Effects of a Lutein and Zeaxanthin Intervention on Cognitive Function: A Randomized, Double-Masked, Placebo-Controlled Trial of Younger Healthy Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Ethics
2.3. Methods
2.3.1. Retinal L + Z Levels
2.3.2. Serum L + Z Levels
2.3.3. Cognitive Function
2.3.4. Statistical Analyses and other Considerations
3. Results
3.1. Retinal L + Z Levels
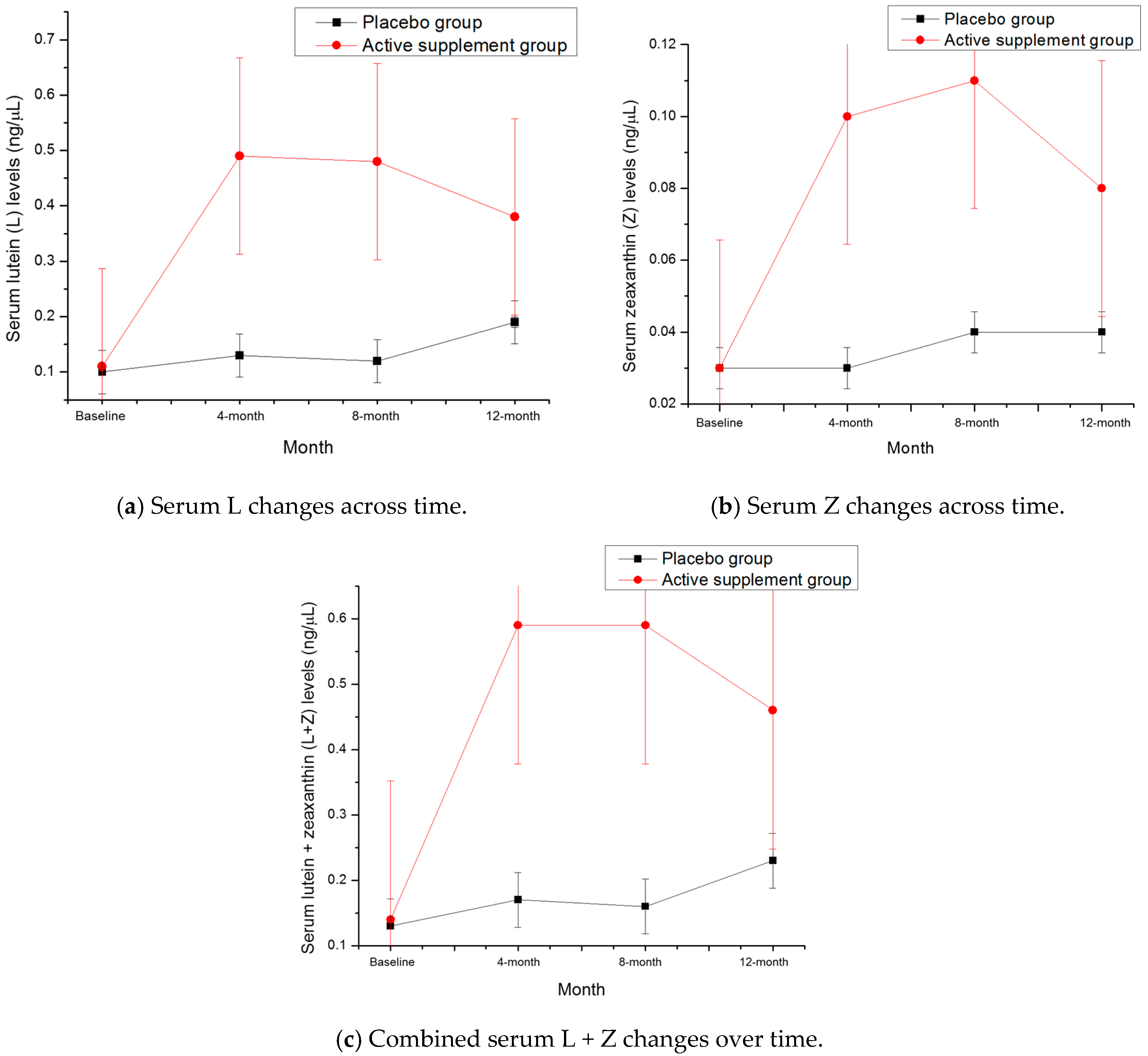
3.2. Serum L + Z Levels
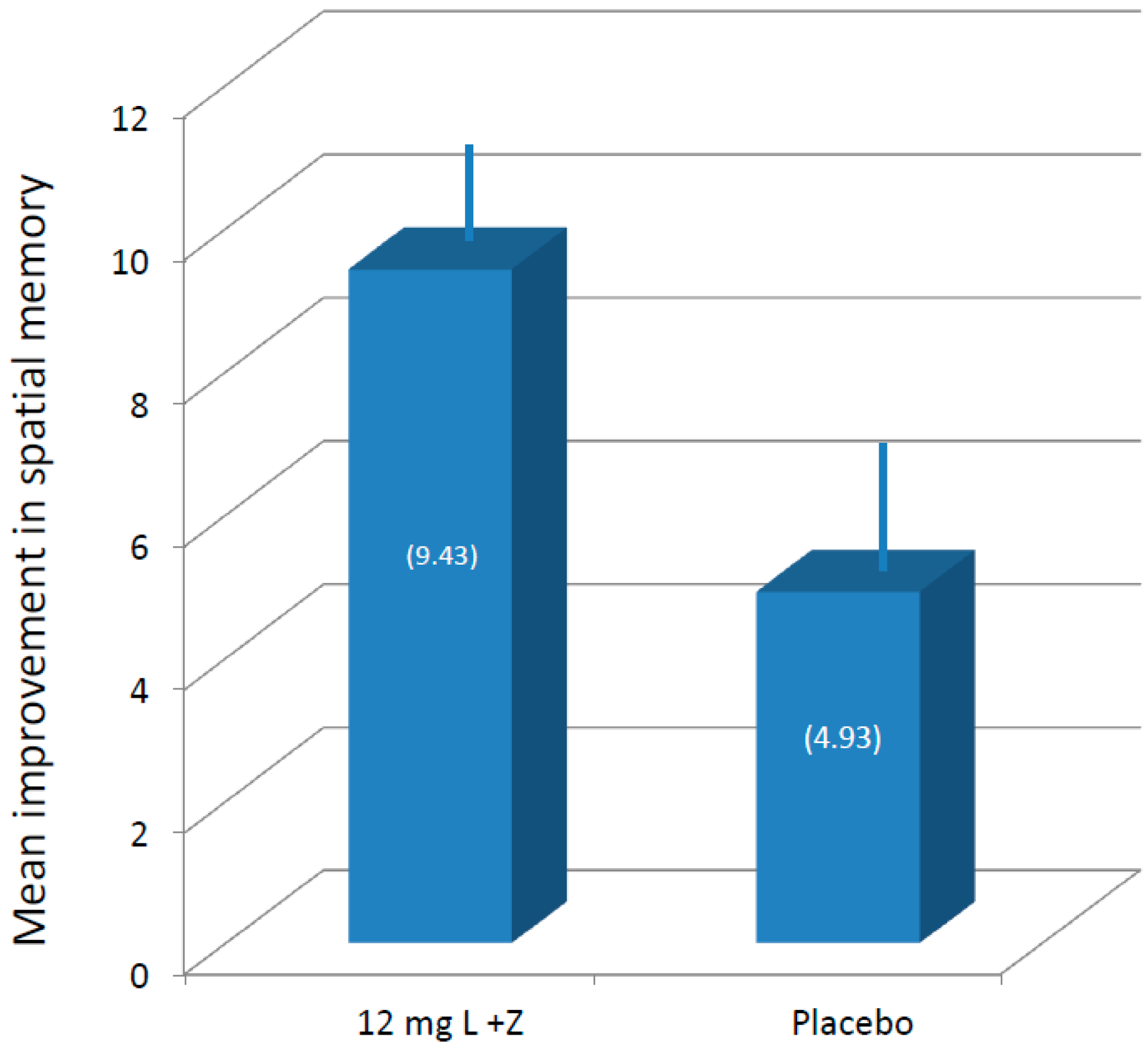
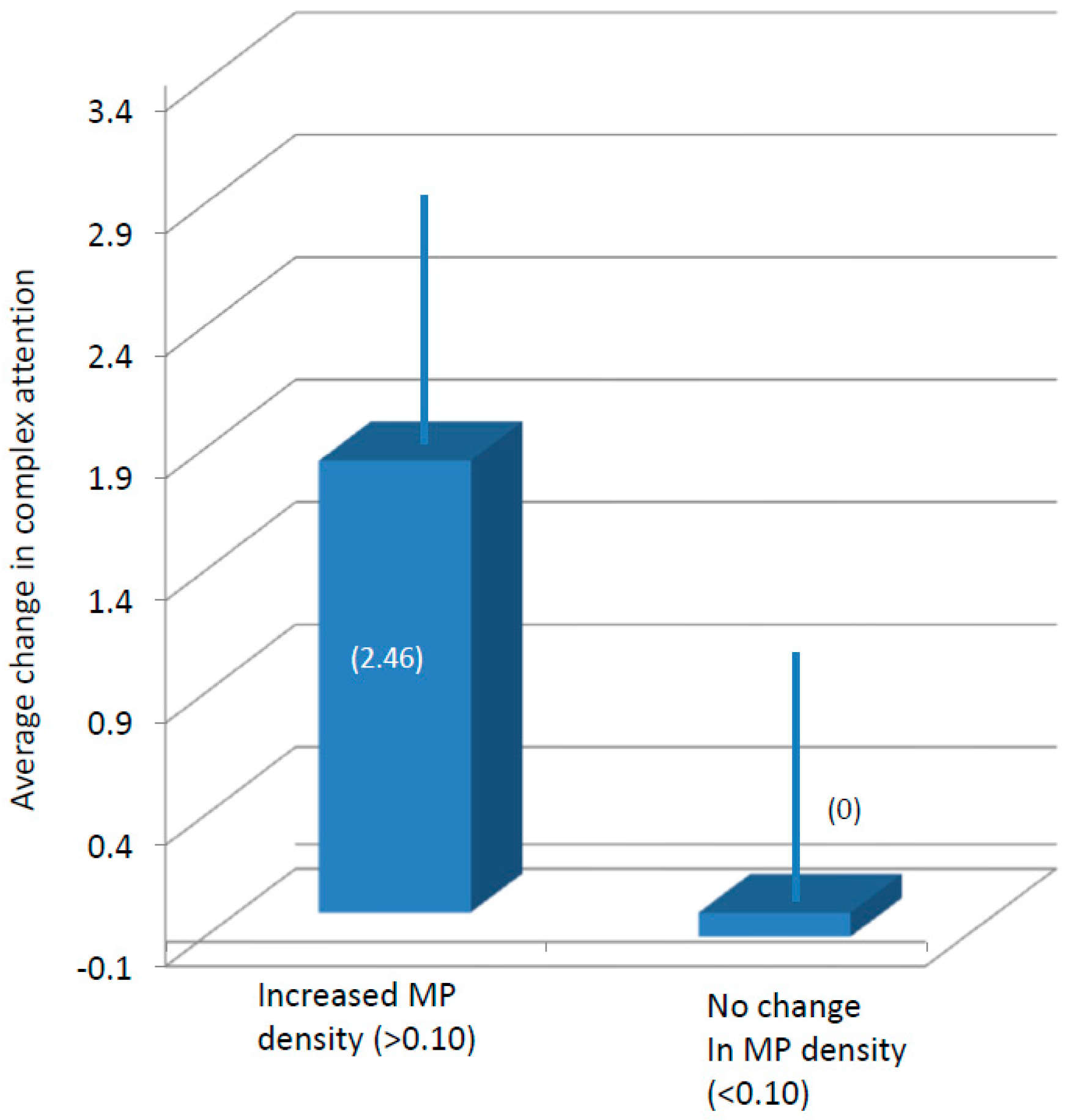
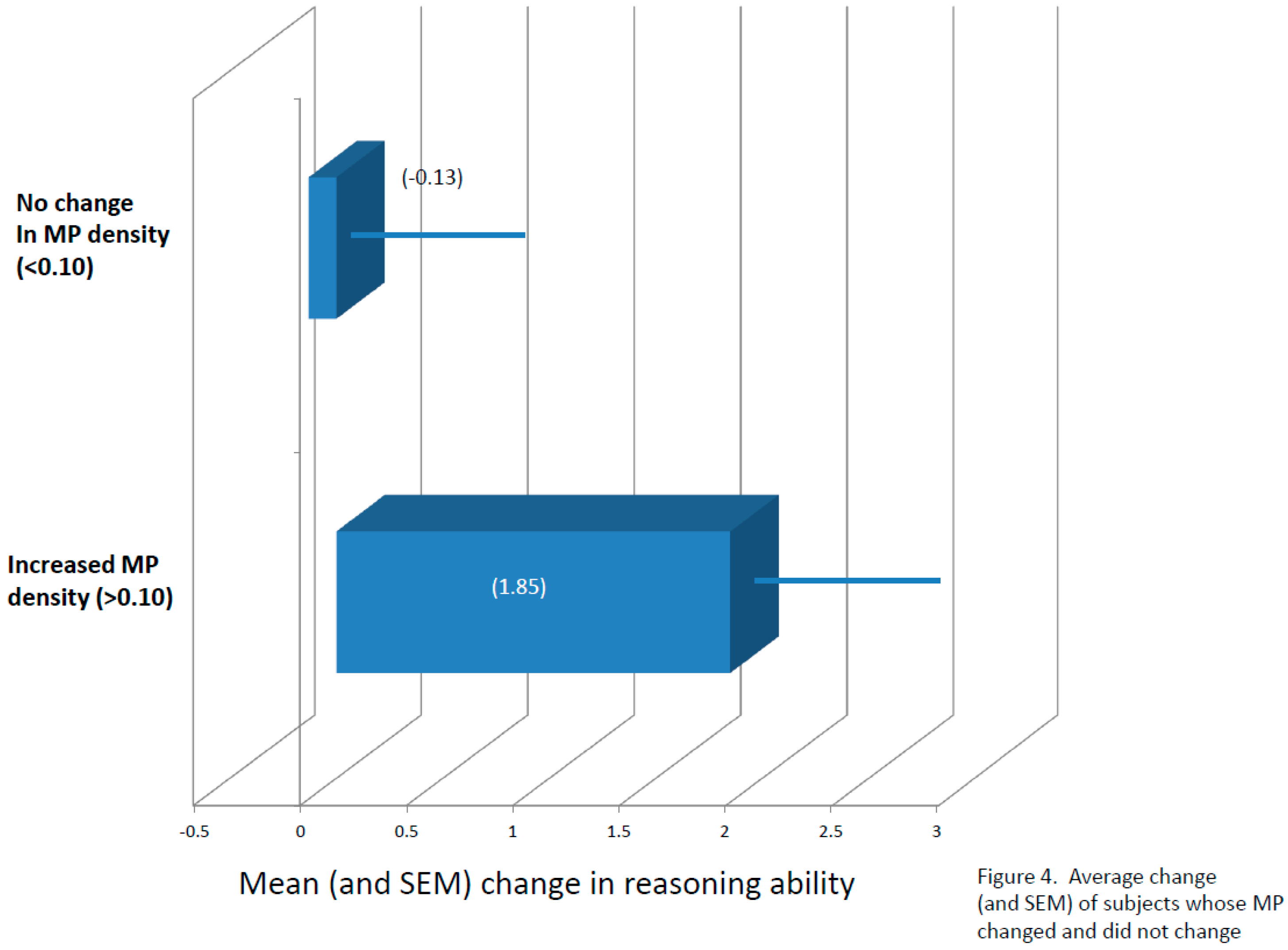
3.3. Cognitive Function
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
Registry Information
Abbreviations
| L | lutein |
| Z | zeaxanthin |
| MPOD | macular pigment optical density |
| MZ | meso-zeaxanthin |
| CNS | central nervous system |
| MP | macular pigment |
| HPLC | high-performance liquid chromatography |
| FFQ | food frequency questionnaire |
| RCI | reliable change index |
| VeM | verbal memory |
| ViM | visual memory |
| R | reasoning |
| EF | executive function |
| PmS | psychomotor speed |
| CA | complex attention |
| CF | cognitive flexibility |
| NVRT | non-verbal reasoning test |
| SAT | shifting attention test |
| FTT | finger tapping test |
| SDC | symbol-digit coding |
| ST | Stroop test |
| CPT | continuous performance task |
References
- Gomez-Pinilla, F. Brain foods: The effects of nutrients on brain function. Nat. Rev. Neurosci. 2008, 9, 568–578. [Google Scholar] [CrossRef] [PubMed]
- Joseph, J.A.; Shukitt-Hale, B.; Casadesus, G.; Fisher, D. Oxidative stress and inflammation in brain aging: Nutritional considerations. Neurochem. Res. 2005, 30, 927–935. [Google Scholar] [CrossRef] [PubMed]
