Health Benefits of Nut Consumption
Abstract
:1. Introduction
2. Nutrient Content of Nuts
| Nuts | Energy (kJ) | Fat (g) | SFA (g) | MUFA (g) | PUFA (g) | LA (g) | ALA (g) | Protein (g) | Fiber (g) | Folate (μg) | PS (mg) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Almonds | 2418 | 50.6 | 3.9 | 32.2 | 12.2 | 12.2 | 0.00 | 21.3 | 8.8 | 29 | 120 |
| Brazil nuts (dried) | 2743 | 66.4 | 15.1 | 24.5 | 20.6 | 20.5 | 0.05 | 14.3 | 8.5 | 22 | NR |
| Cashews | 2314 | 46.4 | 9.2 | 27.3 | 7.8 | 7.7 | 0.15 | 18.2 | 5.9 | 25 | 158 |
| Hazelnuts | 2629 | 60.8 | 4.5 | 45.7 | 7.9 | 7.8 | 0.09 | 15.0 | 10.4 | 113 | 96 |
| Macadamia nuts | 3004 | 75.8 | 12.1 | 58.9 | 1.5 | 1.3 | 0.21 | 7.9 | 6.0 | 11 | 116 |
| Peanuts | 2220 | 49.2 | 6.8 | 24.4 | 15.6 | 15.6 | 0.00 | 25.8 | 8.5 | 145 | 220 |
| Pecans | 2889 | 72.0 | 6.2 | 40.8 | 21.6 | 20.6 | 1.00 | 9.2 | 8.4 | 22 | 102 |
| Pine nuts (dried) | 2816 | 68.4 | 4.9 | 18.8 | 34.1 | 33.2 | 0.16 | 13.7 | 3.7 | 34 | 141 |
| Pistachios | 2332 | 44.4 | 5.4 | 23.3 | 13.5 | 13.2 | 0.25 | 20.6 | 9.0 | 51 | 214 |
| Walnuts | 2738 | 65.2 | 6.1 | 8.9 | 47.2 | 38.1 | 9.08 | 15.2 | 6.4 | 98 | 72 |
| Nuts | Calcium | Magnesium | Sodium | Potassium |
|---|---|---|---|---|
| Almonds | 248 | 275 | 1 | 728 |
| Brazil nuts | 160 | 376 | 3 | 659 |
| Cashew nuts | 37 | 292 | 12 | 660 |
| Hazelnuts | 114 | 163 | 0 | 680 |
| Macadamia nuts | 85 | 130 | 5 | 368 |
| Peanuts | 92 | 168 | 18 | 705 |
| Pecans | 70 | 121 | 0 | 410 |
| Pine nuts | 16 | 251 | 2 | 597 |
| Pistachios | 107 | 121 | 1 | 1025 |
| Walnuts | 98 | 158 | 2 | 441 |
| Other foods | ||||
| Apples (with skin) | 6 | 5 | 1 | 107 |
| Bananas | 5 | 27 | 1 | 358 |
| Beans (white, cooked) | 90 | 63 | 6 | 561 |
| Broccoli (cooked) | 40 | 21 | 41 | 293 |
| Cheese (cheddar) | 721 | 28 | 621 | 98 |
| Chickpeas (cooked) | 49 | 48 | 7 | 291 |
| Ham | 24 | 22 | 1304 | 287 |
| Lettuce (romaine) | 33 | 14 | 8 | 247 |
| Milk | 113 | 10 | 40 | 143 |
| Rice (white, cooked) | 10 | 38 | 1 | 35 |
| Sardines (canned in oil) | 382 | 39 | 505 | 397 |
| Spinach (cooked) | 136 | 87 | 70 | 466 |
| Tomato | 10 | 11 | 5 | 237 |
| Veal (cooked) | 22 | 26 | 87 | 325 |
| Wheat bread | 72 | 86 | 527 | 252 |
3. Epidemiological Studies of Nut Consumption and Health Outcomes
3.1. Nut Consumption and CHD Risk
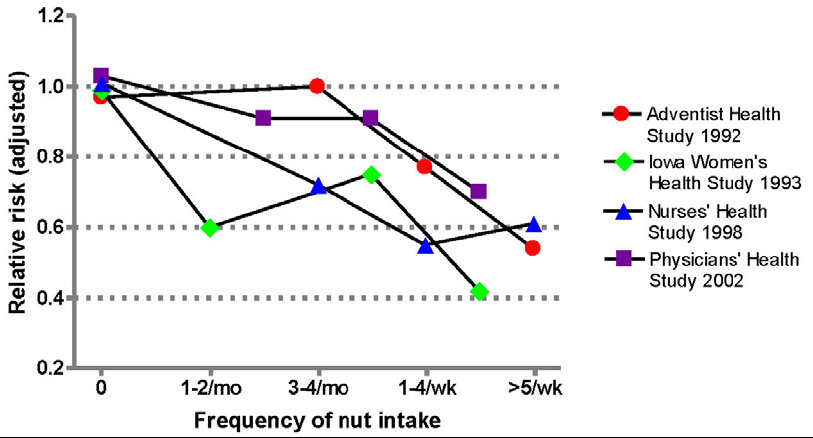
3.2. Nut Consumption and Risk of Type-2 Diabetes
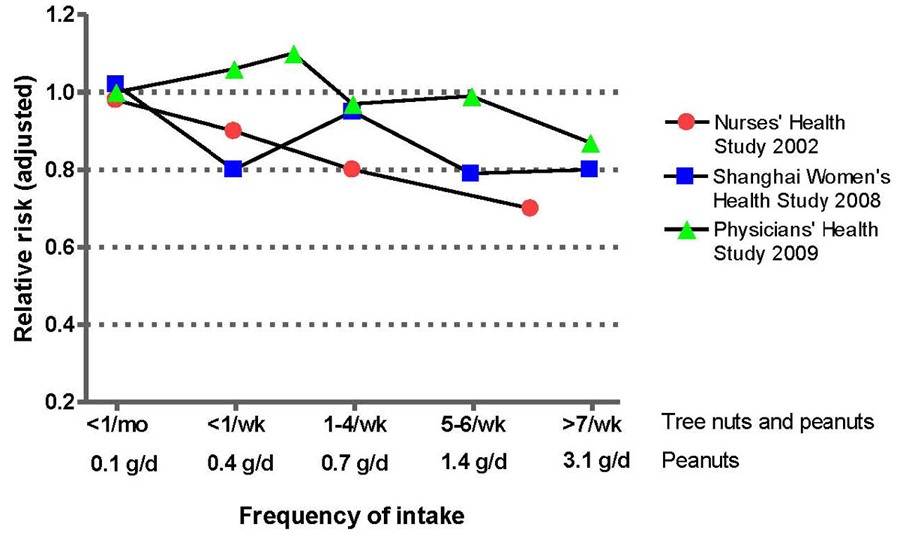
3.3. Nut Consumption and other Health Outcomes
4. Nut Feeding Trials with Outcomes on Cardiovascular Risk Factors
4.1. Effects of Nuts on the Lipid Profile
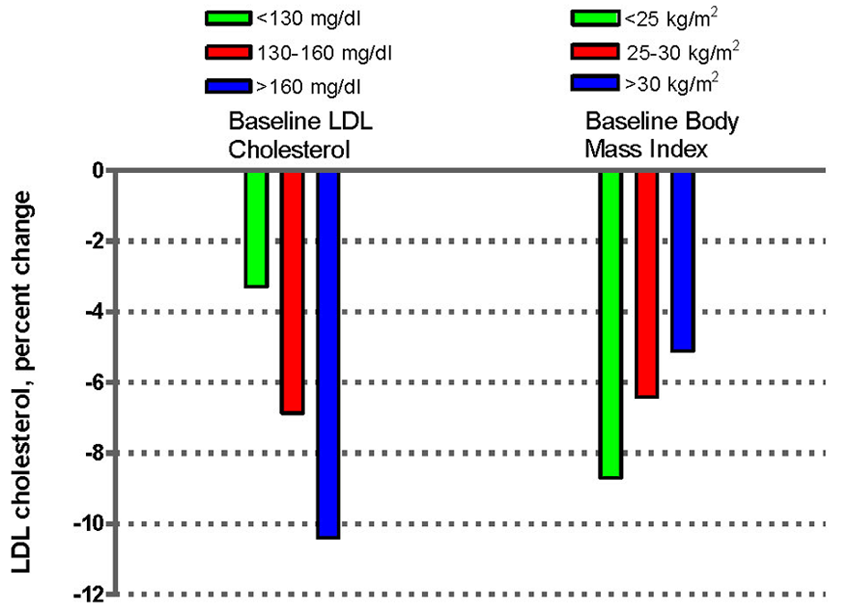
4.2. Nuts, Insulin resistance and Glycemic Control
4.3. Effects of Nuts on Emerging Cardiovascular Risk Factors
4.3.1. Oxidation
4.3.2. Inflammation
4.3.3. Vascular reactivity
5. Safety of Nut Consumption
5.1. Body Weight
5.2. Allergic Reactions
6. Conclusion
| Variables | Effect | Level of evidence |
|---|---|---|
| Epidemiologic studies | ||
| Coronary heart disease | Decrease | ++ |
| Sudden cardiac death | Decrease | + |
| Ischemic stroke | No change | + |
| Heart failure | No change | + |
| Hypertension | Decrease | +/- |
| Diabetes | No change/decrease | +/- |
| Cancer | No change/decrease | +/- |
| Gallstone disease | Decrease | + |
| Complications of diverticular disease | Decrease | + |
