Probiotic Soy Product Supplemented with Isoflavones Improves the Lipid Profile of Moderately Hypercholesterolemic Men: A Randomized Controlled Trial
Abstract
:1. Introduction
2. Experimental Section
2.1. Subjects and Study Design
2.2. Experimental Design and Diets
| Composition in 100 g of Soy Product | ISP | SP | USP |
|---|---|---|---|
| Protein (g) | 3.85 ± 0.00 a | 3.90 ± 0.00 a | 3.85 ± 0.04 a |
| Fat (g) | 2.26 ± 0.09 a | 2.30 ± 0.08 a | 2.32 ± 0.02 a |
| Carbohydrate (g) | 10.06 ± 0.11 a | 9.70 ± 0.15 a | 9.93 ± 0.16 a |
| Ash (g) | 0.90 ± 0.00 a | 0.90 ± 0.07 a | 0.90 ± 0.00 a |
| Moisture (g) | 82.93 ± 0.17 a | 83.20 ± 0.00 a | 83.00 ± 0.16 a |
| Isoflavones (mg) | 51.26 ± 1.12 a | 8.04 ± 0.01 b | 8.03 ± 0.07 b |
2.3. Data Collection
2.4. Lipid Profile
2.5. LDL (‒) and Anti-LDL (‒) Autoantibodies
2.6. C-Reactive Protein (CRP) and Fibrinogen
2.7. Isoflavone Profile
2.8. Gastrointestinal Resistance of E. faecium
2.9. Statistical Analysis
3. Results
3.1. Study Population
| Clinical Characteristics | Groups | ||
|---|---|---|---|
| ISP (n = 17) | SP (n = 17) | USP (n = 15) | |
| Age (y) | 48.1 ± 5.1 a | 46.1 ± 6.1 a | 45.4 ± 5.1 a |
| Height (m) | 1.73 ± 0.08 a | 1.75 ± 0.06 a | 1.72 ± 0.07 a |
| Weight (kg) | 76.7 ± 14.5 a | 81.6 ± 15.6 a | 78.0 ± 14.1 a |
| BMI (kg/m2) | 25.65 ± 4.36 a | 26.41 ± 4.88 a | 26.26 ± 3.98 a |
| WHR | 0.91 ± 0.06 a | 0.90 ± 0.07 a | 0.90 ± 0.05 a |
| Diabetes | 0 | 0 | 0 |
| Hypertension | 1 (5.9%) | 1 (5.9%) | 0 |
