Adiposity Phenotypes and Associated Cardiometabolic Risk Profile in the Inuit Population of Nunavik
Abstract
1. Introduction
2. Methodology
2.1. Setting
2.2. Study Population
2.3. Clinical Assessment, Medical Review and Questionnaires
2.4. Statistical Analysis
3. Results
4. Discussion
Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Batal, M.; Decelles, S. A Scoping Review of Obesity among Indigenous Peoples in Canada. J. Obes. 2019, 2019, 9741090. [Google Scholar] [CrossRef]
- Kellett, S.; Poirier, P.; Dewailly, E.; Sampasa, H.; Chateau-Degat, M.L. Is severe obesity a cardiovascular health concern in the Inuit population? Am. J. Hum. Biol. 2012, 24, 441–445. [Google Scholar] [CrossRef]
- Garnier, R.; Carrière, G.; Sanmartin, C. The Health of First Nations Living Off-Reserve, Inuit, and Métis Adults in Canada: The Impact of Socio-Economic Status on Inequalities in Health; Health Research Working Paper Series; Statistics Canada: Ottawa, ON, Canada, 2010. [Google Scholar]
- Allaire, J.; Levesque, B.; Poirier, P.; Gagnon, C.; Auclair, G.; Lemire, M.; Ayotte, P. Prevalence and determinants of hypertension in the adult Inuit population of Nunavik (northern Quebec, Canada). Can. J. Public Health 2023, 115, 168–179. [Google Scholar] [CrossRef]
- Chateau-Degat, M.L.; Dewailly, E.; Louchini, R.; Counil, E.; Noel, M.; Ferland, A.; Lucas, M.; Valera, B.; Ekoé, J.M.; Ladouceur, R.; et al. Cardiovascular burden and related risk factors among Nunavik (Quebec) Inuit: Insights from baseline findings in the circumpolar Inuit Health in Transition cohort study. Can. J. Cardiol. 2010, 26, 190–196. [Google Scholar] [CrossRef]
- Chateau-Degat, M.L.; Dannenbaum, D.A.; Egeland, G.M.; Nieboer, E.; Laouan Sidi, E.A.; Abdous, B.; Dewailly, E. A comparison of the metabolic response to abdominal obesity in two Canadian Inuit and First Nations population. Obesity 2011, 19, 2254–2260. [Google Scholar] [CrossRef]
- Young, T.K. Obesity, central fat patterning, and their metabolic correlates among the inuit of the central Canadian Arctic. Hum. Biol. 1996, 68, 245–263. [Google Scholar]
- Piche, M.E.; Tchernof, A.; Despres, J.P. Obesity Phenotypes, Diabetes, and Cardiovascular Diseases. Circ. Res. 2020, 126, 1477–1500. [Google Scholar] [CrossRef]
- Tchernof, A.; Despres, J.P. Pathophysiology of human visceral obesity: An update. Physiol. Rev. 2013, 93, 359–404. [Google Scholar] [CrossRef] [PubMed]
- Prince, S.A.; McDonnell, L.A.; Turek, M.A.; Visintini, S.; Nahwegahbow, A.; Kandasamy, S.; Sun, L.Y.; Coutinho, T. The State of Affairs for Cardiovascular Health Research in Indigenous Women in Canada: A Scoping Review. Can. J. Cardiol. 2018, 34, 437–449. [Google Scholar] [CrossRef] [PubMed]
- Hedayat, L.M.A.; Murchison, C.C.; Foulds, H.J.A. A systematic review and meta-analysis of cardiorespiratory fitness among Indigenous populations in North America and circumpolar Inuit populations. Prev. Med. 2018, 109, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Wharton, S.; Lau, D.C.W.; Vallis, M.; Sharma, A.M.; Biertho, L.; Campbell-Scherer, D.; Adamo, K.; Alberga, A.; Bell, R.; Boule, N.; et al. Obesity in adults: A clinical practice guideline. CMAJ 2020, 192, E875–E891. [Google Scholar] [CrossRef] [PubMed]
- Wharton, S.; Pedersen, S.D.; Lau, D.C.; Sharma, A.M.; Diabetes Canada Clinical Practice Guidelines Expert Committee. Weight Management in Diabetes. Can. J. Diabetes 2018, 42, S124–S129. [Google Scholar] [CrossRef] [PubMed]
- Després, J.P.; Carpentier, A.C.; Tchernof, A.; Neeland, I.J.; Poirier, P. Management of Obesity in Cardiovascular Practice: JACC Focus Seminar. J. Am. Coll. Cardiol. 2021, 78, 513–531. [Google Scholar] [CrossRef] [PubMed]
- Aziz, H.; Marchand, M.; Pop, C.; King, A.; Anand, S.S.; Arbour, L.; Atzema, C.; Spaziano, M.; Merveille, N.; Filimon, S.; et al. A Call to Action: Optimizing Indigenous Cardiovascular Health in Canada. Can. J. Cardiol. 2022, 38, 1584–1587. [Google Scholar] [CrossRef] [PubMed]
- Hamel, D.; Hamel, G.; Gagnon, S. Qanuilirpitaa? 2017 Nunavik Inuit Health Survey: Methodological Report. Nunavik Regional Board of Health and Social Services & Institut National de Santé Publique du Québec (INSPQ). Available online: http://nrbhss.ca/en/health-surveys (accessed on 4 January 2024).
