Bioactive Compounds and Their Neuroprotective Effects in Diabetic Complications
Abstract
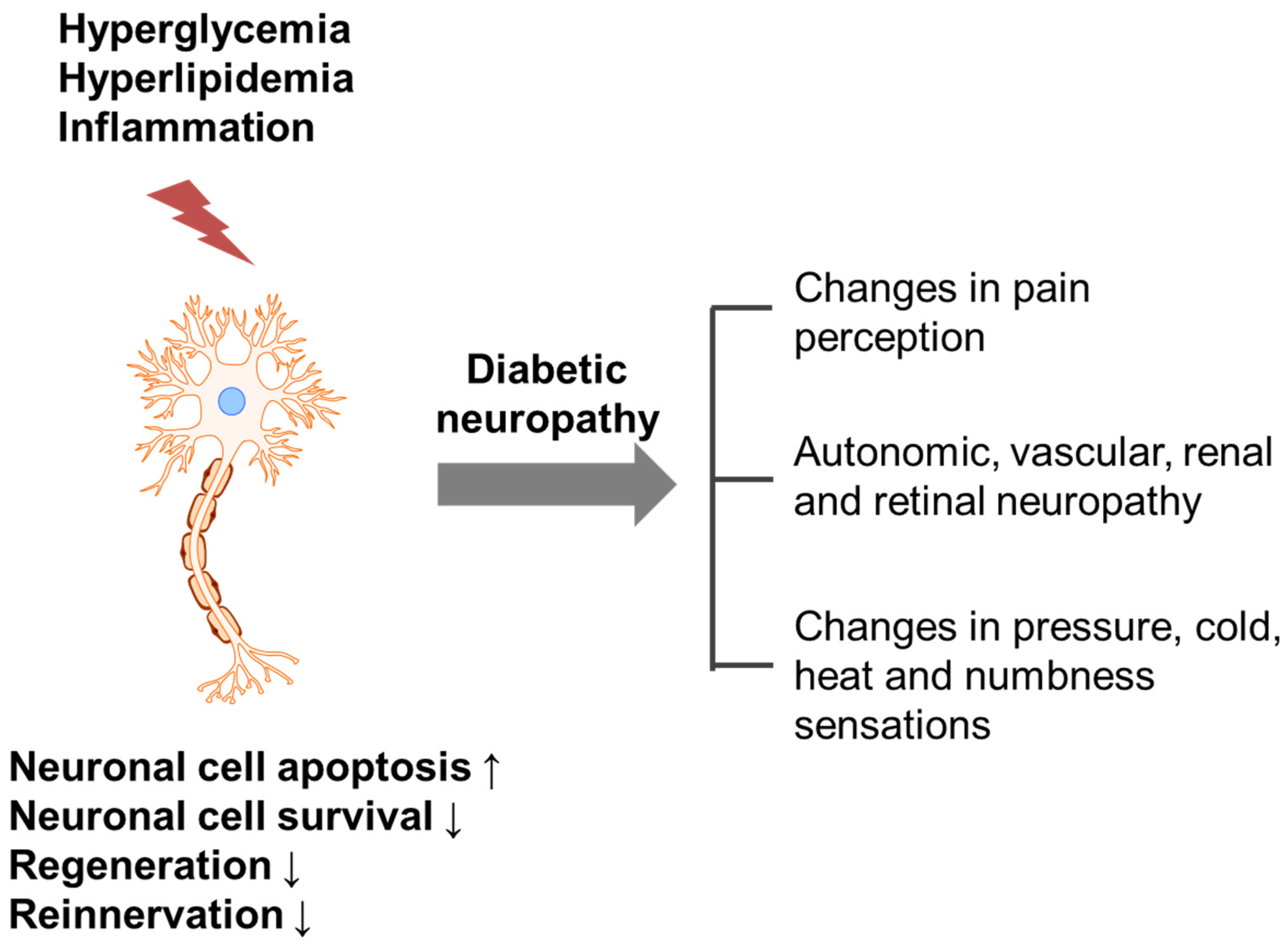
:1. Introduction
2. Causes of Diabetic Neuropathy and Related Pathophysiology
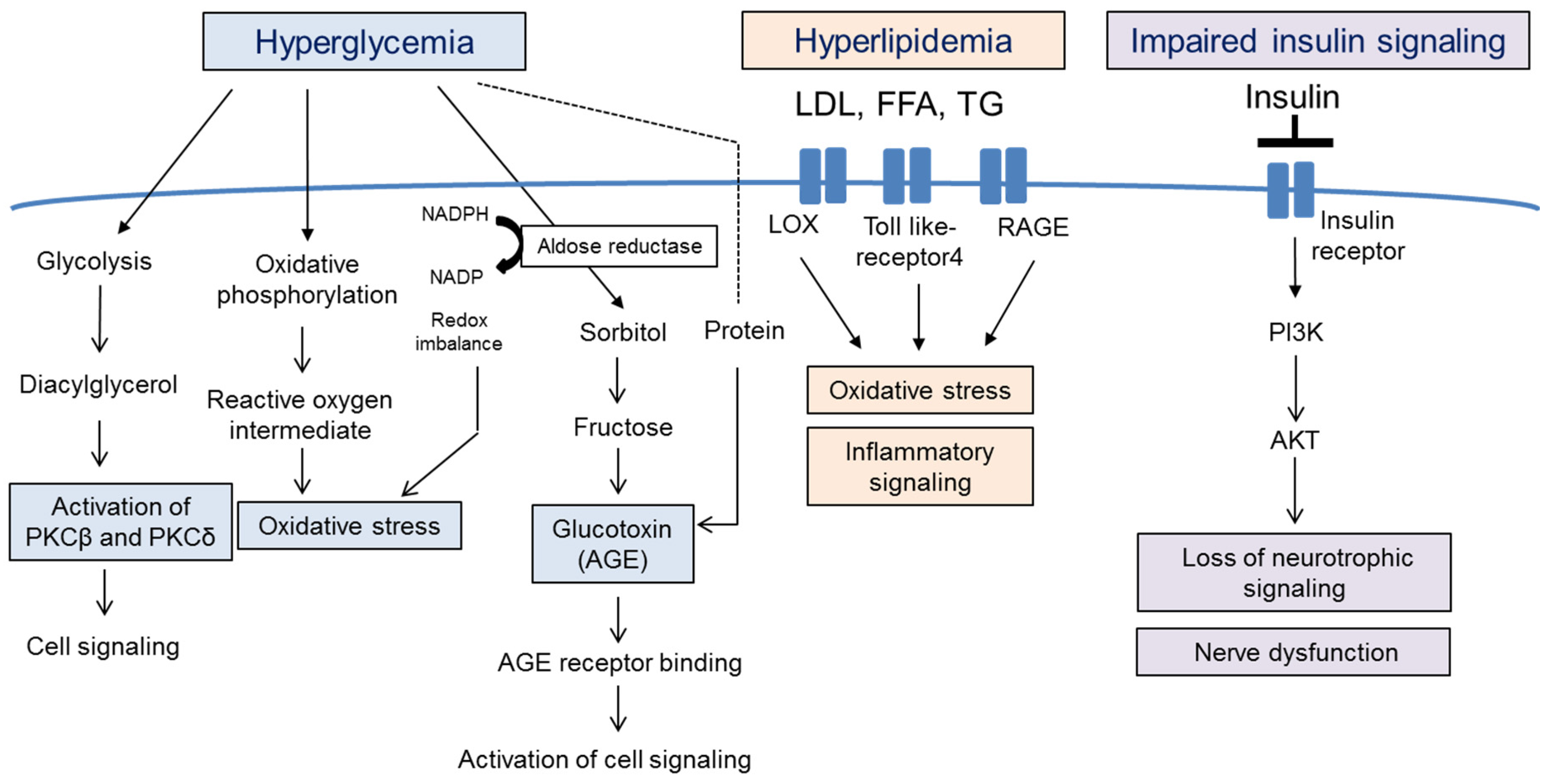
2.1. Hyperglycemia
2.2. Hyperlipidemia
2.3. Impairment of Insulin Signaling
3. Method of Literature Mining
4. Effect of Flavonoids on Neuronal Cell Death and Dysfunction
4.1. Baicalein
4.2. Chrysin
4.3. Diosmin
4.4. Epigallocatechin-3-Gallate
4.5. Hesperidin
4.6. Kaempferol
4.7. Luteolin
4.8. Myricetin
4.9. Naringenin
4.10. Proanthocyanidin
4.11. Puerarin
4.12. Quercetin
4.13. Rutin
4.14. Silibinin
5. Effect of Vitamins on Neuronal Cell Death and Dysfunction
5.1. Vitamin A
5.2. Vitamin C
5.3. Vitamin D
5.4. Vitamin E
6. Conclusions
Acknowledgments
Conflicts of Interest
References
- Atkinson, M.A.; Eisenbarth, G.S. Type 1 diabetes: New perspectives on disease pathogenesis and treatment. Lancet 2001, 358, 221–229. [Google Scholar] [CrossRef]
- Lin, Y.; Sun, Z. Current views on type 2 diabetes. J. Endocrinol. 2010, 204, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Russell, N.D.; Cooper, M.E. 50 years forward: Mechanisms of hyperglycaemia-driven diabetic complications. Diabetologia 2015, 58, 1708–1714. [Google Scholar] [CrossRef] [PubMed]
- Dobretsov, M.; Romanovsky, D.; Stimers, J.R. Early diabetic neuropathy: Triggers and mechanisms. World J. Gastroenterol. WJG 2007, 13, 175–191. [Google Scholar] [CrossRef] [PubMed]
- Callaghan, B.C.; Cheng, H.T.; Stables, C.L.; Smith, A.L.; Feldman, E.L. Diabetic neuropathy: Clinical manifestations and current treatments. Lancet. Neurol. 2012, 11, 521–534. [Google Scholar] [CrossRef]
- Tesfaye, S.; Selvarajah, D. Advances in the epidemiology, pathogenesis and management of diabetic peripheral neuropathy. Diabetes/Metab. Res. Rev. 2012, 28, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Shaikh, A.S.; Somani, R.S. Animal models and biomarkers of neuropathy in diabetic rodents. Indian J. Pharmacol. 2010, 42, 129–134. [Google Scholar] [PubMed]
- Ziegler, D.; Sohr, C.G.; Nourooz-Zadeh, J. Oxidative stress and antioxidant defense in relation to the severity of diabetic polyneuropathy and cardiovascular autonomic neuropathy. Diabetes Care 2004, 27, 2178–2183. [Google Scholar] [CrossRef] [PubMed]
- Sheetz, M.J.; King, G.L. Molecular understanding of hyperglycemia′s adverse effects for diabetic complications. J. Am. Med. Assoc. 2002, 288, 2579–2588. [Google Scholar] [CrossRef]
- Vincent, A.M.; Russell, J.W.; Low, P.; Feldman, E.L. Oxidative stress in the pathogenesis of diabetic neuropathy. Endocr. Rev. 2004, 25, 612–628. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, A.M.; Hofmann, M.; Taguchi, A.; Yan, S.D.; Stern, D.M. Rage: A multiligand receptor contributing to the cellular response in diabetic vasculopathy and inflammation. Semin. Thromb. Hemost. 2000, 26, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Yue, D.K.; Hanwell, M.A.; Satchell, P.M.; Turtle, J.R. The effect of aldose reductase inhibition on motor nerve conduction velocity in diabetic rats. Diabetes 1982, 31, 789–794. [Google Scholar] [CrossRef] [PubMed]
- Cameron, N.E.; Leonard, M.B.; Ross, I.S.; Whiting, P.H. The effects of sorbinil on peripheral nerve conduction velocity, polyol concentrations and morphology in the streptozotocin-diabetic rat. Diabetologia 1986, 29, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Goto, Y.; Hotta, N.; Shigeta, Y.; Sakamoto, N.; Kikkawa, R. Effects of an aldose reductase inhibitor, epalrestat, on diabetic neuropathy. Clinical benefit and indication for the drug assessed from the results of a placebo-controlled double-blind study. Biome. Pharmacother. 1995, 49, 269–277. [Google Scholar] [CrossRef]
- Padilla, A.; Descorbeth, M.; Almeyda, A.L.; Payne, K.; de Leon, M. Hyperglycemia magnifies schwann cell dysfunction and cell death triggered by PA-induced lipotoxicity. Brain Res. 2011, 1370, 64–79. [Google Scholar] [CrossRef] [PubMed]
- Vincent, A.M.; Hayes, J.M.; McLean, L.L.; Vivekanandan-Giri, A.; Pennathur, S.; Feldman, E.L. Dyslipidemia-induced neuropathy in mice: The role of oxLDL/LOX-1. Diabetes 2009, 58, 2376–2385. [Google Scholar] [CrossRef] [PubMed]
- Vincent, A.M.; Perrone, L.; Sullivan, K.A.; Backus, C.; Sastry, A.M.; Lastoskie, C.; Feldman, E.L. Receptor for advanced glycation end products activation injures primary sensory neurons via oxidative stress. Endocrinology 2007, 148, 548–558. [Google Scholar] [CrossRef] [PubMed]
- Lam, K.S.; Cheng, I.K.; Janus, E.D.; Pang, R.W. Cholesterol-lowering therapy may retard the progression of diabetic nephropathy. Diabetologia 1995, 38, 604–609. [Google Scholar] [CrossRef] [PubMed]
- Corrao, G.; Zambon, A.; Bertu, L.; Botteri, E.; Leoni, O.; Contiero, P. Lipid lowering drugs prescription and the risk of peripheral neuropathy: An exploratory case-control study using automated databases. J. Epidemiol. Community Health 2004, 58, 1047–1051. [Google Scholar] [CrossRef] [PubMed]
- Toth, C.; Brussee, V.; Martinez, J.A.; McDonald, D.; Cunningham, F.A.; Zochodne, D.W. Rescue and regeneration of injured peripheral nerve axons by intrathecal insulin. Neuroscience 2006, 139, 429–449. [Google Scholar] [CrossRef] [PubMed]
