Maltol, a Food Flavoring Agent, Attenuates Acute Alcohol-Induced Oxidative Damage in Mice
Abstract
:1. Introduction
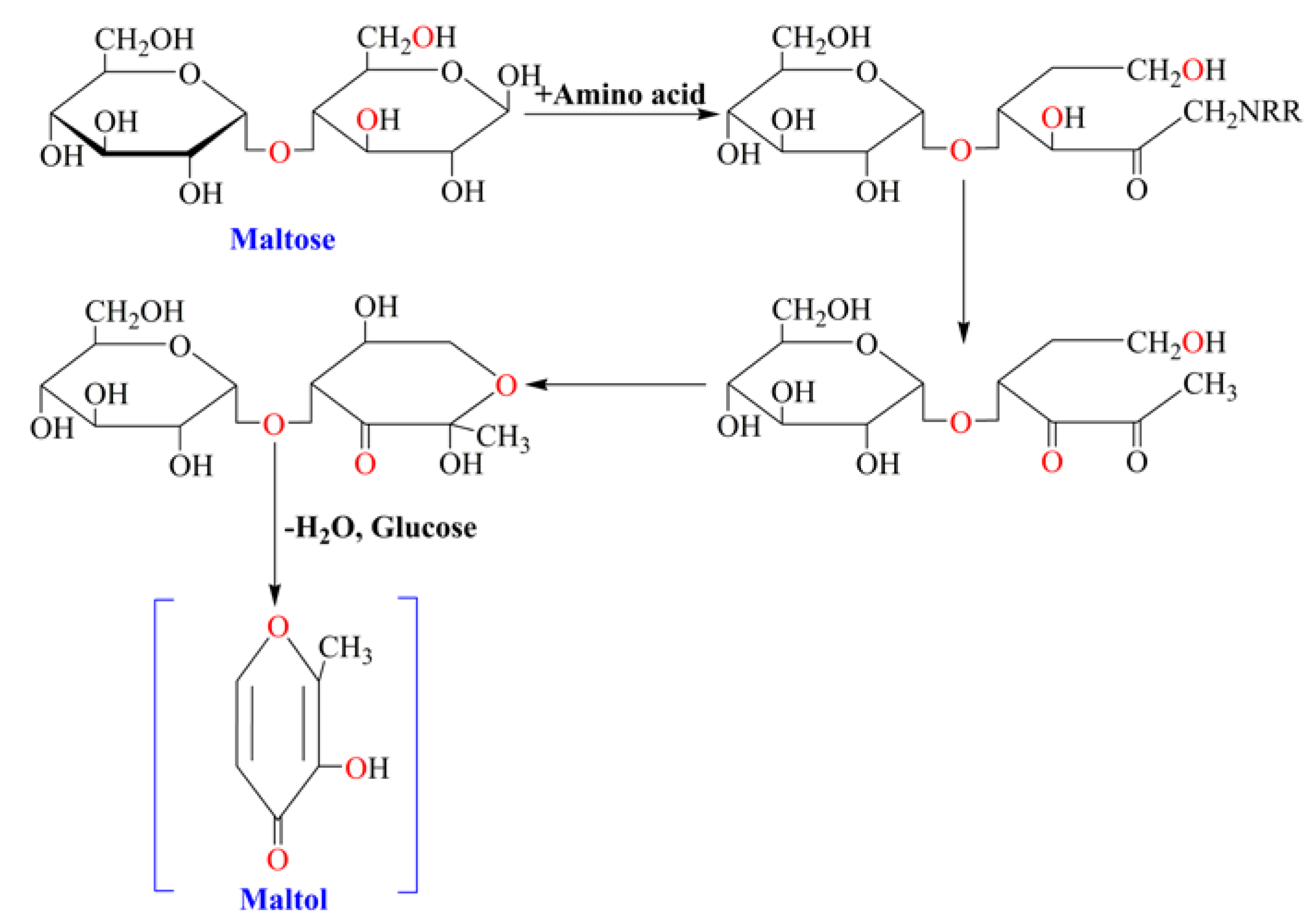
2. Experimental Section
2.1. Chemicals and Reagents
2.2. Identification and Analysis of Maltol
2.3. Animals
2.4. Experimental Groups and Treatment
2.5. Assay for Serum Biochemical Markers
2.6. Assay for Hepatic Antioxidant Activities and Oxidative Stress Marker
2.7. Assay for Inflammatory Markers Activities
2.8. Histopathological Examination
2.9. Statistical Analysis
3. Results
3.1. HPLC and MS Analysis of Maltol in Red Ginseng

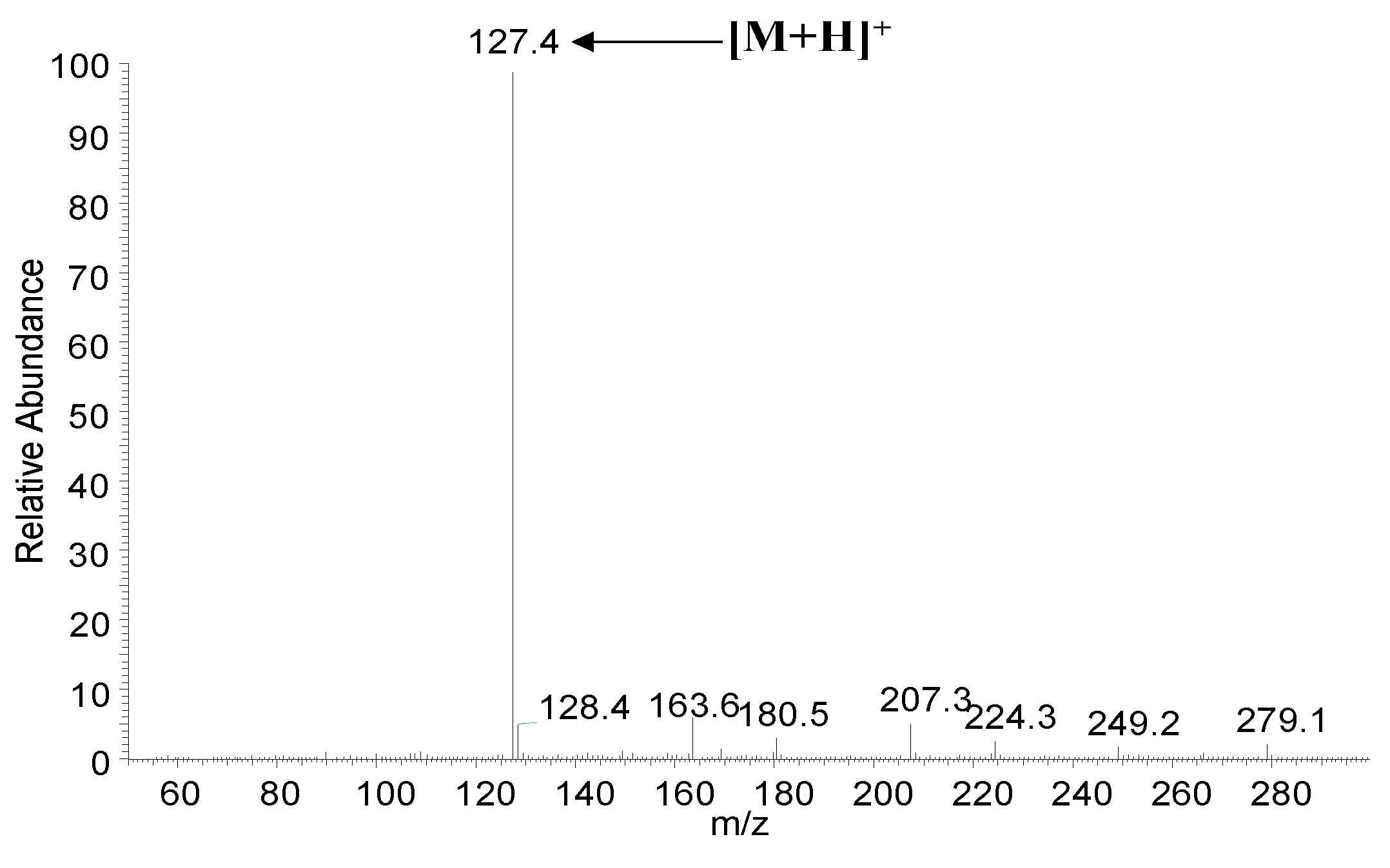
3.2. Effects of Maltol on Body Weight and Organ Weight
| Group | Dosage (mg/kg) | Weight | Weight gain (%) | Organ Weight | ||
|---|---|---|---|---|---|---|
| Initial | Final | Liver (g) | Spleen (mg) | |||
| Normal | 20.18 ± 1.08 | 31.46 ± 1.96 | 55.9 | 1.39 ± 0.12 | 106.96 ± 12.62 | |
| Alcohol | 20.13 ± 1.08 | 29.53 ± 1.67 | 46.7 * | 1.81 ± 0.22 * | 129.93 ± 23.60 * | |
| Alcohol + Silymarin | 50 | 20.07 ± 1.31 | 32.26 ± 2.60 | 60.6 | 1.62 ± 0.14 # | 112.91 ± 16.10 # |
| Alcohol + maltol | 12.5 | 20.97 ± 1.01 | 30.19 ± 2.82 | 43.9 | 1.77 ± 0.12 | 117.74 ± 15.13 |
| 25 | 20.32 ± 1.25 | 30.48 ± 2.89 | 50.0 | 1.70 ± 0.11 # | 103.63 ± 6.14 # | |
| 50 | 20.54 ± 1.05 | 30.73 ± 2.23 | 49.6 | 1.58 ± 0.10 # | 92.19 ± 3.77 # | |
3.3. Effect of Maltol on Serum Biochemical Markers
3.4. Effect of Maltol on Hepatic Biochemical Markers Activities
| Group | Dosage (mg/kg) | ALT (U/L) | AST (U/L) | ALP (U/L) | TG (mmoL/L) |
|---|---|---|---|---|---|
| Normal | 21.26 ± 2.67 | 12.84 ± 3.31 | 23.79 ± 1.65 | 0.71 ± 0.10 | |
| Alcohol | 55.26 ± 5.29 * | 59.29 ± 4.92 * | 61.03 ± 2.74 * | 1.64 ± 0.28 * | |
| Alcohol + Silymarin | 50 | 34.02 ± 5.44 # | 29.06 ± 4.58 # | 25.67 ± 1.15 # | 1.09 ± 0.19 # |
| Alcohol + maltol | 12.5 | 32.62 ± 4.01 # | 27.17 ± 4.61 # | 33.05 ± 0.29 # | 1.29 ± 0.15 |
| 25 | 28.57 ± 5.66 # | 24.52 ± 3.02 # | 28.10 ± 0.88 # | 1.12 ± 0.23 # | |
| 50 | 27.50 ±4.85 # | 26.55 ± 4.23 # | 27.04 ± 2.33 # | 0.80 ± 0.11 # |

3.5. Effect of Maltol on Hepatic Inflammatory Markers

3.6. Pathological Observations and Steatosis Grade

| Groups | Dosage (mg/kg) | Steatosis Grade | Score | Ridit | ||||
|---|---|---|---|---|---|---|---|---|
