Role of Vitamin A/Retinoic Acid in Regulation of Embryonic and Adult Hematopoiesis
Abstract
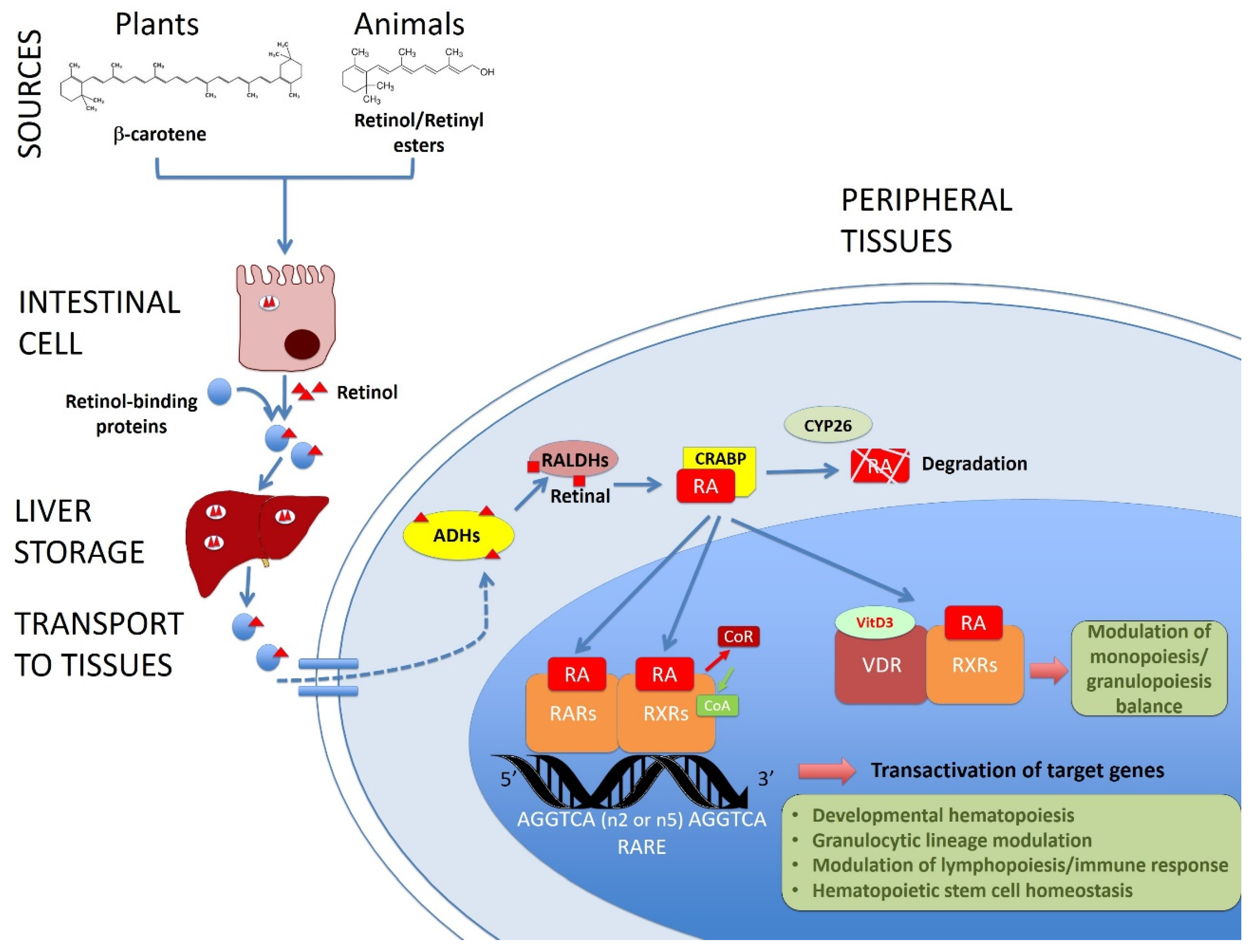
:1. Introduction
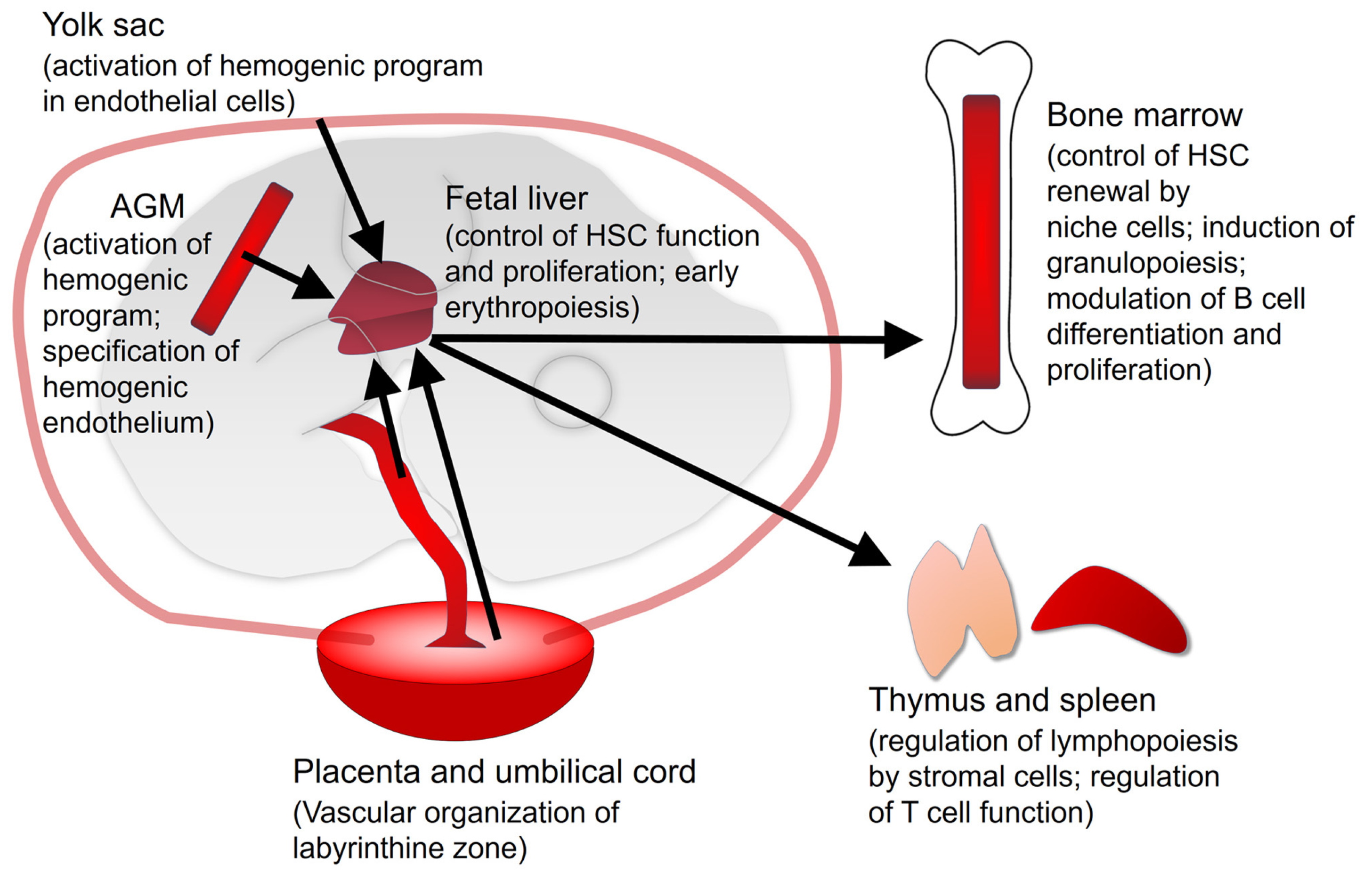
2. Vitamin A/Retinoic Acid in Developmental Hematopoiesis
2.1. Yolk Sac
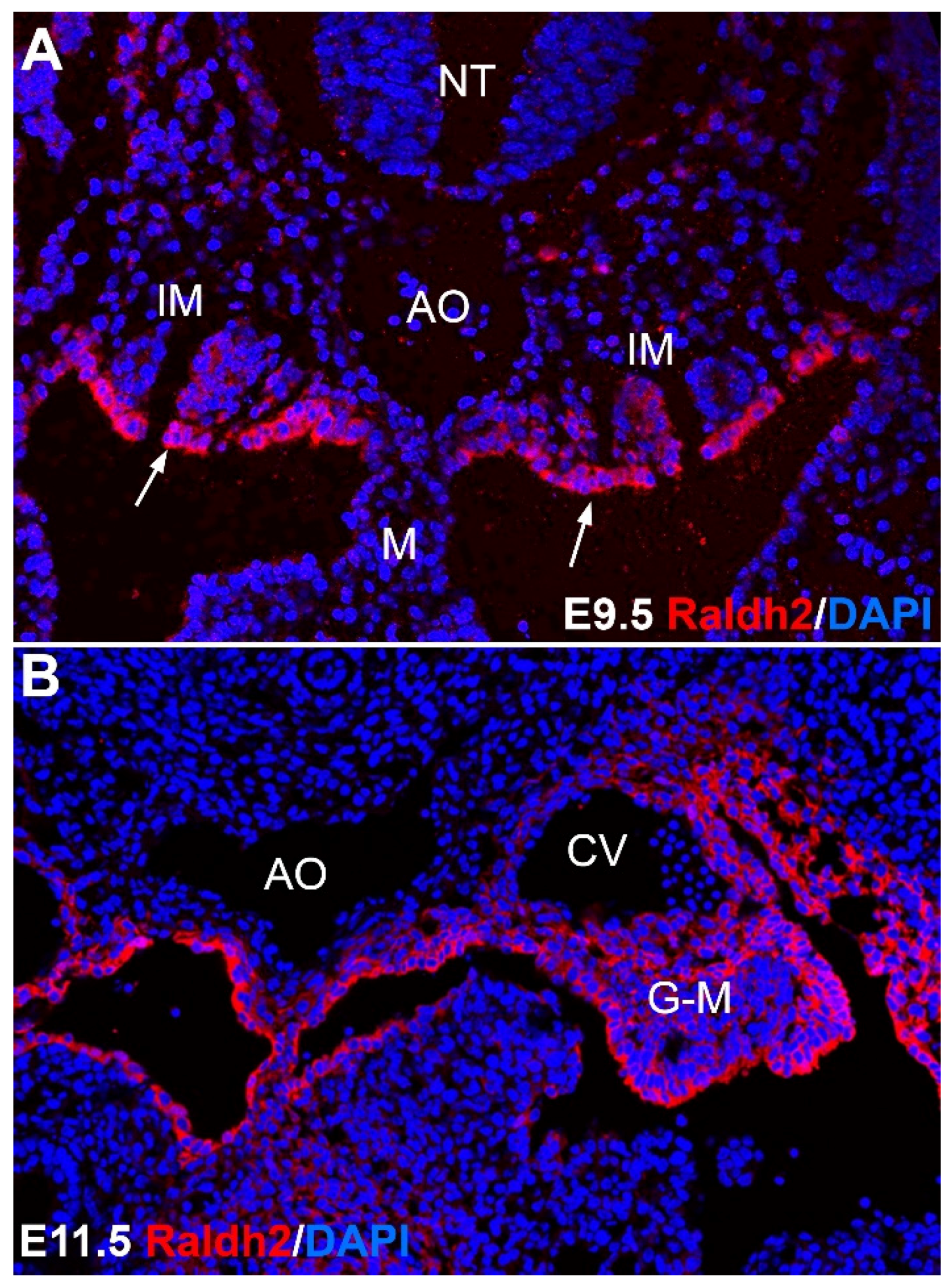
2.2. Aorta-Gonad-Mesonephros (AGM)
2.3. Fetal Liver
2.4. Placenta and Umbilical Cord
2.5. Vitamin A/Retinoic Acid and Developmental Hematopoiesis in Zebrafish
3. Vitamin A/Retinoic Acid in Adult Hematopoiesis
4. RA and Leukemia
5. Epigenetic Modulation of Hematopoiesis by Vitamin A/RA
6. Vitamin A/Vitamin D Interrelationships in Hematopoiesis and Leukemia
7. Vitamin A Deficiency and Anemia
8. Conclusions and Future Directions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Semba, R.D. The discovery of the vitamins. Int. J. Vitam. Nutr. Res. 2012, 823, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Adamo, S.; De Luca, L.M.; Akalovsky, I.; Bhat, P.V. Retinoid-induced adhesion in cultured, transformed mouse fibroblasts. J. Natl. Cancer Inst. 1979, 62, 1473–1478. [Google Scholar] [PubMed]
- Kato, S.; De Luca, L.M. Retinoic acid modulates attachment of mouse fibroblasts to laminin substrates. Exp. Cell Res. 1987, 173, 450–462. [Google Scholar] [CrossRef]
- De Luca, L.M.; Adamo, S.; Kato, S. Retinoids and cell adhesion. Methods Enzymol. 1990, 190, 81–91. [Google Scholar] [PubMed]
- Gerster, H. Vitamin A-functions, dietary requirements and safety in humans. Int. J. Vitam. Nutr. Res. 1997, 67, 71–90. [Google Scholar] [PubMed]
- Tanumihardjo, S.A. Vitamin A: Biomarkers of nutrition for development. Am. J. Clin. Nutr. 2011, 94, 658S–665S. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez-Mazariegos, J.; Theodosiou, M.; Campo-Paysaa, F.; Schubert, M. Vitamin A: A multifunctional tool for development. Semin. Cell Dev. Biol. 2011, 22, 603–610. [Google Scholar] [CrossRef] [PubMed]
- McLaren, D.S.; Kraemer, K. Manual on vitamin A deficiency disorders (VADD). World Rev. Nutr. Diet. 2012, 103, 1–12. [Google Scholar] [PubMed]
- Sanchez, A.M.; Shortrede, J.E.; Vargas-Roig, L.M.; Flamini, M.I. Retinoic acid induces nuclear FAK translocation and reduces breast cancer cell adhesion through Moesin, FAK, and Paxillin. Mol. Cell Endocrinol. 2016, 15, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wen, Q.; Chen, X.L.; Yang, S.J.; Gao, L.; Gao, L.; Zhang, C.; Li, J.L.; Xiang, X.X.; Wan, K.; et al. All-trans retinoic acid arrests cell cycle in leukemic bone marrow stromal cells by increasing intercellular communication through connexin 43-mediated gap junction. J. Hematol. Oncol. 2015, 7, 110. [Google Scholar] [CrossRef] [PubMed]
- Labrecque, J.; Dumas, F.; Lacroix, A.; Bhat, P.V. A novel isoenzyme of aldehyde dehydrogenase specifically involved in the biosynthesis of 9-cis and all-trans retinoic acid. Biochem. J. 1995, 15, 681–684. [Google Scholar] [CrossRef]
- Zhao, D.; McCaffery, P.; Ivins, K.J.; Neve, R.L.; Hogan, P.; Chin, W.W.; Dräger, U.C. Molecular identification of a major retinoic-acid-synthesizing enzyme, a retinaldehyde-specific dehydrogenase. Eur. J. Biochem. 1996, 15, 15–22. [Google Scholar] [CrossRef]
- Mic, F.A.; Molotkov, A.; Fan, X.; Cuenca, A.E.; Duester, G. RALDH3, a retinaldehyde dehydrogenase that generates retinoic acid, is expressed in the ventral retina, otic vesicle and olfactory pit during mouse development. Mech. Dev. 2000, 97, 227–230. [Google Scholar] [CrossRef]
- Duester, G. Families of retinoid dehydrogenases regulating vitamin A function: Production of visual pigment and retinoic acid. Eur. J. Biochem. 2000, 267, 4315–4324. [Google Scholar] [CrossRef] [PubMed]
- Sima, A.; Parisotto, M.; Mader, S.; Bhat, P.V. Kinetic characterization of recombinant mouse retinal dehydrogenase types 3 and 4 for retinal substrates. Biochim. Biophys. Acta 2009, 1790, 1660–1664. [Google Scholar] [CrossRef] [PubMed]
- McKenna, N.J. EMBO Retinoids 2011: Mechanisms, biology and pathology of signaling by retinoic acid and retinoic acid receptors. Nucl. Recept. Signal 2012, 10. [Google Scholar] [CrossRef]
- Clagett-Dame, M.; Knutson, D. Vitamin A in reproduction and development. Nutrients 2011, 3, 385–428. [Google Scholar] [CrossRef] [PubMed]
- De Braekeleer, E.; Douet-Guilbert, N.; De Braekeleer, M. RARA fusion genes in acute promyelocytic leukemia: A review. Expert Rev. Hematol. 2014, 7, 347–357. [Google Scholar] [CrossRef] [PubMed]
- Ablain, J.; De Thé, H. Retinoic acid signaling in cancer: The parable of acute promyelocytic leukemia. Int. J. Cancer 2014, 15, 2262–2272. [Google Scholar] [CrossRef] [PubMed]