- Bourre, J.M. Effects of nutrients (in food) on the structure and function of the nervous system: Update on dietary requirements for brain. Part 1: Micronutrients. J. Nutr. Health Aging 2006, 10, 377–385. [Google Scholar] [PubMed]
- Carlson, S.E.; Carver, J.D.; House, S.G. High fat diets varying in ratios of polyunsaturated to saturated fatty acid and linoleic to linolenic acid: A comparison of rat neural and red cell membrane phospholipids. J. Nutr. 1986, 116, 718–725. [Google Scholar] [PubMed]
- Cook, C.C.; Thomson, A.D. B-complex vitamins in the prophylaxis and treatment of Wernicke-Korsakoff syndrome. Br. J. Hosp. Med. 1997, 57, 461–465. [Google Scholar] [PubMed]
- McCaddon, A.; Regland, B.; Hudson, P.; Davies, G. Functional vitamin B12 deficiency and Alzheimer disease. Neurology 2002, 58, 1395–1399. [Google Scholar] [CrossRef] [PubMed]
- Erdman, J.W.; Smith, J.W.; Kuchan, M.J.; Mohn, E.S.; Johnson, E.J.; Rubakhin, S.S.; Wang, L.; Sweedler, J.V.; Neuringer, M. Lutein and Brain Function. Foods 2015, 4, 547–564. [Google Scholar] [CrossRef] [PubMed]
- Hammond, B.R.J.; Wooten, B.R.; Smollon, B. Assessment of the validity of in vivo methods of measuring human macular pigment optical density. Optom. Vis. Sci. 2005, 82, 387–404. [Google Scholar] [CrossRef] [PubMed]
- Vishwanathan, R.; Schalch, W.; Johnson, E.J. Macular pigment carotenoids in the retina and occipital cortex are related in humans. Nutr. Neurosci. 2015, 19, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Renzi, L.M.; Dengler, M.J.; Puente, A.; Miller, L.S.; Hammond, B.R.J. Relationships between macular pigment optical density and cognitive function in unimpaired and mildly cognitively impaired older adults. Neurobiol. Aging 2014, 35, 1695–1699. [Google Scholar] [CrossRef] [PubMed]
