Valorization of Salicornia patula Duval-Jouve Young Shoots in Healthy and Sustainable Diets
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Moisture Determination
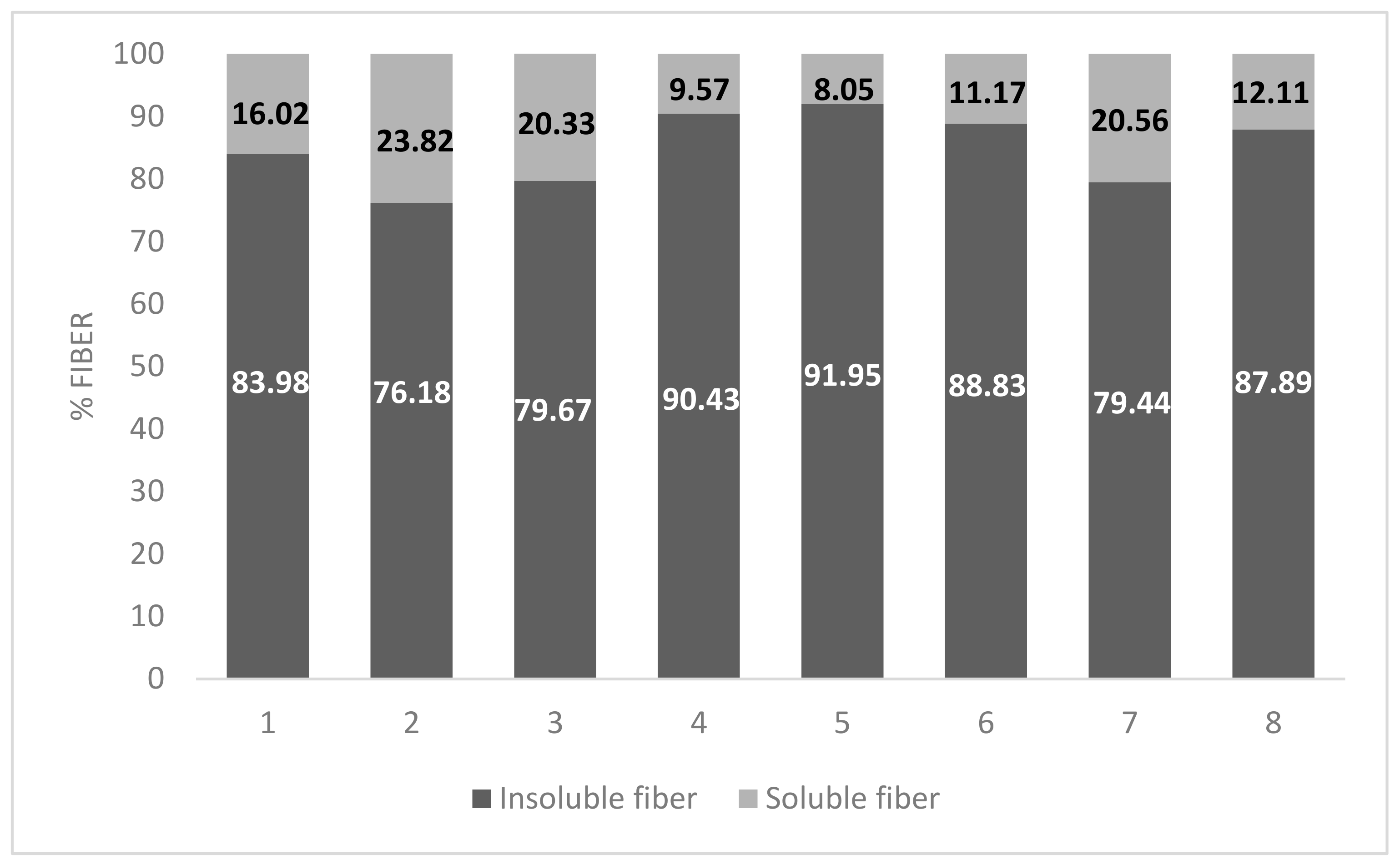
2.3. Fiber Determination
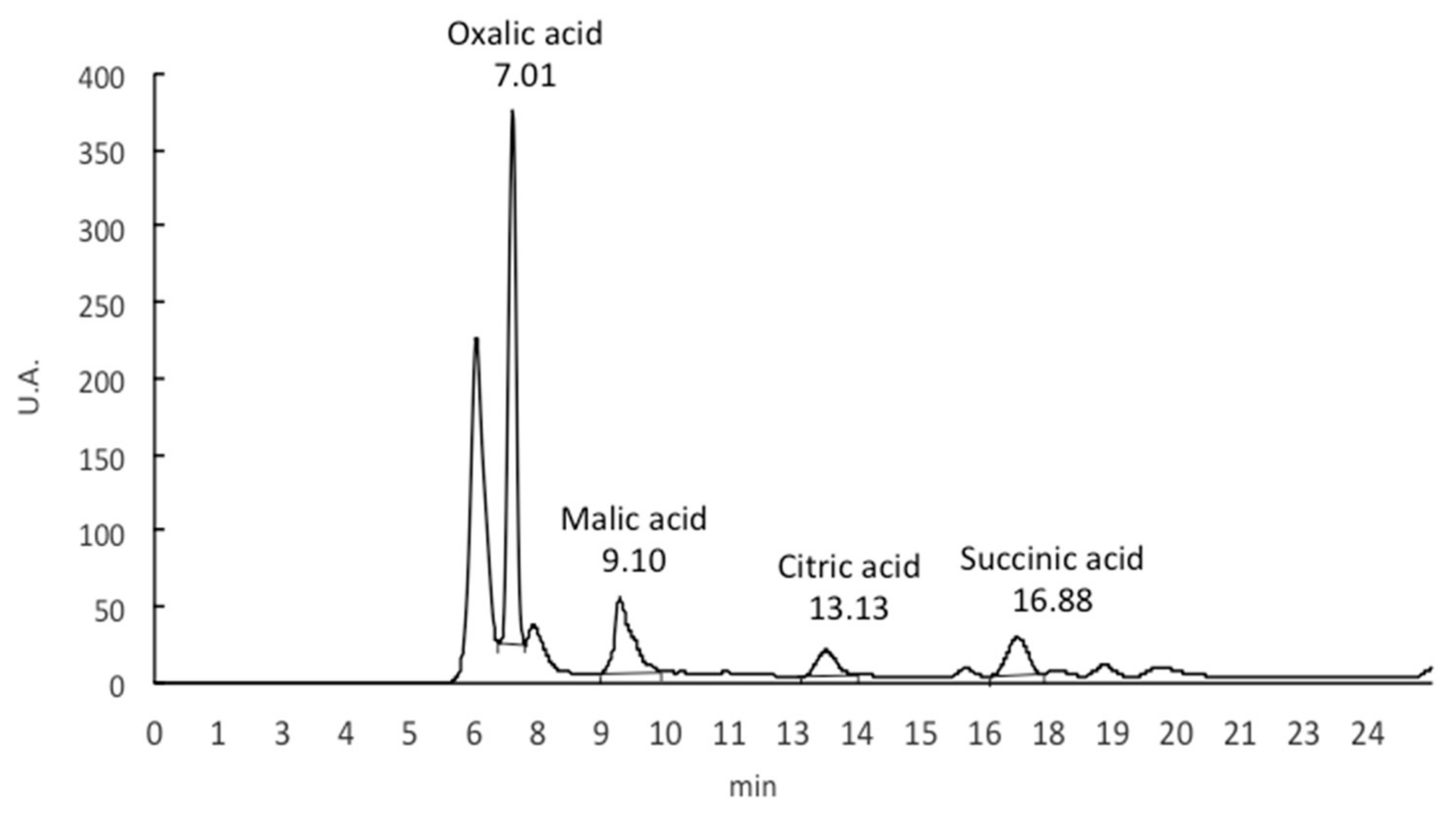
2.4. Determination of Organic Acids
2.5. Statistical Analysis
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yokoyama, Y.; Levin, S.M.; Barnard, N.D. Association between plant-based diets and plasma lipids: A systematic review and meta-analysis. Nutr. Rev. 2017, 75, 683–698. [Google Scholar] [CrossRef]
- Guasch-Ferré, M.; Willett, W.C. The Mediterranean diet and health: A comprehensive overview. J. Intern. Med. 2021, 290, 549–566. [Google Scholar] [CrossRef] [PubMed]
- López García, E.; Bretón Lesmes, I.; Díaz Perales, A.; Moreno-Arribas, V.; Portillo Baquedano, M.P.; Rivas Velasco, A.M.; Fresán Salvo, U.; Tejedor Romero, L.; Ortega Porcel, F.B.; Aznar Laín, S.; et al. Informe del Comité Científico de la Agencia Española de Seguridad Alimentaria y Nutrición (AESAN) sobre recomendaciones dietéticas sostenibles y recomendaciones de actividad física para la población española. Rev. Com. Cient. AESAN 2022, 36, 11–70. [Google Scholar]
- European Food Safety Authority (EFSA). Dietary reference values for nutrients: Summary report. EFSA Support. Publ. 2017, 92, e15121. [Google Scholar] [CrossRef]
- Marlett, J.A.; McBurney, M.I.; Slavin, J.L. Position of the American Dietetic Association: Health implications of dietary fiber. J. Am. Diet. Assoc. 2002, 102, 993–1000. [Google Scholar] [CrossRef] [PubMed]
- Cámara, M.; Fernández-Ruiz, V.; Morales, P.; Cortes Sánchez-Mata, M. Fiber compounds and human health. Curr. Pharm. Des. 2017, 23, 2835–2849. [Google Scholar] [CrossRef]
- García-Herrera, P.; Morales, P.; Cámara, M.; Fernández-Ruiz, V.; Tardío, J.; Sánchez-Mata, M.C. Nutritional and phytochemical composition of Mediterranean wild vegetables after culinary treatment. Foods 2020, 9, 1761. [Google Scholar] [CrossRef] [PubMed]
