Potential of Lactobacillus plantarum CCFM639 in Protecting against Aluminum Toxicity Mediated by Intestinal Barrier Function and Oxidative Stress
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Culture Conditions
2.2. Cell Culture Experiment
2.3. Animals and Experimental Design
2.4. Measurement of Cytotoxicity
2.5. Measurement of Tight Junction Proteins in HT-29 Cell Line
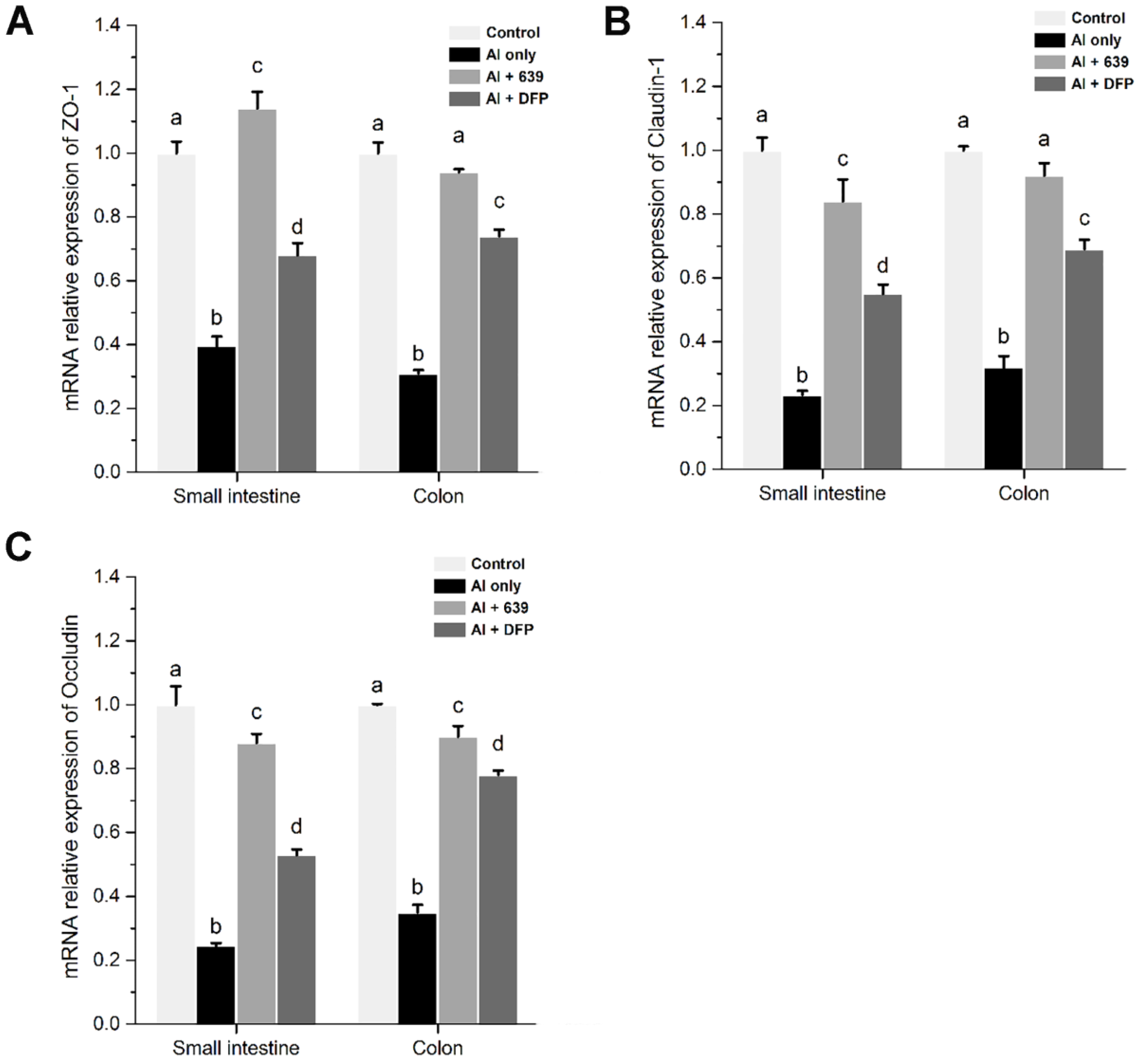
2.6. Measurement of Tight Junction Proteins in Mice Intestinal Tissues
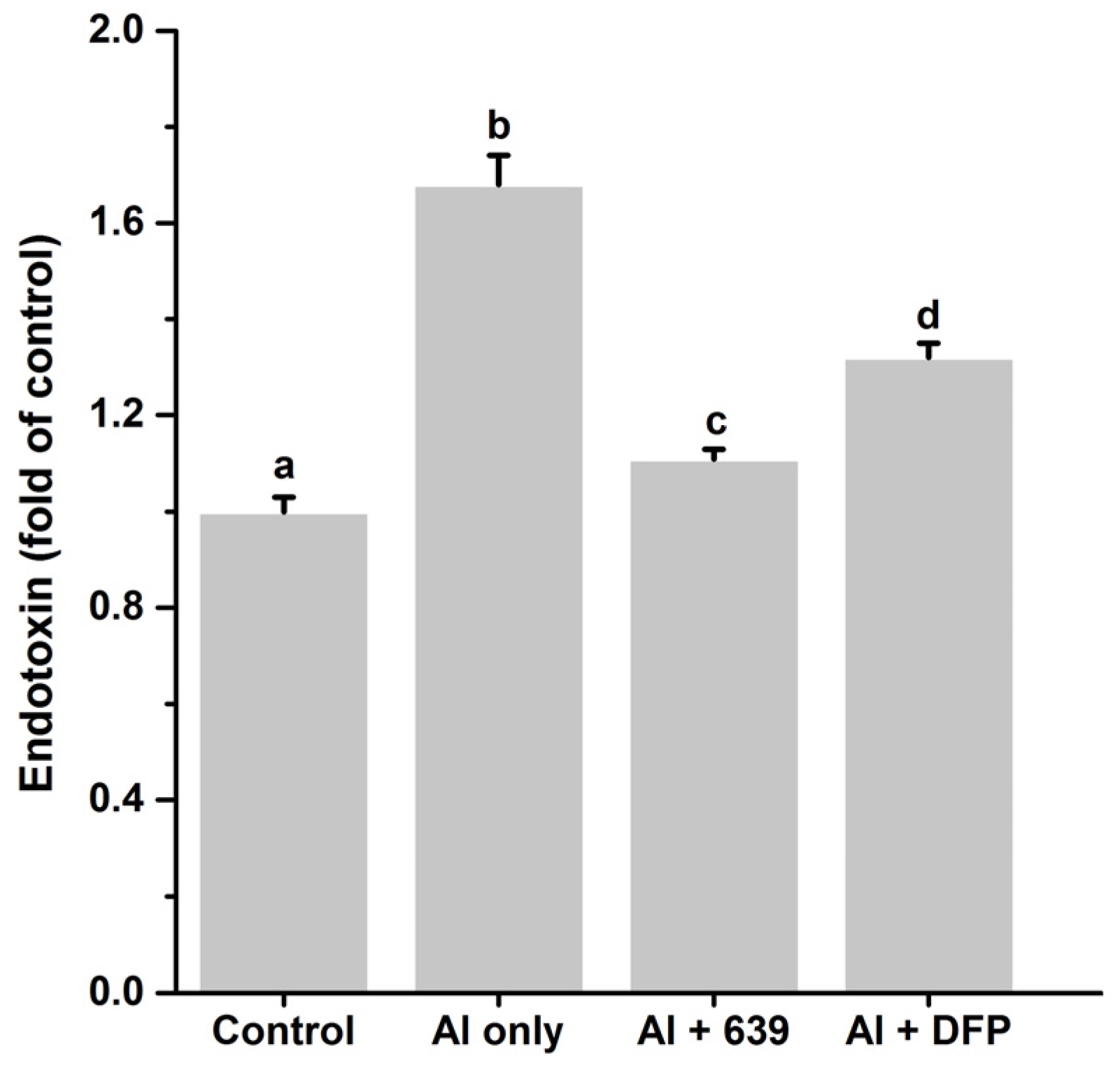
2.7. Measurement of Intestinal Permeability of Mice
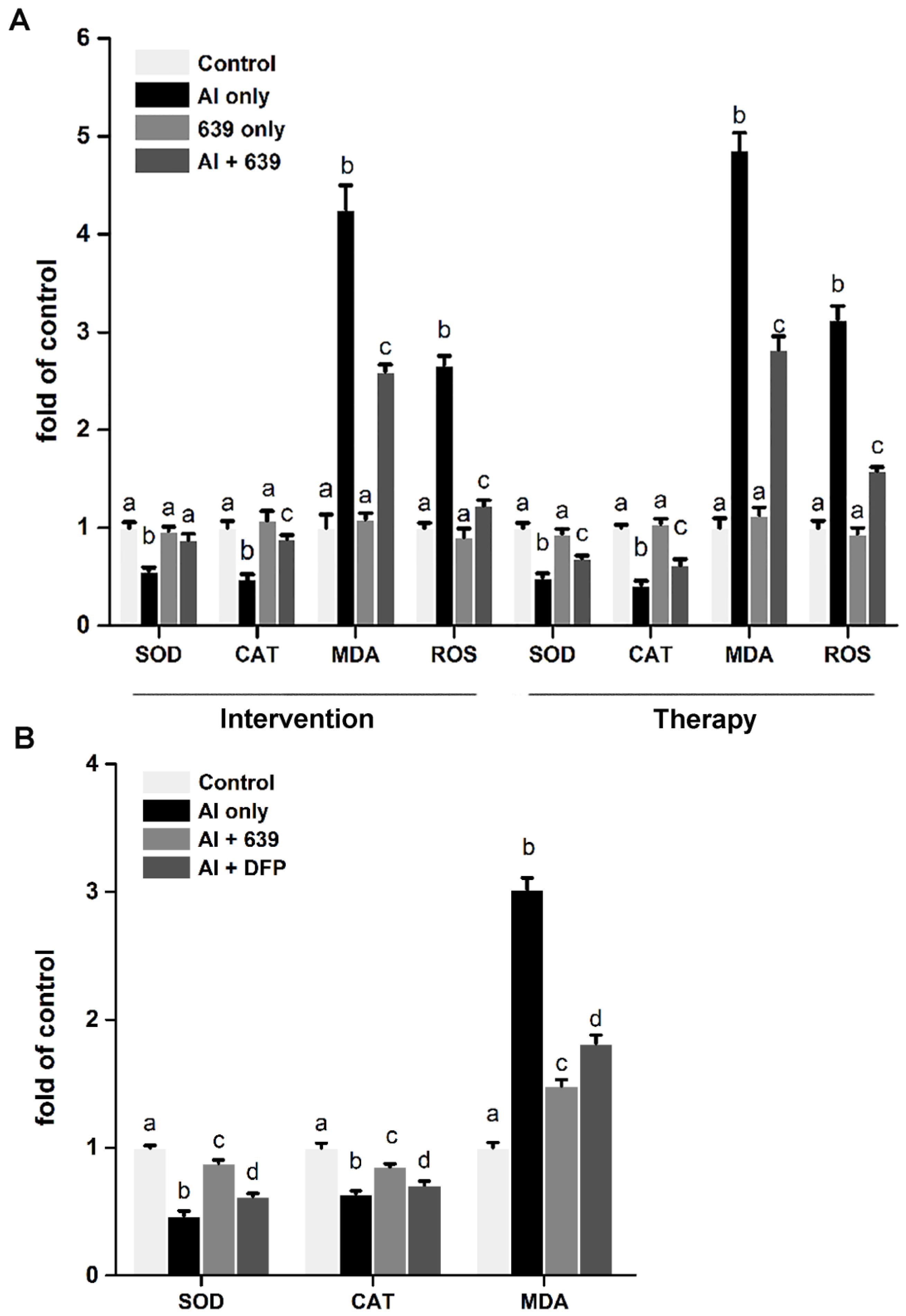
2.8. Measurement of Oxidative-Stress-Related Parameters in HT-29 Cell Cultures and Serums
2.9. Measurement of Pro-Inflammatory Cytokines in HT-29 Cell Cultures and Intestinal Tissues
2.10. Statistical Analysis
3. Results
3.1. Effects of L. plantarum CCFM639 on Al-Induced Cytotoxicity
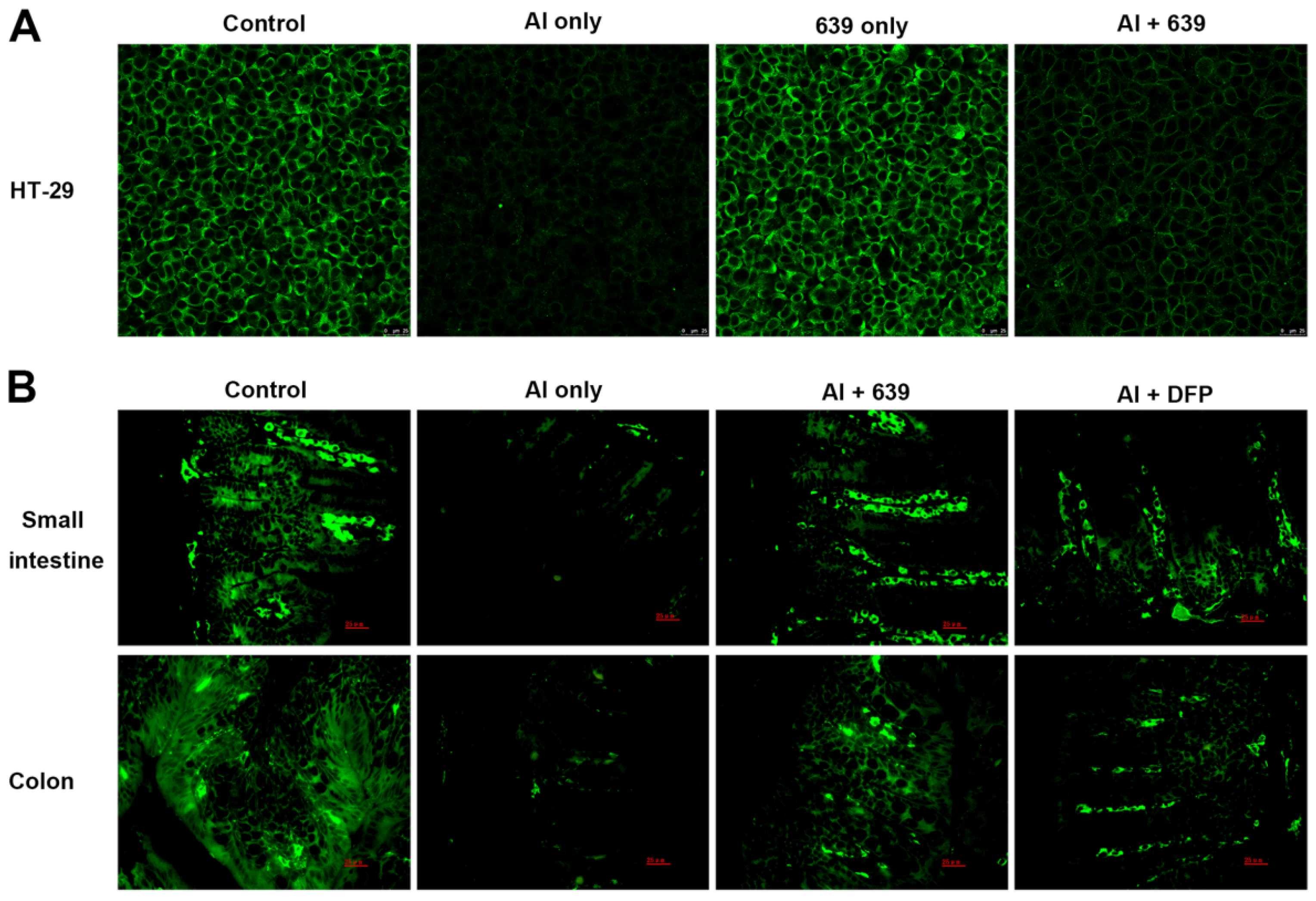
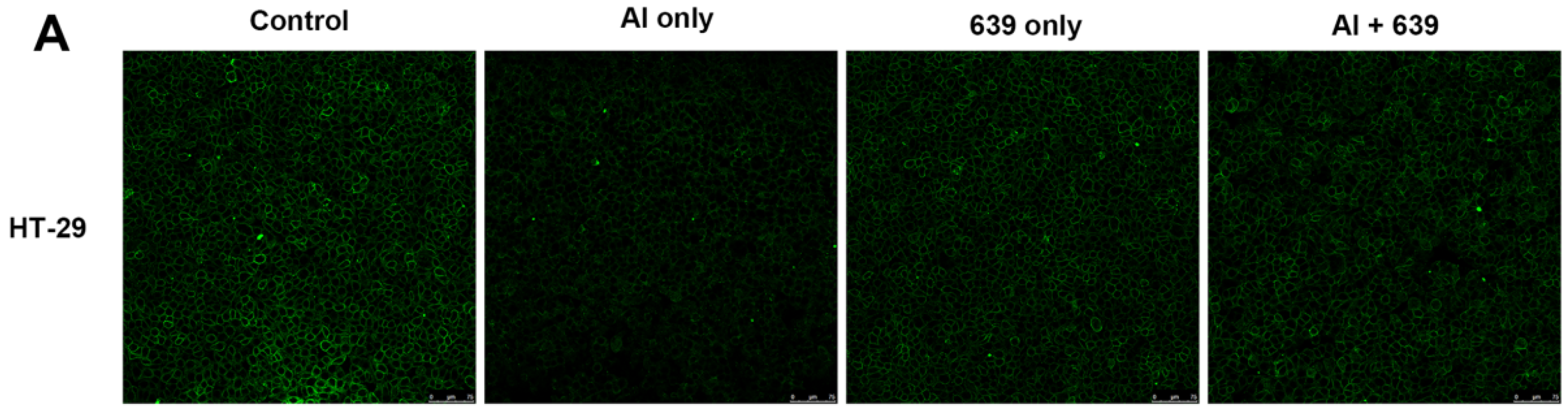
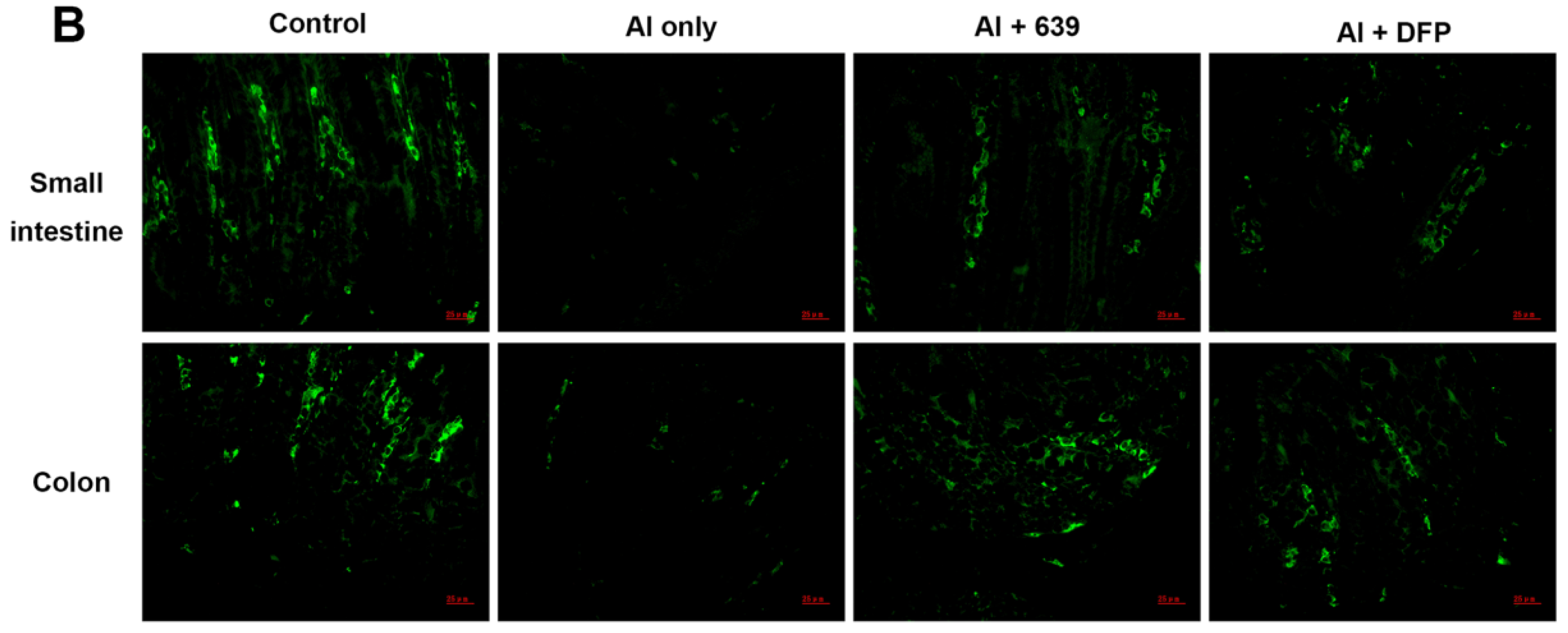
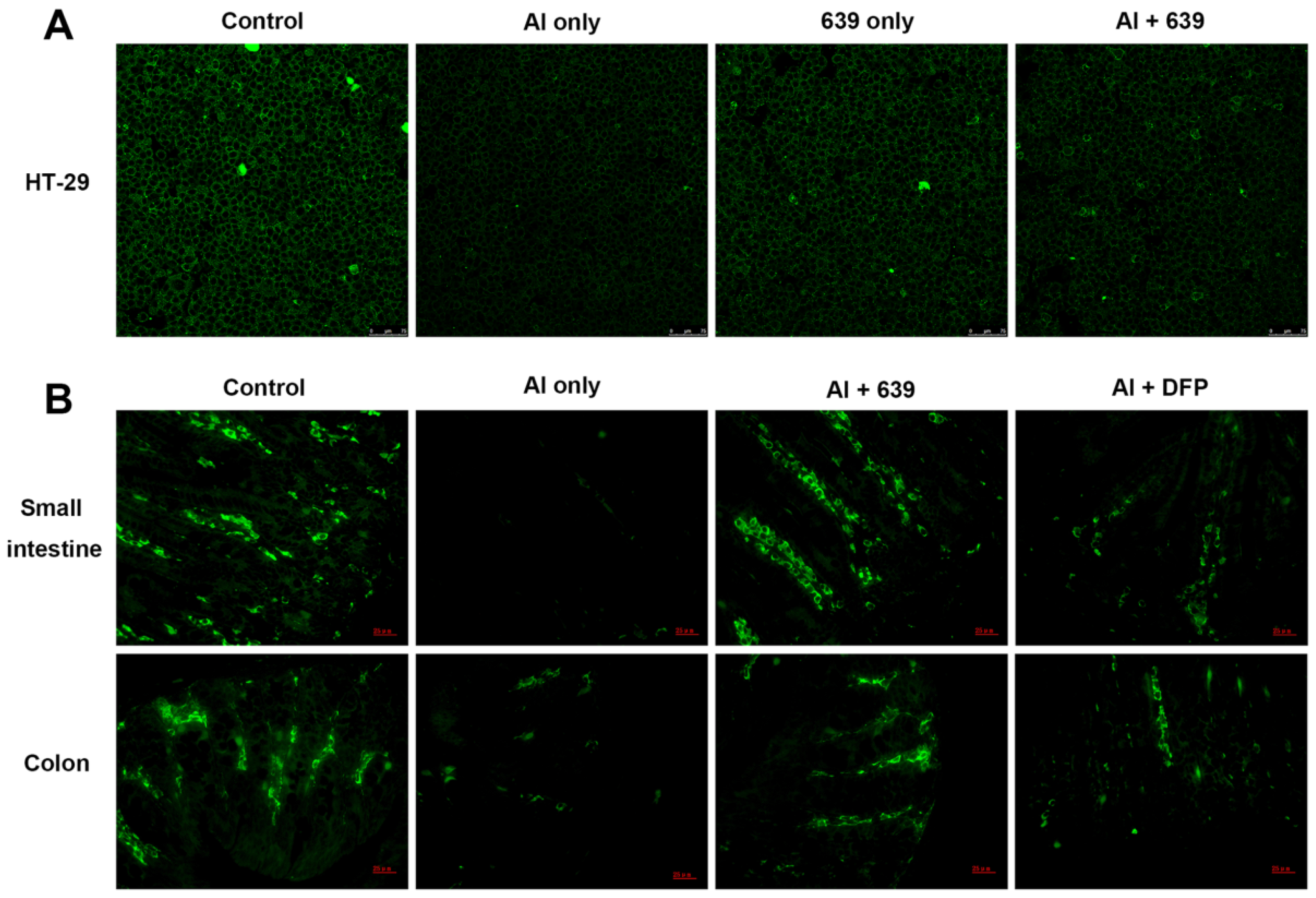
3.2. Effects of L. plantarum CCFM639 on Al-Induced Alterations of Tight Junction Proteins
3.3. Effects of L. plantarum CCFM639 on Gut Permeability
3.4. Effects of L. plantarum CCFM639 on Al-Induced Oxidative Stress In Vitro and In Vivo
3.5. Effects of L. plantarum CCFM639 on Al-Induced Alterations of Pro-Inflammatory Cytokines In Vitro and In Vivo
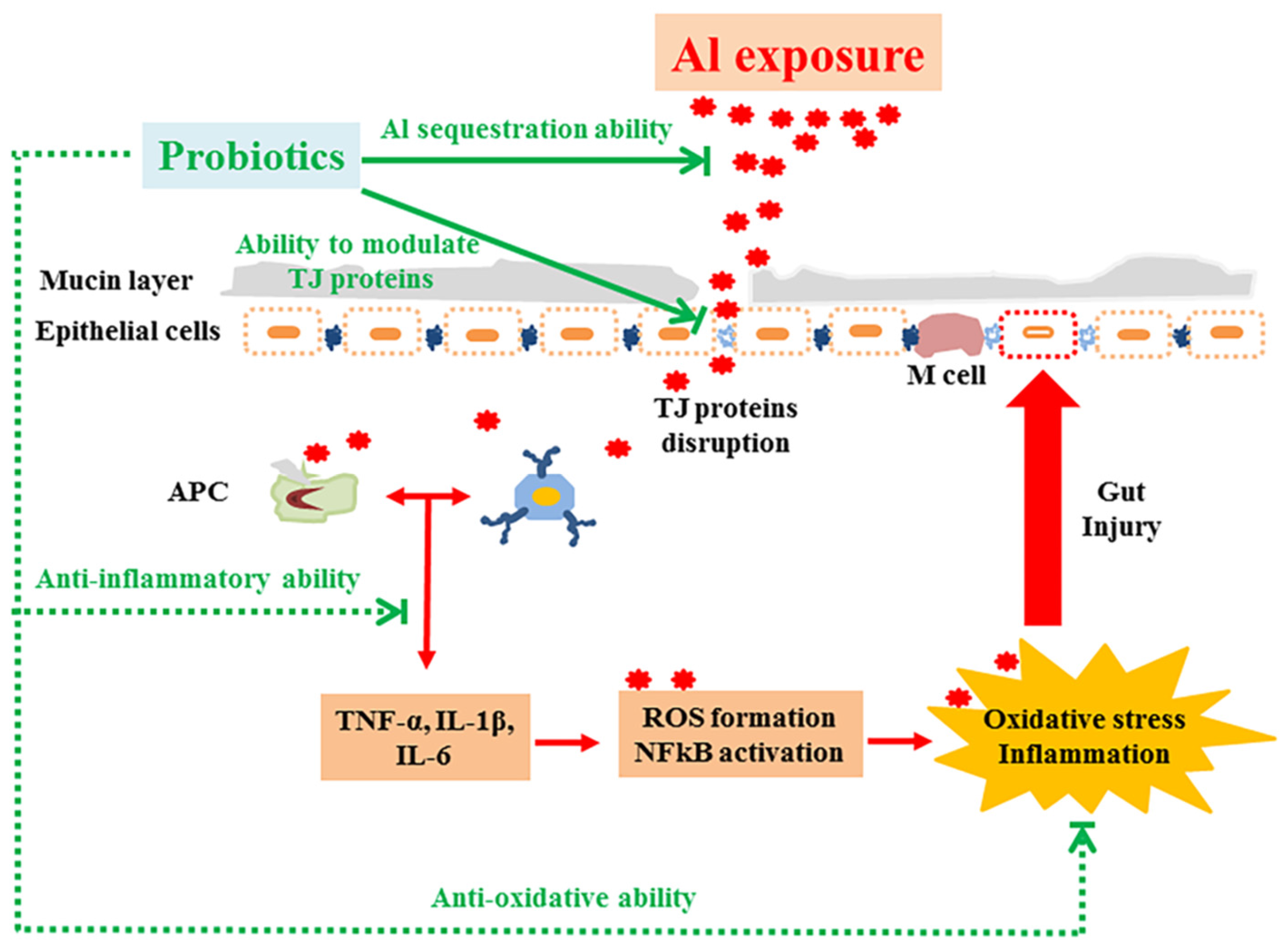
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Nayak, P. Aluminum: Impacts and disease. Environ. Res. 2002, 89, 101–115. [Google Scholar] [CrossRef] [PubMed]
- De Chambrun, G.P.; Body-Malapel, M.; Frey-Wagner, I.; Djouina, M.; Deknuydt, F.; Atrott, K.; Vignal, C. Aluminum enhances inflammation and decreases mucosal healing in experimental colitis in mice. Mucosal Immunol. 2014, 7, 589–601. [Google Scholar] [CrossRef] [PubMed]