- Cano, E.; Ariza, L.; Muñoz-Chápuli, R.; Carmona, R. Signaling by Retinoic Acid in Embryonic and Adult Hematopoiesis. J. Dev. Biol. 2014, 2, 18–33. [Google Scholar] [CrossRef]
- North, T.; Gu, T.L.; Stacy, T.; Wang, Q.; Howard, L.; Binder, M.; Marín-Padilla, M.; Speck, N.A. Cbfa2 is required for the formation of intra-aortic hematopoietic clusters. Development 1999, 126, 2563–2575. [Google Scholar] [PubMed]
- Golub, R.; Cumano, A. Embryonic hematopoiesis. Blood Cells Mol. Dis. 2013, 51, 226–231. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.; Buisman, S.; De Haan, G. Hematopoiesis during development, aging, and disease. Exp. Hematol. 2016, 44, 689–695. [Google Scholar] [CrossRef] [PubMed]
- Bollerot, K.; Pouget, C.; Jaffredo, T. The embryonic origins of hematopoietic stem cells: A tale of hemangioblast and hemogenic endothelium. APMIS 2005, 113, 790–803. [Google Scholar] [CrossRef] [PubMed]
- Dieterlen-Lièvre, F.; Pouget, C.; Bollérot, K.; Jaffredo, T. Are intra-aortic hemopoietic cells derived from endothelial cells during ontogeny? Trends Cardiovasc. Med. 2006, 16, 128–139. [Google Scholar] [CrossRef] [PubMed]
- Dzierzak, E.; Speck, N.A. Of lineage and legacy-the development of mammalian hemopoetic stem cells. Nat. Immunol. 2008, 9, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Hirschi, K.K. Hemogenic endothelium during development and beyond. Blood 2012, 24, 4823–4827. [Google Scholar] [CrossRef] [PubMed]
- Gritz, E.; Hirschi, K.K. Specification and function of hemogenic endothelium during embryogenesis. Cell Mol. Life Sci. 2016, 73, 1547–1567. [Google Scholar] [CrossRef] [PubMed]
- Goldie, L.C.; Lucitti, J.L.; Dickinson, M.E.; Hirschi, K.K. Cell signaling directing the formation and function of hemogenic endothelium during murine embryogenesis. Blood 2008, 15, 3194–3204. [Google Scholar] [CrossRef] [PubMed]
- Niederreither, K.; Subbarayan, V.; Dollé, P.; Chambon, P. Embryonic retinoic acid synthesis is essential for early mouse post-implantation development. Nat. Genet. 1999, 21, 444–448. [Google Scholar] [CrossRef] [PubMed]
- Lai, L.; Bohnsack, B.L.; Niederreither, K.; Hirschi, K.K. Retinoic acid regulates endothelial cell proliferation during vasculogenesis. Development 2003, 130, 6465–6474. [Google Scholar] [CrossRef] [PubMed]
- Bohnsack, B.L.; Lai, L.; Dolle, P.; Hirschi, K.K. Signaling hierarchy downstream of retinoic acid that independently regulates vascular remodeling and endothelial cell proliferation. Genes Dev. 2004, 1, 1345–1358. [Google Scholar] [CrossRef] [PubMed]
- Chanda, B.; Ditadi, A.; Iscove, N.N.; Keller, G. Retinoic acid signaling is essential for embryonic hematopoietic stem cell development. Cell 2013, 155, 215–227. [Google Scholar] [CrossRef] [PubMed]
- Li, E.; Sucov, H.M.; Lee, K.F.; Evans, R.M.; Jaenisch, R. Normal development and growth of mice carrying a targeted disruption of the alpha 1 retinoic acid receptor gene. Proc. Natl. Acad. Sci. USA 1993, 15, 1590–1594. [Google Scholar] [CrossRef]
- Lufkin, T.; Lohnes, D.; Mark, M.; Dierich, A.; Gorry, P.; Gaub, M.P.; LeMeur, M.; Chambon, P. High postnatal lethality and testis degeneration in retinoic acid receptor alpha mutant mice. Proc. Natl. Acad. Sci. USA 1993, 90, 7225–7229. [Google Scholar] [CrossRef] [PubMed]
- Marcelo, K.L.; Sills, T.M.; Coskun, S.; Vasavada, H.; Sanglikar, S.; Goldie, L.C.; Hirschi, K.K. Hemogenic endothelial cell specification requires c-Kit, Notch signaling, and p27-mediated cell-cycle control. Dev. Cell 2013, 9, 504–515. [Google Scholar] [CrossRef] [PubMed]
- Dou, D.R.; Calvanese, V.; Sierra, M.I.; Nguyen, A.T.; Minasian, A.; Saarikoski, P.; Sasidharan, R.; Ramirez, C.M.; Zack, J.A.; Crooks, G.M.; et al. Medial HOXA genes demarcate haematopoietic stem cell fate during human development. Nat. Cell Biol. 2016, 18, 595–606. [Google Scholar] [CrossRef] [PubMed]
- Lebert-Ghali, C.É.; Fournier, M.; Kettyle, L.; Thompson, A.; Sauvageau, G.; Bijl, J.J. Hoxa cluster genes determine the proliferative activity of adult mouse hematopoietic stem and progenitor cells. Blood 2016, 7, 87–90. [Google Scholar] [CrossRef] [PubMed]
- Makita, T.; Duncan, S.A.; Sucov, H.M. Retinoic acid, hypoxia, and GATA factors cooperatively control the onset of fetal liver erythropoietin expression and erythropoietic differentiation. Dev. Biol. 2005, 1, 59–72. [Google Scholar] [CrossRef] [PubMed]
- Ottersbach, K.; Dzierzak, E. The murine placenta contains hematopoietic stem cells within the vascular labyrinth region. Dev. Cell 2005, 8, 377–387. [Google Scholar] [CrossRef] [PubMed]