- Benedetti, F.; Caon, L. Global Soil Laboratory Assessment 2020—Laboratories’ Capacities and Needs; FAO: Rome, Italy, 2021; Available online: https://www.fao.org/3/cb6395en/cb6395en.pdf (accessed on 21 December 2023).
- Hasnain, M.; Abideen, Z.; Ali, F.; Hasanuzzaman, M.; El-Keblawy, A. Potential of Halophytes as Sustainable Fodder Production by Using Saline Resources: A Review of Current Knowledge and Future Directions. Plants 2023, 12, 2150. [Google Scholar] [CrossRef]
- Khondoker, M.; Mandal, S.; Gurav, R.; Hwang, S. Freshwater Shortage, Salinity Increase, and Global Food Production: A Need for Sustainable Irrigation Water Desalination—A Scoping Review. Earth 2023, 4, 223–240. [Google Scholar] [CrossRef]
- Lopes, M.; Silva, A.S.; Séndon, R.; Barbosa-Pereira, L.; Cavaleiro, C.; Ramos, F. Towards the Sustainable Exploitation of Salt-Tolerant Plants: Nutritional Characterisation, Phenolics Composition, and Potential Contaminants Analysis of Salicornia ramosissima and Sarcocornia perennis alpini. Molecules 2023, 28, 2726. [Google Scholar] [CrossRef]
- Flowers, T.J.; Colmer, T.D. Salinity tolerance in halophytes. New Phytol. 2008, 179, 945–963. [Google Scholar] [CrossRef] [PubMed]
- Vizetto-Duarte, C.; Figueiredo, F.; Rodrigues, M.J.; Polo, C.; Rešek, E.; Custódio, L. Sustainable valorization of halophytes from the mediterranean area: A comprehensive evaluation of their fatty acid profile and implications for human and animal nutrition. Sustainability 2019, 11, 2197. [Google Scholar] [CrossRef]
- Hamed, K.B.; Castagna, A.; Ranieri, A.; Garcia-Caparros, P.; Santin, M.; Hernandez, J.A.; Espin, G.B. Halophyte based Mediterranean agriculture in the contexts of food insecurity and global climate change. Environ. Exp. Bot. 2021, 191, 104601. [Google Scholar] [CrossRef]
