An Overview of Sarcopenia: Focusing on Nutritional Treatment Approaches
Abstract
1. Introduction
2. Definition
3. Classification
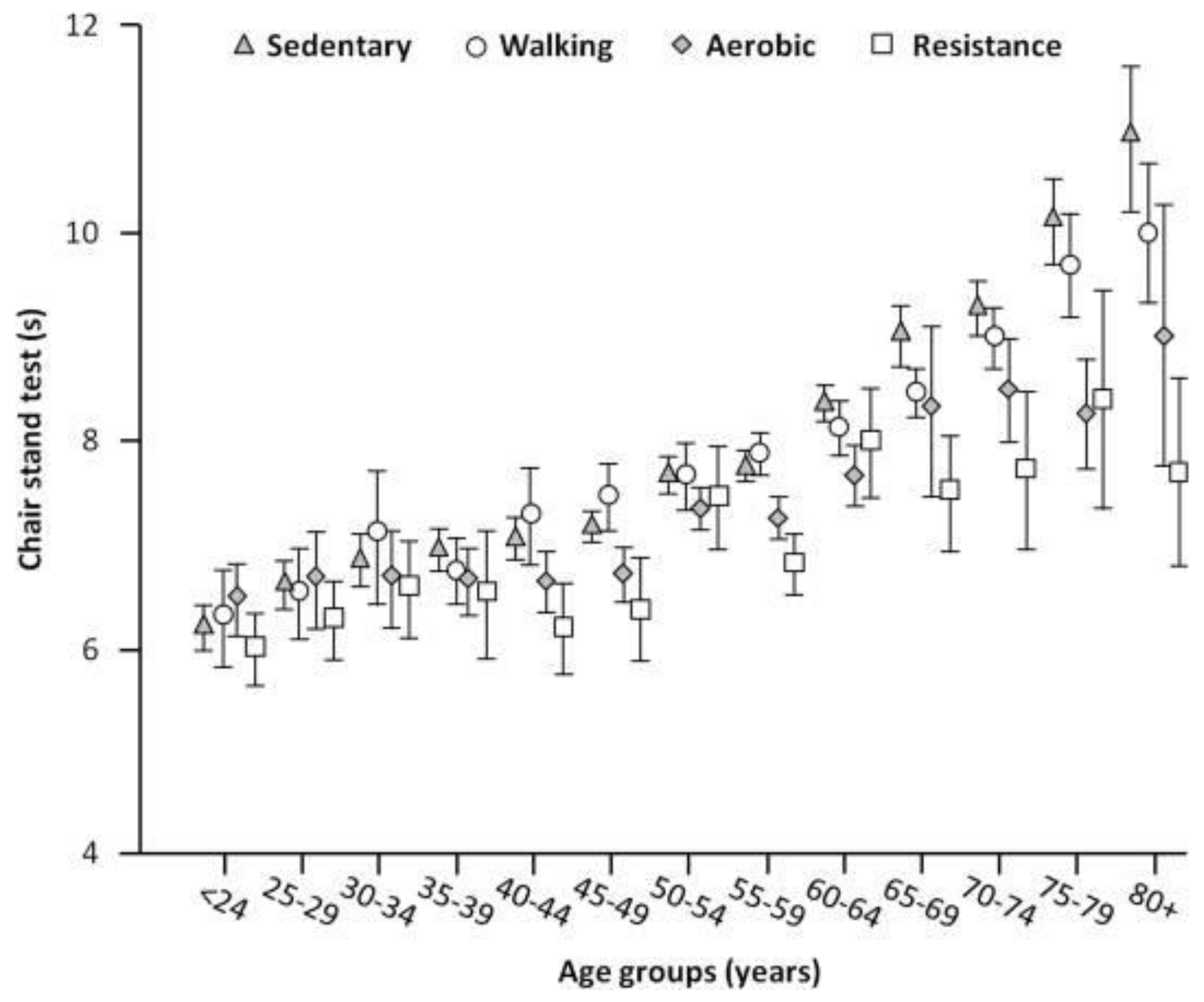
4. Epidemiology
5. Physiopathological Aspects
6. Primary Sarcopenia: The Physiological Process of Aging
7. Secondary Sarcopenia
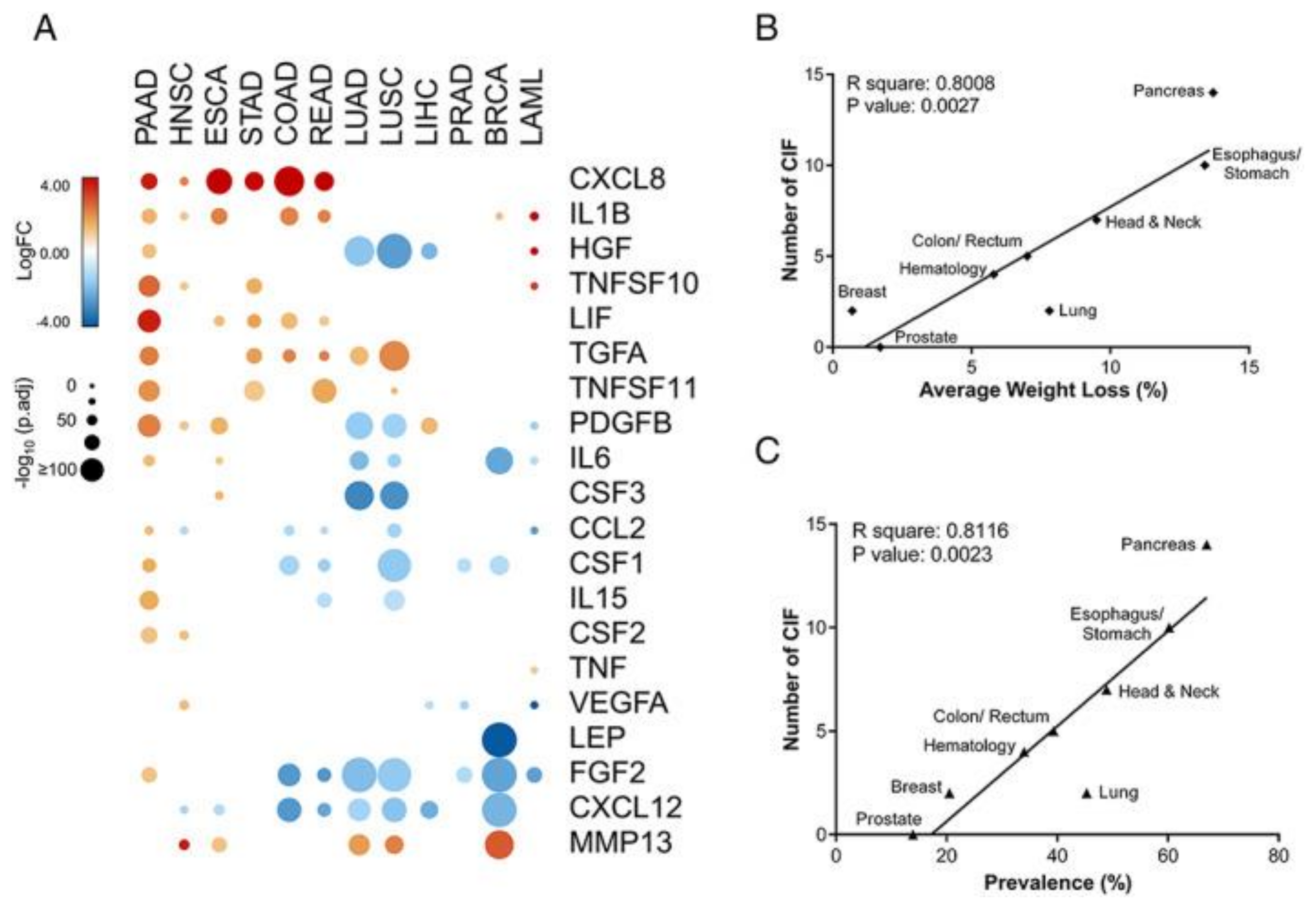
Cancer-Related Sarcopenia
8. The Nutritional Treatment of Sarcopenia
8.1. Specific Dietary Components and Protein Synthesis
8.1.1. Branched-Chain Amino Acids and Whey Proteins
8.1.2. β-Hydroxy-β-Methylbutyrate
8.1.3. Omega-3 Fatty Acids
8.1.4. Vitamin D
8.2. Foods for Special Medical Purposes
9. Disease Specific Aspects and Nutritional Treatment
9.1. Nutritional Treatment in Cancer Patients
9.2. Nutrition in Kidney Diseases and Cirrhosis
10. Future Perspectives
11. Limitations and Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| PI3K | phosphoinositide-3-kinase |
| IGF-1 | insulin-like growth factor |
| PIP3 | phosphoinositide-3,4,5-trisphosphate |
| PDK1 | phosphoinositide-dependent kinase 1 |
| FoxO | Forkhead Box |
| mTOR | mammalian target of rapamycin |
| MuRF1 | muscle ring finger 1 |
| MAFbx | muscle atrophy F-box |
| mTORC1 | mTOR Complex 1 |
| mTORC2 | mTOR Complex 2 |
| GDF-8 | growth and differentiation factor 8 |
| TGF-β | transforming growth factor beta |
| TNF-α | tumor necrosis factor alpha |
| CIFs | cachexia-inducing factors |
| UCP1 | uncoupling protein 1 |
| LMF | lipid-mobilizing factors |
| HMB | β-hydroxy-β-methylbutyrate |
| FMSPs | food for special medical purposes |
| MT-ONS | targeted oral nutritional supplementation |
| PAL | physical activity level |
| REE | resting energy expenditure |
| TEE | total energy expenditure |
| EPA | eicosapentaenoic acid |
References
- Harris, T.; Woteki, C.; Briefel, R.R.; Kleinman, J.C. NHANES III for older persons: Nutrition content and methodological considerations. Am. J. Clin. Nutr. 1989, 50, 1145–1149. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, I.H.; Roubenoff, R. Stalking sarcopenia. Ann. Intern. Med. 1995, 123, 727–728. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.; Boirie, Y.; Cederholm, T.; Landi, F.; Martin, F.C.; Michel, J.P.; Rolland, Y.; Schneider, S.M.; et al. European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.K.; Liu, L.K.; Woo, J.; Assantachai, P.; Auyeung, T.W.; Bahyah, K.S.; Chou, M.Y.; Chen, L.Y.; Hsu, P.S.; Krairit, O.; et al. Sarcopenia in Asia: Consensus report of the Asian Working Group for Sarcopenia. J. Am. Med. Dir. Assoc. 2014, 15, 95–101. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef]
- Smith, C.; Woessner, M.N.; Sim, M.; Levinger, I. Sarcopenia definition: Does it really matter? Implications for resistance training. Ageing Res. Rev. 2022, 78, 101617. [Google Scholar] [CrossRef]
- Goodpaster, B.H.; Park, S.W.; Harris, T.B.; Kritchevsky, S.B.; Nevitt, M.; Schwartz, A.V.; Simonsick, E.M.; Tylavsky, F.A.; Visser, M.; Newman, A.B. The loss of skeletal muscle strength, mass, and quality in older adults: The Health, Aging and Body Composition Study. J. Gerontol. A Biol. Sci. Med. Sci. 2006, 61, 1059–1064. [Google Scholar] [CrossRef]
- Janssen, I.; Baumgartner, R.N.; Ross, R.; Rosenberg, I.H.; Roubenoff, R. Skeletal muscle cutpoints associated with elevated physical disability risk in older men and women. Am. J. Epidemiol. 2004, 159, 413–421. [Google Scholar] [CrossRef]
- Mokdad, A.H.; Bowman, B.A.; Ford, E.S.; Vinicor, F.; Marks, J.S.; Koplan, J.P. The continuing epidemics of obesity and diabetes in the United States. JAMA 2001, 286, 1195–1200. [Google Scholar] [CrossRef]
- Reaven, G.M. Banting lecture 1988. Role of insulin resistance in human disease. Diabetes 1988, 37, 1595–1607. [Google Scholar] [CrossRef]
- Prado, C.M.; Wells, J.C.; Smith, S.R.; Stephan, B.C.; Siervo, M. Sarcopenic obesity: A critical appraisal of the current evidence. Clin. Nutr. 2012, 31, 583–601. [Google Scholar] [CrossRef] [PubMed]
- Johnson Stoklossa, C.A.; Sharma, A.M.; Forhan, M.; Siervo, M.; Padwal, R.S.; Prado, C.M. Prevalence of Sarcopenic Obesity in Adults with Class II/III Obesity Using Different Diagnostic Criteria. J. Nutr. Metab. 2017, 2017, 7307618. [Google Scholar] [CrossRef] [PubMed]
- Kalinkovich, A.; Livshits, G. Sarcopenic obesity or obese sarcopenia: A cross talk between age-associated adipose tissue and skeletal muscle inflammation as a main mechanism of the pathogenesis. Ageing Res. Rev. 2017, 35, 200–221. [Google Scholar] [CrossRef] [PubMed]
- Newman, A.B.; Haggerty, C.L.; Goodpaster, B.; Harris, T.; Kritchevsky, S.; Nevitt, M.; Miles, T.P.; Visser, M.; The Health, Aging, and Body Composition Research Group. Strength and muscle quality in a well-functioning cohort of older adults: The Health, Aging and Body Composition Study. J. Am. Geriatr. Soc. 2003, 51, 323–330. [Google Scholar] [CrossRef]
- Barone, M.; Viggiani, M.T.; Losurdo, G.; Principi, M.; Leandro, G.; Di Leo, A. Systematic review with meta-analysis: Post-operative complications and mortality risk in liver transplant candidates with obesity. Aliment. Pharmacol. Ther. 2017, 46, 236–245. [Google Scholar] [CrossRef]
- Shachar, S.S.; Williams, G.R.; Muss, H.B.; Nishijima, T.F. Prognostic value of sarcopenia in adults with solid tumours: A meta-analysis and systematic review. Eur. J. Cancer 2016, 57, 58–67. [Google Scholar] [CrossRef]
- Shu, X.; Lin, T.; Wang, H.; Zhao, Y.; Jiang, T.; Peng, X.; Yue, J. Diagnosis, prevalence, and mortality of sarcopenia in dialysis patients: A systematic review and meta-analysis. J. Cachexia Sarcopenia Muscle 2022, 13, 145–158. [Google Scholar] [CrossRef]
- Tantai, X.; Liu, Y.; Yeo, Y.H.; Praktiknjo, M.; Mauro, E.; Hamaguchi, Y.; Engelmann, C.; Zhang, P.; Jeong, J.Y.; van Vugt, J.L.A.; et al. Effect of sarcopenia on survival in patients with cirrhosis: A meta-analysis. J. Hepatol. 2022, 76, 588–599. [Google Scholar] [CrossRef]
- Feng, L.; Gao, Q.; Hu, K.; Wu, M.; Wang, Z.; Chen, F.; Mei, F.; Zhao, L.; Ma, B. Prevalence and risk factors of sarcopenia in patients with diabetes: A meta-analysis. J. Clin. Endocrinol. Metab. 2022, 107, 1470–1483. [Google Scholar] [CrossRef]
- Barone, M.; Viggiani, M.T.; Anelli, M.G.; Fanizzi, R.; Lorusso, O.; Lopalco, G.; Cantarini, L.; Di Leo, A.; Lapadula, G.; Iannone, F. Sarcopenia in Patients with Rheumatic Diseases: Prevalence and Associated Risk Factors. J. Clin. Med. 2018, 7, 504. [Google Scholar] [CrossRef]
- Janssen, I.; Shepard, D.S.; Katzmarzyk, P.T.; Roubenoff, R. The healthcare costs of sarcopenia in the United States. J. Am. Geriatr. Soc. 2004, 52, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Abellan van Kan, G. Epidemiology and consequences of sarcopenia. J. Nutr. Health Aging 2009, 13, 708–712. [Google Scholar] [CrossRef] [PubMed]
- Yuan, S.; Larsson, S.C. Epidemiology of sarcopenia: Prevalence, risk factors, and consequences. Metabolism 2023, 144, 155533. [Google Scholar] [CrossRef]