- Mikkola, H.K.; Gekas, C.; Orkin, S.H.; Dieterlen-Lievre, F. Placenta as a site for hematopoietic stem cell development. Exp. Hematol. 2005, 33, 1048–1054. [Google Scholar] [CrossRef] [PubMed]
- Zovein, A.C.; Hofmann, J.J.; Lynch, M.; French, W.J.; Turlo, K.A.; Yang, Y.; Becker, M.S.; Zanetta, L.; Dejana, E.; Gasson, J.C.; et al. Fate tracing reveals the endothelial origin of hematopoietic stem cells. Cell Stem Cell 2008, 4, 625–636. [Google Scholar] [CrossRef] [PubMed]
- Ottersbach, K.; Dzierzak, E. Analysis of the mouse placenta as a hematopoietic stem cell niche. Methods Mol. Biol. 2009, 538, 335–346. [Google Scholar] [PubMed]
- Robin, C.; Bollerot, K.; Mendes, S.; Haak, E.; Crisan, M.; Cerisoli, F.; Lauw, I.; Kaimakis, P.; Jorna, R.; Vermeulen, M.; et al. Human placenta is a potent hematopoietic niche containing hematopoietic stem and progenitor cells throughout development. Cell Stem Cell 2009, 5, 385–395. [Google Scholar] [CrossRef] [PubMed]
- Ottersbach, K.; Dzierzak, E. The placenta as a haematopoietic organ. Int. J. Dev. Biol. 2012, 54, 1099–1106. [Google Scholar] [CrossRef] [PubMed]
- Niu, W.; Huang, H.; Zhao, L.; Li, Z.; He, W.; Liu, B.; Li, L.; Xiong, J. Characterization of hemangioblast in umbilical arteries of mid-gestation mouse embryos. Int. J. Hematol. 2012, 95, 632–639. [Google Scholar] [CrossRef] [PubMed]
- Sapin, V.; Dollé, P.; Hindelang, C.; Kastner, P.; Chambon, P. Defects of the chorioallantoic placenta in mouse RXRα null fetuses. Dev. Biol. 1997, 19, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Wendling, O.; Chambon, P.; Mark, M. Retinoid X receptors are essential for early mouse development and placentogenesis. Proc. Natl. Acad. Sci. USA 1999, 19, 547–551. [Google Scholar] [CrossRef]
- Pereira, C.F.; Chang, B.; Gomes, A.; Bernitz, J.; Papatsenko, D.; Niu, X.; Swiers, G.; Azzoni, E.; De Bruijn, M.F.; Schaniel, C.; et al. Hematopoietic reprogramming in vitro informs in vivo identification of hemogenic precursors to definitive hematopoietic stem cells. Dev. Cell 2016, 36, 525–539. [Google Scholar] [CrossRef] [PubMed]
- Pillay, L.M.; Mackowetzky, K.J.; Widen, S.A.; Waskiewicz, A.J. Somite-Derived Retinoic Acid Regulates Zebrafish Hematopoietic Stem Cell Formation. PLoS ONE 2016, 18, e0166040. [Google Scholar] [CrossRef] [PubMed]
- De Jong, J.L.; Davidson, A.J.; Wang, Y.; Palis, J.; Opara, P.; Pugach, E.; Daley, G.Q.; Zon, L.I. Interaction of retinoic acid and scl controls primitive blood development. Blood 2010, 116, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Samarut, E.; Fraher, D.; Laudet, V.; Gibert, Y. ZebRA: An overview of retinoic acid signaling during zebrafish development. Biochim. Biophys. Acta 1849, 1849, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Liang, D.; Jia, W.; Li, J.; Li, K.; Zhao, Q. Retinoic acid signaling plays a restrictive role in zebrafish primitive myelopoiesis. PLoS ONE 2012, 7, e30865. [Google Scholar] [CrossRef] [PubMed]
- Purton, L.E.; Dworkin, S.; Olsen, G.H.; Walkley, C.R.; Fabb, S.A.; Collins, S.J.; Chambon, P. RAR gamma is critical for maintaining a balance between hematopoietic stem cell self-renewal and differentiation. J. Exp. Med. 2006, 15, 1283–1293. [Google Scholar] [CrossRef] [PubMed]
- Kastner, P.; Chan, S. Function of RAR alpha during the maturation of neutrophils. Oncogene 2001, 29, 7178–7185. [Google Scholar] [CrossRef] [PubMed]
- Dewamitta, S.R.; Joseph, C.; Purton, L.E.; Walkley, C.R. Erythroid-extrinsic regulation of normal erythropoiesis by retinoic acid receptors. Br. J. Haematol. 2014, 164, 280–285. [Google Scholar] [CrossRef] [PubMed]
- Walkley, C.R.; Olsen, G.H.; Dworkin, S.; Fabb, S.A.; Swann, J.; McArthur, G.A.; Westmoreland, S.V.; Chambon, P.; Scadden, D.T.; Purton, L.E. A microenvironment-induced myeloproliferative syndrome caused by retinoic acid receptor gamma deficiency. Cell 2007, 15, 1097–1110. [Google Scholar] [CrossRef] [PubMed]
- Varnum-Finney, B.; Xu, L.; Brashem-Stein, C.; Nourigat, C.; Flowers, D.; Bakkour, S.; Pear, W.S.; Bernstein, I.D. Pluripotent, cytokine-dependent, hematopoietic stem cells are immortalized by constitutive Notch1 signaling. Nat. Med. 2000, 6, 1278–1281. [Google Scholar] [CrossRef] [PubMed]
- Antonchuk, J.; Sauvageau, G.; Humphries, R.K. HOXB4 overexpression mediates very rapid stem cell regeneration and competitive hematopoietic repopulation. Exp. Hematol. 2001, 29, 1125–1134. [Google Scholar] [CrossRef]
