Vitamin A Supplementation Programs and Country-Level Evidence of Vitamin A Deficiency
Abstract
:1. Introduction
2. Materials and Methods
2.1. VAS Programs
2.2. Vitamin A Fortification, Bio-Fortification, and Micronutrient Powder Programs
2.3. Search Strategy for Vitamin A Deficiency Data and Inclusion and Exclusion Criteria
2.4. Deficiency and Coverage Definitions
3. Results
3.1. VAS Coverage and Overlap with Other VA Programs
3.2. Recentness of Vitamin A Deficiency Data
3.3. VAS Program and Deficiency Data Comparison
3.4. All Programs and Deficiency Data Comparison
4. Discussion
4.1. Implications of Outdated/Missing VAD Data
4.2. Filling the Data Gap
4.3. Alignment of Vitamin A Deficiency Results and Supplementation Programs
4.4. Global Debate and Engagement
4.5. Limitations
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- World Health Organization. Global Prevalence of Vitamin A Deficiency in Populations at Risk 1995–2005; World Health Organization: Geneva, Switzerland, 2009; Available online: http://apps.who.int/iris/bitstream/10665/44110/1/9789241598019_eng.pdf (accessed on 17 February 2017).
- Stevens, G.A.; Bennett, J.E.; Hennocq, Q.; Lu, Y.; De-Regil, L.M.; Rogers, L.; Danaei, G.; Li, G.; White, R.A.; Flaxman, S.R.; et al. Trends and mortality effects of vitamin A deficiency in children in 138 low-income and middle-income countries between 1991 and 2013: A pooled analysis of population-based surveys. Lancet Glob. Health 2015, 3, e528–e536. [Google Scholar] [CrossRef]
- Engle-Stone, R.; Haskell, M.J.; Nankap, M.; Ndjebayi, A.O.; Brown, K.H. Breast milk retinol and plasma retinol-binding protein concentrations provide similar estimates of vitamin A deficiency prevalence and identify similar risk groups among women in Cameroon but breast milk retinol underestimates the prevalence of deficiency among young children. J. Nutr. 2014, 144, 209–217. [Google Scholar] [PubMed]
- Stoltzfus, R.J.; Underwood, B.A. Breast-Milk vitamin A as an indicator of the vitamin A status of women and infants. Bull. World Health Organ. 1995, 73, 703–711. [Google Scholar] [PubMed]
- Alvarez, J.O.; Salazarlindo, E.; Kohatsu, J.; Miranda, P.; Stephensen, C.B. Urinary-excretion of retinol in children with acute diarrhea. Am. J. Clin. Nutr. 1995, 61, 1273–1276. [Google Scholar]
- Mitra, A.K.; Alvarez, J.O.; Guay-Woodford, L.; Fuchs, G.J.; Wahed, M.A.; Stephensen, C.B. Urinary retinol excretion and kidney function in children with shigellosis. Am. J. Clin. Nutr. 1998, 68, 1095–1103. [Google Scholar] [PubMed]
- Mitra, A.K.; Alvarez, J.O.; Stephensen, C.B. Increased urinary retinol loss in children with severe infections. Lancet 1998, 351, 1033–1034. [Google Scholar] [CrossRef]
- Beaton, G.; Martorell, R.; Aronson, K.; Edmonston, B.; McCabe, G.; Ross, A.; Harvey, B. Effectiveness of Vitamin A Supplementation in the Control of Young Child Morbidity and Mortality in Developing Countries; Administrative Committee on Coordination–Subcommittee on Nutrition (ACC/SCN): Geneva, Switzerland, 1993. [Google Scholar]
- Palmer, A.C.; West, K.P.; Dalmiya, N.; Schultink, W. The use and interpretation of serum retinol distributions in evaluating the public health impact of vitamin A programmes. Public Health Nutr. 2012, 15, 1201–1215. [Google Scholar] [CrossRef] [PubMed]
- Sommer, A.; Tarwotjo, I.; Djunaedi, E.; West, K.P., Jr.; Loeden, A.A.; Tilden, R.; Mele, L. Impact of vitamin A supplementation on childhood mortality. A randomised controlled community trial. Lancet 1986, 1, 1169–1173. [Google Scholar] [CrossRef]
- Beaton, G.; Martorell, R.; Aronson, K.; Edmonston, B.; McCabe, G.; Ross, A.; Harvey, B. Vitamin A supplementation and child morbidity and mortality in developing countries. Bol. Oficina Santitaria Panam. 1994, 117, 506–517. [Google Scholar]
- Imdad, A.; Herzer, K.; Mayo-Wilson, E.; Yakoob, M.Y.; Bhutta, Z.A. Vitamin A supplementation for preventing morbidity and mortality in children from 6 months to 5 years of age. Cochrane Database Syst. Rev. 2010. [Google Scholar] [CrossRef]
- Mayo-Wilson, E.; Imdad, A.; Herzer, K.; Yakoob, M.Y.; Bhutta, Z.A. Vitamin A supplements for preventing mortality, illness, and blindness in children aged under 5: Systematic review and meta-analysis. BMJ 2011, 343, d5094. [Google Scholar] [CrossRef] [PubMed]
- Awasthi, S.; Peto, R.; Read, S.; Clark, S.; Pande, V.; Bundy, D.; the DEVTA (Deworming and Enhanced Vitamin A) team. Vitamin A supplementation every 6 months with retinol in 1 million pre-school children in north India: DEVTA, a cluster-randomised trial. Lancet 2013, 381, 1469–1477. [Google Scholar] [CrossRef]
- Fisker, A.B.; Bale, C.; Rodrigues, A.; Balde, I.; Fernandes, M.; Jorgensen, M.J.; Danneskiold-Samsoe, N.; Hornshoj, L.; Rasmussen, J.; Christensen, E.D.; et al. High-Dose vitamin A with vaccination after 6 months of age: A randomized trial. Pediatrics 2014, 134, e739–e748. [Google Scholar] [CrossRef] [PubMed]
- Mason, J.; Greiner, T.; Shrimpton, R.; Sanders, D.; Yukich, J. Vitamin A policies need rethinking. Int. J. Epidemiol. 2015, 44, 283–292. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Guideline: Vitamin A Supplementation in Infants and Children 6–59 Months of Age; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- West, K.P.; Sommer, A.; Palmer, A.; Schultink, W.; Habicht, J.P. Commentary: Vitamin A policies need rethinking. Int. J. Epidemiol. 2015, 44, 292–294. [Google Scholar] [CrossRef] [PubMed]
- Bhutta, Z.A.; Baker, S.K. Premature abandonment of global vitamin A supplementation programmes is not prudent! Int. J. Epidemiol. 2015, 44, 297–299. [Google Scholar] [CrossRef] [PubMed]
- Benn, C.S.; Fisker, A.B.; Aaby, P. Response to: J Mason et al. Vitamin A policies need 447 rethinking. Int. J. Epidemiol. 2015, 44, 366–367. [Google Scholar] [CrossRef] [PubMed]
- Global Alliance for Vitamin A (GAVA). Technical Consultation on Guidance to Vitamin A Supplementation Programs for Children 6–59 Months of Age; GAVA: Ottawa, ON, Canada, 2012. [Google Scholar]
- United Nations International Children’s Fund. Data: Monitoring the Situation of Children and Women. 2016. Available online: http://data.unicef.org/nutrition/vitamin-a.html (accessed on 17 February 2017).
