Suppressive Effects of Tea Catechins on Breast Cancer
Abstract
:1. Introduction
2. Epidemiological Evidence
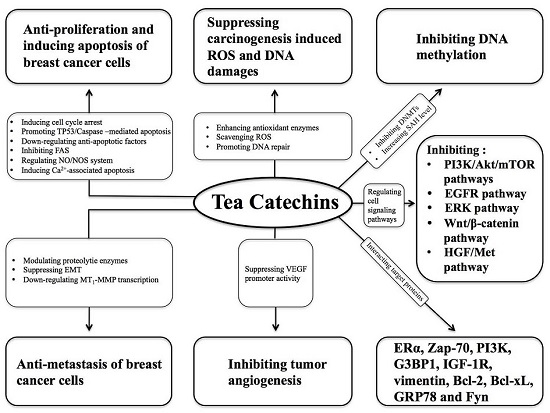
3. Mechanism of Tea Catechins in Suppressing Breast Cancer
3.1. Suppressing Carcinogen-Induced ROS Elevation and DNA Damage
3.2. Regulating Cell Signaling Pathways
3.3. Interacting with Target Proteins
3.4. Inhibiting DNA Methylation
3.5. Inhibiting Tumor Angiogenesis
3.6. Anti-Proliferation and Inducing Breast Cancer Cell Apoptosis
3.7. Anti-Metastasis of Breast Cancer Cells
4. Inconsistent Results and Further Study Suggestions
4.1. Inconsistent Results
4.2. Further Study Suggestions
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Stewart, B.W.; Wild, C.P. World Cancer Report 2014; World Health Organization: French, 2014; Chapters 1.1 and 5.2; Available online: http://www.searo.who.int/publications/bookstore/documents/9283204298/en/ (accessed on 20 May 2016).
- Kushi, L.H.; Doyle, C.; McCullough, M.; Rock, C.L.; Demark-Wahnefried, W.; Bandera, E.V.; Gapstur, S.; Patel, A.V.; Andrews, K.; Gansler, T. American cancer society guidelines on nutrition and physical activity for cancer prevention: Reducing the risk of cancer with healthy food choices and physical activity. CA Cancer J. Clin. 2012, 62, 30–67. [Google Scholar] [CrossRef] [PubMed]
- Thomson, C.A. Diet and breast cancer: Understanding risks and benefits. Nutr. Clin. Pract. 2012, 27, 636–650. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.R.; Ye, Q.; Jin, J.; Liang, H.; Lu, J.L.; Du, Y.Y.; Dong, J.J. Chemical and instrumental assessment of green tea sensory preference. Int. Food Prop. 2008, 11, 258–272. [Google Scholar] [CrossRef]
- Dong, J.J.; Ye, J.H.; Lu, J.L.; Zheng, X.Q.; Liang, Y.R. Isolation of antioxidant catechins from green tea and its decaffeination. Food Bioprod. Process. 2011, 89, 62–66. [Google Scholar] [CrossRef]
- Lin, S.Y.; Chen, Y.L.; Lee, C.L.; Cheng, C.Y.; Roan, S.F.; Chen, I.Z. Monitoring volatile compound profiles and chemical compositions during the process of manufacturing semi-fermented oolong tea. J. Hortic. Sci. Biotechnol. 2013, 88, 159–164. [Google Scholar] [CrossRef]
- Liang, Y.R.; Lu, J.L.; Zhang, L.Y.; Wu, S.; Wu, Y. Estimation of black tea quality by analysis of chemical composition and colour difference of tea infusions. Food Chem. 2003, 80, 283–290. [Google Scholar] [CrossRef]
- Xu, J.Y.; Wu, L.Y.; Zheng, X.Q.; Lu, J.L.; Wu, M.Y.; Liang, Y.R. Green tea polyphenols attenuating ultraviolet b-induced damage to human retinal pigment epithelial cells in vitro. Invest. Ophthalmol. Vis. Sci. 2010, 51, 6665–6670. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Lu, W.; Zheng, Y.; Gu, K.; Chen, Z.; Zheng, W.; Shu, X.O. Exercise, tea consumption, and depression among breast cancer survivors. J. Clin. Oncol. 2010, 28, 991–998. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Tse, L.A.; Chan, W.C.; Kwok, C.H.; Leung, S.L.; Wu, C.; Yu, W.C.; Yu, I.T.S.; Yu, C.H.T.; Wang, F.; et al. Evaluation of breast cancer risk associated with tea consumption by menopausal and estrogen receptor status among Chinese women in Hong Kong. Cancer Epidemiol. 2016, 40, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.; Tsubono, Y.; Nakaya, N.; Suzuki, Y.; Koizumi, Y.; Tsuji, I. Green tea and the risk of breast cancer: Pooled analysis of two prospective studies in Japan. Br. J. Cancer 2004, 90, 1361–1363. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Holman, C.D.A.J.; Huang, J.P.; Xie, X. Green tea and the prevention of breast cancer: A case-control study in Southeast China. Carcinogenesis 2007, 28, 1074–1078. [Google Scholar] [CrossRef] [PubMed]
