Redefining the Role of Nutrition in Infant Food Allergy Prevention: A Narrative Review
Abstract
1. Introduction
2. Food Allergy
2.1. Development
2.2. Allergenic Foods
2.3. Prevalence of Food Allergies in the United States
2.4. The Atopic March
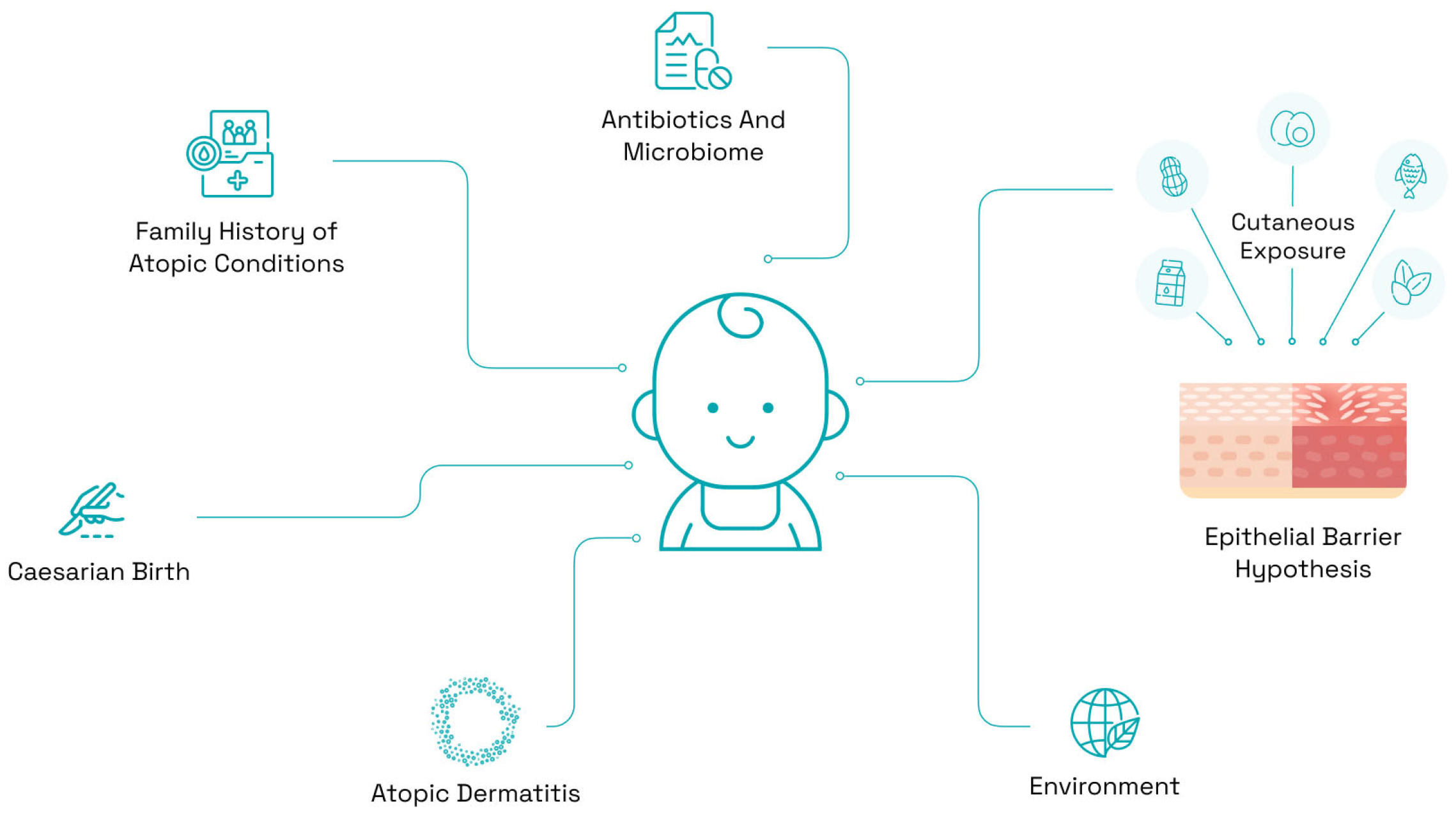
3. Risk Factors for Food Allergy
3.1. Family History of Atopic Conditions
3.2. Epicutaneous Sensitization
3.3. Birth/Mode of Delivery
3.4. Environment
3.5. Vitamin D and Season of Birth
3.6. Epithelial Barrier Hypothesis
3.7. The Microbiome and Antibiotic Usage
3.8. Piecing Together the Puzzle
4. Dietary Factors in Allergic Disease
4.1. Maternal Diet in Pregnancy
4.1.1. Food
4.1.2. Supplements
4.2. Maternal Diet While Breastfeeding
4.3. Infant Diet
4.3.1. Breastfeeding and Formula Feeding
4.3.2. Supplements
4.3.3. Complementary Feeding
4.3.4. Early Allergen Introduction
5. Conclusions
- Sidebar 1. Guidelines on Feeding with Formula (Adapted from the USDA & AAP)
- First Days:
- In total, 1 to 2 ounces of infant formula every 2 to 3 h in the first days of life.
- Give more if showing signs of hunger.
- Most infant formula-fed newborns will feed 8 to 12 times in 24 h.
- First Weeks and Months:
- Feed every 3 to 4 h.
- Babies will generally take what they need at each feeding and stop eating when they are full.
- In total, 3 to 4 ounces (90 to 120 mL) per feed and 32 ounces per day.
- If an infant sleeps longer than 4 to 5 h during the first few weeks after birth and starts missing feedings, parents should wake them up and offer a bottle.
- Provide Vitamin D Supplementation if the infant drinks less than 32 ounces of infant formula a day,
- By 6 months: Your baby will consume 6 to 8 ounces (180–240 mL) at each of 4 or 5 feedings in 24 h.
- Measurements:
- 1 ounce = 30 cc (cubic centimeters) = 30 mL (milliliters)
- 8 fluid ounces = 1 cup
- 32 fluid ounces = 1 quart
- Scoop of powder to every 2 fluid ounces of water.
- In total, 2 fluid ounces of concentrate with 2 fluid ounces of water.
- Sidebar 2. Dietitian’s Role in Food Allergy Prevention
- Support breastfeeding.
- Nutritional guidance to the breast-feeding parent.