- Vignal, C.; Desreumaux, P.; Body-Malapel, M. Gut: An underestimated target organ for Aluminum. Morphologie 2016, 100, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, F.; Autrup, H.; Barlow, S.; Castle, L.; Crebelli, R.; Dekant, W.; Engel, K.-H.; Gontard, N.; Gott, D.; Grilli, S.; et al. Safety of aluminium from dietary intake—Scientific Opinion of the Panel on Food Additives, Flavourings, Processing Aids and Food Contact Materials (AFC). EFSA J. 2008, 754, 1–34. [Google Scholar]
- Becaria, A.; Campbell, A.; Bondy, S.C. Aluminum as a toxicant. Toxicol. Ind. Health 2002, 18, 309–320. [Google Scholar] [CrossRef] [PubMed]
- Cunat, L.; Lanhers, M.C.; Joyeux, M.; Burnel, D. Bioavailability and intestinal absorption of aluminum in rats—Effects of aluminum compounds and some dietary constituents. Biol. Trace Elem. Res. 2000, 76, 31–55. [Google Scholar] [CrossRef]
- Powell, J.J.; Ainley, C.C.; Evans, R.; Thompson, R.P. Intestinal perfusion of dietary levels of aluminium: Association with the mucosa. Gut 1994, 35, 1053–1057. [Google Scholar] [CrossRef] [PubMed]
- Moeser, A.J.; Klok, C.V.; Ryan, K.A.; Wooten, J.G.; Little, D.; Cook, V.L.; Blikslager, A.T. Stress signaling pathways activated by weaning mediate intestinal dysfunction in the pig. Am. J. Physiol. Gastrointest. Liver Physiol. 2007, 292, G173–G181. [Google Scholar] [CrossRef] [PubMed]
- Sharma, D.R.; Wani, W.Y.; Sunkaria, A.; Kandimalla, R.J.L.; Verma, D.; Cameotra, S.S.; Gill, K.D. Quercetin Protects Against Chronic Aluminum-Induced Oxidative Stress and Ensuing Biochemical, Cholinergic, and Neurobehavioral Impairments in Rats. Neurotox. Res. 2013, 23, 336–357. [Google Scholar] [CrossRef] [PubMed]
- Menard, S.; Candalh, C.; Bambou, J.C.; Terpend, K.; Cerf-Bensussan, N.; Heyman, M. Lactic acid bacteria secrete metabolites retaining anti-inflammatory properties after intestinal transport. Gut 2004, 53, 821–828. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Q.; Tian, F.; Zhao, J.; Zhang, H.; Narbad, A.; Chen, W. Oral administration of probiotics inhibits heavy metal cadmium absorption by protecting intestinal barrier. Appl. Environ. Microbiol. 2016, 82, 4429–4440. [Google Scholar] [CrossRef] [PubMed]