- Vitamin and Mineral Nutrition Information System. Summary Tables and Maps on Global Prevalence of Vitamin A Deficiency 1995–2005. Available online: http://www.who.int/vmnis/database/vitamina/status/en/ (accessed on 17 February 2017).
- Global Alliance for Improved Nutrition. Fortifying Our Future—A Snapshot on Food Fortification. 2015. Available online: http://www.gainhealth.org/wp-content/uploads/2015/05/Fortifying-our-Future-A-SnapShot-Report-on-Food-Fortification1.pdf (accessed on 17 February 2017).
- Global Alliance for Improved Nutrition. Programs—Global Tracking. 2016. Available online: http://www.gainhealth.org/programs/initiatives/#global-tracking (accessed on 17 February 2017).
- World Health Organization. Guidelines for Food Fortification. 2006. Available online: http://www.who.int/nutrition/publications/guide_food_fortification_micronutrients.pdf (accessed on 17 February 2017).
- Sight and Life: Food Fortification in Africa. Available online: http://www.sightandlife.org/fileadmin/data/Magazine/2013/27_3_2013/food_fortification_in_africa.pdf2013 (accessed on 17 February 2017).
- Klemm, R.D.W.; West, K.P.; Palmer, A.C.; Johnson, Q.; Randall, P.; Ranum, P.; Northrop-Clewes, C. Vitamin A fortification of wheat flour: Considerations and current recommendations. Food Nutr. Bull. 2010, 31, S47–SS61. [Google Scholar] [CrossRef] [PubMed]
- Food Fortification Initiative. Global Progress. 2016. Available online: http://www.ffinetwork.org/global_progress/index.php (accessed on 17 February 2017).
- United Nations International Children’s Fund; Centers for Disease Control and Prevention. UNICEF-CDC Global Assessment of Home Fortification Interventions, 2011 Micronutrient Powders Data; Home Fortification Technical Advisory Group: Geneva, Switzerland, 2012.
- United Nations Children’s Fund. NutriDash Platform-2016; United Nations Children’s Fund: New York, NY, USA, in press.
- HarvestPlus. Nutritious Staple Food Crops: Who Is Growing What? International Food Policy Research Institute: Washington, DC, USA, 2014. [Google Scholar]
- World Health Organization. Serum Retinol Concentrations for Determining the Prevalence of Vitamin A Deficiency in Populations. Vitamin and Mineral Nutrition Information System. Available online: http://www.who.int/vmnis/indicators/retinol.pdf (accessed on 17 February 2017).
- United Nations Children’s Fund. Vitamin A Supplementation: A Decade of Progress. 2007. Available online: http://www.unicef.org/publications/files/Vitamin_A_Supplementation.pdf (accessed on 17 February 2017).
- United Nations Children’s Fund. National Nutrition Survey Afghanistan (2013); Ministry of Public Health (MoPH) and UNICEF: Karachi, Pakistan, 2013.
- United Nations Children’s Fund. Assessing Vitamin A and Iron Deficiency Anaemia, Nutritional Anaemia among Children Aged 0–60 Months in the Republic of Angola (1998–1999); UNICEF: Luanda, Angola, 2000. [Google Scholar]
- Ministry of Health, United Nations Children’s Fund. Azerbaijan Nutrition Survey (AzNS), 2013; The U.S. Agency for International Development; World Bank Group; UNICEF: Baku, Azerbaijan, 2014.
- The International Centre for Diarrhoeal Disease Research, Bangladesh (ICDDR’B). National Micronutrients Status Survey 2011–12; Final Report; Centre for Nutrition and Food Security; UNICEF; GAIN; Institute of Public Health and Nutrition: Dhaka, Bangladesh, 2013. [Google Scholar]
- United Nations Children’s Fund. Benin: Enquete Nationale sur la Carence en Vitamine a et la Disponibilite en sel Iode Dans les Menages 2000; Ministere de la Sante Publique; Direction de la Sante Familiale; The United Nations Children’s Fund; USAID: New York, NY, USA, 1999.
