Intestinal Barrier Impairment, Preservation, and Repair: An Update
Abstract
1. Introduction
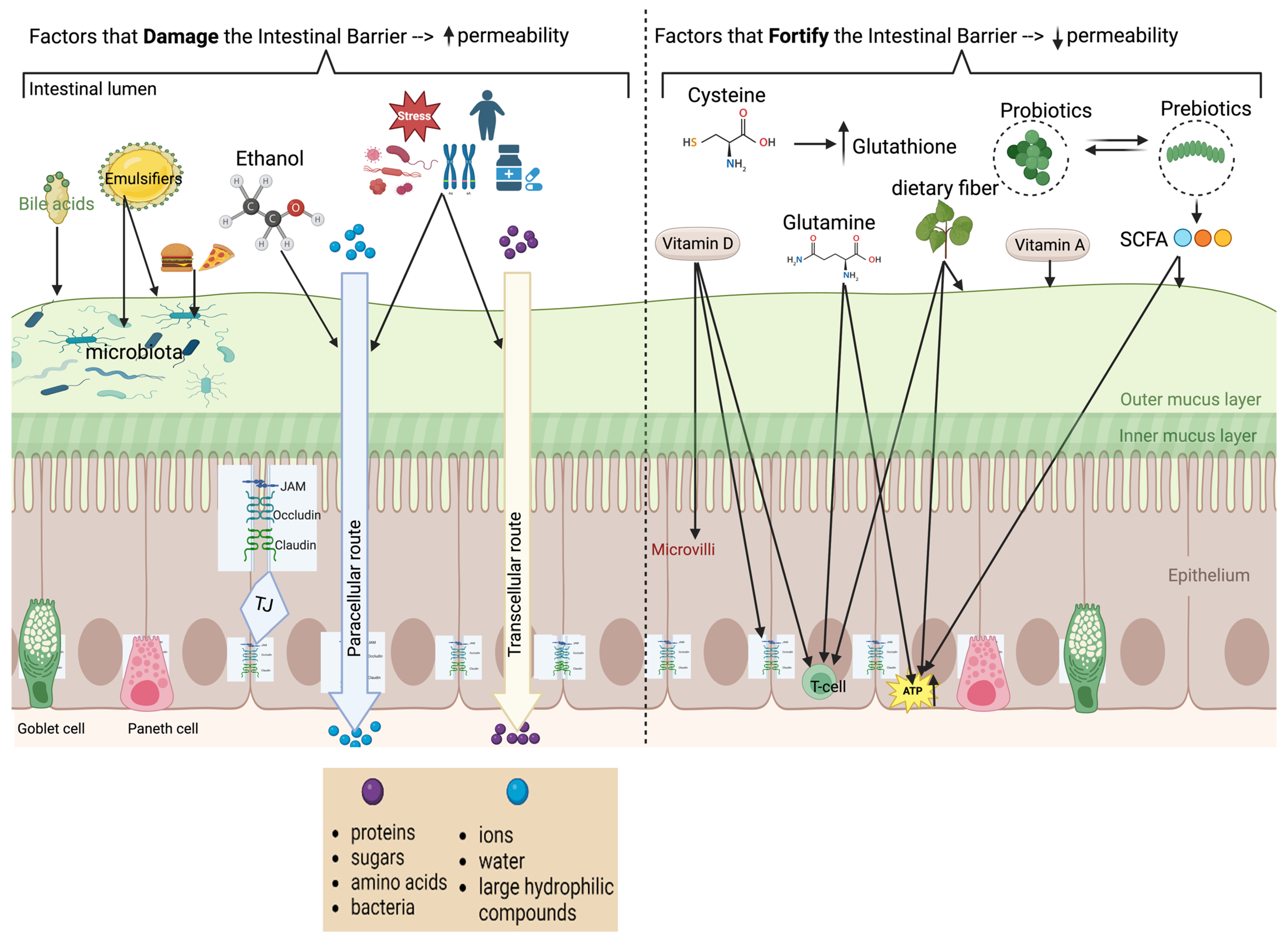
Intestinal Barrier and Permeability
2. Epigenetic Mechanisms Related to Intestinal Permeability
3. Effect of Food Substances and Disease States on Intestinal Permeability
3.1. Food Substances That Decrease Intestinal Barrier Integrity or Increase Permeability
3.2. Altered Permeability and Disease States
4. Effects of Dietary Components That Enhance or Damage the Intestinal Barrier
4.1. Nutrients
4.1.1. Substances That Help Increase Intestinal Barrier Integrity
4.1.2. Dietary Fiber
4.1.3. Polyphenols, Anthocyanins, and Ellagitannins
4.1.4. Glutamine
4.1.5. Vitamin D
4.1.6. Zinc
4.1.7. Emulsifiers
4.2. Microbial Nutrients and Metabolites
4.2.1. Prebiotics
4.2.2. Probiotics
4.2.3. Synbiotics
5. Recommendations and Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Barbara, G.; Barbaro, M.R.; Fuschi, D.; Palombo, M.; Falangone, F.; Cremon, C.; Marasco, G.; Stanghellini, V. Inflammatory and microbiota-related regulation of the intestinal epithelial barrier. Front. Nutr. 2021, 8, 718356. [Google Scholar] [CrossRef] [PubMed]
- Farré, R.; Fiorani, M.; Rahiman, S.A.; Matteoli, G. Intestinal permeability, inflammation and the role of nutrients. Nutrients 2020, 12, 1185. [Google Scholar] [CrossRef]
- de Souza Marinho do Nascimento, D.; Costa Campos Mota, A.C.; da Cruz Carvalho, M.C.; de Oliveira Andrade, E.D.; de Oliveira, E.P.S.F.; Galvão, L.L.P.; Maciel, B.L.L. Can diet alter the intestinal barrier permeability in healthy people? A systematic review. Nutrients 2024, 16, 1871. [Google Scholar] [CrossRef] [PubMed]
- Allam-Ndoul, B.; Castonguay-Paradis, S.; Veilleux, A. Gut microbiota and intestinal trans-epithelial permeability. Int. J. Mol. Sci. 2020, 21, 6402. [Google Scholar] [CrossRef]
- Ahern, P.P.; Maloy, K.J. Understanding immune–microbiota interactions in the intestine. Immunology 2020, 159, 4–14. [Google Scholar] [CrossRef]
- Allaire, J.M.; Crowley, S.M.; Law, H.T.; Chang, S.-Y.; Ko, H.-J.; Vallance, B.A. The intestinal epithelium: Central coordinator of mucosal immunity. Trends Immunol. 2018, 39, 677–696. [Google Scholar] [CrossRef]
- Dunleavy, K.A.; Raffals, L.E.; Camilleri, M. Intestinal barrier dysfunction in inflammatory bowel disease: Underpinning pathogenesis and therapeutics. Dig. Dis. Sci. 2023, 68, 4306–4320. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Shang, W.; Bao, L.; Peng, Z.; Wu, C. Epithelial-immune cell crosstalk for intestinal barrier homeostasis. Eur. J. Immunol. 2024, 54, e2350631. [Google Scholar] [CrossRef]
- Citi, S.; Fromm, M.; Furuse, M.; González-Mariscal, L.; Nusrat, A.; Tsukita, S.; Turner, J.R. A short guide to the tight junction. J. Cell Sci. 2024, 137, jcs261776. [Google Scholar] [CrossRef]
- Imafuku, K.; Iwata, H.; Natsuga, K.; Okumura, M.; Kobayashi, Y.; Kitahata, H.; Kubo, A.; Nagayama, M.; Ujiie, H. Zonula occludens-1 distribution and barrier functions are affected by epithelial proliferation and turnover rates. Cell Prolif. 2023, 56, e13441. [Google Scholar] [CrossRef]
- AlMarzooqi, S.K.; Almarzooqi, F.; Sadida, H.Q.; Jerobin, J.; Ahmed, I.; Abou-Samra, A.-B.; Fakhro, K.A.; Dhawan, P.; Bhat, A.A.; Al-Shabeeb Akil, A.S. Deciphering the complex interplay of obesity, epithelial barrier dysfunction, and tight junction remodeling: Unraveling potential therapeutic avenues. Obes. Rev. 2024, 25, e13766. [Google Scholar] [CrossRef] [PubMed]
- Horowitz, A.; Chanez-Paredes, S.D.; Haest, X.; Turner, J.R. Paracellular permeability and tight junction regulation in gut health and disease. Nat. Rev. Gastroenterol. Hepatol. 2023, 20, 417–432. [Google Scholar] [CrossRef] [PubMed]
- Zuo, L.; Kuo, W.-T.; Turner, J.R. Tight junctions as targets and effectors of mucosal immune homeostasis. Cell. Mol. Gastroenterol. Hepatol. 2020, 10, 327–340. [Google Scholar] [CrossRef]
- Oami, T.; Yumoto, T.; Shimazui, T.; Sarmiento, S.; Klingensmith, N.J.; Chen, C.-W.; Otani, S.; Liang, Z.; Burd, E.M.; Mahdi, Z.K.; et al. Chronic ethanol use worsens gut permeability and alters tight junction expression in a murine sepsis model. Shock 2023, 60, 280–290. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T. Regulation of intestinal epithelial permeability by tight junctions. Cell Mol. Life Sci. 2013, 70, 631–659. [Google Scholar] [CrossRef] [PubMed]
- Buckley, A.; Turner, J.R. Cell biology of tight junction barrier regulation and mucosal disease. Cold Spring Harb. Perspect. Biol. 2018, 10, a029314. [Google Scholar] [CrossRef]
- Lacy, B.E.; Wise, J.L.; Cangemi, D.J. Leaky gut syndrome: Myths and management. Gastroenterol. Hepatol. 2024, 20, 265. [Google Scholar]
- Perez-Diaz-del-Campo, N.; Castelnuovo, G.; Ribaldone, D.G.; Caviglia, G.P. Fecal and circulating biomarkers for the non-invasive assessment of intestinal permeability. Diagnostics 2023, 13, 1976. [Google Scholar] [CrossRef]
- Camilleri, M. Leaky gut: Mechanisms, measurement and clinical implications in humans. Gut 2019, 68, 1516–1526. [Google Scholar] [CrossRef]
- Meyer, F.; Wendling, D.; Demougeot, C.; Prati, C.; Verhoeven, F. Cytokines and intestinal epithelial permeability: A systematic review. Autoimmun. Rev. 2023, 22, 103331. [Google Scholar] [CrossRef]
- Camilleri, M.; Vella, A. What to do about the leaky gut. Gut 2022, 71, 424–435. [Google Scholar] [CrossRef] [PubMed]
