRef] [PubMed]
- Marin, L.; Miguelez, E.M.; Villar, C.J.; Lombo, F. Bioavailability of dietary polyphenols and gut microbiota metabolism: Antimicrobial properties. Biomed. Res. Int. 2015, 2015, 905215. [Google Scholar] [CrossRef] [PubMed]



| Compound | [M − H]− | SRM Transition | S-Lens Value | Quantification |
|---|---|---|---|---|
| Catechol | 109 | 108, 81 | 68 | ND |
| Methylcatechol | 123 | 108, 81 | 68 | ND |
| Pyrogallol | 125 | 124, 81, 97 | 68 | ND |
| Hydroxybenzoic acid | 137 | 91, 93, 45 | 70 | ND |
| Hydroxyphenylacetic acid | 151 | 107 | 51 | ND |
| Dihydroxybenzoic acid | 153 | 108, 109 | 64 | ND |
| 3-(3’-Hydroxyphenyl)propionic acid | 165 | 119, 121 | 48 | ND |
| Vanillic acid | 167 | 152, 108, 123 | 60 | ND |
| Gallic acid | 169 | 125 | 68 | ND |
| Hippuric acid | 178 | 134 | 61 | Hippuric acid |
| (3′-Methoxy, 4′-hydroxyphenyl)acetic acid | 181 | 137 | 64 | ND |
| Dihydrocaffeic acid | 181 | 137, 119 | 64 | ND |
| Methylgallic acid | 183 | 168, 139 | 70 | ND |
| Catechol sulfate | 189 | 109, 81 | 70 | ND |
| (3′-Hydroxyphenyl)-γ-valerolactone | 191 | 147, 106 | 70 | ND |
| (4′-Hydroxyphenyl)-γ-valerolactone | 191 | 147, 173, 103, 107 | 70 | ND |
| Ferulic acid | 193 | 134, 178 | 71 | ND |
| (Hydroxyphenyl)-γ-valeric acid | 193 | 147, 149, 157, 175 | 72 | ND |
| 4-Hydroxyhippuric acid | 194 | 100, 150 | 72 | 4-Hydroxyhippuric acid |
| Dihydroferulic acid | 195 | 136 | 73 | ND |
| Syringic acid | 197 | 153, 182 | 70 | ND |
| Methylcatechol sulfate | 203 | 123, 108, 81 | 70 | ND |
| Pyrogallol sulfate | 205 | 125, 124, 81, 97 | 68 | Dihydrocaffeic acid 3-O-sulfate |
| (3′,5′-Dihydroxyphenyl)-γ-valerolactone | 207 | 163, 123, 121 | 75 | ND |
| (3′,4′-Dihydroxyphenyl)-γ-valerolactone | 207 | 163, 122 | 75 | ND |
| (3′,5′-Dihydroxyphenyl)-γ-valeric acid | 209 | 101, 124, 147 | 63 | ND |
| (3′,4′-Dihydroxyphenyl)-γ-valeric acid | 209 | 151, 165, 191, 194 | 63 | ND |
| Hydroxybenzoic acid sulfate | 217 | 137, 93, 45 | 70 | ND |
| Methyl-trihydroxybenzoic acid sulfate | 219 | 139, 124, 125, 81, 97 | 68 | Dihydroferulic acid 4′-O-sulfate |
| (3′,4′,5′-Trihydroxyphenyl)-γ-valerolactone | 223 | 179, 205, 138 | 75 | ND |
| Dihydroxybenzoic acid sulfate | 233 | 153, 108, 109 | 64 | Dihydrocaffeic acid 3-O-sulfate |
| Hydroxyphenylpropionic acid sulfate | 245 | 165, 121, 119 | 90 | Dihydrocaffeic acid 3-O-sulfate |
| Vanillic acid sulfate | 247 | 167, 152, 108, 123 | 90 | ND |
| Gallic acid sulfate | 249 | 169, 125 | 68 | ND |
| Feruloylglycine | 250 | 206, 134, 162, 191, 177 | 79 | Feruloylglycine |
| Dihydrocaffeic acid sulfate | 261 | 181, 137 | 96 | ND |
| Methylgallic acid sulfate | 263 | 183, 168, 125 | 68 | ND |
| Phenyl-γ-valerolactone-4′-O-sulfate | 271 | 191, 147 | 93 | ND |