- Advice on suitable infant formula (if needed).
- Guidance on responsive feeding styles.
- Advice on nutritionally appropriate foods.
- Advice on suitable textures and texture progression.
- Introduction of food allergens in terms, including:
- ○
- amount;
- ○
- type;
- ○
- frequency.
- Monitoring growth and nutritional status.
- Incorporating food allergens into the usual family diet.
- Overcoming food aversions.
Author Contributions
Funding
Conflicts of Interest
List of Abbreviations
| AAAAI | American Academy of Allergy, Asthma and Immunology |
| AAP | American Academy of Pediatrics |
| ACAAI | American College of Allergy, Asthma, and Immunology |
| AD | Atopic Dermatitis |
| DII | Dietary Inflammation Index |
| FA | Food Allergy |
| FARE | Food Allergy Research and Education |
| FDA | Food and Drug Administration |
| FLG | Filaggrin |
| HEI | Healthy Eating Index |
| IgE | Immunoglobin E |
| IL | Interleukin |
| LEAP | Learning Early About Peanuts |
| RCT | Randomized Controlled Trial |
| sIgE | Specific Immunoglobin E |
| US | United States |
| USDA | United States Department of Agriculture |
| VD | Vaginal Delivery |
| WHO | World Health Organization |
References
- Jackson, K.D.; Howie, L.D.; Akinbami, L.J. Trends in Allergic Conditions among Children: United States, 1997–2011; NCHS Data Brief; US Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics: Hyattsville, MD, USA, 2013; pp. 1–8.
- Keet, C.A.; Savage, J.H.; Seopaul, S.; Peng, R.D.; Wood, R.A.; Matsui, E.C. Temporal trends and racial/ethnic disparity in self-reported pediatric food allergy in the United States. Ann. Allergy Asthma Immunol. 2014, 112, 222–229.e3. [Google Scholar] [CrossRef] [PubMed]
- Clark, S.; Espinola, J.; Rudders, S.A.; Banerji, A.; Camargo, C.A. Frequency of US emergency department visits for food-related acute allergic reactions. J. Allergy Clin. Immunol. 2011, 127, 682–683. [Google Scholar] [CrossRef] [PubMed]
- Bock, S.A.; Muñoz-Furlong, A.; Sampson, H.A. Further fatalities caused by anaphylactic reactions to food, 2001–2006. J. Allergy Clin. Immunol. 2007, 119, 1016–1018. [Google Scholar] [CrossRef] [PubMed]
- Sampson, H.A.; Mendelson, L.; Rosen, J.P. Fatal and near-fatal anaphylactic reactions to food in children and adolescents. N. Engl. J. Med. 1992, 327, 380–384. [Google Scholar] [CrossRef] [PubMed]
- Walker, S.O.; Mao, G.; Caruso, D.; Hong, X.; Pongracic, J.A.; Wang, X. Cardiovascular Risk Factors in Parents of Food-Allergic Children. Medicine 2016, 95, e3156. [Google Scholar] [CrossRef]
- Shemesh, E.; Annunziato, R.A.; Ambrose, M.A.; Ravid, N.L.; Mullarkey, C.; Rubes, M.; Chuang, K.; Sicherer, M.; Sicherer, S.H. Child and parental reports of bullying in a consecutive sample of children with food allergy. Pediatrics 2013, 131, e10–e17. [Google Scholar] [CrossRef]
- Gupta, R.; Holdford, D.; Bilaver, L.; Dyer, A.; Holl, J.L.; Meltzer, D. The economic impact of childhood food allergy in the United States. JAMA Pediatr. 2013, 167, 1026–1031. [Google Scholar] [CrossRef] [PubMed]
- Fleischer, D.M.; Conover-Walker, M.K.; Matsui, E.C.; Wood, R.A. The natural history of tree nut allergy. J. Allergy Clin. Immunol. 2005, 116, 1087–1093. [Google Scholar] [CrossRef]
- Fleischer, D.M.; Conover-Walker, M.K.; Christie, L.; Burks, A.W.; Wood, R.A. Peanut allergy: Recurrence and its management. J. Allergy Clin. Immunol. 2004, 114, 1195–1201. [Google Scholar] [CrossRef]
- Jimenez, J.; Paller, A.S. The atopic march and its prevention. Ann. Allergy Asthma Immunol. 2021, 127, 289–290. [Google Scholar] [CrossRef]
- Bawany, F.; Beck, L.A.; Järvinen, K.M. Halting the March: Primary Prevention of Atopic Dermatitis and Food Allergies. J. Allergy Clin. Immunol. Pract. 2020, 8, 860–875. [Google Scholar] [CrossRef] [PubMed]
- Schroer, B.; Groetch, M.; Mack, D.P.; Venter, C. Practical challenges and considerations for early introduction of potential food allergens for prevention of food allergy. J. Allergy Clin. Immunol. Pract. 2020, 9, 44–56.e1. [Google Scholar] [CrossRef] [PubMed]
- Eiwegger, T.; Hung, L.; Diego, K.E.S.; O’Mahony, L.; Upton, J. Recent developments and highlights in food allergy. Allergy 2019, 74, 2355–2367. [Google Scholar] [CrossRef] [PubMed]
- Żukiewicz-Sobczak, W.A.; Wróblewska, P.; Adamczuk, P.; Kopczyński, P. Causes, symptoms and prevention of food allergy. Adv. Dermatol. Aller-Gology/Postępy Dermatol. I Alergol. 2013, 30, 113–116. [Google Scholar] [CrossRef]
- Shade, K.-T.C.; Conroy, M.E.; Washburn, N.; Kitaoka, M.; Huynh, D.J.; Laprise, E.; Patil, S.U.; Shreffler, W.G.; Anthony, R.M. Sialylation of immunoglobulin E is a determinant of allergic pathogenicity. Nature 2020, 582, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Ballmer-Weber, B.K. Allergic Reactions to Food Proteins. Int. J. Vitam. Nutr. Res. 2011, 81, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Warren, C.M.; Jiang, J.; Gupta, R.S. Epidemiology and Burden of Food Allergy. Curr. Allergy Asthma Rep. 2020, 20, 6. [Google Scholar] [CrossRef] [PubMed]