- Imai, K.; Suga, K.; Nakachi, K. Cancer-Preventive effects of drinking green tea among a Japanese population. Prev. Med. 1997, 26, 769–775. [Google Scholar] [CrossRef] [PubMed]
- Shrubsole, M.J.; Lu, W.; Chen, Z.; Shu, X.O.; Zheng, Y.; Dai, Q.; Cai, Q.; Gu, K.; Ruan, Z.X.; Gao, Y.T.; et al. Drinking Green Tea Modestly Reduces Breast Cancer Risk. J. Nutr. 2009, 139, 310–316. [Google Scholar] [CrossRef] [PubMed]
- Ganmaa, D.; Willett, W.C.; Li, T.Y.; Feskanich, D.; Dam, R.M.V.; Lopez-Garcia, E.; Hunter, D.J.; Holmes, M.D. Coffee, tea, caffeine and risk of breast cancer: A 22-year follow-up. Int. J. Cancer 2008, 122, 2071–2076. [Google Scholar] [CrossRef] [PubMed]
- Dai, Q.; Shu, X.O.; Li, H.L.; Yang, G.; Shrubsole, M.J.; Cai, H.; Wen, W.Q.; Franke, A.; Gao, Y.T.; Zheng, W. Is green tea drinking associated with a later onset of breast cancer? Ann. Epidemiol. 2010, 20, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Huang, J.; Xie, X.; Holman, C.D.A.J. Dietary intakes of mushrooms and green tea combine to reduce the risk of breast cancer in Chinese women. Int. J. Cancer 2009, 124, 1404–1408. [Google Scholar] [CrossRef] [PubMed]
- Wu, A.H.; Yu, M.C.; Tseng, C.C.; Hankin, J.; Pike, M.C. Green tea and risk of breast cancer in Asian Americans. Int. J. Cancer 2003, 106, 574–579. [Google Scholar] [CrossRef] [PubMed]
- Wu, A.H.; Yu, M.C.; Tseng, C.C.; Pike, M.C. Body size, hormone therapy and risk of breast cancer in Asian-American women. Int. J. Cancer 2007, 120, 844–852. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.; Titus-Ernstoff, L.; Newcomb, P.A.; Trentham-Dietz, A.; Anic, G.; Egani, K.M. Tea consumption and risk of breast cancer. Cancer Epidemiol. Biomark. 2009, 18, 341–345. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.M.; Koh, W.P.; Sun, C.L.; Lee, J.P.; Yu, M.C. Green tea intake, ACE gene polymorphism and breast cancer risk among Chinese women in Singapore. Carcinogenesis 2005, 26, 1389–1394. [Google Scholar] [CrossRef] [PubMed]
- Inoue, M.; Robien, K.; Wang, R.; Berg, D.J.V.D.; Koh, W.P.; Yu, M.C. Green tea intake, MTHFR/TYMS genotype and breast cancer risk: The Singapore Chinese Health Study. Carcinogenesis 2008, 29, 1967–1972. [Google Scholar] [CrossRef] [PubMed]
- Ogunleye, A.A.; Xue, F.; Michels, K.B. Green tea consumption and breast cancer risk or recurrence: A meta-analysis. Breast Cancer Res. Treat. 2010, 119, 477–484. [Google Scholar] [CrossRef] [PubMed]
- Guyton, K.Z.; Kensler, T.W. Oxidative mechanisms in carcinogenesis. Br. Med. Bull. 1993, 49, 523–544. [Google Scholar] [PubMed]
- Rathore, K.; Choudhary, S.; Odoi, A.; Wang, H.C.R. Green tea catechin intervention of reactive oxygen species-mediated ERK pathway activation and chronically induced breast cell carcinogenesis. Carcinogenesis 2012, 33, 174–183. [Google Scholar] [CrossRef] [PubMed]
- Ruch, R.J.; Cheng, S.J.; Klaunig, J.E. Prevention of cytotoxicity and inhibition of intercellular communication by antioxidant catechins isolated from Chinese green tea. Carcinogenesis 1989, 10, 1003–1008. [Google Scholar] [CrossRef] [PubMed]
- Kaur, S.; Greaves, P.; Cooke, D.N.; Edwards, R.; Steward, W.P.; Gescher, A.J.; Marczylo, T.H. Breast cancer prevention by green tea catechins and black tea theaflavins in the C3(1) SV40 T,t antigen transgenic mouse model is accompanied by increased apoptosis and a decrease in oxidative DNA adducts. J. Agric. Food Chem. 2007, 55, 3378–3385. [Google Scholar] [CrossRef] [PubMed]