| Phenyl-γ-valerolactone-3′-O-sulfate | 271 | 191, 147, 93, 80, 106 | 93 | Phenyl-γ-valerolactone-3′-O-sulfate |
| Ferulic acid sulfate | 273 | 193, 134, 178 | 92 | ND |
| Phenyl-γ-valeric acid‑O‑sulfate | 273 | 193, 175, 157, 149, 147 | 92 | ND |
| Dihydroferulic acid sulfate | 275 | 195, 136 | 75 | ND |
| (5′-Hydroxyphenyl)-γ-valerolactone-3′-O-sulfate | 287 | 207, 122, 163 | 96 | ND |
| (4′-hydroxyphenyl)-γ-valerolactone-3′-O-sulfate | 287 | 207, 109, 163 | 96 | (4′-Hydroxyphenyl)-γ-valerolactone-3′-O-sulfate |
| (Epi)catechin | 289 | 245, 203, 204.9 | 98 | ND |
| (Hydroxyphenyl)-γ-valeric acid‑O‑sulfate | 289 | 209, 191, 151, 147, 124, 101 | 92 | ND |
| Dihydroxyphenyl-γ-valerolactone-O-sulfate | 303 | 179, 223 | 90 | ND |
| Methyl(epi)catechin | 303 | 288, 245, 205 | 98 | ND |
| Hydroxybenzoic acid glucuronide | 313 | 137, 93, 45 | 70 | ND |
| (Methyl-hydroxyphenyl)-γ-valerolactone-O-sulfate | 317 | 222, 237 | 92 | ND |
| Dihydroxybenzoic acid glucuronide | 329 | 153, 108, 109 | 64 | ND |
| Gallic acid glucuronide | 345 | 169, 125 | 68 | ND |
| Apigenin sulfate | 349 | 269, 225 | 98 | ND |
| Naringenin sulfate | 351 | 271, 151 | 84 | ND |
| 3-Caffeoylquinic acid | 353 | 191, 179, 135 | 85 | ND |
| 5-Caffeoylquinic acid | 353 | 191 | 85 | ND |
| Caffeic acid glucuronide | 355 | 179, 135 | 87 | ND |
| Dihydrocaffeic acid glucuronide | 357 | 181, 137, 113 | 63 | ND |
| Kaempferol sulfate | 365 | 285, 257 | 90 | ND |
| Phenyl-γ-valerolactone-3′-O-glucuronide | 367 | 191, 113, 207 | 93 | ND |
| Phenyl-γ-valerolactone-3′,4′-di-O-sulfate | 367 | 287, 147 | 93 | ND |
| Ferulic acid glucuronide | 369 | 193, 178, 175 | 92 | Isoferulic acid 3′-O-glucuronide |
| (Epi)catechin sulfate | 369 | 289, 245, 203, 205 | 98 | ND |
| Dihydroxyphenyl-γ-valeric acid disulfate | 369 | 209, 191, 151, 147, 124 | 92 | ND |
| Diosmetin sulfate | 379 | 299, 284 | 90 | Quercetin 3′-O-sulfate |
| Quercetin 3′-sulfate | 381 | 301, 151, 179 | 83 | Quercetin 3′-O-sulfate |
| Hesperetin sulfate | 381 | 301, 151, 179 | 115 | Quercetin 3′-O-sulfate |
| (4′-hydroxyphenyl)-γ-valerolactone-3′-O-glucuronide | 383 | 207, 163 | 87 | (5′-hydroxyphenyl)-γ-valerolactone-3′-O-glucuronide |
| Methyl(epi)catechin sulfate | 383 | 303, 288, 245, 205 | 98 | ND |
| (Epi)gallocatechin sulfate | 385 | 305, 179, 221 | 98 | ND |
| Myricetin sulfate | 397 | 317, 316, 179 | 90 | ND |
| Dihydroxyphenyl-γ-valerolactone‑O‑glucuronide | 399 | 223, 175, 179 | 87 | ND |
| Methyl(epi)gallocatechin sulfate | 399 | 319, 304, 179, 221 | 98 | ND |
| Patuletin sulfate | 411 | 331, 316, 209 | 90 | Quercetin 3′-O-sulfate |
| Spinacetin sulfate | 425 | 345, 330 | 90 | ND |
| Apigenin glucuronide | 445 | 269, 225 | 90 | ND |