- Barone, M.; Losurdo, G.; Iannone, A.; Leandro, G.; Di Leo, A.; Trerotoli, P. Assessment of body composition: Intrinsic methodological limitations and statistical pitfalls. Nutrition 2022, 102, 111736. [Google Scholar] [CrossRef]
- Schiaffino, S.; Mammucari, C. Regulation of skeletal muscle growth by the IGF1-Akt/PKB pathway: Insights from genetic models. Skelet. Muscle 2011, 1, 4. [Google Scholar] [CrossRef]
- Williamson, D.L.; Raue, U.; Slivka, D.R.; Trappe, S. Resistance exercise, skeletal muscle FOXO3A, and 85-year-old women. J. Gerontol. A Biol. Sci. Med. Sci. 2010, 65, 335–343. [Google Scholar] [CrossRef]
- Manning, B.D.; Cantley, L.C. AKT/PKB signaling: Navigating downstream. Cell 2007, 129, 1261–1274. [Google Scholar] [CrossRef]
- Stitt, T.N.; Drujan, D.; Clarke, B.A.; Panaro, F.; Timofeyva, Y.; Kline, W.O.; Gonzalez, M.; Yancopoulos, G.D.; Glass, D.J. The IGF-1/PI3K/Akt pathway prevents expression of muscle atrophy-induced ubiquitin ligases by inhibiting FOXO transcription factors. Mol. Cell 2004, 14, 395–403. [Google Scholar] [CrossRef]
- Huang, H.; Tindall, D.J. Dynamic FoxO transcription factors. J. Cell Sci. 2007, 120, 2479–2487. [Google Scholar] [CrossRef]
- Jang, S.W.; Yang, S.J.; Srinivasan, S.; Ye, K. Akt phosphorylates MstI and prevents its proteolytic activation, blocking FOXO3 phosphorylation and nuclear translocation. J. Biol. Chem. 2007, 282, 30836–30844. [Google Scholar] [CrossRef]
- Murton, A.J.; Greenhaff, P.L. Muscle atrophy in immobilization and senescence in humans. Curr. Opin. Neurol. 2009, 22, 500–505. [Google Scholar] [CrossRef] [PubMed]
- Markofski, M.M.; Dickinson, J.M.; Drummond, M.J.; Fry, C.S.; Fujita, S.; Gundermann, D.M.; Glynn, E.L.; Jennings, K.; Paddon-Jones, D.; Reidy, P.T.; et al. Effect of age on basal muscle protein synthesis and mTORC1 signaling in a large cohort of young and older men and women. Exp. Gerontol. 2015, 65, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Wall, B.T.; Gorissen, S.H.; Pennings, B.; Koopman, R.; Groen, B.B.; Verdijk, L.B.; van Loon, L.J. Aging Is Accompanied by a Blunted Muscle Protein Synthetic Response to Protein Ingestion. PLoS ONE 2015, 10, e0140903. [Google Scholar] [CrossRef]
- Tanner, R.E.; Brunker, L.B.; Agergaard, J.; Barrows, K.M.; Briggs, R.A.; Kwon, O.S.; Young, L.M.; Hopkins, P.N.; Volpi, E.; Marcus, R.L.; et al. Age-related differences in lean mass, protein synthesis and skeletal muscle markers of proteolysis after bed rest and exercise rehabilitation. J. Physiol. 2015, 593, 4259–4273. [Google Scholar] [CrossRef]
- Constantin, D.; Menon, M.K.; Houchen-Wolloff, L.; Morgan, M.D.; Singh, S.J.; Greenhaff, P.; Steiner, M.C. Skeletal muscle molecular responses to resistance training and dietary supplementation in COPD. Thorax 2013, 68, 625–633. [Google Scholar] [CrossRef]
- Sanada, F.; Taniyama, Y.; Muratsu, J.; Otsu, R.; Shimizu, H.; Rakugi, H.; Morishita, R. Source of chronic inflammation in aging. Front. Cardiovasc. Med. 2018, 5, 12. [Google Scholar] [CrossRef]
- Kwak, M.K.; Baek, J.Y.; Park, S.J.; Jung, H.W.; Lee, E.; Jang, I.Y.; Ji, E.; Hong, E.G.; Jo, Y.; Ryu, D.; et al. Higher Circulating Resistin Levels Linked to Increased Sarcopenia Risk in Older Adults. J. Clin. Endocrinol. Metab. 2024, dgae894. [Google Scholar] [CrossRef]
- Evans, W.J.; Guralnik, J.; Cawthon, P.; Appleby, J.; Landi, F.; Clarke, L.; Vellas, B.; Ferrucci, L.; Roubenoff, R. Sarcopenia: No consensus, no diagnostic criteria, and no approved indication. How did we get here? Geroscience 2024, 46, 183–190. [Google Scholar] [CrossRef]
- Pedersen, B.K.; Febbraio, M.A. Muscles, exercise and obesity: Skeletal muscle as a secretory organ. Nat. Rev. Endocrinol. 2012, 8, 457–465. [Google Scholar] [CrossRef]
- Lee, J.H.; Jun, H.S. Role of Myokines in Regulating Skeletal Muscle Mass and Function. Front. Physiol. 2019, 10, 42. [Google Scholar] [CrossRef]
- McPherron, A.C.; Lee, S.J. Double muscling in cattle due to mutations in the myostatin gene. Proc. Natl. Acad. Sci. USA 1997, 94, 12457–12461. [Google Scholar] [CrossRef] [PubMed]
- Sharma, M.; Kambadur, R.; Matthews, K.G.; Somers, W.G.; Devlin, G.P.; Conaglen, J.V.; Fowke, P.J.; Bass, J.J. Myostatin, a transforming growth factor-beta superfamily member, is expressed in heart muscle and is upregulated in cardiomyocytes after infarct. J. Cell. Physiol. 1999, 180, 1–9. [Google Scholar] [CrossRef]
- Wilhelmsen, A.; Stephens, F.B.; Bennett, A.J.; Karagounis, L.G.; Jones, S.W.; Tsintzas, K. Skeletal muscle myostatin mRNA expression is upregulated in aged human adults with excess adiposity but is not associated with insulin resistance and ageing. Geroscience 2024, 46, 2033–2049. [Google Scholar] [CrossRef] [PubMed]
- Makiel, K.; Suder, A.; Targosz, A.; Maciejczyk, M.; Haim, A. Effect of Exercise Interventions on Irisin and Interleukin-6 Concentrations and Indicators of Carbohydrate Metabolism in Males with Metabolic Syndrome. J. Clin. Med. 2023, 12, 369. [Google Scholar] [CrossRef]
- Bostrom, P.; Wu, J.; Jedrychowski, M.P.; Korde, A.; Ye, L.; Lo, J.C.; Rasbach, K.A.; Boström, E.A.; Choi, J.H.; Long, J.Z.; et al. A PGC1-alpha-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature 2012, 481, 463–468. [Google Scholar] [CrossRef]
- Jeong, D.; Park, K.; Lee, J.; Choi, J.; Du, H.; Jeong, H.; Li, L.; Sakai, K.; Kang, S. Effects of Resistance Exercise and Essential Amino Acid Intake on Muscle Quality, Myokine, and Inflammation Factors in Young Adult Males. Nutrients 2024, 16, 1688. [Google Scholar] [CrossRef]
- Park, H.S.; Kim, H.C.; Zhang, D.; Yeom, H.; Lim, S.K. The novel myokine irisin: Clinical implications and potential role as a biomarker for sarcopenia in postmenopausal women. Endocrine 2019, 64, 341–348. [Google Scholar] [CrossRef]
- Colaianni, G.; Oranger, A.; Dicarlo, M.; Lovero, R.; Storlino, G.; Pignataro, P.; Fontana, A.; Di Serio, F.; Ingravallo, A.; Caputo, G.; et al. Irisin Serum Levels and Skeletal Muscle Assessment in a Cohort of Charcot-Marie-Tooth Patients. Front. Endocrinol. 2022, 13, 886243. [Google Scholar] [CrossRef]
- Gao, S.; Durstine, J.L.; Koh, H.J.; Carver, W.E.; Frizzell, N.; Carson, J.A. Acute myotube protein synthesis regulation by IL-6-related cytokines. Am. J. Physiol. Cell. Physiol. 2017, 313, C487–C500. [Google Scholar] [CrossRef]
- Pedersen, B.K.; Febbraio, M.A. Muscle as an endocrine organ: Focus on muscle-derived interleukin-6. Physiol. Rev. 2008, 88, 1379–1406. [Google Scholar] [CrossRef]
- Tanaka, T.; Narazaki, M.; Kishimoto, T. IL-6 in inflammation, immunity, and disease. Cold Spring Harb. Perspect. Biol. 2014, 6, a016295. [Google Scholar] [CrossRef]
- Tamura, Y.; Watanabe, K.; Kantani, T.; Hayashi, J.; Ishida, N.; Kaneki, M. Upregulation of circulating IL-15 by treadmill running in healthy individuals: Is IL-15 an endocrine mediator of the beneficial effects of endurance exercise? Endocrine J. 2011, 58, 211–215. [Google Scholar] [CrossRef]
- Garneau, L.; Parsons, S.A.; Smith, S.R.; Mulvihill, E.E.; Sparks, L.M.; Aguer, C. Plasma Myokine Concentrations After Acute Exercise in Non-obese and Obese Sedentary Women. Front. Physiol. 2020, 11, 18. [Google Scholar] [CrossRef]