- Tian, F.; Zhai, Q.; Zhao, J.; Liu, X.; Wang, G.; Zhang, H.; Chen, W. Lactobacillus plantarum CCFM8661 alleviates lead toxicity in mice. Biolog. Trace Elem. Res. 2012, 150, 264–271. [Google Scholar] [CrossRef] [PubMed]
- Shimazu, T.; Villena, J.; Tohno, M.; Fujie, H.; Hosoya, S.; Shimosato, T.; Kitazawa, H. Immunobiotic Lactobacillus jensenii elicits anti-inflammatory activity in porcine intestinal epithelial cells by modulating negative regulators of the Toll-like receptor signaling pathway. Infect. Immun. 2012, 80, 276–288. [Google Scholar] [CrossRef] [PubMed]
- Karczewski, J.; Troost, F.J.; Konings, I.; Dekker, J.; Kleerebezem, M.; Brummer, R.J.; Wells, J.M. Regulation of human epithelial tight junction proteins by Lactobacillus plantarum in vivo and protective effects on the epithelial barrier. Am. J. Physiol. Gastrointest. Liver Physiol. 2010, 298, G851–G859. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Wang, A.; Zeng, X.; Hou, C.; Liu, H.; Qiao, S. Lactobacillus reuteri I5007 modulates tight junction protein expression in IPEC-J2 cells with LPS stimulation and in newborn piglets under normal conditions. BMC Microbiol. 2015, 15. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.-Y.; Roos, S.; Jonsson, H.; Ahl, D.; Dicksved, J.; Lindberg, J.E.; Lundh, T. Effects of Lactobacillus johnsonii and Lactobacillus reuteri on gut barrier function and heat shock proteins in intestinal porcine epithelial cells. Physiol. Rep. 2015, 3. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Zhai, Q.; Liu, X.; Wang, G.; Zhang, Q.; Zhao, J.; Narbad, A.; Zhang, H.; Tian, F.; Chen, W. Lactobacillus plantarum CCFM639 alleviates aluminium toxicity. Appl. Microbiol. Biotechnol. 2016, 100, 1891–1900. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Zhai, Q.; Yin, R.; Li, P.; Tian, F.; Liu, X.; Zhao, J.; Gong, J.; Zhang, H.; Chen, W. Lactobacillus plantarum CCFM639 alleviate trace element imbalance-related oxidative stress in liver and kidney of chronic aluminum exposure mice. Biol. Trace Elem. Res. 2016. [Google Scholar] [CrossRef] [PubMed]