| Gastrointestinal diseases | 1 (5.9%) | 1 (5.9%) | 0 |
| Previous CVD | 0 | 0 | 0 |
| Smoking | 3 (17.6%) | 2 (11.8%) | 3 (20.0%) |
3.2. Lipid Profile
| Time | ISP (n = 17) | SP (n = 17) | USP (n = 15) |
|---|---|---|---|
| TC (mmol/L) | |||
| T0 | 5.47 ± 0.59 a,A | 5.76 ± 1.02 a,A | 5.60 ± 0.76 a,A |
| T30 | 4.65 ± 0.48 b,B | 5.44 ± 0.93 a,A | 5.52 ± 0.76 a,A |
| T42 | 4.72 ± 0.45 b,B | 5.41 ± 0.86 a,A | 5.50 ± 0.78 a,A |
| HDL-C (mmol/L) | |||
| T0 | 1.36 ± 0.22 a,A | 1.38 ± 0.25 a,A | 1.50 ± 0.28 a,A |
| T30 | 1.18 ± 0.23 a,A | 1.33 ± 0.19 a,A | 1.24 ± 0.38 a,A,B |
| T42 | 1.21 ± 0.22 a,A | 1.28 ± 0.18 a,A | 1.13 ± 0.38 a,B |
| LDL-C (mmol/L) | |||
| T0 | 3.23 ± 0.61 a,A | 3.54 ± 0.92 a,A | 3.14 ± 0.95 a,A |
| T30 | 2.75± 0.51 a,A | 3.33 ± 0.84 a,A | 3.41 ± 0.60 a,A |
| T42 | 2.79 ± 0.50 b,A | 3.35 ± 0.80 a,b,A | 3.51 ± 0.63 a,A |
| TG (mmol/L) | |||
| T0 | 1.91 ± 0.98 a,A | 1.88 ± 0.88 a,A | 1.97 ± 1.32 a,A |
| T30 | 1.57 ± 0.74 a,A | 1.83 ± 0.89 a,A | 2.18 ± 1.61 a,A |
| T42 | 1.55 ± 0.70 a,A | 1.86 ± 0.93 a,A | 2.15 ± 1.64 a,A |
| nHDL-C (mmol/L) | |||
| T0 | 4.11 ± 0.65 a,A | 4.38 ± 1.12 a,A | 4.08 ± 0.82 a,A |
| T30 | 3.47 ± 0.48 a,B | 4.11 ± 0.98 a,A | 4.28 ± 0.99 a,A |
| T42 | 3.50 ± 0.46 b,B | 4.14 ± 0.92 a,b,A | 4.37 ± 0.96 a,A |
| TC/HDL-C | |||
| T0 | 4.11 ± 0.78 a,A | 4.28 ± 0.90 a,A | 3.80 ± 0.83 a,A |
| T30 | 4.07 ± 0.75 a,A | 4.18 ± 0.90 a,A | 4.92 ± 1.69 a,A |
| T42 | 4.00 ± 0.71 b,A | 4.32 ± 0.83 a,b,A | 5.47 ± 1.98 a,A |
3.3. LDL (‒) and Anti-LDL (‒) Autoantibodies