- Pérez-López, A.; McKendry, J.; Martin-Rincon, M.; Morales-Alamo, D.; Pérez-Köhler, B.; Valadés, D.; Buján, J.; Calbet, J.A.L.; Breen, L. Skeletal muscle IL-15/IL-15Rα and myofibrillar protein synthesis after resistance exercise. Scand. J. Med. Sci. Sports 2018, 28, 116–125. [Google Scholar] [CrossRef]
- Pérez-López, A.; Martin-Rincon, M.; Santana, A.; Perez-Suarez, I.; Dorado, C.; Calbet, J.A.L.; Morales-Alamo, D. Antioxidants Facilitate High-intensity Exercise IL-15 Expression in Skeletal Muscle. Int. J. Sports Med. 2019, 40, 16–22. [Google Scholar] [CrossRef]
- Quinn, L.S.; Haugk, K.L.; Grabstein, K.H. Interleukin-15: A novel anabolic cytokine for skeletal muscle. Endocrinology 1995, 136, 3669–3672. [Google Scholar] [CrossRef]
- Quinn, L.S.; Anderson, B.G.; Drivdahl, R.H.; Alvarez, B.; Argiles, J.M. Overexpression of interleukin-15 induces skeletal muscle hypertrophy in vitro: Implications for treatment of muscle wasting disorders. Exp. Cell Res. 2002, 280, 55–63. [Google Scholar] [CrossRef]
- Krolopp, J.E.; Thornton, S.M.; Abbott, M.J. IL-15 activates the Jak3/STAT3 signaling pathway to mediate glucose uptake in skeletal muscle cells. Front. Physiol. 2016, 7, 626. [Google Scholar] [CrossRef]
- Carbó, N.; López-Soriano, J.; Costelli, P.; Busquets, S.; Alvarez, B.; Baccino, F.M.; Quinn, L.S.; López-Soriano, F.J.; Argilés, J.M. Interleukin-15 antagonizes muscle protein waste in tumour-bearing rats. Br. J. Cancer 2000, 83, 526–531. [Google Scholar] [CrossRef]
- Kurz, E.; Hirsch, C.A.; Dalton, T.; Shadaloey, S.A.; Khodadadi-Jamayran, A.; Miller, G.; Pareek, S.; Rajaei, H.; Mohindroo, C.; Baydogan, S.; et al. Exercise-induced engagement of the IL-15/IL-15Rα axis promotes anti-tumor immunity in pancreatic cancer. Cancer Cell 2022, 40, 720–737.e5. [Google Scholar] [CrossRef]
- Wu, J.; Huang, C.; Xiao, H.; Tang, Q.; Cai, W. Weight loss and resting energy expenditure in male patients with newly diagnosed esophageal cancer. Nutrition 2013, 29, 1310–1314. [Google Scholar] [CrossRef] [PubMed]
- El Shafey, N.; Guesnon, M.; Simon, F.; Deprez, E.; Cosette, J.; Stockholm, D.; Scherman, D.; Bigey, P.; Kichler, A. Inhibition of the myostatin/Smad signaling pathway by short decorin-derived peptides. Exp. Cell Res. 2016, 341, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Marshall, A.; Salerno, M.S.; Thomas, M.; Davies, T.; Berry, C.; Dyer, K.; Bracegirdle, J.; Watson, T.; Dziadek, M.; Kambadur, R.; et al. Mighty is a novel promyogenic factor in skeletal myogenesis. Exp. Cell Res. 2008, 314, 1013–1029. [Google Scholar] [CrossRef] [PubMed]
- Tapscott, S.J. The circuitry of a master switch: Myod and the regulation of skeletal muscle gene transcription. Development 2005, 132, 2685–2695. [Google Scholar] [CrossRef]
- Chargé, S.B.; Rudnicki, M.A. Cellular and molecular regulation of muscle regeneration. Physiol. Rev. 2004, 84, 209–238. [Google Scholar] [CrossRef]
- Arnold, H.H.; Winter, B. Muscle differentiation: More complexity to the network of myogenic regulators. Curr. Opin. Genet. Dev. 1998, 8, 539–544. [Google Scholar] [CrossRef]
- Ataeinosrat, A.; Saeidi, A.; Abednatanzi, H.; Rahmani, H.; Daloii, A.A.; Pashaei, Z.; Hojati, V.; Basati, G.; Mossayebi, A.; Laher, I.; et al. Intensity Dependent Effects of Interval Resistance Training on Myokines and Cardiovascular Risk Factors in Males with Obesity. Front. Endocrinol. 2022, 10, 895512. [Google Scholar] [CrossRef]
- Lee, S.J. Regulation of muscle mass by myostatin. Annu. Rev. Cell Dev. Biol. 2004, 20, 61–86. [Google Scholar] [CrossRef]
- Amthor, H.; Nicholas, G.; McKinnell, I.; Kemp, C.F.; Sharma, M.; Kambadur, R.; Patel, K. Follistatin complexes Myostatin and antagonizes Myostatin-mediated inhibition of myogenesis. Dev. Biol. 2004, 270, 19–30. [Google Scholar] [CrossRef]
- McPherron, A.C.; Lawler, A.M.; Lee, S.J. Regulation of skeletal muscle mass in mice by a new TGF-beta superfamily member. Nature 1997, 387, 83–90. [Google Scholar] [CrossRef]
- Larsson, L.; Degens, H.; Li, M.; Salviati, L.; Lee, Y.I.; Thompson, W.; Kirkland, J.L.; Sandri, M. Sarcopenia: Aging-Related Loss of Muscle Mass and Function. Physiol. Rev. 2019, 99, 427–511. [Google Scholar] [CrossRef] [PubMed]
- Piasecki, M.; Ireland, A.; Piasecki, J.; Stashuk, D.W.; Swiecicka, A.; Rutter, M.K.; Jones, D.A.; McPhee, J.S. Failure to expand the motor unit size to compensate for declining motor unit numbers distinguishes sarcopenic from non-sarcopenic older men. J. Physiol. 2018, 596, 1627–1637. [Google Scholar] [CrossRef] [PubMed]
- Larsson, L. Histochemical characteristics of human skeletal muscle during aging. Acta Physiol. Scand. 1983, 117, 469–471. [Google Scholar] [CrossRef]
- Luo, X.; Wang, J.; Ju, Q.; Li, T.; Bi, X. Molecular mechanisms and potential interventions during aging-associated sarcopenia. Mech. Ageing Dev. 2025, 223, 112020. [Google Scholar] [CrossRef]
- Evans, W.J. Skeletal muscle loss: Cachexia, sarcopenia, and inactivity. Am. J. Clin. Nutr. 2010, 91, 1123S–1127S. [Google Scholar] [CrossRef]
- Kortebein, P.; Ferrando, A.; Lombeida, J.; Wolfe, R.; Evans, W.J. Effect of 10 days of bed rest on skeletal muscle in healthy older adults. JAMA 2007, 297, 1772–1774. [Google Scholar] [CrossRef]
- Paddon-Jones, D.; Sheffield-Moore, M.; Urban, R.J.; Sanford, A.P.; Aarsland, A.; Wolfe, R.R.; Ferrando, A.A. Essential amino acid and carbohydrate supplementation ameliorates muscle protein loss in humans during 28 days bedrest. J. Clin. Endocrinol. Metab. 2004, 89, 4351–4358. [Google Scholar] [CrossRef]
- Landi, F.; Calvani, R.; Picca, A.; Tosato, M.; Martone, A.M.; D’Angelo, E.; Serafini, E.; Bernabei, R.; Marzetti, E. Impact of habitual physical activity and type of exercise on physical performance across ages in community-living people. PLoS ONE 2018, 13, e0191820. [Google Scholar] [CrossRef]
- Grimby, G.; Saltin, B. The ageing muscle. Clin. Physiol. 1983, 3, 209–218. [Google Scholar] [CrossRef]
- Ketchem, J.M.; Bowman, E.J.; Isales, C.M. Male sex hormones, aging, and inflammation. Biogerontology 2023, 24, 1–25. [Google Scholar] [CrossRef]
- Hellström, L.; Wahrenberg, H.; Hruska, K.; Reynisdottir, S.; Arner, P. Mechanisms behind gender differences in circulating leptin levels. J. Intern. Med. 2000, 247, 457–462. [Google Scholar] [CrossRef] [PubMed]
- Stern, J.H.; Rutkowski, J.M.; Scherer, P.E. Adiponectin, Leptin, and Fatty Acids in the Maintenance of Metabolic Homeostasis through Adipose Tissue Crosstalk. Cell Metab. 2016, 23, 770–784. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Lou, S.; Shi, R. From mitochondria to sarcopenia: Role of 17β-estradiol and testosterone. Front. Endocrinol. 2023, 14, 1156583. [Google Scholar] [CrossRef]
- Remelli, F.; Vitali, A.; Zurlo, A.; Volpato, S. Vitamin D deficiency and sarcopenia in older persons. Nutrients 2019, 11, 2861. [Google Scholar] [CrossRef]