- Parvez, S.; Tabassum, H.; Rehman, H.; Banerjee, B.D.; Athar, M.; Raisuddin, S. Catechin prevents tamoxifen-induced oxidative stress and biochemical perturbations in mice. Toxicology 2006, 225, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Abrahim, N.N.; Kanthimathi, M.S.; Abdul-Aziz, A. Piper betle shows antioxidant activities, inhibits MCF-7 cell proliferation and increases activities of catalase and superoxide dismutase. BMC Complement. Altern. Med. 2012, 12, 220–230. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, M.R.; Nabavi, S.F.; Daglia, M.; Rastrell, L. Epigallocatechin gallate and mitochondria—A story of life and death. Pharmacol. Res. 2016, 104, 70–85. [Google Scholar] [CrossRef] [PubMed]
- Van Aller, G.S.; Carson, J.D.; Tang, W.; Peng, H.; Zhao, L.; Copeland, R.A.; Tummino, P.J.; Luo, L. Epigallocatechingallate (EGCG), a major component of green tea, is a dual phosphoinositide-3-kinase/mTOR inhibitor. Biochem. Biophys. Res. Commun. 2011, 406, 194–199. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.H.; Lin, C.C.; Lin, J.K.; Chen, W.J. Tea polyphenol (−)-epigallocatechin 3-gallate suppresses heregulin-beta 1-induced fatty acid synthase expression in human breast cancer cells by inhibiting phosphatidylinositol 3-kinase/Akt and mitogen-activated protein kinase cascade signaling. J. Agric. Food Chem. 2007, 55, 5030–5037. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Zhang, X.W.; Rieger-Christ, K.M.; Summerhayes, I.C.; Wazer, D.E.; Paulson, K.E.; Yee, A.S. Suppression of Wnt signaling by the green tea compound (−)-epigallocatechin 3-gallate (EGCG) in invasive breast cancer cells-Requirement of the transcriptional repressor HBP1. J. Biol. Chem. 2006, 281, 10865–10875. [Google Scholar] [CrossRef] [PubMed]
- Bigelow, R.L.H.; Cardelli, J.A. The green tea catechins, (−)-epigallocatechin-3-gallate (EGCG) and (−)-epicatechin-3-gallate (ECG), inhibit HGF/Met signaling in immortalized and tumorigenic breast epithelial cells. Oncogene 2006, 25, 1922–1930. [Google Scholar] [CrossRef] [PubMed]
- Pike, M.C.; Spicer, D.V.; Dahmoush, L.; Press, M.F. Estrogens, progestogens, normal breast cell-proliferation, and breast-cancer risk. Epidemiol. Rev. 1992, 15, 17–35. [Google Scholar]
- Haldosen, L.A.; Zhao, C.Y.; Dahlman-Wright, K. Estrogen receptor beta in breast cancer. Mol. Cell. Endocrinol. 2014, 382, 665–672. [Google Scholar] [CrossRef] [PubMed]
- Heldring, N.; Pike, A.; Andersson, S.; Matthews, J.; Cheng, G.; Hartman, J.; Tujague, M.; Strom, A.; Treuter, E.; Warner, M.; et al. Estrogen receptors: How do they signal and what are their targets. Physiol. Rev. 2007, 87, 905–931. [Google Scholar] [CrossRef] [PubMed]
- Kuruto-Niwa, R.; Inoue, S.; Ogawa, S.; Muramatsu, M.; Nozawa, R. Effects of tea catechins on the ERE-regulated estrogenic activity. J. Agric. Food Chem. 2000, 48, 6355–6361. [Google Scholar] [CrossRef] [PubMed]
- Goodin, M.G.; Fertuck, K.C.; Zacharewski, T.R.; Rosengren, R.J. Estrogen receptor-mediated actions of polyphenoliccatechins in vivo and in vitro. Toxicol. Sci. 2002, 69, 354–361. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.Y.; Yuan, Y.Y.; Meeran, S.M.; Tollefsbol, T.O. Synergistic epigenetic reactivation of estrogen receptor-alpha (ERα) by combined green tea polyphenol and histone deacetylase inhibitor in ER alpha-negative breast cancer cells. Mol. Cancer 2010, 9, 274. [Google Scholar] [CrossRef] [PubMed]