| Inflammatory markers | Decrease | + |
| Body weight | No change/decrease | ++ |
| Clinical studies | ||
| Blood cholesterol | Decrease | ++ |
| Insulin sensitivity | No change/increase | +/- |
| Blood pressure | Decrease | + |
| Oxidation | No change/decrease | +/- |
| Inflammation | No change/decrease | +/- |
| Vascular reactivity | Increase | + |
| Body weight | No change | ++ |
| Visceral adiposity | Decrease | + |
Conflict of Interest
Acknowledgements
References
- Fraser, G.E.; Sabaté, J.; Beeson, W.L; Strahan, T.M. A possible protective effect of nut consumption on risk of coronary heart disease. The Adventist Health Study. Arch. Intern. Med. 1992, 152, 1416–1424. [Google Scholar] [PubMed]
- Sabaté, J.; Fraser, G.E.; Burke, K.; Knutsen, S.F.; Bennett, H.; Lindsted, K.D. Effects of walnuts on serum lipid levels and blood pressure in normal men. N. Engl. J. Med. 1993, 328, 603–607. [Google Scholar]
- Sabaté, J.; Salas-Salvadó, J.; Ros, E. Nuts, nutrition and health outcomes. Br. J. Nutr. 2006, 96, S1–S102. [Google Scholar]
- King, J.C.; Rechkemmer, G.; Geiger, C.J. Supplement: 2007 Nuts and Health Symposium. J. Nutr. 2008, 138, 1734S–1765S. [Google Scholar]
- Brufau, G.; Boatella, J.; Rafecas, M. Nuts, source of energy and macronutrients. Br. J. Nutr. 2006, 96, S24–S28. [Google Scholar]
- Ros, E.; Mataix, J. Fatty acid composition of nuts. Implications for cardiovascular health. Br. J. Nutr. 2006, 96, S29–S35. [Google Scholar] [CrossRef] [PubMed]
- Eaton, S.B.; Konner, M. Paleolithic nutrition. A consideration of its nature and current implications. N. Engl. J. Med. 1985, 312, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Dreher, M.L.; Maher, C.V.; Kearney, P. The traditional and emerging role of nuts in healthful diets. Nutr. Rev. 1996, 54, 241–245. [Google Scholar]
- Sabaté, J. Nut consumption, vegetarian diets, ischemic heart disease risk, and all-cause mortality: evidence from epidemiologic studies. Am. J. Clin. Nutr. 1999, 70, 500S–503S. [Google Scholar]
- Hu, F.B.; Willett, W.C. Optimal diets for prevention of coronary heart disease. J. Am. Med. Assoc. 2002, 288, 2569–2578. [Google Scholar]
- US Food and Drug Administration, Qualified Health Claims, Letter of Enforcement Discretion – Nuts and Coronary Heart Disease; US Food & Drug Administration: Rockville, MD, USA, 14 07 2003; pp. 1–4.
- King, J.C.; Blumberg, J.; Ingwersen, L.; Jenab, M.; Tucker, K.L. Tree nuts and peanuts as components of a healthy diet. J. Nutr. 2008, 138, 1736–1740. [Google Scholar] [PubMed]
- Nash, S.D.; Nash, D.T. Nuts as part of a healthy cardiovascular diet. Curr. Atheroscler. Rev. 2008, 10, 529–535. [Google Scholar]
- Lloyd-Jones, D.M.; Hong, Y.; Labarthe, D.; Mozaffarian, D.; Appel, L.J.; Van Horn, L.; Greenlund, K.; Daniels, S.; Nichol, G.; Tomaselli, G.F.; Arnett, D.K.; Fonarow, G.C.; Ho, P.M.; Lauer, M.S.; Masoudi, F.A.; Robertson, R.M.; Roger, V.; Schwamm, L.H.; Sorlie, P.; Yancy, C.W.; Rosamond, W.D. American Heart Association Strategic Planning Task Force and Statistics Committee. American Heart Association Strategic Planning Task Force and Statistics Committee. Defining and setting national goals for cardiovascular health promotion and disease reduction: the American Heart Association's strategic Impact Goal through 2020 and beyond. 121, 586–613.