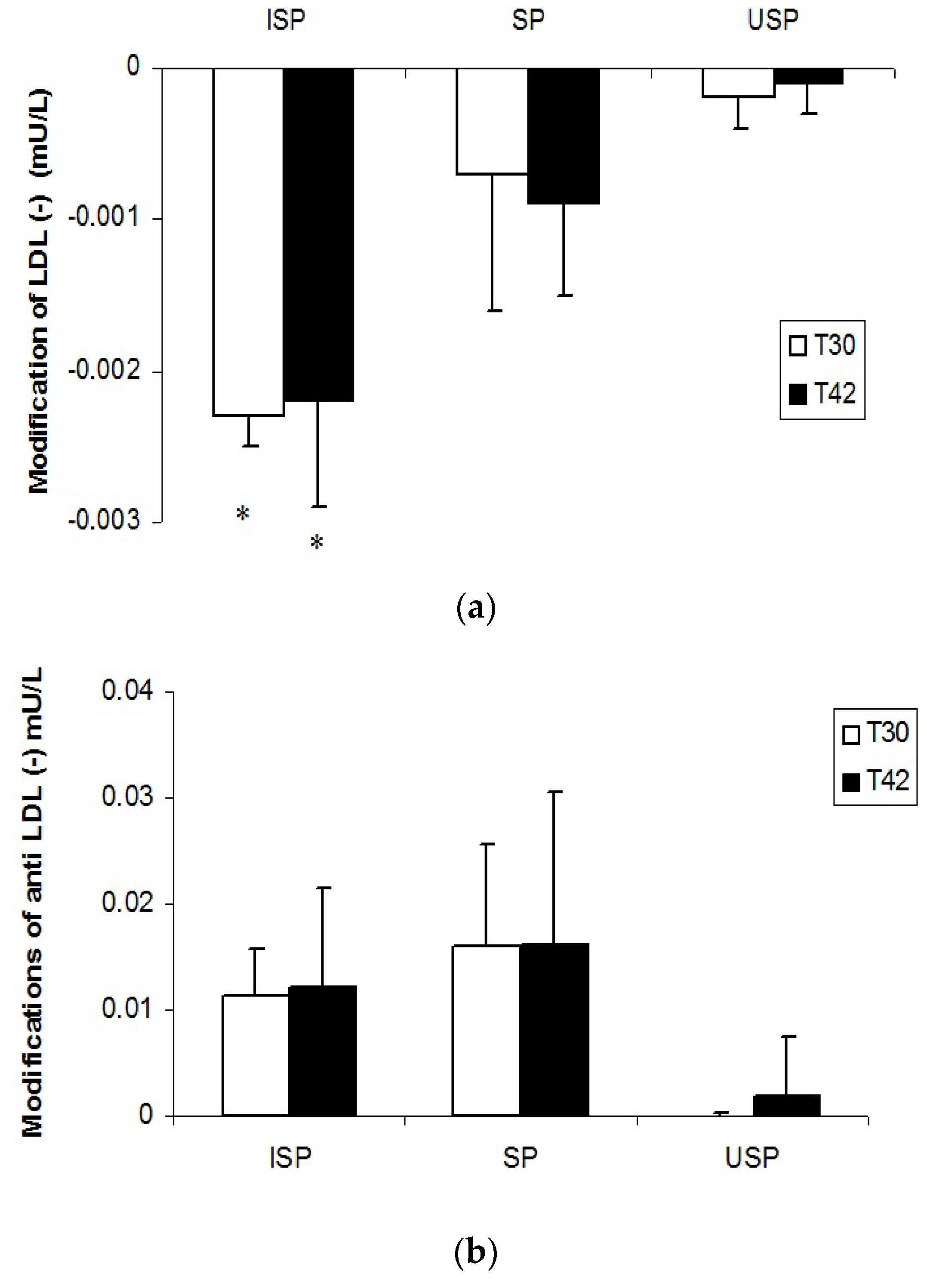
3.4. Fibrinogen and C-Reactive Protein
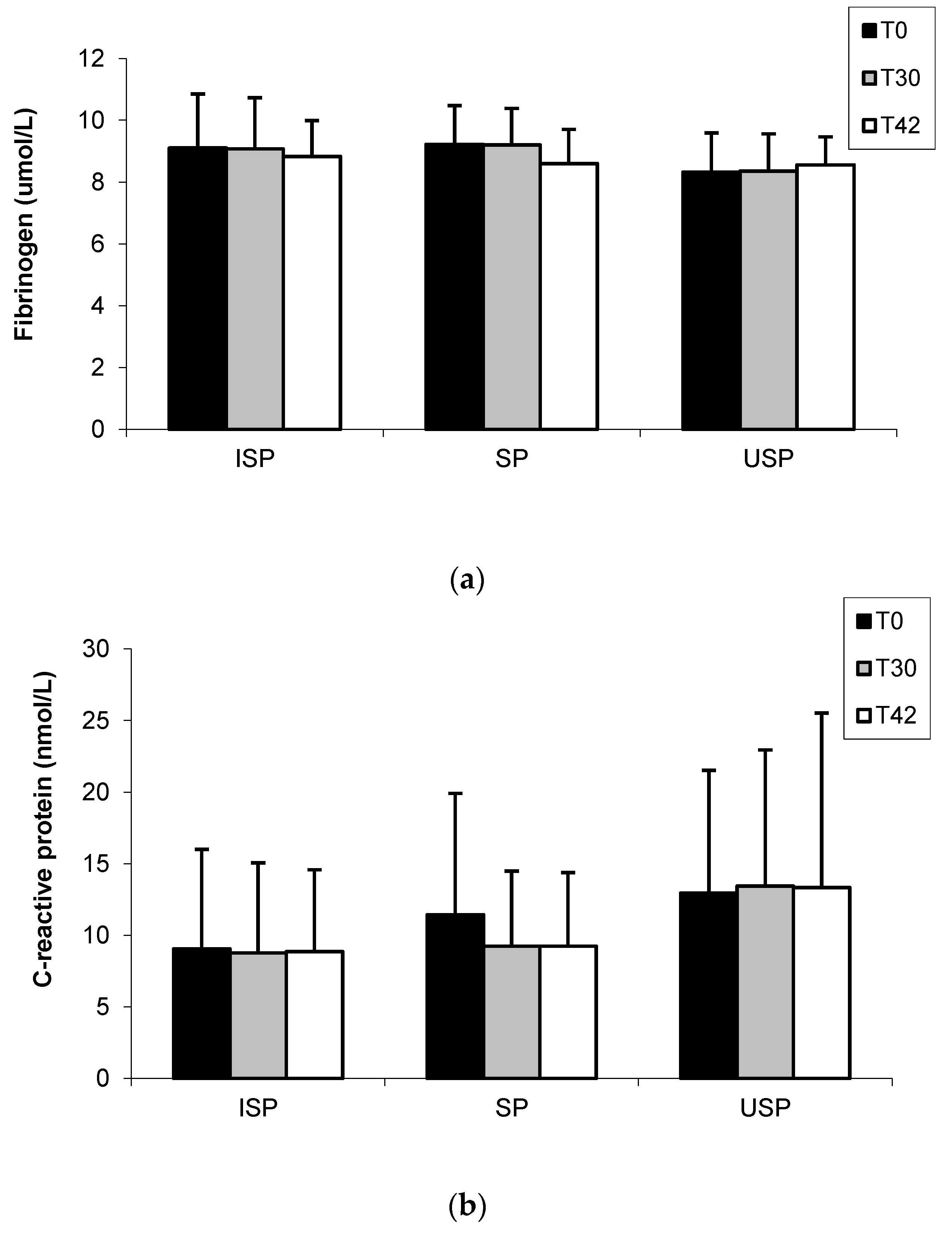
3.5. Urine Isoflavones
| Isoflavones (μmol/L) | ISP (n = 17) | SP (n = 17) | USP (n = 15) |
|---|---|---|---|
| Daidzein | 70.09 ± 25.21 a | 13.02 ± 0.67 b | 6.37 ± 1.9 c |
| Equol | 5.24 ± 2.68 | nd | nd |
| Genistein | 23.42 ± 7.88 a | 6.18 ± 0.11 b | 6.55 ± 0.41 b |
| Total | 75.75 ± 35.78 a | 19.20 ± 0.79 b | 12.92 ± 0.60 c |
3.6. E. faecium Gastrointestinal Survival
| Treatments | Enterococcus Species | |||||
|---|---|---|---|---|---|---|
| T0 | T30 | T42 | ||||
| ISP | E. faecium | 48.36% | E. faecium | 93.97% | E. faecium | 96.09% |
| E. faecalis | 30.19% | E. durans | 6.03% | E. durans | 3.91% | |
| E. durans | 21.45% | |||||
| SP | E. faecium | 54.55% | E. faecium | 83.00% | E. faecium | 83.00% |
| E. gallinarum | 18.55% | E. durans | 17.00% | E. durans | 17.00% | |
| E. durans | 26.90% | |||||
| USP | E. faecium | 52.80% | E. faecium | 50.30% | E. faecium | 45.10% |
| E. durans | 20.07% | E. gallinarum | 25.80% | E. gallinarum | 30.10% | |
| E. faecalis | 7.13% | E. durans | 23.90% | E. durans | 24.80% | |
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- World Health Organization. Global Health Risks. Mortality and Burden of Disease Attributable to Selected Major Risks. Available online: http://www.who.int/healthinfo/global_burden_disease/GlobalHealthRisks_report_full.pdf?ua=1 (accessed on 18 December 2014).