- Schoultz, I.; Keita, A.V. The intestinal barrier and current techniques for the assessment of gut permeability. Cells 2020, 9, 1909. [Google Scholar] [CrossRef] [PubMed]
- Mahurkar-Joshi, S.; Chang, L. Epigenetic mechanisms in irritable bowel syndrome. Front. Psychiatry 2020, 11, 805. [Google Scholar] [CrossRef] [PubMed]
- Wiley, J.W.; Zong, Y.; Zheng, G.; Zhu, S.; Hong, S. Histone H3K9 methylation regulates chronic stress and IL-6-induced colon epithelial permeability and visceral pain. Neurogastroenterol. Motil. 2020, 32, e13941. [Google Scholar] [CrossRef]
- Zhou, Q.; Costinean, S.; Croce, C.M.; Brasier, A.R.; Merwat, S.; Larson, S.A.; Basra, S.; Verne, G.N. MicroRNA 29 targets nuclear factor-κB-repressing factor and Claudin 1 to increase intestinal permeability. Gastroenterology 2015, 148, 158–169.e8. [Google Scholar] [CrossRef]
- Wiley, J.W.; Higgins, G.A.; Hong, S. Chronic psychological stress alters gene expression in rat colon epithelial cells promoting chromatin remodeling, barrier dysfunction and inflammation. PeerJ 2022, 10, e13287. [Google Scholar] [CrossRef]
- Zhou, Q.; Souba, W.W.; Croce, C.M.; Verne, G.N. MicroRNA-29a regulates intestinal membrane permeability in patients with irritable bowel syndrome. Gut 2010, 59, 775–784. [Google Scholar] [CrossRef] [PubMed]
- Chao, G.; Wang, Y.; Zhang, S.; Yang, W.; Ni, Z.; Zheng, X. MicroRNA-29a increased the intestinal membrane permeability of colonic epithelial cells in irritable bowel syndrome rats. Oncotarget 2017, 8, 85828–85837. [Google Scholar] [CrossRef]
- Wohlfarth, C.; Schmitteckert, S.; Härtle, J.D.; Houghton, L.A.; Dweep, H.; Fortea, M.; Assadi, G.; Braun, A.; Mederer, T.; Pöhner, S.; et al. miR-16 and miR-103 impact 5-HT(4) receptor signalling and correlate with symptom profile in irritable bowel syndrome. Sci. Rep. 2017, 7, 14680. [Google Scholar] [CrossRef]
- Martínez, C.; Rodiño-Janeiro, B.K.; Lobo, B.; Stanifer, M.L.; Klaus, B.; Granzow, M.; González-Castro, A.M.; Salvo-Romero, E.; Alonso-Cotoner, C.; Pigrau, M.; et al. miR-16 and miR-125b are involved in barrier function dysregulation through the modulation of claudin-2 and cingulin expression in the jejunum in IBS with diarrhoea. Gut 2017, 66, 1537–1538. [Google Scholar] [CrossRef]
- Hou, Q.; Huang, Y.; Zhu, S.; Li, P.; Chen, X. MiR-144 increases intestinal permeability in IBS-D rats by targeting OCLN and ZO1. Cell. Physiol. Biochem. 2017, 44, 2256–2268. [Google Scholar] [CrossRef] [PubMed]
- Fazio, A.; Bordoni, D.; Kuiper, J.W.P.; Weber-Stiehl, S.; Stengel, S.T.; Arnold, P.; Ellinghaus, D.; Ito, G.; Tran, F.; Messner, B.; et al. DNA methyltransferase 3A controls intestinal epithelial barrier function and regeneration in the colon. Nat. Commun. 2022, 13, 6266. [Google Scholar] [CrossRef] [PubMed]
- Rawat, M.; Nighot, M.; Al-Sadi, R.; Gupta, Y.; Viszwapriva, D.; Yochum, G.; Koltun, W.; Ma, T.Y. IL1B increases intestinal tight junction permeability by up-regulation of MIR200C-3p which degrades occludin, m.R.N.A. Gastroenterology 2020, 159, 1375–1389. [Google Scholar] [CrossRef]
- Bowes, M.M.; Casares-Marfil, D.; Sawalha, H. Intestinal permeability correlates with disease activity and DNA methylation changes in lupus patients. Clin. Immunol. 2024, 262, 110173. [Google Scholar] [CrossRef]
- Higgins, G.A.; Hong, S.; Wiley, J.W. The role of epigenomic regulatory pathways in the gut-brain axis and visceral hyperalgesia. Cell. Mol. Neurobiol. 2022, 42, 361–376. [Google Scholar] [CrossRef]
- Yuan, X.; Tan, Y.; Bajinka, O.; Jammeh, M.L.; Dukureh, A.; Obiegbusi, C.N.; Abdelhalim, K.A. The connection between epigenetics and gut microbiota-current perspective. Cell Biochem. Funct. 2024, 42, e3941. [Google Scholar] [CrossRef]
- Wu, S.-E.; Hashimoto-Hill, S.; Woo, V.; Eshleman, E.M.; Whitt, J.; Engleman, L.; Karns, R.; Denson, L.A.; Haslam, D.B.; Alenghat, T. Microbiota-derived metabolite promotes HDAC3 activity in the gut. Nature 2020, 586, 108–112. [Google Scholar] [CrossRef] [PubMed]
- Miousse, I.R.; Pathak, R.; Garg, S.; Skinner, C.M.; Melnyk, S.; Pavliv, O.; Hendrickson, H.; Landes, R.D.; Lumen, A.; Tackett, A.J.; et al. Short-term dietary methionine supplementation affects one-carbon metabolism and DNA methylation in the mouse gut and leads to altered microbiome profiles, barrier function, gene expression and histomorphology. Genes Nutr. 2017, 12, 22. [Google Scholar] [CrossRef]
- Pepke, M.L.; Hansen, S.B.; Limborg, M.T. Unraveling host regulation of gut microbiota through the epigenome-microbiome axis. Trends Microbiol. 2024. Online ahead of print. [Google Scholar] [CrossRef]
- Speciale, A.; Molonia, M.S.; Muscarà, C.; Cristani, M.; Salamone, F.L.; Saija, A.; Cimino, F. An overview on the cellular mechanisms of anthocyanins in maintaining intestinal integrity and function. Fitoterapia 2024, 175, 105953. [Google Scholar] [CrossRef]
- Jimenez-Rondan, F.R.; Ruggiero, C.H.; Lobean McKinley, K.; Koh, J.; Roberts, J.F.; Triplett, E.W.; Cousins, R.J. Enterocyte-specific deletion of metal transporter Zip14 (Slc39a14) alters intestinal homeostasis through epigenetic mechanisms. Am. J. Physiol. Gastrointest. Liver Physiol. 2023, 324, G159–G176. [Google Scholar] [CrossRef] [PubMed]
- Khoshbin, K.; Camilleri, M. Effects of dietary components on intestinal permeability in health and disease. Am. J. Physiol. Gastrointest. Liver Physiol. 2020, 319, G589–G608. [Google Scholar] [CrossRef] [PubMed]