- Dzhagalov, I.; Chambon, P.; He, Y.W. Regulation of CD8+ T lymphocyte effector function and macrophage inflammatory cytokine production by retinoic acid receptor gamma. J. Immunol. 2007, 15, 2113–2121. [Google Scholar] [CrossRef]
- Joseph, C.; Nota, C.; Fletcher, J.L.; Maluenda, A.C.; Green, A.C.; Purton, L.E. Retinoic Acid Receptor γ Regulates B and T Lymphopoiesis via Nestin-Expressing Cells in the Bone Marrow and Thymic Microenvironments. J. Immunol. 2016, 1, 2132–2144. [Google Scholar] [CrossRef] [PubMed]
- Ghiaur, G.; Yegnasubramanian, S.; Perkins, B.; Gucwa, J.L.; Gerber, J.M.; Jones, R.J. Regulation of human hematopoietic stem cell self-renewal by the microenvironment’s control of retinoic acid signaling. Proc. Natl. Acad. Sci. USA 2013, 1, 16121–16126. [Google Scholar] [CrossRef] [PubMed]
- Chute, J.P.; Muramoto, G.G.; Whitesides, J.; Colvin, M.; Safi, R.; Chao, N.J.; McDonnell, D.P. Inhibition of aldehyde dehydrogenase and retinoid signaling induces the expansion of human hematopoietic stem cells. Proc. Natl. Acad. Sci. USA 2006, 1, 11707–11712. [Google Scholar] [CrossRef] [PubMed]
- Brown, G.; Marchwicka, A.; Cunningham, A.; Toellner, K.M.; Marcinkowska, E. Antagonizing Retinoic Acid Receptors Increases Myeloid Cell Production by Cultured Human Hematopoietic Stem Cells. Arch. Immunol. Ther. Exp. (Warsz.) 2016, 13. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.A.; Cannons, J.L.; Grainger, J.R.; Dos Santos, L.M.; Hand, T.W.; Naik, S.; Wohlfert, E.A.; Chou, D.B.; Oldenhove, G.; Robinson, M.; et al. Essential role for retinoic acid in the promotion of CD4(+) T cell effector responses via retinoic acid receptor alpha. Immunity 2011, 25, 435–447. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.C. Vitamin A and retinoic acid in T cell-related immunity. Am. J. Clin. Nutr. 2012, 96, 1166S–1172S. [Google Scholar] [CrossRef] [PubMed]
- Raverdeau, M.; Mills, K.H. Modulation of T cell and innate immune responses by retinoic acid. J. Immunol. 2014, 1, 2953–2958. [Google Scholar] [CrossRef] [PubMed]
- Cassani, B.; Villablanca, E.J.; De Calisto, J.; Wang, S.; Mora, J.R. Vitamin A and immune regulation: Role of retinoic acid in gut-associated dendritic cell education, immune protection and tolerance. Mol. Asp. Med. 2012, 33, 63–76. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.H.; Taparowsky, E.J.; Kim, C.H. Retinoic Acid Differentially Regulates the Migration of Innate Lymphoid Cell Subsets to the Gut. Immunity 2015, 21, 107–119. [Google Scholar] [CrossRef] [PubMed]
- Bakdash, G.; Vogelpoel, L.T.; Van Capel, T.M.; Kapsenberg, M.L.; De Jong, E.C. Retinoic acid primes human dendritic cells to induce gut-homing, IL-10-producing regulatory T cells. Mucosal Immunol. 2015, 8, 265–278. [Google Scholar] [CrossRef] [PubMed]
- Bono, M.R.; Tejon, G.; Flores-Santibañez, F.; Fernandez, D.; Rosemblatt, M.; Sauma, D. Retinoic Acid as a Modulator of T Cell Immunity. Nutrients 2016, 8, 349. [Google Scholar] [CrossRef] [PubMed]
- Ertesvåg, A.; Naderi, S.; Blomhoff, H.K. Regulation of B cell proliferation and differentiation by retinoic acid. Semin. Immunol. 2009, 21, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.C.; Chen, Q.; Ma, Y. Vitamin A and retinoic acid in the regulation of B-cell development and antibody production. Vitam. Horm. 2011, 86, 103–126. [Google Scholar] [PubMed]
- Holm, K.L.; Indrevaer, R.L.; Myklebust, J.H.; Kolstad, A.; Moskaug, J.Ø.; Naderi, E.H.; Blomhoff, H.K. Myeloid cell leukemia 1 has a vital role in retinoic acid-mediated protection of Toll-like receptor 9-stimulated B cells from spontaneous and DNA damage-induced apoptosis. Immunology 2016, 149, 62–73. [Google Scholar] [CrossRef] [PubMed]
- Testa, U.; Lo-Coco, F. Targeting of leukemia-initiating cells in acute promyelocytic leukemia. Stem Cell Investig. 2015, 29, 2–8. [Google Scholar]
- Cicconi, L.; Divona, M.; Ciardi, C.; Ottone, T.; Ferrantini, A.; Lavorgna, S.; Alfonso, V.; Paoloni, F.; Piciocchi, A.; Avvisati, G.; et al. PML-RARα kinetics and impact of FLT3-ITD mutations in newly diagnosed acute promyelocytic leukemia treated with ATRA and ATO or ATRA and chemotherapy. Leukemia 2016, 30, 1987–1992. [Google Scholar] [CrossRef] [PubMed]
- Cicconi, L.; Lo-Coco, F. Current management of newly diagnosed acute promyelocytic leukemia. Ann. Oncol. 2016, 27, 1474–1481. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Liu, L.; Jin, J.; Lou, Y. All-Trans Retinoic Acid Plus Arsenic Trioxide versus All-Trans Retinoic Acid plus Chemotherapy for Newly Diagnosed Acute Promyelocytic Leukemia: A Meta-Analysis. PLoS ONE 2016, 8. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, M.R.; Tallman, M.S.; Abboud, C.N.; Altman, J.K.; Appelbaum, F.R.; Arber, D.A.; Attar, E.; Borate, U.; Coutre, S.E.; Damon, L.E.; et al. National Comprehensive Cancer Network. Acute myeloid leukemia. J. Natl. Compr. Cancer Netw. 2013, 1, 1047–1055. [Google Scholar]