- De Castellarnau, C.; Sánchez-Quesada, J.L.; Benítez, S.; Rosa, R.; Caveda, L.; Vila, L.; Ordóñez-Llanos, J. Electronegative LDL from normolipidemic subjects induces IL-8 and monocyte chemotactic protein secretion by human endothelial cells. Aterioscler. Thromb. Vasc. Biol. 2000, 20, 2281–2287. [Google Scholar] [CrossRef]
- Mello, A.P.Q.; Silva, I.T.; Abdalla, D.S.P.; Damasceno, N.R.T. Electronegative low-density lipoprotein: Origin and impact on health and disease. Atherosclerosis 2011, 215, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Toshima, S.; Hasegawa, A.; Kurabayashi, M.; Itabe, H.; Takano, T.; Sugano, J.; Shimamura, K.; Kimura, J.; Michishita, I.; Suzuki, T.; et al. Circulating oxidized low density lipoprotein levels. A biochemical risk marker for coronary heart disease. Arterioscler. Thromb. Vasc. Biol. 2000, 20, 2243–2247. [Google Scholar] [CrossRef] [PubMed]
- Fukomoto, M.; Shoji, T.; Emoto, M.; Kawagishi, T.; Okuno, Y.; Nishizawa, Y. Antibodies against oxidized LDL and carotid artery intima-media thickness in a healthy population. Arterioscler. Thromb. Vasc. Biol. 2000, 20, 703–707. [Google Scholar] [CrossRef]
- Inoue, T.; Uchida, T.; Kamishirado, H.; Takayanagi, K.; Takayanagi, K.; Morooka, S. Antibody against oxidized low density lipoprotein may predict progression or regression of atherosclerotic coronary artery disease. J. Am. Coll. Cardiol. 2001, 37, 1871–1876. [Google Scholar] [CrossRef]
- Chen, H.W.; Kuo, C.L.; Huang, C.S.; Kuo, S.J.; Liu, C.S. Oxidized low-density lipoproteins, autoantibodies against oxidized low-density lipoproteins and carotid intima media thickness in a clinically healthy population. Cardiology 2008, 110, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Karvonen, J.; Paivansalo, M.; Kesaniemi, Y.A.; Horkko, S. Immunoglobulin M type of autoantibodies to oxidized low-density lipoprotein has an inverse relation to carotid artery atherosclerosis. Circulation 2003, 108, 2107–2112. [Google Scholar] [CrossRef] [PubMed]
- Kim, P.Y.; Stewart, R.J.; Lipson, S.M.; Nesheim, M.E. The relative kinetics of clotting and lysis provide a biochemical rationale for the correlation between elevated fibrinogen and cardiovascular disease. J. Thromb. Haemost. 2007, 5, 1250–1256. [Google Scholar] [CrossRef] [PubMed]
- Albert, C.M.; Ma, J.; Rifai, N.; Stampfer, M.J.; Ridker, P.M. Prospective study of C-reactive protein, homocysteine, and plasma lipid levels as predictors of sudden cardiac death. Circulation 2002, 105, 2595–2599. [Google Scholar] [CrossRef] [PubMed]
- Lloyd-Jones, D.M.; Hong, Y.; Labarthe, D.; Mozaffarian, D.; Appel, L.J.; van Horn, L.; Greenlund, K.; Daniels, S.; Tomaselli, G.F.; Arnett, D.K.; et al. Defining and setting national goals for cardiovascular health promotion and disease reduction: The American Heart Association’s strategic impact goal through 2020 and beyond. Circulation 2010, 121, 586–613. [Google Scholar] [CrossRef] [PubMed]
- Wierzbicki, A.S.; Hardman, T.C.; Viljoen, A. New lipid-lowering drugs: An update. Int. J. Clin. Pract. 2012, 66, 270–280. [Google Scholar] [CrossRef] [PubMed]
- Ahn-Jarvis, J.; Clinton, S.K.; Rield, J.M.; Vodovotz, Y.; Schawartz, S.J. Impact of food matrix on isoflavone metabolism and cardiovascularbiomarkers in adults with hypercholesterolemia. Food Funct. 2012, 3, 1051–1058. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Liu, X.M.; Zhang, Q.X.; Shen, Z.; Tian, F.W.; Zhang, H.; Sun, Z.H.; Zhang, H.P.; Chen, W. Influence of consumption of probiotics on the plasma lipid profile: A meta-analysis of randomised controlled trials. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 844–850. [Google Scholar] [CrossRef] [PubMed]
- Messina, M. A brief historical overview of the past two decades of soy and isoflavone research. J. Nutr. 2010, 140, 1350S–1354S. [Google Scholar] [CrossRef] [PubMed]
- Adans, M.R.; Golden, D.L.; Williams, J.K.; Franke, A.A.; Register, T.C.; Kaplan, J.R. Soy protein containing isoflavones reduces the size of atherosclerotic plaques without affecting coronary artery reactivity in adult male monkey. J. Nutr. 2005, 135, 2852–2856. [Google Scholar]