- Sima, A.A.; Zhang, W.; Grunberger, G. Type 1 diabetic neuropathy and c-peptide. Exp. Diabesity Res. 2004, 5, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Pittenger, G.; Vinik, A. Nerve growth factor and diabetic neuropathy. Exp. Diabesity Res 2003, 4, 271–285. [Google Scholar] [CrossRef] [PubMed]
- Campbell, C.M.; Kipnes, M.S.; Stouch, B.C.; Brady, K.L.; Kelly, M.; Schmidt, W.K.; Petersen, K.L.; Rowbotham, M.C.; Campbell, J.N. Randomized control trial of topical clonidine for treatment of painful diabetic neuropathy. Pain 2012, 153, 1815–1823. [Google Scholar] [CrossRef] [PubMed]
- Havsteen, B.H. The biochemistry and medical significance of the flavonoids. Pharmacol. Ther. 2002, 96, 67–202. [Google Scholar] [CrossRef]
- Testa, R.; Bonfigli, A.R.; Genovese, S.; de Nigris, V.; Ceriello, A. The possible role of flavonoids in the prevention of diabetic complications. Nutrients 2016, 8, 310. [Google Scholar] [CrossRef] [PubMed]
- Lebeau, A.; Esclaire, F.; Rostene, W.; Pelaprat, D. Baicalein protects cortical neurons from beta-amyloid (25–35) induced toxicity. Neuroreport 2001, 12, 2199–2202. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Huang, K.; Xu, H. Protective effects of flavonoids in the roots of scutellaria baicalensis georgi against hydrogen peroxide-induced oxidative stress in Hs-SY5Y cells. Pharmacol. Res. 2001, 43, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Li, F.Q.; Wang, T.; Pei, Z.; Liu, B.; Hong, J.S. Inhibition of microglial activation by the herbal flavonoid baicalein attenuates inflammation-mediated degeneration of dopaminergic neurons. J. Neural Transm. 2005, 112, 331–347. [Google Scholar] [CrossRef] [PubMed]
- Stavniichuk, R.; Drel, V.R.; Shevalye, H.; Maksimchyk, Y.; Kuchmerovska, T.M.; Nadler, J.L.; Obrosova, I.G. Baicalein alleviates diabetic peripheral neuropathy through inhibition of oxidative-nitrosative stress and p38 MAPK activation. Exp. Neurol. 2011, 230, 106–113. [Google Scholar] [CrossRef] [PubMed]
- Izuta, H.; Shimazawa, M.; Tazawa, S.; Araki, Y.; Mishima, S.; Hara, H. Protective effects of Chinese propolis and its component, chrysin, against neuronal cell death via inhibition of mitochondrial apoptosis pathway in SH-SY5Y cells. J. Agric. Food Chem. 2008, 56, 8944–8953. [Google Scholar] [CrossRef] [PubMed]
- Lapidot, T.; Walker, M.D.; Kanner, J. Antioxidant and prooxidant effects of phenolics on pancreatic beta-cells in vitro. J. Agric. Food Chem. 2002, 50, 7220–7225. [Google Scholar] [CrossRef] [PubMed]
- Ha, S.K.; Moon, E.; Kim, S.Y. Chrysin suppresses LPS-stimulated proinflammatory responses by blocking NF-kappaB and JNK activations in microglia cells. Neurosci. Lett. 2010, 485, 143–147. [Google Scholar] [CrossRef] [PubMed]
- Gresa-Arribas, N.; Serratosa, J.; Saura, J.; Sola, C. Inhibition of CCAAT/enhancer binding protein delta expression by chrysin in microglial cells results in anti-inflammatory and neuroprotective effects. J. Neurochem. 2010, 115, 526–536. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Zang, A.; Zhang, L.; Zhang, H.; Zhao, L.; Qi, Z.; Wang, H. Chrysin ameliorates diabetes-associated cognitive deficits in wistar rats. Neurol. Sci. 2014, 35, 1527–1532. [Google Scholar] [CrossRef] [PubMed]
- Dholakiya, S.L.; Benzeroual, K.E. Protective effect of diosmin on LPS-induced apoptosis in PC12 cells and inhibition of TNF-alpha expression. Toxicol. Vitro 2011, 25, 1039–1044. [Google Scholar] [CrossRef] [PubMed]
- Jain, D.; Bansal, M.K.; Dalvi, R.; Upganlawar, A.; Somani, R. Protective effect of diosmin against diabetic neuropathy in experimental rats. J. Integr. Med. 2014, 12, 35–41. [Google Scholar] [CrossRef]
- Srinivasan, S.; Pari, L. Ameliorative effect of diosmin, a citrus flavonoid against streptozotocin-nicotinamide generated oxidative stress induced diabetic rats. Chem. Biol. Interact. 2012, 195, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.T.; Jung, C.H.; Lee, S.R.; Bae, J.H.; Baek, W.K.; Suh, M.H.; Park, J.; Park, C.W.; Suh, S.I. The green tea polyphenol (−)-epigallocatechin gallate attenuates beta-amyloid-induced neurotoxicity in cultured hippocampal neurons. Life Sci. 2001, 70, 603–614. [Google Scholar] [CrossRef]
- Baluchnejadmojarad, T.; Roghani, M. Chronic oral epigallocatechin-gallate alleviates streptozotocin-induced diabetic neuropathic hyperalgesia in rat: Involvement of oxidative stress. Iranian J. Pharm. Res. 2012, 11, 1243–1253. [Google Scholar]
- Raposo, D.; Morgado, C.; Pereira-Terra, P.; Tavares, I. Nociceptive spinal cord neurons of laminae I-III exhibit oxidative stress damage during diabetic neuropathy which is prevented by early antioxidant treatment with epigallocatechin-gallate (EGCG). Brain Res. Bull. 2015, 110, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.M.; Li, S.Q.; Zhu, X.Y.; Wang, Y.; Wu, W.L.; Zhang, X.J. Protective effects of hesperidin against amyloid-beta (Abeta) induced neurotoxicity through the voltage dependent anion channel 1 (VDAC1)-mediated mitochondrial apoptotic pathway in PC12 cells. Neurochem. Res. 2013, 38, 1034–1044. [Google Scholar] [CrossRef] [PubMed]
- Nones, J.; TC, E.S.; Gomes, F.C. Hesperidin, a flavone glycoside, as mediator of neuronal survival. Neurochem. Res. 2011, 36, 1776–1784. [Google Scholar] [CrossRef] [PubMed]
- Visnagri, A.; Kandhare, A.D.; Chakravarty, S.; Ghosh, P.; Bodhankar, S.L. Hesperidin, a flavanoglycone attenuates experimental diabetic neuropathy via modulation of cellular and biochemical marker to improve nerve functions. Pharm. Biol. 2014, 52, 814–828. [Google Scholar] [CrossRef] [PubMed]