- Barone, M.; Viggiani, M.T.; Avolio, A.W.; Iannone, A.; Rendina, M.; Di Leo, A. Obesity as predictor of postoperative outcomes in liver transplant candidates: Review of the literature and future perspectives. Dig. Liver Dis. 2017, 49, 957–966. [Google Scholar] [CrossRef]
- Biolo, G.; Fleming, R.Y.; Maggi, S.P.; Nguyen, T.T.; Herndon, D.N.; Wolfe, R.R. Inverse regulation of protein turnover and amino acid transport in skeletal muscle of hypercatabolic patients. J. Clin. Endocrinol. Metab. 2002, 87, 3378–3384. [Google Scholar] [CrossRef]
- Wolfe, R.R.; Martini, W.Z. Changes in intermediary metabolism in severe surgical illness. World J. Surg. 2000, 24, 639–647. [Google Scholar] [CrossRef]
- Mijnarends, D.M.; Koster, A.; Schols, J.M.; Meijers, J.M.; Halfens, R.J.; Gudnason, V.; Eiriksdottir, G.; Siggeirsdottir, K.; Sigurdsson, S.; Jónsson, P.V.; et al. Physical activity and incidence of sarcopenia: The population-based AGES-Reykjavik Study. Age Ageing 2016, 45, 614–620. [Google Scholar] [CrossRef]
- Barone, M.; Leo, A.D.; de van der Schueren, M.A.E. Malnutrition assessment by Global Leadership Initiative on Malnutrition criteria in patients with amyotrophic lateral sclerosis. Nutrition 2023, 109, 111997. [Google Scholar] [CrossRef]
- Biolo, G.; Cederholm, T.; Muscaritoli, M. Muscle contractile and metabolic dysfunction is a common feature of sarcopenia of aging and chronic diseases: From sarcopenic obesity to cachexia. Clin. Nutr. 2014, 33, 737–748. [Google Scholar] [CrossRef]
- Argilés, J.M.; Busquets, S.; Stemmler, B.; López-Soriano, F.J. Cancer cachexia: Understanding the molecular basis. Nat. Rev. Cancer 2014, 14, 754–762. [Google Scholar] [CrossRef] [PubMed]
- Dhaliwal, A.; Quinlan, J.I.; Overthrow, K.; Greig, C.; Lord, J.M.; Armstrong, M.J.; Cooper, S.C. Sarcopenia in Inflammatory Bowel Disease: A Narrative Overview. Nutrients 2021, 13, 656. [Google Scholar] [CrossRef] [PubMed]
- Ebadi, M.; Bhanji, R.A.; Mazurak, V.C.; Montano-Loza, A.J. Sarcopenia in cirrhosis: From pathogenesis to interventions. J. Gastroenterol. 2019, 54, 845–859. [Google Scholar] [CrossRef] [PubMed]
- Saltzman, J.R.; Russell, R.M. Nutritional consequences of intestinal bacterial overgrowth. Compr. Ther. 1994, 20, 523–530. [Google Scholar]
- Balestrieri, P.; Ribolsi, M.; Guarino, M.P.L.; Emerenziani, S.; Altomare, A.; Cicala, M. Nutritional Aspects in Inflammatory Bowel Diseases. Nutrients 2020, 12, 372. [Google Scholar] [CrossRef]
- Metsios, G.S.; Fenton, S.A.; Moe, H.R.; van der Esch, M.; van Zanten, J.V.; Koutedakis, Y.; Vitalis, P.; Kennedy, N.; Brodin, N.C.; Tzika, A.; et al. IMPACT-RMD Consortium. Implementation of Physical Activity into Routine Clinical Practice in Rheumatic Musculoskeletal Disease: The IMPACT-RMD Study Protocol and Rationale. Mediterr. J. Rheumatol. 2020, 30, 231–236. [Google Scholar] [CrossRef]
- Fearon, K.C. The 2011 ESPEN Arvid Wretlind lecture: Cancer cachexia: The potential impact of translational research on patient-focused outcomes. Clin. Nutr. 2012, 31, 577–582. [Google Scholar] [CrossRef]
- Freire, P.P.; Fernandez, G.J.; de Moraes, D.; Cury, S.S.; Dal Pai-Silva, M.; Dos Reis, P.P.; Rogatto, S.R.; Carvalho, R.F. The expression landscape of cachexia-inducing factors in human cancers. J. Cachexia Sarcopenia Muscle 2020, 11, 947–961. [Google Scholar] [CrossRef]
- Demark-Wahnefried, W.; Peterson, B.L.; Winer, E.P.; Marks, L.; Aziz, N.; Marcom, P.K.; Blackwell, K.; Rimer, B.K. Changes in weight, body composition, and factors influencing energy balance among premenopausal breast cancer patients receiving adjuvant chemotherapy. J. Clin. Oncol. 2001, 19, 2381–2389. [Google Scholar] [CrossRef]
- Brown, J.C.; Caan, B.J.; Meyerhardt, J.A.; Weltzien, E.; Xiao, J.; Cespedes Feliciano, E.M.; Kroenke, C.H.; Castillo, A.; Kwan, M.L.; Prado, C.M. The deterioration of muscle mass and radiodensity is prognostic of poor survival in stage I-III colorectal cancer: A population-based cohort study (C-SCANS). J. Cachexia Sarcopenia Muscle 2018, 9, 664–672. [Google Scholar] [CrossRef]
- Sanders, P.M.; Tisdale, M.J. Role of lipid-mobilising factor (LMF) in protecting tumour cells from oxidative damage. Br. J. Cancer 2004, 90, 1274–1278. [Google Scholar] [CrossRef] [PubMed]
- Stephens, N.A.; Skipworth, R.J.; Macdonald, A.J.; Greig, C.A.; Ross, J.A.; Fearon, K.C. Intramyocellular lipid droplets increase with progression of cachexia in cancer patients. J. Cachexia Sarcopenia Muscle 2011, 2, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Molfino, A.; Beltrà, M.; Amabile, M.I.; Belli, R.; Birolo, G.; Belloni, E.; De Lucia, S.; Garcia-Castillo, L.; Penna, F.; Imbimbo, G.; et al. Small non-coding RNA profiling in patients with gastrointestinal cancer. J. Cachexia Sarcopenia Muscle 2023, 14, 2692–2702. [Google Scholar] [CrossRef]
- Robinson, S.; Granic, A.; Cruz-Jentoft, A.J.; Sayer, A.A. The role of nutrition in the prevention of sarcopenia. Am. J. Clin. Nutr. 2023, 118, 852–864. [Google Scholar] [CrossRef]
- Srivastava, S.; Pandey, V.K.; Singh, A.; Dar, A.H. Exploring the Potential of Treating Sarcopenia through Dietary Interventions. J. Food Biochem. 2024, 3018760. [Google Scholar] [CrossRef]
- Castillo, E.C.; Vázquez-Garza, E.; Yee-Trejo, D.; García-Rivas, G.; Torre-Amione, G. What Is the Role of Inflammation in the Pathogenesis of Heart Failure? Curr. Cardiol. Rep. 2020, 22, 139. [Google Scholar] [CrossRef]
- Gualtierotti, R. Bridging the gap: Time to integrate sex and gender differences into research and clinical practice for improved health outcomes. Eur. J. Intern Med. 2025, S0953-6205(25)00040-8. [Google Scholar] [CrossRef]
- Conforto, R.; Rizzo, V.; Russo, R.; Mazza, E.; Maurotti, S.; Pujia, C.; Succurro, E.; Arturi, F.; Ferro, Y.; Sciacqua, A.; et al. Advances in body composition and gender differences in susceptibility to frailty syndrome: Role of osteosarcopenic obesity. Metabolism 2024, 161, 156052. [Google Scholar] [CrossRef]
- Jakubowicz, D.; Rosenblum, R.C.; Wainstein, J.; Twito, O. Influence of fasting until noon (extended postabsorptive state) on clock gene mRNA expression and regulation of body weight and glucose metabolism. Int. J. Mol. Sci. 2023, 24, 7154. [Google Scholar] [CrossRef]
- Bird, S.P.; Tarpenning, K.M. Influence of circadian time structure on acute hormonal responses to a single bout of heavy-resistance exercise in weight-trained men. Chronobiol. Int. 2004, 21, 131–146. [Google Scholar] [CrossRef]
- Moschou, D.; Krikelis, M.; Georgakopoulos, C.; Mole, E.; Chronopoulos, E.; Tournis, S.; Mavragani, C.; Makris, K.; Dontas, I.; Gazi, S. Sarcopenia in rheumatoid arthritis. A narrative review. J. Frailty Sarcopenia Falls 2023, 8, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Calvani, R.; Picca, A.; Coelho-Júnior, H.J.; Tosato, M.; Marzetti, E.; Landi, F. Diet for the prevention and management of sarcopenia. Metabolism 2023, 146, 155637. [Google Scholar] [CrossRef] [PubMed]
- Wirt, A.; Collins, C.E. Diet quality—What is it and does it matter? Public Health Nutr. 2009, 12, 2473–2492. [Google Scholar] [CrossRef] [PubMed]
