Metabolic Effects of a 24-Week Energy-Restricted Intervention Combined with Low or High Dairy Intake in Overweight Women: An NMR-Based Metabolomics Investigation
Abstract
:1. Introduction
2. Material and Methods
2.1. Subjects
2.2. Anthropometrics and Blood Pressure
2.3. Biochemical Analysis
2.4. NMR Analysis
2.5. NMR Data Preprocessing
2.6. Multivariate Data Analysis
2.7. Identification and Quantification of Metabolites
2.8. Statistical Analysis
3. Results and Discussion
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Nguyen, D.M.; El-Serag, H.B. The epidemiology of obesity. Gastroenterol. Clin. North Am. 2010, 39, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Stevens, G.A.; Singh, G.M.; Lu, Y.; Danaei, G.; Lin, J.K.; Finucane, M.M.; Bahalim, A.N.; McIntire, R.K.; Gutierrez, H.R.; Cowan, M.; et al. National, regional, and global trends in adult overweight and obesity prevalences. Popul. Health Metr. 2012, 10, 22. [Google Scholar] [CrossRef] [PubMed]
- Giskes, K.; Van Lenthe, F.; Avendano-Pabon, M.; Brug, J. A systematic review of environmental factors and obesogenic dietary intakes among adults: Are we getting closer to understanding obesogenic environments? Obes. Rev. 2011, 12, e95–e106. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Hao, T.; Rimm, E.B.; Willett, W.C.; Hu, F.B. Changes in diet and lifestyle and long-term weight gain in women and men. N. Eng. J. Med. 2011, 364, 2392–2404. [Google Scholar] [CrossRef] [PubMed]
- Schaafsma, G. The Western Diet with a Special Focus on Dairy Products; Institut Danone: Paris, France, 1997. [Google Scholar]
- Bray, G.A.; Paeratakul, S.; Popkin, B.M. Dietary fat and obesity: A review of animal, clinical and epidemiological studies. Physiol. Behav. 2004, 83, 549–555. [Google Scholar] [CrossRef] [PubMed]
- Lanou, A.J.; Barnard, N.D. Dairy and weight loss hypothesis: An evaluation of the clinical trials. Nutr. Rev. 2008, 66, 272–279. [Google Scholar] [CrossRef] [PubMed]
- Louie, J.C.Y.; Flood, V.M.; Hector, D.J.; Rangan, A.M.; Gill, T.P. Dairy consumption and overweight and obesity: A systematic review of prospective cohort studies. Obes. Rev. 2011, 12, e582–e592. [Google Scholar] [CrossRef] [PubMed]