- Shim, J.H.; Choi, H.S.; Pugliese, A.; Lee, S.Y.; Chae, J.I.; Choi, B.Y.; Bode, A.M.; Dong, Z. (−)-Epigallocatechin gallate regulates CD3-mediated T cell receptor signaling in leukemia through the inhibition of ZAP-70 kinase. J. Biol. Chem. 2008, 283, 28370–28379. [Google Scholar] [CrossRef] [PubMed]
- Umeda, D.; Yano, S.; Yamada, K.; Tachibana, H. Green tea polyphenol epigallocatechin-3-gallate signaling pathway through 67-kDa laminin receptor. J. Biol. Chem. 2008, 283, 3050–3058. [Google Scholar] [CrossRef] [PubMed]
- Shim, J.H.; Su, Z.Y.; Chae, J.I.; Kim, D.J.; Zhu, F.; Ma, W.Y.; Bode, A.M.; Yang, C.S.; Dong, Z. Epigallocatechin gallate suppresses lung cancer cell growth through Ras-GTPase-activating protein SH3 domain-binding protein 1. Cancer Prev. Res. (Phila) 2010, 3, 670–679. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; He, Z.; Ermakova, S.; Zheng, D.; Tang, F.; Cho, Y.Y.; Zhu, F.; Ma, W.Y.; Sham, Y.; Rogozin, E.A.; et al. Direct inhibition of insulin-like growth factor-I receptor kinase activity by (−)-epigallocatechin-3-gallate regulates cell transformation. Cancer Epidemiol. Biomark. Prev. 2007, 16, 598–605. [Google Scholar] [CrossRef] [PubMed]
- Ermakova, S.; Choi, B.Y.; Choi, H.S.; Kang, B.S.; Bode, A.M.; Dong, Z. The intermediate filament protein vimentin is a new target for epigallocatechin gallate. J. Biol. Chem. 2005, 280, 16882–16890. [Google Scholar] [CrossRef] [PubMed]
- Leone, M.; Zhai, D.; Sareth, S.; Kitada, S.; Reed, J.C.; Pellecchia, M. Cancer prevention by tea polyphenols is linked to their direct inhibition of antiapoptotic Bcl-2-family proteins. Cancer Res. 2003, 63, 8118–8121. [Google Scholar] [PubMed]
- Ermakova, S.P.; Kang, B.S.; Choi, B.Y.; Choi, H.S.; Schuster, T.F.; Ma, W.Y.; Bode, A.M.; Dong, Z. (-)-Epigallocatechin gallate overcomes resistance to etoposide-induced cell death by targeting the molecular chaperone glucose-regulated protein 78. Cancer Res. 2006, 66, 9260–9269. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Tang, F.; Ermakova, S.; Li, M.; Zhao, Q.; Cho, Y.Y.; Ma, W.Y.; Choi, H.S.; Bode, A.M.; Yang, C.S.; et al. Fyn is a novel target of (−)-epigallocatechin gallate in the inhibition of JB6 Cl41 cell transformation. Mol. Carcinog. 2008, 47, 172–183. [Google Scholar] [CrossRef] [PubMed]
- Jones, P.A.; Takai, D. The role of DNA methylation in mammalian epigenetics. Science 2001, 293, 1068–1070. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.L.; Shim, J.Y.; Zhu, B.T. Mechanisms for the inhibition of DNA methyltransferases by tea catechins and bioflavonoids. Mol. Pharmacol. 2005, 68, 1018–1030. [Google Scholar] [CrossRef] [PubMed]
- Fang, M.Z.; Wang, Y.; Ai, N.; Hou, Z.; Sun, Y.; Lu, H.; Welsh, W.; Yang, C.S. Tea polyphenol (−)-epigallocatechin-3-gallate inhibits DNA methyltransferase and reactivates methylation-silenced genes in cancer cell lines. Cancer Res. 2003, 63, 7563–7570. [Google Scholar] [PubMed]
- Huo, C.; Yang, H.; Cui, Q.C.; Dou, Q.P.; Chan, T.H. Proteasome inhibition in human breast cancer cells with high catechol-o-methyltransferase activity by green tea polyphenol EGCG analog. Bioorg. Med. Chem. 2010, 18, 1252–1258. [Google Scholar] [CrossRef] [PubMed]
- Landis-Piwowar, K.R.; Wan, S.B.; Wiegand, R.A.; Kuhn, D.J.; Chan, T.H.; Dou, Q.P. Methylation suppresses the proteasome-inhibitory function of green tea polyphenols. J. Cell. Physiol. 2007, 213, 256–260. [Google Scholar] [CrossRef] [PubMed]
- Landis-Piwowar, K.R.; Chen, D.I.; Chan, T.H.; Dou, Q.P. Inhibition of catechol-O-methyltransferase activity in human breast cancer cells enhances the biological effect of the green tea polyphenol (−)-EGCG. Oncol. Rep. 2010, 24, 563–569. [Google Scholar] [PubMed]