- Lo-Coco, F.; Orlando, S.M.; Platzbecker, U. Treatment of acute promyelocytic leukemia. N. Engl. J. Med. 2013, 10, 1472. [Google Scholar]
- Zhu, H.H.; Wu, D.P.; Jin, J.; Li, J.Y.; Ma, J.; Wang, J.X.; Chen, S.J.; Huang, X.J. Long-term survival of acute promyelocytic leukemia patients treated with arsenic and retinoic acid. Br. J. Haematol. 2016, 174, 820–822. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, H.; Watanabe, T.; Kinoshita, A.; Yuza, Y.; Moritake, H.; Terui, K.; Iwamoto, S.; Nakayama, H.; Shimada, A.; Kudo, K.; et al. High event-free survival rate with minimum-dose-anthracycline treatment in childhood acute promyelocytic leukemia: A nationwide prospective study by the Japanese Paediatric Leukemia/Lymphoma Study Group. Br. J. Haematol. 2016, 174, 437–443. [Google Scholar] [CrossRef] [PubMed]
- Platzbecker, U.; Avvisati, G.; Cicconi, L.; Thiede, C.; Paoloni, F.; Vignetti, M.; Ferrara, F.; Divona, M.; Albano, F.; Efficace, F.; et al. Improved outcomes with Retinoic Acid and Arsenic Trioxide compared with Retinoic Acid and chemotherapy in Non-High-Risk Acute Promyelocytic Leukemia: Final results of the Randomized Italian-German APL0406 Trial. J. Clin. Oncol. 2017, 35, 605–612. [Google Scholar] [CrossRef] [PubMed]
- Norsworthy, K.J.; Altman, J.K. Optimal treatment strategies for high-risk acute promyelocytic leukemia. Curr. Opin. Hematol. 2016, 23, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Podoltsev, N.A.; Stahl, M.; Zeidan, A.M.; Gore, S.D. Selecting initial treatment of acute myeloid leukemia in older adults. Blood Rev. 2016, 8. [Google Scholar] [CrossRef]
- Wang, X.; Lin, Q.; Lv, F.; Liu, N.; Xu, Y.; Liu, M.; Chen, Y.; Yi, Z. LG-362B targets PML-RARα and blocks ATRA resistance of acute promyelocytic leukemia. Leukemia 2016, 30, 1465–1474. [Google Scholar] [CrossRef] [PubMed]
- Wallace, A.S.; Supnick, H.T.; Bunaciu, R.P.; Yen, A. RRD-251 enhances all-trans retinoic acid (RA)-induced differentiation of HL-60 myeloblastic leukemia cells. Oncotarget 2016, 17. [Google Scholar] [CrossRef] [PubMed]
- Atashrazm, F.; Lowenthal, R.M.; Dickinson, J.L.; Holloway, A.F.; Woods, G.M. Fucoidan enhances the therapeutic potential of arsenic trioxide and all-trans retinoic acid in acute promyelocytic leukemia, in vitro and in vivo. Oncotarget 2016, 14. [Google Scholar] [CrossRef] [PubMed]
- Shao, X.; Liu, Y.; Li, Y.; Xian, M.; Zhou, Q.; Yang, B.; Ying, M.; He, Q. The HER2 inhibitor TAK165 Sensitizes Human Acute Myeloid Leukemia Cells to Retinoic Acid-Induced Myeloid Differentiation by activating MEK/ERK mediated RARα/STAT1 axis. Sci. Rep. 2016, 14. [Google Scholar] [CrossRef] [PubMed]
- Montesinos, P.; Sanz, M.A. The differentiation syndrome in patients with acute promyelocytic leukemia: Experience of the pethema group and review of the literature. Mediterr. J. Hematol. Infect. Dis. 2011, 3, e2011059. [Google Scholar] [CrossRef] [PubMed]
- Sanz, M.A.; Montesinos, P. How we prevent and treat differentiation syndrome in patients with acute promyelocytic leukemia. Blood 2014, 1, 2777–2782. [Google Scholar] [CrossRef] [PubMed]
- Schwager, J.; Bompard, A.; Weber, P.; Raederstorff, D. Ascorbic acid modulates cell migration in differentiated HL-60 cells and peripheral blood leukocytes. Mol. Nutr. Food Res. 2015, 59, 1513–1523. [Google Scholar] [CrossRef] [PubMed]
- Sumi, D.; Suzukawa, K.; Himeno, S. Arsenic trioxide augments all-trans retinoic acid-induced differentiation of HL-60 cells. Life Sci. 2016, 15, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Weng, X.Q.; Sheng, Y.; Ge, D.Z.; Wu, J.; Shi, L.; Cai, X. RAF-1/MEK/ERK pathway regulates ATRA-induced differentiation in acute promyelocytic leukemia cells through C/EBPβ, C/EBPε and PU.1. Leuk. Res. 2016, 45, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; Jiang, Q.; Liu, W.; Bai, Y. All-trans retinoic acid inhibits HOXA7 expression in leukemia cell NB4. Cell Mol. Biol. 2016, 22, 63–66. [Google Scholar]
- Liu, W.J.; Zhang, T.; Guo, Q.L.; Liu, C.Y.; Bai, Y.Q. Effect of ATRA on the expression of HOXA5 gene in K562 cells and its relationship with cell cycle and apoptosis. Mol. Med. Rep. 2016, 13, 4221–4228. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Jia, X.; Wang, J.; Li, Y.; Xie, S. Knockdown of homeobox A5 by small hairpin RNA inhibits proliferation and enhances cytarabine chemosensitivity of acute myeloid leukemia cells. Mol. Med. Rep. 2015, 12, 6861–6866. [Google Scholar] [CrossRef] [PubMed]