| 0 | I | II | III | IV | ||||
| Normal | 4 | 3 | 1 | 0 | 0 | 5 | 0.348 | |
| Alcohol | 0 | 1 | 2 | 3 | 2 | 19 | 0.835 * | |
| Alcohol + Silymarin | 50 | 2 | 4 | 1 | 1 | 0 | 9 | 0.484 # |
| Alcohol + maltol | 12.5 | 2 | 3 | 2 | 1 | 0 | 13 | 0.520 # |
| 25 | 3 | 3 | 2 | 0 | 0 | 9 | 0.425 # | |
| 50 | 3 | 3 | 2 | 0 | 0 | 7 | 0.389 # | |
4. Discussions
5. Conclusions
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Liu, S.; Hou, W.; Yao, P.; Zhang, B.; Sun, S.; Nussler, A.K.; Liu, L. Quercetin protects against ethanol-induced oxidative damage in rat primary hepatocytes. Toxicol. in Vitro 2010, 24, 516–522. [Google Scholar]
- Das, S.K.; Vasudevan, D.M. Alcohol-induced oxidative stress. Life Sci. 2007, 81, 177–187. [Google Scholar]
- Droge, W. Free radicals in the physiological control of cell function. Physiol. Rev. 2002, 82, 47–95. [Google Scholar]
- Gu, X.; Manautou, J.E. Molecular mechanisms underlying chemical liver injury. Expert Rev. Mol. Med. 2012, 14, e4. [Google Scholar] [CrossRef]
- Kim, Y.; You, Y.; Yoon, H.G.; Lee, Y.H.; Kim, K.; Lee, J.; Kim, M.S.; Kim, J.C.; Jun, W. Hepatoprotective effects of fermented Curcuma longa l. On carbon tetrachloride-induced oxidative stress in rats. Food Chem. 2014, 151, 148–153. [Google Scholar]
- Wang, M.; Zhu, P.; Jiang, C.; Ma, L.; Zhang, Z.; Zeng, X. Preliminary characterization, antioxidant activity in vitro and hepatoprotective effect on acute alcohol-induced liver injury in mice of polysaccharides from the peduncles of Hovenia dulcis. Food Chem. Toxicol. 2012, 50, 2964–2970. [Google Scholar]
- Tatiya, A.U.; Surana, S.J.; Sutar, M.P.; Gamit, N.H. Hepatoprotective effect of poly herbal formulation against various hepatotoxic agents in rats. Pharmacogn. Res. 2012, 4, 50–56. [Google Scholar]
- Kang, K.S.; Ham, J.; Kim, Y.J.; Park, J.H.; Cho, E.J.; Yamabe, N. Heat-processed Panax ginseng and diabetic renal damage: Active components and action mechanism. J. Ginseng Res. 2013, 37, 379–388. [Google Scholar]
- Lee, K.G.; Shibamoto, T. Antioxidant properties of aroma compounds isolated from soybeans and mung beans. J. Agric. Food Chem. 2000, 48, 4290–4293. [Google Scholar]