- Folkman, J. Angiogenesis in cancer, vascular, rheumatoid and other disease. Nat. Med. 1995, 1, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, N.; Davis-Smyth, T. The biology of vascular endothelial growth factor. Endocr. Rev. 1997, 18, 4–25. [Google Scholar] [CrossRef] [PubMed]
- Mukhtar, H.; Katiyar, S.K.; Agarwal, R. Green tea and skin-anticarcinogenic effects. J. Invest. Dermatol. 1994, 102, 3–7. [Google Scholar] [CrossRef] [PubMed]
- Stoner, G.D.; Mukhtar, H. Polyphenols as cancer chemopreventive agents. J. Cell. Biochem. 1995, 22, 169–180. [Google Scholar] [CrossRef]
- Tang, F.Y.; Meydani, M. Green tea catechins and vitamin E inhibit angiogenesis of human microvascular endothelial cells through suppression of IL-8 production. Nutr. Cancer 2001, 41, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Maryam, R.S.; Shao, Z.M.; David, H. Green tea inhibits vascular endothelial growth factor (VEGF) induction inhuman breast cancer cells. J. Nutr. 2002, 132, 2307–2311. [Google Scholar]
- Shaun, K.R.; Guo, W.M.; Liu, L.P. Green tea catechin, epigallocatechin-3-gallate, inhibits vascular endothelial growth factor angiogenic signaling by disrupting the formation of a receptor complex. J. Cancer 2006, 118, 1635–1644. [Google Scholar]
- Seeram, N.P.; Zhang, Y.; Nair, M.G. Inhibition of proliferation of human cancer cells and cyclooxygenase enzymes by anthocyanidins and catechins. Nutr. Cancer 2003, 46, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Deguchi, H.; Fujii, T.; Nakagawa, S.; Koga, T.; Shirouzu, K. Analysis of cell growth inhibitory effects of catechin through MAPK in human breast cancer cell line T47D. Int. J. Oncol. 2002, 21, 1301–1305. [Google Scholar] [CrossRef] [PubMed]
- Alshatwi, A.A. Catechin hydrate suppresses MCF-7 proliferation through TP53/Caspase-mediated apoptosis. J. Exp. Clin. Canc. Res. 2010, 29, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Afsar, T.; Trembley, J.H.; Salomon, C.E.; Razak, S.; Khan, M.R.; Ahmed, K. Growth inhibition and apoptosis in cancer cells induced by polyphenolic compounds of Acacia hydaspica: Involvement of multiple signal transduction pathways. Sci. Rep. 2016, 6, 23077. [Google Scholar] [CrossRef] [PubMed]
- Jo, E.H.; Lee, S.J.; Ahn, N.S.; Park, J.S.; Hwang, J.W.; Kim, S.H.; Aruoma, O.I.; Lee, Y.S.; Kang, K.S. Induction of apoptosis in MCF-7 and MDA-MB-231 breast cancer cells by Oligonol is mediated by Bcl-2 family regulation and MEK/ERK signaling. Eur. J. Cancer Prev. 2007, 16, 342–347. [Google Scholar] [CrossRef] [PubMed]
- Nifli, A.P.; Kampa, M.; Alexaki, V.I.; Notas, G.; Castanas, E. Polyphenol interaction with the T47D human breast cancer cell line. J. Dairy Res. 2005, 72, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Palmer, A.E.; Jin, C.; Reed, J.C.; Tsien, R.Y. Bcl-2-Mediated alterations in endoplasmic reticulum Ca2t analyzed with an improved genetically encoded fluorescent sensor. Proc. Natl. Acad. Sci. USA 2004, 101, 17404–17409. [Google Scholar] [CrossRef] [PubMed]
- Hsu, Y.C.; Liou, Y.M. The Anti-cancer effects of (−)-epigalocathine-3-gallate on the signaling pathways associated with membrane receptors in MCF-7 cells. J. Cell. Physiol. 2011, 226, 2721–2730. [Google Scholar] [CrossRef] [PubMed]