- Dorrington, N.; Fallaize, R.; Hobbs, D.; Weech, M.; Lovegrove, J.A. Diet Quality Index for older adults (DQI-65): Development and use in predicting adherence to dietary recommendations and health markers in the UK National Diet and Nutrition Survey. Br. J. Nutr. 2022, 128, 2193–2207. [Google Scholar] [CrossRef]
- Bloom, I.; Shand, C.; Cooper, C.; Robinson, S.; Baird, J. Diet Quality and Sarcopenia in Older Adults: A Systematic Review. Nutrients 2018, 10, 308. [Google Scholar] [CrossRef]
- Ramadas, A.; Law, H.H.; Krishnamoorthy, R.; Ku, J.W.S.; Mohanty, P.; Lim, M.Z.C.; Shyam, S. Diet quality and measures of sarcopenia in developing economies: A systematic review. Nutrients 2022, 14, 868. [Google Scholar] [CrossRef]
- Ghoreishy, S.M.; Koujan, S.E.; Hashemi, R.; Heshmat, R.; Motlagh, A.D.; Esmaillzadeh, A. Relationship between healthy eating index and sarcopenia in elderly people. BMC Geriatr. 2023, 23, 25. [Google Scholar] [CrossRef]
- Krznarić, Ž.; Karas, I.; Ljubas Kelečić, D.; Vranešić Bender, D. The Mediterranean and Nordic Diet: A Review of Differences and Similarities of Two Sustainable, Health-Promoting Dietary Patterns. Front. Nutr. 2021, 8, 683678. [Google Scholar] [CrossRef]
- Cacciatore, S.; Calvani, R.; Marzetti, E.; Picca, A.; Coelho-Júnior, H.J.; Martone, A.M.; Massaro, C.; Tosato, M.; Landi, F. Low Adherence to Mediterranean Diet Is Associated with Probable Sarcopenia in Community-Dwelling Older Adults: Results from the Longevity Check-Up (Lookup) 7+ Project. Nutrients 2023, 15, 1026. [Google Scholar] [CrossRef]
- Coelho-Júnior, H.J.; Trichopoulou, A.; Panza, F. Cross-sectional and longitudinal associations between adherence to Mediterranean diet with physical performance and cognitive function in older adults: A systematic review and meta-analysis. Ageing Res. Rev. 2021, 70, 101395. [Google Scholar] [CrossRef]
- Granic, A.; Dismore, L.; Hurst, C.; Robinson, S.M.; Sayer, A.A. Myoprotective whole foods, muscle health and sarcopenia: A systematic review of observational and intervention studies in older adults. Nutrients 2020, 12, 2257. [Google Scholar] [CrossRef] [PubMed]
- Landi, F.; Liperoti, R.; Fusco, D.; Mastropaolo, S.; Quattrociocchi, D.; Proia, A.; Russo, A.; Bernabei, R.; Onder, G. Prevalence and risk factors of sarcopenia among nursing home older residents. J. Gerontol. A Biol. Sci. Med. Sci. 2012, 67, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Serafini, E.; Marzetti, E.; Calvani, R.; Picca, A.; Tosato, M.; Bernabei, R.; Landi, F. Nutritional approach to sarcopenia. J. Gerontol. Geriatr. 2019, 67, 52–61. [Google Scholar]
- Paddon-Jones, D.; Rasmussen, B.B. Dietary protein recommendations and the prevention of sarcopenia. Curr. Opin. Clin. Nutr. Metab. Care 2009, 12, 86–90. [Google Scholar] [CrossRef]
- Fujita, S.; Rasmussen, B.B.; Cadenas, J.G.; Drummond, M.J.; Glynn, E.L.; Sattler, F.R.; Volpi, E. Aerobic exercise overcomes the age-related insulin resistance of muscle protein metabolism by improving endothelial function and Akt/mammalian target of rapamycin signaling. Diabetes 2007, 56, 1615–1622. [Google Scholar] [CrossRef]
- Palmer, K.; Vetrano, D.L.; Marengoni, A.; Tummolo, A.; Villani, E.; Acampora, N.; Bernabei, R.; Onder, G. The relationship between anaemia and frailty: A systematic review and meta-analysis of observational studies. J. Nutr. Health Aging 2018, 22, 965–974. [Google Scholar] [CrossRef]
- Keller, U. Nutritional laboratory markers in malnutrition. J. Clin. Med. 2019, 8, 775. [Google Scholar] [CrossRef]
- Baird, M.F.; Graham, S.M.; Baker, J.S.; Bickerstaff, G.F. Creatine-kinase-and exercise-related muscle damage implications for muscle performance and recovery. J. Nutr. Metab. 2012, 960363. [Google Scholar] [CrossRef]
- Milla, S.P.; Luna, P.P.G.; Casariego, A.V.; González, F.V.; Folgueras, T.M.; Jáuregui, O.I.; Rey, S.G.; Fernández, A.C.; Plaza, B.L.; Quintana, T.C.; et al. Adherence and impact of an oral nutritional supplement enriched in leucine, EVOO, EPA and DHA, and beta-glucans on the coverage of energy and protein requirements in patients with cancer and malnutrition: Alisenoc study. Nutrition 2024, 120, 112355. [Google Scholar] [CrossRef]
- Ganapathy, A.; Nieves, J.W. Nutrition and Sarcopenia-What Do We Know? Nutrients 2020, 12, 1755. [Google Scholar] [CrossRef]
- Zhao, J.; Huang, Y.; Yu, X. A Narrative Review of Gut-Muscle Axis and Sarcopenia: The Potential Role of Gut Microbiota. Int. J. Gen. Med. 2021, 14, 1263–1273. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Jia, S.; Xue, X.; Guo, C.; Dong, K. Gut microbiota: A novel target for exercise-mediated regulation of NLRP3 inflammasome activation. Front. Microbiol. 2025, 15, 1476908. [Google Scholar] [CrossRef] [PubMed]
- Frampton, J.; Murphy, K.G.; Frost, G.; Chambers, E.S. Short-chain fatty acids as potential regulators of skeletal muscle metabolism and function. Nat. Metab. 2020, 2, 840–848. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Lei, P. Efficacy of probiotic supplements in the treatment of sarcopenia: A systematic review and meta-analysis. PLoS ONE 2025, 20, e0317699. [Google Scholar] [CrossRef]
- Borack, M.S.; Volpi, E. Efficacy and Safety of Leucine Supplementation in the Elderly. J. Nutr. 2016, 146, 2625S–2629S. [Google Scholar] [CrossRef]
- Wang, X.; Proud, C.G. The mTOR pathway in the control of protein synthesis. Physiology 2006, 21, 362–369. [Google Scholar] [CrossRef]
- Blomstrand, E.; Eliasson, J.; Karlsson, H.K.; Köhnke, R. Branched-chain amino acids activate key enzymes in protein synthesis after physical exercise. J. Nutr. 2006, 136, 269S–273S. [Google Scholar] [CrossRef]
- Wandrag, L.; Brett, S.J.; Frost, G.; Hickson, M. Impact of supplementation with amino acids or their metabolites on muscle wasting in patients with critical illness or other muscle wasting illness: A systematic review. J. Hum. Nutr. Diet. 2015, 28, 313–330. [Google Scholar] [CrossRef]
- Teixeira, F.J.; Santos, H.O.; Howell, S.L.; Pimentel, G.D. Whey protein in cancer therapy: A narrative review. Pharmacol. Res. 2019, 144, 245–256. [Google Scholar] [CrossRef]
- Zhang, X.; Kaneko, M.; Liu, W.; Stephen, O.A.; Nakamura, K.; Yamaguchi, M.; Yoshida, C.; Oishi, T.; Kobayashi, S.; Mizoi, K.; et al. Analysis of Protein Digestion and Absorption Using a Ussing Chamber to Simulate the Environment in the Digestive Tract. J. Nutr. Sci. Vitaminol. 2024, 70, 158–163. [Google Scholar] [CrossRef]
- Rogers, L.M.; Belfield, A.E.; Korzepa, M.; Gritsas, A.; Churchward-Venne, T.A.; Breen, L. Postprandial plasma aminoacidemia and indices of appetite regulation following pea-rice blend, pea isolate and whey protein ingestion in healthy young adults. Br. J. Nutr. 2024, 132, 691–700. [Google Scholar] [CrossRef] [PubMed]