- Amar, J.; Burcelin, R.; Ruidavets, J.B.; Cani, P.D.; Fauvel, J.; Alessi, M.C.; Chamontin, B.; Ferriéres, J. Energy intake is associated with endotoxemia in apparently healthy men. Am. J. Clin. Nutr. 2008, 87, 1219–1223. [Google Scholar] [CrossRef]
- Leutner, M.; Göbl, C.; Wielandner, A.; Howorka, E.; Prünner, M.; Bozkurt, L.; Schlager, O.; Charwat-Resl, S.; Kautzky-Willer, A. Clinical and metabolic characteristics of treated hyperlipidemic patients additionally affected by subclinical hyperglycemia. Lipids Health Dis. 2016, 15, 10. [Google Scholar] [CrossRef]
- Bowser, S.M.; McMillan, R.P.; Boutagy, N.E.; Tarpey, M.D.; Smithson, A.T.; Osterberg, K.L.; Neilson, A.P.; Davy, B.M.; Davy, K.P.; Hulver, M.W. Serum endotoxin, gut permeability and skeletal muscle metabolic adaptations following a short term high fat diet in humans. Metabolism 2020, 103, 154041. [Google Scholar] [CrossRef]
- Roberts, C.L.; Keita, A.V.; Duncan, S.H.; O’Kennedy, N.; Söderholm, J.D.; Rhodes, J.M.; Campbell, B.J. Translocation of Crohns disease Escherichia coli across M-cells: Contrasting effects of soluble plant fibres and emulsifiers. Gut 2010, 59, 1331–1339. [Google Scholar] [CrossRef]
- Mistry, R.H.; Liu, F.; Borewicz, K.; Lohuis, M.A.M.; Smidt, H.; Verkade, H.J.; Tietge, U.J.F. Long-term β-galacto-oligosaccharides supplementation decreases the development of obesity and insulin resistance in mice fed a western-type diet. Mol. Nutr. Food Res. 2020, 64, e1900922. [Google Scholar] [CrossRef]
- Gondalia, S.V.; Wymond, B.; Benassi-Evans, B.; Berberzy, P.; Bird, A.R.; Belobrajdic, D.P. Substitution of refined conventional wheat flour with wheat high in resistant starch modulates the intestinal microbiota and fecal metabolites in healthy adults: A randomized, controlled trial. J. Nutr. 2022, 152, 1426–1437. [Google Scholar] [CrossRef]
- Yamamoto, E.A.; Jørgensen, T.N. Relationships between vitamin D, gut microbiome, and systemic autoimmunity. Front. Immunol. 2020, 10, 499337. [Google Scholar] [CrossRef]
- Thomas, R.L.; Jiang, L.; Adams, J.S.; Xu, Z.Z.; Shen, J.; Janssen, S.; Ackermann, G.; Vanderschueren, D.; Pauwels, S.; Knight, R.; et al. Vitamin D metabolites and the gut microbiome in older men. Nat. Commun. 2020, 11, 5997. [Google Scholar] [CrossRef]
- Koren, O.; Tako, E. Chronic dietary zinc deficiency alters gut microbiota composition and function. Proceedings 2020, 61, 16. [Google Scholar] [CrossRef]
- Skalny, A.V.; Aschner, M.; Lei, X.G.; Gritsenko, V.A.; Santamaria, A.; Alekseenko, S.I.; Prakash, N.T.; Chang, J.-S.; Sizova, E.A.; Chao, J.C.J.; et al. Gut microbiota as a mediator of essential and toxic effects of zinc in the intestines and other tissues. Int. J. Mol. Sci. 2021, 22, 13074. [Google Scholar] [CrossRef] [PubMed]
- Abdulrahman, A.O.; Alzubaidi, M.Y.; Nadeem, M.S.; Khan, J.A.; Rather, I.A.; Khan, M.I. Effects of urolithins on obesity-associated gut dysbiosis in rats fed on a high-fat diet. Int. J. Food Sci. Nutr. 2021, 72, 923–934. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Yan, Y.; Yin, B.; Zhang, L.; Qin, W.; Niu, Y.; Tang, Y.; Zhou, S.; Yan, X.; Ma, L. Dietary glycyl-glutamine supplementation ameliorates intestinal integrity, inflammatory response, and oxidative status in association with the gut microbiota in LPS-challenged piglets. Food Funct. 2021, 12, 3539–3551. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.; Hu, M.; Li, M.; Hou, J.; Zhang, X.; Gao, Y.; Chachar, B.; Li, X. Dietary bioactive peptide alanyl-glutamine attenuates dextran sodium sulfate-induced colitis by modulating gut microbiota. Oxid. Med. Cell Longev. 2021, 2021, 5543003. [Google Scholar] [CrossRef]
- Tian, S.; Chu, Q.; Ma, S.; Ma, H.; Song, H. Dietary fiber and its potential role in obesity: A focus on modulating the gut microbiota. J. Agric. Food Chem. 2023, 71, 14853–14869. [Google Scholar] [CrossRef]
- Rezende, E.S.V.; Lima, G.C.; Naves, M.M.V. Dietary fibers as beneficial microbiota modulators: A proposed classification by prebiotic categories. Nutrition 2021, 89, 111217. [Google Scholar] [CrossRef]
- Ye, S.; Shah, B.R.; Li, J.; Liang, H.; Zhan, F.; Geng, F.; Li, B. A critical review on interplay between dietary fibers and gut microbiota. Trends Food Sci. Technol. 2022, 124, 237–249. [Google Scholar] [CrossRef]
- Fusco, W.; Lorenzo, M.B.; Cintoni, M.; Porcari, S.; Rinninella, E.; Kaitsas, F.; Lener, E.; Mele, M.C.; Gasbarrini, A.; Collado, M.C.; et al. Short-chain fatty-acid-producing bacteria: Key components of the human gut microbiota. Nutrients 2023, 15, 2211. [Google Scholar] [CrossRef]
- Blaak, E.E.; Canfora, E.E.; Theis, S.; Frost, G.; Groen, A.K.; Mithieux, G.; Nauta, A.; Scott, K.; Stahl, B.; van Harsselaar, J.; et al. Short chain fatty acids in human gut and metabolic health. Benef. Microbes 2020, 11, 411–455. [Google Scholar] [CrossRef]
- Nogal, A.; Louca, P.; Zhang, X.; Wells, P.M.; Steves, C.J.; Spector, T.D.; Falchi, M.; Valdes, A.M.; Menni, C. Circulating levels of the short-chain fatty acid acetate mediate the effect of the gut microbiome on visceral fat. Front. Microbiol. 2021, 12, 711359. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Li, Q.; Yang, Y.; Guo, A. Biological function of short-chain fatty acids and its regulation on intestinal health of poultry. Front. Vet. Sci. 2021, 8, 736739. [Google Scholar] [CrossRef] [PubMed]