- Luo, K.; Koa, C.H.; Yue, G.G.L.; Lee, J.K.M.; Li, K.K.; Lee, M.; Li, G.; Fung, K.P.; Leung, P.C.; Lau, C.B.S. Green tea (Camellia sinensis) extract inhibits both the metastasis and osteolytic components of mammary cancer 4T1 lesions in mice. J. Nutr. Biochem. 2014, 25, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Slivova, V.; Zaloga, G.; DeMichele, S.J.; Mukerji, P.; Huang, Y.S.; Siddiqui, R.; Harvey, K.; Valachovicova, T.; Sliva, D. Green tea polyphenols modulate secretion of urokinase plasminogen activator (uPA) and inhibit invasive behavior of breast cancer cells. Nutr. Cancer 2005, 52, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Han, G.; Fan, B.; Zhou, Y.; Zhou, X.; Wei, L.; Zhang, J. Green tea (−)-epigallocatechin-3-gallate down-regulates VASP expression and inhibits breast cancer cell migration and invasion by attenuating Rac1 activity. Eur. J. Pharmacol. 2009, 606, 172–179. [Google Scholar] [CrossRef] [PubMed]
- Annabi, B.; Lachambre, M.P.; Bousquet-Gagnon, N.; Page, M.; Gingras, D.; Beliveau, R. Green tea polyphenol (−)-epigallocatechin 3-gallate inhibits MMP-2 secretion and MT1-MMP-driven migration in glioblastoma cells. Biochim. Biophys. Acta 2002, 1542, 209–220. [Google Scholar] [CrossRef]
- Sen, T.; Chatterjee, A. Epigallocatechin-3-gallate (EGCG) downregulates EGF-induced MMP-9 in breast cancer cells: Involvement of integrin receptor alpha 5 beta 1 in the process. Eur. J. Nutr. 2011, 50, 465–478. [Google Scholar] [CrossRef] [PubMed]
- Xiaokaiti, Y.; Wu, H.M.; Chen, Y.; Yang, H.P.; Duan, J.H.; Li, X.; Pan, Y.; Tie, L.; Zhang, L.R.; Li, X.J. EGCG reverses human neutrophil elastase-induced migration in A549 cells by directly binding to HNE and by regulating alpha1-AT. Sci. Rep. 2015, 5, 11494. [Google Scholar] [CrossRef] [PubMed]
- Farabegoli, F.; Papi, A.; Orlandi, M. (−)-Epigallocatechin-3-gallate down-regulates EGFR, MMP-2, MMP-9 and EMMPRIN and inhibits the invasion of MCF-7 tamoxifen-resistant cells. Biosci. Rep. 2010, 31, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Li, J.P.; Zhang, C. Green tea consumption and breast cancer risk: Three recent meta-analyses. Breast Cancer Res. Treat. 2011, 127, 581–582. [Google Scholar] [CrossRef] [PubMed]
- Ogunleye, A.A.; Xue, F.; Michels, K.B. Green tea consumption and breast cancer risk: Three recent meta-analyses Rebuttal. Breast Cancer Res. Treat. 2011, 127, 583. [Google Scholar]
- Baker, J.A.; Beehler, G.P.; Sawant, A.C.; Jayaprakash, V.; McCann, S.E.; Moysich, K.B. Consumption of coffee, but not black tea, is associated with decreased risk of premenopausal breast cancer. J. Nutr. 2006, 136, 166–171. [Google Scholar] [PubMed]
- Rosenblatt, K.A.; Thomas, D.B.; Jimenez, L.M.; Fish, B.; McTiernan, A.; Stalsberg, H.; Stemhagen, A.; Thompson, W.D.; Curnen, M.G.M.; Satariano, W.; et al. The relationship between diet and breast cancer in men (United States). Cancer Cause Control 1999, 10, 107–113. [Google Scholar] [CrossRef]
- Weng, Z.; Greenhaw, J.; Salminen, W.F.; Shi, Q. Mechanisms for epigallocatechin gallate induced inhibition of drug metabolizing enzymes in rat liver microsomes. Toxicol. Lett. 2012, 214, 328–338. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, M.; Paul, F.; Moobed, M.; Baumann, G.; Zimmermann, B.F.; Stangl, K.; Stangl, V. The activity of catechol-O-methyltransferase (COMT) is not impaired by high doses of epigallocatechin-3-gallate (EGCG) in vivo. Eur. J. Pharmacol. 2014, 740, 645–651. [Google Scholar] [CrossRef] [PubMed]
- Suganuma, M.; Okabe, S.; Sueoka, N.; Sueoka, E.; Matsuyama, S.; Imai, K.; Nakachi, K.; Fujiki, H. Green tea and cancer chemoprevention. Mutat. Res. 1999, 428, 339–344. [Google Scholar] [CrossRef]