- Faria, S.L.; Faria Op Fau-Cardeal, M.D.A.; Cardeal Md Fau-Ito, M.K.; Ito, M.K. Validation study of multi-frequency bioelectrical impedance with dual-energy X-ray absorptiometry among obese patients. Obes. Surg. 2014, 24, 1476–1480. [Google Scholar] [CrossRef] [PubMed]
- National Heart, Lung and Blood Institute. Managing Overweight and Obesity in Adults: Systematic Evidence Review from the Obesity Expert Panel, 2013. US Department of Health and Human Services: National Institutes of Health. 2013. Available online: https://www.nhlbi.nih.gov/health-topics/managing-overweight-obesity-in-adults (accessed on 4 January 2024).
- Utter, A.C.; Nieman, D.C.; Ward, A.N.; Butterworth, D.E. Use of the leg-to-leg bioelectrical impedance method in assessing body-composition change in obese women. Am. J. Clin. Nutr. 1999, 69, 603–607. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Liu, Y.A.-O.; Yan, C.; Yang, R.; Xu, L.; Guo, Z.; Yu, A.; Cheng, X.A.-O.; Ma, L.; Hu, C.; et al. Measurement of visceral fat and abdominal obesity by single-frequency bioelectrical impedance and CT: A cross-sectional study. BJM Open 2021, 11, e048221. [Google Scholar] [CrossRef]
- Schorr, M.; Dichtel, L.E.; Gerweck, A.V.; Valera, R.D.; Torriani, M.; Miller, K.K.; Bredella, M.A. Sex differences in body composition and association with cardiometabolic risk. Biol. Sex. Differ 2018, 9, 28. [Google Scholar] [CrossRef]
- Lear, S.A.; James, P.T.; Ko, G.T.; Kumanyika, S. Appropriateness of waist circumference and waist-to-hip ratio cutoffs for different ethnic groups. Eur. J. Clin. Nutr. 2010, 64, 42–61. [Google Scholar] [CrossRef]
- Chooi, Y.C.; Ding, C.; Magkos, F. The epidemiology of obesity. Metabolism 2019, 92, 6–10. [Google Scholar] [CrossRef]
- Whelton, P.K.; Carey, R.M.; Aronow, W.S.; Casey, D.E.; Collins, K.J.; Dennison Himmelfarb, C.; DePalma, S.M.; Gidding, S.; Jamerson, K.A.; Jones, D.W.; et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: Executive Summary: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 2018, 71, e127–e248. [Google Scholar] [PubMed]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Manjoo, P.; Dannenbaum, D.; Joseph, L.; Torrie, J.; Dasgupta, K. Utility of current obesity thresholds in signaling diabetes risk in the James Bay Cree of Eeyou Istchee. BMJ Open Diabetes Res. Care 2015, 3, e000114. [Google Scholar] [CrossRef] [PubMed]
- Punthakee, Z.; Goldenberg, R.; Katz, P.; Diabetes Canada Clinical Practice Guidelines Expert Committee. Definition, Classification and Diagnosis of Diabetes, Prediabetes and Metabolic Syndrome. Can. J. Diabetes 2018, 42, S10–S15. [Google Scholar] [CrossRef] [PubMed]
- Pearson, G.J.; Thanassoulis, G.; Anderson, T.J.; Barry, A.R.; Couture, P.; Dayan, N.; Francis, G.A.; Genest, J.; Grégoire, J.; Grover, S.A.; et al. 2021 Canadian Cardiovascular Society Guidelines for the Management of Dyslipidemia for the Prevention of Cardiovascular Disease in Adults. Can. J. Cardiol. 2021, 37, 1129–1150. [Google Scholar] [CrossRef] [PubMed]
- Stevens, P.E.; Levin, A.; Kidney Disease: Improving Global Outcomes Chronic Kidney Disease Guideline Development Work Group Members. Evaluation and management of chronic kidney disease: Synopsis of the kidney disease: Improving global outcomes 2012 clinical practice guideline. Ann. Intern. Med. 2013, 158, 825–830. [Google Scholar] [CrossRef] [PubMed]
- Bedogni, G.; Bellentani, S.; Miglioli, L.; Masutti, F.; Passalacqua, M.; Castiglione, A.; Tiribelli, C. The Fatty Liver Index: A simple and accurate predictor of hepatic steatosis in the general population. BMC Gastroenterol. 2006, 6, 33. [Google Scholar] [CrossRef]