- Javed, H.; Vaibhav, K.; Ahmed, M.E.; Khan, A.; Tabassum, R.; Islam, F.; Safhi, M.M. Effect of hesperidin on neurobehavioral, neuroinflammation, oxidative stress and lipid alteration in intracerebroventricular streptozotocin induced cognitive impairment in mice. J. Neurol. Sci. 2015, 348, 51–59. [Google Scholar] [CrossRef] [PubMed]
- El-Marasy, S.A.; Abdallah, H.M.; El-Shenawy, S.M.; El-Khatib, A.S.; El-Shabrawy, O.A.; Kenawy, S.A. Anti-depressant effect of hesperidin in diabetic rats. Can. J. Physiol. Pharm. 2014, 92, 945–952. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.N.; Chi, C.W.; Lin, Y.L.; Chen, C.F.; Shiao, Y.J. The neuroprotective effects of phytoestrogens on amyloid beta protein-induced toxicity are mediated by abrogating the activation of caspase cascade in rat cortical neurons. J. Biol. Chem. 2001, 276, 5287–5295. [Google Scholar] [CrossRef] [PubMed]
- Al-Numair, K.S.; Chandramohan, G.; Veeramani, C.; Alsaif, M.A. Ameliorative effect of kaempferol, a flavonoid, on oxidative stress in streptozotocin-induced diabetic rats. Redox Rep. Commun. Free Radic. Res. 2015, 20, 198–209. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.Y.; Hsieh, M.T.; Tsai, F.S.; Wu, C.R.; Chiu, C.S.; Lee, M.M.; Xu, H.X.; Zhao, Z.Z.; Peng, W.H. Neuroprotective effect of luteolin on amyloid beta protein(25–35)-induced toxicity in cultured rat cortical neurons. Phytother. Res. 2010, 24 (Suppl. 1), S102–S108. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.W.; Wu, M.J.; Liu, I.Y.; Su, J.D.; Yen, J.H. Neurotrophic and cytoprotective action of luteolin in PC12 cells through ERK-dependent induction of NRF2-driven Ho-1 expression. J. Agric. Food Chem. 2010, 58, 4477–4486. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Tian, X.; Gou, L.; Sun, L.; Ling, X.; Yin, X. Luteolin attenuates diabetes-associated cognitive decline in rats. Brain Res. Bull. 2013, 94, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Li, Q.; Zhao, Q.; Zhang, J.; Lin, J. Luteolin improves the impaired nerve functions in diabetic neuropathy: Behavioral and biochemical evidences. Int. J. Exp. Pathol. 2015, 8, 10112–10120. [Google Scholar]
- Shimmyo, Y.; Kihara, T.; Akaike, A.; Niidome, T.; Sugimoto, H. Three distinct neuroprotective functions of myricetin against glutamate-induced neuronal cell death: Involvement of direct inhibition of caspase-3. J. Neurosci. Res. 2008, 86, 1836–1845. [Google Scholar] [CrossRef] [PubMed]
- Vafeiadou, K.; Vauzour, D.; Lee, H.Y.; Rodriguez-Mateos, A.; Williams, R.J.; Spencer, J.P. The citrus flavanone naringenin inhibits inflammatory signalling in glial cells and protects against neuroinflammatory injury. Arch. Biochem. Biophys. 2009, 484, 100–109. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.H.; Lin, C.; Lin, H.Y.; Liu, Y.S.; Wu, C.Y.; Tsai, C.F.; Chang, P.C.; Yeh, W.L.; Lu, D.Y. Naringenin suppresses neuroinflammatory responses through inducing suppressor of cytokine signaling 3 expression. Mol. Neurobiol. 2016, 53, 1080–1091. [Google Scholar] [CrossRef] [PubMed]
- Hasanein, P.; Fazeli, F. Role of naringenin in protection against diabetic hyperalgesia and tactile allodynia in male wistar rats. J. Physiol. Biochem. 2014, 70, 997–1006. [Google Scholar] [CrossRef] [PubMed]
- Al-Rejaie, S.S.; Aleisa, A.M.; Abuohashish, H.M.; Parmar, M.Y.; Ola, M.S.; Al-Hosaini, A.A.; Ahmed, M.M. Naringenin neutralises oxidative stress and nerve growth factor discrepancy in experimental diabetic neuropathy. Neurol. Res. 2015, 37, 924–933. [Google Scholar] [CrossRef] [PubMed]
- Roychowdhury, S.; Wolf, G.; Keilhoff, G.; Bagchi, D.; Horn, T. Protection of primary glial cells by grape seed proanthocyanidin extract against nitrosative/oxidative stress. Nitric Oxide 2001, 5, 137–149. [Google Scholar] [CrossRef] [PubMed]
- Bagchi, D.; Garg, A.; Krohn, R.L.; Bagchi, M.; Bagchi, D.J.; Balmoori, J.; Stohs, S.J. Protective effects of grape seed proanthocyanidins and selected antioxidants against TPA-induced hepatic and brain lipid peroxidation and DNA fragmentation, and peritoneal macrophage activation in mice. Gen. Pharmacol. 1998, 30, 771–776. [Google Scholar] [CrossRef]
- Cui, X.P.; Li, B.Y.; Gao, H.Q.; Wei, N.; Wang, W.L.; Lu, M. Effects of grape seed proanthocyanidin extracts on peripheral nerves in streptozocin-induced diabetic rats. J. Nutr. Sci. Vitaminol. 2008, 54, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Dai, X.; Zhang, Z.; Jiang, Y.; Ma, X.; Cai, X.; Li, Y. Proanthocyanidins protect against early diabetic peripheral neuropathy by modulating endoplasmic reticulum stress. J. Nutr. Biochem. 2014, 25, 765–772. [Google Scholar] [CrossRef] [PubMed]
- He, Q.; Yang, S.Y.; Wang, W.; Wu, Z.J.; Ma, H.L.; Lu, Y. Proanthocyanidins affects the neurotoxicity of Abeta25–35 on C57/bl6 mice. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 679–684. [Google Scholar] [PubMed]
- Jiang, B.; Liu, J.H.; Bao, Y.M.; An, L.J. Hydrogen peroxide-induced apoptosis in pc12 cells and the protective effect of puerarin. Cell Biol. Int. 2003, 27, 1025–1031. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Leng, W.; Zhang, J. Protective effect of puerarin against oxidative stress injury of neural cells and related mechanisms. Med. Sci. Monit. 2016, 22, 1244–1249. [Google Scholar] [CrossRef] [PubMed]