- Sudharson, S.; Kalic, T.; Hafner, C.; Breiteneder, H. Newly defined allergens in the WHO/IUIS Allergen Nomenclature Database during 01/2019–03/2021. Allergy 2021, 76, 3359–3373. [Google Scholar] [CrossRef]
- Foong, R.-X.; Santos, A.F. Biomarkers of diagnosis and resolution of food allergy. Pediatr. Allergy Immunol. 2021, 32, 223–233. [Google Scholar] [CrossRef]
- National Academies of Sciences. Finding a Path to Safety in Food Allergy: Assessment of the Global Burden, Causes, Prevention, Management, and Public Policy; Oria, M.P., Stallings, V.A., Eds.; National Academies Press (US): Washington, DC, USA, 2016. [Google Scholar]
- Gupta, R.S.; Warren, C.M.; Smith, B.M.; Blumenstock, J.A.; Jiang, J.; Davis, M.M.; Nadeau, K.C. The Public Health Impact of Parent-Reported Childhood Food Allergies in the United States. Pediatrics 2018, 142, e20181235. [Google Scholar] [CrossRef]
- Adatia, A.; Clarke, A.E.; Yanishevsky, Y.; Ben-Shoshan, M. Sesame allergy: Current perspectives. J. Asthma Allergy 2017, 10, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Warren, C.M.; Chadha, A.S.; Sicherer, S.H.; Jiang, J.; Gupta, R.S. Prevalence and Severity of Sesame Allergy in the United States. JAMA Netw. Open 2019, 2, e199144. [Google Scholar] [CrossRef] [PubMed]
- Peters, R.L.; Krawiec, M.; Koplin, J.J.; Santos, A.F. Update on food allergy. Pediatr. Allergy Immunol. 2021, 32, 647–657. [Google Scholar] [CrossRef]
- Tham, E.H.; Leung, A.S.Y.; Pacharn, P.; Lee, S.Y.; Ebisawa, M.; Lee, B.W.; Wong, G.W.K.; The APAPARI Anaphylaxis Study Group. Anaphylaxis—Lessons learnt when East meets West. Pediatr. Allergy Immunol. 2019, 30, 681–688. [Google Scholar] [CrossRef] [PubMed]
- Botha, M.; Basera, W.; Facey-Thomas, H.E.; Gaunt, B.; Gray, C.L.; Ramjith, J.; Watkins, A.; Levin, M.E. Rural and urban food allergy prevalence from the South African Food Allergy (SAFFA) study. J. Allergy Clin. Immunol. 2019, 143, 662–668.e2. [Google Scholar] [CrossRef] [PubMed]
- Santos, A.F.; Riggioni, C.; Agache, I.; Akdis, C.A.; Akdis, M.; Alvarez-Perea, A.; Alvaro-Lozano, M.; Ballmer-Weber, B.; Barni, S.; Beyer, K.; et al. EAACI guidelines on the diagnosis of IgE-mediated food allergy. Allergy 2023, 78, 3057–3076. [Google Scholar] [CrossRef]
- Paller, A.S.; Spergel, J.M.; Mina-Osorio, P.; Irvine, A.D. The atopic march and atopic multimorbidity: Many trajectories, many pathways. J. Allergy Clin. Immunol. 2019, 143, 46–55. [Google Scholar] [CrossRef]
- Illi, S.; von Mutius, E.; Lau, S.; Nickel, R.; Grüber, C.; Niggemann, B.; Wahn, U.; The Multicenter Allergy Study Group. The natural course of atopic dermatitis from birth to age 7 years and the association with asthma. J. Allergy Clin. Immunol. 2004, 113, 925–931. [Google Scholar] [CrossRef]
- Ziyab, A.H.; Hankinson, J.; Ewart, S.; Schauberger, E.; Kopec-Harding, K.; Zhang, H.; Custovic, A.; Arshad, H.; Simpson, A. Epistasis between FLG and IL4R Genes on the Risk of Allergic Sensitization: Results from Two Population-Based Birth Cohort Studies. Sci. Rep. 2018, 8, 3221. [Google Scholar] [CrossRef]
- van der Hulst, A.E.; Klip, H.; Brand, P.L. Risk of developing asthma in young children with atopic eczema: A systematic review. J. Allergy Clin. Immunol. 2007, 120, 565–569. [Google Scholar] [CrossRef]
- Tran, M.M.; Lefebvre, D.L.; Dharma, C.; Dai, D.; Lou, W.Y.; Subbarao, P.; Becker, A.B.; Mandhane, P.J.; Turvey, S.E.; Sears, M.R.; et al. Predicting the atopic march: Results from the Canadian Healthy Infant Longitudinal Development Study. J. Allergy Clin. Immunol. 2018, 141, 601–607.e8. [Google Scholar] [CrossRef] [PubMed]
- Abo-Zaid, G.; Sharpe, R.A.; Fleming, L.E.; Depledge, M.; Osborne, N.J. Association of Infant Eczema with Childhood and Adult Asthma: Analysis of Data from the 1958 Birth Cohort Study. Int. J. Environ. Res. Public Health 2018, 15, 1415. [Google Scholar] [CrossRef] [PubMed]
- Spergel, J.M.; Toit, G.D.; Davis, C.M. Might biologics serve to interrupt the atopic march? J. Allergy Clin. Immunol. 2023, 151, 590–594. [Google Scholar] [CrossRef] [PubMed]
- Cergan, R.; Berghi, O.N.; Dumitru, M.; Vrinceanu, D.; Manole, F.; Serboiu, C.S. Biologics for Chronic Rhinosinusitis—A Modern Option for Therapy. Life 2023, 13, 2165. [Google Scholar] [CrossRef] [PubMed]
- E Childs, C.; Munblit, D.; Ulfman, L.; Gómez-Gallego, C.; Lehtoranta, L.; Recker, T.; Salminen, S.; Tiemessen, M.; Collado, M.C. Potential Biomarkers, Risk Factors, and Their Associations with IgE-Mediated Food Allergy in Early Life: A Narrative Review. Adv. Nutr. Int. Rev. J. 2022, 13, 633–651. [Google Scholar] [CrossRef] [PubMed]
- Koplin, J.J.; Allen, K.J.; Gurrin, L.C.; Peters, R.L.; Lowe, A.J.; Tan, H.-T.T.; Dharmage, S.C. The impact of family history of allergy on risk of food allergy: A population-based study of infants. Int. J. Environ. Res. Public Health 2013, 10, 5364–5377. [Google Scholar] [CrossRef] [PubMed]