- Feeney, J.; Finucane, C.; Savva, G.M.; Cronin, H.; Beatty, S.; Nolan, J.M.; Kenny, R.A. Low macular pigment optical density is associated with lower cognitive performance in a large, population-based sample of older adults. Neurobiol. Aging 2013, 34, 2449–2456. [Google Scholar] [CrossRef] [PubMed]
- Kelly, D.; Coen, R.F.; Akuffo, K.O.; Beatty, S.; Dennison, J.; Moran, R.; Stack, J.; Howard, A.N.; Mulcahy, R.; Nolan, J.M. Cognitive Function and Its Relationship with Macular Pigment Optical Density and Serum Concentrations of its Constituent Carotenoids. J. Alzheimers Dis. 2015, 48, 261–277. [Google Scholar] [CrossRef] [PubMed]
- Vishwanathan, R.; Iannaccone, A.; Scott, T.M.; Kritchevsky, S.B.; Jennings, B.J.; Carboni, G.; Forma, G.; Satterfield, S.; Harris, T.; Johnson, K.C.; et al. Macular pigment optical density is related to cognitive function in older people. Age Ageing 2014, 43, 271–275. [Google Scholar] [CrossRef] [PubMed]
- Bovier, E.R.; Renzi, L.M.; Hammond, B.R. A double-blind, placebo-controlled study on the effects of lutein and zeaxanthin on neural processing speed and efficiency. PLoS ONE 2014, 9, e108178. [Google Scholar] [CrossRef] [PubMed]
- Bovier, E.R.; Hammond, B.R. A randomized placebo-controlled study on the effects of lutein and zeaxanthin on visual processing speed in young healthy subjects. Arch. Biochem. Biophys. 2015, 572, 54–57. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.; Vishwanathan, R.; Mohn, E.; Haddock, J.; Rasmussen, H.; Scott, T. Avocado Consumption Increases Neural Lutein and Improves Cognitive Function. FASEB J. 2015, 29, 32.8. [Google Scholar]