- Kelly, J.H. Jr; Sabaté, J. Nuts and coronary heart disease, an epidemiological perspective. Br. J. Nutr. 2006, 96, S61–S67. [Google Scholar] [CrossRef] [PubMed]
- Sabaté, J.; Angt, Y. Nuts and health outcomes, new epidemiologic evidence. Am. J. Clin. Nutr. 2009, 89, 1643S–1648S. [Google Scholar]
- Feldman, E.B. The scientific evidence for a beneficial health relationship between walnuts and coronary heart disease. J. Nutr. 2002, 132, 1062S–1101S. [Google Scholar]
- Mukuddem-Petersen, J.; Oosthuizen, W.; Jerling, J. A systematic review of the effects of nuts on blood lipid profiles in humans. J. Nutr. 2005, 135, 2082–2089. [Google Scholar]
- Griel, A.E.; Kris-Etherton, P.M. Tree nuts and the lipid profile, a review of clinical studies. Br. J. Nutr. 2006, 96, S68–S78. [Google Scholar]
- Coates, A.M.; Howe, P.R. Edible nuts and metabolic health. Curr. Opin. Lipidol. 2007, 18, 25–30. [Google Scholar]
- Ros, E. Nuts and novel biomarkers of cardiovascular disease. Am. J. Clin. Nutr. 2009, 89, 1649S–1656S. [Google Scholar]
- López-Uriarte, P.; Bulló, M.; Casas-Agustench, P.; Babio, N.; Salas-Salvadó, J. Nuts and oxidation, a systematic review. Nutr. Rev. 2009, 67, 497–508. [Google Scholar]
- Salas-Salvadó, J.; Bulló, M.; Pérez-Heras, A.; Ros, E. Dietary fibre, nuts and cardiovascular disease. Br. J. Nutr. 2006, 96, S45–S51. [Google Scholar]
- Segura, R.; Javierre, C.; Lizarraga, M.A.; Ros, E. Other relevant components of nuts, phytosterols, folate and mineral. Br. J. Nutr. 2006, 96, S36–S44. [Google Scholar]
- Blomhoff, R.; Carlsen, M.H.; Frost Andersen, L.; Jacobs, D.R. Jr. Health benefits of nuts, potential role of antioxidants. Br. J. Nutr. 2006, 96, S52–S60. [Google Scholar]
- Rajaram, S.; Sabaté, J. Nuts, body weight and insulin resistance. Br. J. Nutr. 2006, 96, S79–S86. [Google Scholar]
- Mattes, R.D.; Kris-Etherton, P.M.; Foster, G.D. Impact of peanuts and tree nuts on body weight and healthy weight loss in adults. J. Nutr. 2008, 138, 1741S–1745S. [Google Scholar]
- Exler, J.; Weihrauch, J.L. Provisional Table on the Content of Omega-3 Fatty Acids and Other Fat Components in Selected Foods. US Department of Agriculture: Washington, DC, USA, 1986; (Publication HNIS/PT-103). [Google Scholar]
- Huynh, N.N.; Chin-Dusting, J. Amino acids, arginase and nitric oxide in vascular health. Clin. Exp. Pharmacol. Physiol. 2006, 33, 1–8. [Google Scholar]
- Welch, G.N.; Loscalzo, J. Homocysteine and atherothrombosis. N. Engl. J. Med. 1998, 338, 1042–1050. [Google Scholar]
- Wagner, K.H.; Kamal-Eldin, A.; Elmadfa, I.U. Gamma-tocopherol - An underestimated vitamin? Ann. Nutr. Metab. 2004, 48, 169–188. [Google Scholar] [CrossRef] [PubMed]
- Milbury, P.E.; Chen, C.Y.; Dolnikowski, G.G.; Blumberg, J.B. Determination of flavonoids and phenolics and their distribution in almonds. J. Agric. Food Chem. 2006, 54, 5027–5033. [Google Scholar]
- Chen, C.Y.; Milbury, P.E.; Lapsley, K.; Blumberg, J.B. Flavonoids from almond skins are bioavailable and act synergistically with vitamins C and E to enhance hamster and human LDL resistance to oxidation. J. Nutr. 2005, 135, 1366–1373. [Google Scholar]
- Lou, H.; Yuan, H.; Ma, B.; Ren, D.; Ji, M.; Oka, S. Polyphenols from peanut skins and their free radical-scavenging effects. Phytochemistry 2004, 65, 2391–2399. [Google Scholar]
- Seeram, N.P.; Zhang, Y.; Henning, S.M.; Lee, R.; Niu, Y.; Lin, G.; Heber, D. Pistachio skin phenolics are destroyed by bleaching resulting in reduced antioxidative capacities. J. Agric. Food Chem. 2006, 54, 7036–7040. [Google Scholar]
- Garrido, I.; Monagas, M.; Gómez-Cordovés, C.; Bartolomé, B. Polyphenols and antioxidant properties of almond skins: influence of industrial processing. Food. Chem. 2008, 73, C106–C115. [Google Scholar]
- Hartmann, M.A. Plant sterols and the membrane environment. Trends Plant. Sci. 1998, 3, 170–175. [Google Scholar]
- Ostlund, R.E. Jr. Phytosterols in human nutrition. Annu. Rev. Nutr. 2002, 22, 533–549. [Google Scholar]
- Cordain, L.; Eaton, S.B.; Sebastian, A.; Mann, N.; Lindeberg, S.; Watkins, B.A.; O’Keefe, J.H.; Brand-Miller, J. Origins and evolution of the Western diet: health implications for the 21st century. Am. J. Clin. Nutr. 2005, 81, 341–354. [Google Scholar]
- Jacobs, D.Rr Jr.; Gross, M.D.; Tapsell, L.C. Food synergy: an operational concept for understanding nutrition. Am. J. Clin. Nutr. 2009, 89, 1543–1548. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.B.; Stampfer, M.J.; Manson, J.E.; Rimm, E.B.; Colditz, G.A.; Rosner, B.A.; Speizer, F.E.; Hennekens, C.H.; Willett, W.C. Frequent nut consumption and risk of coronary heart disease in women. Prospective cohort study. Br. Med. J. 1998, 317, 1341–1345. [Google Scholar]
- Kushi, L.H.; Folsom, A.R.; Prineas, R.J.; Mink, P.J.; Wu, Y.; Bostick, R.M. Dietary antioxidant vitamins and death from coronary heart disease in postmenopausal women. N. Engl. J. Med. 1996, 334, 1156–1162. [Google Scholar]
- Albert, C.M.; Gaziano, J.M.; Willett, W.C.; Manson, J.E. Nut consumption and decreased risk of sudden cardiac death in the Physicians' Health Study. Arch. Intern. Med. 2002, 162, 1382–1387. [Google Scholar]
- Li, T.Y.; Brennan, A.M.; Wedick, N.M.; Mantzoros, C.; Rifai, N.; Hu, F.B. Regular consumption of nuts is associated with a lower risk of cardiovascular disease in women with type 2 diabetes. J. Nutr. 2009, 139, 1333–1338. [Google Scholar]