- Kanchan, K.; Clay, S.; Irizar, H.; Bunyavanich, S.; Mathias, R.A. Current insights into the genetics of food allergy. J. Allergy Clin. Immunol. 2021, 147, 15–28. [Google Scholar] [CrossRef]
- Madore, A.-M.; Vaillancourt, V.T.; Asai, Y.; Alizadehfar, R.; Ben-Shoshan, M.; Michel, D.L.; Kozyrskyj, A.L.; Becker, A.; Chan-Yeung, M.; E Clarke, A.; et al. HLA-DQB1*02 and DQB1*06:03P are associated with peanut allergy. Eur. J. Hum. Genet. 2013, 21, 1181–1184. [Google Scholar] [CrossRef]
- Asai, Y.; Eslami, A.; van Ginkel, C.D.; Akhabir, L.; Wan, M.; Ellis, G.; Ben-Shoshan, M.; Martino, D.; Ferreira, M.A.; Allen, K.; et al. Genome-wide association study and meta-analysis in multiple populations identifies new loci for peanut allergy and establishes C11orf30/EMSY as a genetic risk factor for food allergy. J. Allergy Clin. Immunol. 2018, 141, 991–1001. [Google Scholar] [CrossRef]
- Fleischer, D.M.; Chan, E.S.; Venter, C.; Spergel, J.M.; Abrams, E.M.; Stukus, D.; Groetch, M.; Shaker, M.; Greenhawt, M. A consensus approach to the primary prevention of food allergy through nutrition: Guidance from the American Academy of Allergy, Asthma, and Immunology; American College of Allergy, Asthma, and Immunology; and the Canadian Society for Al-lergy and Clinical Immunology. J. Allergy Clin. Immunol. Pract. 2021, 9, 22–43.e4. [Google Scholar] [CrossRef]
- Lopez-Ojeda, W.; Pandey, A.; Alhajj, M.; Oakley, A.M. Anatomy, Skin (Integument); StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Sozener, Z.C.; Ozturk, B.O.; Cerci, P.; Turk, M.; Akin, B.G.; Akdis, M.; Altiner, S.; Ozbey, U.; Ogulur, I.; Mitamura, Y.; et al. Epithelial barrier hypothesis: Effect of the external exposome on the microbiome and epithelial barriers in allergic disease. Allergy 2022, 77, 1418–1449. [Google Scholar] [CrossRef] [PubMed]
- Medline. FLG Gene. Available online: https://medlineplus.gov/genetics/gene/flg/#:~:text=Filaggrin%20plays%20an%20important%20role,to%20create%20a%20strong%20barrier (accessed on 9 November 2023).
- Akdis, M.; Aab, A.; Altunbulakli, C.; Azkur, K.; Costa, R.A.; Crameri, R.; Duan, S.; Eiwegger, T.; Eljaszewicz, A.; Ferstl, R.; et al. Interleukins (from IL-1 to IL-38), interferons, transforming growth factor β, and TNF-α: Receptors, functions, and roles in diseases. J. Allergy Clin. Immunol. 2016, 138, 984–1010. [Google Scholar] [CrossRef] [PubMed]
- Sicherer, S.H.; Wood, R.A.; Vickery, B.P.; Jones, S.M.; Liu, A.H.; Fleischer, D.M.; Dawson, P.; Mayer, L.; Burks, A.W.; Grishin, A.; et al. The natural history of egg allergy in an observational cohort. J. Allergy Clin. Immunol. 2014, 133, 492–499.e8. [Google Scholar] [CrossRef] [PubMed]
- Coelho, G.D.P.; Ayres, L.F.A.; Barreto, D.S.; Henriques, B.D.; Prado, M.R.M.C.; Dos Passos, C.M. Acquisition of microbiota according to the type of birth: An integrative review. Rev. Latino-Americana Enferm. 2021, 29, e3446. [Google Scholar] [CrossRef] [PubMed]
- Pyrhönen, K.; Kulmala, P. Delivery mode and the incidence of atopic sensitization and food allergy in a Finnish child population. Pediatr. Allergy Immunol. 2022, 33, e13584. [Google Scholar] [CrossRef] [PubMed]
- Currell, A.; Koplin, J.J.; Lowe, A.J.; Perrett, K.P.; Ponsonby, A.-L.; Tang, M.L.; Dharmage, S.C.; Peters, R.L. Mode of Birth Is Not Associated with Food Allergy Risk in Infants. J. Allergy Clin. Immunol. Pract. 2022, 10, 2135–2143.e3. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Zhou, C.; Guo, C.; Wang, J.; Chen, I.; Wen, S.W.; Krewski, D.; Yue, L.; Xie, R. The prevalence of food allergy in cesarean-born children aged 0–3 years: A systematic review and meta-analysis of cohort studies. Front. Pediatr. 2023, 10, 1044954. [Google Scholar] [CrossRef]
- Peters, R.L.; Mavoa, S.; Koplin, J.J. An Overview of Environmental Risk Factors for Food Allergy. Int. J. Environ. Res. Public Health 2022, 19, 722. [Google Scholar] [CrossRef]
- Krempski, J.W.; Dant, C.; Nadeau, K.C. The origins of allergy from a systems approach. Ann. Allergy Asthma Immunol. 2020, 125, 507–516. [Google Scholar] [CrossRef]
- Berghi, O.N.; Vrinceanu, D.; Cergan, R.; Dumitru, M.; Costache, A. Solanum melongena allergy (A comprehensive review). Exp. Ther. Med. 2021, 22, 1061. [Google Scholar] [CrossRef]
- Allen, K.J.; Koplin, J.J. Prospects for prevention of food allergy. J. Allergy Clin. Immunol. Pract. 2016, 4, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Giannetti, A.; Bernardini, L.; Cangemi, J.; Gallucci, M.; Masetti, R.; Ricci, G. Role of Vitamin D in Prevention of Food Allergy in Infants. Front. Pediatr. 2020, 8, 447. [Google Scholar] [CrossRef] [PubMed]