- Lindbergh, C.A.; Renzi-Hammond, L.M.; Hammond, B.R.; Terry, D.P.; Mewborn, C.M.; Puente, A.N.; Miller, L.S. Lutein and Zeaxanthin Influence Brain Function in Older Adults: A Randomized Controlled Trial. J. Int. Neuropsychol. Soc. 2017, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Hammond, B.R.; Miller, L.S.; Bello, M.O.; Lindbergh, C.A.; Mewborn, C.; Renzi-Hammond, L.M. Effects of Lutein/Zeaxanthin Supplementation on the Cognitive Function of Community Dwelling Older Adults: A Randomized, Double-Masked, Placebo-Controlled Trial. Front. Aging Neurosci. 2017, 9, 254. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.J.; McDonald, K.; Caldarella, S.M.; Chung, H.-Y.; Troen, A.M.; Snodderly, D.M. Cognitive findings of an exploratory trial of docosahexaenoic acid and lutein supplementation in older women. Nutr. Neurosci. 2008, 11, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Zlody, R.L. The Relationship between Critical Flicker Frequency (CFF) and Several Intellectual Measures. Am. J. Psychol. 1965, 78, 596. [Google Scholar] [CrossRef] [PubMed]
- Owsley, C. Visual processing speed. Vis. Res. 2013, 90, 52–56. [Google Scholar] [CrossRef] [PubMed]
- Satlhouse, D.J.; Timothy, A.; Madden, D.J. Information Processing Speed and Aging. In Information Processing Speed in Clinical Populations; DeLuca, J., Kalmar, J.H., Eds.; Taylor and Frances: New York, NY, USA, 2008; pp. 221–242. [Google Scholar]
