Vitamin D in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome After COVID-19 or Vaccination: A Randomized Controlled Trial
Abstract
1. Introduction
2. Methods
2.1. Study Design
2.2. Sample Size Calculation
2.3. Randomization
2.4. Safety Monitoring
2.5. Data Collection and Management
2.6. Statistical Analysis
3. Results
3.1. Participant Characteristics
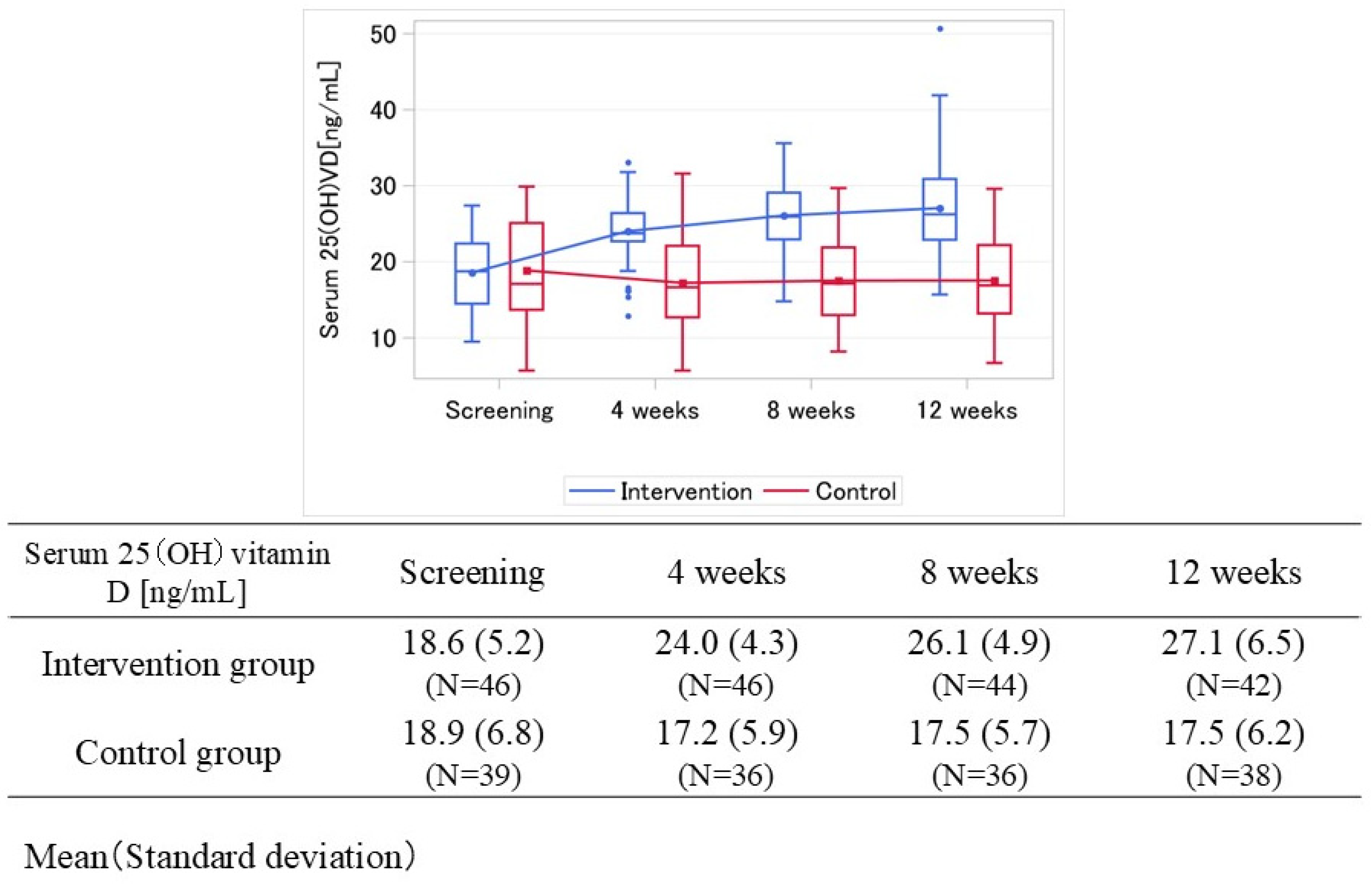
3.2. Temporal Changes in Laboratory Values and Symptoms
3.3. Primary Endpoint Results
3.4. Secondary Endpoints Results