- Winter, A.; MacAdams, J.; Chevalier, S. Normal protein anabolic response to hyperaminoacidemia in insulin-resistant patients with lung cancer cachexia. Clin. Nutr. 2012, 31, 765–773. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, R.R. Skeletal muscle protein metabolism and resistance exercise. J. Nutr. 2006, 136, 525S–528S. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, D.J.; Hossain, T.; Hill, D.S.; Phillips, B.E.; Crossland, H.; Williams, J.; Loughna, P.; Churchward-Venne, T.A.; Breen, L.; Phillips, S.M.; et al. Effects of leucine and its metabolite β-hydroxy-β-methylbutyrate on human skeletal muscle protein metabolism. J. Physiol. 2013, 591, 2911–2923. [Google Scholar] [CrossRef]
- Van Kovering, M.; Nissen, S.L. Oxidation of leucine and alpha-ketoisocaproate to β-hydroxy-β-methylbutyrate in vivo. Am. J. Physiol. Endocrinol. Metab. 1992, 262, E27. [Google Scholar] [CrossRef]
- Nissen, S.; Sharp, R.L.; Panton, L.; Vukovich, M.; Trappe, S.; Fuller, J.C., Jr. β-Hydroxy-β-Methylbutyrate (HMB) Supplementation in Humans Is Safe and May Decrease Cardiovascular Risk Factors. J. Nutr. 2000, 130, 1937–1945. [Google Scholar] [CrossRef]
- Argilés, J.M.; Campos, N.; Lopez-Pedrosa, J.M.; Rueda, R.; Rodriguez-Manas, L. Skeletal Muscle Regulates Metabolism via Interorgan Crosstalk: Roles in Health and Disease. J. Am. Med. Dir. Assoc. 2016, 17, 789–796. [Google Scholar] [CrossRef]
- Giacosa, A.; Barrile, G.C.; Mansueto, F.; Rondanelli, M. The nutritional support to prevent sarcopenia in the elderly. Front. Nutr. 2024, 11, 1379814. [Google Scholar] [CrossRef]
- Wilson, J.M.; Fitschen, P.J.; Campbell, B.; Wilson, G.J.; Zanchi, N.; Taylor, L.; Antonio, J. International Society of Sports Nutrition Position Stand: Beta-hydroxy-beta-methylbutyrate (HMB). J. Int. Soc. Sports Nutr. 2013, 10, 6. [Google Scholar] [CrossRef]
- Smith, H.J.; Khal, J.; Tisdale, M.J. Downregulation of ubiquitin-dependent protein degradation in murine myotubes during hyperthermia by eicosapentaenoic acid. Biochem. Biophys Res. Commun. 2005, 332, 83–88. [Google Scholar] [CrossRef]
- Rathmacher, J.A.; Pitchford, L.M.; Stout, J.R.; Townsend, J.R.; Jäger, R.; Kreider, R.B.; Campbell, B.I.; Kerksick, C.M.; Harty, P.S.; Candow, D.G.; et al. International society of sports nutrition position stand: β-hydroxy-β-methylbutyrate (HMB). J. Int. Soc. Sports Nutr. 2025, 22, 2434734. [Google Scholar] [CrossRef] [PubMed]
- Shobeiri, P.; Seyedmirzaei, H.; Karimi, N.; Rashidi, F.; Teixeira, A.L.; Brand, S.; Sadeghi-Bahmani, D.; Rezaei, N. IL-6 and TNF-α responses to acute and regular exercise in adult individuals with multiple sclerosis (MS): A systematic review and meta-analysis. Eur. J. Med. Res. 2022, 27, 185. [Google Scholar] [CrossRef] [PubMed]
- Therdyothin, A.; Prokopidis, K.; Galli, F.; Witard, O.C.; Isanejad, M. The effects of omega-3 polyunsaturated fatty acids on muscle and whole-body protein synthesis: A systematic review and meta-analysis. Nutr. Rev. 2025, 83, e131–e143. [Google Scholar] [CrossRef]
- Smith, G.I.; Atherton, P.; Reeds, D.N.; Mohammed, B.S.; Rankin, D.; Rennie, M.J.; Mittendorfer, B. Dietary omega-3 fatty acid supplementation increases the rate of muscle protein synthesis in older adults: A randomized controlled trial. Am. J. Clin. Nutr. 2011, 93, 402–412. [Google Scholar] [CrossRef]
- Engelen, M.P.K.J.; Jonker, R.; Sulaiman, H.; Fisk, H.L.; Calder, P.C.; Deutz, N.E.P. ω-3 polyunsaturated fatty acid supplementation improves postabsorptive and prandial protein metabolism in patients with chronic obstructive pulmonary disease: A randomized clinical trial. Am. J. Clin. Nutr. 2022, 116, 686–698. [Google Scholar] [CrossRef]
- Jans, A.; van Hees, A.M.; Gjelstad, I.M.; Sparks, L.M.; Tierney, A.C.; Risérus, U.; Drevon, C.A.; Schrauwen, P.; Roche, H.M.; Blaak, E.E. Impact of dietary fat quantity and quality on skeletal muscle fatty acid metabolism in subjects with the metabolic syndrome. Metabolism 2012, 61, 1554–1565. [Google Scholar] [CrossRef]
- Kavyani, Z.; Musazadeh, V.; Fathi, S.; Hossein Faghfouri, A.; Dehghan, P.; Sarmadi, B. Efficacy of the omega-3 fatty acids supplementation on inflammatory biomarkers: An umbrella meta-analysis. Int. Immunopharmacol. 2022, 111, 109104. [Google Scholar] [CrossRef]
- Guo, Y.; Ma, B.; Li, X.; Hui, H.; Zhou, Y.; Li, N.; Xie, X. n-3 PUFA can reduce IL-6 and TNF levels in patients with cancer. Br. J. Nutr. 2023, 129, 54–65. [Google Scholar] [CrossRef]
- Allaire, J.; Couture, P.; Leclerc, M.; Charest, A.; Marin, J.; Lépine, M.C.; Talbot, D.; Tchernof, A.; Lamarche, B. A randomized, crossover, head-to-head comparison of eicosapentaenoic acid and docosahexaenoic acid supplementation to reduce inflammation markers in men and women: The Comparing EPA to DHA (ComparED) Study. Am. J. Clin. Nutr. 2016, 104, 280–287. [Google Scholar] [CrossRef]
- Bischoff-Ferrari, H.A.; Borchers, M.; Gudat, F.; Durmuller, U.; Stahelin, H.B.; Dick, W. Vitamin D receptor expression in human muscle tissue decreases with age. J. Bone Miner. Res. 2004, 19, 265–269. [Google Scholar] [CrossRef]
- Ceglia, L.; Harris, S.S. Vitamin D and its role in skeletal muscle. Calcif. Tissue Int. 2013, 92, 151–162. [Google Scholar] [CrossRef] [PubMed]
- Okuno, J.; Tomura, S.; Yabushita, N.; Kim, M.J.; Okura, T.; Tanaka, K.; Yanagi, H. Effects of serum 25-hydroxyvitamin D(3) levels on physical fitness in community-dwelling frail women. Arch. Gerontol. Geriatr. 2010, 50, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Bischoff-Ferrari, H.A.; Dawson-Hughes, B.; Staehelin, H.B.; Orav, J.E.; Stuck, A.E.; Theiler, R.; Wong, J.B.; Egli, A.; Kiel, D.P.; Henschkowski, J. Fall prevention with supplemental and active forms of vitamin D: A meta-analysis of randomised controlled trials. BMJ 2009, 339, b3692. [Google Scholar] [CrossRef]
- Visser, M.; Deeg, D.J.; Lips, P. Low vitamin D and high parathyroid hormone levels as determinants of loss of muscle strength and muscle mass (sarcopenia): The Longitudinal Aging Study Amsterdam. J. Clin. Endocrinol. Metab. 2003, 88, 5766–5772. [Google Scholar] [CrossRef] [PubMed]
- Dawson-Hughes, B. Vitamin D and muscle function. J. Steroid Biochem. Mol. Biol. 2017, 173, 313–316. [Google Scholar] [CrossRef]
- Ceglia, L.; Niramitmahapanya, S.; da Silva Morais, M.; Rivas, D.A.; Harris, S.S.; Bischoff-Ferrari, H.; Fielding, R.A.; Dawson-Hughes, B. A randomized study on the effect of vitamin D3 supplementation on skeletal muscle morphology and vitamin D receptor concentration in older women. J. Clin. Endocrinol. Metab. 2013, 98, E1927–E1935. [Google Scholar] [CrossRef]
- Stockton, K.A.; Mengersen, K.; Paratz, J.D.; Kandiah, D.; Bennell, K.L. Effect of vitamin D supplementation on muscle strength: A systematic review and meta-analysis. Osteoporos Int. 2011, 22, 859–871. [Google Scholar] [CrossRef]
- Tomlinson, P.B.; Joseph, C.; Angioi, M. Effects of vitamin D supplementation on upper and lower body muscle strength levels in healthy individuals. A systematic review with meta-analysis. J. Sci. Med. Sport. 2015, 18, 575–580. [Google Scholar] [CrossRef]
- Tabrizi, R.; Hallajzadeh, J.; Mirhosseini, N.; Lankarani, K.B.; Maharlouei, N.; Akbari, M.; Asemi, Z. The effects of vitamin D supplementation on muscle function among postmenopausal women: A systematic review and meta-analysis of randomized controlled trials. EXCLI J. 2019, 18, 591–603. [Google Scholar] [CrossRef]