- Stringham, J.M.; Hammond, B.R.; Nolan, J.M.; Wooten, B.R.; Mammen, A.; Smollon, W.; Snodderly, D.M. The utility of using customized heterochromatic flicker photometry (cHFP) to measure macular pigment in patients with age-related macular degeneration. Exp. Eye Res. 2008, 87, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Wooten, B.R.; Hammond, B.R.; Land, R.I.; Snodderly, D.M. A practical method for measuring macular pigment optical density. Investig. Ophthalmol. Vis. Sci. 1999, 40, 2481–2489. [Google Scholar]
- Lindbergh, C.A.; Mewborn, C.M.; Hammond, B.R.; Renzi-Hammond, L.M.; Curran-Celentano, J.M.; Miller, L.S. Relationship of Lutein and Zeaxanthin Levels to Neurocognitive Functioning: An fMRI Study of Older Adults. J. Int. Neuropsychol. Soc. 2017, 23, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Gualtieri, C.T.; Johnson, L.G. Reliability and validity of a computerized neurocognitive test battery, CNS Vital Signs. Arch. Clin. Neuropsychol. 2006, 21, 623–643. [Google Scholar] [CrossRef] [PubMed]
- Hammond, B.R., Jr.; Johnson, E.J.; Russell, R.M.; Krinsky, N.I.; Yeum, K.J.; Edwards, R.B.; Snodderly, D.M. Dietary modification of human macular pigment density. Investig. Ophthalmol. Vis. Sci. 1997, 38, 1795–1801. [Google Scholar]
- Hammond, B.R.; Caruso-Avery, M. Macular pigment optical density in a Southwestern sample. Investig. Ophthalmol. Vis. Sci. 2000, 41, 1492–1497. [Google Scholar]
- Curran-Celentano, J.; Hammond, B.R.; Ciulla, T.A.; Cooper, D.A.; Pratt, L.M.; Danis, R.B. Relation between dietary intake, serum concentrations, and retinal concentrations of lutein and zeaxanthin in adults in a Midwest population. Am. J. Clin. Nutr. 2001, 74, 796–802. [Google Scholar] [PubMed]
- Craft, N.; Dorey, C.K. Carotenoid, tocopherol, and retinal concentrations in elderly human brain. J. Nutr. Health Aging 2004, 8, 156–162. [Google Scholar] [PubMed]
