Cocoa and Heart Health: A Historical Review of the Science
Abstract
:1. Introduction
2. Linkage between Cocoa and Heart Health: Early Beginnings
My heart trouble will keep me here in Paris for another few days at least, until my doctors are in complete agreement about my immediate treatment. Isn’t it the irony of fate that I have been prescribed N/G 1(nitroglycerine), to be taken internally! They call it Trinitrin, so as not to scare the chemist and the public.[7]
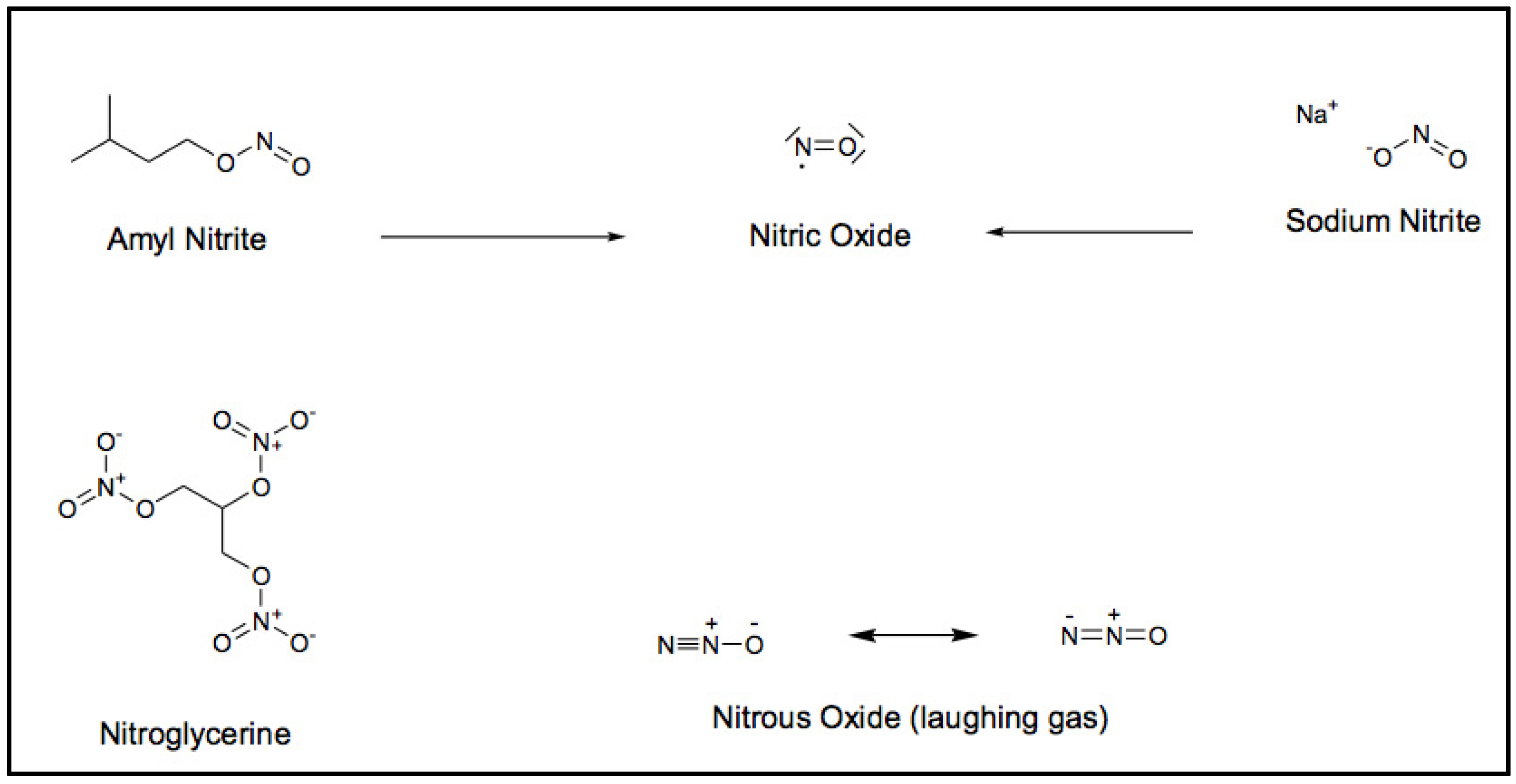
3. Linkage between Cocoa and Heart Health: Recent Findings
4. Concluding Comments on the Relationship between Cocoa and Heart Health
Acknowledgments
Conflicts of Interest
References
- Dillinger, T.L.; Barriga, P.; Escarcega, S.; Jimenez, M.; Salazar Lowe, D.; Grivetti, L.E. Food of the gods: Cure for humanity? A cultural history of the medicinal and ritual use of chocolate. J. Nutr. 2000, 130, 2057S–2072S. [Google Scholar]
- Messerli, F.H. Chocolate consumption, cognitive function, and Nobel laureates. N. Engl. J. Med. 2012, 367, 1562–1564. [Google Scholar] [CrossRef]
- Alpert, J.S. Coronary heart disease: Where have we been and where are we going? Lancet 1999, 353, 1540–1541. [Google Scholar] [CrossRef]
- Hippocrates, C. The Aphorisms of Hippocrates Classics of Medicine Library; Gryphon Editions: Birmingham, AL, USA, 1982; p. 38. [Google Scholar]
- Marsh, N.; Marsh, A. A short history of nitroglycerine and nitric oxide in pharmacology and physiology. Clin. Exp. Pharmacol. Physiol. 2000, 27, 313–319. [Google Scholar] [CrossRef]
- Hughes, R.; Dake, J.P. A Cyclopaedia of Drug Pathogenesy; Boericke and Tafel: New York, NY, USA, 1886; p. 347. [Google Scholar]
- Ringertz, N. Alfred Nobel’s Health and Interest in Medicine. 2007. Available online: http://nobelprize.org/alfred_nobel/biographical/articles/ringertz/index.html (accessed on 5 June 2013).
- Richardson, B.W. Report of the physiological action of nitrate of amyl. Br. Assoc. Adv. Sci. 1864, 34, 120–129. [Google Scholar]
- Murrell, W. Nitro-glycerine as a remedy for angina pectoris. Lancet 1879, 113, 80–81. [Google Scholar] [CrossRef]
- Murrell, W. Nitro-glycerine as a remedy for angina pectoris. Lancet 1879, 113, 113–115. [Google Scholar] [CrossRef]