- Jiang, R.; Jacobs, D.R. Jr; Mayer-Davis, E.; Szklo, M.; Herrington, D.; Jenny, N.S.; Kronmal, R.; Barr, R.G. Nut and seed consumption and inflammatory markers in the Multi-Ethnic Study of Atherosclerosis. Am. J. Epidemiol. 2006, 163, 222–231. [Google Scholar] [PubMed]
- Mantzoros, C.S.; Williams, C.J.; Manson, J.E.; Meigs, J.B.; Hu, F.B. Adherence to the Mediterranean dietary pattern is positively associated with plasma adiponectin concentrations in diabetic women. Am. J. Clin. Nutr. 2006, 84, 328–335. [Google Scholar]
- Salas-Salvadó, J.; Garcia-Arellano, A.; Estruch, R.; Márquez-Sandoval, F.; Corella, D.; Fiol, M; Gómez-Gracia, E.; Vinyoles, E.; Arós, F; Herrera, C.; Lahoz, C; Lapetra, J.; Perona, J.S.; Muñoz-Aguado, D.; Martínez-González, M.A.; Ros, E. PREDIMED Investigators. Components of the Mediterranean-type food pattern and serum inflammatory markers among patients at high risk for cardiovascular disease. Eur. J. Clin. Nutr. 2008, 62, 651–659. [Google Scholar] [PubMed]
- Jiang, R.; Manson, J.E.; Stampfer, M.J.; Liu, S.; Willett, W.C.; Hu, F.B. Nut and peanut butter consumption and risk of type 2 diabetes in women. J. Am. Med. Assoc. 2002, 288, 2554–2560. [Google Scholar]
- Parker, E.D.; Harnack, L.J.; Folsom, A.R. Nut consumption and risk of type 2 diabetes. J. Am. Med. Assoc. 2003, 290, 38–39. [Google Scholar]
- Villegas, R.; Gao, Y.T.; Yang, G.; Li, H.L.; Elasy, T.A.; Zheng, W.; Shu, X.O. Legume and soy food intake and the incidence of type 2 diabetes in the Shanghai Women Health Study. Am. J. Clin. Nutr. 2008, 87, 162–167. [Google Scholar]
- Kochar, J.; Gaziano, J.M.; Djoussé, L. Nut consumption and risk of type II diabetes in the Physicians’ Health Study. Eur. J. Clin. Nutr. 2010, 64, 75–79. [Google Scholar]
- Djoussé, L.; Rudich, T.; Gaziano, J.M. Nut consumption and risk of hypertension in US male physicians. Clin. Nutr. 2009, 28, 10–14. [Google Scholar]
- Martínez-Lapiscina, E.H.; Pimenta, A.M.; Beunza, J.J.; Bes-Rastrollo, M.; Martínez, J.A.; Martínez-González, M.A. Nut consumption and incidence of hypertension: The SUN prospective cohort. Nutr. Metab. Cardiovasc. Dis. 2009. [Epub ahead of print]. [Google Scholar] [PubMed]
- Djoussé, L.; Gaziano, J.M.; Kase, C.S.; Kurth, T. Nut consumption and risk of stroke in US male physicians. Clin. Nutr. 2010. [Epub ahead of print]. [Google Scholar] [PubMed]
- Djoussé, L.; Rudich, T.; Gaziano, J.M. Nut consumption and risk of heart failure in the Physicians’ Health Study I. Am. J. Clin. Nutr. 2008, 88, 930–933. [Google Scholar]
- Gonzalez, C.A.; Salas-Salvadó, J. The potential of nuts in the prevention of cancer. Br. J. Nutr. 2006, 96, S87–S94. [Google Scholar]
- Mills, P.K.; Beeson, W.L.; Phillips, R.L.; Fraser, G.E. Cohort study of diet, lifestyle and prostate cancer in Adventist men. Cancer 1989, 64, 598–604. [Google Scholar]
- Heilbrun, L.K.; Nomura, A.; Hankin, J.H.; Stemmermann, G.N. Diet and colorectal cancer with special reference to fiber intake. Int. J. Cancer 1989, 44, 1–6. [Google Scholar]
- Singh, P.N.; Fraser, G.E. Dietary risk factors for colon cancer in a low-risk population. Am. J. Epidemiol. 1998, 148, 761–774. [Google Scholar]
- Petridou, E.; Kedikoglou, S.; Koukoulomatis, P.; Dessypris, N.; Trichopoulos, D. Diet in relation to endometrial cancer risk: a case control study in Greece. Nutr. Cancer 2002, 44, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Jenab, M.; Ferrari, P.; Slimani, N.; Norat, T.; Casagrande, C.; Overad, K.; Olsen, A.; Stripp, C.; Tjønneland, A.; Boutron-Ruault, M.C.; Clavel-Chapelon, F.; Kesse, E.; Nieters, A.; Bergmann, M.; Boeing, H.; Naska, A.; Trichopoulou, A.; Palli, D.; Krogh, V.; Celentano, E.; Tumino, R.; Sacerdote, C.; Bueno-de-Mesquita, H.B.; Ocké, M.C.; Peeters, P.H.; Engeset, D.; Quirós, J.R.; González, C.A.; Martínez, C.; Chirlaque, M.D.; Ardanaz, E.; Dorronsoro, M.; Wallström, P.; Palmqvist, R.; Van Guelpen, B.; Bingham, S.; San Joaquin, M.A.; Saracci, R.; Kaaks, R.; Riboli, E. Association of nut and seed intake with colorectal cancer risk in the European Prospective Investigation into Cancer and Nutrition. Cancer Epidemiol. Biomarkers Prev. 2004, 13, 1595–1603. [Google Scholar]
- Yeh, C.C.; You, S.L.; Chen, C.J.; Sung, F.C. Peanut consumption and reduced risk of colorectal cancer in women: A prospective study in Taiwan. World J. Gastroenterol. 2006, 12, 222–227. [Google Scholar]
- Tsai, C.Y.; Leitzmann, M.F.; Hu, F.B.; Willett, W.C.; Giovannucci, E.L. Frequent nut consumption and decreased risk of cholecystectomy in women. Am. J. Clin. Nutr. 2004, 80, 76–81. [Google Scholar]
- Spaccarotella, K.J.; Kris-Etherton, P.M.; Stone, W.L.; Bagshaw, D.M.; Fishell, V.K.; West, S.G.; Lawrence, F.R.; Hartman, T.J. The effect of walnut intake on factors related to prostate and vascular health in older men. Nutr. J. 2008, 7, 13. [Google Scholar]
- Carvalho, M.; Ferreira, P.J.; Mendes, V.S.; Silva, R.; Pereira, J.A.; Jerónimo, C.; Silva, B.M. Human cancer cell antiproliferative and antioxidant activities of Juglans regia L. Food Chem. Toxicol. 2010, 48, 441–447. [Google Scholar]
- Hardman, W.E.; Ion, G. Suppression of implanted MDA-MB 231 human breast cancer growth in nude mice by dietary walnut. Nutr. Cancer 2008, 60, 666–674. [Google Scholar]