- Lavefve, L.; Howard, L.R.; Carbonero, F. Berry polyphenols metabolism and impact on human gut microbiota and health. Food Funct. 2020, 11, 45–65. [Google Scholar] [CrossRef] [PubMed]
- Aravind, S.M.; Wichienchot, S.; Tsao, R.; Ramakrishnan, S.; Chakkaravarthi, S. Role of dietary polyphenols on gut microbiota, their metabolites and health benefits. Food Res. Int. 2021, 142, 110189. [Google Scholar] [CrossRef]
- Bouyahya, A.; El Omari, N.; El Hachlafi, N.; El Jemly, M.; Hakkour, M.; Balahbib, A.; El Menyiy, N.; Bakrim, S.; Mrabti, H.N.; Khouchlaa, A.; et al. Chemical compounds of berry-derived polyphenols and their effects on gut microbiota, inflammation, and cancer. Molecules 2022, 27, 3286. [Google Scholar] [CrossRef]
- García-Villalba, R.; Tomás-Barberán, F.A.; Iglesias-Aguirre, C.E.; Giménez-Bastida, J.A.; González-Sarrías, A.; Selma, M.V.; Espín, J.C. Ellagitannins, urolithins, and neuroprotection: Human evidence and the possible link to the gut microbiota. Mol. Asp. Med. 2023, 89, 101109. [Google Scholar] [CrossRef]
- García-Villalba, R.; Giménez-Bastida, J.A.; Cortés-Martín, A.; Ávila-Gálvez, M.A.; Tomás-Barberán, F.A.; Selma, M.V.; Espín, J.C.; González-Sarrías, A. Urolithins: A comprehensive update on their metabolism, bioactivity, and associated gut microbiota. Mol. Nutr. Food Res. 2022, 66, e2101019. [Google Scholar] [CrossRef]
- Bellerba, F.; Muzio, V.; Gnagnarella, P.; Facciotti, F.; Chiocca, S.; Bossi, P.; Cortinovis, D.; Chiaradonna, F.; Serrano, D.; Raimondi, S.; et al. The association between vitamin D and gut microbiota: A systematic review of human studies. Nutrients 2021, 13, 3378. [Google Scholar] [CrossRef]
- Chen, L.; Wang, Z.; Wang, P.; Yu, X.; Ding, H.; Wang, Z.; Feng, J. Effect of long-term and short-term imbalanced Zn manipulation on gut microbiota and screening for microbial markers sensitive to zinc status. Microbiol. Spectr. 2021, 9, e00483-21. [Google Scholar] [CrossRef]
- Pajarillo, E.A.B.; Lee, E.; Kang, D.-K. Trace metals and animal health: Interplay of the gut microbiota with iron, manganese, zinc, and copper. Anim. Nutr. 2021, 7, 750–761. [Google Scholar] [CrossRef]
- Ghosh, S.; Whitley, C.S.; Haribabu, B.; Jala, V.R. Regulation of intestinal barrier function by microbial metabolites. Cell. Mol. Gastroenterol. Hepatol. 2021, 11, 1463–1482. [Google Scholar] [CrossRef] [PubMed]
- Levine, A.R.; Picoraro, J.A.; Dorfzaun, S.; LeLeiko, N.S. Emulsifiers and intestinal health: An introduction. J. Pediatr. Gastroenterol. Nutr. 2022, 74, 314–319. [Google Scholar] [CrossRef] [PubMed]
- Naimi, S.; Viennois, E.; Gewirtz, A.T.; Chassaing, B. Direct impact of commonly used dietary emulsifiers on human gut Microbiota. Microbiome 2021, 9, 66. [Google Scholar] [CrossRef]
- Bancil, A.S.; Sandall, A.M.; Rossi, M.; Chassaing, B.; Lindsay, J.O.; Whelan, K. Food additive emulsifiers and their impact on gut microbiome, permeability, and inflammation: Mechanistic insights in inflammatory bowel disease. J. Crohns. Colitis 2021, 15, 1068–1079. [Google Scholar] [CrossRef]
- Ogulur, I.; Yazici, D.; Pat, Y.; Bingöl, E.N.; Babayev, H.; Ardicli, S.; Heider, A.; Rückert, B.; Sampath, V.; Dhir, R.; et al. Mechanisms of gut epithelial barrier impairment caused by food emulsifiers polysorbate 20 and polysorbate 80. Allergy 2023, 78, 2441–2455. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, J.A.; Gibson, P.R.; Taylor, K.M.; Halmos, E.P. The effect of dietary emulsifiers and thickeners on intestinal barrier function and its response to acute stress in healthy adult humans: A randomised controlled feeding study. Aliment. Pharmacol. Ther. 2024. Online ahead of print. [Google Scholar] [CrossRef]
- Gibson, G.R.; Hutkins, R.; Sanders, M.E.; Prescott, S.L.; Reimer, R.A.; Salminen, S.J.; Scott, K.; Stanton, C.; Swanson, K.S.; Cani, P.D.; et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 491–502. [Google Scholar] [CrossRef] [PubMed]
- Makki, K.; Deehan, E.C.; Walter, J.; Bäckhed, F. The Impact of dietary fiber on gut microbiota in host health and disease. Cell Host Microbe 2018, 23, 705–715. [Google Scholar] [CrossRef]
- Looijer-Van Langen, M.A.C.; Dieleman, L.A. Prebiotics in chronic intestinal inflammation. Inflamm. Bowel Dis. 2008, 15, 454–462. [Google Scholar] [CrossRef]
- Francavilla, R.; Castellaneta, S.; Masciale, A.; Straziuso, S.; Polimeno, L.; Gagliardi, F.; Bohem, G.; Miniello, V. Intestinal permeability and faecal flora of infants fed with a prebiotic supplemented formula: A double blind placebo controlled study. J. Pediatr. Gastroenterol. Nutr. 2006, 42, E96–E97. [Google Scholar] [CrossRef]
- Westerbeek, E.A.; van den Berg, J.P.; Lafeber, H.N.; Fetter, W.P.; Boehm, G.; Twisk, J.W.; van Elburg, R.M. Neutral and acidic oligosaccharides in preterm infants: A randomized, double-blind, placebo-controlled trial1234. Am. J. Clin. Nutr. 2010, 91, 679–686. [Google Scholar] [CrossRef] [PubMed]
- Ho, J.; Nicolucci, A.C.; Virtanen, H.; Schick, A.; Meddings, J.; Reimer, R.A.; Huang, C. Effect of prebiotic on microbiota, intestinal permeability, and glycemic control in children with type 1 diabetes. J. Clin. Endocrinol. Metab. 2019, 104, 4427–4440. [Google Scholar] [CrossRef]
- Olguin, F.; Araya, M.; Hirsch, S.; Brunser, O.; Ayala, V.; Rivera, R.; Gotteland, M. Prebiotic ingestion does not improve gastrointestinal barrier function in burn patients. Burns 2005, 31, 482–488. [Google Scholar] [CrossRef]