- United Nations Children’s Fund. United Nations Children’s Fund. Micronutrient Malnutrition in Botswana. In A National Survey to Assess the Status of Iodine, Iron, and Vitamin A, 1996; Ministry of Health Wellness: Gaborone, Botswana, 1996. [Google Scholar]
- United Nations Children’s Fund. United Nations Children’s Fund. Burundi: Rapport de l’enquete Nationale de Nutrition de la Population, 2005. In Ministere de la Sante Publique, Programme de Lutte Contre les Maladies Transmissibles es Carentielles, 2006; The United Nations Children’s Fund: New York, NY, USA, 2005. [Google Scholar]
- National Institute of Statistics; Directorate General for Health; ICF International. Cambodia Demographic and Health Survey, 2014; National Institute of Statistics; Directorate General for Health; The DHS Program: Phnom Penh, Cambodia, 2015. [Google Scholar]
- Engle-Stone, R.; Ndjebayi, A.O.; Nankap, M.; Brown, K.H. Consumption of potentially fortifiable foods by women and young children varies by ecological zone and socio-economic status in Cameroon. J. Nutr. 2012, 142, 555–565. [Google Scholar] [CrossRef] [PubMed]
- United Nations Children’s Fund. Republique Centrafricaine, Enquête Nationale sur L’avitaminose A, la Carence en fer et la Consommation du sel iode, 2000; Ministere Delegue a l’Economie au Plan et a la Cooperation Internationale; Ministere de la Sante et de la Population; The United Nations Children’s Fund: New York, NY, USA, 2000.
- Rohner, F.; Northrop-Clewes, C.; Tschannen, A.B.; Bosso, P.E.; Kouassi-Gohou, V.; Erhardt, J.G.; Bui, M.; Zimmermann, M.B.; Mascie-Taylor, C.G. Prevalence and public health relevance of micronutrient deficiencies and undernutrition in pre-school children and women of reproductive age in Cote d’Ivoire, West Africa. Public Health Nutr. 2014, 17, 2016–2028. [Google Scholar] [CrossRef] [PubMed]
- Ministère de la Santé; Bureau National TDCI; UNICEF; CEPLANUT. Importance de la Carence en Vitamine A en Republique Democratique du Congo; Ministère de la Santé: Kinshasa, Democratic Republic of the Congo, 2000.
- The United Nations International Children’s Fund (UNICEF). National Survey for Assessment of Vitamin A Status in Egypt, 1995; Nutrition Institute, The United Nations International Children’s Fund: New York, NY, USA, 1995. [Google Scholar]
- Demissie, T.; Ali, A.; Mekonen, Y.; Haider, J.; Umeta, M. Magnitude and distribution of vitamin A deficiency in Ethiopia. Food Nutr. Bull. 2010, 31, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Bah, A.; Semega-Janneh, I.; Prentice, A.; Bates, C. Nationwide Survey on the Prevalence of Vitamin A and Iron Deficiency in Women and Children in the Gambia; National Agency: Lyon, France, 2001. [Google Scholar]
- Ministerio de Salud Publica y Asistencia Social (MSPAS). Guatemala: Encuesta Nacional de Micronutrientes 2009–2010, ENMICRON; Ministerio de Salud Publica y Asistencia Social: Guatemala City, Guatemala, 2012.
- Ministere de la Sante Publique et de la Population. Enquete sur la Prevlence de la Carence en Vitamine A et de la Deficience end iode end Haiti; Ministere de la Sante Publique et de la Population; United Nations Children’s Fund: Port-au-Prince, Haiti, 2005.
- Nestel, P.; Melara, A.; Rosado, J.; Mora, J.O. Vitamin A deficiency and anemia among children 12–71 months old in Honduras. Rev. Panam. Salud Publica 1999, 6, 34–43. [Google Scholar] [CrossRef] [PubMed]
- Sandjaja, S.; Budiman, B.; Harahap, H.; Ernawati, F.; Soekatri, M.; Widodo, Y.; Sumedi, E.; Rustan, E.; Sofia, G.; Syarief, S.N.; Khouw, I. Food consumption and nutritional and biochemical status of 0.5–12-year-old Indonesian children: the SEANUTS study. Br. J. Nutr. 2013, 110, S11–S20. [Google Scholar] [CrossRef] [PubMed]
- Kenya Medical Research Institute and Ministry of Health. The Kenya National Micronutrient Survey 2011; Kenya Medical Research Institute and Ministry of Health: Nairobi, Kenya, 2016.
- Ministry of Health, National Statistics Committee, CDC, UNICEF. Follow-Up Survey of Nutritional Status in Children 6–29 Months of Age, Kyrgyz Republic 2013; United Nations Children’s Fund: Bishkek, Kyrgyz Republic, 2015.
- Ministry of Health, National Institute of Public Health. Report on National Health Survey: Health Status of the People of LAO PDR; Ministry of Health: Vientiane, Lao PDR, 2001.
- Wolde-Gebriel, Z. National Survey on Iodine, Vitamin A and Iron Status of Women and Children in Lesotho; Ministry of Health, UNICEF: Maseru, Lesotho, 1994.
- United Nations Children’s Fund. Liberia National Micronutrient Survey 2011- Selected Preliminary Findings; The United Nations Children’s Fund; Liberia Institute of Statistics: Monrovia, Liberia, 2011. [Google Scholar]
- U.S. Agency for International Development Micronutrient Progam (MOST). Enquête sur la Carence en Vitamine A chez les Femmes et les Enfants et Enquête sur l’Anémie chez les Ecoliers de 6 à 14 Ans-Madagascar 2000. Available online: http://pdf.usaid.gov/pdf_docs/Pnadc603.pdf (accessed on 17 February 2017).
- Ministry of Health. Malawi- National Micronutrient Survey 2009; Department of Nutrition, HIV and AIDS in the Office of President and Cabinet (DHNA-OPC); Ministry of Health (MOH); National Statistics Office (NSO); UNICEF; CDC: Lilongwe, Malawi, 2011.