3.5. Subgroup Analyses
3.6. Safety Evaluation
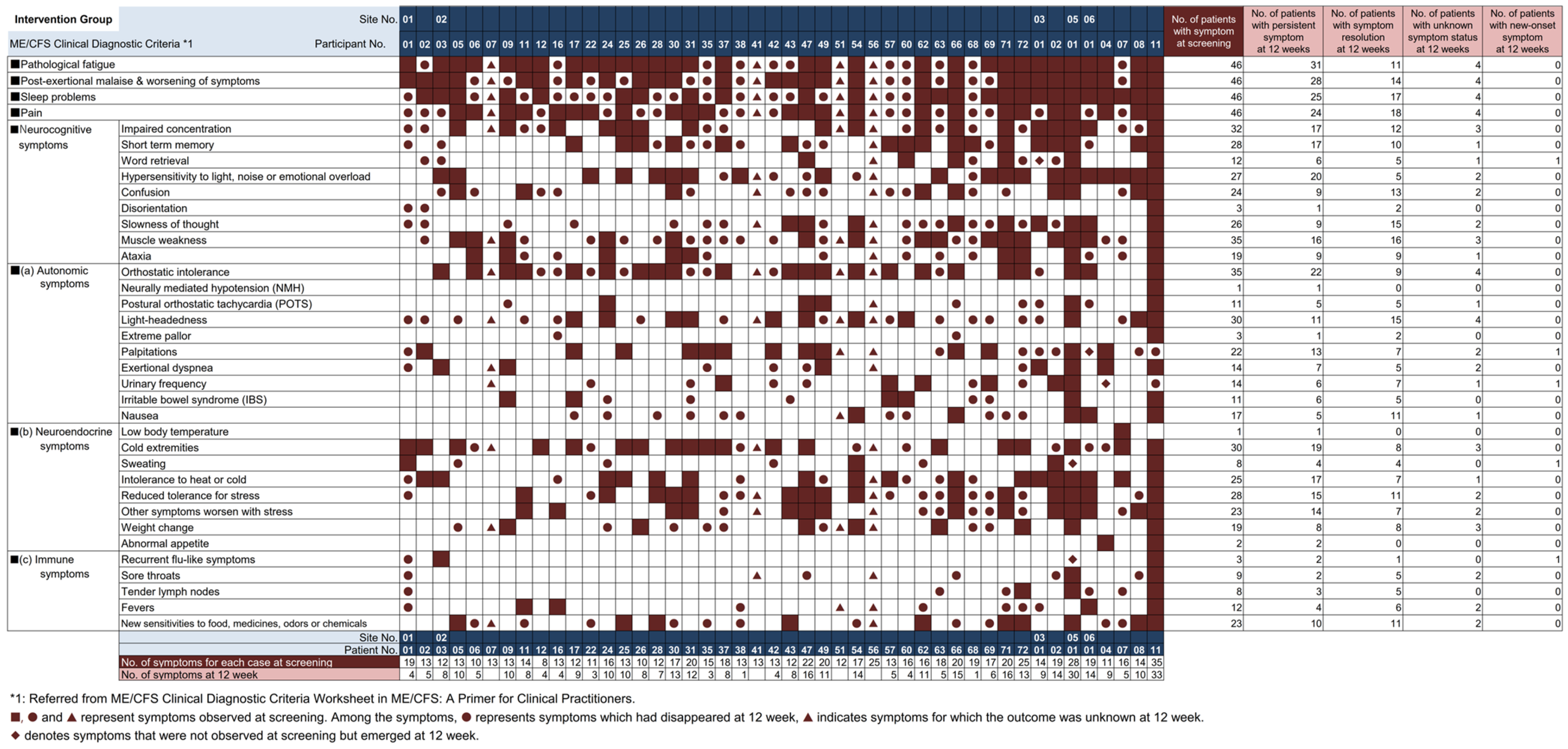
3.7. Symptoms at Screening and Week 12
3.8. Factors Affecting Time to Achievement of <8 ME/CFS Symptoms
4. Discussion
5. Limitations of the Study
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization (WHO). WHO Director-General’s Opening Remarks at the Media Briefing on COVID-19—11 March 2020. Available online: https://www.who.int/news-room/speeches/item/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020 (accessed on 26 November 2025).