- Russo, F.; Linsalata, M.; Clemente, C.; Chiloiro, M.; Orlando, A.; Marconi, E.; Chimienti, G.; Riezzo, G. Inulin-enriched pasta improves intestinal permeability and modifies the circulating levels of zonulin and glucagon-like peptide 2 in healthy young volunteers. Nutr. Res. 2012, 32, 940–946. [Google Scholar] [CrossRef] [PubMed]
- Salden, B.N.; Troost, F.J.; Wilms, E.; Truchado, P.; Vilchez-Vargas, R.; Pieper, D.H.; Jáuregui, R.; Marzorati, M.; van de Wiele, T.; Possemiers, S.; et al. Reinforcement of intestinal epithelial barrier by arabinoxylans in overweight and obese subjects: A randomized controlled trial: Arabinoxylans in gut barrier. Clin. Nutr. 2018, 37, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Müller, M.; Hermes, G.D.A.; Emauel, E.C.; Holst, J.J.; Zoetendal, E.G.; Smidt, H.; Troost, F.; Schaap, F.G.; Damink, S.O.; Jocken, J.W.E.; et al. Effect of wheat bran derived prebiotic supplementation on gastrointestinal transit, gut microbiota, and metabolic health: A randomized controlled trial in healthy adults with a slow gut transit. Gut Microbes 2020, 12, 1704141. [Google Scholar] [CrossRef]
- Ganda Mall, J.P.; Löfvendahl, L.; Lindqvist, C.M.; Brummer, R.J.; Keita, Å.V.; Schoultz, I. Differential effects of dietary fibres on colonic barrier function in elderly individuals with gastrointestinal symptoms. Sci. Rep. 2018, 8, 13404. [Google Scholar] [CrossRef]
- Machado, A.M.; da Silva, N.B.M.; de Freitas, R.M.P.; de Freitas, M.B.D.; Chaves, J.B.P.; Oliveira, L.L.; Martino, H.S.D.; de Cássia Goncalves Alfenas, R. Effects of yacon flour associated with an energy restricted diet on intestinal permeability, fecal short chain fatty acids, oxidative stress and inflammation markers levels in adults with obesity or overweight: A randomized, double blind, placebo controlled clinical trial. Arch. Endocrinol. Metab. 2021, 64, 597–607. [Google Scholar]
- Pedersen, C.; Gallagher, E.; Horton, F.; Ellis, R.J.; Ijaz, U.Z.; Wu, H.; Jaiyeola, E.; Diribe, O.; Duparc, T.; Cani, P.D.; et al. Host–microbiome interactions in human type 2 diabetes following prebiotic fibre (galacto-oligosaccharide) intake. Br. J. Nutr. 2016, 116, 1869–1877. [Google Scholar] [CrossRef]
- Jain, P.K.; McNaught, C.E.; Anderson, A.D.G.; MacFie, J.; Mitchell, C.J. Influence of synbiotic containing Lactobacillus acidophilus La5, Bifidobacterium lactis Bb 12, Streptococcus thermophilus, Lactobacillus bulgaricus and oligofructose on gut barrier function and sepsis in critically ill patients: A randomised controlled trial. Clin. Nutr. 2004, 23, 467–475. [Google Scholar]
- Cao, S.; Shaw, E.L.; Quarles, W.R.; Sasaki, G.Y.; Dey, P.; Hodges, J.K.; Pokala, A.; Zeng, M.; Bruno, R.S. Daily inclusion of resistant starch-containing potatoes in a dietary guidelines for Americans dietary pattern does not adversely affect cardiometabolic risk or intestinal permeability in adults with metabolic syndrome: A randomized controlled trial. Nutrients 2022, 14, 1545. [Google Scholar] [CrossRef] [PubMed]
- Vaghef-Mehrabani, E.; Harouni, R.; Behrooz, M.; Ranjbar, F.; Asghari-Jafarabadi, M.; Ebrahimi-Mameghani, M. Effects of inulin supplementation on inflammatory biomarkers and clinical symptoms of women with obesity and depression on a calorie-restricted diet: A randomised controlled clinical trial. Br. J. Nutr. 2023, 129, 1897–1907. [Google Scholar] [CrossRef] [PubMed]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef]
- Sanders, M.E.; Merenstein, D.J.; Reid, G.; Gibson, G.R.; Rastall, R.A. Probiotics and prebiotics in intestinal health and disease: From biology to the clinic. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 605–616. [Google Scholar] [CrossRef] [PubMed]
- Gou, H.Z.; Zhang, Y.L.; Ren, L.F.; Li, Z.J.; Zhang, L. How do intestinal probiotics restore the intestinal barrier? Front. Microbiol. 2022, 13, 929346. [Google Scholar] [CrossRef]
- Isolauri, E.; Majamaa, H.; Arvola, T.; Rantala, I.; Virtanen, E.; Arvilommi, H. Lactobacillus casei strain GG reverses increased intestinal permeability induced by cow milk in suckling rats. Gastroenterology 1993, 105, 1643–1650. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, Y.; Sidhu, A.; Ma, Z.; McClain, C.; Feng, W. Lactobacillus rhamnosus GG culture supernatant ameliorates acute alcohol-induced intestinal permeability and liver injury. Am. J. Physiol. Gastrointest. Liver Physiol. 2012, 303, G32–G41. [Google Scholar] [CrossRef]
- Capurso, L. Thirty years of Lactobacillus rhamnosus GG: A review. J. Clin. Gastroenterol. 2019, 53 (Suppl. 1), S1–S41. [Google Scholar] [CrossRef] [PubMed]
- Ramezani Ahmadi, A.; Sadeghian, M.; Alipour, M.; Ahmadi Taheri, S.; Rahmani, S.; Abbasnezhad, A. The effects of probiotic/synbiotic on serum level of zonulin as a biomarker of intestinal permeability: A systematic review and meta-analysis. Iran J. Public Health 2020, 49, 1222–1231. [Google Scholar] [CrossRef]
- Stratiki, Z.; Costalos, C.; Sevastiadou, S.; Kastanidou, O.; Skouroliakou, M.; Giakoumatou, A.; Petrohilou, V. The effect of a bifidobacter supplemented bovine milk on intestinal permeability of preterm infants. Early Hum. Dev. 2007, 83, 575–579. [Google Scholar] [CrossRef]