- United Nations Children’s Fund. National Micronutrient Survey 2007—Maldives. Available online: http://www.unicef.org/maldives/micro_final_report-15_Aug_2010.pdf (accessed on 17 February 2017).
- Palafox, N.A.; Gamble, M.V.; Dancheck, B.; Ricks, M.O.; Briand, K.; Semba, R.D. Vitamin A deficiency, iron deficiency, and anemia among preschool children in the Republic of the Marshall Islands. Nutrition 2003, 19, 405–408. [Google Scholar] [CrossRef]
- Villalpando, S.; Cruz Vde, L.; Shamah-Levy, T.; Rebollar, R.; Contreras-Manzano, A. Nutritional status of iron, vitamin B12, folate, retinol and anemia in children 1 to 11 years old: Results of the Ensanut 2012. Salud Publica Mex. 2015, 57, 372–384. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Health. Nutrition Status of Mongolian Population; Fourth National Nutrition Survey Report, 2010; Minstry of Health; Public Health Institute; Nutrition Research Centre; UNICEF; WHO: Ulaanbaatar, Mongolia, 2011.
- Nasri, I.; El Bouhali, B.; Aguenaou, H.; Mokhtar, N. Vitamin A deficiency among Moroccan women and children. Afr. Health Sci. 2004, 4, 3–8. [Google Scholar] [PubMed]
- United Nations Children’s Fund. Mozambique: National Survey on Vitamin A Deficiency, Anemia, and Malaria; The United Nations Children’s Fund; Ministry of Health; World Health Organization: Geneva, Switzerland, 2002.
- United Nations Children’s Fund. Iodine Deficiency Disorders in Namibia and Data in the Status of Vitamin A and Iron; International Council for the Control of Iodine Deficiency Disorders (ICCIDD); Ministry of Health and Social Services (Namibia); United Nations Children’s Fund: New York, NY, USA, 1992.
- Gorstein, J.; Shreshtra, R.K.; Pandey, S.; Adhikari, R.K.; Pradhan, A. Current status of vitamin A deficiency and the National Vitamin A Control Program in Nepal: results of the 1998 National Micronutrient Status Survey. Asia Pac. J. Clin. Nutr. 2003, 12, 96–103. [Google Scholar] [PubMed]
- Gurdián, M.; Kontorovsky, I.; Alvarado, E.; Ramírez, S.; Hernández, R. Sistema Integrado de Vigilancia de Intervenciones Nutricionales (SIVIN). In Informe de Progreso. Nicaragua, Ministerio de Salud, 2005; Comision Tecnica de Seguridad Alimentaria y Nutricional: Managua, Nicaragua, 2005. [Google Scholar]
- Maziya-Dixon, B.B.; Akinyele, I.O.; Sanusi, R.A.; Oguntona, T.E.; Nokoe, S.K.; Harris, E.W. Vitamin A deficiency is prevalent in children less than 5 years of age in Nigeria. J. Nutr. 2006, 136, 2255–2261. [Google Scholar] [PubMed]
- Bhutta, Z.A. National Nutrition Survey, Pakistan, 2011. Available online: http://gilanifoundation.com/homepage/Free_Pub/HEALTH/National_Nutrition_Survey_of_Pakistan_2011.pdf (accessed on 17 February 2017).
- Department of Public Health. National Micronutrient Survey Papua New Guinea, 2005—Summary Report; UNICEF; Department of Public Health of Papua New Guinea; University of Papua New Guinea; Center for Disease Control and Prevention: Port Moresby, Papua New Guinea, 2006.
- Food and Nutrition Research Institute (FNRI); Metro Manila, Philippines. National Nutrition Survey Philippines 2013. Personal communication, 2013. [Google Scholar]
- United Nations Children’s Fund. National Nutrition Survey of Women and Children in Rwanda in 1996; Ministry of Health (Rwanda); United Nations Children’s Fund; World Health Organization (WHO): Kigali, Rwanda, 1997.
- Laboratoire de Nutrition-Université Cheikh Anta DIOP (UCAD), Comité Sénégalais Pour la Fortification des Aliments en Micronutriments (COSFAM), Micronutrient Initiative. Situation de Base du Statut en Vitamine A et en fer chez les Enfants de 12–59 Mois et Chez les Femmes en age de Procreer (15–49 ans) Dans le Cadre du Programme de Fortification des Aliments en Micronutriments au Senegal; Republique du Senegal: Dakar, Senegal, 2011.
- Ministry of Health and Sanitation. 2013 Sierra Leone Micronutrient Survey. Available online: http://groundworkhealth.org/wp-content/uploads/2016/03/SLMS-Report_FINAL_151203.pdf (accessed on 17 February 2017).
- Food Security and Nutrition Analysis Unit. National Micronutrient and Anthropometric Nutrition Survey, Somalia 2009; Food Security and Nutrition Analysis Unit; SIDA; WHO; UNICEF; Ministry of Health: Wellington, Australia, 2009.
- Shisana, O.; Labadarios, D.; Rehle, T.; Simbayi, L.; Zuma, K.; Dhansay, A.; Reddy, P.; Parker, W.; Hoosain, E.; Naidoo, P.; et al. The South African National Helath and Nutrition Examination Survey, 2012 SANHANES-1: The Health and Nutritional Status of the Nation; HSRC Press: Cape Town, South Africa, 2014. [Google Scholar]
- United Nations Children’s Fund. Vitamin A Nutrition Status in Sri Lanka 2006; Ministry of Healthcare and Nutrition; The United Nations Children’s Fund: New York, NY, USA, 2006.
- National Bureau of Statistics (NBS). Micronutrients: Results of the 2010 Tanzania Demographic and Health Survey. Available online: http://pdf.usaid.gov/pdf_docs/PNADY494.pdf (accessed on 17 February 2017).
- United Nations Children’s Fund. Timor-Leste Food and Nutrition Survey 2013, Ministry of Health Final Report; United Nations Chilren’s Fund; Ministry of Health: New York, NY, USA, 2015.