- Murray, E.A. What have ablation studies told us about the neural substrates of stimulus memory? Semin. Neurosci. 1996, 8, 13–22. [Google Scholar] [CrossRef]
- Downing, P.E. Interactions between visual working memory and selective attention. Psychol. Sci. 2000, 11, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Lean, G.; (Ken) Clements, M.A. Spatial ability, visual imagery, and mathematical performance. Educ. Stud. Math. 1981, 12, 267–299. [Google Scholar] [CrossRef]
- Mewborn, C.; Terry, D.; Lindbergh, C.; Renzi-Hammond, L.; Hammond, B.; Miller, L. Aging and Dementia-2Retinal and Serum Lutein and Zeaxanthin: Relation to white Matter Integrity in Younger and Older Adults. Arch. Clin. Neuropsychol. 2016, 31, 573. [Google Scholar] [CrossRef][Green Version]
- Renzi, L.M.; Hammond, B.R. The relation between the macular carotenoids, lutein and zeaxanthin, and temporal vision. Ophthalmic Physiol. Opt. 2010, 30, 351–357. [Google Scholar] [CrossRef] [PubMed]
| Group | Age (Years) | Body Mass Index | Gender | Ethnicity | Race | Years of Education | MP Optical Density |
|---|---|---|---|---|---|---|---|
| Active, Mean ± SD | 21.50 ± 2.69 | 24.15 ± 3.86 | 21 M, 16 F | n = 35 non-Hispanic n = 2 Hispanic | n = 30 White n = 4 Black n = 1 pan-Asian n = 2 Latino | 12+ | 0.47 ± 0.18 |
| Placebo, Mean ± SD | 20.50 ± 1.91 | 23.03 ± 3.98 | 8 M, 6 F | n = 14 non-Hispanic | n = 10 White n = 2 Black n = 2 pan-Asian | 12+ | 0.39 ± 0.12 |
| Domain | Corresponding Tests | Computation Procedure |
|---|---|---|
| Verbal Memory (VeM) | Verbal Memory Test | Correct hits for presented words + correct passes on distractors for tests immediately after presentation and after a 30-min delay. |