- Xing, G.; Dong, M.; Li, X.; Zou, Y.; Fan, L.; Wang, X.; Cai, D.; Li, C.; Zhou, L.; Liu, J.; et al. Neuroprotective effects of puerarin against beta-amyloid-induced neurotoxicity in PC12 cells via a PI3K-dependent signaling pathway. Brain Res. Bull. 2011, 85, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Hong, B.; Fan, L.; Zhou, L.; Liu, Y.; Wu, Q.; Zhang, X.; Dong, M. Protective effect of puerarin against beta-amyloid-induced oxidative stress in neuronal cultures from rat hippocampus: Involvement of the GSK-3beta/Nrf2 signaling pathway. Free Radic. Res. 2013, 47, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Liao, K.; Yu, C.; Li, X.; Liu, S.; Yang, S. Puerarin alleviates neuropathic pain by inhibiting neuroinflammation in spinal cord. Mediat. Inflamm. 2014, 2014, 485927. [Google Scholar] [CrossRef] [PubMed]
- Xi, J.; Zhang, B.; Luo, F.; Liu, J.; Yang, T. Quercetin protects neuroblastoma SH-SY5Y cells against oxidative stress by inhibiting expression of kruppel-like factor 4. Neurosci. Lett. 2012, 527, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Liang, X.C.; Zhang, H.; Wu, Q.L.; Qu, L.; Sun, Q. Quercetin protects rat dorsal root ganglion neurons against high glucose-induced injury in vitro through NRF-2/HO-1 activation and NF-kB inhibition. Acta Pharmacol. Sin. 2013, 34, 1140–1148. [Google Scholar] [CrossRef] [PubMed]
- Qu, L.; Liang, X.; Gu, B.; Liu, W. Quercetin alleviates high glucose-induced schwann cell damage by autophagy. Neural Regen. Res. 2014, 9, 1195–1203. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Huang, C.Y.; Tsai, F.J.; Tsai, C.C.; Yao, C.H.; Chen, Y.S. Growth-promoting effects of quercetin on peripheral nerves in rats. Int. J. Artif. Organs 2011, 34, 1095–1105. [Google Scholar] [CrossRef] [PubMed]
- Xia, S.F.; Xie, Z.X.; Qiao, Y.; Li, L.R.; Cheng, X.R.; Tang, X.; Shi, Y.H.; Le, G.W. Differential effects of quercetin on hippocampus-dependent learning and memory in mice fed with different diets related with oxidative stress. Physiol. Behav. 2015, 138, 325–331. [Google Scholar] [CrossRef] [PubMed]
- Kamalakkannan, N.; Prince, P.S. Antihyperglycaemic and antioxidant effect of rutin, a polyphenolic flavonoid, in streptozotocin-induced diabetic wistar rats. Basic Clin. Pharmacol. Toxicol. 2006, 98, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Tian, R.; Yang, W.; Xue, Q.; Gao, L.; Huo, J.; Ren, D.; Chen, X. Rutin ameliorates diabetic neuropathy by lowering plasma glucose and decreasing oxidative stress via nrf2 signaling pathway in rats. Eur. J. Pharmacol. 2016, 771, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Yin, F.; Liu, J.; Ji, X.; Wang, Y.; Zidichouski, J.; Zhang, J. Silibinin: A novel inhibitor of abeta aggregation. Neurochem. Int. 2011, 58, 399–403. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Li, Y.J.; Ding, Y.; Zhang, H.N.; Sun, T.; Zhang, K.; Yang, L.; Guo, Y.Y.; Liu, S.B.; Zhao, M.G.; et al. Silibinin prevents autophagic cell death upon oxidative stress in cortical neurons and cerebral ischemia-reperfusion injury. Mol. Neurobiol. 2016, 53, 932–943. [Google Scholar] [CrossRef] [PubMed]
- Marrazzo, G.; Bosco, P.; La Delia, F.; Scapagnini, G.; Di Giacomo, C.; Malaguarnera, M.; Galvano, F.; Nicolosi, A.; Li Volti, G. Neuroprotective effect of silibinin in diabetic mice. Neurosc. Lett. 2011, 504, 252–256. [Google Scholar] [CrossRef] [PubMed]
- Guleria, R.S.; Pan, J.; Dipette, D.; Singh, U.S. Hyperglycemia inhibits retinoic acid-induced activation of rac1, prevents differentiation of cortical neurons, and causes oxidative stress in a rat model of diabetic pregnancy. Diabetes 2006, 55, 3326–3334. [Google Scholar] [CrossRef] [PubMed]
- De Bittencourt Pasquali, M.A.; de Ramos, V.M.; Albanus, R.D.; Kunzler, A.; de Souza, L.H.; Dalmolin, R.J.; Gelain, D.P.; Ribeiro, L.; Carro, L.; Moreira, J.C. Gene expression profile of NF-kappaB, NRF2, glycolytic, and p53 pathways during the SH-SY5Y neuronal differentiation mediated by retinoic acid. Mol. Neurobiol. 2016, 53, 423–435. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; May, J.M. Ascorbic acid protects SH-SY5Y neuroblastoma cells from apoptosis and death induced by beta-amyloid. Brain Res. 2006, 1097, 52–58. [Google Scholar] [CrossRef] [PubMed]
- May, J.M.; Jayagopal, A.; Qu, Z.C.; Parker, W.H. Ascorbic acid prevents high glucose-induced apoptosis in human brain pericytes. Biochem. Biophys. Res. Commun. 2014, 452, 112–117. [Google Scholar] [CrossRef] [PubMed]
- Saporito, M.S.; Brown, E.R.; Hartpence, K.C.; Wilcox, H.M.; Vaught, J.L.; Carswell, S. Chronic 1,25-dihydroxyvitamin D3-mediated induction of nerve growth factor mRNA and protein in l929 fibroblasts and in adult rat brain. Brain Res. 1994, 633, 189–196. [Google Scholar] [CrossRef]
- Neveu, I.; Naveilhan, P.; Baudet, C.; Brachet, P.; Metsis, M. 1,25-dihydroxyvitamin D3 regulates NT-3, NT-4 but not bdnf mrna in astrocytes. Neuroreport 1994, 6, 124–126. [Google Scholar] [CrossRef] [PubMed]
- Naveilhan, P.; Neveu, I.; Wion, D.; Brachet, P. 1,25-dihydroxyvitamin D3, an inducer of glial cell line-derived neurotrophic factor. Neuroreport 1996, 7, 2171–2175. [Google Scholar] [CrossRef] [PubMed]
- Riaz, S.; Malcangio, M.; Miller, M.; Tomlinson, D.R. A vitamin D(3) derivative (CB1093) induces nerve growth factor and prevents neurotrophic deficits in streptozotocin-diabetic rats. Diabetologia 1999, 42, 1308–1313. [Google Scholar] [CrossRef] [PubMed]