- Murrell, W. Nitro-glycerine as a remedy for angina pectoris. Lancet 1879, 113, 151–152. [Google Scholar] [CrossRef]
- Murrell, W. Nitro-glycerine as a remedy for angina pectoris. Lancet 1879, 113, 225–227. [Google Scholar] [CrossRef]
- Murrell, W. Nitro-glycerine tablets. Lancet 1885, 126, 546–547. [Google Scholar] [CrossRef]
- Murrell, W.; Martindale, W.M. On the dosage of some of the vasomotor dilators. Lancet 1896, 148, 634–635. [Google Scholar] [CrossRef]
- Brunton, T.L. On the action of nitrite of amyl on the circulation. J. Anat. Physiol. 1871, 5, 92–101. [Google Scholar]
- Bodo, R. The effect of the “heart-tonics” and other drugs upon the heart-tone and coronary circulation. J. Physiol. 1928, 64, 365–387. [Google Scholar]
- Katsuki, S.; Arnold, W.; Mittal, C.; Murad, F. Stimulation of guanylate cyclase by sodium nitroprusside, nitroglycerin and nitric oxide in various tissue preparations and comparison to the effects of sodium azide and hydroxylamine. J. Cycl. Nucleotide Res. 1977, 3, 23–35. [Google Scholar]
- Murad, F.; Arnold, W.P.; Mittal, C.K.; Braughler, J.M. Properties and regulation of guanylase cyclase and some proposed functions of cyclic GMP. Adv. Cycl. Nucl. Prot. Phosphoryl. Res. 1979, 11, 175–204. [Google Scholar]
- Cherry, P.D.; Furchgott, R.F.; Zawadzki, J.V.; Jothianandan, D. Role of endothelial cells in relaxation of isolated arteries by bradykinin. Proc. Natl. Acad. Sci. USA 1982, 79, 2106–2110. [Google Scholar] [CrossRef]
- Ignarro, L.J. Biological actions and properties of endothelium-derived nitric oxide formed and released from artery and vein. Circ. Res. 1989, 65, 1–21. [Google Scholar] [CrossRef]
- Ignarro, L.J.; Byrns, R.E.; Buga, G.M.; Wood, K.S. Endothelium-derived relaxing factor from pulimary artery and vein possesses pharmacologic and chemical properties identical to those of nitric oxide radical. Circ. Res. 1987, 61, 866–879. [Google Scholar] [CrossRef]
- Palmer, R.M.; Ferrige, A.G.; Moncada, S. Nitric oxide release accounts for the biological activity of endothelium-derived relaxing factor. Nature 1987, 327, 524–526. [Google Scholar] [CrossRef]
- The 1998 Nobel Prize in Physiology or Medicine. 1998. Available online: http://nobelprize.org/nobel_prizes/lists/1998.html (accessed on 2 June 2013).