- Dougkas, A.; Reynolds, C.K.; Givens, I.D.; Elwood, P.C.; Minihane, A.M. Associations between dairy consumption and body weight: A review of the evidence and underlying mechanisms. Nutr. Res. Rev. 2011, 24, 72–95. [Google Scholar] [CrossRef] [PubMed]
- Abargouei, A.S.; Janghorbani, M.; Salehi-Marzijarani, M.; Esmaillzadeh, A. Effect of dairy consumption on weight and body composition in adults: A systematic review and meta-analysis of randomized controlled clinical trials. Int. J. Obes. 2012, 36, 1485–1493. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Pan, A.; Malik, V.S.; Hu, F.B. Effects of dairy intake on body weight and fat: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2012, 96, 735–747. [Google Scholar] [CrossRef] [PubMed]
- Paddon-Jones, D.; Westman, E.; Mattes, R.D.; Wolfe, R.R.; Astrup, A.; Westerterp-Plantenga, M. Protein, weight management, and satiety. Am. J. Clin. Nutr. 2008, 87, 1558S–1561S. [Google Scholar] [PubMed]
- Zemel, M.B.; Richards, J.; Milstead, A.; Campbell, P. Effects of calcium and dairy on body composition and weight loss in African-American adults. Obes. Res. 2005, 13, 1218–1225. [Google Scholar] [CrossRef] [PubMed]
- Melanson, E.L.; Sharp, T.A.; Schneider, J.; Donahoo, W.T.; Grunwald, G.K.; Hill, J.O. Relation between calcium intake and fat oxidation in adult humans. Int. J. Obes. 2003, 27, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Christensen, R.; Lorenzen, J.K.; Svith, C.R.; Bartels, E.M.; Melanson, E.L.; Saris, W.H.; Tremblay, A.; Astrup, A. Effect of calcium from dairy and dietary supplements on faecal fat excretion: A meta-analysis of randomized controlled trials. Obes. Rev. 2009, 10, 475–486. [Google Scholar] [CrossRef] [PubMed]
- St-Onge, M.P.; Farnworth, E.R.; Jones, P.J. Consumption of fermented and nonfermented dairy products: Effects on cholesterol concentrations and metabolism. Am. J. Clin. Nutr. 2000, 71, 674–681. [Google Scholar] [PubMed]
- Greiner, T.; Bäckhed, F. Effects of the gut microbiota on obesity and glucose homeostasis. Trends Endocrinol. Metab. 2011, 22, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, S.D.; Amer, B.; Young, J.F.; Mortensen, G.; Petersen, R.K.; Kristiansen, K.; Dalsgaard, T.K. Medium chain fatty acids from milk induce angiopoietin-like 4 (ANGPTL4) gene expression. Int. Dairy J. 2015, 42, 34–41. [Google Scholar] [CrossRef]