- Sindhu, K.N.C.; Sowmyanarayanan, T.V.; Paul, A.; Babji, S.; Ajjampur, S.S.R.; Priyadarshini, S.; Sarkar, R.; Balasubramanian, K.A.; Wanke, C.A.; Ward, H.D.; et al. Immune response and intestinal permeability in children with acute gastroenteritis treated with Lactobacillus rhamnosus GG: A randomized, double-blind, placebo-controlled trial. Clin. Infect. Dis. 2014, 58, 1107–1115. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.; Andrew, H.; Kirschner, B.S.; Guandalini, S. Is Lactobacillus GG helpful in children with Crohn’s disease? Results of a preliminary, open-label study. J. Pediatr. Gastroenterol. Nutr. 2000, 31, 453–457. [Google Scholar]
- Rosenfeldt, V.; Benfeldt, E.; Valerius, N.H.; Paerregaard, A.; Fleischer Michaelsen, K. Effect of probiotics on gastrointestinal symptoms and small intestinal permeability in children with atopic dermatitis. J. Pediatr. 2004, 145, 612–616. [Google Scholar] [CrossRef]
- Sentongo, T.A.; Cohran, V.; Korff, S.; Sullivan, C.; Iyer, K.; Zheng, X. Intestinal permeability and effects of Lactobacillus rhamnosus therapy in children with short bowel syndrome. J. Pediatr. Gastroenterol. Nutr. 2008, 46, 41–47. [Google Scholar] [CrossRef]
- Gotteland, M.; Cruchet, S.; Verbeke, S. Effect of Lactobacillus ingestion on the gastrointestinal mucosal barrier alterations induced by indometacin in humans. Aliment. Pharmacol. Ther. 2001, 15, 11–17. [Google Scholar] [CrossRef]
- Montalto, M.; Gallo, A.; Curigliano, V.; D’Onofrio, F.; Santoro, L.; Covino, M.; Dalvai, S.; Gasbarrini, A.; Gasbrarrini, G. Clinical trial: The effects of a probiotic mixture on non-steroidal anti-inflammatory drug enteropathy—A randomized, double-blind, cross-over, placebo-controlled study. Aliment. Pharmacol. Ther. 2010, 32, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Akama, F.; Nishino, R.; Makino, S.; Kobayashi, K.; Kamikaseda, K.; Nagano, J.; Koga, Y. The effect of probiotics on gastric mucosal permeability in humans administered with aspirin. Scand. J. Gastroenterol. 2011, 46, 831–836. [Google Scholar] [CrossRef] [PubMed]
- Judkins, T.C.; Solch-Ottaiano, R.J.; Ceretto-Clark, B.; Nieves, C., Jr.; Colee, J.; Wang, Y.; Tompkins, T.A.; Caballero-Calero, S.E.; Langkamp-Henken, B. The effect of an acute aspirin challenge on intestinal permeability in healthy adults with and without prophylactic probiotic consumption: A double-blind, placebo-controlled, randomized trial. BMC Gastroenterol. 2024, 24, 4. [Google Scholar] [CrossRef]
- Garcia Vilela, E.; De Lourdes De Abreu Ferrari, M.; Da Gama Torres, H.O.; Gurerra Pinto, A.; Carneiro Aguirre, A.C.; Martins, F.P.; Andrade Goulart, E.M.; Da Cunha, A.S. Influence of Saccharomyces boulardii on the intestinal permeability of patients with Crohn’s disease in remission. Scand. J. Gastroenterol. 2008, 43, 842–848. [Google Scholar] [CrossRef]
- Wegh, C.A.M.; de Roos, N.M.; Hovenier, R.; Meijerink, J.; Besseling-van der Vaart, I.; van Hemert, S.; Witteman, B.J.M. Intestinal permeability measured by urinary sucrose excretion correlates with serum zonulin and faecal calprotectin concentrations in UC patients in remission. J. Nutr. Metab. 2019, 2019, 2472754. [Google Scholar] [CrossRef]
- Zeng, J.; Li, Y.-Q.; Zuo, X.-L.; Zhen, Y.-B.; Yang, J.; Liu, C.-H. Clinical trial: Effect of active lactic acid bacteria on mucosal barrier function in patients with diarrhoea-predominant irritable bowel syndrome. Aliment. Pharmacol. Ther. 2008, 28, 994–1002. [Google Scholar] [CrossRef] [PubMed]
- Bonfrate, L.; Di Palo, D.M.; Celano, G.; Algert, A.; Vitellio, P.; De Angelis, M.; Gobbetti, M.; Portincasa, P. Effects of Bifidobacterium longum BB536 and Lactobacillus rhamnosus HN001 in IBS patients. Eur. J. Clin. Investig. 2020, 50, e13201. [Google Scholar] [CrossRef] [PubMed]
- Boonma, P.; Shapiro, J.M.; Hollister, E.B.; Badu, S.; Wu, Q.; Weidler, E.M.; Abraham, B.P.; Devaraj, S.; Luna, R.A.; Versalovic, J.; et al. Probiotic VSL#3 treatment reduces colonic permeability and abdominal pain symptoms in patients with irritable bowel syndrome. Front. Pain Res. 2021, 2, 691689. [Google Scholar]
- Ait Abdellah, S.; Gal, C.; Laterza, L.; Velenza, V.; Settanni, C.R.; Napoli, M.; Schiavoni, E.; Mora, V.; Petito, V.; Gasbarrini, A. Effect of a multistrain probiotic on leaky gut in patients with diarrhea-predominant irritable bowel syndrome: A pilot study. Dig. Dis. 2023, 41, 489–499. [Google Scholar] [CrossRef]
- Marchix, J.; Quénéhervé, L.; Bordron, P.; Aubert, P.; Durand, T.; Oullier, T.; Blondeau, C.; Ait Abdellah, S.; Bruley des Varannes, S.; Chaffron, S.; et al. Could the microbiota be a predictive factor for the clinical response to probiotic supplementation in IBS-D? A cohort study. Microorganisms 2023, 11, 277. [Google Scholar] [CrossRef]
- Zhong, C.; Qu, C.; Wang, B.; Liang, S.; Zeng, B. Probiotics for preventing and treating small intestinal bacterial overgrowth: A meta-analysis and systematic review of current evidence. J. Clin. Gastroenterol. 2017, 51, 300–311. [Google Scholar] [CrossRef] [PubMed]
- Leber, B.; Tripolt, N.J.; Bladdl, D.; Eder, M.; Wascher TCPieber, T.R.; Stauber, R.; Sourij, H.; Oettl, K.; Stadlbauer, V. The influence of probiotic supplementation on gut permeability in patients with metabolic syndrome: An open label, randomized pilot study. Eur. J. Clin. Nutr. 2012, 66, 1110–1115. [Google Scholar] [CrossRef]
- DiMattia, Z.; Damani, J.J.; Van Syoc, E.; Rogers, C.J. Effect of probiotic supplementation on intestinal permeability in overweight and obesity: A systematic review of randomized controlled trials and animal studies. Adv. Nutr. 2024, 15, 100162. [Google Scholar] [CrossRef] [PubMed]