- Braunwald, E. Cardiovascular medicine at the turn of the millennium: Triumphs, concerns, and opportunities. N. Engl. J. Med. 1997, 337, 1360–1369. [Google Scholar] [CrossRef]
- Mccullough, M.; Chevaux, K.; Jackson, L.; Preston, M.; Martinez, G.; Schmitz, H.; Coletti, C.; Campos, H.; Hollenberg, N. Hypertension, the Kuna, and the epidemiology of flavanols. J. Cardiovasc. Pharmacol. 2006, 47, S103–S109. [Google Scholar] [CrossRef]
- Hollenberg, N. Vascular action of cocoa flavanols in humans: The roots of the story. J. Cardiovasc. Pharmacol. 2006, 47, S99–S102. [Google Scholar] [CrossRef]
- Fisher, N.D.L.A.; Hollenberg, N.K.B. Aging and vascular responses to flavanol-rich cocoa. J. Hypertens. 2006, 24, 1575–1580. [Google Scholar] [CrossRef]
- Bayard, V.; Chamorro, F.; Motta, J.; Hollenberg, N.K. Does flavanol intake influence mortality from nitric oxide-dependent processes? Ischemic heart disease, stroke, diabetes mellitus, and cancer in Panama. Int. J. Med. Sci. 2007, 4, 53–58. [Google Scholar]
- Buijsse, B.; Feskens, E.J.; Kok, F.J.; Kromhout, D. Cocoa intake, blood pressure, and cardiovascular mortality: The Zutphen Elderly Study. Arch. Intern. Med. 2006, 166, 411–417. [Google Scholar]
- Hertog, M.G.; Holland, P.C.; Katan, M.B. Content of potenially anticarcinogenic flavonoids of 28 vegetables and 9 fruits commonly consumed in the Netherlands. J. Agric. Food Chem. 1992, 40, 2379–2383. [Google Scholar] [CrossRef]
- Manach, C.; Scalbert, A.; Morand, C.; Remesy, C.; Jimenez, L. Polyphenols: Food sources and bioavailbility. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar]
- Arts, I.C.; Hollman, P.H.; Kromhout, D. Chocolate as a source of tea flavonoids. Lancet 1999, 354, 488. [Google Scholar] [CrossRef]
- Dreosti, I.E. Antioxidant polyphenols in tea, cocoa, and wine. Nutrition 2000, 16, 692–694. [Google Scholar] [CrossRef]
- Borchers, A.T.; Keen, C.L.; Hannum, S.M.; Gershwin, M.E. Cocoa and chocolate: Composition, bioavailability, and health implications. J. Med. Foods 2000, 3, 77–105. [Google Scholar] [CrossRef]
- Caligiani, A.; Cirlini, M.; Palla, G.; Ravaglia, R.; Arlorio, M. GC-MS detection of chiral markers in cocoa beans of different quality and geographic origin. Chirality 2007, 19, 329–334. [Google Scholar] [CrossRef]
- Schwan, R.F.; Wheals, A.E. The microbiology of cocoa fermentation and its role in chocolate quality. J. Food Sci. 2004, 59, 152–154. [Google Scholar]
- Jinap, S.; Jamilah, B.; Nazamid, S. Effects of incubation and polyphenol oxidase enrichment on colour, fermentation index, procyanidins and astringency of unfermented and partly fermented cocoa beans. Int. J. Food Sci. Tech. 2003, 38, 285–295. [Google Scholar] [CrossRef]
- Misnawi, J.S.; Jamilah, B.; Nazamid, S. Sensory properties of cocoa liquor as affected by polyphenol concentration and duration of roasting. Food Qual. Prefer. 2004, 15, 403–409. [Google Scholar] [CrossRef]
- De Brito, E.S.; Garcia, N.H.P.; Amancio, A.L.C.; Valente, A.L.P.; Pini, G.F.; Augusto, F. Effect of autoclaving cocoa nibs before roasting on the precursors of the Maillard reaction and pyrazines. Int. J. Food Sci. Technol. 2001, 36, 625–630. [Google Scholar] [CrossRef]