- Anthony, M.S.; Clarkson, T.B.; Hughes, C.L.; Morgan, T.M.; Burke, G.L. Soybean isoflavones improve cardiovascular risk factors without affecting the reproductive system of peripubertal rhesus monkeys. J. Nutr. 1996, 126, 43–50. [Google Scholar] [PubMed]
- Zhuo, X.G.; Melby, M.K.; Watanabe, S. Soy isoflavone intake lowers serum LDL cholesterol: A meta-analysis of 8 randomized controls trials in humans. J. Nutr. 2004, 134, 2395–2400. [Google Scholar] [PubMed]
- Liu, Z.; Ho, S.C.H.; Chen, Y.; Ho, S.; To, K.; Tomlison, B.; Woo, J. Whole soy, but not purified daidzein, had a favorable effect on improvement of cardiovascular risks: A 6-month randomized, double-blind, and placebo-controlled trial in equol-producing postmenopausal women. Mol. Nutr. Food Res. 2014, 58, 709–717. [Google Scholar] [CrossRef] [PubMed]
- Pereira, D.I.A.; Gibson, G.R. Cholesterol assimilation by lactic acid bacteria and bifidobacteria isolated from the human gut. Appl. Environ. Microbiol. 2002, 68, 4689–4693. [Google Scholar] [CrossRef] [PubMed]
- Rossi, E.A.; Giori, G.S.; Holgado, A.P.R.; Valdez, G.F. In vitro effect of Enterococcus faecium and Lactobacillus acidophilus on cholesterol. Microbiol. Aliment. Nutr. 1994, 12, 267–270. [Google Scholar]
- Rossi, E.A.; Vendramini, R.C.; Carlos, I.Z.; Ueiji, I.S.; Squinzari, M.M.; Silva Júnior, S. Effects of a novel fermented soy product on the serum lipids of hypercholesterolemic rabbits. Arq. Bras. Cardiol. 2000, 74, 213–216. [Google Scholar] [CrossRef]
- Rossi, E.A.; Vendramini, R.C.; Carlos, I.Z.; Oliveira, M.N.; Valdez, G.F. Efeito de um novo produto fermentado de soja sobre lípides séricos de homens adultos normocolesterolêmicos. Arch. Latinoam. Nutr. 2003, 53, 47–51. (in Portuguese). [Google Scholar] [PubMed]
- Rossi, E.A.; Rosier, I.; Dâmaso, A.R.; Vendramini, R.C.; Carlos, I.Z.; Abdalla, D.S.P. Determinação de isoflavonas nas diversas etapas de processamento de “iogurte” de soja. Aliment. Nutr. 2004, 15, 93–99. (in Portuguese). [Google Scholar]
- Cavallini, D.C.U.; Abdalla, D.S.P.; Bondespacho, L.Q.; Bedani, R.; Pauly-Silveira, N.D.; Valdez, G.F.; Rossi, E.A. Effects of probiotic bacteria, isoflavones and simvastatin on lipid profile and atherosclerosis in cholesterol-fed rabbits: A randomized double-blind study. Lipids Health Dis. 2009, 8. [Google Scholar] [CrossRef] [PubMed]
- Cavallini, D.C.U.; Abdalla, D.S.P.; Vendramini, R.C.; Bedani, R.; Bondespacho, L.Q.; Pauly-Silveira, N.D.; Valdez, G.F.; Rossi, E.A. Effects of isoflavone-supplemented soy yogurt on lipid parameters and atherosclerosis development in hypercholesterolemic rabbits: A randomized double-blind study. Lipids Health Dis 2009, 8. [Google Scholar] [CrossRef] [PubMed]
- Cavallini, D.C.U.; Suzuki, J.Y.; Abdalla, D.S.P.; Vendramini, R.C.; Pauly-Silveira, N.D.; Roselino, M.N.; Pinto, E.A.; Rossi, E.A. Influence of a probiotic soy product on fecal microbiota and its association with cardiovascular risk factors in an animal model. Lipids Health Dis. 2011, 10. [Google Scholar] [CrossRef] [PubMed]
- Rossi, E.A.; Carlos, I.Z.; Pei, Y.C.; Valdez, G.F. Development of a novel fermented soymilk product with potential probiotic properties. Eur. Food Res. Technol. 1999, 209, 305–307. [Google Scholar] [CrossRef]
- Allain, C.A.; Poon, L.S.; Chan, C.S.G.; Richmond, W.; Fu, P.C. Enzymatic determination of total serum cholesterol. Clin. Chem. 1974, 20, 470–475. [Google Scholar] [PubMed]
- Bergmeyer, H.U. Methods of Enzymatic Analysis, 3rd ed.; Academic Press: London, UK, 1983. [Google Scholar]
- Fossati, P.; Prencipe, L. Serum triglycerides determined colorimetrically with an enzyme that produce hydrogen peroxide. Clin. Chem. 1982, 29, 2077–2080. [Google Scholar]
- Friedwald, W.T.; Levy, R.J.; Frederickson, D.S. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of preparative centrifuge. Clin. Chem. 1972, 18, 499–509. [Google Scholar]