| Visual Memory (ViM) | Visual Memory Test | Correct hits for presented shapes and symbols + correct passes on distractors for tests immediately after presentation and after a 30-min delay. |
| Reasoning (R) | Non-Verbal Reasoning Test (NVRT) | Correct responses on the NVRT − commission errors on the NVRT. |
| Executive Function (EF) | Shifting Attention Test (SAT) | Correct responses on the SAT − errors on the SAT. |
| Psychomotor Speed (PmS) | Finger Tapping Test (FTT) Symbol-Digit Coding Test (SDC) | Average number of taps on the FTT with the right hand + average number of taps with the left hand + number of correct responses on the SDC |
| Complex Attention (CA) | Stroop Test (ST) SAT Continuous Performance Task (CPT) | Commission errors on the ST + Errors on the SAT + Commission and omission errors on the CPT |
| Cognitive Flexibility (CF) | SAT ST | Correct responses on the SAT − errors on the SAT − Commission errors on the ST |
| Group | Serum Lutein (ng/μL) | Serum Zeaxanthin (ng/μL) | Lutein + Zeaxanthin (ng/μL) |
|---|---|---|---|
| Active, Mean ± SD | 0.11 ± 0.07 | 0.03 ± 0.02 | 0.14 ± 0.08 |
| Placebo, Mean ± SD | 0.10 ± 0.03 | 0.03 ± 0.02 | 0.13 ± 0.04 |
| Group | Serum Lutein (ng/μL) | Serum Zeaxanthin (ng/μL) | Lutein + Zeaxanthin (ng/μL) |
|---|---|---|---|
| Active, Mean ± SD | 0.38 ± 0.28 | 0.08 ± 0.05 | 0.46 ± 0.32 |
| Placebo, Mean ± SD | 0.19 ± 0.19 | 0.04 ± 0.04 | 0.23 ± 0.23 |
| Group | Verbal Memory | Visual Memory | Reasoning Ability | Executive Function | Complex Attention * | Cognitive Flexibility |
|---|---|---|---|---|---|---|
| Baseline Mean, all subjects | 54.22 | 41.04 | 9.06 | 53.73 | 7.38 | 52.02 |