- Behl, C. Vitamin E protects neurons against oxidative cell death in vitro more effectively than 17-beta estradiol and induces the activity of the transcription factor NF-kappaB. J. Neural. Transm. 2000, 107, 393–407. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.; Conrad, C.C.; Dai, R.; Malakowsky, C.A.; Talent, J.M.; Carroll, C.A.; Weintraub, S.T.; Gracy, R.W. Vitamin E prevents oxidation of antiapoptotic proteins in neuronal cells. Proteomics 2003, 3, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Chang, T.I.; Horal, M.; Jain, S.K.; Wang, F.; Patel, R.; Loeken, M.R. Oxidant regulation of gene expression and neural tube development: Insights gained from diabetic pregnancy on molecular causes of neural tube defects. Diabetologia 2003, 46, 538–545. [Google Scholar] [PubMed]
- Baydas, G.; Nedzvetskii, V.S.; Tuzcu, M.; Yasar, A.; Kirichenko, S.V. Increase of glial fibrillary acidic protein and S-100B in hippocampus and cortex of diabetic rats: Effects of vitamin E. Eur. J. Pharmacol. 2003, 462, 67–71. [Google Scholar] [CrossRef]
- Huang, Y.; Tsang, S.Y.; Yao, X.; Chen, Z.Y. Biological properties of baicalein in cardiovascular system. Curr. Drug Targets Cardiovasc. Haematol. Disord. 2005, 5, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.; Yun, C.W.; Park, W.K.; Kong, J.Y.; Kim, K.S.; Park, Y.; Lee, S.; Kim, B.K. Modulation of the activity of pro-inflammatory enzymes, COX-2 and inos, by chrysin derivatives. Pharmacol. Res. 2004, 49, 37–43. [Google Scholar] [CrossRef]
- Kang, S.S.; Lee, J.Y.; Choi, Y.K.; Kim, G.S.; Han, B.H. Neuroprotective effects of flavones on hydrogen peroxide-induced apoptosis in SH-SY5Y neuroblostoma cells. Bioorg. Med. Chem. Lett. 2004, 14, 2261–2264. [Google Scholar] [CrossRef] [PubMed]
- Akaishi, T.; Morimoto, T.; Shibao, M.; Watanabe, S.; Sakai-Kato, K.; Utsunomiya-Tate, N.; Abe, K. Structural requirements for the flavonoid fisetin in inhibiting fibril formation of amyloid beta protein. Neurosci. Lett. 2008, 444, 280–285. [Google Scholar] [CrossRef] [PubMed]
- Benavente-Garcia, O.; Castillo, J. Update on uses and properties of citrus flavonoids: New findings in anticancer, cardiovascular, and anti-inflammatory activity. J. Agric. Food Chem. 2008, 56, 6185–6205. [Google Scholar] [CrossRef] [PubMed]
- Manuel y Keenoy, B.; Vertommen, J.; de Leeuw, I. The effect of flavonoid treatment on the glycation and antioxidant status in type 1 diabetic patients. Diabetes Nutr. Metab. 1999, 12, 256–263. [Google Scholar] [PubMed]
- Kang, K.S.; Wen, Y.; Yamabe, N.; Fukui, M.; Bishop, S.C.; Zhu, B.T. Dual beneficial effects of (−)-epigallocatechin-3-gallate on levodopa methylation and hippocampal neurodegeneration: In vitro and in vivo studies. PLoS ONE 2010, 5, e11951. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Zhang, J.; Liang, M.; Zhang, W.; Yan, S.; Lin, M. Simultaneous analysis of eight bioactive compounds in danning tablet by HPLC-ESI-MS and HPLC-UV. J. Pharm. Biomed. Anal. 2007, 43, 1007–1012. [Google Scholar] [CrossRef] [PubMed]
- Galati, E.M.; Monforte, M.T.; Kirjavainen, S.; Forestieri, A.M.; Trovato, A.; Tripodo, M.M. Biological effects of hesperidin, a citrus flavonoid. (note I): Antiinflammatory and analgesic activity. Farmaco 1994, 40, 709–712. [Google Scholar] [PubMed]
- Monforte, M.T.; Trovato, A.; Kirjavainen, S.; Forestieri, A.M.; Galati, E.M.; Lo Curto, R.B. Biological effects of hesperidin, a citrus flavonoid. (note II): Hypolipidemic activity on experimental hypercholesterolemia in rat. Farmaco 1995, 50, 595–599. [Google Scholar] [PubMed]
- Kim, J.K.; Choi, S.J.; Cho, H.Y.; Hwang, H.J.; Kim, Y.J.; Lim, S.T.; Kim, C.J.; Kim, H.K.; Peterson, S.; Shin, D.H. Protective effects of kaempferol (3,4′,5,7-tetrahydroxyflavone) against amyloid beta peptide (Abeta)-induced neurotoxicity in ICR mice. Biosci. Biotechnol. Biochem. 2010, 74, 397–401. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Gao, H.Y.; Baba, M.; Okada, Y.; Okuyama, T.; Wu, L.J.; Zhan, L.B. Extracts and compounds with anti-diabetic complications and anti-cancer activity from castanea mollissina blume (Chinese chestnut). BMC Complement. Altern. Med. 2014, 14, 422. [Google Scholar] [CrossRef] [PubMed]
- Arts, I.C.; Hollman, P.C. Polyphenols and disease risk in epidemiologic studies. Am. J. Clin. Nutr. 2005, 81, 317S–325S. [Google Scholar] [PubMed]
- Nazari, Q.A.; Kume, T.; Takada-Takatori, Y.; Izumi, Y.; Akaike, A. Protective effect of luteolin on an oxidative-stress model induced by microinjection of sodium nitroprusside in mice. J. Pharm. Sci. 2013, 122, 109–117. [Google Scholar] [CrossRef]
- Xu, Y.; Zhang, J.; Liu, J.; Li, S.; Li, C.; Wang, W.; Ma, R.; Liu, Y. Luteolin attenuate the d-galactose-induced renal damage by attenuation of oxidative stress and inflammation. Nat. Prod. Res. 2015, 29, 1078–1082. [Google Scholar] [CrossRef] [PubMed]
- Harnly, J.M.; Doherty, R.F.; Beecher, G.R.; Holden, J.M.; Haytowitz, D.B.; Bhagwat, S.; Gebhardt, S. Flavonoid content of U.S. Fruits, vegetables, and nuts. J. Agric. Food Chem. 2006, 54, 9966–9977. [Google Scholar] [CrossRef] [PubMed]
- Mira, L.; Fernandez, M.T.; Santos, M.; Rocha, R.; Florencio, M.H.; Jennings, K.R. Interactions of flavonoids with iron and copper ions: A mechanism for their antioxidant activity. Free Radic. Res. 2002, 36, 1199–1208. [Google Scholar] [CrossRef] [PubMed]