- Tsai, C.Y.; Leitzmann, M.F.; Hu, F.B.; Willett, W.C.; Giovannucci, E.L. A prospective cohort study of nut consumption and the risk of gallstone disease in men. Am. J. Epidemiol. 2004, 160, 961–968. [Google Scholar]
- Strate, L.L.; Liu, Y.L.; Syngal, S.; Aldoori, W.H.; Giovannucci, E.L. Nut, corn, and popcorn consumption and the incidence of diverticular disease. J. Am. Med. Assoc. 2008, 300, 907–914. [Google Scholar]
- Sabaté, J.; Oda, K.; Ros, E. Nut consumption and blood lipids: a pooled analysis of 25 intervention trials. Arch. Intern. Med. 2010, 170, 821–827. [Google Scholar]
- Banel, D.K.; Hu, F.B. Effects of walnut consumption on blood lipids and other cardiovascular risk factors: a meta-analysis and systematic review. Am. J. Clin. Nutr. 2009, 90, 56–63. [Google Scholar]
- Rajaram, S.; Haddad, E.H.; Mejia, A.; Sabaté, J. Walnuts and fatty fish influence different serum lipid fractions in normal to mildly hyperlipidemic individuals: a randomized controlled study. Am. J. Clin. Nutr. 2009, 89, 1657S–1663S. [Google Scholar]
- Torabian, S.; Haddad, E.; Cordero-MacIntyre, Z.; Tanzman, J.; Fernandez, M.L.; Sabaté, J. Long-term walnut supplementation without dietary advice induces favorable serum lipid changes in free-living individuals. Eur. J. Clin. Nutr. 2010, 64, 274–279. [Google Scholar]
- Tapsell, L.C.; Batterham, M.J.; Teuss, G.; Tan, S.Y.; Dalton, S.; Quick, C.J.; Gillen, L.J.; Charlton, K.E. Long-term effects of increased dietary polyunsaturated fat from walnuts on metabolic parameters in type II diabetes. Eur. J. Clin. Nutr. 2009, 63, 1008–1015. [Google Scholar]
- Ma, Y.; Njike, V.Y.; Millet, J.; Dutta, S.; Doughty, K.; Treu, J.A.; Katz, D.L. Effects of walnut consumption on endothelial function in type 2 diabetic subjects: a randomized controlled crossover trial. Diabetes Care 2010, 33, 227–232. [Google Scholar]
- Jenkins, D.J.; Kendall, C.W.; Marchie, A.; Josse, A.R.; Nguyen, T.H.; Faulkner, D.A.; Lapsley, K.G.; Blumberg, J. Almonds reduce biomarkers of lipid peroxidation in older hyperlipidemic subjects. J. Nutr. 2008, 138, 908–913. [Google Scholar]
- Mercanligil, S.M.; Arslan, P.; Alasalvar, C.; Okut, E.; Akgül, E.; Pinar, E.; Geyik, P.O.; Tokgözoğlu, L.; Shahidi, F. Effects of hazelnut-enriched diet on plasma cholesterol and lipoprotein profiles in hypercholesterolemic adult men. Eur. J. Clin. Nutr. 2007, 61, 212–220. [Google Scholar]
- Gebauer, S.K.; West, S.G.; Kay, C.D.; Alaupovic, P.; Bagshaw, D.; Kris-Etherton, P.M. Effects of pistachios on cardiovascular disease risk factors and potential mechanisms of action, a dose-response study. Am. J. Clin. Nutr. 2008, 88, 651–659. [Google Scholar]
- Griel, A.E.; Cao, Y.; Bagshaw, D.D.; Cifelli, A.M.; Holub, B.; Kris-Etherton, P.M. A macadamia nut-rich diet reduces total and LDL-cholesterol in mildly hypercholesterolemic men and women. J. Nutr. 2008, 138, 761–767. [Google Scholar]
- Lokko, P.; Lartey, A.; Armar-Klemesu, M.; Mattes, R.D. Regular peanut consumption improves plasma lipid levels in healthy Ghanaians. Int. J. Food Sci. Nutr. 2007, 58, 190–200. [Google Scholar]
- Estruch, R.; Martínez-González, M.A.; Corella, D.; Salas-Salvadó, J.; Ruiz-Gutiérrez, V.; Covas, M.I.; Fiol, M.; Gómez-Gracia, E.; López-Sabater, M.C.; Vinyoles, E.; Arós, F.; Conde, M.; Lahoz, C.; Lapetra, J.; Sáez, G.; Ros, E. Effects of a Mediterranean-style diet on cardiovascular risk factors. A randomized trial. Ann. Intern. Med. 2006, 145, 1–11. [Google Scholar] [PubMed]
- Mukuddem-Petersen, J.; Stonehouse, W.; Jerling, J.C.; Hanekom, S.M.; White, Z. Effects of a high walnut and high cashew nut diet on selected markers of the metabolic syndrome, a controlled feeding trial. Br. J. Nutr. 2007, 97, 1144–1153. [Google Scholar]
- Casas-Agustench, P.; López-Uriarte, P.; Bulló, M.; Ros, E.; Cabré-Vila, J.J.; Salas-Salvadó, J. Effects of one serving of mixed nuts on serum lipids, insulin resistance and inflammatory markers in patients with the metabolic syndrome. Nutr. Metab. Cardiovasc. Dis. 2009. [Epub ahead of print]. [Google Scholar] [PubMed]
- Lefevre, M.; Champagne, C.M.; Tulley, R.T.; Rood, J.C.; Most, M.M. Individual variability in cardiovascular disease risk factor responses to low-fat and low-saturated-fat diets in men: body mass index, adiposity, and insulin resistance predict changes in LDL cholestero. Am. J. Clin. Nutr. 2005, 82, 957–963. [Google Scholar]
- Knopp, R.H.; Retzlaff, B.; Fish, B.; Walden, C.; Wallick, S.; Anderson, M.; Aikawa, K.; Kahn, S.E. Effects of insulin resistance and obesity on lipoproteins and sensitivity to egg feeding. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 1437–1443. [Google Scholar]
- Hannah, J.S.; Jablonshki, K.A.; Howard, B.V. The relationship between weight and response to cholesterol lowering diets in women. Int. J. Obes. Relat. Metab. Disord. 1997, 21, 445–450. [Google Scholar]
- Jansen, A.; Lopez-Miranda, J.; Salas, J.; Castro, P.; Paniagua, J.A.; Lopez-Segura, F.; Ordovas, J.M.; Jimenez-Pereperez, J.A.; Blanco, A.; Perez-Jimenez, F. Plasma lipid response to hypolipidemic diets in young health non-obese men varies with body mass index. J. Nutr. 1998, 128, 1144–1149. [Google Scholar]
- Denke, M.A.; Adams-Huet, B.; Nguyen, A.T. Individual cholesterol variation in response to a margarine- or butter-based diet. A study in families. J. Am. Med. Assoc. 2000, 284, 2740–2747. [Google Scholar]