| Naringenin 4′-glucuronide | 447 | 271 151, 379, 119 | 112 | Naringenin 4′-O-glucuronide |
| Naringenin 7-glucuronide | 447 | 271, 151 | 84 | ND |
| Kaempferol glucuronide | 461 | 285, 257 | 90 | Quercetin 3-O-glucuronide |
| Phenyl-γ-valerolactone‑3′,4′-O-sulfate‑O-glucuronide | 463 | 163, 207, 287, 383 | 87 | ND |
| (Epi)catechin glucuronide | 465 | 289, 245, 205 | 98 | ND |
| Diosmetin glucuronide | 475 | 299, 284 | 90 | ND |
| Quercetin 3-glucuronide | 477 | 301, 151, 179 | 91 | Quercetin 3-O-glucuronide |
| Hesperetin 3′-glucuronide | 477 | 301, 113 | 115 | ND |
| Hesperetin 7-glucuronide | 477 | 301, 151 | 115 | Hesperetin 7-O-glucuronide |
| Methyl(epi)catechin glucuronide | 479 | 303, 288, 245, 205 | 98 | ND |
| Hydroxyphenyl-γ-valerolactone‑O-sulfate‑O-glucuronide | 479 | 303, 223, 175, 259 | 91 | ND |
| (Epi)gallocatechin glucuronide | 481 | 305, 179, 221 | 98 | ND |
| Myricetin glucuronide | 493 | 317, 316, 209 | 90 | Quercetin 3-O-glucuronide |
| Methyl(epi)gallocatechin glucuronide | 495 | 319, 304, 179, 221 | 98 | ND |
| Patuletin glucuronide | 507 | 331, 316, 209 | 90 | ND |
| Spinacetin glucuronide | 521 | 345, 330 | 90 | ND |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bresciani, L.; Martini, D.; Mena, P.; Tassotti, M.; Calani, L.; Brigati, G.; Brighenti, F.; Holasek, S.; Malliga, D.-E.; Lamprecht, M.; et al. Absorption Profile of (Poly)Phenolic Compounds after Consumption of Three Food Supplements Containing 36 Different Fruits, Vegetables, and Berries. Nutrients 2017, 9, 194. https://doi.org/10.3390/nu9030194
Bresciani L, Martini D, Mena P, Tassotti M, Calani L, Brigati G, Brighenti F, Holasek S, Malliga D-E, Lamprecht M, et al. Absorption Profile of (Poly)Phenolic Compounds after Consumption of Three Food Supplements Containing 36 Different Fruits, Vegetables, and Berries. Nutrients. 2017; 9(3):194. https://doi.org/10.3390/nu9030194
Chicago/Turabian StyleBresciani, Letizia, Daniela Martini, Pedro Mena, Michele Tassotti, Luca Calani, Giacomo Brigati, Furio Brighenti, Sandra Holasek, Daniela-Eugenia Malliga, Manfred Lamprecht, and et al. 2017. "Absorption Profile of (Poly)Phenolic Compounds after Consumption of Three Food Supplements Containing 36 Different Fruits, Vegetables, and Berries" Nutrients 9, no. 3: 194. https://doi.org/10.3390/nu9030194
APA StyleBresciani, L., Martini, D., Mena, P., Tassotti, M., Calani, L., Brigati, G., Brighenti, F., Holasek, S., Malliga, D.-E., Lamprecht, M., & Del Rio, D. (2017). Absorption Profile of (Poly)Phenolic Compounds after Consumption of Three Food Supplements Containing 36 Different Fruits, Vegetables, and Berries. Nutrients, 9(3), 194. https://doi.org/10.3390/nu9030194