- Nwaru, B.I.; Ahonen, S.; Kaila, M.; Erkkola, M.; Haapala, A.; Kronberg-Kippilä, C.; Veijola, R.; Ilonen, J.; Simell, O.; Knip, M.; et al. Maternal diet during pregnancy and allergic sensitization in the offspring by 5 yrs of age: A prospective cohort study. Pediatr. Allergy Immunol. 2010, 21, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Allen, K.J.; Koplin, J.J.; Ponsonby, A.-L.; Gurrin, L.C.; Wake, M.; Vuillermin, P.; Martin, P.; Matheson, M.; Lowe, A.; Robinson, M.; et al. Vitamin D insufficiency is associated with challenge-proven food allergy in infants. J. Allergy Clin. Immunol. 2013, 131, 1109–1116.e6. [Google Scholar] [CrossRef] [PubMed]
- Molloy, J.; Koplin, J.J.; Allen, K.J.; Tang, M.L.K.; Collier, F.; Carlin, J.B.; Saffery, R.; Burgner, D.; Ranganathan, S.; Dwyer, T.; et al. Vitamin D insufficiency in the first 6 months of infancy and challenge-proven IgE-mediated food allergy at 1 year of age: A case-cohort study. Allergy 2017, 72, 1222–1231. [Google Scholar] [CrossRef] [PubMed]
- Norizoe, C.; Akiyama, N.; Segawa, T.; Tachimoto, H.; Mezawa, H.; Ida, H.; Urashima, M. Increased food allergy and vitamin D: Randomized, double-blind, placebo-controlled trial. Pediatr. Int. 2014, 56, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Rueter, K.; Jones, A.P.; Siafarikas, A.; Lim, E.-M.; Prescott, S.L.; Palmer, D.J. In “High-Risk” Infants with Sufficient Vitamin D Status at Birth, Infant Vitamin D Supplementation Had No Effect on Allergy Outcomes: A Randomized Controlled Trial. Nutrients 2020, 12, 1747. [Google Scholar] [CrossRef]
- Rosendahl, J.; Pelkonen, A.S.; Helve, O.; Hauta-Alus, H.; Holmlund-Suila, E.; Valkama, S.; Enlund-Cerullo, M.; Viljakainen, H.; Hytinantti, T.; Mäkitie, O.; et al. High-Dose Vitamin D Supplementation Does Not Prevent Allergic Sensitization of Infants. J. Pediatr. 2019, 209, 139–145.e1. [Google Scholar] [CrossRef]
- Zhong, Y.; Samuel, M.; van Bever, H.; Tham, E.H. Emollients in infancy to prevent atopic dermatitis: A systematic review and meta-analysis. Allergy 2022, 77, 1685–1699. [Google Scholar] [CrossRef]
- Loraud, C.; de Ménonville, C.T.; Bourgoin-Heck, M.; Cottel, N.; Wanin, S.; Just, J. Emergence of pollen food allergy syndrome in asthmatic children in Paris. Pediatr. Allergy Immunol. 2021, 32, 702–708. [Google Scholar] [CrossRef]
- Chen, N.T.; Chen, M.J.; Wu, C.D.; Guo, Y.L. Emergency room visits for childhood atopic dermatitis are associated with floods? Sci. Total Environ. 2021, 773, 145435. [Google Scholar] [CrossRef]
- Renz, H.; Skevaki, C. Early life microbial exposures and allergy risks: Opportunities for prevention. Nat. Rev. Immunol. 2021, 21, 177–191. [Google Scholar] [CrossRef] [PubMed]
- Bunyavanich, S.; Berin, M.C. Food allergy and the microbiome: Current understandings and future directions. J. Allergy Clin. Immunol. 2019, 144, 1468–1477. [Google Scholar] [CrossRef] [PubMed]
- Petersen, C.; Dai, D.L.; Boutin, R.C.; Sbihi, H.; Sears, M.R.; Moraes, T.J.; Becker, A.B.; Azad, M.B.; Mandhane, P.J.; Subbarao, P.; et al. A rich meconium metabolome in human infants is associated with early-life gut microbiota composition and reduced allergic sensitization. Cell Rep. Med. 2021, 2, 100260. [Google Scholar] [CrossRef] [PubMed]
- Joseph, C.L.; Sitarik, A.R.; Kim, H.; Huffnagle, G.; Fujimura, K.; Yong, G.J.M.; Levin, A.M.; Zoratti, E.; Lynch, S.; Ownby, D.R.; et al. Infant gut bacterial community composition and food-related manifestation of atopy in early childhood. Pediatr. Allergy Immunol. 2022, 33, e13704. [Google Scholar] [CrossRef] [PubMed]
- Roduit, C.; Frei, R.; Ferstl, R.; Loeliger, S.; Westermann, P.; Rhyner, C.; Schiavi, E.; Barcik, W.; Rodriguez-Perez, N.; Wawrzyniak, M.; et al. High levels of butyrate and propionate in early life are associated with protection against atopy. Allergy 2019, 74, 799–809. [Google Scholar] [CrossRef] [PubMed]
- de Silva, D.; Halken, S.; Singh, C.; Muraro, A.; Angier, E.; Arasi, S.; Arshad, H.; Beyer, K.; Boyle, R.; Toit, G.D.; et al. Preventing food allergy in infancy and childhood: Systematic review of randomised controlled trials. Pediatr. Allergy Immunol. 2020, 31, 813–826. [Google Scholar] [CrossRef] [PubMed]
- Landau, T.; Gamrasni, K.; Barlev, Y.; Elizur, A.; Benor, S.; Mimouni, F.; Brandwein, M. A machine learning approach for stratifying risk for food allergies utilizing electronic medical record data. Allergy 2023, 79, 499–502. [Google Scholar] [CrossRef]
- Sugiura, S.; Hiramitsu, Y.; Futamura, M.; Kamioka, N.; Yamaguchi, C.; Umemura, H.; Ito, K.; Camargo, C.A. Development of a prediction model for infants at high risk of food allergy. Asia Pac. Allergy 2021, 11, e5. [Google Scholar] [CrossRef]
- Delvert, R.; Ghozal, M.; Adel-Patient, K.; Kadawathagedara, M.; Heude, B.; Charles, M.-A.; Annesi-Maesano, I.; Tafflet, M.; Leynaert, B.; Varraso, R.; et al. Maternal Diet Quality during Pregnancy and Allergic and Respiratory Multimorbidity Clusters in Children from the EDEN Mother–Child Cohort. Nutrients 2023, 15, 146. [Google Scholar] [CrossRef]