- Simonen, P.; Gylling, H.; Howard, A.N.; Miettinen, T.A. Introducing a new component of the metabolic syndrome: low cholesterol absorption. Am. J. Clin. Nutr. 2000, 72, 82–88. [Google Scholar]
- Pihlajamäki, J.; Gylling, H.; Miettinen, T.A.; Laakso, M. Insulin resistance is associated with increased cholesterol synthesis and decreased cholesterol absorption in normoglycemic men. J. Lipid Res. 2004, 45, 507–512. [Google Scholar]
- Mensink, R.P.; Katan, M.B. Effect of dietary fatty acids on serum lipids and lipoproteins. A meta-analysis of 27 trials. Arterioscler. Thromb. 1992, 12, 911–919. [Google Scholar] [CrossRef] [PubMed]
- Escurriol, V.; Cofán, M.; Serra, M.; Bulló, M.; Basora, J.; Salas-Salvadó, J.; Corella, D.; Zazpe, I.; Martínez-González, M.A.; Ruiz-Gutiérrez, V; Estruch, R.; Ros, E. Serum sterol responses to increasing plant sterol intake from natural foods in the Mediterranean diet. Eur. J. Nutr. 2009, 48, 373–382. [Google Scholar] [PubMed]
- Lovejoy, J.C.; Most, M.M.; Lefevre, M.; Greenway, F.L.; Rood, J.C. Effect of diets enriched in almonds on insulin action and serum lipids in adults with normal glucose tolerance or type 2 diabetes. Am. J. Clin. Nutr. 2002, 76, 1000–1006. [Google Scholar]
- Scott, L.W.; Balasubramanyam, A.; Kimball, K.T.; Aherns, A.K.; Fordis, C.M. JR; Ballantyne, C.M. Long-term, randomized clinical trial of two diets in the metabolic syndrome and type 2 diabetes. Diabetes Care 2003, 26, 2481–2482. [Google Scholar] [CrossRef] [PubMed]
- Tapsell, L.C.; Gillen, L.J.; Patch, C.S.; Batterham, M.; Owen, A.; Baré, M.; Kennedy, M. Including walnuts in a low-fat/modified-fat diet improves HDL cholesterol-to-total cholesterol ratios in patients with type 2 diabetes. Diabetes Care 2004, 27, 2777–2783. [Google Scholar]
- Jenkins, D.J.; Kendall, C.W.; Marchie, A.; Josse, A.R.; Nguyen, T.H.; Faulkner, D.A.; Lapsley, K.G.; Singer, W. Effect of almonds on insulin secretion and insulin resistance in nondiabetic hyperlipidemic subjects: a randomized controlled crossover trial. Metabolism 2008, 57, 882–887. [Google Scholar]
- Wien, M.A.; Sabaté, J.M.; Iklé, D.N.; Cole, S.E.; Kandeel, F.R. Almonds vs. complex carbohydrates in a weight reduction program. Int. J. Obes. Relat. Metab. Disord. 2003, 7, 1365–1372, Erratum in: Int. J. Obes. Relat. Metab. Disord. 2004, 28, 459. [Google Scholar]
- Pieters, M.; Oosthuizen, W.; Jerling, J.C.; Loots, D.T.; Mukuddem-Petersen, J.; Hanekom, S.M. Clustering of haemostatic variables and the effect of high cashew and walnut diets on these variables in metabolic syndrome patients. Blood Coagul. Fibrinolysis 2005, 16, 429–437. [Google Scholar]
- Brennan, A.M.; Sweeney, L.L.; Liu, X; Mantzoros, C.S. Walnut consumption increases satiation but has no effect on insulin resistance or the metabolic profile over a 4-day period. Obesity(Silver Spring) 2010, 18, 1176–1182. [Google Scholar] [PubMed]
- Jenkins, D.J.; Kendall, C.W.; Josse, A.R.; Salvatore, S.; Brighenti, F.; Augustin, L.S.; Ellis, P.R.; Vidgen, E.; Rao, A.V. lmonds decrease postprandial glycemia, insulinemia, and oxidative damage in healthy individuals. J. Nutr. 2006, 136, 2987–2992. [Google Scholar] [PubMed]
- Josse, A.R.; Kendall, C.W.; Augustin, L.S.; Ellis, P.R.; Jenkins, D.J. Almonds and postprandial glycemia-a dose-response study. Metabolism 2007, 56, 400–404. [Google Scholar]
- Reiter, R.J.; Manchester, L.C.; Tan, D.X. Melatonin in walnuts: Influence on levels of melatonin and total antioxidant capacity of blood. Nutrition 2005, 21, 920–924. [Google Scholar]
- Reaven, P.D.; Witzum, J.L. Oxidized low density lipoproteins in atherogenesis: role of dietary modification. Ann. Rev. Nutr. 1996, 16, 51–71. [Google Scholar]
- Cortés, B.; Núñez, I.; Cofán, M.; Gilabert, R.; Pérez-Heras, A.; Casals, E.; Deulofeu, R.; Ros, E. Acute effects of high-fat meals enriched with walnuts or olive oil on postprandial endothelial function in healthy subjects and patients with hypercholesterolemia. J. Am. Coll. Cardiol. 2006, 48, 1666–1671. [Google Scholar]
- Berry, S.E.; Tydeman, E.A.; Lewis, H.B.; Phalora, R.; Rosborough, J.; Picout, J.R.; Ellis, P.R. Manipulation of lipid bioaccessibility of almond seeds influences postprandial lipemia in healthy human subjects. Am. J. Clin. Nutr. 2008, 88, 922–929. [Google Scholar]
- Torabian, S.; Haddad, E.; Rajaram, S.; Banta, J.; Sabaté, J. Acute effect of nut consumption on plasma total polyphenols, antioxidant capacity and lipid peroxidation. J. Hum. Nutr. Diet 2009, 22, 64–71. [Google Scholar]
- Fitó, M.; Guxens, M.; Corella, D.; Sáez, G.; Estruch, R.; de la Torre, R.; Francés, F.; Cabezas, C.; López-Sabater, M.C.; Marrugat, J; García-Arellano, A; Arós, F.; Ruiz-Gutierrez, V.; Ros, E; Salas-Salvadó, J.; Fiol, M.; Solá, R.; Covas, M.I., on behalf of the PREDIMED Study Investigators. Effect of a traditional Mediterranean diet on lipoprotein oxidation. A randomized, controlled trial. Arch. Intern. Med. 2007, 167, 1195–1203. [Google Scholar] [PubMed]
- López-Uriarte, P.; Nogués, R.; Saez, G.; Bulló, M.; Romeu, M.; Masana, L.; Tormos, C.; Casas-Agustench, P.; Salas-Salvadó, J. Effect of nut consumption on oxidative stress and the endothelial function in metabolic syndrome. Clin. Nutr. 2010, 29, 373–380. [Google Scholar]
