Effects of β-Hydroxy-β-methylbutyrate-free Acid Supplementation on Strength, Power and Hormonal Adaptations Following Resistance Training
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Experimental Protocol
2.3. Testing Procedures
2.3.1. Anthropometric Measures
2.3.2. Strength and Power Measures
2.3.3. Blood Measures and Analysis
2.4. HMB-FA Supplementations
2.5. Diet Control
2.6. Training Program
2.7. Statistical Analyses
3. Results
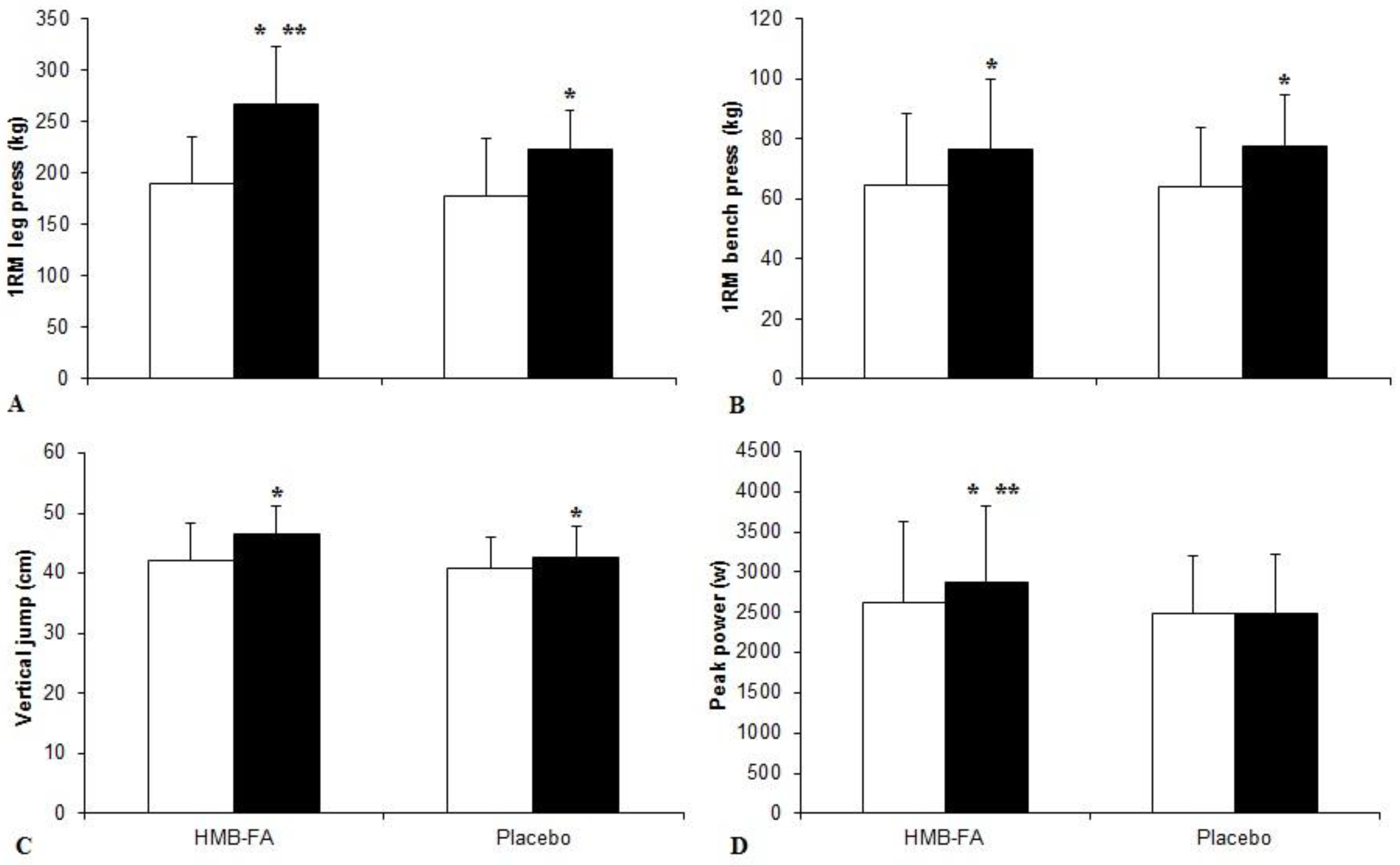
3.1. Strength and Power Measures
3.2. Hormonal Measures
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hakkinen, K.; Pakarinen, A. Acute hormonal responses to two different fatiguing heavy-resistance protocols in male athletes. J. Appl. Physiol. 1993, 74, 882–887. [Google Scholar] [PubMed]
- Hakkinen, K.; Pakarinen, A. Serum hormones and strength development during strength training in middle-aged and elderly males and females. Acta. Physiol. Scand. 1994, 150, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, W.J.; Ratamess, N.A. Endocrine responses and adaptations to strength and power training. In Strength and Power in Sport, 2nd ed.; Komi, P.V., Ed.; Malden (MA): Blackwell Science Ltd.: Oxford, UK, 2003; pp. 331–342. [Google Scholar]
- Arazi, H.; Damirchi, A.; Asadi, A. Age-related muscle circumference, strength development and hormonal adaptations with 8 weeks moderate intensity resistance training. Ann. Endocrinol. 2013, 74, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, R.R. Effects of amino acid intake on anabolic processes. Can. J. Appl. Physiol. 2001, 26, 220–227. [Google Scholar] [CrossRef]
- Blomstrand, E.; Eliasson, J.; Karlsson, H.K. Branched-chain amino acids activate key enzymes in protein synthesis after physical exercise. J. Nutr. 2006, 136, 269–273. [Google Scholar]
- Arazi, H.; Rohani, H.; Ghiasi, A. Resistance training & beta-hydroxy-beta-methylbutirate supplementation on hormones. Braz. J. Sports Med. 2015, 21, 386–389. [Google Scholar]
- Wilson, J.M.; Lowery, R.P.; Joy, J.M.; Andersen, J.C.; Wilson, S.M.C.; Stout, J.R.; Duncan, N.; Fuller, J.C.; Baier, S.M.; Naimo, M.A.; et al. The effects of 12 weeks of beta-hydroxy-beta-methylbutyrate free acid supplementation on muscle mass, strength and power in resistance-trained indeviduals: A randomized, double-blind, placebo-controlled study. Eur. J. Appl. Physiol. 2014, 114, 1217–1227. [Google Scholar] [CrossRef] [PubMed]
- Nissen, S.L.; Sharp, R.L. Effect of dietary supplements on lean mass and strength gains with resistance exercise: A meta-analysis. J. Appl. Physiol. 2003, 94, 651–659. [Google Scholar] [CrossRef] [PubMed]
- Townsend, J.R.; Hoffman, J.R.; Gonzalez, A.M.; Jajtner, A.R.; Boone, C.H.; Robinson, E.H.; Mangine, G.T.; Wells, A.J.; Fragala, M.S.; Fukuda, D.H.; et al. Effects of β-Hydroxy-β-methylbutyrate (HMB)-free acid ingestion and resistance exercise on the acute endocrine response. Int. J. End. 2015. [Google Scholar] [CrossRef]