- Ghouri, Y.A.; Richards, D.M.; Rahimi, E.F.; Krill, J.T.; Jelinek, K.A.; DuPont, A.W. Systematic review of randomized controlled trials of probiotics, prebiotics, and synbiotics in inflammatory bowel disease. Clin. Exp. Gastroenterol. 2014, 7, 473–487. [Google Scholar] [PubMed]
- Lin, M.Y.; Yen, C.L. Antioxidative ability of lactic acid bacteria. J. Agric. Food Chem. 1999, 47, 1460–1466. [Google Scholar] [CrossRef] [PubMed]
- Echabaane, M.; Rouis, A.; Bonnamour, I.; Ouada, H.B. Studies of aluminum (III) ion-selective optical sensor based on a chromogenic calix[4]arene derivative. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2013, 115, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Prakash, D.; Gopinath, K.; Sudhandiran, G. Fisetin enhances behavioral performances and attenuates reactive gliosis and inflammation during aluminum chloride-induced neurotoxicity. Neuromol. Med. 2013, 15, 192–208. [Google Scholar] [CrossRef] [PubMed]
- Johnson, V.J.; Kim, S.H.; Sharma, R.P. Aluminum-maltolate induces apoptosis and necrosis in neuro-2a cells: Potential role for p53 signaling. Toxicol. Sci. 2005, 83, 329–339. [Google Scholar] [CrossRef] [PubMed]
- Braniste, V.; Al-Asmakh, M.; Kowal, C.; Anuar, F.; Abbaspour, A.; Toth, M.; Pettersson, S. The gut microbiota influences blood-brain barrier permeability in mice. Sci. Transl. Med. 2014, 6, 263. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, J.; Mizukure, T.; Park, S.B.; Kishino, S.; Kimura, I.; Hirano, K.; Tanabe, S.A. gut microbial metabolite of linoleic acid, 10-hydroxy-cis-12-octadecenoic acid, ameliorates intestinal epithelial barrier impairment partially via GPR40-MEK-ERK pathway. J. Biol. Chem. 2015, 290, 2902–2918. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Possemiers, S.; de Wiele, T.V.; Guiot, Y.; Everard, A.; Rottier, O.; Delzenne, N.M. Changes in gut microbiota control inflammation in obese mice through a mechanism involving GLP-2-driven improvement of gut permeability. Gut 2009, 58, 1091–1103. [Google Scholar] [CrossRef] [PubMed]