- Uganda Bureau of Statistics; ICF International. Uganda Demographic and Health Survey 2011—Addendum to Chapter 11; Uganda Bureau of Statistics; ICF International: Kampala, Uganda, 2012.
- Laillou, A.; Pham, T.V.; Tran, N.T.; Le, H.T.; Wieringa, F.; Rohner, F.; Fortin, S.; Le, M.B.; Tran do, T.; Moench-Pfanner, R.; et al. Micronutrient deficits are still public health issues among women and young children in Vietnam. PLoS ONE 2012, 7, e34906. [Google Scholar] [CrossRef] [PubMed]
- USAID. Report of the National Survery to Evaluate the Impact of Vitamin A Interventions in Zambia, July and November 2003; Micronutrient Operational Strategies and Technologies (MOST); Centers for Disease Control and Prevention (CDC); Food and Nutrition Commision of Zambia; University of Zambia; The United Nations Children’s Fund: New York, NY, USA, 2003.
- United Nations Children’s Fund. Zimbabwe National Micronutrient Survey: 1999; Ministry of Health and Child Welfare; Nutrition Unit; The United Nations International Children’s Fund: New York, NY, USA, 2001.
- Tanumihardjo, S.A. Vitamin A fortification efforts require accurate monitoring of population vitamin A status to prevent excessive intakes. Procedia Chem. 2015, 14, 398–407. [Google Scholar] [CrossRef]
- Sablah, M.; Klopp, J.; Steinberg, D.; Touaoro, Z.; Laillou, A.; Baker, S. Thriving public-private partnership to fortify cooking oil in the West African Economic and Monetary Union (UEMOA) to control vitamin A deficiency: Faire Tache d’Huile en Afrique de l’Ouest. Food Nutr. Bull. 2012, 33, S310–S320. [Google Scholar] [CrossRef] [PubMed]
- Rohner, F.; Leyvraz, M.; Konan, A.G.; Esso, L.J.C.E.; Wirth, J.P.; Norte, A.; Adiko, A.F.; Bonfoh, B.; Aaron, G.J. The Potential of Food Fortification to Add Micronutrients in Young Children and Women of Reproductive Age—Findings from a Cross-Sectional Survey in Abidjan, Cote d’Ivoire. PLoS ONE 2016, 11, e0158552. [Google Scholar] [CrossRef] [PubMed]
- Luthringer, C.L.; Rowe, L.A.; Vossenaar, M.; Garrett, G.S. Regulatory Monitoring of Fortified Foods: Identifying Barriers and Good Practices. Glob. Health Sci. Pract. 2015, 3, 446–461. [Google Scholar] [CrossRef] [PubMed]
- Tanumihardjo, S.A.; Russe, R.M.; Stephensen, C.B.; Gannon, B.M.; Craft, N.E.; Haskell, M.J.; Lietz, G.; Schulze, K.; Raiten, D.J. Biomarkers of Nutrition for Development (BOND)-Vitamin A Review. J. Nutr. 2016, 146, 1816–1848. [Google Scholar] [CrossRef] [PubMed]
- Raiten, D.J.; Ashour, F.A.S.; Ross, A.C.; Meydani, S.N.; Dawson, H.D.; Stephensen, C.B.; Brabin, B.J.; Suchdev, P.S.; van Ommen, B.; Grp, I.C. Inflammation and Nutritional Science for Programs/Policies and Interpretation of Research Evidence (INSPIRE). J. Nutr. 2015, 145, 1039s–1108s. [Google Scholar] [CrossRef] [PubMed]
- Thurnham, D.I.; Northrop-Clewes, C.A.; Knowles, J. The Use of Adjustment Factors to Address the Impact of Inflammation on Vitamin A and Iron Status in Humans. J. Nutr. 2015, 145, 1137s–1143s. [Google Scholar] [CrossRef] [PubMed]
- Larson, L.; Addo, O.Y.; Sandalinas, F.; Baawo, K.; Faigao, K.; Kupka, R.; Flores-Ayala, R.; Suchdev, P. Accounting for the Influence of Inflammation on Retinol Binding Protein in a Population Survey of Liberian Preschool-Aged Children. FASEB J. 2016. [Google Scholar] [CrossRef]
- Greig, A.; Neufeld, L. A Framework for Shifting from Universal Vitamin A Supplementation: Technical Consultation on Guidance for VAS Programs for Children 6–59 Months of Age. Sight Life 2013, 27, 76–77. [Google Scholar]
- Tanumihardjo, S.A.; Mokhtar, N.; Haskell, M.J.; Brown, K.H. Assessing the Safety of Vitamin A Delivered Through Large-Scale Intervention Programs: Workshop Report on Setting the Research Agenda. Food Nutr. Bull. 2016, 37, S63–S74. [Google Scholar] [CrossRef] [PubMed]
- Sandjaja, J.I.; Jahari, A.B.; Ifrad, H.M.K.; Tilden, R.L.; Soekarjo, D.; Utomo, B.; Moench-Pfanner, R.; Soekirman; Korenromp, E.L. Vitamin A-fortified cooking oil reduces vitamin A deficiency in infants, young children and women: results from a programme evaluation in Indonesia. Public Health Nutr. 2015, 18, 2511–2522. [Google Scholar] [CrossRef] [PubMed]
- Global Alliance for Improved Nutrition. Fortification of Vegetable Oil in Indonesia. Available online: http://www.gainhealth.org/knowledge–centre/project/fortification–vegetable–oil–indonesia/ (accessed on 17 February 2017).
- Home Fortification Technical Advisory Group. Home Fortification with Micronutrient Powders (MNP). Available online: http://hftag.org/2157_Projects.asp?tax_product_type=mnp&wpas=1&product–label=Micronutrient%20powders%20(MNP) (accessed on 17 February 2017).