- Hoffman, J.R.; Cooper, J.; Wendell, M.; Im, J.; Kang, J. Effects of β-Hydroxy-β-methylbutyrate on power performance and indices of muscle damage and stress during high-intensity training. J. Strength Cond. Res. 2004, 18, 747–752. [Google Scholar] [PubMed]
- Slater, G.J.; Jenkins, D. Beta-hydroxy-beta-methylbutyrate (HMB) supplementation and the promotion of muscle growth and strength. Sports Med. 2000, 30, 105–116. [Google Scholar] [CrossRef] [PubMed]
- Eley, H.L.; Russel, S.T.; Baxter, J.H.; Mukerji, P.; Tisdale, M.J. Signaling pathways initiated by β-Hydroxy-β-methylbutyrate to attenuate the depression of protein synthesis in skeletal muscle in response to cachectic stimuli. Am. J. Physiol. Endocrinol. Metab. 2007, 293, 923–931. [Google Scholar] [CrossRef] [PubMed]
- Fuller, J.C., Jr.; Sharp, R.L.; Angus, H.F.; Baier, S.M.; Rathmacher, J.A. Free acid gel form of β-Hydroxy-β-methylbutyrate (HMB) improves HMB clearance from plasma in human subjects compared with the calcium HMB salt. Br. J. Nutr. 2011, 105, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, A.M.; Fragala, M.S.; Jajtner, A.R.; Townsend, J.R.; Wells, A.J.; Beyer, K.S.; Boone, C.H.; Pruna, G.J.; Mangine, G.T.; Bohner, J.D.; et al. Effects of β-Hydroxy-β-methylbutyrate (HMB)-free acid and cold water immersion on expression of CR3 and MIP-1β following resistance exercise. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2014, 306, 483–489. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.M.; Lowery, R.P.; Joy, J.M.; Walters, J.A.; Baier, S.M.; Fuller, J.C.; Stout, J.R.; Norton, L.E.; Sikorski, E.M.; Wilson, S.M.C.; et al. β-Hydroxy-β-methylbutyrate free acid reduces markers of exercise induced muscle damage and improve recovery in resistance trained men. Br. J. Nutr. 2012, 110, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.M.; Kim, J.; Lee, S.; Rathmacher, J.A.; Dalmau, B.; Kingsley, J.D.; Koch, H.; Manninen, A.H.; Saadat, R.; Panton, L.B. Acute and timing effects of β-Hydroxy-β-methylbutyrate (HMB) on indirect markers of skeletal muscle damage. Nutr. Metab. 2009, 6, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, A.M.; Stout, J.R.; Jajtner, A.R.; Townsend, J.R.; Wells, A.J.; Beyer, K.S.; Boone, C.H.; Pruna, G.J.; Mangine, G.T.; Scanlon, T.M.; et al. Effects of β-Hydroxy-β-methylbutyrate (HMB)-free acid and cold water immersion on post exercise markers of muscle damage. J. Amino Acids 2014, 46, 1501–1511. [Google Scholar] [CrossRef] [PubMed]
- Faul, F.; Erdfelder, E.; Lang, A.G.; Buchner, A. G*Power 3: A flexible statistical power analysis program for the social, behavioural and biomedical sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, W.J.; Fry, A. ACSM’s Guidelines for Exercise Testing and Prescription, 6th ed.; Human Kinetics: Champaign, IL, USA, 1995. [Google Scholar]