- Soriano, J.B.; Murthy, S.; Marshall, J.C.; Relan, P.; Diaz, J.V.; WHO Clinical Case Definition Working Group on Post-COVID-19 Condition. A clinical case definition of post-COVID-19 condition by a Delphi consensus. Lancet Infect. Dis. 2022, 22, e102–e107. [Google Scholar] [CrossRef]
- Thaweethai, T.; Jolley, S.E.; Karlson, E.W.; Levitan, E.B.; Levy, B.; McComsey, G.A.; McCorkell, L.; Nadkarni, G.N.; Parthasarathy, S.; Singh, U.; et al. Development of a Definition of Postacute Sequelae of SARS-CoV-2 Infection. JAMA 2023, 329, 1934–1946. [Google Scholar] [CrossRef] [PubMed]
- Bonilla, H.; Quach, T.C.; Tiwari, A.; Bonilla, A.E.; Miglis, M.; Yang, P.C.; Eggert, L.E.; Sharifi, H.; Horomanski, A.; Subramanian, A.; et al. Myalgic Encephalomyelitis/Chronic Fatigue Syndrome is common in post-acute sequelae of SARS-CoV-2 infection (PASC): Results from a post-COVID-19 multidisciplinary clinic. Front. Neurol. 2023, 14, 1090747. [Google Scholar] [CrossRef] [PubMed]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Moreira, E.D.; Zerbini, C.; et al. Safety and Efficacy of the BNT162b2 mRNA COVID-19 Vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef] [PubMed]
- Konishi, N.; Hirai, Y.; Hikota, H.; Miyahara, S.; Fujisawa, A.; Motohashi, H.; Ueda, J.; Inoue, M.; Fukushima, M. Side effects of COVID-19 vaccines: Academic reports presented at medical conferences in Japan and a PubMed Survey. Clin. Eval. 2024, 51, 479–521. (In Japanese) [Google Scholar]
- Kodama, S.; Konishi, N.; Hirai, Y.; Fujisawa, A.; Nakata, M.; Teramukai, S.; Fukushima, M. Efficacy of vitamin D replacement therapy on 28 cases of myalgic encephalomyelitis/chronic fatigue syndrome after COVID-19 vaccination. Nutrition 2025, 134, 112718. [Google Scholar] [CrossRef]
- Ruiz-Pablos, M.; Paiva, B.; Zabaleta, A. Hypocortisolemic ASIA: A vaccine- and chronic infection-induced syndrome behind the origin of long COVID and myalgic encephalomyelitis. Front. Immunol. 2024, 15, 1422940. [Google Scholar] [CrossRef]
- di Filippo, L.; Frara, S.; Nannipieri, F.; Cotellessa, A.; Locatelli, M.; Rovere Querini, P.; Giustina, A. Low Vitamin D Levels Are Associated With Long COVID Syndrome in COVID-19 Survivors. J. Clin. Endocrinol. Metab. 2023, 108, e1106–e1116. [Google Scholar] [CrossRef]
- International Association for Chronic Fatigue Syndrome/Myalgic Encephalomyelitis. Chronic Fatigue Syndrome Myalgic Encephalomyelitis: A Primer for Clinical Practitioners 2014 Edition [in Japanese] n.d; Development of Medical Network Model for Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS), Japan Agency for Medical Research and Development (AMED), Translator; International Association for Chronic Fatigue Syndrome/Myalgic Encephalomyelitis (IACFS/ME): Chicago, IL, USA, 2014. [Google Scholar]
- International Association for Chronic Fatigue Syndrome/Myalgic Encephalomyelitis; Friedburg, F.; Bateman, L.; Bested, A.C.; Davenport, T.; Friedman, K.J.; Gurwitt, A.; Jason, L.A.; Lapp, C.W.; Stevens, S.R.; et al. ME/CFS: A Primer for Clinical Practitioners 2014 Edition; International Association for Chronic Fatigue Syndrome/Myalgic Encephalomyelitis: Stony Brook, NY, USA, 2014. [Google Scholar]
- Teijin Pharma Limited. Onealfa®Tablets 0.25µg, 0.5µg, 1.0µg Package Insert; Teijin Pharma Limited: Tokyo, Japan, 2024. (In Japanese) [Google Scholar]