- Campbell, D.I.; Elia, M.; Lunn, P.G. Growth faltering in rural Gambian infants is associated with impaired small intestinal barrier function, leading to endotoxemia and systemic inflammation. J. Nutr. 2003, 133, 1332–1338. [Google Scholar] [PubMed]
- Ward, R.J.; Zhang, Y.; Crichton, R.R. Aluminium toxicity and iron homeostasis. J. Inorg. Biochem. 2001, 87, 9–14. [Google Scholar] [CrossRef]
- Zatta, P.; Kiss, T.; Suwalsky, M.; Berthon, G. Aluminium(III) as a promoter of cellular oxidation. Coord. Chem. Rev. 2002, 228, 271–284. [Google Scholar] [CrossRef]
- Gomes, A.C.; Bueno, A.A.; de Souza, R.G.M.; Mota, J.F. Gut microbiota, probiotics and diabetes. Nutr. J. 2014, 13, 60. [Google Scholar] [CrossRef] [PubMed]
- Andersson, U.; Bränning, C.; Ahrné, S.; Molin, G.; Alenfall, J.; Önning, G.; Nyman, M.; Holm, C. Probiotics lower plasma glucose in the high-fat fed C57BL/6J mouse. Benef. Microbes 2010, 1, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Moroti, C.; Magri, L.F.S.; de Rezende Costa, M.; Cavallini, D.C.; Sivieri, K. Effect of the consumption of a new symbiotic shake on glycemia and cholesterol levels in elderly people with type 2 diabetes mellitus. Lipids Health Dis. 2012, 11, 1. [Google Scholar] [CrossRef] [PubMed]
- Greger, J.L. Aluminum metabolism. Annu. Rev. Nutr. 1993, 13, 43–63. [Google Scholar] [CrossRef] [PubMed]
- Greger, J.L.; Sutherland, J.E. Aluminum exposure and metabolism. Crit. Rev. Clin. Lab. Sci. 1997, 34, 439–474. [Google Scholar] [CrossRef] [PubMed]
- Exley, C.; Burgess, E.; Day, J.P.; Jeffery, E.H.; Melethil, S.; Yokel, R.A. Aluminum toxicokinetics. J. Toxicol. Environ. Health 1996, 48, 569–584. [Google Scholar] [PubMed]
- Miyauchi, E.; O’Callaghan, J.; Butto, L.F.; Hurley, G.; Melgar, S.; Tanabe, S.; O’Toole, P.W. Mechanism of protection of transepithelial barrier function by Lactobacillus salivarius: Strain dependence and attenuation by bacteriocin production. Am. J. Physiol. Gastrointest. Liver Physiol. 2012, 303, G1029–G1041. [Google Scholar] [CrossRef] [PubMed]