- Ventura, Y.; Wuddineh, W.A.; Myrzabayeva, M.; Alikulov, Z.; Khozin-Goldberg, I.; Shpigel, M.; Samocha, T.; Sagi, M. Effect of seawater concentration on the productivity and nutritional value of annual Salicornia and perennial Sarcocornia halophytes as leafy vegetable crops. Sci. Hortic. 2011, 128, 189–196. [Google Scholar] [CrossRef]
- Petropoulos, S.A.; Karkanis, A.; Martins, N.; Ferreira, I.C. Halophytic herbs of the Mediterranean basin: An alternative approach to health. Food Chem. Toxicol. 2018, 114, 155–169. [Google Scholar] [CrossRef]
- García-Herrera, P.; Sánchez-Mata, M.D.C. The contribution of wild plants to dietary intakes of micronutrients (II): Mineral Elements. In Mediterranean Wild Edible Plants: Ethnobotany and Food Composition Tables; Sánchez-Mata, M., Tardío, J., Eds.; Springer: New York, NY, USA, 2016; pp. 141–171. [Google Scholar]
- Cárdenas-Pérez, S.; Piernik, A.; Chanona-Pérez, J.J.; Grigore, M.N.; Perea-Flores, M.J. An overview of the emerging trends of the Salicornia L. genus as a sustainable crop. Environ. Exp. Bot. 2021, 191, 104606. [Google Scholar] [CrossRef]
- Loconsole, D.; Cristiano, G.; De Lucia, B. Glassworts: From wild salt marsh species to sustainable edible crops. Agriculture 2019, 9, 14. [Google Scholar] [CrossRef]
- Ríos, S.; Obón, C.; Martínez-Francés, V.; Verde, A.; Ariza, D.; Laguna, E. Halophytes as Food: Gastroethnobotany of Halophytes. In Handbook of Halophytes: From Molecules to Ecosystems towards Biosaline Agriculture; Springer: Berlin/Heidelberg, Germany, 2020; pp. 1–36. [Google Scholar]
- De la Cruz, M. 1510 Estepas salinas mediterráneas (Limonietalia) (*). In Bases Ecológicas Preliminares Para la Conservación de los Tipos de Hábitat de Interés Comunitario en España; Ministerio de Medio Ambiente, y Medio Rural y Marino: Madrid, Spain, 2009; 78p. [Google Scholar]
- Rivas Martínez, S.; Herrera, M. Data on Salicornia L. (Chenopodiaceae) in Spain. An. Jard. Bot. Madr. 1996, 54, 149–154. [Google Scholar]