- Krishnakumar, V.; Barathi, D.; Mathammal, R.; Balamani, J.; Jayamani, N. Spectroscopic properties, NLO, HOMO-LUMO and NBO of maltol. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2014, 121, 245–253. [Google Scholar]
- Anwar-Mohamed, A.; El-Kadi, A.O. Induction of cytochrome p450 1a1 by the food flavoring agent, maltol. Toxicol. in Vitro 2007, 21, 685–690. [Google Scholar]
- Domotor, O.; Aicher, S.; Schmidlehner, M.; Novak, M.S.; Roller, A.; Jakupec, M.A.; Kandioller, W.; Hartinger, C.G.; Keppler, B.K.; Enyedy, E.A. Antitumor pentamethylcyclopentadienyl rhodium complexes of maltol and allomaltol: Synthesis, solution speciation and bioactivity. J. Inorg. Biochem. 2014, 134, 57–65. [Google Scholar]
- Kandioller, W.; Hartinger, C.G.; Nazarov, A.A.; Bartel, C.; Skocic, M.; Jakupec, M.A.; Arion, V.B.; Keppler, B.K. Maltol-derived ruthenium-cymene complexes with tumor inhibiting properties: The impact of ligand-metal bond stability on anticancer activity in vitro. Chemistry 2009, 15, 12283–12291. [Google Scholar]
- Hong, S.; Iizuka, Y.; Lee, T.; Kim, C.Y.; Seong, G.J. Neuroprotective and neurite outgrowth effects of maltol on retinal ganglion cells under oxidative stress. Mol. Vis. 2014, 20, 1456–1462. [Google Scholar]
- Kang, K.S.; Yamabe, N.; Kim, H.Y.; Yokozawa, T. Role of maltol in advanced glycation end products and free radicals: In-vitro and in vivo studies. J. Pharm. Pharmacol. 2008, 60, 445–452. [Google Scholar]
- Patton, S. The formation of maltol in certain carbohydrate-glycine systems. J. Biol. Chem. 1950, 184, 131–134. [Google Scholar]
- Li, M.; Lu, Y.; Hu, Y.; Zhai, X.; Xu, W.; Jing, H.; Tian, X.; Lin, Y.; Gao, D.; Yao, J. Salvianolic acid B protects against acute ethanol-induced liver injury through SIRT1-mediated deacetylation of p53 in rats. Toxico. Lett. 2014, 228, 67–74. [Google Scholar]
- Liu, H.; Qi, X.; Cao, S.; Li, P. Protective effect of flavonoid extract from Chinese bayberry (Myrica rubra Sieb. et Zucc.) fruit on alcoholic liver oxidative injury in mice. J. Nal. Med. 2014, 68, 521–529. [Google Scholar]
- Kang, K.S.; Kim, H.Y.; Pyo, J.S.; Yokozawa, T. Increase in the free radical scavenging activity of ginseng by heat-processing. Bio. Pharm. Bull. 2006, 29, 750–754. [Google Scholar]
- Kang, K.S.; Kim, H.Y.; Yamabe, N.; Nagai, R.; Yokozawa, T. Protective effect of sun ginseng against diabetic renal damage. Bio. Pharm. Bull. 2006, 29, 1678–1684. [Google Scholar]