| 12-Month Mean, all subjects | 56.26 | 49.24 | 10.20 | 62.59 | 5.98 | 60.65 |
| Average change between baseline and 12 months, placebo group participants | 3.57 | 4.93 | 1.64 | 8.86 | 0.46 | 8.43 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Renzi-Hammond, L.M.; Bovier, E.R.; Fletcher, L.M.; Miller, L.S.; Mewborn, C.M.; Lindbergh, C.A.; Baxter, J.H.; Hammond, B.R. Effects of a Lutein and Zeaxanthin Intervention on Cognitive Function: A Randomized, Double-Masked, Placebo-Controlled Trial of Younger Healthy Adults. Nutrients 2017, 9, 1246. https://doi.org/10.3390/nu9111246
Renzi-Hammond LM, Bovier ER, Fletcher LM, Miller LS, Mewborn CM, Lindbergh CA, Baxter JH, Hammond BR. Effects of a Lutein and Zeaxanthin Intervention on Cognitive Function: A Randomized, Double-Masked, Placebo-Controlled Trial of Younger Healthy Adults. Nutrients. 2017; 9(11):1246. https://doi.org/10.3390/nu9111246
Chicago/Turabian StyleRenzi-Hammond, Lisa M., Emily R. Bovier, Laura M. Fletcher, L. Stephen Miller, Catherine M. Mewborn, Cutter A. Lindbergh, Jeffrey H. Baxter, and Billy R. Hammond. 2017. "Effects of a Lutein and Zeaxanthin Intervention on Cognitive Function: A Randomized, Double-Masked, Placebo-Controlled Trial of Younger Healthy Adults" Nutrients 9, no. 11: 1246. https://doi.org/10.3390/nu9111246
APA StyleRenzi-Hammond, L. M., Bovier, E. R., Fletcher, L. M., Miller, L. S., Mewborn, C. M., Lindbergh, C. A., Baxter, J. H., & Hammond, B. R. (2017). Effects of a Lutein and Zeaxanthin Intervention on Cognitive Function: A Randomized, Double-Masked, Placebo-Controlled Trial of Younger Healthy Adults. Nutrients, 9(11), 1246. https://doi.org/10.3390/nu9111246