- Urbano, M.; Tomaselli, V.; Bisignano, V.; Veronico, G.; Hammer, K.; Laghetti, G. Salicornia patula Duval-Jouve: From gathering of wild plants to some attempts of cultivation in Apulia region (southern Italy). Genet. Resour. Crop Evol. 2017, 64, 1465–1472. [Google Scholar] [CrossRef]
- Accogli, R.; Tomaselli, V.; Direnzo, P.; Perrino, E.V.; Albanese, G.; Urbano, M.; Laghetti, G. Edible halophytes and halo-tolerant species in Apulia region (Southeastern Italy): Biogeography, traditional food use and potential sustainable crops. Plants 2023, 12, 549. [Google Scholar] [CrossRef]
- Clavel-Coibrié, E.; Sales, J.R.; da Silva, A.M.; Barroca, M.J.; Sousa, I.; Raymundo, A. Sarcocornia perennis: A salt substitute in savory snacks. Foods 2021, 10, 3110. [Google Scholar] [CrossRef]
- Barroca, M.J.; Flores, C.; Ressurreição, S.; Guiné, R.; Osório, N.; Moreira da Silva, A. Re-Thinking Table Salt Reduction in Bread with Halophyte Plant Solutions. Appl. Sci. 2023, 13, 5342. [Google Scholar] [CrossRef]
- Peris, J.B.; Guillen, A.; Roselló, R.; Laguna, E.; Ferrer-Gallego, P.P.; Gómez-Navarro, J. Les plantes utilitzades en les ensalades campestres valencianes. Nemus 2019, 9, 46–64. [Google Scholar]
- Singh, D.; Buhmann, A.K.; Flowers, T.J.; Seal, C.E.; Papenbrock, J. Salicornia as a crop plant in temperate regions: Selection of genetically characterized ecotypes and optimization of their cultivation conditions. AoB Plants 2014, 10, 6. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Gavilán, I.; Rufo, L.; Rodríguez, N.; de la Fuente, V. On the elemental composition of the Mediterranean euhalophyte Salicornia patula Duval-Jouve (Chenopodiaceae) from saline habitats in Spain (Huelva, Toledo and Zamora). Environ. Sci. Pollut. Res. 2021, 28, 2719–2727. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Gavilán, I.; Ramírez, E.; de la Fuente, V. Bioactive Compounds in Salicornia patula Duval-Jouve: A Mediterranean Edible Euhalophyte. Foods 2021, 10, 410. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Mata, M.C.; Cabrera Loera, R.D.; Morales, P.; Fernández-Ruiz, V.; Cámara, M.; Díez Marqués, C.; Pardo de Santayana, M.; Tardío, J. Wild vegetables of the Mediterranean area as valuable sources of bioactive compounds. Genet. Resour. Crop Evol. 2012, 59, 431–443. [Google Scholar] [CrossRef]
- Horowitz, W.; Latimer, G.W. Official Methods of Analysis of AOAC International, 18th ed.; AOAC International: Gaithersburg, MD, USA, 2006. [Google Scholar]
- AOAC. Official Methods of Analysis (Supplement March 1995); Ref. 993.19. Vitamins and Other Nutrients; AOAC International: Gaithersburg, MD, USA, 1993. [Google Scholar]
- Maieves, H.A.; López-Froilán, R.; Morales, P.; Pérez-Rodríguez, M.L.; Ribani, R.H.; Cámara, M.; Sánchez-Mata, M.C. Antioxidant phytochemicals of Hovenia dulcis Thunb. peduncles in different maturity stages. J. Funct. Foods 2015, 18, 1117–1124. [Google Scholar] [CrossRef]
- Arias-Rico, J.; Cruz-Cansino, N.D.S.; Cámara-Hurtado, M.; López-Froilán, R.; Pérez-Rodríguez, M.L.; Sánchez-Mata, M.D.C.; Jaramillo-Morales, O.A.; Barrera-Gálvez, R.; Ramírez-Moreno, E. Study of xoconostle (Opuntia spp.) powder as source of dietary fiber and antioxidants. Foods 2020, 9, 403. [Google Scholar] [CrossRef]
- Igual, M.; García-Herrera, P.; Cámara, R.M.; Martínez-Monzó, J.; García-Segovia, P.; Cámara, M. Bioactive compounds in rosehip (Rosa canina) powder with encapsulating agents. Molecules 2022, 27, 4737. [Google Scholar] [CrossRef]
- Zaier, M.M.; Ciudad-Mulero, M.; Cámara, M.; Pereira, C.; Ferreira, I.C.; Achour, L.; Kacem, A.; Morales, P. Revalorization of Tunisian wild Amaranthaceae halophytes: Nutritional composition variation at two different phenotypes stages. J. Food Compos. Anal. 2020, 89, 103463. [Google Scholar] [CrossRef]
- BEDCA Database. Available online: https://www.bedca.net/bdpub/ (accessed on 21 December 2023).