- Gray, A.M.; Flatt, P.R. Nature’s own pharmacy: The diabetes perspective. Proc. Nutr. Soc. 1997, 56, 507–517. [Google Scholar] [CrossRef] [PubMed]
- Kawanishi, K.; Ueda, H.; Moriyasu, M. Aldose reductase inhibitors from the nature. Curr. Med. Chem. 2003, 10, 1353–1374. [Google Scholar] [CrossRef] [PubMed]
- Urios, P.; Grigorova-Borsos, A.M.; Sternberg, M. Flavonoids inhibit the formation of the cross-linking AGE pentosidine in collagen incubated with glucose, according to their structure. Eur. J. Nutr. 2007, 46, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.N.; Kang, M.J.; Lee, S.J.; Kim, J.I. Ameliorative effect of myricetin on insulin resistance in mice fed a high-fat, high-sucrose diet. Nutr. Res. Pract. 2014, 8, 544–549. [Google Scholar] [CrossRef] [PubMed]
- Liu, I.M.; Liou, S.S.; Lan, T.W.; Hsu, F.L.; Cheng, J.T. Myricetin as the active principle of abelmoschus moschatus to lower plasma glucose in streptozotocin-induced diabetic rats. Planta Med. 2005, 71, 617–621. [Google Scholar] [CrossRef] [PubMed]
- Tjolsen, A.; Berge, O.G.; Hunskaar, S.; Rosland, J.H.; Hole, K. The formalin test: An evaluation of the method. Pain 1992, 51, 5–17. [Google Scholar] [CrossRef]
- Freshwater, J.D.; Svensson, C.I.; Malmberg, A.B.; Calcutt, N.A. Elevated spinal cyclooxygenase and prostaglandin release during hyperalgesia in diabetic rats. Diabetes 2002, 51, 2249–2255. [Google Scholar] [CrossRef] [PubMed]
- O′Connor, A.B.; Dworkin, R.H. Treatment of neuropathic pain: An overview of recent guidelines. Am. J. Med. 2009, 122, S22–S32. [Google Scholar] [CrossRef] [PubMed]
- Rahigude, A.; Bhutada, P.; Kaulaskar, S.; Aswar, M.; Otari, K. Participation of antioxidant and cholinergic system in protective effect of naringenin against type-2 diabetes-induced memory dysfunction in rats. Neuroscience 2012, 226, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Cos, P.; de Bruyne, T.; Hermans, N.; Apers, S.; Berghe, D.V.; Vlietinck, A.J. Proanthocyanidins in health care: Current and new trends. Curr. Med. Chem. 2004, 11, 1345–1359. [Google Scholar] [CrossRef] [PubMed]
- Natella, F.; Belelli, F.; Gentili, V.; Ursini, F.; Scaccini, C. Grape seed proanthocyanidins prevent plasma postprandial oxidative stress in humans. J. Agric. Food Chem. 2002, 50, 7720–7725. [Google Scholar] [CrossRef] [PubMed]
- Prasain, J.K.; Jones, K.; Brissie, N.; Moore, R.; Wyss, J.M.; Barnes, S. Identification of puerarin and its metabolites in rats by liquid chromatography-tandem mass spectrometry. J. Agric. Food Chem. 2004, 52, 3708–3712. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.Y.; Liu, Y.H.; Wang, H.Q.; Xu, J.H.; Hu, H.T. Puerarin protects PC12 cells against beta-amyloid-induced cell injury. Cell. Biol. Int. 2008, 32, 1230–1237. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Zhang, X.; Zhang, B. Efficacy and safety of puerarin injection in treatment of diabetic peripheral neuropathy: A systematic review and meta-analysis of randomized controlled trials. J. Tradit. Chin. Med. 2014, 34, 401–410. [Google Scholar] [CrossRef]
- Nijveldt, R.J.; van Nood, E.; van Hoorn, D.E.; Boelens, P.G.; van Norren, K.; van Leeuwen, P.A. Flavonoids: A review of probable mechanisms of action and potential applications. Am. J. Clin. Nutr. 2001, 74, 418–425. [Google Scholar] [PubMed]
- Formica, J.V.; Regelson, W. Review of the biology of quercetin and related bioflavonoids. Food Chem. Toxicol. 1995, 33, 1061–1080. [Google Scholar] [CrossRef]
- Sasaki, M.; Nakamura, H.; Tsuchiya, S.; Horie, S.; Kashiwayanagi, M.; Saito, T.; Murayama, T. Quercetin-induced PC12 cell death accompanied by caspase-mediated DNA fragmentation. Biol. Pharm. Bull. 2007, 30, 682–686. [Google Scholar] [CrossRef] [PubMed]
- Anjaneyulu, M.; Chopra, K. Quercetin, a bioflavonoid, attenuates thermal hyperalgesia in a mouse model of diabetic neuropathic pain. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2003, 27, 1001–1005. [Google Scholar] [CrossRef]
- Ferreira, P.E.; Lopes, C.R.; Alves, A.M.; Alves, E.P.; Linden, D.R.; Zanoni, J.N.; Buttow, N.C. Diabetic neuropathy: An evaluation of the use of quercetin in the cecum of rats. World J. Gastroenterol. 2013, 19, 6416–6426. [Google Scholar] [CrossRef] [PubMed]
- Valensi, P.; Le Devehat, C.; Richard, J.L.; Farez, C.; Khodabandehlou, T.; Rosenbloom, R.A.; LeFante, C. A multicenter, double-blind, safety study of QR-333 for the treatment of symptomatic diabetic peripheral neuropathy. A preliminary report. J. Diabetes Complicat. 2005, 19, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Bravo, L. Polyphenols: Chemistry, dietary sources, metabolism, and nutritional significance. Nutr. Rev. 1998, 56, 317–333. [Google Scholar] [CrossRef] [PubMed]
- Hosseinzadeh, H.; Nassiri-Asl, M. Review of the protective effects of rutin on the metabolic function as an important dietary flavonoid. J. Endocrinol. Invest. 2014, 37, 783–788. [Google Scholar] [CrossRef] [PubMed]
- Na, J.Y.; Kim, S.; Song, K.; Kwon, J. Rutin alleviates prion peptide-induced cell death through inhibiting apoptotic pathway activation in dopaminergic neuronal cells. Cell. Mol. Neurobiol. 2014, 34, 1071–1079. [Google Scholar] [CrossRef] [PubMed]