- Sterling, R.K.; Lissen, E.; Clumeck, N.; Sola, R.; Correa, M.C.; Montaner, J.; Sulkowski, M.S.; Torriani, F.J.; Dieterich, D.T.; Thomas, D.L.; et al. Development of a simple noninvasive index to predict significant fibrosis in patients with HIV/HCV coinfection. Hepatology 2006, 43, 1317–1325. [Google Scholar] [CrossRef]
- Shah, A.G.; Lydecker, A.; Murray, K.; Tetri, B.N.; Contos, M.J.; Sanyal, A.J.; Nash Clinical Research Network. Comparison of noninvasive markers of fibrosis in patients with nonalcoholic fatty liver disease. Clin. Gastroenterol. Hepatol. 2009, 7, 1104–1112. [Google Scholar] [CrossRef]
- Muhammad, A.G.; Hansen, F.O.; Gantzel, R.H.; Rex, K.F.; Villadsen, G.E.; Gronbaek, H.; Pedersen, M.L. Non-alcoholic fatty liver disease in patients with type 2 diabetes in Greenland: A register-based cross-sectional study. Int. J. Circumpolar Health 2022, 81, 2065755. [Google Scholar] [CrossRef]
- DeLong, E.R.; DeLong, D.M.; Clarke-Pearson, D.L. Comparing the areas under two or more correlated receiver operating characteristic curves: A nonparametric approach. Biometrics 1988, 44, 837–845. [Google Scholar] [CrossRef]
- Hughes, G. Youden’s index and the weight of evidence. Methods Inf. Med. 2015, 54, 198–199. [Google Scholar] [CrossRef]
- Riediger, N.; Ian, C. Prevalence of metabolic syndrome in the Canadian adult population. CMAJ 2011, 183, E1127–E1133. [Google Scholar] [CrossRef]
- Erem, C.; Hacıhasanoglu, A.; Deger, O.; Topbaş, M.; Hosver, I.; Ersoz, H.O.; Can, G. Prevalence of metabolic syndrome and associated risk factors among Turkish adults: Trabzon MetS study. Endocrine 2008, 33, 9–20. [Google Scholar] [CrossRef]
- Esteghamati, A.; Meysamie, A.; Khalilzadeh, O.; Rashidi, A.; Haghazali, M.; Asgari, F.; Kamgar, M.; Gouya, M.M.; Abbasi, M. Third national Surveillance of Risk Factors of Non-Communicable Diseases (SuRFNCD-2007) in Iran: Methods and results on prevalence of diabetes, hypertension, obesity, central obesity, and dyslipidemia. BMC Public Health 2009, 9, 167. [Google Scholar] [CrossRef]
- Rowe, A.; Hernandez, P.; Kuhle, S.; Kirkland, S. The association between anthropometric measures and lung function in a population-based study of Canadian adults. Respir. Med. 2017, 131, 199–204. [Google Scholar] [CrossRef]
- Statistics Canada. Overweight and Obese Adults, 2018. Health Fact Sheet 2019, 82, 65. [Google Scholar]
- Ronn, P.F.; Andersen, G.S.; Lauritzen, T.; Christensen, D.L.; Aadahl, M.; Carstensen, B.; Grarup, N.; Jorgensen, M.E. Abdominal visceral and subcutaneous adipose tissue and associations with cardiometabolic risk in Inuit, Africans and Europeans: A cross-sectional study. BMJ Open 2020, 10, e038071. [Google Scholar] [CrossRef]
- Ronn, P.F.; Andersen, G.S.; Lauritzen, T.; Christensen, D.L.; Aadahl, M.; Carstensen, B.; Jorgensen, M.E. Ethnic differences in anthropometric measures and abdominal fat distribution: A cross-sectional pooled study in Inuit, Africans and Europeans. J. Epidemiol. Community Health 2017, 71, 536–543. [Google Scholar] [CrossRef]
- Lajeunesse-Trempe, F.; Dufour, R.; du Souich, P.; Paquette, M.; Kaduka, L.U.; Christensen, D.L. Anthropometric measures and their association with risk factors for cardio-metabolic diseases in Kenyan adults. Ann. Hum. Biol. 2018, 45, 486–495. [Google Scholar] [CrossRef]
- Statistics Canada. Niveaux de cholesterol chez les adultes 2012 à 2013, 2018. Health Fact Sheet 2014, 82, 65. [Google Scholar]
- Statistics Canada. Diabetes in Canada: Facts and Figures from a Public Health Perspective. Available online: https://www.canada.ca/en/public-health/services/chronic-diseases/reports-publications/diabetes/diabetes-canada-facts-figures-a-public-health-perspective.html (accessed on 7 January 2024).