- Song, Z.; Deaciuc, I.; Song, M.; Lee, D.Y.; Liu, Y.; Ji, X.; McClain, C. Silymarin protects against acute ethanol-induced hepatotoxicity in mice. Alcohol Clin. Exp. Res. 2006, 30, 407–413. [Google Scholar]
- Das, S.K.; Vasudevan, D.M. Protective effects of silymarin, a milk thistle (Silybium marianum) derivative on ethanol-induced oxidative stress in liver. Indian J. Biochem. Biophys. 2006, 43, 306–311. [Google Scholar]
- Draper, H.H.; Hadley, M. Malondialdehyde determination as index of lipid peroxidation. Methods Enzymol. 1990, 186, 421–431. [Google Scholar]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar]
- Lee, S.K.; Kim, J.H.; Sohn, H.J.; Yang, J.W. Changes in aroma characteristics during the preparation of red ginseng estimated by electronic nose, sensory evaluation and gas chromatography/mass spectrometry. Sens. Actuators B Chem. 2005, 106, 7–12. [Google Scholar]
- Galicia-Moreno, M.; Gutierrez-Reyes, G. The role of oxidative stress in the development of alcoholic liver disease. Rev. Gastroenterol. Mex. 2014, 79, 135–144. [Google Scholar]
- Albano, E. Alcohol, oxidative stress and free radical damage. Proc. Nutr. Soc. 2006, 65, 278–290. [Google Scholar]
- Wu, D.; Cederbaum, A.I. Alcohol, oxidative stress, and free radical damage. Alcohol Res. Health 2003, 27, 277–284. [Google Scholar]
- Noh, J.R.; Kim, Y.H.; Gang, G.T.; Hwang, J.H.; Lee, H.S.; Ly, S.Y.; Oh, W.K.; Song, K.S.; Lee, C.H. Hepatoprotective effects of chestnut (Castanea crenata) inner shell extract against chronic ethanol-induced oxidative stress in C57BL/6 mice. Food Chem. Toxicol. 2011, 49, 1537–1543. [Google Scholar]
- Yang, Y.; Wang, J.; Xu, C.; Pan, H.; Zhang, Z. Maltol inhibits apoptosis of human neuroblastoma cells induced by hydrogen peroxide. J. Biochem. Mol. Biol. 2006, 39, 145–149. [Google Scholar]
- Song, Z.; Zhou, Z.; Deaciuc, I.; Chen, T.; McClain, C.J. Inhibition of adiponectin production by homocysteine: A potential mechanism for alcoholic liver disease. Hepatology 2008, 47, 867–879. [Google Scholar]
- Zeng, T.; Guo, F.F.; Zhang, C.L.; Zhao, S.; Dou, D.D.; Gao, X.C.; Xie, K.Q. The anti-fatty liver effects of garlic oil on acute ethanol-exposed mice. Chem. Biol. Interact. 2008, 176, 234–242. [Google Scholar]