- Lafferty, M.J.; Bradford, K.C.; Erie, D.A.; Neher, S.B. Angiopoietin-like protein 4 inhibition of lipoprotein lipase evidence for reversible complex formation. J. Biol. Chem. 2013, 288, 28524–28534. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.P.; Park, Y.; Ziegler, T.R. Nutritional metabolomics: Progress in addressing complexity in diet and health. Ann. Rev. Nutr. 2012, 32, 183–202. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Yde, C.C.; Clausen, M.R.; Kristensen, M.; Lorenzen, J.; Astrup, A.; Bertram, H.C. Metabolomics investigation to shed light on cheese as a possible brick in the French paradox puzzle. J. Agric. Food Chem. 2015, 63, 2830–2839. [Google Scholar] [CrossRef] [PubMed]
- Savorani, F.; Tomasi, G.; Engelsen, S.B. icoshift: A versatile tool for the rapid alignment of 1D NMR spectra. J. Magn. Reson. 2010, 202, 190–202. [Google Scholar] [CrossRef] [PubMed]
- Biosystems data analysis. Available online: http://www.bdagroup.nl/Home.php (accessed on 11 March 2014).
- Zheng, H.; Yde, C.C.; Arnberg, K.; Mølgaard, C.; Michaelsen, K.F.; Larnkjær, A.; Bertram, H.C. NMR-based metabolomic profiling of overweight adolescents: An elucidation of the effects of inter-/intraindividual differences, gender, and pubertal development. BioMed Res. Int. 2014, 2014, 537157. [Google Scholar] [CrossRef] [PubMed]
- Psychogios, N.; Hau, D.D.; Peng, J.; Guo, A.C.; Mandal, R.; Bouatra, S.; Sinelnikov, I.; Krishnamurthy, R.; Eisner, R.; Gautam, B.; et al. The human serum metabolome. PLoS ONE 2011, 6, e16957. [Google Scholar] [CrossRef] [PubMed]
- Heinzmann, S.S.; Brown, I.J.; Chan, Q.; Bictash, M.; Dumas, M.E.; Kochhar, S.; Stamler, J.; Holmes, E.; Elliott, P.; Nicholson, J.K. Metabolic profiling strategy for discovery of nutritional biomarkers: Proline betaine as a marker of citrus consumption. Am. J. Clin. Nutr. 2010, 92, 436–443. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, A.; Gibney, M.J.; Brennan, L. Dietary intake patterns are reflected in metabolomic profiles: Potential role in dietary assessment studies. Am. J. Clin. Nutr. 2011, 93, 314–321. [Google Scholar] [CrossRef] [PubMed]