- Liu, J.; Sempos, C.; Donahue, R.P.; Dorn, J.; Trevisan, M.; Grundy, S.M. Joint distribution of Non-HDL and LDL cholesterol and coronary heart disease risk prediction among individuals with and without diabetes. Diabetes Care 2005, 28, 1916–1921. [Google Scholar] [CrossRef] [PubMed]
- Song, T.; Lee, S.O.; Murphy, P.A.; Hendrich, S. Soy protein with or without isoflavones, soy germ and soy germ extract, and daidzein lessen plasma cholesterol levels in golden syrian hamsters. Exp. Biol. Med. 2006, 228, 1063–1068. [Google Scholar]
- Lobo, J.C.; Mafra, D.; Farage, N.E.; Faulin, T.A.S.; Abdalla, D.S.P.; Nóbrega, A.C.L.; Torres, J.P.M. Increased electronegative LDL and decreased antibodies against electronegative LDL levels correlate with inflammatory markers and adhesion molecules in hemodialysed patients. Clin. Chim. Acta 2011, 412, 1788–1792. [Google Scholar] [CrossRef] [PubMed]
- Faulin, T.E.S.; de Sena-Evangelista, K.C.; Pacheco, D.B.; Augusto, E.M.; Abdalla, D.S. Development of immunoassays for anti-electronegative LDL autoantibodies and immune complexes. Clin. Chim. Acta 2012, 413, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Bedani, R.; Pauly-Silveira, N.D.; Cano, V.S.P.; Valentini, S.R.; de Valdez, G.F.; Rossi, E.A. Effect of ingestion of soy yogurt on intestinal parameters of rats fed on a beef-based animal diet. Braz. J. Microbiol. 2011, 42, 1238–1247. [Google Scholar] [CrossRef] [PubMed]
- Coppola, T.M.; Parente, J.E.; Dumontet, S.; la Peccerella, A. The microflora of natural whey cultures utilized as stars in the manufacture of Mozzarella cheese from water buffalo milk. Le Lait 1988, 68, 295–310. [Google Scholar] [CrossRef]
- Sivieri, K.; Cano, V.P.S.; Valentini, S.R.; Rossi, E.A. Gastrointestinal survival of Enterococcus faecium CRL 183 in rats. Le Lait 2007, 87, 59–69. [Google Scholar]
- Langa, S.; Fernández, A.; Martín, R.; Reviriego, C.; Marin, M.; Fernandez, L. Differentiation of Enterococcus. faecium from Lactobacillus delbrueckii subsp. bulgaricus and Streptococcus thermophilus strains by PCR and dot-blot hybridization. Int. J. Food Microbiol. 2003, 88, 197–200. [Google Scholar]
- Perk, J.; de Backer, G.; Gohlke, H.; Graham, I.; Reiner, Z.; Verschuren, M.; Albus, C.; Benlian, P.; Boysen, G.; Cifkova, R.; et al. European Guidelines on cardiovascular disease prevention in clinical practice (version 2012). The Fifth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice (constituted by representatives of nine societies and by invited experts). Developed with the special contribution of the European Association for Cardiovascular Prevention & Rehabilitation (EACPR). Eur. Heart J. 2012, 33, 1635–1701. [Google Scholar] [PubMed]
- Setchell, K.D.R.; Cole, S.J. Method of defining equol-producer status and its frequency among vegetarians. J. Nutr. 2006, 136, 2188–2193. [Google Scholar] [PubMed]
- Naruszewicz, M.; Johansson, M.L.; Zapolska-Downar, D.; Bukowska, H. Effect of Lactobacillus plantarum 299v on cardiovascular disease risk factor in smokers. Am. J. Clin. Nutr. 2002, 76, 1249–1255. [Google Scholar] [PubMed]
- Hlivak, P.; Odraska, J.; Ferencik, M.; Ebringer, L.; Mikes, Z. One-year application of probiotic strain Enterococcus faecium M-74 decreases serum cholesterol levels. Bratisl. Lek. Listy 2005, 106, 67–72. [Google Scholar] [PubMed]
- Ejtahed, H.S.; Mohtadi-Nia, J.; Homayouni-Rad, A.; Niafar, M.; Asghari-Jafarabadi, M.; Mofid, V.; Akbarian-Moghari, A. Effect of probiotic yogurt containing Lactobacillus acidophilus and Bifidobacterium lactis on lipid profile in individuals with type 2 diabetes mellitus. J. Dairy Sci. 2011, 94, 3288–3294. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, M.C.; Lajo, T.; Carrión, J.M.; Cuné, J. Cholesterol-lowering efficacy of Lactobacillus plantarum CECT 7527, 7528 and 7529 in hypercholesterolaemic adults. Br. J. Nutr. 2013, 109, 1866–1872. [Google Scholar] [CrossRef] [PubMed]
- Jahreis, G.; Vogelsang, H.; Kiessling, W.; Shubert, R.; Bunte, C.; Hammes, W. Influence of probiotic sausage (Lactobacillus paracasei) on blood lipids and immunological parameters of healthy volunteers. J. Food Technol. 2002, 35, 133–138. [Google Scholar] [CrossRef]