- Du, J.; He, D.; Sun, L.N.; Han, T.; Zhang, H.; Qin, L.P.; Rahman, K. Semen hoveniae extract protects against acute alcohol-induced liver injury in mice. Pharm. Biol. 2010, 48, 953–958. [Google Scholar]
- Obogwu, M.B.; Akindele, A.J.; Adeyemi, O.O. Hepatoprotective and in vivo antioxidant activities of the hydroethanolic leaf extract of Mucuna pruriens (fabaceae) in antitubercular drugs and alcohol models. Chin. J. Nat. Med. 2014, 12, 273–283. [Google Scholar]
- Qiu, L.; Chen, Y.; Gao, M.; Zheng, C.; Zhao, Q. Phagocytic uptake and ROS-mediated cytotoxicity in human hepatic cell line of amphiphilic polyphosphazene nanoparticles. J. Biomed. Mater. Res. A 2013, 101, 285–297. [Google Scholar]
- Purohit, V.; Gao, B.; Song, B.J. Molecular mechanisms of alcoholic fatty liver. Alcohol Clin. Exp. Res. 2009, 33, 191–205. [Google Scholar]
- Rukkumani, R.; Aruna, K.; Varma, P.S.; Menon, V.P. Influence of ferulic acid on circulatory prooxidant-antioxidant status during alcohol and PUFA induced toxicity. J. Physiol. Pharmacol. 2004, 55, 551–561. [Google Scholar]
- Venukumar, M.R.; Latha, M.S. Antioxidant effect of Coscinium fenestratum in carbon tetrachloride treated rats. Indian J. Physiol. Pharmacol. 2002, 46, 223–228. [Google Scholar]
- Wong, P.S.; Eiserich, J.P.; Reddy, S.; Lopez, C.L.; Cross, C.E.; van der Vliet, A. Inactivation of glutathione S-transferases by nitric oxide-derived oxidants: Exploring a role for tyrosine nitration. Arch. Biochem. Biophys. 2001, 394, 216–228. [Google Scholar]
- Singh, R.; Sharma, P. Hepatoprotective effect of curcumin on lindane-induced oxidative stress in male wistar rats. Toxicol. Int. 2011, 18, 124–129. [Google Scholar]
- You, Y.; Yoo, S.; Yoon, H.G.; Park, J.; Lee, Y.H.; Kim, S.; Oh, K.T.; Lee, J.; Cho, H.Y.; Jun, W. In vitro and in vivo hepatoprotective effects of the aqueous extract from Taraxacum officinale (dandelion) root against alcohol-induced oxidative stress. Food Chem. Toxicol. 2010, 48, 1632–1637. [Google Scholar]
- Balasubramaniyan, V.; Kalaivani Sailaja, J.; Nalini, N. Role of leptin on alcohol-induced oxidative stress in swiss mice. Pharmacol. Res. 2003, 47, 211–216. [Google Scholar]