- Forester, S.C.; Lambert, J.D. The catechol-O-methyltransferase inhibitor, tolcapone, increases the bioavailability of unmethylated (−)-epigallocatechin-3-gallate in mice. J. Funct. Foods 2015, 17, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Krupkova, O.; Ferguson, S.J.; Wuertz-Kozak, K. Stability of (−)-epigallocatechin gallate and its activity in liquid formulations and delivery systems. J. Nutr. Biochem. 2016, 37, 1–12. [Google Scholar] [CrossRef]
- Dube, A.; Nicolazzo, J.A.; Larson, I. Chitosan nanoparticles enhance the intestinal absorption of the green tea catechins (+)-catechin and (−)-epigallocatechingallate. Eur. J. Pharm. Sci. 2014, 41, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Yadav, R.; Kumar, D.; Kumari, A.; Yadav, S.K. Encapsulation of catechin and epicatechin on BSA NPs improved their stability and antioxidant potential. EXCLI J. 2014, 13, 331–346. [Google Scholar] [PubMed]
- Liang, J.; Cao, L.; Zhang, L.; Wan, X. Preparation, characterization, and in vitro antitumor activity of folate conjugated chitosan coated EGCG nanoparticles. Food Sci. Biotechnol. 2014, 23, 569–575. [Google Scholar] [CrossRef]
- Fangueiro, J.F.; Calpena, A.C.; Clares, B.; Andreani, T.; Egea, M.A.; Veiga, F.J.; Garcia, M.L.; Silva, A.M.; Souto, E.B. Biopharmaceutical evaluation of epigallocatechin gallate-loaded cationic lipid nanoparticles (EGCG-LNs): In vivo, in vitro and ex vivo studies. Int. J. Pharm. 2016, 502, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Tagashira, T.; Choshi, T.; Hibino, S.; Kamishikiryou, J.; Sugihara, N. Influence of gallate and pyrogallol moieties on the intestinal absorption of (−)-epicatechin and (−)-epicatechingallate. J. Food Sci. 2012, 77, H208–H215. [Google Scholar] [CrossRef] [PubMed]
- Naumovski, N.; Blades, B.L.; Roach, P.D. Food inhibits the oral bioavailability of the major green tea antioxidant epigallocatechingallate in humans. Antioxidants 2015, 4, 373–393. [Google Scholar] [CrossRef] [PubMed]
- Son, Y.R.; Chung, J.H.; Ko, S.; Shim, S.M. Combinational enhancing effects of formulation and encapsulation on digestive stability and intestinal transport of green tea catechins. J. Microencapsul. 2016, 33, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Aditya, N.P.; Aditya, S.; Yang, H.; Kim, H.W.; Park, S.O.; Ko, S. Co-Delivery of hydrophobic curcumin and hydrophilic catechin by a water-in-oil-in-water double emulsion. Food Chem. 2015, 173, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Garcia, J.P.D.; Hsieh, M.F.; Doma, B.T.; Peruelo, D.C.; Chen, I.H.; Lee, H.M. Synthesis of gelatin-γ-polyglutamic acid-based hydrogel for the in vitro controlled release of epigallocatechin gallate (EGCG) from Camellia sinensis. Polymers 2014, 6, 39–58. [Google Scholar] [CrossRef]

| Type of Study | Location | Number of Subjects | Main Results | References |
|---|---|---|---|---|
| Population-based cohort study | Shanghai, China | 1399 women with breast cancer | Drinking tea regularly (>100 g dried tea per month) was inversely associated with overall depression. | Chen et al. (2010) [9] |
| Hospital-based case–control study | Hong Kong, China | Cases: 439 Controls: 434 | Habitual tea drinking was significantly associated with a lower risk for breast cancer in premenopausal women (OR = 0.62, 95%CI: 0.40–0.97). | Li et al. (2016) [10] |
| Case–control study | Southeast China | Cases: 1009 Controls: 1009 | Green tea consumption was associated with a reduced risk of breast cancer. | Zhang et al. (2007) [12] |
| Prospective cohort study | Saitama Prefecture, Japan | 9 years of follow-up study (71,248.5 person-years) | Drinking green tea had a potentially preventive effect on breast cancer | Imai et al. (1997) [13] |