- Seth, A.; Yan, F.; Polk, D.B.; Rao, R. Probiotics ameliorate the hydrogen peroxide-induced epithelial barrier disruption by a PKC-and MAP kinase-dependent mechanism. Am. J. Physiol. Gastrointest. Liver Physiol. 2008, 294, G1060–G1069. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Q.; Narbad, A.; Chen, W. Dietary Strategies for the Treatment of Cadmium and Lead Toxicity. Nutrients 2015, 7, 552–571. [Google Scholar] [CrossRef] [PubMed]
- Chow, J. Probiotics and prebiotics: A brief overview. J. Ren. Nutr. 2002, 12, 76–86. [Google Scholar] [CrossRef] [PubMed]
- Dicksved, J.; Schreiber, O.; Willing, B.; Petersson, J.; Rang, S.; Phillipson, M.; Roos, S. Lactobacillus reuteri maintains a functional mucosal barrier during DSS treatment despite mucus layer dysfunction. PLoS ONE 2012, 7, e46399. [Google Scholar] [CrossRef] [PubMed]
- Aspenstrom-Fagerlund, B.; Sundstrom, B.; Tallkvist, J.; Ilback, N.G.; Glynn, A.W. Fatty acids increase paracellular absorption of aluminium across Caco-2 cell monolayers. Chem. Biol. Interact. 2009, 181, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Exley, C.; Mold, M.J. The binding, transport and fate of aluminium in biological cells. J. Trace Elem. Med. Biol. 2014, 30, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Zalups, R.K.; Ahmad, S. Molecular handling of cadmium in transporting epithelia. Toxicol. Appl. Pharmacol. 2003, 186, 163–188. [Google Scholar] [CrossRef]
- Sung, M.K.; Park, M.Y. Nutritional modulators of ulcerative colitis: Clinical efficacies and mechanistic view. World J. Gastroenterol. 2013, 19, 994–1004. [Google Scholar] [CrossRef] [PubMed]