- Cheynier, V. Polyphenols in foods are more complex than often thought. Am. J. Clin. Nutr. 2005, 81, 223S–229S. [Google Scholar]
- Lesschaeve, I.; Noble, A.C. Polyphenols: Factors influencing their sensory properties and their effects on food and beverage preferences. Am. J. Clin. Nutr. 2005, 81, 330S–335S. [Google Scholar]
- Teixerira, S.; Siquet, C.; Alves, C.; Boal, I.; Marques, M.P. Structure-property studies on the antioxidant acitivity of flavonoids present in diet. Free Radic. Biol. Med. 2005, 39, 1099–1108. [Google Scholar] [CrossRef]
- Cooper, K.A.; Donovan, J.L.; Waterhouse, A.L.; Williamson, G. Cocoa and health: A decade of research. Br. J. Nutr. 2008, 99, 1–11. [Google Scholar]
- Hercberg, S. The history of β-carotene and cancers: From observational to intervention studies. What lessons can be drawn for future research on polyphenols? Am. J. Clin. Nutr. 2005, 81, 218S–222S. [Google Scholar]
- Mennen, L.I.; Sapinho, D.; Ito, H.; Bertrais, S.; Galan, P.; Hercherg, S.; Scalbert, A. Urinary flavonoids and phenolic acids as biomarkers of intake of polyphenol-rich foods. Br. J. Nutr. 2006, 96, 191–198. [Google Scholar] [CrossRef]
- Harnly, J.M.; Doherty, R.F.; Beecher, G.R.; Holden, J.M.; Haytowitz, D.B.; Bhagwat, S.; Gebhardt, S. Flavonoid content of U.S. fruits, vegetables, and nuts. J. Agric. Food Chem. 2006, 54, 9966–9977. [Google Scholar]
- Nutrient Data Laboratory, USDA Database for the Flavonoid Content of Selected Foods, Release 2.1; United States Department of Agriculture: Washington, DC, USA, 2007; p. 128.
- Manach, C.; Williamson, G.; Morand, C.; Scalbert, A.; Remesy, C. Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am. J. Clin. Nutr. 2005, 81, 230S–242S. [Google Scholar]
- Urquiaga, I.; Leighton, F. Plant polyphenol antioxidants and oxidative stress. Biol. Res. 2000, 33, 55–64. [Google Scholar]
- Williamson, G.; Barron, D.; Shimoi, K.; Terao, J. In vitro biological properties of flavonoid conjugates found in vivo. Free Radic. Res. 2005, 39, 457–469. [Google Scholar] [CrossRef]
- Kroon, P.A.; Clifford, M.N.; Crozier, A.; Day, A.J.; Donovan, J.L.; Manach, C.; Williamson, G. How should we assess the effects of exposure to dietary polyphenols in vitro? Am. J. Clin. Nutr. 2004, 80, 15–21. [Google Scholar]
- Lila, M.A.; Yousef, G.G.; Jiang, Y.; Weaver, C.M. Sorting out bioactivity in flavonoid mixtures. J. Nutr. 2005, 135, 1231–1235. [Google Scholar]
- Low, S.Y. Application of pharmaceuticals to nitric oxide. Mol. Asp. Med. 2005, 26, 97–138. [Google Scholar] [CrossRef]
- Lotito, S.B.; Frei, B. Consumption of flavonoid-rich foods and increased plasma antioxidant capacity in humans: Cause, consequence, or epiphenomenon? Free Radi. Biol. Med. 2006, 41, 1727–1746. [Google Scholar] [CrossRef]
- Lotito, S.B.; Actis-Goretta, L.; Renart, L.; Caligiuri, M.; Rein, D.; Schmitz, H.H.; Steinberg, F.M.; Keen, C.L.; Fraga, C.G. Influence of oligomer chain length on the antioxidant activity of procyanidins. Biochem. Biophys. Res. Commun. 2000, 276, 945–951. [Google Scholar] [CrossRef]
- Zhu, Q.; Holt, R.; Lazarus, S.; Orozco, T.; Keen, C. Inhibitory effects of cocoa flavanols and procyanidin oligomers on free radical-induced erythrocyte hemolysis. Exp. Biol. Med. 2002, 227, 321–329. [Google Scholar]