- Okazaki, R.; Ozono, K.; Fukumoto, S.; Inoue, D.; Yamauchi, M.; Minagawa, M.; Michigami, T.; Takeuchi, Y.; Matsumoto, T.; Sugimoto, T. Assessment criteria for vitamin D deficiency/insufficiency in Japan: Proposal by an expert panel supported by the Research Program of Intractable Diseases, Ministry of Health, Labour and Welfare, Japan, the Japanese Society for Bone and Mineral Research and the Japan Endocrine Society [Opinion]. J. Bone Miner. Metab. 2017, 35, 1–5. [Google Scholar] [CrossRef]
- Bested, A.C.; Marshall, L.M. Review of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: An evidence-based approach to diagnosis and management by clinicians. Rev. Environ. Health 2015, 30, 223–249. [Google Scholar] [CrossRef]
- Kingdon, C.; Lowe, A.; Shepherd, C.; Nacul, L. What Primary Care Practitioners Need to Know about the New NICE Guideline for Myalgic Encephalomyelitis/Chronic Fatigue Syndrome in Adults. Healthcare 2022, 10, 2438. [Google Scholar] [CrossRef]
- Sherif, Z.A.; Gomez, C.R.; Connors, T.J.; Henrich, T.J.; Reeves, W.B. RECOVER Mechanistic Pathway Task Force. Pathogenic mechanisms of post-acute sequelae of SARS-CoV-2 infection (PASC). eLife 2023, 12, e86002. [Google Scholar] [CrossRef] [PubMed]
- Mishra, R.; Banerjea, A.C. SARS-CoV-2 Spike Targets USP33-IRF9 Axis via Exosomal miR-148a to Activate Human Microglia. Front. Immunol. 2021, 12, 656700. [Google Scholar] [CrossRef] [PubMed]
- Parry, P.I.; Lefringhausen, A.; Turni, C.; Neil, C.J.; Cosford, R.; Hudson, N.J.; Gillespie, J. ‘Spikeopathy’: COVID-19 Spike Protein Is Pathogenic, from Both Virus and Vaccine mRNA. Biomedicines 2023, 11, 2287. [Google Scholar] [CrossRef]
- Miller, C.M.; Moen, J.K.; Iwasaki, A. The lingering shadow of epidemics: Post-acute sequelae across history. Trends Immunol. 2026, 47, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Santos Guedes de Sa, K.; Silva, J.; Bayarri-Olmos, R.; Brinda, R.; Alec Rath Constable, R.; Colom Diaz, P.A.; Kwon, D.-I.; Rodrigues, G.; Wenxue, L.; Baker, C.; et al. A causal link between autoantibodies and neurological symptoms in long COVID. medRxiv 2024. [Google Scholar] [CrossRef]
- Fujii, H.; Sato, W.; Kimura, Y.; Matsuda, H.; Ota, M.; Maikusa, N.; Suzuki, F.; Amano, K.; Shin, I.; Yamamura, T.; et al. Altered Structural Brain Networks Related to Adrenergic/Muscarinic Receptor Autoantibodies in Chronic Fatigue Syndrome. J. Neuroimaging 2020, 30, 822–827. [Google Scholar] [CrossRef]
- Sato, W.; Ono, H.; Matsutani, T.; Nakamura, M.; Shin, I.; Amano, K.; Suzuki, R.; Yamamura, T. Skewing of the B cell receptor repertoire in myalgic encephalomyelitis/chronic fatigue syndrome. Brain Behav. Immun. 2021, 95, 245–255. [Google Scholar] [CrossRef]
- Chen, T.-B.; Chang, C.-M.; Yang, C.-C.; Tsai, I.-J.; Wei, C.-Y.; Yang, H.-W.; Yang, C.-P. Neuroimmunological Effect of Vitamin D on Neuropsychiatric Long COVID Syndrome: A Review. Nutrients 2023, 15, 3802. [Google Scholar] [CrossRef]
- Jeffery, L.E.; Burke, F.; Mura, M.; Zheng, Y.; Qureshi, O.S.; Hewison, M.; Walker, L.S.K.; A Lammas, D.; Raza, K.; Sansom, D.M. 1,25-Dihydroxyvitamin D3 and IL-2 combine to inhibit T cell production of inflammatory cytokines and promote development of regulatory T cells expressing CTLA-4 and FoxP3. J. Immunol. 2009, 183, 5458–5467. [Google Scholar] [CrossRef]
- Chen, S.; Sims, G.P.; Chen, X.X.; Gu, Y.Y.; Chen, S.; Lipsky, P.E. Modulatory effects of 1,25-dihydroxyvitamin D3 on human B cell differentiation. J. Immunol. 2007, 179, 1634–1647. [Google Scholar] [CrossRef] [PubMed]
- Penna, G.; Adorini, L. 1 Alpha,25-dihydroxyvitamin D3 inhibits differentiation, maturation, activation, and survival of dendritic cells leading to impaired alloreactive T cell activation. J. Immunol. 2000, 164, 2405–2411. [Google Scholar] [CrossRef] [PubMed]