- Farhadi, A.; Keshavarzian, A.; Ranjbaran, Z.; Fields, J.Z.; Banan, A. The role of protein kinase C isoforms in modulating injury and repair of the intestinal barrier. J. Pharmacol. Exp. Ther. 2006, 316, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Yan, F.; Cao, H.; Cover, T.L.; Whitehead, R.; Washington, M.K.; Polk, D.B. Soluble proteins produced by probiotic bacteria regulate intestinal epithelial cell survival and growth. Gastroenterology 2007, 132, 562–575. [Google Scholar] [CrossRef] [PubMed]
- Banan, A.; Keshavarzian, A.; Zhang, L.; Shaikh, M.; Forsyth, C.; Tang, Y.; Fields, J. NF-κB activation as a key mechanism in ethanol-induced disruption of the F-actin cytoskeleton and monolayer barrier integrity in intestinal epithelium. Alcohol 2007, 41, 447–460. [Google Scholar] [CrossRef] [PubMed]
- O’Hara, A.M.; Shanahan, F. The gut flora as a forgotten organ. EMBO Rep. 2006, 7, 688–693. [Google Scholar] [CrossRef] [PubMed]
- Foligné, B.; Daniel, C.; Pot, B. Probiotics from research to market: The possibilities, risks and challenges. Curr. Opin. Microbiol. 2013, 16, 284–292. [Google Scholar] [CrossRef] [PubMed]
| Cell Experiment | Animal Experiment | |||||
|---|---|---|---|---|---|---|
| Group | Intervention Assay | Therapy Assay | Group | Intervention Assay | ||
| 0–24 h | 0–24 h | 24–48 h | 0–8 Weeks | 8–14 Weeks | ||
| Control | Medium | Medium | Medium | Control | SM + PW | SM + PW |
| Al only | Medium + Al | Medium + Al | Medium | Al only | SM + Al | SM + PW |
| 639 only | Medium + 639 | Medium | Medium + 639 | Al + 639 | SM + Al + 639 | SM + PW + 639 |
| Al + 639 | Medium + Al + 639 | Medium + Al | Medium + 639 | Al + DFP | SM + Al + DFP | PW + DFP |
| Group | TNF-α (pg/mL) | IL-1β (pg/mL) | IL-6 (pg/mL) | |||
|---|---|---|---|---|---|---|
| Intervention | Therapy | Intervention | Therapy | Intervention | Therapy | |
| Control | 25.12 ± 2.14 a | 27.82 ± 1.46 a | 6.90 ± 0.45 a | 6.12 ± 0.56 a | 39.49 ± 0.71 a | 40.04 ± 1.18 a |
| Al only | 43.66 ± 2.91 b | 49.56 ± 4.37 b | 13.23 ± 1.23 b | 14.69 ± 1.94 b | 52.49 ± 1.58 b | 55.82 ± 2.16 b |
| 639 only | 23.84 ± 1.78 a | 29.50 ± 1.81 a | 6.46 ± 0.78 a | 6.89 ± 0.23 a | 37.80 ± 1.63 a | 41.12 ± 1.35 a |