- Uchitomi, R.; Oyabu, M.; Kamei, Y. Vitamin D and Sarcopenia: Potential of Vitamin D Supplementation in Sarcopenia Prevention and Treatment. Nutrients 2020, 12, 3189. [Google Scholar] [CrossRef]
- Latham, C.M.; Brightwell, C.R.; Keeble, A.R.; Munson, B.D.; Thomas, N.T.; Zagzoog, A.M.; Fry, C.S.; Fry, J.L. Vitamin D Promotes Skeletal Muscle Regeneration and Mitochondrial Health. Front. Physiol. 2021, 12, 660498. [Google Scholar] [CrossRef] [PubMed]
- Roseland, J.M.; Phillips, K.M.; Patterson, K.Y.; Pehrsson, P.R.; Taylor, C.L. Vitamin D in foods: An evolution of knowledge. In Vitamin D, 4th ed.; Feldman, D., Pike, J.W., Bouillon, R., Giovannucci, E., Goltzman, D., Hewison, M., Eds.; Academic Press: Cambridge, MA, USA, 2018; Volume 2: Health, Disease and Therapeutics, pp. 41–78. [Google Scholar]
- Institute of Medicine, Food and Nutrition Board. Dietary Reference Intakes for Calcium and Vitamin D; National Academy Press: Washington, DC, USA, 2010. [Google Scholar]
- Calvo, M.S.; Whiting, S.J.; Barton, C.N. Vitamin D fortification in the United States and Canada: Current status and data needs. Am. J. Clin. Nutr. 2004, 80, 1710S–1716S. [Google Scholar] [CrossRef] [PubMed]
- European Parliament, Council of the European Union. Regulation (EU) No 609/2013 of the European Parliament and of the Council of 12 June 2013 on Food Intended for Infants and Young Children, Food for Special Medical Purposes, and Total Diet Replacement for Weight Control. Off. J. Eur. Union 2013, 181, 35–56. [Google Scholar]
- Chereches, M.C.; Finta, H.; Popa, C.O.; Stefanescu, D.; Muntean, D.L. Qualitative Study on the Factors Influencing the Utilisation of Products Labelled “Food for Special Medicinal Use” (FSMP). Nutrients 2023, 15, 2582. [Google Scholar] [CrossRef]
- An, H.J.; Tizaoui, K.; Terrazzino, S.; Cargnin, S.; Lee, K.H.; Nam, S.W.; Kim, J.S.; Yang, J.W.; Lee, J.Y.; Smith, L. Sarcopenia in autoimmune and rheumatic diseases: A comprehensive review. Int. J. Mol. Sci. 2020, 21, 5678. [Google Scholar] [CrossRef]
- Lynch, C.J.; Adams, S.H. Branched-chain amino acids in metabolic signaling and insulin resistance. Nat. Rev. Endocrinol. 2014, 10, 723–736. [Google Scholar] [CrossRef]
- Fekete, M.; Lehoczki, A.; Csípő, T.; Fazekas-Pongor, V.; Szappanos, Á.; Major, D.; Mózes, N.; Dósa, N.; Varga, J.T. The role of trace elements in COPD: Pathogenetic mechanisms and therapeutic potential of zinc, iron, magnesium, selenium, manganese, copper, and calcium. Nutrients 2024, 16, 4118. [Google Scholar] [CrossRef]
- Rondanelli, M.; Donini, L.M.; Giusti, A. Sarcopenia: Definition, diagnosis and nutritional treatment. In SIGG Monographs: Geriatrics; Pacini Editore Medicina: Pisa, Italy, 2024; Available online: https://www.sigg.it/assets/monografie/2024/Sarcopenia%20definizione%20diagnosi%20e%20trattamento%20nutrizionale.pdf (accessed on 8 March 2025).
- Nasimi, N.; Sohrabi, Z.; Nunes, E.A.; Sadeghi, E.; Jamshidi, S.; Gholami, Z.; Akbarzadeh, M.; Faghih, S.; Akhlaghi, M.; Phillips, S.M. Whey Protein Supplementation with or without Vitamin D on Sarcopenia-Related Measures: A Systematic Review and Meta-Analysis. Adv. Nutr. 2023, 14, 762–773. [Google Scholar] [CrossRef]
- Cuyul-Vásquez, I.; Pezo-Navarrete, J.; Vargas-Arriagada, C.; Ortega-Díaz, C.; Sepúlveda-Loyola, W.; Hirabara, S.M.; Marzuca-Nassr, G.N. Effectiveness of Whey Protein Supplementation during Resistance Exercise Training on Skeletal Muscle Mass and Strength in Older People with Sarcopenia: A Systematic Review and Meta-Analysis. Nutrients 2023, 15, 3424. [Google Scholar] [CrossRef]
- Cereda, E.; Pisati, R.; Rondanelli, M.; Caccialanza, R. Whey Protein, Leucine- and Vitamin-D-Enriched Oral Nutritional Supplementation for the Treatment of Sarcopenia. Nutrients 2022, 14, 1524. [Google Scholar] [CrossRef]
- Prado, C.M.; Purcell, S.A.; Laviano, A. Nutrition interventions to treat low muscle mass in cancer. J. Cachexia Sarcopenia Muscle 2020, 11, 366–380. [Google Scholar] [CrossRef] [PubMed]
- Ryan, A.M.; Power, D.G.; Daly, L.; Cushen, S.J.; Ní Bhuachalla, Ē.; Prado, C.M. Cancer-associated malnutrition, cachexia and sarcopenia: The skeleton in the hospital closet 40 years later. Proc. Nutr. Soc. 2016, 75, 199–211. [Google Scholar] [PubMed]
- Moses, A.W.; Slater, C.; Preston, T.; Barber, M.D.; Fearon, K.C. Reduced total energy expenditure and physical activity in cachectic patients with pancreatic cancer can be modulated by an energy and protein dense oral supplement enriched with n-3 fatty acids. Br. J. Cancer 2004, 90, 996–1002. [Google Scholar] [CrossRef] [PubMed]
- Viggiani, M.T.; Lorusso, O.; Natalizio, F.; Principi, M.; Di Leo, A.; Barone, M. Influence of chemotherapy on total energy expenditure in patients with gastrointestinal cancer: A pilot study. Nutrition 2017, 42, 7–11. [Google Scholar] [CrossRef]
- Muscaritoli, M.; Arends, J.; Bachmann, P.; Baracos, V.; Barthelemy, N.; Bertz, H.; Bozzetti, F.; Hütterer, E.; Isenring, E.; Kaasa, S. ESPEN Practical Guideline: Clinical Nutrition in Cancer. Clin. Nutr. 2021, 40, 2898–2913. [Google Scholar] [CrossRef]
- Arends, J.; Bachmann, P.; Baracos, V.; Barthelemy, N.; Bertz, H.; Bozzetti, F.; Fearon, K.; Hütterer, E.; Isenring, E.; Kaasa, S.; et al. ESPEN guidelines on nutrition in cancer patients. Clin. Nutr. 2017, 36, 11–48. [Google Scholar] [CrossRef]
- Prado, C.M.; Lieffers, J.R.; McCargar, L.J.; Reiman, T.; Sawyer, M.B.; Martin, L.; Baracos, V.E. Prevalence and clinical implications of sarcopenic obesity in patients with solid tumours of the respiratory and gastrointestinal tracts: A population-based study. Lancet Oncol. 2008, 9, 629–635. [Google Scholar] [CrossRef]
- Op den Kamp, C.M.; Langen, R.C.; Haegens, A.; Schols, A.M. Muscle atrophy in cachexia: Can dietary protein tip the balance? Curr. Opin. Clin. Nutr. Metab. Care 2009, 12, 611–616. [Google Scholar] [CrossRef]
- Ford, K.L.; Arends, J.; Atherton, P.J.; Engelen, M.P.K.J.; Gonçalves, T.J.M.; Laviano, A.; Lobo, D.N.; Phillips, S.M.; Ravasco, P.; Deutz, N.E.P.; et al. The importance of protein sources to support muscle anabolism in cancer: An expert group opinion. Clin. Nutr. 2022, 41, 192–201. [Google Scholar] [CrossRef]
- Bozzetti, F.; Bozzetti, V. Is the intravenous supplementation of amino acid to cancer patients adequate? A critical appraisal of literature. Clin. Nutr. 2013, 32, 142–146. [Google Scholar] [CrossRef]
- Morais, J.A.; Jacob, K.W.; Chevalier, S. Effects of aging and insulin resistant states on protein anabolic responses in older adults. Exp. Gerontol. 2018, 108, 262–268. [Google Scholar] [CrossRef] [PubMed]
- Martin, W.F.; Armstrong, L.E.; Rodriguez, N.R. Dietary protein intake and renal function. Nutr. Metab. 2005, 2, 25. [Google Scholar] [CrossRef]
- Arends, J.; Baracos, V.; Bertz, H.; Bozzetti, F.; Calder, P.C.; Deutz, N.E.P.; Erickson, N.; Laviano, A.; Lisanti, M.P.; Lobo, D.N.; et al. ESPEN expert group recommendations for action against cancer-related malnutrition. Clin. Nutr. 2017, 36, 1187–1196. [Google Scholar] [CrossRef] [PubMed]