- Centers for Disease Control and Prevention: International Micronutrient Malnutrition Prevention and Control (IMMPaCt) —Kyrgyzstan. 2015. Available online: http://www.cdc.gov/immpact/projects/kyrgyzstan.html (accessed on 17 February 2017).
- Hotz, C.; Chileshe, J.; Siamusantu, W.; Palaniappan, U.; Kafwembe, E. Vitamin A intake and infection are associated with plasma retinol among pre-school children in rural Zambia. Public Health Nutr. 2012, 15, 1688–1696. [Google Scholar] [CrossRef] [PubMed]
- Bresnahan, K.A.; Chileshe, J.; Arscott, S.; Nuss, E.; Surles, R.; Masi, C.; Kafwembe, E.; Tanumihardjo, S.A. The Acute Phase Response Affected Traditional Measures of Micronutrient Status in Rural Zambian Children during a Randomized, Controlled Feeding Trial. J. Nutr. 2014, 144, 972–978. [Google Scholar] [CrossRef] [PubMed]
- Gannon, B.; Kaliwile, C.; Arscott, S.A.; Schmaelzle, S.; Chileshe, J.; Kalungwana, N.; Mosonda, M.; Pixley, K.; Masi, C.; Tanumihardjo, S.A. Biofortified orange maize is as efficacious as a vitamin A supplement in Zambian children even in the presence of high liver reserves of vitamin A: A community-based, randomized placebo-controlled trial. Am. J. Clin. Nutr. 2014, 100, 1541–1550. [Google Scholar] [CrossRef] [PubMed]
- Tanumihardjo, S.A.; Gannon, B.M.; Kaliwile, C.; Chileshe, J. Hypercarotenodermia in Zambia: Which children turned orange during mango season? Eur. J. Clin. Nutr. 2015, 69, 1346–1349. [Google Scholar] [CrossRef] [PubMed]
- Palmer, A.C.; Siamusantu, W.; Chileshe, J.; Schulze, K.J.; Barffour, M.; Craft, N.E.; Molobeka, N.; Kalungwana, N.; Arguello, M.A.; Mitra, M.; et al. Provitamin A-biofortified maize increases serum beta-carotene, but not retinol, in marginally nourished children: A cluster-randomized trial in rural Zambia. Am. J. Clin. Nutr. 2016, 104, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Sesay, F.F.; Hodges, M.H.; Kamara, H.I.; Turay, M.; Wolfe, A.; Samba, T.T.; Koroma, A.S.; Kamara, W.; Fall, A.; Mitula, P.; et al. High coverage of vitamin A supplementation and measles vaccination during an integrated Maternal and Child Health Week in Sierra Leone. Int. Health 2015, 7, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Klemm, R.D.W.; Palmer, A.C.; Greig, A.; Engle-Stone, R.; Dalmiya, N. A Changing Landscape for Vitamin A Programs: Implications for Optimal Intervention Packages, Program Monitoring, and Safety. Food Nutr. Bull. 2016, 37, S75–S86. [Google Scholar] [CrossRef] [PubMed]
- Tanumihardjo, S.; Ball, A.; Kaliwile, C.; Pixley, K. The research and implementation continuum of biofortified sweetpotato and maize in Africa. Ann. N. Y. Acad. Sci. 2017. [Google Scholar] [CrossRef] [PubMed]
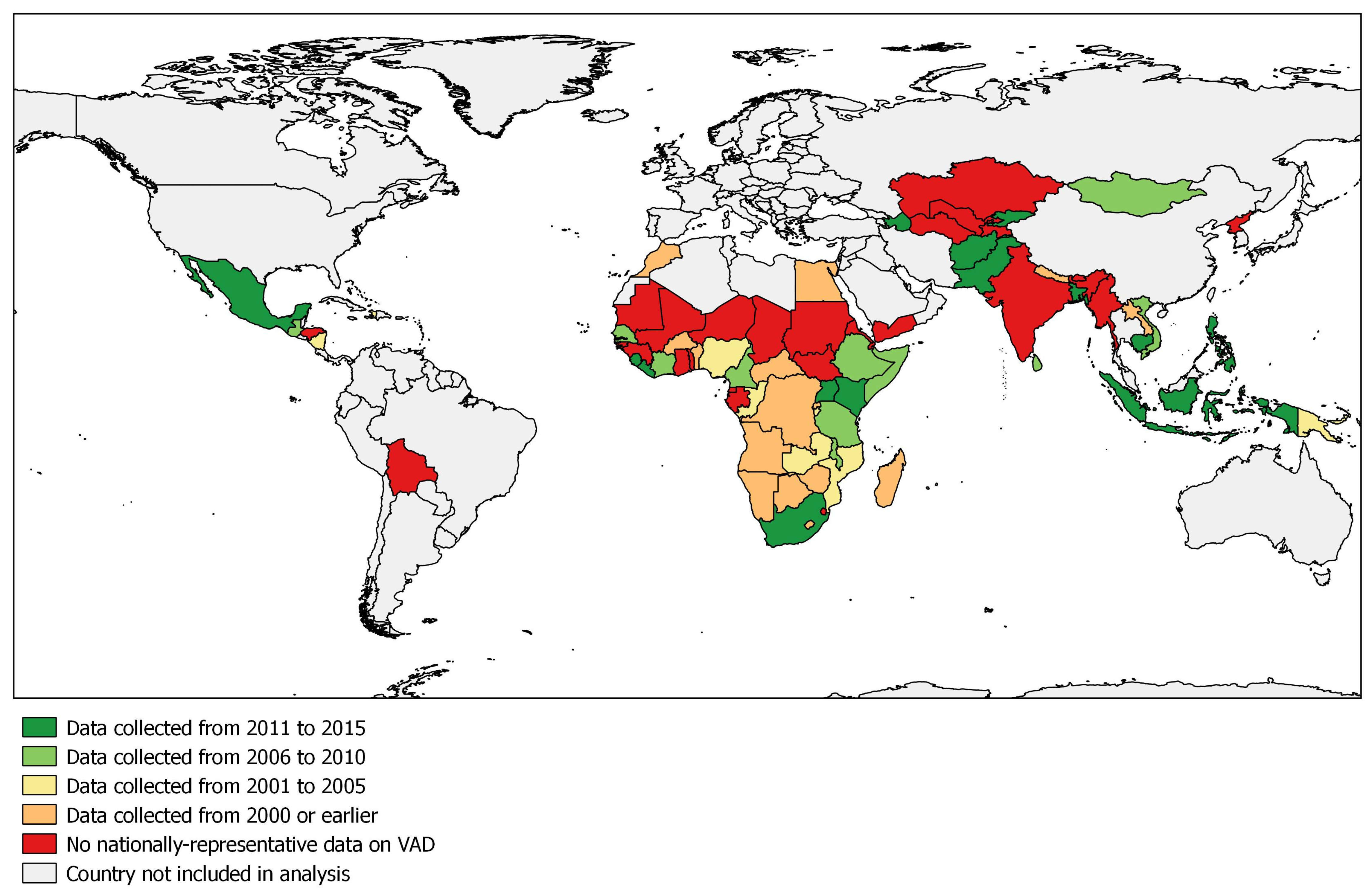
| Countries and Territories | Coverage (%) of VAS Program | Year of Coverage Estimate | VA Fortification, Biofortification, and MNP Programs 2 | Year of Most Recent Nationally-Representative VAD Survey | Biomarker 3 | VAD Prevalence (%) 4 | Severity of VAD | Source |
|---|---|---|---|---|---|---|---|---|
| Afghanistan | 95 | 2014 | fVO (v), fW (v) | 2013 | ROH | 50.4 | Severe | [35] |