- Mihara, K.; Yoshida, T.; Ishida, S.; Takei, Y.; Kitanaka, A.; Shimoda, K.; Morishita, K.; Takihara, Y.; Ichinohea, T. All-trans retinoic acid and interferon-α increase CD38 expression on adult T-cell leukemia cells and sensitize them to T cells bearing anti-CD38 chimeric antigen receptors. Blood Cancer J. 2016, 13, 421. [Google Scholar] [CrossRef] [PubMed]
- Sashida, G.; Iwama, A. Epigenetic regulation of hematopoiesis. Int. J. Hematol. 2012, 96, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Di Croce, L. Chromatin modifying activity of leukemia associated fusion proteins. Hum. Mol. Genet. 2005, 15, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Rice, K.L.; Hormaeche, I.; Licht, J.D. Epigenetic regulation of normal and malignant hematopoiesis. Oncogene 2007, 15, 6697–6714. [Google Scholar] [CrossRef] [PubMed]
- Fazi, F.; Zardo, G.; Gelmetti, V.; Travaglini, L.; Ciolfi, A.; Di Croce, L.; Rosa, A.; Bozzoni, I.; Grignani, F.; Lo-Coco, F.; et al. Heterochromatic gene repression of the retinoic acid pathway in acute myeloid leukemia. Blood 2007, 15, 4432–4440. [Google Scholar] [CrossRef] [PubMed]
- Przespolewski, A.; Wang, E.S. Inhibitors of LSD1 as a potential therapy for acute myeloid leukemia. Expert Opin. Investig. Drugs 2016, 25, 771–780. [Google Scholar] [CrossRef] [PubMed]
- Guidez, F.; Parks, S.; Wong, H.; Jovanovic, J.V.; Mays, A.; Gilkes, A.F.; Mills, K.I.; Guillemin, M.C.; Hobbs, R.M.; Pandolfi, P.P.; et al. RARα-PLZF overcomes PLZF-mediated repression of CRABPI, contributing to retinoid resistance in t(11;17) acute promyelocytic leukemia. Proc. Natl. Acad. Sci. USA 2007, 20, 18694–18699. [Google Scholar] [CrossRef] [PubMed]
- Choi, W.I.; Kim, M.Y.; Jeon, B.N.; Koh, D.I.; Yun, C.O.; Li, Y.; Lee, C.E.; Oh, J.; Kim, K.; Hur, M.W. Role of promyelocytic leukemia zinc finger (PLZF) in cell proliferation and cyclin-dependent kinase inhibitor 1A (p21WAF/CDKN1A) gene repression. J. Biol. Chem. 2014, 4, 18625–18640. [Google Scholar] [CrossRef] [PubMed]
- Choi, W.I.; Yoon, J.H.; Kim, M.Y.; Koh, D.I.; Licht, J.D.; Kim, K.; Hur, M.W. Promyelocytic leukemia zinc finger-retinoic acid receptor α (PLZF-RARα), an oncogenic transcriptional repressor of cyclin-dependent kinase inhibitor 1A (p21WAF/CDKN1A) and tumor protein p53 (TP53) genes. J. Biol. Chem. 2014, 4, 18641–18656. [Google Scholar] [CrossRef] [PubMed]
- Jones, L.C.; Tefferi, A.; Idos, G.E.; Kumagai, T.; Hofmann, W.K.; Koeffler, H.P. RARbeta2 is a candidate tumor suppressor gene in myelofibrosis with myeloid metaplasia. Oncogene 2004, 14, 7846–7853. [Google Scholar] [CrossRef] [PubMed]
- Nervi, C.; Fazi, F.; Rosa, A.; Fatica, A.; Bozzoni, I. Emerging role for microRNAs in acute promyelocytic leukemia. Curr. Top. Microbiol. Immunol. 2007, 313, 73–84. [Google Scholar] [PubMed]
- Haussler, M.R.; Haussler, C.A.; Jurutka, P.W.; Thompson, P.D.; Hsieh, J.C.; Remus, L.S.; Selznick, S.H.; Whitfield, G.K. The vitamin D hormone and its nuclear receptor: Molecular actions and disease states. J. Endocrinol. 1997, 154, S57–S73. [Google Scholar] [PubMed]
- Thompson, P.D.; Jurutka, P.W.; Haussler, C.A.; Whitfield, G.K.; Haussler, M.R. Heterodimeric DNA binding by the vitamin D receptor and retinoid X receptors is enhanced by 1,25-dihydroxyvitamin D3 and inhibited by 9-cis-retinoic acid. Evidence for allosteric receptor interactions. J. Biol. Chem. 1998, 273, 8483–8491. [Google Scholar] [CrossRef] [PubMed]
- Luong, Q.T.; Koeffler, H.P. Vitamin D compounds in leukemia. J. Steroid Biochem. Mol. Biol. 2005, 97, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Kelly, J.L.; Friedberg, J.W.; Calvi, L.M.; Van Wijngaarden, E.; Fisher, S.G. A case-control study of ultraviolet radiation exposure, vitamin D, and lymphoma risk in adults. Cancer Causes Control 2010, 21, 1265–1275. [Google Scholar] [CrossRef] [PubMed]
- Jeanson, N.T.; Scadden, D.T. Vitamin D receptor deletion leads to increased hematopoietic stem and progenitor cells residing in the spleen. Blood 2010, 18, 4126–4129. [Google Scholar] [CrossRef] [PubMed]
- Bunce, C.M.; Brown, G.; Hewison, M. Vitamin D and hematopoiesis. Trends Endocrinol. Metab. 1997, 8, 245–251. [Google Scholar] [CrossRef]