- Jerschow, E.; McGinn, A.P.; de Vos, G.; Vernon, N.; Jariwala, S.; Hudes, G.; Rosenstreich, D. Dichlorophenol-containing pesticides and allergies: Results from the US National Health and Nutrition Examination Survey 2005–2006. Ann. Allergy Asthma Immunol. 2012, 109, 420–425. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, H.; Matsuhara, H.; Miotani, S.; Sako, Y.; Matsui, T.; Tanaka, H.; Inagaki, N. Artificial sweeteners and mixture of food additives cause to break oral tolerance and induce food allergy in murine oral tolerance model for food allergy. Clin. Exp. Allergy 2017, 47, 1204–1213. [Google Scholar] [CrossRef] [PubMed]
- Togias, A.; Cooper, S.F.; Acebal, M.L.; Assa’Ad, A.; Baker, J.R.; Beck, L.A.; Block, J.; Byrd-Bredbenner, C.; Chan, E.S.; Eichenfield, L.F.; et al. Addendum guidelines for the prevention of peanut allergy in the United States: Report of the National Institute of Allergy and Infectious Diseases–sponsored expert panel. J. Allergy Clin. Immunol. 2017, 139, 29–44. [Google Scholar] [CrossRef] [PubMed]
- Greer, F.R.; Sicherer, S.H.; Burks, A.W. Effects of early nutritional interventions on the development of atopic disease in infants and children: The role of maternal dietary restriction, breastfeeding, timing of introduction of complementary foods, and hydrolyzed formulas. Pediatrics 2008, 121, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Venter, C.; Palumbo, M.P.; Glueck, D.H.; Sauder, K.A.; O’Mahony, L.; Fleischer, D.M.; Ben-Abdallah, M.; Ringham, B.M.; Dabelea, D. The maternal diet index in pregnancy is associated with offspring allergic diseases: The Healthy Start study. Allergy 2022, 77, 162–172. [Google Scholar] [CrossRef] [PubMed]
- Bodén, S.; Lindam, A.; Domellöf, M.; Venter, C.; West, C.E. Diet diversity in pregnancy and early allergic manifestations in the offspring. Clin. Exp. Allergy 2023, 53, 963–968. [Google Scholar] [CrossRef]
- Moonesinghe, H.; Patil, V.K.; Dean, T.; Arshad, S.; Glasbey, G.; Grundy, J.; Venter, C. Association between healthy eating in pregnancy and allergic status of the offspring in childhood. Ann. Allergy Asthma Immunol. 2016, 116, 163–165. [Google Scholar] [CrossRef] [PubMed]
- Hanson, C.; Rifas-Shiman, S.L.; Shivappa, N.; Wirth, M.D.; Hebert, J.R.; Gold, D.; Camargo, C.A.; Sen, S.; Sordillo, J.E.; Oken, E.; et al. Associations of Prenatal Dietary Inflammatory Potential with Childhood Respiratory Outcomes in Project Viva. J. Allergy Clin. Immunol. Pract. 2020, 8, 945–952.e4. [Google Scholar] [CrossRef]
- Venter, C.; Agostoni, C.; Arshad, S.H.; Ben-Abdallah, M.; Toit, G.D.; Fleischer, D.M.; Greenhawt, M.; Glueck, D.H.; Groetch, M.; Lunjani, N.; et al. Dietary factors during pregnancy and atopic outcomes in childhood: A systematic review from the European Academy of Allergy and Clinical Immunology. Pediatr. Allergy Immunol. 2020, 31, 889–912. [Google Scholar] [CrossRef]
- Hoppenbrouwers, T.; Cvejić Hogervorst, J.H.; Garssen, J.; Wichers, H.J.; Willemsen, L.E.M. Long Chain Polyunsaturated Fatty Acids (LCPUFAs) in the Prevention of Food Allergy. Front. Immunol. 2019, 10, 1118. [Google Scholar] [CrossRef]
- Anandan, C.; Nurmatov, U.; Sheikh, A. Omega 3 and 6 oils for primary prevention of allergic disease: Systematic review and meta-analysis. Allergy 2009, 64, 840–848. [Google Scholar] [CrossRef] [PubMed]
- Vahdaninia, M.; Mackenzie, H.; Dean, T.; Helps, S. ω-3 LCPUFA supplementation during pregnancy and risk of allergic outcomes or sensitization in offspring: A systematic review and meta-analysis. Ann. Allergy Asthma Immunol. 2019, 122, 302–313.e2. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Larsen, V.; Ierodiakonou, D.; Jarrold, K.; Cunha, S.; Chivinge, J.; Robinson, Z.; Geoghegan, N.; Ruparelia, A.; Devani, P.; Trivella, M.; et al. Diet during pregnancy and infancy and risk of allergic or autoimmune disease: A systematic review and meta-analysis. PLoS Med. 2018, 15, e1002507. [Google Scholar] [CrossRef] [PubMed]
- Abrams, E.M.; Shaker, M.S.; Chan, E.S.; A Brough, H.; Greenhawt, M. Prevention of food allergy in infancy: The role of maternal interventions and exposures during pregnancy and lactation. Lancet Child Adolesc. Health 2023, 7, 358–366. [Google Scholar] [CrossRef] [PubMed]
- Cuello-Garcia, C.A.; Brożek, J.L.; Fiocchi, A.; Pawankar, R.; Yepes-Nuñez, J.J.; Terracciano, L.; Gandhi, S.; Agarwal, A.; Zhang, Y.; Schünemann, H.J. Probiotics for the prevention of allergy: A systematic review and meta-analysis of randomized controlled trials. J. Allergy Clin. Immunol. 2015, 136, 952–961. [Google Scholar] [CrossRef] [PubMed]
- Burris, A.D.; Pizzarello, C.; Järvinen, K.M. Immunologic components in human milk and allergic diseases with focus on food allergy. Semin. Perinatol. 2021, 45, 151386. [Google Scholar] [CrossRef] [PubMed]