- Zhu, Q.Y.; Schramm, D.D.; Gross, H.B.; Holt, R.R.; Kim, S.H.; Yamaguchi, T.; Kwik-Uribe, C.L.; Keen, C.L. Influence of cocoa flavanols and procyanidins on free radical-induced human erythrocyte hemolysis. Clin. Dev. Immunol. 2005, 12, 27–34. [Google Scholar] [CrossRef]
- Zhang, J.; Stanley, R.A.; Adaim, A.; Melton, L.D.; Skinner, M.A. Free radical scavenging and cytoprotective activities of phenolic antioxidants. Mol. Nutr. Food Res. 2006, 50, 996–1005. [Google Scholar] [CrossRef]
- Moskaug, J.O.; Carlsen, H.; Myhrstad, M.C.W.; Blomhoff, R. Polyphenols and glutathione synthesis regulation. Am. J. Clin. Nutr. 2005, 81, 277S–283S. [Google Scholar]
- Halliwell, B.; Rafter, J.; Jenner, A. Health promotion by flavonoids, tocopherols, tocotrienols, and other phenols: Direct or indirect effects? Antioxidant or not? Am. J. Clin. Nutr. 2005, 81, 268S–276S. [Google Scholar]
- Verstraeten, S.V.; Keen, C.L.; Schmitz, H.H.; Fraga, C.G.; Oteiza, P.I. Flavan-3-ols and procyanidins protect liposomes against lipid oxidation and disruption of the bilayer structure. Free Radic. Biol. Med. 2003, 34, 84–92. [Google Scholar] [CrossRef]
- Mennen, L.I.; Walker, R.; Bennetau-Pelissero, C.; Scalbert, A. Risks and safety of polyphenol consumption. Am. J. Clin. Nutr. 2005, 81, 326S–329S. [Google Scholar]
- Prior, R.L.; Gu, L. Occurrence and biological significance of proanthocyanidins in the American diet. Phytochemistry 2005, 66, 2264–2280. [Google Scholar] [CrossRef]
- Ramiro-Puig, E.; Urpi-Sarda, M.; Perez-Cano, F.J.; Franch, A.; Castellote, C.; Andres-Lacueva, C.; Izquierdo-Pulido, M.; Castell, M. Cocoa-enriched diet enhances antioxidant enzyme activity and modulates lymphocyte composition in thymus from young rats. J. Agric. Food Chem. 2007, 55, 6431–6438. [Google Scholar] [CrossRef]
- Ramiro-Puig, E.; Casadesús, G.; Lee, H.-G.; Zhu, X.; Mcshea, A.; Perry, G.; Pérez-Cano, F.J.; Smith, M.A.; Castell, M. Neuroprotective effect of cocoa flavonoids on in vitro oxidative stress. Eur. J. Nutr. 2009, 48, 54–61. [Google Scholar] [CrossRef]
- Vinson, J.A.; Proch, J.; Bose, P.; Muchler, S.; Taffera, P.; Shuta, D.; Samman, N.; Agbor, G.A. Chocolate is a powerful ex vivo and in vivo antioxidant, an antiatherosclerotic agent in an animal model, and a significant contributor to antioxidants in the European and American diets. J. Agric. Food Chem. 2006, 54, 8071–8076. [Google Scholar] [CrossRef]
- Scalbert, A.; Johnson, I.T.; Saltmarsh, M. Polyphenols: Antioxidants and beyond. Am. J. Clin. Nutr. 2005, 81, 215S–217S. [Google Scholar]
- Murad, F. Discovery of some of the biological effects of nitric oxide and its role in cell signaling. Biosci. Rep. 2004, 24, 452–474. [Google Scholar] [CrossRef]
- Heiss, C.; Schroeter, H.; Balzer, J.; Kleinbongard, P.; Matern, S.; Sies, H.; Kelm, M. Endothelial function, nitric oxide, and cocoa flavanols. J. Cardiovasc. Pharmcol. 2006, 47, S128–S135. [Google Scholar] [CrossRef]
- Ferri, C.; Grassi, D.; Grassi, G. Cocoa beans, endothelial function and aging: An unexpected friendship? J. Hypertens. 2006, 24, 1471–1474. [Google Scholar] [CrossRef]