| Angola | 6 | 2014 | bSP (v) | 1999 | ROH | 64.3 | Severe | [36] * |
| Azerbaijan | 58 | 2014 | 2013 | RBP | 8.0 | Mild | [37] | |
| Bangladesh | 0 | 2014 | fVO (m), fW (v), MNP (v) | 2011 | ROH | 20.5 | Severe | [38] |
| Benin | 99 | 2014 | fVO (m) | 1999 | ROH | 82.0 | Severe | [39] * |
| Bhutan | 45 | 2013 | ||||||
| Bolivia | 40 | 2013 | fVO (m), MNP (v) | |||||
| Botswana | 70 | 2014 | 1994 | ROH | 35.4 | Severe | [40] * | |
| Burkina Faso | 98 | 2014 | fVO (m), bSP (v) | |||||
| Burundi | 68 | 2014 | fVO (v), bP (v) | 2005 | ROH | 27.9 | Severe | [41] |
| Cambodia | 71 | 2014 | fVO (v), MNP (v) | 2014 | RBP | 9.2 | Mild | [42] |
| Cameroon | 96 | 2014 | fVO (m), bP (v) | 2009 | RBP | 35.0 †† | Severe | [43] |
| Central African Rep | 34 | 2014 | 1999 | ROH | 68.2 | Severe | [44] * | |
| Chad | 96 | 2014 | ||||||
| Comoros | 14 | 2014 | ||||||
| Congo | 99 | 2014 | ||||||
| Côte d’Ivoire | 99 | 2014 | fVO (m) | 2007 | RBP | 24.1 | Severe | [45] |
| DRC | 99 | 2014 | bC (v), bP (v) | 1998/99 | -ROH | 61.1 | Severe | [46] |
| Djibouti | 66 | 2013 | MNP (v) | |||||
| Egypt | 68 | 2008 | fVO (v) | 1995 | ROH | 11.9 | Moderate | [47] * |
| Equatorial Guinea | ||||||||
| Eritrea | 49 | 2014 | ||||||
| Ethiopia | 71 | 2014 | bSP (v) | 2006 ‡ | ROH | 37.7 | Severe | [48] |
| Gabon | 2 | 2012 | ||||||
| Gambia | 27 | 2014 | 1999 | ROH | 64.0 | Severe | [49] * | |
| Ghana | 23 | 2014 | fVO (m), fW (m), bSP (v), bM (v) | |||||
| Guatemala | 19 | 2014 | fMG (m), fS (m), MNP (v) | 2009/10 | ROH | 0.3 | None | [50] |
| Guinea | 99 | 2012 | fVO (m) | |||||
| Guinea-Bissau | 98 | 2014 | fVO (m) | |||||
| Haiti | 30 | 2014 | MNP (v) | 2005 | ROH | 32.0 | Severe | [51] * |
| Honduras | 40 | 2005 | fMG (m), fS (m) | 1996 | ROH | 13.8 | Moderate | [52] |
| India | 61 | 2014 | fVO (v), bSP (v) | |||||
| Indonesia | 84 | 2014 | fVO (m), fW (m) | 2011 | ROH | <1 | None | [53] |
| Kazakhstan | ||||||||
| Kenya | 28 | 2014 | fVO (m), fS (v), bSP (v) | 2012 | RBP | 9.2 | Mild | [54] |
| Kiribati | 54 | 2006 | ||||||
| Kyrgyzstan | 97 | 2010 | MNP (v) | 2013 | RBP | 7.8 | Mild | [55] |
| Laos | 89 | 2014 | 2000 | ROH | 44.7 | Severe | [56] | |
| Lesotho | 67 | 2014 | fW (v) | 1993 | ROH | 78.0 | Severe | [57] * |
| Liberia | 0 | 2014 | fVO (m), fS (m) | 2011 | RBP | 13.2 | Moderate | [58] |
| Madagascar | 99 | 2014 | bSP (v), MNP (v) | 2000 | ROH | 42.1 | Severe | [59] |
| Malawi | 41 | 2014 | fVO (m), fS (m), bSP (v), bC (v), MNP (v) | 2009 | RBP | 22.0 †† | Severe | [60] |
| Maldives | 76 | 2013 | 2007 | ROH | 5.1 | Mild | [61] | |
| Mali | 98 | 2013 | fVO (v), bM (v) | |||||
| Marshall Islands | 39 | 2007 | 1995 | ROH | 59.9 | Severe | [62] | |
| Mauritania | 89 | 2014 | fVO (m) | |||||
| Mexico | fMG (m), MNP (v) | 2011/12 | ROH | 15.7 | Moderate | [63] | ||
| Micronesia | 68 | 2006 | ||||||
| Mongolia | 79 | 2014 | MNP (v) | 2010 | RBP | 32.4 | Severe | [64] |
| Morocco | fVO (m) | 1996 | ROH | 40.4 | Severe | [65] | ||
| Mozambique | 99 | 2014 | fVO (m), bSP (v), MNP (v) | 2002 | ROH | 68.8 | Severe | [66] |
| Myanmar | 94 | 2014 | MNP (v) | |||||