- Guinan, E.M.; Doyle, S.L.; O’Neill, L.; Dunne, M.R.; Foley, E.K.; O’Sullivan, J.; Reynolds, J.V.; Hussey, J. Effects of a multimodal rehabilitation programme on inflammation and oxidative stress in oesophageal cancer survivors: The ReStOre feasibility study. Support Care Cancer 2017, 25, 749–756. [Google Scholar] [CrossRef]
- Shenkin, A. The key role of micronutrients. Clin. Nutr. 2006, 25, 1–13. [Google Scholar] [CrossRef]
- Ströhle, A.; Zänker, K.; Hahn, A. Nutrition in oncology: The case of micronutrients (review). Oncol. Rep. 2010, 24, 815–828. [Google Scholar] [CrossRef]
- Corry, J.; Poon, W.; McPhee, N.; Milner, A.D.; Cruickshank, D.; Porceddu, S.V.; Rischin, D.; Peters, L.J. Randomized study of percutaneous endoscopic gastrostomy versus nasogastric tubes for enteral feeding in head and neck cancer patients treated with (chemo)radiation. J. Med. Imaging Radiat. Oncol. 2008, 52, 503–510. [Google Scholar] [CrossRef]
- Nugent, B.; Parker, M.J.; McIntyre, I.A. Nasogastric tube feeding and percutaneous endoscopic gastrostomy tube feeding in patients with head and neck cancer. J. Hum. Nutr. Diet. 2010, 23, 277–284. [Google Scholar] [CrossRef]
- Bozzetti, F.; Santarpia, L.; Pironi, L.; Thul, P.; Klek, S.; Gavazzi, C.; Tinivella, M.; Joly, F.; Jonkers, C.; Baxter, J.; et al. The prognosis of incurable cachectic cancer patients on home parenteral nutrition: A multi-centre observational study with prospective follow-up of 414 patients. Ann. Oncol. 2014, 25, 487–493. [Google Scholar] [CrossRef]
- Muscaritoli, M.; Corsaro, E.; Molfino, A. Awareness of Cancer-Related Malnutrition and Its Management: Analysis of the Results from a Survey Conducted Among Medical Oncologists. Front. Oncol. 2021, 11, 682999. [Google Scholar] [CrossRef]
- Cano, N.; Fiaccadori, E.; Tesinsky, P.; Toigo, G.; Druml, W.; Kuhlmann, M.; Mann, H.; Hörl, W.H.; DGEM (German Society for Nutritional Medicine); ESPEN (European Society for Parenteral and Enteral Nutrition). ESPEN Guidelines on Enteral Nutrition: Adult renal failure. Clin. Nutr. 2006, 25, 295–310. [Google Scholar] [CrossRef] [PubMed]
- Aquilani, R.; D’Antona, G.; Baiardi, P.; Gambino, A.; Iadarola, P.; Viglio, S.; Pasini, E.; Verri, M.; Barbieri, A.; Boschi, F. Essential amino acids and exercise tolerance in elderly muscle-depleted subjects with chronic diseases: A rehabilitation without rehabilitation? Biomed. Res. Int. 2014, 2014, 341603. [Google Scholar] [CrossRef] [PubMed]
- Ikizler, T.A.; Burrowes, J.D.; Byham-Gray, L.D.; Campbell, K.L.; Carrero, J.J.; Chan, W.; Fouque, D.; Friedman, A.N.; Ghaddar, S.; Goldstein-Fuchs, D.J.; et al. KDOQI Clinical Practice Guideline for Nutrition in CKD: 2020 Update. Am. J. Kidney Dis. 2020, 76, S1–S107. [Google Scholar] [CrossRef]
- European Association for the Study of the Liver. EASL Clinical Practice Guidelines on nutrition in chronic liver disease. J. Hepatol. 2019, 70, 172–193. [Google Scholar] [CrossRef]
- Glass, C.; Hipskind, P.; Tsien, C.; Malin, S.K.; Kasumov, T.; Shah, S.N.; Kirwan, J.P.; Dasarathy, S. Sarcopenia and a physiologically low respiratory quotient in patients with cirrhosis: A prospective controlled study. J. Appl. Physiol. 2013, 114, 559–565. [Google Scholar] [CrossRef]
- Tsien, C.D.; McCullough, A.J.; Dasarathy, S. Late evening snack: Exploiting a period of anabolic opportunity in cirrhosis. J. Gastroenterol. Hepatol. 2012, 27, 430–441. [Google Scholar] [CrossRef]
- Vaisman, N.; Katzman, H.; Carmiel-Haggai, M.; Lusthaus, M.; Niv, E. Breakfast improves cognitive function in cirrhotic patients with cognitive impairment. Am. J. Clin. Nutr. 2010, 92, 137–140. [Google Scholar] [CrossRef]
- Plank, L.D.; Gane, E.J.; Peng, S.; Muthu, C.; Mathur, S.; Gillanders, L.; McIlroy, K.; Donaghy, A.J.; McCall, J.L. Nocturnal nutritional supplementation improves total body protein status of patients with liver cirrhosis: A randomized 12-month trial. Hepatology 2008, 48, 557–566. [Google Scholar] [CrossRef]
- Rendina, M.; Viggiani, M.T.; Di Leo, A.; Barone, M. Malnutrition, sarcopenia, and refractory ascites in end stage liver diseases: Is there a way to climb back up? Dig. Liver Dis. 2019, 51, 1513–1514. [Google Scholar] [CrossRef]
- Yoshida, T.; Muto, Y.; Moriwaki, H.; Yamato, M. Effect of long-term oral supplementation with branched-chain amino acid granules on the prognosis of liver cirrhosis. Gastroenterol. Jpn. 1989, 24, 692–698. [Google Scholar] [CrossRef]
- Nakaya, Y.; Harada, N.; Kakui, S.; Okada, K.; Takahashi, A.; Inoi, J.; Ito, S. Severe catabolic state after prolonged fasting in cirrhotic patients: Effect of oral branched-chain amino-acid-enriched nutrient mixture. J. Gastroenterol. 2002, 37, 531–536. [Google Scholar] [CrossRef] [PubMed]
- Rolland, Y.; Dray, C.; Vellas, B.; Barreto, P.S. Current and investigational medications for the treatment of sarcopenia. Metabolism 2023, 149, 155597. [Google Scholar] [CrossRef] [PubMed]
- Kucharski, R.; Maleszka, J.; Foret, S.; Maleszka, R. Nutritional control of reproductive status in honeybees via DNA methylation. Science 2008, 319, 1827–1830. [Google Scholar] [CrossRef] [PubMed]


| Diagnostic Phase | Main Parameter | Description | Type of Measurement |
|---|---|---|---|
| Suspected sarcopenia | Muscle strength | The reduction is an early indicator of sarcopenia. | Handgrip test |
| Confirmation of sarcopenia | Muscle mass | The reduction confirms the diagnosis of sarcopenia. | DEXA, BIA |
| High-grade sarcopenia | Physical performance | When it is associated with a reduction in function and mobility. | Walk speed test |
| Type of Sarcopenia | Methods of Identification |
|---|---|
| Primary Sarcopenia | |
| Age-related sarcopenia | Absence of other causes. |
| Secondary Sarcopenia | |
| Sarcopenia related to physical activity | Sedentary lifestyle, bed rest syndrome. |
| Sarcopenia related to diseases | From advanced organ failure, inflammatory disease, neoplasia, or endocrine disease. |
| Sarcopenia related to nutritional aspects | From inadequate energy and/or protein intake, intestinal malabsorption, or anorexia. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barone, M.; Baccaro, P.; Molfino, A. An Overview of Sarcopenia: Focusing on Nutritional Treatment Approaches. Nutrients 2025, 17, 1237. https://doi.org/10.3390/nu17071237
Barone M, Baccaro P, Molfino A. An Overview of Sarcopenia: Focusing on Nutritional Treatment Approaches. Nutrients. 2025; 17(7):1237. https://doi.org/10.3390/nu17071237
Chicago/Turabian StyleBarone, Michele, Palmina Baccaro, and Alessio Molfino. 2025. "An Overview of Sarcopenia: Focusing on Nutritional Treatment Approaches" Nutrients 17, no. 7: 1237. https://doi.org/10.3390/nu17071237
APA StyleBarone, M., Baccaro, P., & Molfino, A. (2025). An Overview of Sarcopenia: Focusing on Nutritional Treatment Approaches. Nutrients, 17(7), 1237. https://doi.org/10.3390/nu17071237