- Karim, M.; Mccormick, K.; Kappagoda, C.T. Effects of cocoa procyanidins on endothelium-dependent relaxation. J. Nutr. 2000, 130, 2105S–2108S. [Google Scholar]
- Fisher, N.L.; Hughes, M.; Gerhard-Herman, M.; Hollenberg, N.K. Flavanol-rich cocoa induces nitric-oxide-dependent vasodilation in healthy humans. J. Hypertens. 2003, 21, 2281–2286. [Google Scholar] [CrossRef]
- Fisher, N.D.L.; Hurwitz, S.; Hollenberg, N.K. Habitual flavonoid intake and endothelial function in healthy humans. J. Am. Coll. Nutr. 2012, 31, 275–279. [Google Scholar] [CrossRef]
- Heiss, C.; Kleinbongard, P.; Dejam, A.; Perre, S.; Schroeter, H.; Sies, H.; Kelm, M. Acute consumption of flavanol-rich cocoa and the reversal of endothelial dysfunction in smokers. J. Am. Coll. Cardiol. 2005, 46, 1276–1283. [Google Scholar] [CrossRef]
- Schroeter, H.; Heiss, C.; Balzer, J.; Kleinbongard, P.; Keen, C.L.; Hollenberg, N.K.; Sies, H.; Kwik-Uribe, C.; Schmitz, H.H.; Kelm, M. (−)-Epicatechin mediates beneficial effects of flavanol-rich cocoa on vascular function in humans. Proc. Natl. Acad. Sci. USA 2006, 103, 1024–1029. [Google Scholar] [CrossRef]
- Heiss, C.; Finis, D.; Kleinbongard, P.; Hoffmann, A.; Rassaf, T.; Kelm, M.; Sies, H. Sustained increase in flow-mediated dilation after daily intake of high-flavanol cocoa drink over 1 week. J. Cardiovasc. Pharmacol. 2007, 49, 74–80. [Google Scholar] [CrossRef]
- Farouque, H.M.O.; Leung, M.; Hope, S.A.; Baldi, M.; Schechter, C.; Cameron, J.D.; Meredith, I.T. Acute and chronic effects of flavanol-rich cocoa on vascular function in subjects with coronary artery disease: A randomized double-blind placebo-controlled study. Clin. Sci. 2006, 111, 71–80. [Google Scholar] [CrossRef]
- Wang-Polagruto, J.F.; Villablanca, A.C.; Polagruto, J.A.; Lee, L.; Holt, R.R.; Schrader, H.R.; Ensunsa, J.L.; Steinberg, F.M.; Schmitz, H.H.; Keen, C.L. Chronic consumption of flavanol-rich cocoa improves endothelial function and decreases vascular cell adhesion molecule in hypercholesterolemic postmenopausal women. J. Cardiovasc. Pharmacol. 2006, 47, S177–S186. [Google Scholar] [CrossRef]
- Husain, S.; Andrews, N.P.; Mulcahy, D.; Panza, J.A.; Quyyumi, A.A. Aspirin improves endothelial dysfunction in atherosclerosis. Circulation 1998, 97, 716–720. [Google Scholar] [CrossRef]
- Heptinstall, S.; May, J.; Fox, S.; Kwik-Uribe, C.L.; Zhao, L. Cocoa flavanols and platelet and leukocyte function: Recent in vitro and ex vivo studies in healthy adults. J. Cardiovasc. Pharmacol. 2006, 47, S197–S205. [Google Scholar]
- Vita, J.A. Polyphenols and cardiovascular disease: Effects on endothelial and platelet function. Am. J. Clin. Nutr. 2005, 81, 292S–297S. [Google Scholar]
- Pearson, D.; Paglieroni, T.; Rein, D.; Wun, T.; Schramm, D.D.; Wang, J.F.; Holt, R.R.; Gosselin, R.; Schmitz, H.H.; Keen, C.L. The effects of flavanol-rich cocoa and aspirin on ex vivo platelet function. Thromb. Res. 2002, 106, 191–197. [Google Scholar] [CrossRef]
- Rein, D.; Paglieroni, T.G.; Wun, T.; Pearson, D.A.; Schmitz, H.H.; Gosselin, R.; Keen, C.L. Cocoa inhibits platelet activation and function. Am. J. Clin. Nutr. 2000, 72, 30–35. [Google Scholar]