- Kurotori, I.; Sasao, W.; Abe, M. Recovery from Myalgic Encephalomyelitis/Chronic Fatigue syndrome developed after severe acute respiratory syndrome coronavirus 2 vaccination: A case report. PCN Rep. 2025, 4, e70111. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Vitamin D: Important for prevention of osteoporosis, cardiovascular heart disease, type 1 diabetes, autoimmune diseases, and some cancers. South Med. J. 2005, 98, 1024–1027. [Google Scholar] [CrossRef]
- Khanorkar, P.; Gupta, S.; Jani, P.; Deepak, A.; Podder, A. Evaluation of serum Vitamin-D levels in non-specific chronic musculoskeletal pain. Bioinformation 2025, 21, 2740–2743. [Google Scholar] [CrossRef]
- Holick, M.F. The Vitamin D Deficiency Pandemic: A Forgotten Hormone Important for Health. Public Health Rev. 2010, 32, 267–283. [Google Scholar] [CrossRef]
- Bischoff-Ferrari, H.A.; Willett, W.C.; Orav, E.J.; Lips, P.; Meunier, P.J.; Lyons, R.A.; Flicker, L.; Wark, J.; Jackson, R.D.; Cauley, J.A.; et al. A pooled analysis of vitamin D dose requirements for fracture prevention. N. Engl. J. Med. 2012, 367, 40–49. [Google Scholar] [CrossRef]
- Reddy, P.; Edwards, L.R. Magnesium Supplementation in Vitamin D Deficiency. Am. J. Ther. 2019, 26, e124–e132. [Google Scholar] [CrossRef]
- Miyamoto, H.; Kawakami, D.; Hanafusa, N.; Nakanishi, T.; Miyasaka, M.; Furutani, Y.; Ikeda, Y.; Ito, K.; Kato, T.; Yokoyama, K.; et al. Determination of a Serum 25-Hydroxyvitamin D Reference Ranges in Japanese Adults Using Fully Automated Liquid Chromatography–Tandem Mass Spectrometry. J. Nutr. 2023, 153, 1253–1264. [Google Scholar] [CrossRef]
- Funaki, T.; Sanpei, M.; Morisaki, N.; Mizoue, T.; Yamaguchi, K. Serious vitamin D deficiency in healthcare workers during the COVID-19 pandemic. BMJ Nutr. Prev. Health 2022, 5, 134–136. [Google Scholar] [CrossRef]
- Papadimitriou, D.T. The Big Vitamin D Mistake. JPMPH 2017, 50, 278–281. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Health, Labour and Welfare (MHLW), Japan. 1-6 Vitamin: Vitamin D. In A Report of the Study Group for Formulation of “Dietary Reference Intakes in Japanese (2020)”; Ministry of Health, Labour and Welfare (MHLW): Tokyo, Japan, 2019; pp. 178–187. (In Japanese) [Google Scholar]
- Sobczak, M.; Pawliczak, R. Effect of Vitamin D3 Supplementation on Severe COVID-19: A Meta-Analysis of Randomized Clinical Trials. Nutrients 2024, 16, 1402. [Google Scholar] [CrossRef] [PubMed]
- Peramaiyan, R.; Anthony, J.; Varalakshmi, S.; Sekar, A.K.; Ali, E.M.; A, A.H.S.; Abdallah, B.M. Comparison of the role of vitamin D in normal organs and those affected by COVID-19. Int. J. Med. Sci. 2025, 22, 240–251. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Huang, Q.; Gu, J.; Hu, W.; Liu, Y.; Hu, Y.; Zhang, Z.-L. Comparison of the effects of cholecalciferol and calcitriol on calcium metabolism and bone turnover in Chinese postmenopausal women with vitamin D insufficiency. Acta Pharmacol. Sin. 2012, 33, 490–495. [Google Scholar] [CrossRef]
- Cozzolino, M.; Bernard, L.; Csomor, P.A. Active vitamin D increases the risk of hypercalcaemia in non-dialysis chronic kidney disease patients with secondary hyperparathyroidism: A systematic review and meta-analysis. Clin. Kidney J. 2021, 14, 2437–2443. [Google Scholar] [CrossRef]
- Shankar, V.; Wilhelmy, J.; Curtis, E.J.; Michael, B.; Cervantes, L.; Mallajosyula, V.; Davis, R.W.; Snyder, M.; Younis, S.; Robinson, W.H.; et al. Oxidative stress is a shared characteristic of ME/CFS and Long COVID. Proc. Natl. Acad. Sci. USA 2025, 122, e2426564122. [Google Scholar] [CrossRef]