- European Parliament. Regulation (EC) No. 1924/2006 of the European Parliament and of the Council of 20 December 2006 on nutrition and health claims made on foods. Off. J. Eur. Union 2006, 404, 9. [Google Scholar]
- Morales, P.; Ferreira, I.C.; Carvalho, A.M.; Sánchez-Mata, M.C.; Cámara, M.; Fernández-Ruiz, V.; Pardo de Santayana, M.; Tardío, J. Mediterranean non-cultivated vegetables as dietary sources of compounds with antioxidant and biological activity. LWT-Food Sci. Technol. 2014, 55, 389–396. [Google Scholar] [CrossRef]
- Concon, J.M. Toxicology. Principles and Concepts; Marcel Dekker: New York, NY, USA, 1988. [Google Scholar]
- Derache, R. Toxicología y Seguridad de los Alimentos; Omega: Barcelona, Spain, 1990. [Google Scholar]
- Magni, N.N.; Veríssimo, A.C.; Silva, H.; Pinto, D.C. Metabolomic Profile of Salicornia perennis Plant’s Organs under Diverse in Situ Stress: The Ria de Aveiro Salt Marshes Case. Metabolites 2023, 13, 280. [Google Scholar] [CrossRef] [PubMed]
- Antunes, M.D.; Gago, C.; Guerreiro, A.; Sousa, A.R.; Julião, M.; Miguel, M.G.; Faleiro, M.L.; Panagopoulos, T. Nutritional characterization and storage ability of Salicornia ramosissima and Sarcocornia perennis for fresh vegetable salads. Horticulturae 2021, 7, 6. [Google Scholar] [CrossRef]
- Lu, D.; Zhang, M.; Wang, S.; Cai, J.; Zhou, X.; Zhu, C. Nutritional characterization and changes in quality of Salicornia bigelovii Torr. during storage. LWT-Food Sci. Technol. 2010, 43, 519–524. [Google Scholar] [CrossRef]
- Srivarathan, S.; Phan, A.D.T.; Hong, H.T.; Netzel, G.; Wright, O.R.; Sultanbawa, Y.; Netzel, M.E. Nutritional composition and anti-nutrients of underutilized Australian indigenous edible halophytes—Saltbush, Seablite and Seapurslane. J. Food Compos. Anal. 2023, 115, 104876. [Google Scholar] [CrossRef]
- Kumari, A.; Das, P.; Parida, A.K.; Agarwal, P.K. Proteomics, metabolomics, and ionomics perspectives of salinity tolerance in halophytes. Front. Plant Sci. 2015, 6, 537. [Google Scholar] [CrossRef]
- Jiang, Z.; Huang, Q.; Jia, D.; Zhong, M.; Tao, J.; Liao, G.; Huang, C.; Xu, X. Characterization of Organic Acid Metabolism and Expression of Related Genes During Fruit Development of Actinidia eriantha ‘Ganmi 6’. Plants 2020, 9, 332. [Google Scholar] [CrossRef]
| Sample ID | Site | Grid Reference MGRS |
|---|---|---|
| S1 | Huelva, La Rábida | 29SPB8320 |
| S2 | Huelva, Moguer | 29SPB7729 |
| S3 | Huelva, El Terrón | 29SPB4717 |
| S4 | Huelva, San Juan del Puerto | 29SPB9230 |
| S5 | Huelva, Monumento a Colón | 29SPB8220 |
| S6 | Valladolid Aldeamayor de San Martín | 30TUL6196 |
| S7 | Madrid, Colmenar de Oreja | 31TVK5143 |
| S8 | Toledo, Laguna larga de Villacañas | 30SYH0928 |