- Regueiro, J.; Vallverdú-Queralt, A.; Simal-Gándara, J.; Estruch, R.; Lamuela-Raventós, R.M. Urinary tartaric acid as a potential biomarker for the dietary assessment of moderate wine consumption: A randomised controlled trial. Br. J. Nutr. 2014, 111, 1680–1685. [Google Scholar] [CrossRef] [PubMed]
- Van Dorsten, F.A.; Grün, C.H.; van Velzen, E.J.; Jacobs, D.M.; Draijer, R.; van Duynhoven, J.P. The metabolic fate of red wine and grape juice polyphenols in humans assessed by metabolomics. Mol. Nutr. Food Res. 2010, 54, 897–908. [Google Scholar] [CrossRef] [PubMed]
- Ogegbo, O.L.; Dissanyake, W.; Crowder, J.; Asekun, O.; Familoni, O.; Branford-White, C.J.; Annie Bligh, S.W. Urinary 1H-NMR metabonomics study on intervention effects of soya milk in Africans. Phytother. Res. 2012, 26, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Moazzami, A.A.; Bondia-Pons, I.; Hanhineva, K.; Juntunen, K.; Antl, N.; Poutanen, K.; Mykkänen, H. Metabolomics reveals the metabolic shifts following an intervention with rye bread in postmenopausal women-a randomized control trial. Nutr. J. 2012, 11, 88. [Google Scholar] [CrossRef] [PubMed]
- Jansen, J.J.; Hoefsloot, H.C.; van der Greef, J.; Timmerman, M.E.; Westerhuis, J.A.; Smilde, A.K. ASCA: Analysis of multivariate data obtained from an experimental design. J. Chemometr. 2005, 19, 469–481. [Google Scholar] [CrossRef]
- Yde, C.C.; Jansen, J.J.; Theil, P.K.; Bertram, H.C.; Knudsen, K.E.B. Different metabolic and absorption patterns of betaine in response to dietary intake of whole-wheat grain, wheat aleurone or rye aleurone in catheterized pigs. Eur. Food Res. Technol. 2012, 235, 939–949. [Google Scholar] [CrossRef]
- Walsh, M.C.; Brennan, L.; Malthouse, J.P.G.; Roche, H.M.; Gibney, M.J. Effect of acute dietary standardization on the urinary, plasma, and salivary metabolomic profiles of healthy humans. Am. J. Clin. Nutr. 2006, 84, 531–539. [Google Scholar] [PubMed]
- McHarg, T.; Rodgers, A.; Charlton, K. Influence of cranberry juice on the urinary risk factors for calcium oxalate kidney stone formation. BJU Int. 2003, 92, 765–768. [Google Scholar] [CrossRef] [PubMed]