- Rahman, I.; Biswas, S.K.; Kirkham, P.A. Regulation of inflammation and redox signaling by dietary polyphenols. Biochem. Pharmacol. 2006, 72, 1439–1452. [Google Scholar]
- Zhao, G.; Etherton, T.D.; Martin, K.R.; West, S.G.; Gillies, P.J.; Kris-Etherton, P.M. Dietary alpha-linolenic acid reduces inflammatory and lipid cardiovascular risk factors in hypercholesterolemic men and women. J. Nutr. 2004, 134, 2991–2997. [Google Scholar]
- Zhao, G.; Etherton, T.D.; Martin, K.R.; Gillies, P.J.; West, S.G.; Kris-Etherton, P.M. Dietary α-linolenic acid inhibits proinflammatory cytokine production by peripheral blood mononuclear cells in hypercholesterolemic subjects. Am. J. Clin. Nutr. 2004, 85, 385–391. [Google Scholar]
- Papoutsi, Z.; Kassi, E.; Chinou, I.; Halabalaki, M.; Skaltsounis, L.A.; Moutsatsou, P. Walnut extract (Juglans regia L.) and its component ellagic acid exhibit anti-inflammatory activity in human aorta endothelial cells and osteoblastic activity in the cell line KS483. Br. J. Nutr. 2008, 99, 715–722. [Google Scholar] [PubMed]
- Karlsson, S.; Nånberg, E.; Fjaeraa, C.; Wijkander, J. Ellagic acid inhibits lipopolysaccharide-induced expression of enzymes involved in the synthesis of prostaglandin E2 in human monocytes. Br. J. Nutr. 2010, 103, 1102–1109. [Google Scholar]
- Ros, E.; Núñez, I.; Pérez-Heras, A.; Serra, M.; Gilabert, R.; Casals, E.; Deulofeu, R. A walnut diet improves endothelial function in hypercholesterolemic subjects. Circulation 2004, 109, 1609–1614. [Google Scholar]
- Rajaram, S.; Connell, K.M.; Sabaté, J. Effect of almond-enriched high-monounsaturated fat diet on selected markers of inflammation: a randomised, controlled, crossover study. Br. J. Nutr. 2010, 103, 907–912. [Google Scholar]
- Jiménez-Gómez, Y.; López-Miranda, J.; Blanco-Colio, L.M.; Marín, C.; Pérez-Martínez, P.; Ruano, J.; Paniagua, J.A.; Rodríguez, F.; Egido, J.; Pérez-Jiménez, F. Olive oil and walnut breakfasts reduce the postprandial inflammatory response in mononuclear cells compared with a butter breakfast in healthy men. Atherosclerosis 2009, 204, e70–e76. [Google Scholar]
- Mena, M.P.; Sacanella, E.; Vázquez-Agell, M.; Morales, M.; Fitó, M.; Escoda, R.; Serrano-Martínez, M.; Serra, M.; Salas-Salvadó, J.; Benages, N.; Casas, R.; Lamuela-Raventós, R.M.; Masanes, F.; Ros, E.; Estruch, R. Inhibition of circulating immune cell activation, a molecular antiinflammatory effect of the Mediterranean diet. Am. J. Clin. Nutr. 2009, 89, 248–256. [Google Scholar]
- Deanfield, J.E.; Halcox, J.P.; Rabelink, T.J. Endothelial function and dysfunction. Testing and clinical relevance. Circulation 2007, 115, 1285–1295. [Google Scholar]
- Brown, A.A.; Hu, F.B. Dietary modulation of endothelial function: implications for cardiovascular disease. Am. J. Clin. Nutr. 2001, 73, 673–686. [Google Scholar]
- West, S.G. Effect of diet on vascular reactivity: an emerging marker for vascular risk. Curr. Atheroscler. Rep. 2001, 3, 446–455. [Google Scholar]
- Sanderson, P.; Olthof, M.; Grimble, R.F.; Calder, P.C.; Griffin, B.A.; de Roos, N.M.; Belch, J.J.; Muller, D.P.; Vita, J.A. Dietary lipids and vascular function: UK Food Standards Agency workshop report. Br. J. Nutr. 2004, 91, 491–500. [Google Scholar]
- De Koning, E.J.P.; Rabelink, T.J. Endothelial function in the postprandial state. Atherosclerosis Suppl. 2002, 3, 11–16. [Google Scholar]
- Kay, C.D.; Kris-Etherton, P.M.; West, S.G. Effects of antioxidant-rich foods on vascular reactivity: review of the clinical evidence. Curr. Atheroscler. Rep. 2006, 8, 510–522. [Google Scholar]
- Vita, J.A. Polyphenols and cardiovascular disease: effects on endothelial and platelet function. Am. J. Clin. Nutr. 2005, 81, 292S–297S. [Google Scholar]
- Gornik, H.L.; Creager, M.A. Arginine and endothelial and vascular health. J. Nutr. 2004, 134, 2880S–2887S. [Google Scholar]
- West, S.G.; Hecker, K.D.; Mustad, V.A.; Nicholson, S.; Schoemer, S.L.; Wagner, P.; Hinderliter, A.L.; Ulbrecht, J.; Ruey, P.; Kris-Etherton, P.M. Acute effects of monounsaturated fatty acids with and without omega-3 fatty acids on vascular reactivity in individuals with type 2 diabetes. Diabetologia 2005, 48, 113–122. [Google Scholar]
- Sari, I.; Baltaci, Y.; Bagci, C.; Davutoglu, V.; Erel, O.; Celik, H.; Ozer, O.; Aksoy, N.; Aksoy, M. Effect of pistachio diet on lipid parameters, endothelial function, inflammation, and oxidative status: a prospective study. Nutrition 2010, 26, 399–404. [Google Scholar]
- Davis, P.; Valacchi, G.; Pagnin, E.; Shao, Q.; Gross, H.B.; Calo, L.; Yokoyama, W. Walnuts reduce aortic ET-1 mRNA levels in hamsters fed a high-fat, atherogenic diet. J. Nutr. 2006, 136, 428–432. [Google Scholar]
- Barceló, F.; Perona, J.S.; Prades, J.; Funari, S.S.; Gómez-Gracia, E.; Conde, M.; Estruch, R.; Ruiz-Gutiérrez, V. Mediterranean-style diet effect on the structural properties of the erythrocyte cell membrane of hypertensive patients: the Prevención con Dieta Mediterránea Study. Hypertension 2009, 54, 1143–1150. [Google Scholar]
- Molyneux, R.J.; Mahoney, N.; Kim, J.H.; Campbell, B.C. Mycotoxins in edible tree nuts. Int. J. Food Microbiol. 2007, 119, 72–78. [Google Scholar]
- Dorner, J.W. Management and prevention of mycotoxins in peanuts. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess 2008, 25, 203–208. [Google Scholar]