| Sample | Moisture | Insoluble Fiber | Soluble Fiber | Total Fiber |
|---|---|---|---|---|
| S1 | 90.121 ± 0.621 a | 4.417 ± 0.101 a | 0.841± 0.323 a | 5.269 ± 0.336 a |
| S2 | 89.876 ± 1.123 b | 4.800 ± 0.441 ab | 1.409 ± 0.134 b | 6.301 ± 0.438 b |
| S3 | 90.725 ± 0.731 a | 4.603 ± 0.614 ab | 1.114 ± 0.190 b | 5.782 ± 0.811 a |
| S4 | 91.041 ± 0.775 ab | 4.612 ± 0.140 a | 0.488 ± 0.182 a | 5.104 ± 0.276 ab |
| S5 | 90.183 ± 1.411 a | 5.660 ± 0.312 b | 0.519 ± 0.081 a | 6.185 ± 0.290 a |
| S6 | 91.290 ± 1.012 ab | 5.682 ± 0.129 a | 0.793 ± 0.073 a | 6.307 ± 0.273 ab |
| S7 | 90.763 a ± 1.356 a | 3.996 ± 0.181 a | 1.034 ± 0.372 b | 5.038 ± 0.543 a |
| S8 | 91.111 ± 0.813 ab | 4.229 ± 0.163 a | 0.568 ± 0.051 a | 4.812 ± 0.221 ab |
| Sample | Oxalic Acid | Malic Acid | Citric Acid | Succinic Acid | Oxalic Acid/Ca |
|---|---|---|---|---|---|
| S1 | 0.8224 ± 0.061 a | 0.016 ± 0.002 a | 0.072 ± 0.008 b | 0.033 ± 0.008 a | 8.132 ± 0.241 a |
| S2 | 1.060 ± 0.018 b | 0.015 ± 0.001 a | 0.020 ± 0.003 a | 0.012 ± 0.004 b | 11.381 ± 1.313 b |
| S3 | 1.691 ± 0.007 b | Nd | Nd | Nd | 34.006 ± 3.711 ab |
| S4 | 1.187 ± 0.067 b | 0.087 ± 0.053 b | Nd | 0.009 ± 0.003 a | 11.082 ± 1.192 a |
| S5 | 1.533± 0.045 b | 0.016 ± 0.011 a | Nd | 0.077 ± 0.001 b | 7.581 a ± 0.237 a |
| S6 | 0.151 ± 0.023 ab | 0.024 ± 0.071 a | Nd | Nd | 6.689 ± 1.052 a |
| S7 | 0.758 ± 0.013 a | 0.019 ± 0.017 a | 0.004 ± 0.000 a | Nd | 8.061 ± 1.133 a |
| S8 | 0.877 ± 0.555 a | Nd | 0.003 ± 0.000 a | Nd | 4.763 ± 0.920 ab |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez Gavilán, I.; Velázquez Ybarzabal, D.; de la Fuente, V.; Cámara, R.M.; Sánchez-Mata, M.C.; Cámara, M. Valorization of Salicornia patula Duval-Jouve Young Shoots in Healthy and Sustainable Diets. Nutrients 2024, 16, 358. https://doi.org/10.3390/nu16030358
Sánchez Gavilán I, Velázquez Ybarzabal D, de la Fuente V, Cámara RM, Sánchez-Mata MC, Cámara M. Valorization of Salicornia patula Duval-Jouve Young Shoots in Healthy and Sustainable Diets. Nutrients. 2024; 16(3):358. https://doi.org/10.3390/nu16030358
Chicago/Turabian StyleSánchez Gavilán, Irene, Daniela Velázquez Ybarzabal, Vicenta de la Fuente, Rosa M. Cámara, María Cortes Sánchez-Mata, and Montaña Cámara. 2024. "Valorization of Salicornia patula Duval-Jouve Young Shoots in Healthy and Sustainable Diets" Nutrients 16, no. 3: 358. https://doi.org/10.3390/nu16030358
APA StyleSánchez Gavilán, I., Velázquez Ybarzabal, D., de la Fuente, V., Cámara, R. M., Sánchez-Mata, M. C., & Cámara, M. (2024). Valorization of Salicornia patula Duval-Jouve Young Shoots in Healthy and Sustainable Diets. Nutrients, 16(3), 358. https://doi.org/10.3390/nu16030358