- Hall, A.C.; Brown, M.B. The role of vitamin D in hematologic disease and stem cell transplantation. Nutrients 2013, 18, 2206–2221. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Bruce, D.; Froicu, M.; Weaver, V.; Cantorna, M.T. Failure of T cell homing, reduced CD4/CD8αα intraepithelial lymphocytes, and inflammation in the gut of vitamin D receptor KO mice. Proc. Natl. Acad. Sci. USA 2008, 30, 20834–20839. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Cantorna, M.T. Epigenetic reduction in invariant NKT cells following in utero vitamin D deficiency in mice. J. Immunol. 2011, 1, 1384–1390. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Zhao, J.; Cantorna, M.T. Invariant NKT cell defects in vitamin D receptor knockout mice prevents experimental lung inflammation. J. Immunol. 2011, 1, 4907–4912. [Google Scholar] [CrossRef] [PubMed]
- Grande, A.; Montanari, M.; Tagliafico, E.; Manfredini, R.; Zanocco Marani, T.; Siena, M.; Tenedini, E.; Gallinelli, A.; Ferrari, S. Physiological levels of 1alpha, 25 dihydroxyvitamin D3 induce the monocytic commitment of CD34+ hematopoietic progenitors. J. Leuk. Biol. 2002, 71, 641–651. [Google Scholar]
- Kim, M.; Mirandola, L.; Pandey, A.; Nguyen, D.D.; Jenkins, M.R.; Turcel, M.; Cobos, E.; Chiriva-Internati, M. Application of vitamin D and derivatives in hematological malignancies. Cancer Lett. 2012, 1, 8–22. [Google Scholar] [CrossRef] [PubMed]
- Marchwicka, A.; Cebrat, M.; Sampath, P.; Snieżewski, L.; Marcinkowska, E. Perspectives of differentiation therapies of acute myeloid leukemia: The search for the molecular basis of patients’ variable responses to 1,25-dihydroxyvitamin d and vitamin d analogs. Front. Oncol. 2014, 27, 125. [Google Scholar] [CrossRef] [PubMed]
- Gocek, E.; Marchwicka, A.; Baurska, H.; Chrobak, A.; Marcinkowska, E. Opposite regulation of vitamin D receptor by ATRA in AML cells susceptible and resistant to vitamin D-induced differentiation. J. Steroid Biochem. Mol. Biol. 2012, 132, 220–226. [Google Scholar] [CrossRef] [PubMed]
- Marchwicka, A.; Cebrat, M.; Łaszkiewicz, A.; Śnieżewski, Ł.; Brown, G.; Marcinkowska, E. Regulation of vitamin D receptor expression by retinoic acid receptor alpha in acute myeloid leukemia cells. J. Steroid Biochem. Mol. Biol. 2016, 159, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Kuwata, T.; Wang, I.M.; Tamura, T.; Ponnamperuma, R.M.; Levine, R.; Holmes, K.L.; Morse, H.C.; De Luca, L.M.; Ozato, K. Vitamin A deficiency in mice causes a systemic expansion of myeloid cells. Blood 2000, 1, 3349–3356. [Google Scholar]
- Walkley, C.R.; Shea, J.M.; Sims, N.A.; Purton, L.E.; Orkin, S.H. Rb regulates interactions between hematopoietic stem cells and their bone marrow microenvironment. Cell 2007, 15, 1081–1095. [Google Scholar] [CrossRef] [PubMed]
- Da Cunha, M.S.; Siqueira, E.M.; Trindade, L.S.; Arruda, S.F. Vitamin A deficiency modulates iron metabolism via ineffective erythropoiesis. J. Nutr. Biochem. 2014, 25, 1035–1044. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Wang, C.X.; Lan, L.; Zhao, D. Vitamin a deficiency aggravates iron deficiency by upregulating the expression of iron regulatory protein-2. Nutrition 2012, 28, 281–287. [Google Scholar] [CrossRef] [PubMed]
- Citelli, M.; Bittencourt, L.; Da Silva, S.; Pierucci, A.; Pedrosa, C. Vitamin a modulates the expression of genes involved in iron bioavailability. Biol. Trace Element Res. 2012, 149, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Wiseman, E.M.; Bar-El Dadon, S.; Reifen, R. The vicious cycle of vitamin A deficiency: A review. Crit. Rev. Food Sci. Nutr. 2016, 29. [Google Scholar] [CrossRef] [PubMed]
- Michelazzo, F.B.; Oliveira, J.M.; Stefanello, J.; Luzia, L.A.; Rondó, P.H. The influence of vitamin A supplementation on iron status. Nutrients 2013, 7, 4399–4413. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, S.; Ahmed, A.; Randhawa, M.A.; Atukorala, S.; Arlappa, N.; Ismail, T.; Ali, Z. Prevalence of vitamin A deficiency in South Asia: Causes, outcomes, and possible remedies. J. Health Popul. Nutr. 2013, 31, 413–423. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.A.; Grainger, J.R.; Spencer, S.P.; Belkaid, Y. The role of retinoic acid in tolerance and immunity. Immunity 2011, 22, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Sommer, A.; Vyas, K.S. A global clinical view on vitamin A and carotenoids. Am. J. Clin. Nutr. 2012, 96, 1204S–1206S. [Google Scholar] [CrossRef] [PubMed]
- Micronutrient Deficiencies: Vitamin A Deficiency. Available online: http://www.who.int/nutrition/topics/vad/en/ (accessed on 9 January 2017).
- WHO Global Database on Vitamin A Deficiency. Available online: http://www.who.int/vmnis/database/vitamina/en/ (accessed on 9 January 2017).