- Arazi, H.; Asadi, A.; Roohi, S. Enhancing muscular performance in women: Compound versus complex, traditional resistance and plyometric training alone. J. Musculoskelet. Res. 2014, 17, 1–10. [Google Scholar] [CrossRef]
- Sayers, S.P.; Harackiewicz, D.V.; Harman, E.A. Cross-validation of three jump power equations. Med. Sci. Sports Exerc. 1999, 31, 572–577. [Google Scholar] [CrossRef] [PubMed]
- Hakkinen, K.; Pakarinen, A.; Kraemer, W.J.; Häkkinen, A.; Alen, M. Selective muscle hypertrophy, changes in EMG, and force, and serum hormones during strength training in older women. J. Appl. Physiol. 2001, 91, 569–580. [Google Scholar] [PubMed]
- Seitz, L.B.; Reyes, A.; Tran, T.T.; Saez de Villarreal, E.S.; Haff, G.G. Increases in lower-body strength transfer positively to sprint performance: A systematic review with meta-analysis. Sports Med. 2014, 44, 1693–1702. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.M.; Duncan, N.M.; Marin, P.J. Meta-analysis of post activation potentiation and power: Effects of conditioning activity, volume, gender, rest periods, and training status. J. Strength Con. Res. 2012, 27, 854–859. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, W.J.; Mazzetti, S.A.; Nindl, B.C.; Gotshalk, L.A.; Volek, J.S.; Bush, J.A.; Marx, J.O.; Dohi, K.; Gómez, A.L.; Miles, M.; et al. Effect of resistance training on women’s strength/power and occupational performances. Med. Sci. Sports Exerc. 2001, 33, 1011–1025. [Google Scholar] [CrossRef] [PubMed]
- Silva, V.R.; Belozo, F.L.; Micheletti, T.O.; Conrado, M.; Stout, J.R. β-Hydroxy-β-methylbutyrate free acid supplementation may improve recovery and muscle adaptations after resistance training: A systematic review. Nutr. Res. 2017, 45, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Portal, S.; Zadik, Z.; Rabinowitz, J.; Pilz-Burstein, R.; Adler-Portal, D.; Meckel, Y.; Cooper, D.M.; Eliakim, A.; Nemet, D. The effect of HMB supplementation on body composition, fitness, hormonal and inflammatory mediators in elite adolescent volleyball players: A prospective randomized, double-blind, placebo-controlled study. Eur. J. Appl. Physiol. 2011, 111, 2261–2269. [Google Scholar] [CrossRef] [PubMed]
- Slater, G.; Jenkins, D.; Logan, P.; Lee, H.; Vukovich, M.; Rathmacher, J.A.; Hahn, A.G. Beta-hydroxy-beta-methylbutyrate (HMB) supplementation does not affect changes in strength or body composition during resistance training in trained men. Int. J. Sport. Nutr. Exerc. Metab. 2001, 11, 384–396. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, D.M.; Crowe, M.J. Effects of six weeks of betahydroxy- beta-methylbutyrate (HMB) and HMB/creatine supplementation on strength, power, and anthropometry of highly trained athletes. J. Strength Cond. Res. 2007, 21, 419–423. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, W.J.; Hakkinen, K.; Newton, R.U.; Nindl, B.C.; Volek, J.S.; McCormick, M.; Gotshalk, L.A.; Gordon, S.E.; Fleck, S.J.; Campbell, W.W.; et al. Effects of heavy-resistance training on hormonal response patterns in younger vs. older men. J. Appl. Physiol. 1999, 87, 982–992. [Google Scholar] [PubMed]