- Perico, L.; Morigi, M.; Pezzotta, A.; Locatelli, M.; Imberti, B.; Corna, D.; Cerullo, D.; Benigni, A.; Remuzzi, G. SARS-CoV-2 spike protein induces lung endothelial cell dysfunction and thrombo-inflammation depending on the C3a/C3a receptor signalling. Sci. Rep. 2023, 13, 11392. [Google Scholar] [CrossRef]
- Wirth, K.J.; Löhn, M. Microvascular Capillary and Precapillary Cardiovascular Disturbances Strongly Interact to Severely Affect Tissue Perfusion and Mitochondrial Function in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Evolving from the Post COVID-19 Syndrome. Medicina 2024, 60, 194. [Google Scholar] [CrossRef]
- Barshtein, G.; Arbell, D.; Gural, A.; Livshits, L. Distribution of Red Blood Cells Deformability: Study on Density-Separated Cell Subpopulations. Colloids Interfaces 2024, 8, 47. [Google Scholar] [CrossRef]
- Saha, A.K.; Schmidt, B.R.; Wilhelmy, J.; Nguyen, V.; Abugherir, A.; Do, J.K.; Nemat-Gorgani, M.; Davis, R.W.; Ramasubramanian, A.K. Red blood cell deformability is diminished in patients with Chronic Fatigue Syndrome. Clin. Hemorheol. Microcirc. 2019, 71, 113–116. [Google Scholar] [CrossRef]
- Diederich, L.; Suvorava, T.; Sansone, R.; Keller, T.C.S.; Barbarino, F.; Sutton, T.R.; Kramer, C.M.; Lückstädt, W.; Isakson, B.E.; Gohlke, H.; et al. On the Effects of Reactive Oxygen Species and Nitric Oxide on Red Blood Cell Deformability. Front. Physiol. 2018, 9, 332. [Google Scholar] [CrossRef]
- Ježek, J.; Cooper, K.F.; Strich, R. Reactive Oxygen Species and Mitochondrial Dynamics: The Yin and Yang of Mitochondrial Dysfunction and Cancer Progression. Antioxidants 2018, 7, 13. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, N.; Yokokawa, H.; Mori, H.; Hiki, M.; Tabe, Y.; Takahashi, K.; Naito, T. Association Between Serum Zinc Concentration Levels And Severity Of Coronavirus Disease 2019 (COVID-19) In Japanese Inpatients. Int. J. Gen. Med. 2024, 17, 4745–4753. [Google Scholar] [CrossRef] [PubMed]
- Mitsui, J.; Matsukawa, T.; Uemura, Y.; Kawahara, T.; Chikada, A.; Porto, K.J.L.; Naruse, H.; Tanaka, M.; Ishiura, H.; Toda, T.; et al. High-dose ubiquinol supplementation in multiple-system atrophy: A multicentre, randomised, double-blinded, placebo-controlled phase 2 trial. eClinicalMedicine 2023, 59, 101920. [Google Scholar] [CrossRef] [PubMed]
- Oki, R.; Izumi, Y.; Fujita, K.; Miyamoto, R.; Nodera, H.; Sato, Y.; Sakaguchi, S.; Nokihara, H.; Kanai, K.; Tsunemi, T.; et al. Efficacy and Safety of Ultrahigh-Dose Methylcobalamin in Early-Stage Amyotrophic Lateral Sclerosis: A Randomized Clinical Trial. JAMA Neurol. 2022, 79, 575–583. [Google Scholar] [CrossRef]
- Bjornevik, K.; Cortese, M.; Furtado, J.D.; Paganoni, S.; Schwarzschild, M.A.; Cudkowicz, M.E.; Ascherio, A. Association of Polyunsaturated Fatty Acids and Clinical Progression in Patients With ALS: Post Hoc Analysis of the EMPOWER Trial. Neurology 2023, 101, e690–e698. [Google Scholar] [CrossRef]
- Chaubey, K.; Vázquez-Rosa, E.; Tripathi, S.J.; Shin, M.K.; Yu, Y.; Dhar, M.; Chakraborty, S.; Yamakawa, M.; Wang, X.; Sridharan, P.S.; et al. Pharmacologic reversal of advanced Alzheimer’s disease in mice and identification of potential therapeutic nodes in human brain. Cell Rep. Med. 2026, 7, 102535. [Google Scholar] [CrossRef]
- Heng, B.; Gunasegaran, B.; Krishnamurthy, S.; Bustamante, S.; Pires, A.S.; Chow, S.; Ahn, S.B.; Paul-Heng, M.; Maciver, Y.; Smith, K.; et al. Mapping the complexity of ME/CFS: Evidence for abnormal energy metabolism, altered immune profile, and vascular dysfunction. Cell Rep. Med. 2025, 6, 102514. [Google Scholar] [CrossRef]