- Järvinen, K.M.; Konstantinou, G.N.; Pilapil, M.; Arrieta, M.; Noone, S.; Sampson, H.A.; Meddings, J.; Nowak-Węgrzyn, A. Intestinal permeability in children with food allergy on specific elimination diets. Pediatr. Allergy Immunol. 2013, 24, 589–595. [Google Scholar] [CrossRef] [PubMed]
- Venter, C.; Smith, P.K.; Fleischer, D.M. Food allergy prevention: Where are we in 2023? Asia Pac. Allergy 2023, 13, 15–27. [Google Scholar] [CrossRef]
- Furuhjelm, C.; Warstedt, K.; Larsson, J.; Fredriksson, M.; Böttcher, M.F.; Fälth-Magnusson, K.; Duchén, K. Fish oil supplementation in pregnancy and lactation may decrease the risk of infant allergy. Acta Paediatr. 2009, 98, 1461–1467. [Google Scholar] [CrossRef]
- Rekima, A.; Macchiaverni, P.; Turfkruyer, M.; Holvoet, S.; Dupuis, L.; Baiz, N.; Annesi-Maesano, I.; Mercenier, A.; Nutten, S.; Verhasselt, V. Long-term reduction in food allergy susceptibility in mice by combining breastfeeding-induced tolerance and TGF-β-enriched formula after weaning. Clin. Exp. Allergy 2017, 47, 565–576. [Google Scholar] [CrossRef]
- Macchiaverni, P.; Rekima, A.; van den Elsen, L.; Renz, H.; Verhasselt, V. Allergen shedding in human milk: Could it be key for immune system education and allergy prevention? J. Allergy Clin. Immunol. 2021, 148, 679–688. [Google Scholar] [CrossRef] [PubMed]
- Landau, T.; Vakulenko-Lagun, B.; Brandwein, M. The protective effect of moderate maternal peanut consumption on peanut sensitization and allergy. Ann. Allergy Asthma Immunol. 2023, 131, 231–238. [Google Scholar] [CrossRef] [PubMed]
- Azad, M.B.; Dharma, C.; Simons, E.; Tran, M.; Reyna, M.E.; Dai, R.; Becker, A.B.; Marshall, J.; Mandhane, P.J.; Turvey, S.E.; et al. Reduced peanut sensitization with maternal peanut consumption and early peanut introduction while breastfeeding. J. Dev. Orig. Health Dis. 2021, 12, 811–818. [Google Scholar] [CrossRef] [PubMed]
- Järvinen, K.M.; Martin, H.; Oyoshi, M.K. Immunomodulatory effects of breast milk on food allergy. Ann. Allergy Asthma Immunol. 2019, 123, 133–143. [Google Scholar] [CrossRef]
- Greer, F.R.; Sicherer, S.H.; Burks, A.W. The Effects of Early Nutritional Interventions on the Development of Atopic Disease in Infants and Children: The Role of Maternal Dietary Restriction, Breastfeeding, Hydrolyzed Formulas, and Timing of Introduction of Allergenic Complementary Foods. Pediatrics 2019, 143. [Google Scholar] [CrossRef] [PubMed]
- Tuokkola, J.; Luukkainen, P.; Tapanainen, H.; Kaila, M.; Vaarala, O.; Kenward, M.G.; Virta, L.J.; Veijola, R.; Simell, O.; Ilonen, J.; et al. Maternal diet during pregnancy and lactation and cow’s milk allergy in offspring. Eur. J. Clin. Nutr. 2016, 70, 554–559. [Google Scholar] [CrossRef] [PubMed]
- WHO. Infant and Young Child Feeding. 2021. Available online: https://www.who.int/news-room/fact-sheets/detail/infant-and-young-child-feeding (accessed on 29 November 2023).
- NHMRC. Supporting Women to Breastfeed. 2018. Available online: https://www.nhmrc.gov.au/about-us/news-centre/supporting-women-breastfeed#:~:text=It%20is%20recommended%20that%20infants,the%20mother%20and%20child%20desire (accessed on 29 November 2023).
- USDA. MyPlate—Infants. Available online: https://www.myplate.gov/life-stages/infants (accessed on 3 December 2023).
- AAP Parenting Website. Available online: https://www.healthychildren.org/English/Pages/default.aspx (accessed on 7 April 2020).
- Lachover-Roth, I.; Cohen-Engler, A.; Furman, Y.; Shachar, I.; Rosman, Y.; Meir-Shafrir, K.; Mozer-Mandel, M.; Farladansky-Gershnabel, S.; Biron-Shental, T.; Mandel, M.; et al. Early, continuing exposure to cow’s milk formula and cow’s milk allergy: The COMEET study, a single center, prospective interventional study. Ann. Allergy Asthma Immunol. 2023, 130, 233–239.e4. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food and Drug Administration. Infant Formula Guidance Documents & Regulatory Information. 2023. Available online: https://www.fda.gov/food/guidance-documents-regulatory-information-topic-food-and-dietary-supplements/infant-formula-guidance-documents-regulatory-information (accessed on 29 November 2023).
- Psaroulaki, E.; Katsaras, G.N.; Samartzi, P.; Chatziravdeli, V.; Psaroulaki, D.; Oikonomou, E. Association of food allergy in children with vitamin D insufficiency: A systematic review and meta-analysis. Eur. J. Pediatr. 2023, 182, 1533–1554. [Google Scholar] [CrossRef]
- Wagner, C.L.; Greer, F.R.; The Section on Breastfeeding and Committee on Nutrition. Prevention of rickets and vitamin D deficiency in infants, children, and adolescents. Pediatrics 2008, 122, 1142–1152. [Google Scholar] [CrossRef]
- Vitamin D Supplementation for Infants. Biological, Behavioural and Contextual Rationale; Hope Alberta Weiler; School of Human Nutrition; McGill University. July 2017. Available online: https://www.who.int/tools/elena/bbc/vitamind-infants (accessed on 29 November 2023).