- Simons, L.A.; Amansec, S.G.; Conway, P. Effect of Lactobacillus fermentum on serum lipids in subjects with elevated serum cholesterol. Nutr. Metab. Cardiovasc. Dis. 2006, 16, 531–535. [Google Scholar] [CrossRef] [PubMed]
- Kekkonen, R.A.; Sysi-Aho, M.; Seppanen-Laakson, T.; Julkunen, I.; Vapaatalo, H.; Oresic, M.; Korpela, R. Effect of probiotic Lactobacillus rhamnosus GG intervention on global serum lipidomic profiles in healthy adults. World J. Gastroenterol. 2008, 14, 3188–3194. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Yang, Z.; Chen, X.; Huang, Y.; Yin, B.; Guo, F.; Zhao, H.; Zhao, T.; Qu, H.; Huang, J.; et al. The effect of Lactobacillus rhamnosus hsryfm 1301 on the intestinal microbiota of a hyperlipidemic rat model. BCM Complement. Altern. Med. 2014, 14, 386. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.R.; Yang, H. Progress in the effect of probiotics on cholesterol and its mechanism. Wei Sheng Wu Xue Bao 2005, 45, 315–319. [Google Scholar] [PubMed]
- Pirillo, A.; Norata, G.D.; Catapano, A.L. LOX-1, OxLDL, and atherosclerosis. Mediat. Inflamm. 2013, 2013, 152786. [Google Scholar] [CrossRef] [PubMed]
- Grosso, D.M.; Ferderbar, S.; Wanschel, A.C.; Krieger, M.H.; Higushi, M.L.; Abdalla, D.S. Antibodies against electronegative LDL inhibit atherosclerosisin LDLr−/− mice. Braz. J. Med. Biol. Res. 2008, 41, 1086–1092. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Teixeira, S.R.; Potter, S.M.; Weigel, R.; Hannum, S.; Erdman, J.W., Jr.; Hasler, C.M. Effects of feeding 4 levels of soy protein for 3 and 6 wk on blood lipids and apolipoproteins in moderately hypercholesterolemic men. Am. J. Clin. Nutr. 2000, 71, 1077–1084. [Google Scholar] [PubMed]
- Wong, W.W.; Smith, E.O.; Stuff, J.E.; Hachey, D.L.; Heird, W.C.; Pownell, H.J. Cholesterol-lowering effect of soy protein in normocholesterolemic and hypercholesterolemic men. Am. J. Clin. Nutr. 1998, 68, 1385S–1389S. [Google Scholar] [PubMed]
- Sacks, F.M.; Lichtenstein, A.; van Horn, L.; Harris, W.; Kris-Etherton, P.; Winston, M. Soy protein. isoflavones and cardiovascular health. An americam heart association science advisory for professionals from nutrition committee. Circulation 2006, 113, 1034–1044. [Google Scholar] [CrossRef] [PubMed]
- Hodgson, J.M.; Puddey, I.B.; Beilin, J.L.; Mori, T.A.; Croft, K.D. Supplementation with isoflavonoid phytoestrogens does not alter serum lipid concentrations: A randomized controlled trial in humans. J. Nutr. 1998, 128, 728–732. [Google Scholar] [PubMed]
- West, S.G.; Hilpert, K.F.; Juturu, V.; Bordi, P.L.; Lampe, J.W.; Mousa, S.A.; Kris-Etherson, P.M. Effects of including soy protein in a blood cholesterol lowering diet on markers of cardiac risk in men and postmenopausal women +/− hormone replacement therapy. J. Womens Health 2005, 14, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Mendelsohn, M.E.; Karas, R.H. The protective effects of estrogen on the cardiovascular system. N. Engl. J. Méd. 1999, 340, 1801–1811. [Google Scholar] [CrossRef]
- Rimbach, G.; Boesch-Saadatmandi, C.; Frank, J.; Fuchs, D.; Wenzel, U.; Daniel, H.; Hall, W.L.; Weinberg, P.D. Dietary isoflavones in the prevention of cardiovascular disease—A molecular perspective. Food Chem. Toxicol. 2008, 46, 1308–1319. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Bowen, R.; Cai, Q.; Barnes, S.; Wang, Y. Antioxidant and antipromotional effects of the soybean isoflavone genistein. Proc. Soc. Exp. Biol. Med. 1995, 208, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, Y.; Tsurugasaki, W.; Nakamura, S.; Osada, K. Comparison of regulative functions between dietary soy isoflavones aglycone and glucoside on lipid metabolism in rats fed cholesterol. J. Nutr. Biochem. 2005, 16, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Setchell, K.D.; Brown, N.M.; Lydeking-Olsen, E. The clinical importance of the metabolite equol–A clue to the effectiveness of soy and its isoflavones. J. Nutr. 2002, 132, 3577–3584. [Google Scholar] [PubMed]