- Government of Canada. Heart Disease in Canada: Highlights from the Canadian Chronic Disease Surveillance System. Available online: https://www.canada.ca/en/public-health/services/publications/diseases-conditions/heart-disease-canada-fact-sheet.html (accessed on 6 January 2024).
- Swain, M.G.; Ramji, A.; Patel, K.; Sebastiani, G.; Shaheen, A.A.; Tam, E.; Marotta, P.; Elkhashab, M.; Bajaj, H.S.; Estes, C.; et al. Burden of nonalcoholic fatty liver disease in Canada, 2019–2030: A modelling study. CMAJ Open 2020, 8, E429–E436. [Google Scholar] [CrossRef]
- Nazare, J.A.; Smith, J.D.; Borel, A.L.; Haffner, S.M.; Balkau, B.; Ross, R.; Massien, C.; Almeras, N.; Despres, J.P. Ethnic influences on the relations between abdominal subcutaneous and visceral adiposity, liver fat, and cardiometabolic risk profile: The International Study of Prediction of Intra-Abdominal Adiposity and Its Relationship With Cardiometabolic Risk/Intra-Abdominal Adiposity. Am. J. Clin. Nutr. 2012, 96, 714–726. [Google Scholar] [CrossRef]
- Guerrero, R.; Vega, G.L.; Grundy, S.M.; Browning, J.D. Ethnic differences in hepatic steatosis: An insulin resistance paradox? Hepatology 2009, 49, 791–801. [Google Scholar] [CrossRef]
- Labonté, M.È.; Dewailly, É.; Lucas, M.; Chateau-Degat, M.L.; Couture, P.; Lamarche, B. Traditional dietary pattern is associated with elevated cholesterol among the Inuit of Nunavik. J. Acad. Nutr. Diet. 2014, 114, 1208–1215.e3. [Google Scholar] [CrossRef]
- Lewis, J.A.-O.X.; Lind, M.V.; Møller, G.; Hansen, T.; Pedersen, H.; Christensen, M.M.B.; Laursen, J.C.; Nielsen, S.; Ottendahl, C.B.; Larsen, C.V.L.; et al. The effect of traditional diet on glucose homoeostasis in carriers and non-carriers of a common TBC1D4 variant in Greenlandic Inuit: A randomised crossover study. Br. J. Nutr. 2023, 130, 1871–1884. [Google Scholar] [CrossRef]