- Alen, M.; Pakarinen, A.; Hakkinen, K.; Komi, P.V. Responses of serum anabolic and catabolic hormones to prolonged strength training. Int. J. Sports Med. 1998, 9, 229–233. [Google Scholar] [CrossRef]
- McCall, G.E.; Byrnes, W.C.; Fleck, S.J. Acute and chronic hormonal responses to resistance training designed to promote muscle hypertrophy. Can. J. Appl. Physiol. 1999, 24, 96–107. [Google Scholar] [CrossRef] [PubMed]
- Ahtiainen, J.P.; Pakarinen, A.; Alen, M. Muscle hypertrophy, hormonal adaptations and strength development during strength training in strength-trained and untrained men. Eur. J. Appl. Physiol. 2003, 89, 555–563. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, W.J.; Hatfield, D.L.; Volek, J.S.; Fragala, M.S.; Vingren, J.L.; Anderson, J.M.; Spiering, B.A.; Thomas, G.A.; Ho, J.Y.; Quann, E.E.; et al. Effects of amino acids supplement on physiological adaptations to resistance training. Med. Sci. Sport Exerc. 2009, 41, 1111–1121. [Google Scholar] [CrossRef] [PubMed]
- Hakkinen, K.; Pakarinen, A. Serum hormones in male strength athletes during intensive short term strength training. Eur. J. Appl. Physiol. 1991, 63, 191–199. [Google Scholar] [CrossRef]
- Di Pasquale, M. Essential amino acids. In Amino Acids and Proteins for the Athlete; Wolinsky, I., Ed.; CRC Press: Boca Raton, FL, USA, 1997; pp. 105–125. [Google Scholar]
| HMB-FA (n = 8) | Placebo (n = 8) | |
|---|---|---|
| Age (year) | 21.5 ± 0.5 | 21.3 ± 0.9 |
| Height (cm) | 181.7 ± 4.1 | 179.5 ± 4.5 |
| Body mass (kg) | 79.2 ± 13.0 | 78.9 ± 13.3 |
| BMI (kg/m2) | 23.9 ± 3.2 | 24.4 ± 4 |
| HMB-FA (n = 8) | Placebo (n = 8) | ||
|---|---|---|---|
| Energy intake (kcal) | Before | 2532 ± 210 | 2421 ± 176 |
| After | 2891 ± 198 | 2782 ± 215 | |
| Carbohydrate (g) | Before | 260 ± 23 | 259 ± 29 |
| After | 282 ± 31 | 278 ± 41 | |
| Fat (g) | Before | 75 ± 11 | 79 ± 13 |
| After | 84 ± 14 | 81 ± 17 | |
| Protein (g) | Before | 98 ± 19 | 93 ± 21 |
| After | 119 ± 22 | 115 ± 34 | |
| Vitamin E (mg) | Before | 8.9 ± 1.0 | 9.0 ± 2.0 |
| After | 10.0 ± 2.0 | 9.5 ± 1.6 | |
| Vitamin C (mg) | Before | 65 ± 22 | 67 ± 17 |
| After | 77 ± 18 | 78 ± 15 |
| HMB-FA (n = 8) | Placebo (n = 8) | Significance | |||
|---|---|---|---|---|---|
| GH (ng/mL) | |||||
| Pre | 0.10 ± 0.03 | 0.11 ± 0.04 | G = 0.98 | ||
| Post | 0.19 ± 0.09 * | 0.17 ± 0.07 * | T = 0.02 | ||
| Effect Size | 1.41 (−0.25, 2.42) | Very large | 0.12 (−0.87, 1.10) | Trivial | G × T= 0.51 |
| IGF-1 (ng/mL) | |||||
| Pre | 256.2 ± 28.5 | 250.0 ± 36.9 | G = 0.55 | ||
| Post | 284.0 ± 37.9 * | 266.5 ± 43.6 * | T = 0.05 | ||