- Murphy, K.J.; Chronopoulos, A.K.; Singh, I.; Francis, M.A.; Moriarty, H.; Pike, M.J.; Turner, A.H.; Mann, N.; Sinclair, A. Dietary flavanols and procyanidin oligomers from cocoa (Theobroma cacao) inhibit platelet function. Am. J. Clin. Nutr. 2003, 77, 1466–1473. [Google Scholar]
- Selmi, C.; Mao, T.K.; Keen, C.L.; Schmitz, H.H.; Eric, G.M. The anti-inflammatory properties of cocoa flavanols. J. Cardiovasc. Pharmacol. 2006, 47, S163–S171. [Google Scholar] [CrossRef]
- Hodgson, J.M.; Puddey, I.B. Dietary flavonoids and cardiovascular disease: Does the emperor have any clothes? J. Hypertens. 2005, 23, 1461–1463. [Google Scholar] [CrossRef]
- Pockley, A.G. Heat shock proteins, inflammation and cardiovascular disease. Circulation 2002, 105, 1012–1017. [Google Scholar] [CrossRef]
- Carr, A.; Mccall, M.R.; Frei, B. Oxidation of LDL by myeloperoxidase and reactive nitrogen species: Reaction pathways and antioxidant protection. Arterioscler. Thromb. Vasc. Biol. 2000, 20, 1716–1723. [Google Scholar] [CrossRef]
- Osakabe, N.; Sanbongi, C.; Yamagishi, M.; Takizawa, T.; Osawa, T. Effects of polyphenol substances derived from Theobroma cacao on gastric mucsal lesion by ethanol. Biosci. Biotechnol. Biochem. 1998, 62, 1535–1538. [Google Scholar] [CrossRef]
- Keen, C.L.; Holt, R.R.; Oteiza, P.I.; Fraga, C.G.; Schmitz, H.H. Cocoa antioxidants and cardiovascular health. Am. J. Clin. Nutr. 2005, 81, 298S–303S. [Google Scholar]
- Kenny, T.P.; Keen, C.L.; Schmitz, H.H.; Gershwin, M.E. Immune effects of cocoa procyanidin oligomers on peripheral blood mononuclear cells. Exp. Biol. Med. 2007, 232, 293–300. [Google Scholar]
- Peluso, M.R. Flavonoids attenuate cardiovascular disease, inhibit phosphodiesterase, and modulate lipid homeostasis in adipose tissue and liver. Exp. Biol. Med. 2006, 231, 1287–1299. [Google Scholar]
- Baba, S.; Natsume, M.; Yasuda, A.; Nakamura, Y.; Tamura, T.; Osakabe, N.; Kanegae, M.; Kondo, K. Plasma LDL and HDL cholesterol and oxidized LDL concentrations are altered in normo- and hypercholesterolemic humans after intake of different levels of cocoa powder. J. Nutr. 2007, 137, 1436–1441. [Google Scholar]
- Kurosawa, T.; Itoh, F.; Nozaki, A.; Nakano, Y.; Katsuda, S.; Osakabe, N.; Tsubone, H.; Kondo, K.; Itakura, H. Suppressive effects of cacao liquor polyphenols (CLP) on LDL oxidation and the development of atherosclerosis in Kurosawa and Kusanagi-hypercholesterolemic rabbits. Atherosclerosis 2005, 179, 237–246. [Google Scholar] [CrossRef]
- Lecumberri, E.; Goya, L.; Mateos, R.; Alia, M.; Ramos, S.; Izquierdo-Pulido, M.; Bravo, L. A diet rich in dietary fiber from cocoa improves lipid profile and reduces malondialdehyde in hypercholesterolemic rats. Nutrition 2007, 23, 332–341. [Google Scholar] [CrossRef]
- Grassi, D.; Lippi, C.; Necozione, S.; Desideri, G.; Ferri, C. Short-term administration of dark chocolate is followed by a significant increase in insulin sensitivity and a decrease in blood pressure in healthy persons. Am. J. Clin. Nutr. 2005, 81, 611–614. [Google Scholar]
- Graham, R.; Ahn, A.; Davis, R.; O’Conner, B.; Eisenberg, D.; Phillips, R. Use of complementary and alternative medical therapies among racial and ethnic minority adults: Results from the 2002 National Health Interview Survey. J. Natl. Med. Assoc. 2005, 97, 535–545. [Google Scholar]