- Marushka, L.; Batal, M.; David, W.; Schwartz, H.; Ing, A.; Fediuk, K.; Sharp, D.; Black, A.; Tikhonov, C.; Chan, H.M. Association between fish consumption, dietary omega-3 fatty acids and persistent organic pollutants intake, and type 2 diabetes in 18 First Nations in Ontario, Canada. Environ. Res. 2017, 156, 725–737. [Google Scholar] [CrossRef]
- Girard, C.A.-O.; Tromas, N.; Amyot, M.; Shapiro, B.A.-O. Gut Microbiome of the Canadian Arctic Inuit. Microorganisms 2023, 11, 625. [Google Scholar] [CrossRef]
- Singh, K.; Chan, H.M. Persistent organic pollutants and diabetes among Inuit in the Canadian Arctic. Environ. Int. 2017, 101, 183–189. [Google Scholar] [CrossRef]
- Manousaki, D.; Kent, J.W., Jr.; Haack, K.; Zhou, S.; Xie, P.; Greenwood, C.M.; Brassard, P.; Newman, D.E.; Cole, S.; Umans, J.G.; et al. Toward Precision Medicine: TBC1D4 Disruption Is Common Among the Inuit and Leads to Underdiagnosis of Type 2 Diabetes. Diabetes Care 2016, 39, 1889–1895. [Google Scholar] [CrossRef]
- Galloway, T.; Chateau-Degat, M.L.; Egeland, G.M.; Young, T.K. Does sitting height ratio affect estimates of obesity prevalence among Canadian Inuit? Results from the 2007–2008 Inuit Health Survey. Am. J. Hum. Biol. 2011, 23, 655–663. [Google Scholar] [CrossRef] [PubMed]
- Charbonneau-Roberts, G.; Saudny-Unterberger, H.; Kuhnlein, H.V.; Egeland, G.M. Body mass index may overestimate the prevalence of overweight and obesity among the Inuit. Int. J. Circumpolar Health 2005, 64, 163–169. [Google Scholar] [CrossRef] [PubMed]
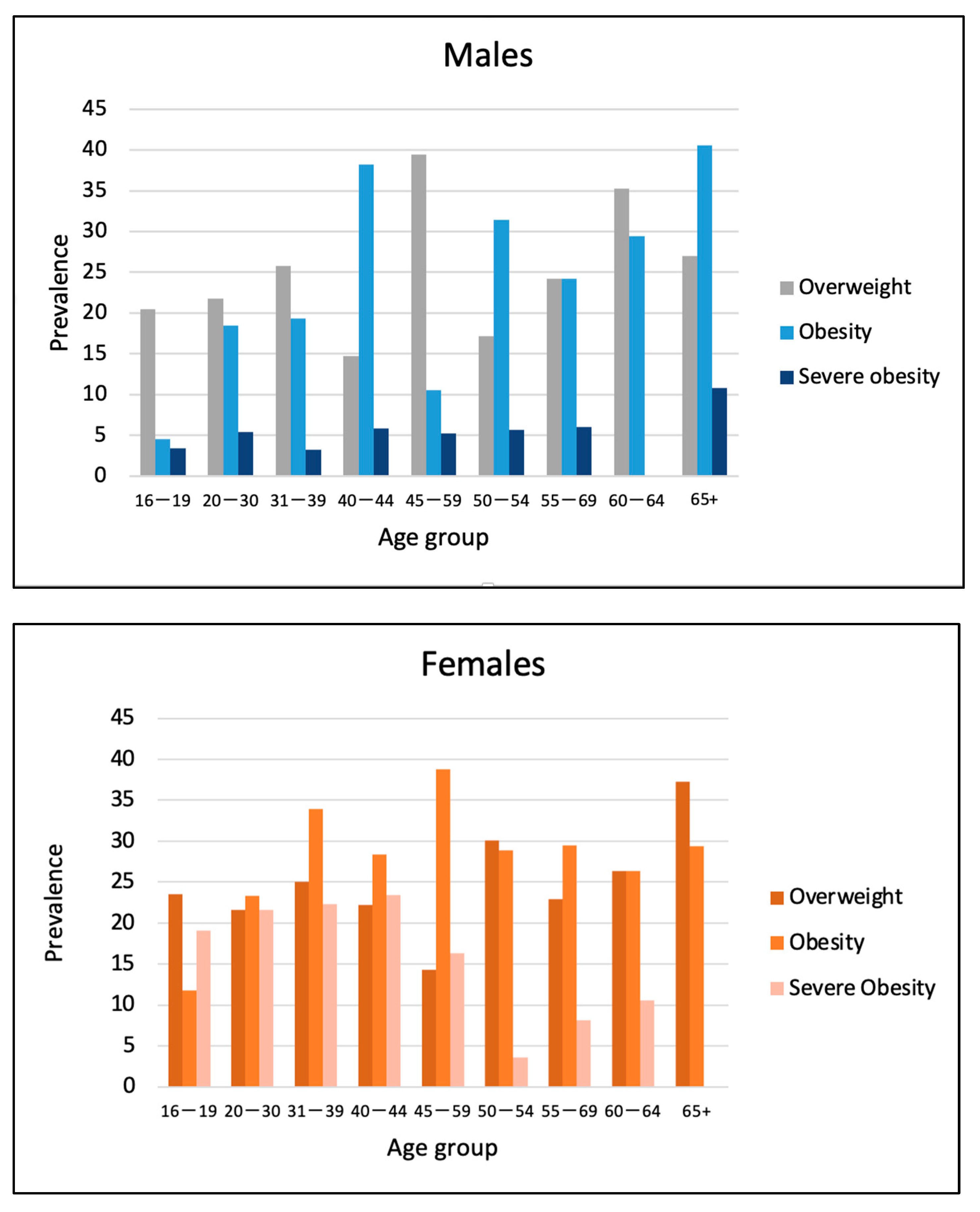
| Male | Female | p-Value | ||||
|---|---|---|---|---|---|---|
| N | 453 | 843 | ||||
| Age | n (%) | n | % | n | % | |
| 16–19 years | 88 | 30.0 | 136 | 26.6 | 0.2 | |
| 20–30 years | 92 | 20.3 | 232 | 27.5 | <0.05 | |
| 31–39 years | 62 | 13.7 | 112 | 13.3 | 0.9 | |
| 40–44 years | 34 | 7.5 | 81 | 9.5 | 0.2 | |
| 45–49 years | 38 | 8.4 | 49 | 5.8 | 0.1 | |
| 50–54 years | 35 | 7.7 | 83 | 9.7 | 0.3 | |
| 55–59 years | 33 | 7.3 | 61 | 7.5 | 0.9 | |
| 60–65 years | 34 | 7.5 | 38 | 4.5 | <0.05 | |
| 65+ years | 37 | 8.2 | 51 | 6.0 | 0.2 | |
| Weight | kg | 73.8 | 17.9 | 67.6 | 16.9 | <0.05 |
| Height | cm | 166.9 | 6.6 | 155.3 | 6.1 | <0.05 |