- Van Dorsten, F.A.; Daykin, C.A.; Mulder, T.P.; van Duynhoven, J.P. Metabonomics approach to determine metabolic differences between green tea and black tea consumption. J. Agric. Food Chem. 2006, 54, 6929–6938. [Google Scholar] [CrossRef] [PubMed]
- Tormo, M.; Izco, J.M. Alternative reversed-phase high-performance liquid chromatography method to analyse organic acids in dairy products. J. Chromatogr. A 2004, 1033, 305–310. [Google Scholar] [CrossRef] [PubMed]
- Lillefosse, H.H.; Clausen, M.R.; Yde, C.C.; Ditlev, D.B.; Zhang, X.; Du, Z.Y.; Bertram, H.C.; Madsen, L.; Kristiansen, K.; Liaset, B. Urinary loss of tricarboxylic acid cycle intermediates as revealed by metabolomics studies: An underlying mechanism to reduce lipid accretion by whey protein ingestion? J. Proteome Res. 2014, 13, 2560–2570. [Google Scholar] [CrossRef] [PubMed]
- Bingham, S.A. Urine nitrogen as a biomarker for the validation of dietary protein intake. J. Nutr. 2003, 133, 921S–924S. [Google Scholar] [PubMed]
- Zheng, H.; Yde, C.C.; Dalsgaard, T.K.; Arnberg, K.; Mølgaard, C.; Michaelsen, K.F.; Larnkjær, A.; Bertram, H.C. Nuclear magnetic resonance-based metabolomics reveals that dairy protein fractions affect urinary urea excretion differently in overweight adolescents. Eur. Food Res. Technol. 2015, 240, 489–497. [Google Scholar] [CrossRef]
- Morens, C.; Bos, C.; Pueyo, M.E.; Benamouzig, R.; Gausserès, N.; Luengo, C.; Tomè, D.; Gaudichon, C. Increasing habitual protein intake accentuates differences in postprandial dietary nitrogen utilization between protein sources in humans. J. Nutr. 2003, 133, 2733–2740. [Google Scholar] [PubMed]
- Keshaviah, P.R.; Nolph, K.D.; Moore, H.L.; Prowant, B.; Emerson, P.F.; Meyer, M.; Twardowski, Z.J.; Khanna, R.; Ponferrada, L.; Collins, A. Lean body mass estimation by creatinine kinetics. J. Am. Soc. Nephrol. 1994, 4, 1475–1485. [Google Scholar] [PubMed]
- Gowda, S.; Desai, P.B.; Kulkarni, S.S.; Hull, V.V.; Math, A.A.; Vernekar, S.N. Markers of renal function tests. N. Am. J. Med. Sci. 2010, 2, 170–173. [Google Scholar] [PubMed]