| Population-based study | Shanghai, China | Cases: 3454 Controls: 3474 | Drinking green tea regularly was weakly associated with a decreased risk of breast cancer. | Shrubsole et al. (2009) [14] |
| Long-term cohort study (1980–2002) | Boston, USA | 85,987 female participants | There was a significant inverse association of caffeine intake with breast cancer among postmenopausal women | Ganmaa et al. (2008) [15] |
| Population-based cohort study | Shanghai, China | 74,942 Chinese women | Women who started drinking tea at 25 years of age or younger had a hazard ratio 0.69 (CI: 0.41–1.17) to develop premenopausal breast cancer, compared with non-tea drinkers | Dai et al. (2010) [16] |
| Case–control study | Southeast China | Cases: 1009 Controls: 1009 | Green tea intake was associated with decreased breast cancer risk in premenopausal and postmenopausal Chinese women, and there was an additional decreased risk from the joint effect of green tea and mushrooms | Zhang et al. (2009) [17] |
| Population-based, case–control study | Los Angeles, USA | Cases: 501 Controls: 594 | Green tea consumption showed a significantly reduced risk of breast cancer, while black tea consumption was not associated with the risk of breast cancer | Wu et al. (2003) [18] |
| Population-based case–control study | Massachusetts, USA | Cases: 5082 Controls: 4501 | Among women less than 50 years old, those who drank three or more cups of tea per day had a 37% reduced breast cancer risk compared to their counterparts that did not drink tea | Kumar et al. (2009) [20] |
| Nested case–control study | Singapore | Cases: 297 Controls: 665 | There was significant association between green tea intake frequency and decreased risk of breast cancer in the women with high-activity of angiotensin-converting enzyme (ACE) genotype (p = 0.039) | Yuan et al. (2005) [21] |
| Nested case–control study | Singapore | Cases: 380 Controls: 662 | Green tea intake was inversely associated with decreased breast cancer risk among women with low folate intake or high-activity MTHFR/TYMS genotypes | Inoue et al. (2008) [22] |
| Meta-analysis | Boston, USA | Cases: 5617 | Increased green tea consumption (>3 cups/day) was inversely associated with recurrence (Pooled RR = 0.73, 95% CI: 0.56–0.96). An analysis of case–control studies of incidence suggested an inverse association with a pooled RR of 0.81 (95% CI: 0.75, 0.88) while no association was found among cohort studies of incidence | Ogunleye et al. (2010) [23] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xiang, L.-P.; Wang, A.; Ye, J.-H.; Zheng, X.-Q.; Polito, C.A.; Lu, J.-L.; Li, Q.-S.; Liang, Y.-R. Suppressive Effects of Tea Catechins on Breast Cancer. Nutrients 2016, 8, 458. https://doi.org/10.3390/nu8080458
Xiang L-P, Wang A, Ye J-H, Zheng X-Q, Polito CA, Lu J-L, Li Q-S, Liang Y-R. Suppressive Effects of Tea Catechins on Breast Cancer. Nutrients. 2016; 8(8):458. https://doi.org/10.3390/nu8080458
Chicago/Turabian StyleXiang, Li-Ping, Ao Wang, Jian-Hui Ye, Xin-Qiang Zheng, Curt Anthony Polito, Jian-Liang Lu, Qing-Sheng Li, and Yue-Rong Liang. 2016. "Suppressive Effects of Tea Catechins on Breast Cancer" Nutrients 8, no. 8: 458. https://doi.org/10.3390/nu8080458
APA StyleXiang, L.-P., Wang, A., Ye, J.-H., Zheng, X.-Q., Polito, C. A., Lu, J.-L., Li, Q.-S., & Liang, Y.-R. (2016). Suppressive Effects of Tea Catechins on Breast Cancer. Nutrients, 8(8), 458. https://doi.org/10.3390/nu8080458