| BMI | kg/m2 | 26.31 | 5.7 | 28.0 | 6.4 | <0.05 |
| Waist circumference | cm | 91.9 | 16.6 | 94.0 | 16.1 | <0.05 |
| Waist to Height ratio | 0.55 | 0.10 | 0.61 | 0.10 | <0.05 | |
| Body Fat Mass | kg | 18.15 | 12.2 | 26.06 | 12.35 | <0.05 |
| Percentage of body fat | % | 22.73 | 10.0 | 36.76 | 9.56 | <0.05 |
| Skeletal Muscle Mass | 31.05 | 4.8 | 22.4 | 3.5 | <0.05 | |
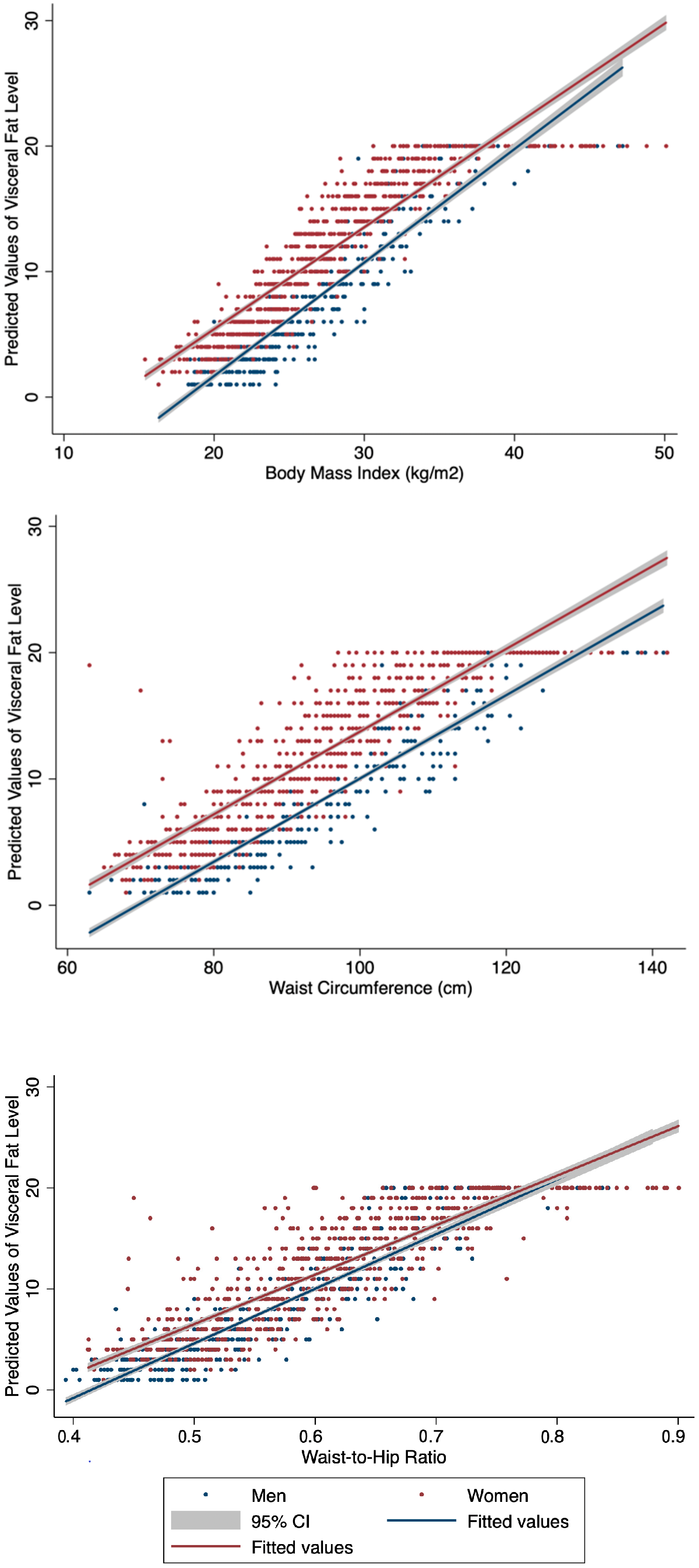
| Visceral fat level * | 5 | 3–11 | 12 | 6–17 | <0.05 | |
| High visceral fat level 1 | 91 | 20.09 | 459 | 54.44 | <0.05 | |
| Basal metabolic rate | kcal/day | 1566.43 | 170.03 | 1260.29 | 125.44 | <0.05 |
| Blood pressure | ||||||
| Systolic | mmHg | 128 | 13 | 120 | 14 | <0.05 |
| Diastolic | mmHg | 80 | 10 | 75 | 9 | <0.05 |
| Smoking status | n (%) No | 113 | 25.0 | 169 | 20.0 | 0.08 |
| Occasional | 41 | 9.1 | 70 | 8.3 | <0.05 | |
| Daily | 292 | 64.5 | 592 | 70.2 | <0.05 | |
| Alcohol consumption | n (%) | |||||
| Daily | 19 | 4.2 | 39 | 4.6 | <0.05 | |
| 3 to 6 per week | 50 | 11.0 | 119 | 14.1 | <0.05 | |
| 1–2 per week | 96 | 21.1 | 169 | 20.0 | <0.05 | |
| 1–3 per month | 105 | 23.2 | 176 | 20.6 | <0.05 | |
| <1 a month | 104 | 22.9 | 142 | 16.6 | <0.05 | |
| Never | 55 | 12.1 | 140 | 16.4 | <0.05 | |
| Degree of adiposity | n (%) | |||||
| Overweight | 110 | 24.3 | 203 | 24.1 | <0.05 | |
| Obesity | 94 | 20.8 | 217 | 25.7 | <0.05 | |
| Severe Obesity | 22 | 4.9 | 144 | 17.1 | <0.05 | |
| Abdominal obesity | n (%) | 210 | 46.4 | 664 | 78.8 | <0.05 |
| Dyslipidemia | n (%) | 164 | 36.2 | 249 | 29.5 | <0.05 |
| Insulin resistance | n (%) | 134 | 55.4 | 709 | 67.3 | 0.9 |
| Pre-diabetes | n (%) | 41 | 9.1 | 84 | 10.0 | <0.05 |
| Type 2 diabetes | n (%) | 27 | 6.0 | 62 | 7.3 | <0.05 |
| Chronic renal disease (EGFR ≤ 90 mL/min) | n (%) | 119 | 26.3 | 145 | 17.2 | <0.05 |
| Hypertension | n (%) | 117 | 25.8 | 126 | 14.9 | <0.05 |
| Non-alcoholic Fatty Liver Disease 2 | n (%) | 14 | 3.1 | 20 | 2.4 | 0.6 |
| Advanced stage of liver fibrosis 3 | n (%) | 1 | 0.2 | 1 | 0.1 | 0.7 |