- Wang, Z.; Klipfell, E.; Bennett, B.J.; Koeth, R.; Levison, B.S.; DuGar, B.; Feldstein, A.E.; Britt, E.B.; Fu, X.; Chung, Y.M.; et al. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature 2011, 472, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Johri, A.M.; Heyland, D.K.; Hétu, M.F.; Crawford, B.; Spence, J.D. Carnitine therapy for the treatment of metabolic syndrome and cardiovascular disease: Evidence and controversies. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 808–814. [Google Scholar] [CrossRef] [PubMed]
- Collins, H.L.; Drazul-Schrader, D.; Sulpizio, A.C.; Koster, P.D.; Williamson, Y.; Adelman, S.J.; Owen, K.; Sanli, T.; Bellamine, A. l-Carnitine intake and high trimethylamine N-oxide plasma levels correlate with low aortic lesions in ApoE−/− transgenic mice expressing CETP. Atherosclerosis 2016, 244, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Lees, H.J.; Swann, J.R.; Wilson, I.D.; Nicholson, J.K.; Holmes, E. Hippurate: The natural history of a mammalian-microbial cometabolite. J. Proteome Res. 2013, 12, 1527–1546. [Google Scholar] [CrossRef] [PubMed]
- Schmedes, M.S.; Yde, C.C.; Svensson, U.; Håkansson, J.; Baby, S.; Bertram, H.C. Impact of a 6-week very low-calorie diet and weight reduction on the serum and fecal metabolome of overweight subjects. Eur. Food Res. Technol. 2015, 240, 583–594. [Google Scholar] [CrossRef]
- Demigné, C.; Morand, C.; Levrat, M.A.; Besson, C.; Moundras, C.; Rémésy, C. Effect of propionate on fatty acid and cholesterol synthesis and on acetate metabolism in isolated rat hepatocytes. Br. J. Nutr. 1995, 74, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Marcil, V.; Delvin, E.; Garofalo, C.; Levy, E. Butyrate impairs lipid transport by inhibiting microsomal triglyceride transfer protein in Caco-2 cells. J. Nutr. 2003, 133, 2180–2183. [Google Scholar] [PubMed]
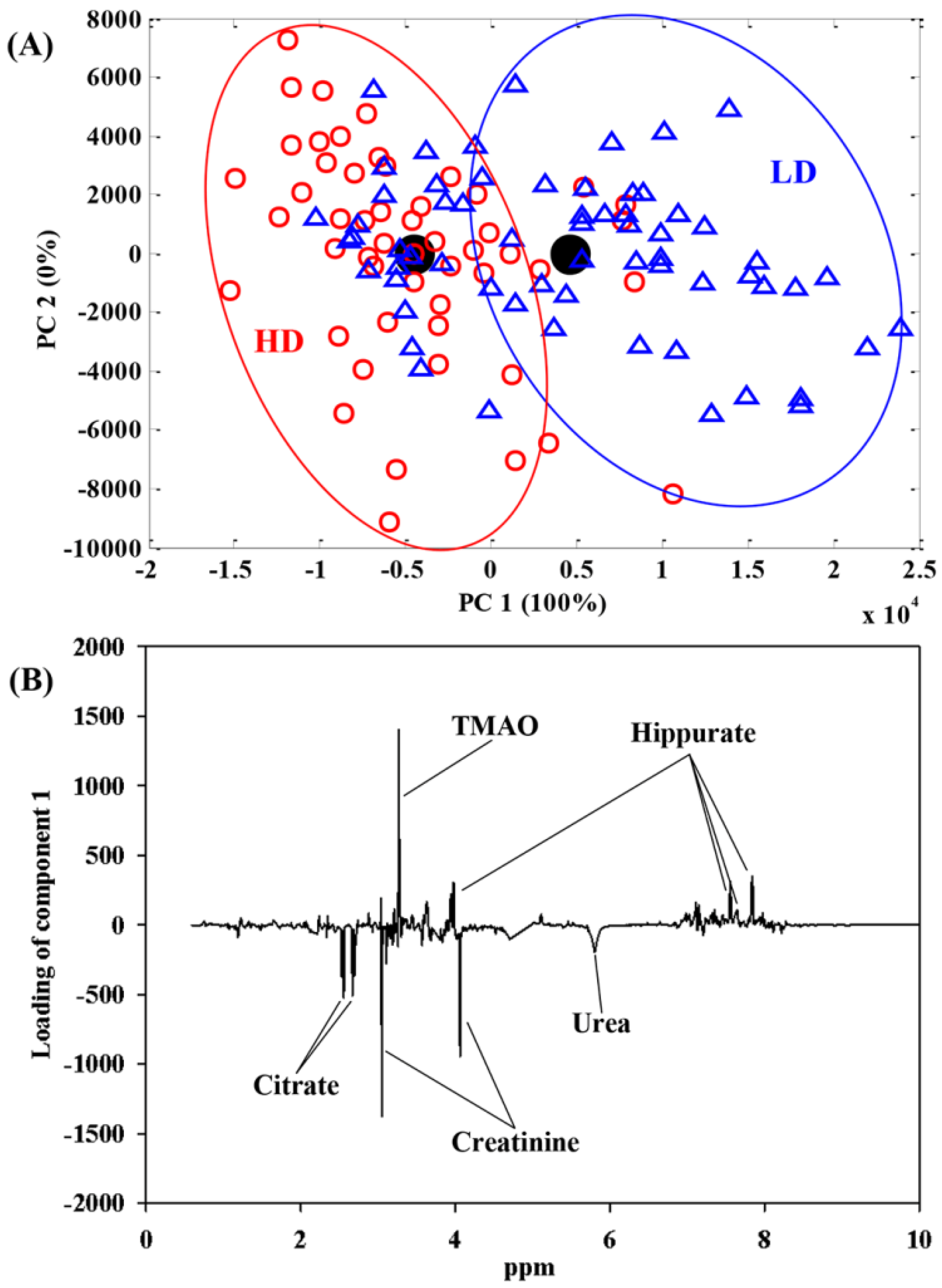
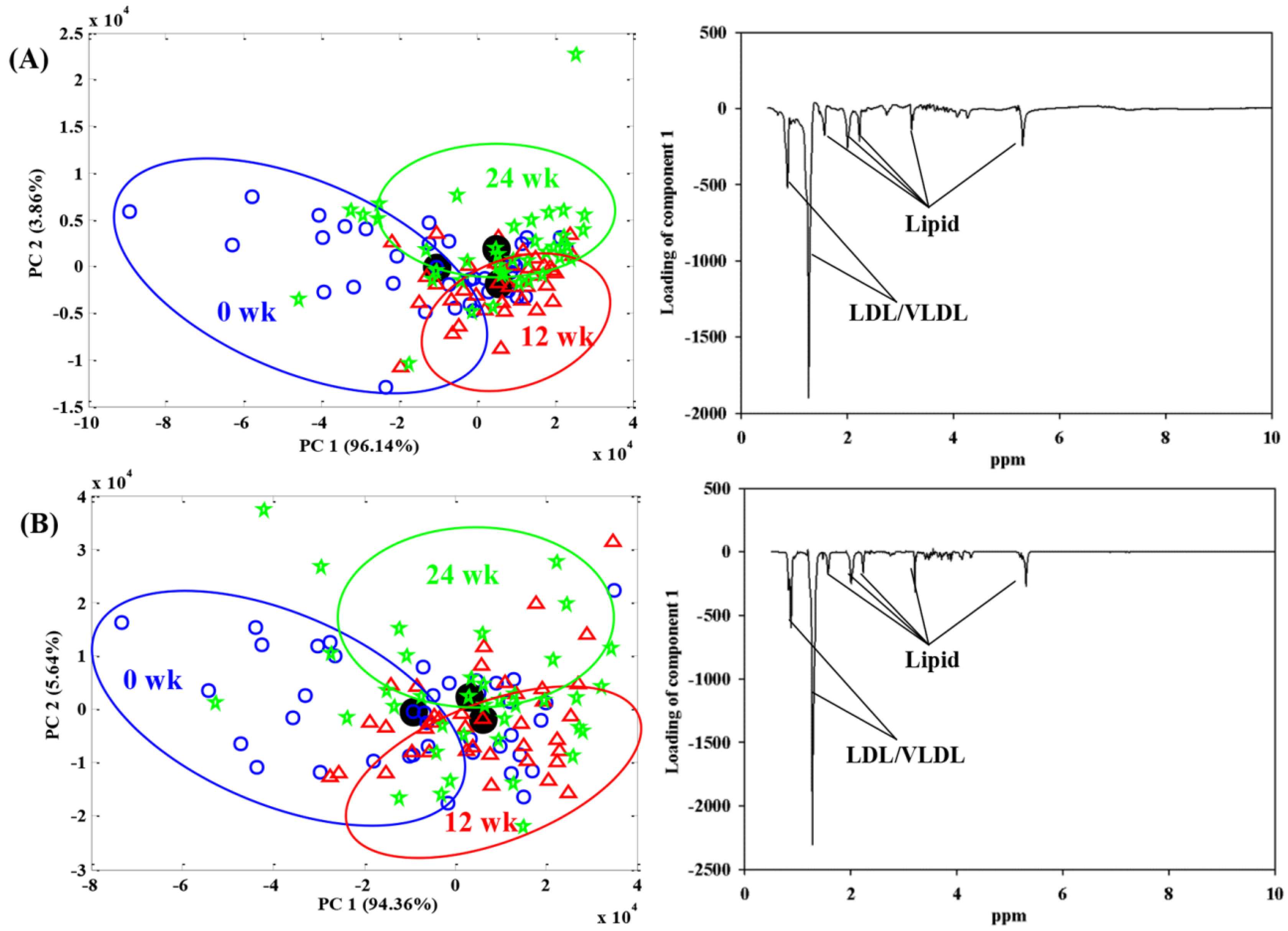
| LD (n = 21) | HD (n = 17) | p | |
|---|---|---|---|
| Age (year) | 45.2 ± 2.9 | 41.3 ± 2.7 | 0.13 |
| BMI (kg/m2) | 30.4 ± 1.5 | 30.0 ± 1.6 | 0.68 |
| FM (kg) | 36.1 ± 2.2 | 36.3 ± 2.6 | 0.91 |
| FFM (kg) | 44.0 ± 2.3 | 47.2 ± 2.3 | 0.50 |
| WC (cm) | 96.3 ± 3.0 | 97.5 ± 3.0 | 0.66 |
| HC (cm) | 110.9 ± 2.5 | 109.5 ± 2.7 | 0.54 |