- Song, K.; Kim, S.; Na, J.Y.; Park, J.H.; Kim, J.K.; Kim, J.H.; Kwon, J. Rutin attenuates ethanol-induced neurotoxicity in hippocampal neuronal cells by increasing aldehyde dehydrogenase 2. Food Chem. Toxicol. 2014, 72, 228–233. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.S.; Han, D.; Packer, L. Antioxidants and herbal extracts protect HT-4 neuronal cells against glutamate-induced cytotoxicity. Free Radic. Res. 2000, 32, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Tongjaroenbuangam, W.; Ruksee, N.; Chantiratikul, P.; Pakdeenarong, N.; Kongbuntad, W.; Govitrapong, P. Neuroprotective effects of quercetin, rutin and okra (Abelmoschus esculentus Linn.) in dexamethasone-treated mice. Neurochem. Int. 2011, 59, 677–685. [Google Scholar] [CrossRef] [PubMed]
- Alenazi, M.M.; Alkhamees, O.A.; Alroujayee, A.S.; Ahmed, M.M. Protective effects of combined therapy of rutin with silymarin on experimentally induced diabetic neuropathy in rats. Diabetic Med. 2014, 31, 57. [Google Scholar]
- Jangra, A.; Kasbe, P.; Pandey, S.N.; Dwivedi, S.; Gurjar, S.S.; Kwatra, M.; Mishra, M.; Venu, A.K.; Sulakhiya, K.; Gogoi, R.; et al. Hesperidin and silibinin ameliorate aluminum-induced neurotoxicity: Modulation of antioxidants and inflammatory cytokines level in mice hippocampus. Biol. Trace Elem. Res. 2015, 168, 462–471. [Google Scholar] [CrossRef] [PubMed]
- Geed, M.; Garabadu, D.; Ahmad, A.; Krishnamurthy, S. Silibinin pretreatment attenuates biochemical and behavioral changes induced by intrastriatal MPP+ injection in rats. Pharmacol. Biochem. Behav. 2014, 117, 92–103. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Park, H.R.; Chun, H.J.; Lee, J. Silibinin prevents dopaminergic neuronal loss in a mouse model of parkinson’s disease via mitochondrial stabilization. J. Neurosci. Res. 2015, 93, 755–765. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Pedro, N.; Ordonez, G.; Ortiz-Plata, A.; Palencia-Hernandez, G.; Garcia-Ulloa, A.C.; Flores-Estrada, D.; Sotelo, J.; Arrieta, O. All-trans retinoic acid induces nerve regeneration and increases serum and nerve contents of neural growth factor in experimental diabetic neuropathy. Transl. Res. 2008, 152, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Arrieta, O.; Garcia-Navarrete, R.; Zuniga, S.; Ordonez, G.; Ortiz, A.; Palencia, G.; Morales-Espinosa, D.; Hernandez-Pedro, N.; Sotelo, J. Retinoic acid increases tissue and plasma contents of nerve growth factor and prevents neuropathy in diabetic mice. Eur. J. Clin. Invest. 2005, 35, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Rice, M.E. Ascorbate regulation and its neuroprotective role in the brain. Trends Neurosci. 2000, 23, 209–216. [Google Scholar] [CrossRef]
- May, J.M. Vitamin C transport and its role in the central nervous system. Subcell. Biochem. 2012, 56, 85–103. [Google Scholar] [PubMed]
- Harrison, F.E.; May, J.M. Vitamin C function in the brain: Vital role of the ascorbate transporter SVCT2. Free Radic. Biol. Med. 2009, 46, 719–730. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.N.; Yang, L.Y.; Wang, J.Y.; Lai, C.C.; Chiu, C.T. l-Ascorbate protects against methamphetamine-induced neurotoxicity of cortical cells via inhibiting oxidative stress, autophagy, and apoptosis. Mol. Neurobiol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimzadeh-Bideskan, A.R.; Hami, J.; Alipour, F.; Haghir, H.; Fazel, A.R.; Sadeghi, A. Protective effects of ascorbic acid and garlic extract against lead-induced apoptosis in developing rat hippocampus. Metab. Brain Dis. 2016. [Google Scholar] [CrossRef] [PubMed]
- Soderstrom, L.H.; Johnson, S.P.; Diaz, V.A.; Mainous, A.G. Association between vitamin D and diabetic neuropathy in a nationally representative sample: Results from 2001 to 2004 nhanes. Diabet. Med. 2012, 29, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.; Chen, R. Vitamin D as an analgesic for patients with type 2 diabetes and neuropathic pain. Arch. Int. Med. 2008, 168, 771–772. [Google Scholar] [CrossRef] [PubMed]
- Burton, G.W.; Traber, M.G. Vitamin E: Antioxidant activity, biokinetics, and bioavailability. Annu. Rev. Nutr. 1990, 10, 357–382. [Google Scholar] [CrossRef] [PubMed]
- Skalska, S.; Kyselova, Z.; Gajdosikova, A.; Karasu, C.; Stefek, M.; Stolc, S. Protective effect of stobadine on ncv in streptozotocin-diabetic rats: Augmentation by vitamin E. Gen. Physiol. Biophys. 2008, 27, 106–114. [Google Scholar] [PubMed]
- Van Dam, P.S.; Bravenboer, B.; van Asbeck, B.S.; Marx, J.J.; Gispen, W.H. High rat food vitamin E content improves nerve function in streptozotocin-diabetic rats. Eur. J. Pharmacol. 1999, 376, 217–222. [Google Scholar] [CrossRef]
- Ogbera, A.O.; Ezeobi, E.; Unachukwu, C.; Oshinaike, O. Treatment of diabetes mellitus-associated neuropathy with vitamin E and eve primrose. Indian J. Endocrinol. Metab. 2014, 18, 846–849. [Google Scholar] [CrossRef] [PubMed]
| Bioactive Compounds | Models | Effects | Specific Mechanisms of Action | Reference |
|---|---|---|---|---|
| Flavonoids (Subclass) | ||||
| Baicalein (flavones) | Primary rat cortical neurons | ↓Aβ-induced cell death | ↓12-lipoxygenase | [26] |
| SH-SY5Y cells | ↓H2O2-induced cell death | ↓oxidative stress | [27] | |
| Primary dopaminergic neurons | ↓LPS-induced cell injury | ↓NO, free radicals | [28] | |
| STZ-induced diabetic mice | ↑nerve conductive velocity | ↓oxidative-nitrosative stress and p38 MAPK | [29] | |
| Chrysin (flavones) | SH-SY5Y cells | ↓ER stress cell death | ↑mitochondrial membrane potential | [30] |