- Kitamura, K.; Watanabe, Y.; Kabasawa, K.; Saito, T.; Takahashi, A.; Kobayashi, R.; Oshiki, R.; Watanabe, K.; Takachi, R.; Tsugane, S.; et al. Plasma 25-hydroxyvitamin D concentrations, vitamin D deficiency and mortality in community-dwelling Japanese adults. Br. J. Nutr. 2025, 134, 634–644. [Google Scholar] [CrossRef]
- Niedermaier, T.; Gredner, T.; Kuznia, S.; Schöttker, B.; Mons, U.; Brenner, H. Vitamin D supplementation to the older adult population in Germany has the cost-saving potential of preventing almost 30 000 cancer deaths per year. Mol. Oncol. 2021, 15, 1986–1994. [Google Scholar] [CrossRef]





| Intervention Group (n = 46) | Control Group (n = 39) | Total (n = 85) | |||
|---|---|---|---|---|---|
| Site | Kamata Yoshino Clinic | 2 (4.3%) | 0 (0.0%) | 2 (2.4%) | |
| Kodama Hospital/Kodama Clinic | 36 (78.3%) | 35 (89.7%) | 71 (83.5%) | ||
| Honbetsu Cardiovascular Medicine Clinic | 2 (4.3%) | 0 (0.0%) | 2 (2.4%) | ||
| Kobayashi Clinic | 1 (2.2%) | 0 (0.0%) | 1 (1.2%) | ||
| Tokachi Mutsumino Clinic | 5 (10.9%) | 4 (10.3%) | 9 (10.6%) | ||
| Number of ME/CFS Symptoms | ≤10 | 3 (6.5%) | 4 (10.3%) | 7 (8.2%) | |
| ≥11 | 43 (93.5%) | 35 (89.7%) | 78 (91.8%) | ||
| Sex | Male | 17 (37.0%) | 13 (33.3%) | 30 (35.3%) | |
| Female | 29 (63.0%) | 26 (66.7%) | 55 (64.7%) | ||
| PVS/PASC | PVS | 33 (71.7%) | 23 (59.0%) | 56 (65.9%) | |
| PASC | 13 (28.3%) | 16 (41.0%) | 29 (34.1%) | ||
| Age at consent (years) | Mean (SD) | 48.0 (19.6) | 51.3 (19.8) | 49.5 (19.7) | |
| Days since onset | Median (range) | 1040 (211–1284) | 891 (3–1490) | 983 (3–1490) | |
| Height (cm) | Mean (SD) | 161.9 (8.8) | 160.4 (10.5) | 161.2 (9.5) | |
| Weight (kg) | Mean (SD) | 60.3 (12.5) | 57.0 (13.4) | 58.8 (12.9) | |
| BMI (kg/m2) | Mean (SD) | 22.9 (3.7) | 22.0 (3.8) | 22.5 (3.8) | |
| Comorbidities | No | 26 (56.5%) | 21 (53.8%) | 47 (55.3%) | |
| Yes | 20 (43.5%) | 18 (46.2%) | 38 (44.7%) | ||
| Medical history | No | 37 (80.4%) | 29 (74.4%) | 66 (77.6%) | |
| Yes | 9 (19.6%) | 10 (25.6%) | 19 (22.4%) | ||
| COVID-19 infection history | No | 15 (32.6%) | 12 (30.8%) | 27 (31.8%) | |
| Yes | 31 (67.4%) | 27 (69.2%) | 58 (68.2%) | ||
| Number of infections | 1 time | 24 (77.4%) | 19 (70.4%) | 43 (74.1%) | |
| 2 times | 7 (22.6%) | 7 (25.9%) | 14 (24.1%) | ||
| 3 times | 0 (0.0%) | 1 (3.7%) | 1 (1.7%) | ||
| Time from most recent infection to consent | ≤3 months | 3 (9.7%) | 6 (22.2%) | 9 (15.5%) | |
| 4–6 months | 4 (12.9%) | 2 (7.4%) | 6 (10.3%) | ||
| 7–12 months | 3 (9.7%) | 6 (22.2%) | 9 (15.5%) | ||
| >1 year | 21 (67.7%) | 10 (37.0%) | 31 (53.4%) | ||
| Unknown | 0 (0.0%) | 3 (11.1%) | 3 (5.2%) | ||
| COVID-19 vaccination history | No | 4 (8.7%) | 6 (15.4%) | 10 (11.8%) | |
| Yes | 42 (91.3%) | 33 (84.6%) | 75 (88.2%) | ||
| Number of vaccinations | 1 time | 5 (11.9%) | 0 (0.0%) | 5 (6.7%) | |
| 2 times | 14 (33.3%) | 8 (24.2%) | 22 (29.3%) | ||
| 3 times | 11 (26.2%) | 14 (42.4%) | 25 (33.3%) | ||
| 4 times | 12 (28.6%) | 11 (33.3%) | 23 (30.7%) | ||
| Time from most recent vaccination to consent | 7–12 months | 0 (0.0%) | 1 (3.0%) | 1 (1.3%) | |
| >1 year | 42 (100.0%) | 32 (97.0%) | 74 (98.7%) | ||
| Vaccine manufacturer | Pfizer (Comirnaty) | 34 (81.0%) | 29 (87.9%) | 63 (84.0%) | |
| Takeda/Moderna (Moderna/Spikevax) | 12 (28.6%) | 8 (24.2%) | 20 (26.7%) | ||
| Moderna (Spikevax bivalent) | 4 (9.5%) | 3 (9.1%) | 7 (9.3%) | ||
| Other vaccine | 1 (2.4%) | 0 (0.0%) | 1 (1.3%) | ||