| REE (kcal) | 1389.0 ± 14.3 | 1447.8 ± 12.9 | 0.34 |
| RQ | 0.83 ± 0.17 | 0.83 ± 0.21 | 0.43 |
| SBP (mmHg) | 120.2 ± 3.7 | 119.1 ± 3.6 | 0.81 |
| DBP (mmHg) | 80.0 ± 3.3 | 79.5 ± 3.0 | 0.89 |
| TC (mmol/L) | 5.2 ± 1.0 | 5.2 ± 0.8 | 0.81 |
| LDLC (mmol/L) | 3.1 ± 0.9 | 3.1 ± 0.8 | 0.87 |
| HDLC (mmol/L) | 1.3 ± 0.4 | 1.4 ± 0.6 | 0.64 |
| TG (mmol/L) | 1.3 ± 0.8 | 1.2 ± 0.8 | 0.75 |
| Plasma glucose (mmol/L) | 5.6 ± 0.8 | 5.5 ± 0.6 | 0.73 |
| Serum insulin (pmol/L) | 52.7 ± 4.9 | 42.6 ± 5.8 | 0.28 |
| FFA (μmol/L) | 726.9 ± 15.9 | 619.4 ± 14.7 | 0.18 |
| Samples | Diet | Time | Diet × Time |
|---|---|---|---|
| Urine | 0.004 | 0.66 | 0.25 |
| Blood ZGPR a | 0.39 | 0.003 | 0.96 |
| Blood CPMG b | 0.89 | 0.0005 | 0.95 |
| Feces WSE c | 0.09 | 0.52 | 0.98 |
| Feces LE d | 0.06 | 0.33 | 0.87 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, H.; Lorenzen, J.K.; Astrup, A.; Larsen, L.H.; Yde, C.C.; Clausen, M.R.; Bertram, H.C. Metabolic Effects of a 24-Week Energy-Restricted Intervention Combined with Low or High Dairy Intake in Overweight Women: An NMR-Based Metabolomics Investigation. Nutrients 2016, 8, 108. https://doi.org/10.3390/nu8030108
Zheng H, Lorenzen JK, Astrup A, Larsen LH, Yde CC, Clausen MR, Bertram HC. Metabolic Effects of a 24-Week Energy-Restricted Intervention Combined with Low or High Dairy Intake in Overweight Women: An NMR-Based Metabolomics Investigation. Nutrients. 2016; 8(3):108. https://doi.org/10.3390/nu8030108
Chicago/Turabian StyleZheng, Hong, Janne K. Lorenzen, Arne Astrup, Lesli H. Larsen, Christian C. Yde, Morten R. Clausen, and Hanne Christine Bertram. 2016. "Metabolic Effects of a 24-Week Energy-Restricted Intervention Combined with Low or High Dairy Intake in Overweight Women: An NMR-Based Metabolomics Investigation" Nutrients 8, no. 3: 108. https://doi.org/10.3390/nu8030108
APA StyleZheng, H., Lorenzen, J. K., Astrup, A., Larsen, L. H., Yde, C. C., Clausen, M. R., & Bertram, H. C. (2016). Metabolic Effects of a 24-Week Energy-Restricted Intervention Combined with Low or High Dairy Intake in Overweight Women: An NMR-Based Metabolomics Investigation. Nutrients, 8(3), 108. https://doi.org/10.3390/nu8030108