- Kwon, O.; Eck, P.; Chen, S.; Corpe, C.P.; Lee, J.-H.; Kruhlak, M.; Levine, M. Inhibition of the intestinal glucose transporter GLUT2 by flavonoids. FASEB J. 2007, 21, 366–377. [Google Scholar] [CrossRef]
- Zayachkivska, O.S.; Konturek, S.J.; Drozdowicz, D.; Konturek, P.C.; Brzozowski, T.; Ghegotsky, M.R. Gastroprotective effects of flavonoids in plant extracts. J. Physiol. Pharmacol. 2005, 56, S219–S231. [Google Scholar]
- Francis, S.T.P.; Head, K.B.; Morris, P.G.P.; Macdonald, I.A.P. The effect of flavanol-rich cocoa on the fMRI response to a cognitive task in healthy young people. J. Cardiovasc. Pharmacol. 2006, 47, S215–S220. [Google Scholar] [CrossRef]
- Heinrich, U.; Neukam, K.; Tronnier, H.; Sies, H.; Stahl, W. Long-term ingestion of high flavanol cocoa provides photoprotection against UV-induced erythema and improves skin condition in women. J. Nutr. 2006, 136, 1565–1569. [Google Scholar]
- Ramljak, D.; Romanczyk, L.J.; Metheny-Barlow, L.J.; Thompson, N.; Knezevic, V.; Galperin, M.; Ramesh, A.; Dickson, R.B. Pentameric procyanidin from Theobroma cacao selectively inhibits growth of human breast cancer cells. Mol. Cancer Ther. 2005, 4, 537–546. [Google Scholar]
- Fraga, C.G. Cocoa, diabetes, and hypertension: Should we eat more chocolate? Am. J. Clin. Nutr. 2005, 81, 541–542. [Google Scholar]
- Stubbe, H. The Indian Nectar, or, a Discourse Concerning Chocolata the Nature of Cacao-Nut and the Other Ingredients of That Composition Is Examined and Stated According to the Judgment and Experience of the Indian and Spanish Writers Its Effects as to Its Alimental and Venereal Quality as Well as Medicinal (Especially in Hypochondrial Melancholy) Are Fully Debated: Together with a Spagyrical Analysis of the Cacao-Nut, Performed by That Excellent Chymist Monsieur le Febure, Chymist to His Majesty; J.C. fdor Andrew Cook: London, UK, 1662; p. 184. [Google Scholar]
- Latif, R. Chocolate/cocoa and human health: A review. Neth. J. Med. 2013, 71, 63–68. [Google Scholar]
- Ellam, S.; Williamson, G. Cocoa and human health. Annu. Rev. Nutr. 2013, 33, 105–128. [Google Scholar] [CrossRef]
- Barnard, K. Flavanols, cocoa, and cardiolog. Circulation 2006, 114, f189–f192. [Google Scholar]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific Opinion on the substantiation of a health claim related to cocoa flavanols and maintenance of normal endothelium-dependent vasodilation pursuant to Article 13(5) of Regulation (EC) No 1924/2006. EFSA J. 2012, 10, 2809.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Pucciarelli, D.L. Cocoa and Heart Health: A Historical Review of the Science. Nutrients 2013, 5, 3854-3870. https://doi.org/10.3390/nu5103854
Pucciarelli DL. Cocoa and Heart Health: A Historical Review of the Science. Nutrients. 2013; 5(10):3854-3870. https://doi.org/10.3390/nu5103854
Chicago/Turabian StylePucciarelli, Deanna L. 2013. "Cocoa and Heart Health: A Historical Review of the Science" Nutrients 5, no. 10: 3854-3870. https://doi.org/10.3390/nu5103854
APA StylePucciarelli, D. L. (2013). Cocoa and Heart Health: A Historical Review of the Science. Nutrients, 5(10), 3854-3870. https://doi.org/10.3390/nu5103854