- Akaza, H.; Miyanaga, N.; Takashima, N.; Naito, S.; Hirao, Y.; Tsukamoto, T.; Fujoika, T.; Mori, M.; Kim, W.J.; Song, J.M.; et al. Comparisons of percent equol producers between prostate cancer patients and control: Case-controlled studies of isoflavones in Japanese Korean and american residents. Jpn. J. Clin. Oncol. 2004, 34, 86–89. [Google Scholar] [CrossRef] [PubMed]
- Kelly, G.E.; Joannou, G.E.; Reeder, A.Y.; Nelson, C.; Waring, M.A. The variable metabolic response to dietary isoflavones in humans. Proc. Soc. Exp. Biol. Med. 1995, 208, 40–43. [Google Scholar] [CrossRef] [PubMed]
- Roweland, I.R.; Wiseman, H.; Sanders, T.A.; Adlercreutz, H.; Bowey, E.A. Interindividual variation in metabolism of soy isoflavones and lignans: Influence of habitual diset on equol production by the gut flora. Nutr. Cancer 2000, 36, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Setchell, K.D.; Clerici, C.; Lephart, E.D.; Cole, S.J.; Heenan, C.; Castellani, D.; Wolfe, B.E.; Nechemias-Zimmer, L.; Brown, N.M.; Lund, T.D.; et al. S-equol, a potent ligand for estrogen receptor β, is the exclusive enantiomeric form of the soy isoflavone metabolite produced by human intestinal bacterial flora. Am. J. Clin. Nutr. 2005, 81, 1072–1079. [Google Scholar] [PubMed]
- Hur, H.G.; Lay, J.O.; Beger, R.D.; Freeman, J.P.; Rafji, F. Isolation of human intestinal bacteria metabolizing the natural isoflavone glycosides daidzin and genistin. Arch. Microbiol. 2000, 174, 422–428. [Google Scholar] [CrossRef] [PubMed]
- Wong, J.M.; Kendall, C.W.; Marchie, A.; Liu, Z.; Vidgen, E.; Holmes, C.; Jackson, C.J.; Josse, R.G.; Penscharz, P.B.; Vuksan, V.; et al. Equol status and blood lipid profile in hyperlipidemia after consumption of diets containing soy foods. Am. J. Clin. Nutr. 2012, 95, 564–571. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Calvo, J.M.; Rodríguez-Iglesias, M.A.; Molinillo, J.M.G.; Macías, F.A. Soy isoflavones and their relationship with microflora: Beneficial effects on human health in equol producers. Phytochem. Rev. 2013, 12, 979–1000. [Google Scholar] [CrossRef]
- Soccol, C.R.; Vandenberghe, L.P.S.; Spier, M.R.; Medeiros, A.B.P.; Yamaguishi, C.T.; Lindner, J.D.D. The potential of probiotics: A review. Food Technol. Biotechnol. 2010, 48, 413–434. [Google Scholar]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cardoso Umbelino Cavallini, D.; Jovenasso Manzoni, M.S.; Bedani, R.; Roselino, M.N.; Celiberto, L.S.; Vendramini, R.C.; De Valdez, G.F.; Saes Parra Abdalla, D.; Aparecida Pinto, R.; Rosetto, D.; et al. Probiotic Soy Product Supplemented with Isoflavones Improves the Lipid Profile of Moderately Hypercholesterolemic Men: A Randomized Controlled Trial. Nutrients 2016, 8, 52. https://doi.org/10.3390/nu8010052
Cardoso Umbelino Cavallini D, Jovenasso Manzoni MS, Bedani R, Roselino MN, Celiberto LS, Vendramini RC, De Valdez GF, Saes Parra Abdalla D, Aparecida Pinto R, Rosetto D, et al. Probiotic Soy Product Supplemented with Isoflavones Improves the Lipid Profile of Moderately Hypercholesterolemic Men: A Randomized Controlled Trial. Nutrients. 2016; 8(1):52. https://doi.org/10.3390/nu8010052
Chicago/Turabian StyleCardoso Umbelino Cavallini, Daniela, Marla Simone Jovenasso Manzoni, Raquel Bedani, Mariana Nougalli Roselino, Larissa Sbaglia Celiberto, Regina Célia Vendramini, Graciela Font De Valdez, Dulcinéia Saes Parra Abdalla, Roseli Aparecida Pinto, Daniella Rosetto, and et al. 2016. "Probiotic Soy Product Supplemented with Isoflavones Improves the Lipid Profile of Moderately Hypercholesterolemic Men: A Randomized Controlled Trial" Nutrients 8, no. 1: 52. https://doi.org/10.3390/nu8010052
APA StyleCardoso Umbelino Cavallini, D., Jovenasso Manzoni, M. S., Bedani, R., Roselino, M. N., Celiberto, L. S., Vendramini, R. C., De Valdez, G. F., Saes Parra Abdalla, D., Aparecida Pinto, R., Rosetto, D., Roberto Valentini, S., & Antonio Rossi, E. (2016). Probiotic Soy Product Supplemented with Isoflavones Improves the Lipid Profile of Moderately Hypercholesterolemic Men: A Randomized Controlled Trial. Nutrients, 8(1), 52. https://doi.org/10.3390/nu8010052