| Male | Female | p-Value | ||||
|---|---|---|---|---|---|---|
| N | 453 | 843 | ||||
| White blood count | WBC per microliter | 7.70 | 2.13 | 7.53 | 2.16 | 0.16 |
| Hemoglobin | nmol/L | 142.55 | 12.13 | 128.37 | 11.11 | <0.05 |
| Platelets | Platelets per microliter | 305.28 | 66.82 | 346.85 | 69.26 | <0.05 |
| Creatinine | mg/dL | 83.02 | 15.42 | 61.24 | 11.97 | <0.05 |
| 24 H micro-albuminuria | mg/24 h | 16.41 | 35.91 | 24.41 | 86.58 | 0.06 |
| EGFR | 101.38 | 19.97 | 107.59 | 19.18 | <0.05 | |
| GGT | U/L | 33.51 | 37.77 | 28.93 | 31.67 | <0.05 |
| ALT | U/L | 12.71 | 10.19 | 11.66 | 12.27 | 0.1 |
| AST | U/L | 24.35 | 44.22 | 20.25 | 15.81 | <0.05 |
| ALB | g/L | 44.35 | 3.00 | 43.64 | 2.96 | <0.05 |
| Bilirubin | μmol/L | 5.60 | 3.25 | 4.17 | 2.16 | <0.05 |
| Apolipoprotein B | mg/dL | 0.89 | 0.27 | 0.89 | 0.25 | 0.6 |
| TG | mmol/L | 1.62 | 1.78 | 1.47 | 0.98 | 0.06 |
| LDL-Chol | mmol/L | 2.58 | 0.93 | 2.64 | 0.83 | 0.2 |
| TOTAL Chol | mmol/L | 4.73 | 1.09 | 4.97 | 1.06 | <0.05 |
| HDL-Chol | mmol/L | 1.43 | 0.42 | 1.63 | 0.50 | <0.05 |
| Total Chol/HDL Ratio | 3.58 | 1.62 | 3.24 | 1.06 | <0.05 | |
| HbA1C | % | 5.56 | 0.60 | 5.47 | 0.74 | <0.05 |
| Random glucose | mmol/L | 5.70 | 1.32 | 5.75 | 1.83 | 0.6 |
| Fasting glucose | N | 206 | 329 | |||
| mmol/L | 5.50 | 0.76 | 5.36 | 0.70 | <0.05 | |
| Fasting insulin 1 | N | 213 | 336 | |||
| mmol/L | 48.25 | 30.37–79.71 | 60.53 | 41.56–92.98 | 0.2 | |
| HOMA-IR 1 | N | 206 | 329 | |||
| 2.83 | 2.94 | 3.06 | 2.77 | 0.4 |
| Weight | BMI | WC | WHtR | VFL | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AUROC | 95% IC | AUROC | 95% IC | AUROC | 95% IC | AUROC | 95% IC | AUROC | 95% IC | ||||||
| Male | |||||||||||||||
| Diabetes | 0.75 | 0.63 | 0.86 | 0.78 | 0.70 | 0.87 | 0.77 | 0.66 | 0.88 | 0.79 | 0.67 | 0.89 | 0.77 | 0.66 | 0.87 |
| Prediabetes | 0.71 | 0.62 | 0.80 | 0.74 | 0.66 | 0.82 | 0.75 | 0.66 | 0.84 | 0.76 | 0.68 | 0.85 | 0.74 | 0.65 | 0.83 |