| Namibia | 62 | 2013 | 1992 | ROH | 23.5 | Severe | [67] * | |
| Nepal | 85 | 2014 | MNP (v) | 1998 ‡ | ROH | 32.3 | Severe | [68] |
| Nicaragua | 4 | 2014 | fS (m), MNP (v) | 2004 | ROH | 2.1 | Mild | [69] |
| Niger | 95 | 2014 | fVO (m), fS (m), bSP (v) | |||||
| Nigeria | 80 | 2014 | fVO (m), fW (m), bSP (v), bM (v), bC (v), bP (v) | 2001 | ROH | 29.5 | Severe | [70] |
| DPR Korea | 99 | 2014 | MNP (v) | |||||
| Pakistan | 96 | 2014 | fVO (m) | 2011 | ROH | 54.0 | Severe | [71] |
| Papua New Guinea | 15 | 2012 | 2005 | RBP | 15.7 | Moderate | [72] | |
| Philippines | 83 | 2014 | fVO (m), fW (m), MNP (v) | 2013 | ROH | 20.4 | Severe | [73] |
| Rwanda | 95 | 2014 | fVO (m), fS (m), bSP (v), MNP (v) | 1996 | ROH | 6.4 | Moderate | [74] |
| Sao Tome and Principe | 56 | 2013 | ||||||
| Senegal | 89 | 2014 | fVO (m), bSP (v), MNP (v) | 2010 | ROH | 17.7 | Severe | [75] |
| Sierra Leone | 8 | 2014 | fVO (m), bC (v) | 2013 | RBP | 17.4 | Moderate | [76] |
| Somalia | 30 | 2014 | MNP (v) | 2009 | RBP | 33.3 †† | Severe | [77] |
| South Africa | 42 | 2013 | fMG (v), fW (m) | 2012 | ROH | 43.6 | Severe | [78] |
| South Sudan | 18 | 2014 | ||||||
| Sri Lanka | 72 | 2014 | MNP (v) | 2006 | ROH | 29.3 | Severe | [79] |
| Sudan | 99 | 2014 | ||||||
| Swaziland | 43 | 2014 | ||||||
| Tajikistan | 99 | 2014 | ||||||
| Tanzania | 88 | 2014 | fVO (m), fS (m), bSP (v), MNP (v) | 2010 | RBP | 33.0 †† | Severe | [80] |
| Timor-Leste | 40 | 2013 | 2013 | RBP | 9.7 | Mild | [81] | |
| Turkmenistan | ||||||||
| Togo | 61 | 2013 | fVO (m) | |||||
| Uganda | 66 | 2014 | fVO (m), fW (m), bSP (v) | 2011 | RBP | 32.6 †† | Severe | [82] |
| Uzbekistan | 99 | 2014 | ||||||
| Vietnam | 94 | 2014 | fVO (m) | 2010 | ROH | 10.1 | Moderate | [83] |
| Yemen | 7 | 2014 | ||||||
| Zambia | 93 | 2013 | fVO (v), fS (m), bSP (v), bM (v) | 2003 | ROH | 54.1 | Severe | [84] |
| Zimbabwe | 32 | 2014 | fVO (v) | 1999 | ROH | 35.8 | Severe | [85] * |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wirth, J.P.; Petry, N.; Tanumihardjo, S.A.; Rogers, L.M.; McLean, E.; Greig, A.; Garrett, G.S.; Klemm, R.D.W.; Rohner, F. Vitamin A Supplementation Programs and Country-Level Evidence of Vitamin A Deficiency. Nutrients 2017, 9, 190. https://doi.org/10.3390/nu9030190
Wirth JP, Petry N, Tanumihardjo SA, Rogers LM, McLean E, Greig A, Garrett GS, Klemm RDW, Rohner F. Vitamin A Supplementation Programs and Country-Level Evidence of Vitamin A Deficiency. Nutrients. 2017; 9(3):190. https://doi.org/10.3390/nu9030190
Chicago/Turabian StyleWirth, James P., Nicolai Petry, Sherry A. Tanumihardjo, Lisa M. Rogers, Erin McLean, Alison Greig, Greg S. Garrett, Rolf D. W. Klemm, and Fabian Rohner. 2017. "Vitamin A Supplementation Programs and Country-Level Evidence of Vitamin A Deficiency" Nutrients 9, no. 3: 190. https://doi.org/10.3390/nu9030190
APA StyleWirth, J. P., Petry, N., Tanumihardjo, S. A., Rogers, L. M., McLean, E., Greig, A., Garrett, G. S., Klemm, R. D. W., & Rohner, F. (2017). Vitamin A Supplementation Programs and Country-Level Evidence of Vitamin A Deficiency. Nutrients, 9(3), 190. https://doi.org/10.3390/nu9030190