- Bailey, R.L.; West, K.P., Jr.; Black, R.E. The epidemiology of global micronutrient deficiencies. Ann. Nutr. Metab. 2015, 66, 22–33. [Google Scholar] [CrossRef] [PubMed]
- Mayo-Wilson, E.; Imdad, A.; Herzer, K.; Yakoob, M.Y.; Bhutta, Z.A. Vitamin A supplements for preventing mortality, illness, and blindness in children aged under 5: Systematic review and meta-analysis. BMJ 2011, 25. [Google Scholar] [CrossRef] [PubMed]
- Mason, J.; Greiner, T.; Shrimpton, R.; Sanders, D.; Yukich, J. Vitamin A policies need rethinking. Int. J. Epidemiol. 2015, 44, 283–292. [Google Scholar] [CrossRef] [PubMed]
- West, K.P., Jr.; Gernand, A.; Sommer, A. Vitamin A in Nutritional Anemia. In Nutritional Anemia; Kraemer, K., Zimmermann, M.B., Eds.; Sight and Life Press: Basel, Switzerland, 2007; pp. 133–153. [Google Scholar]
- Zhang, X.; Chen, K.; Qu, P.; Liu, Y.X.; Li, T.Y. Effect of biscuits fortified with different doses of vitamin A on indices of vitamin A status, haemoglobin and physical growth levels of pre-school children in Chongqing. Public Health Nutr. 2010, 13, 1462–1471. [Google Scholar] [CrossRef] [PubMed]
- Palmer, A.C.; Schulze, K.J.; Khatry, S.K.; De Luca, L.M.; West, K.P., Jr. Maternal vitamin A supplementation increases natural antibody concentrations of preadolescent offspring in rural Nepal. Nutrition 2015, 31, 813–819. [Google Scholar] [CrossRef] [PubMed]
- Arguello, M.A.; Schulze, K.J.; Wu, L.S.; Dreyfuss, M.L.; Khatry, S.K.; Christian, P.; West, K.P. Circulating IGF-1 may mediate improvements in haemoglobin associated with vitamin A status during pregnancy in rural Nepalese women. Asia Pac. J. Clin. Nutr. 2015, 24, 128–137. [Google Scholar] [PubMed]
- Gebremedhin, S.; Enquselassie, F.; Umeta, M. Prevalence and correlates of maternal anemia in rural Sidama, Southern Ethiopia. Afr. J. Reprod. Health 2014, 18, 44–53. [Google Scholar] [PubMed]
- Cardoso, M.A.; Augusto, R.A.; Bortolini, G.A.; Oliveira, C.S.; Tietzman, D.C.; Sequeira, L.A.; Hadler, M.C.; Peixoto- Mdo, R.; Muniz, P.T.; Vitolo, M.R.; et al. Effect of Providing Multiple Micronutrients in Powder through Primary Healthcare on Anemia in Young Brazilian Children: A Multicentre Pragmatic Controlled Trial. PLoS ONE 2016, 14, e0151097. [Google Scholar]
- Thompson, B.; Amoroso, L. (Eds.) Combating Micronutrient Deficiencies: Food-based Approaches; CAB International and FAO: Oxfordshire, UK, 2011; pp. 5–26.
- Nair, M.K.; Augustine, L.F.; Konapur, A. Food-Based Interventions to Modify Diet Quality and Diversity to Address Multiple Micronutrient Deficiency. Front. Public Health 2016, 5, 277. [Google Scholar] [CrossRef] [PubMed]
- Awasthi, S.; Peto, R.; Read, S.; Richards, S.M.; Pande, V.; Bundy, D.; DEVTA (Deworming and Enhanced Vitamin A) Team. Population deworming every 6 months with albendazole in 1 million pre-school children in North India: DEVTA, a cluster-randomised trial. Lancet 2013, 27, 1478–1486. [Google Scholar] [CrossRef]
- Sommer, A.; West, K.P., Jr.; Martorell, R. Vitamin A supplementation in Indian children. Lancet 2013, 17, 591. [Google Scholar] [CrossRef]
- Benn, C.S.; Aaby, P.; Arts, R.J.; Jensen, K.J.; Netea, M.G.; Fisker, A.B. An enigma: Why vitamin A supplementation does not always reduce mortality even though vitamin A deficiency is associated with increased mortality. Int. J. Epidemiol. 2015, 44, 906–918. [Google Scholar] [CrossRef] [PubMed]
- Dary, O.; Mora, J.O. International Vitamin A Consultative Group. Food fortification to reduce vitamin A deficiency: International Vitamin A Consultative Group recommendations. J. Nutr. 2002, 132, 2927S–2933S. [Google Scholar] [PubMed]
- Gannon, B.; Kaliwile, C.; Arscott, S.A.; Schmaelzle, S.; Chileshe, J.; Kalungwana, N.; Mosonda, M.; Pixley, K.; Masi, C.; Tanumihardjo, S.A. Biofortified orange maize is as efficacious as a vitamin A supplement in Zambian children even in the presence of high liver reserves of vitamin A: A community-based, randomized placebo-controlled trial. Am. J. Clin. Nutr. 2014, 100, 1541–1550. [Google Scholar] [CrossRef] [PubMed]
- Palmer, A.C.; Healy, K.; Barffour, M.A.; Siamusantu, W.; Chileshe, J.; Schulze, K.J.; West, K.P., Jr.; Labrique, A.B. Provitamin A Carotenoid-Biofortified Maize Consumption Increases Pupillary Responsiveness among Zambian Children in a Randomized Controlled Trial. J. Nutr. 2016, 146, 2551–2558. [Google Scholar] [CrossRef] [PubMed]
- Talsma, E.F.; Brouwer, I.D.; Verhoef, H.; Mbera, G.N.; Mwangi, A.M.; Demir, A.Y.; Maziya-Dixon, B.; Boy, E.; Zimmermann, M.B.; Melse-Boonstra, A. Biofortified yellow cassava and vitamin A status of Kenyan children: A randomized controlled trial. Am. J. Clin. Nutr. 2016, 103, 258–267. [Google Scholar] [CrossRef] [PubMed]
- Paine, J.A.; Shipton, C.A.; Chaggar, S.; Howells, R.M.; Kennedy, M.J.; Vernon, G.; Wright, S.Y.; Hinchliffe, E.; Adams, J.L.; Silverstone, A.L.; et al. Improving the nutritional value of Golden Rice through increased pro-vitamin A content. Nat. Biotechnol. 2005, 23, 482–487. [Google Scholar] [CrossRef] [PubMed]
- Moghissi, A.A.; Pei, S.; Liu, Y. Golden rice: scientific, regulatory and public information processes of a genetically modified organism. Crit. Rev. Biotechnol. 2016, 36, 535–541. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cañete, A.; Cano, E.; Muñoz-Chápuli, R.; Carmona, R. Role of Vitamin A/Retinoic Acid in Regulation of Embryonic and Adult Hematopoiesis. Nutrients 2017, 9, 159. https://doi.org/10.3390/nu9020159
Cañete A, Cano E, Muñoz-Chápuli R, Carmona R. Role of Vitamin A/Retinoic Acid in Regulation of Embryonic and Adult Hematopoiesis. Nutrients. 2017; 9(2):159. https://doi.org/10.3390/nu9020159
Chicago/Turabian StyleCañete, Ana, Elena Cano, Ramón Muñoz-Chápuli, and Rita Carmona. 2017. "Role of Vitamin A/Retinoic Acid in Regulation of Embryonic and Adult Hematopoiesis" Nutrients 9, no. 2: 159. https://doi.org/10.3390/nu9020159
APA StyleCañete, A., Cano, E., Muñoz-Chápuli, R., & Carmona, R. (2017). Role of Vitamin A/Retinoic Acid in Regulation of Embryonic and Adult Hematopoiesis. Nutrients, 9(2), 159. https://doi.org/10.3390/nu9020159