- Roduit, C.; Frei, R.; Depner, M.; Schaub, B.; Loss, G.; Genuneit, J.; Pfefferle, P.; Hyvärinen, A.; Karvonen, A.M.; Riedler, J.; et al. Increased food diversity in the first year of life is inversely associated with allergic diseases. J. Allergy Clin. Immunol. 2014, 133, 1056–1064.e7. [Google Scholar] [CrossRef]
- Nwaru, B.I.; Takkinen, H.; Niemelä, O.; Kaila, M.; Erkkola, M.; Ahonen, S.; Tuomi, H.; Haapala, A.; Kenward, M.G.; Pekkanen, J.; et al. Introduction of complementary foods in infancy and atopic sensitization at the age of 5 years: Timing and food diversity in a Finnish birth cohort. Allergy 2013, 68, 507–516. [Google Scholar] [CrossRef]
- Maslin, K.; Pickett, K.; Ngo, S.; Anderson, W.; Dean, T.; Venter, C. Dietary diversity during infancy and the association with childhood food allergen sensitization. Pediatr. Allergy Immunol. 2022, 33, e13650. [Google Scholar] [CrossRef]
- Venter, C.; Maslin, K.; Holloway, J.W.; Silveira, L.J.; Fleischer, D.M.; Dean, T.; Arshad, S.H. Different Measures of Diet Diversity during Infancy and the Association with Childhood Food Allergy in a UK Birth Cohort Study. J. Allergy Clin. Immunol. Pract. 2020, 8, 2017–2026. [Google Scholar] [CrossRef] [PubMed]
- Zhong, C.; Guo, J.; Tan, T.; Wang, H.; Lin, L.; Gao, D.; Li, Q.; Sun, G.; Xiong, G.; Yang, X.; et al. Increased food diversity in the first year of life is inversely associated with allergic outcomes in the second year. Pediatr. Allergy Immunol. 2022, 33, e13707. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.R.; Jung, H.-I.; Kim, S.K.; Kwon, M.; Kim, H.; Jung, M.; Kyung, Y.; Kim, B.E.; Choi, S.-J.; Oh, S.-Y.; et al. Dietary Diversity during Early Infancy Increases Microbial Diversity and Prevents Egg Allergy in High-Risk Infants. Immune Netw. 2022, 22, e17. [Google Scholar] [CrossRef] [PubMed]
- Altmann, T.R.; Hill, D.L. Caring for Your Baby and Young Child: Birth to Age 5; American Academy of Pediatrics: Itasca, IL, USA, 2019. [Google Scholar]
- D’auria, E.; Pediatrics, O.B.O.T.I.S.O.; Bergamini, M.; Staiano, A.; Banderali, G.; Pendezza, E.; Penagini, F.; Zuccotti, G.V.; Peroni, D.G. Baby-led weaning: What a systematic review of the literature adds on. Ital. J. Pediatr. 2018, 44, 1–11. [Google Scholar] [CrossRef]
- Petrus, R.R.; Sobral, P.J.D.A.; Tadini, C.C.; Gonçalves, C.B. The NOVA classification system: A critical perspective in food science. Trends Food Sci. Technol. 2021, 116, 603–608. [Google Scholar] [CrossRef]
- Katidi, A.; Xanthopoulou, S.; Vlassopoulos, A.; Noutsos, S.; Priftis, K.; Kapsokefalou, M. Food Allergens in Ultra-Processed Foods According to the NOVA Classification System: A Greek Branded Food Level Analysis. Nutrients 2023, 15, 2767. [Google Scholar] [CrossRef]
- Melo, B.; Rezende, L.; Machado, P.; Gouveia, N.; Levy, R. Associations of ultra-processed food and drink products with asthma and wheezing among Brazilian adolescents. Pediatr. Allergy Immunol. 2018, 29, 504–511. [Google Scholar] [CrossRef]
- Knight, T.; Smith, P.K.; Soutter, V.; Oswald, E.; Venter, C. Is the low pH of infant and toddler foods a concern? Pediatr. Allergy Immunol. 2021, 32, 1103–1106. [Google Scholar] [CrossRef]
- Kulis, M.D.; Smeekens, J.M.; Immormino, R.M.; Moran, T.P. The airway as a route of sensitization to peanut: An update to the dual allergen exposure hypothesis. J. Allergy Clin. Immunol. 2021, 148, 689–693. [Google Scholar] [CrossRef]
- Haahtela, T. A biodiversity hypothesis. Allergy 2019, 74, 1445–1456. [Google Scholar] [CrossRef]
- Ierodiakonou, D.; Garcia-Larsen, V.; Logan, A.; Groome, A.; Cunha, S.; Chivinge, J.; Robinson, Z.; Geoghegan, N.; Jarrold, K.; Reeves, T.; et al. Timing of allergenic food introduction to the infant diet and risk of allergic or autoimmune disease: A systematic review and meta-analysis. JAMA 2016, 316, 1181–1192. [Google Scholar] [CrossRef]
- Bolton, E.; Ferguson, A.; Simpson, B.; Al-Naqeeb, J.; Simpson, E. 420 The effects of bathing, showering and petrolatum on skin barrier function. J. Investig. Dermatol. 2017, 137, S73. [Google Scholar] [CrossRef][Green Version]
- Sampath, V.; Abrams, E.M.; Adlou, B.; Akdis, C.; Akdis, M.; Brough, H.A.; Chan, S.; Chatchatee, P.; Chinthrajah, R.S.; Cocco, R.R.; et al. Food allergy across the globe. J. Allergy Clin. Immunol. 2021, 148, 1347–1364. [Google Scholar] [CrossRef]
- US Department of Agriculture and US Department of Health and Human Services. Dietary Guidelines for Americans, 2020–2025, 9th ed.; US Government Publishing Office: Washington, DC, USA, 2020. Available online: https://www.dietaryguidelines.gov/resources/2020-2025-dietary-guidelines-online-materials (accessed on 29 November 2023).

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brandwein, M.; Enten Vissoker, R.; Jackson, H.; Rogan, T.; Pitcock, J.; Krinkin, E.; Venter, C. Redefining the Role of Nutrition in Infant Food Allergy Prevention: A Narrative Review. Nutrients 2024, 16, 838. https://doi.org/10.3390/nu16060838
Brandwein M, Enten Vissoker R, Jackson H, Rogan T, Pitcock J, Krinkin E, Venter C. Redefining the Role of Nutrition in Infant Food Allergy Prevention: A Narrative Review. Nutrients. 2024; 16(6):838. https://doi.org/10.3390/nu16060838
Chicago/Turabian StyleBrandwein, Michael, Roni Enten Vissoker, Helen Jackson, Tavierney Rogan, Jana Pitcock, Esther Krinkin, and Carina Venter. 2024. "Redefining the Role of Nutrition in Infant Food Allergy Prevention: A Narrative Review" Nutrients 16, no. 6: 838. https://doi.org/10.3390/nu16060838
APA StyleBrandwein, M., Enten Vissoker, R., Jackson, H., Rogan, T., Pitcock, J., Krinkin, E., & Venter, C. (2024). Redefining the Role of Nutrition in Infant Food Allergy Prevention: A Narrative Review. Nutrients, 16(6), 838. https://doi.org/10.3390/nu16060838