| Effect Size | 0.83 (−0.23, 1.80) | Large | 0.41 (−0.60, 1.38) | Small | G × T = 0.331 |
| TEST (ng/mL) | |||||
| Pre | 5.47 ± 0.77 | 5.33 ± 1.10 | G = 0.08 | ||
| Post | 6.11 ± 1.40 | 5.41 ± 0.75 | T = 0.33 | ||
| Effect Size | 0.57 (−0.46, 1.53) | Moderate | 0.08 (−0.90, 1.38) | Trivial | G × T= 0.53 |
| CORT (µg/dl) | |||||
| Pre | 23.0 ± 4.6 | 23.9 ± 2.9 | G = 0.07 | ||
| Post | 16.6 ± 4.3 *,** | 24.5 ± 5.4 * | T = 0.02 | ||
| Effect Size | −1.89 (−2.94, 0.62) | Very large | 0.14 (−0.85,1.11) | Trivial | G × T = 0.05 |
| ACTH (pg/mL) | |||||
| Pre | 58.1 ± 10.8 | 68.8 ± 5.4 | G = 0.06 | ||
| Post | 38.1 ± 12.1 *,** | 60.1 ± 10.5 * | T = 0.002 | ||
| Effect Size | −1.76 (−2.8, 0.52) | Very large | −1.04 (−2.02, 0.05) | Large | G × T = 0.004 |
| Cr (mg/dL) | |||||
| Pre | 0.98 ± 0.06 | 1.01 ± 0.09 | G = 0.16 | ||
| Post | 0.98 ± 0.09 | 1.06 ± 0.08 | T = 0.16 | ||
| Effect Size | 0.01 (−0.98, 1.00) | Trivial | 0.59 (−0.44, 1.55) | Moderate | G × T = 0.27 |
| Urea (mg/dL) | |||||
| Pre | 11.6 ± 1.9 | 11.6 ± 1.6 | G = 0.70 | ||
| Post | 12.8 ± 2.0 | 13.6 ± 3.2 | T = 0.06 | ||
| Effect Size | 0.61 (−0.42, 1.58) | Moderate | 0.8 (−0.26, 1.77) | Moderate | G × T = 0.24 |
| 1RM Leg Press (kg) | 1RM Bench Press (kg) | Vertical Jump (cm) | Peak Power (w) | |
|---|---|---|---|---|
| GH (ng/mL) | r = 0.242 p = 0.366 | r = 0.031 p = 0.911 | r = 0.262 p = 0.328 | r = −0.194 p = 0.472 |
| IGF-1 (ng/mL) | r = 0.649 p = 0.006 | r = 0.623 p = 0.01 | r = 0.272 p = 0.307 | r = 0.155 p = 0.567 |
| TEST (ng/mL) | r = 0.00 p = 0.999 | r =0. 093 p = 0.732 | r = 0.001 p = 0.997 | r = 0.278 p = 0.297 |
| CORT (µg/dl) | r =−0.126 p = 0.641 | r = 0.135 p =0.618 | r = −0.299 p = 0.261 | r = −0.094 p = 0.730 |
| ACTH (pg/ml) | r = -0.178 p = 0.509 | r = −0.120 p = 0.659 | r = −0.330 p = 0.211 | r = −0.081 p = 0.766 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Asadi, A.; Arazi, H.; Suzuki, K. Effects of β-Hydroxy-β-methylbutyrate-free Acid Supplementation on Strength, Power and Hormonal Adaptations Following Resistance Training. Nutrients 2017, 9, 1316. https://doi.org/10.3390/nu9121316
Asadi A, Arazi H, Suzuki K. Effects of β-Hydroxy-β-methylbutyrate-free Acid Supplementation on Strength, Power and Hormonal Adaptations Following Resistance Training. Nutrients. 2017; 9(12):1316. https://doi.org/10.3390/nu9121316
Chicago/Turabian StyleAsadi, Abbas, Hamid Arazi, and Katsuhiko Suzuki. 2017. "Effects of β-Hydroxy-β-methylbutyrate-free Acid Supplementation on Strength, Power and Hormonal Adaptations Following Resistance Training" Nutrients 9, no. 12: 1316. https://doi.org/10.3390/nu9121316
APA StyleAsadi, A., Arazi, H., & Suzuki, K. (2017). Effects of β-Hydroxy-β-methylbutyrate-free Acid Supplementation on Strength, Power and Hormonal Adaptations Following Resistance Training. Nutrients, 9(12), 1316. https://doi.org/10.3390/nu9121316