| Primary microglia/microglia cell line | ↓LPS-induced NO, TNF-α and IL-1β | ↓JNK, NF-κB and CEBPβ | [31,32,33] | |
| STZ-induced diabetic rats | ↑learning and memory function | ↑CAT, SOD, GSH/ ↓MDA | [34] | |
| Diosmin (flavones) | PC12 cells | ↓LPS-induced apoptosis | ↓TNF-α | [35] |
| High-fat diet-/STZ-induced diabetic mice | ↓glucose level and body weight ↑nerve function | ↓oxidative stress enzyme activity | [36] | |
| STZ-/nicotinamide-induced diabetic mice | ↓glucose level | ↑antioxidants (vitamin c, vitamin E) and GSH | [37] | |
| EGCG (flavanol) | Hippocampal neuronal cells | ↓Aβ-induced injury | ↑MDA and caspase activity | [38] |
| STZ-induced diabetic rats | ↓hyperalgesia | ↓TBARS and NO ↑SOD | [39] | |
| STZ-induced diabetic rats | ↓hypersensitivity | ↓oxidative stress damage | [40] | |
| Hesperidin (flavanones) | PC12 cells | ↓Aβ-induced apoptosis | ↑GSK3β-mediated VDAC | [41] |
| Cortical progenitors | ↓cell death | ↑PI3K and MAPK | [42] | |
| STZ-induced diabetic rats | ↓hyperglycemia and hyperlipidemia ↑nerve function | ↓free radical generation and proinflammatory cytokines | [43] | |
| STZ-induced diabetic mice | ↑nerve function | ↑AchE and GSH ↓TBARS, NF-κB, iNOS and COX-2 | [44,45] | |
| Kaempferol (flavonols) | HT22 cells | ↓H2O2-induced apoptosis | ↓ROS production | [46] |
| STZ-induced diabetic mice | ↓glucose level | ↓lipid peroxidation | [47] | |
| Luteolin (flavones) | Primary cortical neurons | ↓Aβ-induced cell death | ↓ERK, JNK, p38 MAPK | [48] |
| SH-SY5Y cells | ↑neurite outgrowth | ↑ERK-dependent Nrf2 pathway | [49] | |
| STZ-induced diabetic rats | ↓neuronal injury ↑cognitive performance | ↓oxidative stress and ChE activity | [50] | |
| STZ-induced diabetic rats | ↑nerve conduction | ↑Nrf2 and HO-1 | [51] | |
| Myricetin (flavonols) | Rat cortical neurons | ↓Aβ-induced cell injury | ↓AGE | [52] |
| Naringenin (flavones) | Primary microglial cells | ↓LPS-induced cytokine release | ↓p38 MAPK, STAT-1 ↑SOCS3 | [53,54] |
| STZ-induced diabetic rats | ↓glucose level ↑NGF, IGF | ↑SOD, CAT, GPx | [55,56] | |
| Proanthocyanidin (flavanols) | Mouse primary microglia cells and PC12 | ↓H2O2-induced cell death | ↓lactate dehydrogenase | [57,58] |
| STZ-induced diabetic rats | ↓glucose level ↑nerve conductive velocity | ↑SOD, ↓AGE and MDA | [59] | |
| STZ-/high carbohydrate-/high-fat diet-induced diabetic rats | ↓LDL ↑nerve conductive velocity | ↓ER stress protein | [60] | |
| Aβ-induced diabetic mice | ↓neuronal apoptosis ↑synaptic density | ↑antioxidant level | [61] | |
| Puerarin (isoflavones) | PC12 cells | ↓H2O2-induced cell death | ↑caspase-3, caspase-9 ↑SOD, GSHAKT/PI3K | [62,63] |
| PC12 cells | ↓Aβ-induced cell death | ↑AKT/PI3K | [64] | |
| Primary rat hippocampal neurons | ↓Aβ-induced oxidative stress | ↑Nrf-2/HO-1 | [65] | |
| STZ-induced diabetic rats | ↓pain sensitivity | ↓inflammatory cytokines | [66] | |
| Quercetin (flavonols) | SH-SY5Y cells | ↓H2O2-induced cell death | ↓KLF4 | [67] |
| Dorsal root ganglion cells, primary Schwann cells and RSC96 cells | ↓high-glucose injury | ↑Nrf-2/HO-1, ↓NF-kB | [68,69] | |
| Schwann cells | ↑growth ↓high glucose-induced damage | ↑autophagy | [69,70] | |
| High-fat diet-induced diabetic mice | ↑cognitive function | ↓oxidative stress enzyme activity | [71] | |
| Rutin (flavonols) | STZ-induced diabetic rats | ↓glucose level | ↓TBARS and lipid hydroperoxides | [72] |
| STZ-induced diabetic rats | ↓glucose level ↑nerve function | ↑Nrf-2 | [73] | |
| Silibinin | SH-SY5Y cells | ↓Aβ induced cytotoxicity | ↓oxidative stress | [74] |
| Mouse cortical neurons | ↓H2O2-induced cell death | ↓beclin-1, LC3-II expression | [75] | |
| db/db mice | ↓oxidative stress ↑DNA protection | ↑HO-1 | [76] | |
| Vitamins | ||||
| Vitamin A | Rat embryonic cortical neurons | ↑neurite outgrowth | ↑RAC1 | [77] |
| SH-SY5Y cells | ↑neuronal differentiation | ↑glycolytic pathway and antioxidant pathway | [78] | |
| Vitamin C | SH-SY5Y cells | ↓Aβ induced cytotoxicity | ↓oxidative stress | [79] |
| Human brain pericytes | ↓high glucose induced apoptosis | ↓advanced glycation end production | [80] | |
| Vitamin D | Primary astrocytes/C6 glioma cells | ↑NGF, GDNF and neurotrophin | - | [81,82,83] |
| STZ-induced diabetic rats | no changes in glucose levels | ↑NGF level | [84] | |
| Vitamin E | HT22 cells/rat cerebellar granule neurons | ↓Aβ- and H2O2-induced cell death | ↑NF-κB activity ↓HSP60 and vimentin | [85,86] |
| Ex vivo embryo tissues | ↓high glucose-induced neuronal tube defect | ↑Pax-3 expression | [87] | |
| STZ-induced diabetic rats | ↓reactive astrocytosis | ↓lipid peroxidation | [88] | |
© 2016 by the author; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oh, Y.S. Bioactive Compounds and Their Neuroprotective Effects in Diabetic Complications. Nutrients 2016, 8, 472. https://doi.org/10.3390/nu8080472
Oh YS. Bioactive Compounds and Their Neuroprotective Effects in Diabetic Complications. Nutrients. 2016; 8(8):472. https://doi.org/10.3390/nu8080472
Chicago/Turabian StyleOh, Yoon Sin. 2016. "Bioactive Compounds and Their Neuroprotective Effects in Diabetic Complications" Nutrients 8, no. 8: 472. https://doi.org/10.3390/nu8080472
APA StyleOh, Y. S. (2016). Bioactive Compounds and Their Neuroprotective Effects in Diabetic Complications. Nutrients, 8(8), 472. https://doi.org/10.3390/nu8080472