| Unknown | 1 (2.4%) | 5 (15.2%) | 6 (8.0%) | ||
| Symptoms at Screening | Achievement of <8 | Intervention Group (n = 42) | Control Group (n = 38) | Total (n = 80) | p-Value * |
|---|---|---|---|---|---|
| ≤10 (N = 7) † | Yes | 2 (66.7%) | 0 (0.0%) | 2 (28.6%) | <0.001 |
| No | 1 (33.3%) | 4 (100.0%) | 5 (71.4%) | ||
| ≥11 (N = 73) † | Yes | 14 (35.9%) | 1 (2.9%) | 15 (20.5%) | |
| No | 25 (64.1%) | 33 (97.1%) | 58 (79.5%) |
| ME/CFS Clinical Diagnostic Criteria | Patients with Symptoms at Screening | Patients with Continued Symptoms at Week 12 | Patients with Improved Symptoms at Week 12 | Patients with Unknown Symptoms at Week 12 | Symptom Improvement Rate | p-Value * | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Intervention Group | Control Group | Intervention Group | Control Group | Intervention Group | Control Group | Intervention Group | Control Group | Intervention Group | Control Group | ||
| Pathological fatigue | 46 | 39 | 31 | 37 | 11 | 1 | 4 | 1 | 23.9% | 2.6% | 0.005 |
| Post-exertional malaise & worsening of symptoms | 46 | 39 | 28 | 38 | 14 | 0 | 4 | 1 | 30.4% | 0.0% | <0.001 |
| Sleep problems | 46 | 39 | 25 | 32 | 17 | 6 | 4 | 1 | 37.0% | 15.4% | 0.03 |
| Pain | 46 | 38 | 24 | 32 | 18 | 5 | 4 | 1 | 39.1% | 13.2% | 0.01 |
| Neurocognitive symptoms | 46 | 39 | 37 | 38 | 5 | 0 | 4 | 1 | 10.9% | 0.0% | 0.06 |
| Autonomic symptoms | 46 | 39 | 33 | 37 | 9 | 1 | 4 | 1 | 19.6% | 2.6% | 0.02 |
| Neuroendocrine symptoms | 46 | 39 | 35 | 38 | 7 | 0 | 4 | 1 | 15.2% | 0.0% | 0.01 |
| Immune symptoms | 33 | 25 | 14 | 18 | 15 | 6 | 4 | 1 | 45.5% | 24.0% | 0.11 |
| Hazard Ratio | 95% CI | p-Value | ||
|---|---|---|---|---|
| Number of symptoms at baseline | Per symptom | 0.87 | 0.74–1.01 | 0.07 |
| Treatment group | Control group vs. Intervention group | 0.06 | 0.01–0.43 | 0.006 |
| PVS/PASC | PASC vs. PVS | 0.29 | 0.07–1.09 | 0.07 |
| Comorbidities | Yes vs. No | 0.29 | 0.09–0.89 | 0.03 |
| MCHC | Per 1% | 0.51 | 0.30–0.85 | 0.010 |
| T-Bil | Per 1 mg/dL | 7.03 | 0.99–50.0 | 0.05 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Kodama, S.; Nakata, M.; Konishi, N.; Yoshino, M.; Fujisawa, A.; Naganuma, M.; Kobayashi, Y.; Hirai, Y.; Kitagawa, A.; Miyokawa, M.; et al. Vitamin D in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome After COVID-19 or Vaccination: A Randomized Controlled Trial. Nutrients 2026, 18, 521. https://doi.org/10.3390/nu18030521
Kodama S, Nakata M, Konishi N, Yoshino M, Fujisawa A, Naganuma M, Kobayashi Y, Hirai Y, Kitagawa A, Miyokawa M, et al. Vitamin D in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome After COVID-19 or Vaccination: A Randomized Controlled Trial. Nutrients. 2026; 18(3):521. https://doi.org/10.3390/nu18030521
Chicago/Turabian StyleKodama, Shinichiro, Mitsuko Nakata, Nafuko Konishi, Masato Yoshino, Akinori Fujisawa, Mutsuo Naganuma, Yuki Kobayashi, Yuriko Hirai, Akiko Kitagawa, Mariko Miyokawa, and et al. 2026. "Vitamin D in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome After COVID-19 or Vaccination: A Randomized Controlled Trial" Nutrients 18, no. 3: 521. https://doi.org/10.3390/nu18030521
APA StyleKodama, S., Nakata, M., Konishi, N., Yoshino, M., Fujisawa, A., Naganuma, M., Kobayashi, Y., Hirai, Y., Kitagawa, A., Miyokawa, M., Mishima, R., Teramukai, S., & Fukushima, M. (2026). Vitamin D in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome After COVID-19 or Vaccination: A Randomized Controlled Trial. Nutrients, 18(3), 521. https://doi.org/10.3390/nu18030521