- Tandon, P.; Moncrief, K.; Madsen, K.; Arrieta, M.C.; Owen, R.J.; Bain, V.G.; Wong, W.W.; Ma, M.M. Effects of probiotic therapy on portal pressure in patients with cirrhosis: A pilot study. Liver Int. 2009, 29, 1110–1115. [Google Scholar] [CrossRef]
- Jayakumar, S.; Carbonneau, M.; Hotte, N.; Befus, A.D.; St Laurent, C.; Owen, R.; McCarthy, M.; Madsen, K.; Bailey, R.J.; Ma, M.; et al. VSL#3® probiotic therapy does not reduce portal pressures in patients with decompensated cirrhosis. Liver Int. 2013, 33, 1470–1477. [Google Scholar]
- Kwak, D.S.; Jun, D.W.; Seo, J.G.; Chung WSPark, S.-E.; Lee, K.N.; Khalid-Saeed, W.; Lee, H.L.; Lee, O.Y.; Yoon, B.C.; Choi, H.S. Short-term probiotic therapy alleviates small intestinal bacterial overgrowth, but does not improve intestinal permeability in chronic liver disease. Eur. J. Gastroenterol. Hepatol. 2014, 26, 1353–1359. [Google Scholar] [CrossRef] [PubMed]
- Horvath, A.; Leber, B.; Schmerboeck, B.; Tawdrous, M.; Zettel, G.; Hartl, A.; Madl, T.; Stryeck, S.; Fuchs, D.; Lemesch, S.; et al. Randomised clinical trial: The effects of a multispecies probiotic vs. placebo on innate immune function, bacterial translocation and gut permeability in patients with cirrhosis. Aliment. Pharmacol. Ther. 2016, 44, 926–935. [Google Scholar] [CrossRef] [PubMed]
- Horvath, A.; Durdevic, M.; Leber, B.; di Vora, K.; Rainer, F.; Krones, E.; Douschan, P.; Spindelboeck, W.; Durchschein, F.; Zollner, G.; et al. Changes in the intestinal microbiome during a multispecies probiotic intervention in compensated cirrhosis. Nutrients 2020, 12, 1874. [Google Scholar] [CrossRef] [PubMed]
- Ayob, N.; Nawawi, K.N.M.; Nor, M.H.M.; Ali, R.A.F.; Ahmad, H.F.; Oon, S.F.; Mokhtar, N.M. The effects of probiotics on small intestinal microbiota composition, inflammatory cytokines and intestinal permeability in patients with non-alcoholic fatty liver disease. Biomedicines 2023, 11, 640. [Google Scholar] [CrossRef]
- Qin, H.-L.; Zheng, J.-J.; Tong, D.-N.; Chen, W.-X.; Fan, X.-B.; Hang, X.-M.; Jiang, Y.-Q. Effect of Lactobacillus plantarum enteral feeding on the gut permeability and septic complications in the patients with acute pancreatitis. Eur. J. Clin. Nutr. 2008, 62, 923–930. [Google Scholar] [CrossRef] [PubMed]
- Besselink, M.G.; van Santvoort, H.C.; Renooij, W.; de Smet, M.B.; Boermeester, M.A.; Fischer, K.; Timmerman Hmali, U.A.; Cirkel, G.A.; Bollen, T.L.; van Ramshorst, B.; et al. Intestinal barrier dysfunction in a randomized trial of a specific probiotic composition in acute pancreatitis. Ann. Surg. 2009, 250, 712–719. [Google Scholar] [CrossRef]
- McNaught, C.E.; Woodcock, N.P.; Anderson, A.D.G.; MacFie, J. A prospective randomised trial of probiotics in critically ill patients. Clin. Nutr. 2005, 24, 211–219. [Google Scholar] [CrossRef]
- Alberda, C.; Gramlich, L.; Meddings, J.; Field, C.; McCargar, L.; Kutsogiannis, D.; Fedorak, R.; Madsen, K. Effects of probiotic therapy in critically ill patients: A randomized, double-blind, placebo-controlled trial. Am. J. Clin. Nutr. 2007, 85, 816–823. [Google Scholar] [CrossRef]
- Presti, R.M.; Yeh, E.; Williams, B.; Landay, A.; Jacobson, J.M.; Wilson, C.; Fichtenbaum, C.J.; Utay, N.S.; Dube, M.P.; Klingman, K.L.; et al. A randomized, placebo-controlled trial assessing the effect of VISBIOME ES probiotic in people with HIV on antiretroviral therapy. Open Forum Infect. Dis. 2021, 8, ofab550. [Google Scholar] [CrossRef]
- Mokkala, K.; Pussinen, P.; Houttu, N.; Koivuniemi, E.; Vahlberg, T.; Laitinen, K. The impact of probiotics and n-3 long-chain polyunsaturated fatty acids on intestinal permeability in pregnancy: A randomised clinical trial. Benef. Microbes 2018, 9, 199–208. [Google Scholar] [CrossRef]
- Chaiyasut, C.; Sivamaruthi, B.S.; Lailerd, N.; Sirilun, S.; Khongtan, S.; Fukngoen, P.; Peerajan, S.; Saelee, M.; Chaiyasut, K.; Kesika, P.; et al. Probiotics supplementation improves intestinal permeability, obesity index and metabolic biomarkers in elderly Thai subjects: A randomized controlled trial. Foods 2022, 11, 268. [Google Scholar] [CrossRef] [PubMed]
- de Roos, N.M.; van Hemert, S.; Rovers, J.M.P.; Smits, M.G.; Witteman, B.J.M. The effects of a multispecies probiotic on migraine and markers of intestinal permeability–results of a randomized placebo-controlled study. Eur. J. Clin. Nutr. 2017, 71, 1455–1462. [Google Scholar] [CrossRef] [PubMed]
- Swanson, K.S.; Gibson, G.R.; Hutkins, R.; Reimer, R.A.; Reid, G.; Verbeke, K.; Scott KPHolscher, H.D.; Azad, M.B.; Delzenne, N.M.; Sanders, M.E. The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of synbiotics. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 687–701. [Google Scholar] [CrossRef]
- Camilleri, M. Human intestinal barrier: Effects of stressors, diet, prebiotics, and probiotics. Clin. Transl. Gastroenterol. 2021, 12, e00308. [Google Scholar] [CrossRef] [PubMed]
- Reddy, B.S.; Macfie, J.; Gatt, M.; Larsen, C.N.; Jensen, S.S.; Leser, T.D. Randomized clinical trial of effect of synbiotics, neomycin and mechanical bowel preparation on intestinal barrier function in patients undergoing colectomy. Br. J. Surg. 2007, 94, 546–554. [Google Scholar] [CrossRef]
- Spindler-Vesel, A.; Bengmark, S.; Vovk, I.; Cerovic, O.; Kompan, L. Synbiotics, prebiotics, glutamine, or peptide in early enteral nutrition: A randomized study in trauma patients. J. Parenter. Enter. Nutr. 2007, 31, 119–126. [Google Scholar] [CrossRef]