| Al + 639 | 30.20 ± 3.73 a | 37.08 ± 2.35 c | 9.39 ± 0.86 c | 11.16 ± 1.05 c | 43.66 ± 0.88 c | 47.61 ± 1.03 c |
| Group | Mean Level (pg/mL) | ||
|---|---|---|---|
| TNF-α | IL-1β | IL-6 | |
| small intestine | |||
| Control | 897.35 ± 51.33 a | 45.37 ± 4.33 a | 133.23 ± 3.36 a |
| Al only | 1577.37 ± 143.73 b | 116.48 ± 8.53 b | 169.34 ± 9.53 b |
| Al + 639 | 1125.26 ± 63.36 c | 83.37 ± 3.07 c | 138.71 ± 5.07 a |
| Al + DFP | 1199.36 ± 54.38 c | 92.16 ± 4.33 d | 160.25 ± 7.13 b |
| colon | |||
| Control | 881.23 ± 25.19 A | 44.73 ± 2.19 A | 114.35 ± 5.19 A |
| Al only | 1446.78 ± 90.59 B | 133.47 ± 5.71 B | 148.37 ± 10.71 B |
| Al + 639 | 1083.19 ± 59.08 C | 66.36 ± 6.32 C | 136.74 ± 4.32 B |
| Al + DFP | 1304.37 ± 33.21 D | 96.32 ± 3.41 D | 145.28 ± 6.41 B |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, L.; Zhai, Q.; Tian, F.; Liu, X.; Wang, G.; Zhao, J.; Zhang, H.; Narbad, A.; Chen, W. Potential of Lactobacillus plantarum CCFM639 in Protecting against Aluminum Toxicity Mediated by Intestinal Barrier Function and Oxidative Stress. Nutrients 2016, 8, 783. https://doi.org/10.3390/nu8120783
Yu L, Zhai Q, Tian F, Liu X, Wang G, Zhao J, Zhang H, Narbad A, Chen W. Potential of Lactobacillus plantarum CCFM639 in Protecting against Aluminum Toxicity Mediated by Intestinal Barrier Function and Oxidative Stress. Nutrients. 2016; 8(12):783. https://doi.org/10.3390/nu8120783
Chicago/Turabian StyleYu, Leilei, Qixiao Zhai, Fengwei Tian, Xiaoming Liu, Gang Wang, Jianxin Zhao, Hao Zhang, Arjan Narbad, and Wei Chen. 2016. "Potential of Lactobacillus plantarum CCFM639 in Protecting against Aluminum Toxicity Mediated by Intestinal Barrier Function and Oxidative Stress" Nutrients 8, no. 12: 783. https://doi.org/10.3390/nu8120783
APA StyleYu, L., Zhai, Q., Tian, F., Liu, X., Wang, G., Zhao, J., Zhang, H., Narbad, A., & Chen, W. (2016). Potential of Lactobacillus plantarum CCFM639 in Protecting against Aluminum Toxicity Mediated by Intestinal Barrier Function and Oxidative Stress. Nutrients, 8(12), 783. https://doi.org/10.3390/nu8120783