- Park, H.Y.; Choi, H.D.; Eom, H.; Choi, I. Enzymatic modification enhances the protective activity of citrus flavonoids against alcohol-induced liver disease. Food Chem. 2013, 139, 231–240. [Google Scholar]
- Balkwill, F.; Charles, K.A.; Mantovani, A. Smoldering and polarized inflammation in the initiation and promotion of malignant disease. Cancer Cell 2005, 7, 211–217. [Google Scholar]
- Schumann, J.; Tiegs, G. Pathophysiological mechanisms of TNF during intoxication with natural or man-made toxins. Toxicology 1999, 138, 103–126. [Google Scholar]
- Candel, S.; de Oliveira, S.; Lopez-Munoz, A.; Garcia-Moreno, D.; Espin-Palazon, R.; Tyrkalska, S.D.; Cayuela, M.L.; Renshaw, S.A.; Corbalan-Velez, R.; Vidal-Abarca, I.; et al. TNF-α signaling through Tnfr2 protects skin against oxidative stress-induced inflammation. PLoS Biol. 2014, 12. [Google Scholar] [CrossRef]
- Krishnaveni, M.; Jayachandran, S. Inhibition of map kinases and down regulation of TNF-α, IL-β and cox-2 genes by the crude extracts from Marine bacteria. Biomed. Pharmacother. 2009, 63, 469–476. [Google Scholar]
- Berenbaum, F. Proinflammatory cytokines, prostaglandins, and the chondrocyte: Mechanisms of intracellular activation. Jt. B. Spine 2000, 67, 561–564. [Google Scholar]
- Chen, Y.L.; Peng, H.C.; Hsieh, Y.C.; Yang, S.C. Epidermal growth factor improved alcohol-induced inflammation in rats. Alcohol 2014, 48, 701–706. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, Y.; Xu, Q.; Hu, J.-n.; Han, X.-y.; Li, W.; Zhao, L.-c. Maltol, a Food Flavoring Agent, Attenuates Acute Alcohol-Induced Oxidative Damage in Mice. Nutrients 2015, 7, 682-696. https://doi.org/10.3390/nu7010682
Han Y, Xu Q, Hu J-n, Han X-y, Li W, Zhao L-c. Maltol, a Food Flavoring Agent, Attenuates Acute Alcohol-Induced Oxidative Damage in Mice. Nutrients. 2015; 7(1):682-696. https://doi.org/10.3390/nu7010682
Chicago/Turabian StyleHan, Ye, Qi Xu, Jiang-ning Hu, Xin-yue Han, Wei Li, and Li-chun Zhao. 2015. "Maltol, a Food Flavoring Agent, Attenuates Acute Alcohol-Induced Oxidative Damage in Mice" Nutrients 7, no. 1: 682-696. https://doi.org/10.3390/nu7010682
APA StyleHan, Y., Xu, Q., Hu, J.-n., Han, X.-y., Li, W., & Zhao, L.-c. (2015). Maltol, a Food Flavoring Agent, Attenuates Acute Alcohol-Induced Oxidative Damage in Mice. Nutrients, 7(1), 682-696. https://doi.org/10.3390/nu7010682