- Bes-Rastrollo, M.; Sabaté, J.; Gomez-Gracia, E.; Alonso, A.; Martinez, J.A.; Martinez-Gonzalez, M.A. Nut consumption and weight gain in a Mediterranean cohort: the SUN Study. Obesity (Silver Spring) 2007, 15, 107–116. [Google Scholar] [PubMed]
- Bes-Rastrollo, M.; Wedick, N.M.; Martinez-Gonzalez, M.A.; Li, T.Y.; Sampson, L.; Hu, F.B. Prospective study of nut consumption, long-term weight change, and obesity risk in women. Am. J. Clin. Nutr. 2009, 89, 1913–1919. [Google Scholar]
- Casas-Agustench, P.; Bulló, M.; Ros, E.; Basora, J.; Salas-Salvadó, J. on behalf of the Nureta-PREDIMED investigators. Cross-sectional association of nut intake with adiposity in a Mediterranean population. Nutr. Metab. Cardiovasc. Dis. 2010. [Epub ahead of print]. [Google Scholar] [PubMed]
- Salas-Salvadó, J.; Fernández-Ballart, J.; Ros, E.; Martínez-González, M.A.; Fitó, M.; Estruch, R.; Corella, D.; Fiol, M.; Gómez-Gracia, E.; Arós, F.; Flores, G.; Lapetra, J.; Lamuela-Raventós, R.; Ruiz-Gutiérrez, V.; Bulló, M.; Basora, J.; Covas, M.I. PREDIMED Study Investigators. A Mediterranean diet supplemented with nuts improves metabolic syndrome status. One-year results of the PREDIMED randomized trial. Arch. Intern. Med. 2008, 158, 2449–2458. [Google Scholar]
- Alper, C.M.; Mattes, R.D. Effects of chronic peanut consumption on energy balance and hedonics. Int. J. Obesity Relat. Metab. Disord. 2002, 26, 1129–1137. [Google Scholar]
- Fraser, G.E.; Bennett, H.W.; Jaceldo, K.B.; Sabaté, J. Effect on body weight of a free 76 Kilojoule (320 calorie) daily supplement of almonds for six months. J. Am. Coll. Nutr. 2002, 21, 275–283. [Google Scholar]
- Sabaté, J.; Cordero-Macintyre, Z.; Siapco, G.; Torabian, S.; Haddad, E. Does regular walnut consumption lead to weight gain? Br. J. Nutr. 2005, 94, 859–864. [Google Scholar] [CrossRef] [PubMed]
- Hollis, J.; Mattes, R. Effect of chronic consumption of almonds on body weight in healthy humans. Br. J. Nutr. 2007, 98, 651–656. [Google Scholar]
- McManus, K.; Antinoro, L.; Sacks, F. A randomized controlled trial of a moderate-fat, low-energy diet compared with a low fat, low-energy diet for weight loss in overweight adult. Int. J. Obesity Relat. Metab. Disord. 2001, 25, 1503–1511. [Google Scholar]
- Wien, M.A.; Sabaté, J.; Iklé, D.N.; Cole, S.E.; Kandeel, F.R. Almonds vs. complex carbohydrates in a weight reduction program. Int. J. Obesity Relat. Metab. Disord. 2003, 7, 1365–1372. [Google Scholar]
- Jaceldo-Siegl, K.; Sabaté, J.; Rajaram, S.; Fraser, G.E. Long-term almond supplementation without advice on food replacement induces favourable nutrient modifications to the habitual diets of free-living individuals. Br. J. Nutr. 2004, 92, 533–540. [Google Scholar]
- Casas-Agustench, P.; López-Uriarte, P.; Bulló, M.; Ros, E.; Gómez-Flores, A.; Salas-Salvadó, J. Acute effects of high-fat meals on energy expenditure, substrate oxidation and satiety: polyunsaturated (walnuts) vs. monounsaturated (virgin olive oil) vs. saturated (fat-rich dairy products) fatty acids. Clin. Nutr. 2009, 28, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Ellis, P.R.; Kendall, C.W.; Ren, Y.; Parker, C.; Pacy, J.F.; Waldron, K.W.; Jenkins, D.J. Role of cell walls in the bioaccessibility of lipids in almond seeds. Am. J. Clin. Nutr. 2004, 80, 604–613. [Google Scholar]
- Cassady, B.A.; Hollis, J.H.; Fulford, A.D.; Considine, R.V.; Mattes, R.D. Mastication of almonds: effects of lipid bioaccessibility, appetite, and hormone respons. Am. J. Clin. Nutr. 2009, 89, 794–800. [Google Scholar]
- Crespo, J.F.; James, J.M.; Fernandez-Rodriguez, C.; Rodriguez, J. Food allergy: nuts and tree nuts. Br. J. Nutr. 2006, 96, S95–S102. [Google Scholar]
- Zuidmeer, L.; Goldhahn, K.; Rona, R.J.; Gislason, D.; Madsen, C.; Summers, C.; Sodergren, E.; Dahlstrom, J.; Lindner, T.; Sigurdardottir, S.T.; McBride, D.; Keil, T. The prevalence of plant food allergies: A systematic review. J. Allergy Clin. Immunol. 2008, 121, 1210–1218. [Google Scholar]
- Summers, C.W.; Pumphrey, R.S.; Woods, C.N.; McDowell, G.; Pemberton, P.W.; Arkwright, P.D. Factors predicting anaphylaxis to peanuts and tree nuts in patients referred to a specialist center. J. Allergy Clin. Immunol. 2008, 121, 632–638. [Google Scholar]
- Sabaté, J. The contribution of vegetarian diets to health and disease: a paradigm shift? Am. J. Clin. Nutr. 2003, 78, S502–S507. [Google Scholar]
- Sofi, F.; Cesari, F.; Abbate, R.; Gensini, G.F.; Casini, A. Adherence to Mediterranean diet and health status: meta-analysis. Br. Med. J. 2008, 337, a1344. [Google Scholar]
- Mente, A.; de Koning, L.; Shannon, H.S.; Anand, S.S. A systematic review of the evidence supporting a causal link between dietary factors and coronary heart disease. Arch. Intern. Med. 2009, 169, 659–669. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an Open Access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ros, E. Health Benefits of Nut Consumption. Nutrients 2010, 2, 652-682. https://doi.org/10.3390/nu2070652
Ros E. Health Benefits of Nut Consumption. Nutrients. 2010; 2(7):652-682. https://doi.org/10.3390/nu2070652
Chicago/Turabian StyleRos, Emilio. 2010. "Health Benefits of Nut Consumption" Nutrients 2, no. 7: 652-682. https://doi.org/10.3390/nu2070652
APA StyleRos, E. (2010). Health Benefits of Nut Consumption. Nutrients, 2(7), 652-682. https://doi.org/10.3390/nu2070652