| Insulin resistance | 0.75 | 0.70 | 0.79 | 0.73 | 0.68 | 0.78 | 0.73 | 0.68 | 0.78 | 0.70 | 0.65 | 0.75 | 0.71 | 0.66 | 0.76 |
| Hypertension | 0.70 | 0.65 | 0.75 | 0.73 | 0.69 | 0.78 | 0.75 | 0.71 | 0.80 | 0.76 | 0.72 | 0.80 | 0.74 | 0.70 | 0.79 |
| Dyslipidemia | 0.71 | 0.66 | 0.76 | 0.73 | 0.68 | 0.79 | 0.75 | 0.70 | 0.79 | 0.75 | 0.70 | 0.79 | 0.75 | 0.70 | 0.80 |
| Female | |||||||||||||||
| Diabetes | 0.66 | 0.57 | 0.76 | 0.71 | 0.62 | 0.81 | 0.66 | 0.55 | 0.78 | 0.68 | 0.56 | 0.80 | 0.69 | 0.60 | 0.79 |
| Prediabetes | 0.64 | 0.57 | 0.70 | 0.70 | 0.64 | 0.76 | 0.68 | 0.62 | 0.75 | 0.71 | 0.65 | 0.78 | 0.69 | 0.63 | 0.76 |
| Insulin resistance | 0.71 | 0.67 | 0.76 | 0.72 | 0.68 | 0.77 | 0.72 | 0.67 | 0.76 | 0.71 | 0.66 | 0.76 | 0.70 | 0.66 | 0.75 |
| Hypertension | 0.64 | 0.59 | 0.68 | 0.65 | 0.61 | 0.70 | 0.66 | 0.62 | 0.70 | 0.66 | 0.61 | 0.70 | 0.66 | 0.62 | 0.70 |
| Dyslipidemia | 0.65 | 0.61 | 0.70 | 0.69 | 0.65 | 0.74 | 0.70 | 0.67 | 0.75 | 0.72 | 0.68 | 0.76 | 0.69 | 0.64 | 0.73 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lajeunesse-Trempe, F.; Piché, M.-E.; Poirier, P.; Tchernof, A.; Ayotte, P. Adiposity Phenotypes and Associated Cardiometabolic Risk Profile in the Inuit Population of Nunavik. Nutrients 2024, 16, 725. https://doi.org/10.3390/nu16050725
Lajeunesse-Trempe F, Piché M-E, Poirier P, Tchernof A, Ayotte P. Adiposity Phenotypes and Associated Cardiometabolic Risk Profile in the Inuit Population of Nunavik. Nutrients. 2024; 16(5):725. https://doi.org/10.3390/nu16050725
Chicago/Turabian StyleLajeunesse-Trempe, Fannie, Marie-Eve Piché, Paul Poirier, André Tchernof, and Pierre Ayotte. 2024. "Adiposity Phenotypes and Associated Cardiometabolic Risk Profile in the Inuit Population of Nunavik" Nutrients 16, no. 5: 725. https://doi.org/10.3390/nu16050725
APA StyleLajeunesse-Trempe, F., Piché, M.-E., Poirier, P., Tchernof, A., & Ayotte, P. (2024). Adiposity Phenotypes and Associated Cardiometabolic Risk Profile in the Inuit Population of Nunavik. Nutrients, 16(5), 725. https://doi.org/10.3390/nu16050725