- Sharma, B.; Srivastava, A.; Singh, N.; Sachdev, V.; Kapur, S.; Saraya, A. Role of probiotics on gut permeability and endotoxemia in patients with acute pancreatitis: A double-blind randomized controlled trial. J. Clin. Gastroenterol. 2011, 45, 442–448. [Google Scholar] [CrossRef]
- West, N.P.; Pyne, D.B.; Cripps, A.W.; Christophersen, C.T.; Conlon, M.A.; Fricker, P.A. Gut Balance, a synbiotic supplement, increases fecal Lactobacillus paracasei but has little effect on immunity in healthy physically active individuals. Gut Microbes 2012, 3, 221–227. [Google Scholar] [CrossRef]
- Yokoyama, Y.; Nishigaki, E.; Abe, T.; Fukaya, M.; Asahara, T.; Nomoto, K.; Nagino, M. Randomized clinical trial of the effect of perioperative synbiotics versus no synbiotics on bacterial translocation after oesophagectomy. Br. J. Surg. 2014, 101, 189–199. [Google Scholar] [CrossRef]
- Wilms, E.; Gerritsen, J.; Smidt, H.; Besseling-van der Vaart, I.; Rijkers, G.T.; Garcia Fuentes, A.R.; Masclee, A.A.M.; Troost, F.J. Effects of supplementation of the synbiotic Ecologic® 825/FOS P6 on intestinal barrier function in healthy humans: A randomized controlled trial. PLoS ONE 2016, 11, e0167775. [Google Scholar] [CrossRef]
- Stenman, L.K.; Lehtinen, M.J.; Meland, N.; Christensen, J.E.; Yeung, N.; Saarinen, M.T.; Courtney, M.; Burcelin, R.; Lähdeaho, M.-L.; Linros, J.; et al. Probiotic with or without fiber controls body fat mass, associated with serum zonulin, in overweight and obese adults-randomized controlled trial. EBioMedicine 2016, 13, 190–200. [Google Scholar] [CrossRef] [PubMed]
- Ferolla, S.M.; Couto, C.A.; Costa-Silva, L.; Armiliato, G.N.A.; Pereira, C.A.S.; Martins, F.S.; de Lourdes AFerrari, M.; Vilela, E.G.; Tores, H.O.G.; Cunha, A.S.; et al. Beneficial effect of synbiotic supplementation on hepatic steatosis and anthropometric parameters, but not on gut permeability in a population with nonalcoholic steatohepatitis. Nutrients 2016, 8, 397. [Google Scholar] [CrossRef] [PubMed]
- Moser, A.M.; Spindelboeck, W.; Halwachs, B.; Strohmaier, H.; Kump, P.; Gorkiewicz, G.; Högenauer, C. Effects of an oral synbiotic on the gastrointestinal immune system and microbiota in patients with diarrhea-predominant irritable bowel syndrome. Eur. J. Nutr. 2019, 58, 2767–2778. [Google Scholar] [CrossRef] [PubMed]
- Horvath, A.; Leber, B.; Feldbacher, N.; Steinwender, M.; Komarova, I.; Rainer, F.; Blesl, A.; Stadlbauer, V. The effects of a multispecies synbiotic on microbiome-related side effects of long-term proton pump inhibitor use: A pilot study. Sci. Rep. 2020, 10, 2723. [Google Scholar] [CrossRef]
- Horvath, A.; Leber, B.; Feldbacher, N.; Tripolt, N.; Rainer, F.; Blesl, A.; Trieb, M.; Marsche, G.; Sourij, H.; Stadlbauer, V. Effects of a multispecies synbiotic on glucose metabolism, lipid marker, gut microbiome composition, gut permeability, and quality of life in diabesity: A randomized, double-blind, placebo-controlled pilot study. Eur. J. Nutr. 2020, 59, 2969–2983. [Google Scholar] [CrossRef]
- Cosola, C.; Rocchetti, M.T.; di Bari, I.; Acquaviva, P.M.; Maranzano, V.; Corciulo, S.; Di Ciaula, A.; Di Palo, D.M.; La Forgia, F.M.; Fontana, S.; et al. An innovative synbiotic formulation decreases free serum indoxyl sulfate, small intestine permeability and ameliorates gastrointestinal symptoms in a randomized pilot trial in stage IIIb-IV CKD patients. Toxins 2021, 13, 334. [Google Scholar] [CrossRef]
- Ghavami, A.; Khorvash, F.; Heidari, Z.; Khalesi, S.; Askari, G. Effect of synbiotic supplementation on migraine characteristics and inflammatory biomarkers in women with migraine: Results of a randomized controlled trial. Pharmacol. Res. 2021, 169, 105668. [Google Scholar] [CrossRef]
- Liu, M.; Tandorost, A.; Moludi, J.; Dey, P. Prebiotics plus probiotics may favorably impact on gut permeability, endocannabinoid receptors, and inflammatory biomarkers in patients with coronary artery diseases: A clinical trial. Food Sci. Nutr. 2024, 12, 1207–1217. [Google Scholar] [CrossRef]
| Participants | Treatment/Duration/Method | Main Results | Ref. |
|---|---|---|---|
| 1015 healthy people randomly recruited; in vivo | Three-day food record; plasma LPS tested in 201 male participants. | Association between food intake (positively correlated with fat intake) and plasma LPS. | Amar et al., 2008 [43] |
| 20 healthy adults (mean age 25 ± 3.2 y); in vivo | Control diet (olive oil, 20%) HFD with omega-3 (fish oil, 35%) HFD with omega-6 (grape seed oil, 35%); diet rich in saturated fat (coconut oil, 35%); serum endotoxin concentration | Serum endotoxin concentration increased during postprandial period after consumption of a high-saturated-fat meal, but decreased after the meal with omega-3. | Lyte et al., 2016 [44] |
| 13 normal weight, sedentary adult males (mean age 22.2 ± 1.6 y); all weight-stable; in vivo | Two-week control diet [55% CHO, 30% fat (9% saturated), and 15% protein]; HFD for 5 days [55% fat (25% saturated fat), 30% CHO, and 15% protein]; HFM challenge [820 kcal: 25% CHO, 12% protein, 63% fat (~26% saturated fat)] pre- and post-5-day HFD; urine excretion of probe molecules. | No significant changes in gastroduodenal, small intestinal (p = 0.084 LM ratio), or colonic permeability following HFD. | Bowser et al., 2020 [45] |
| M-cell monolayers, human Peyer’s patches | Soluble NSP and food emulsifiers. Polysorbate-80 at 0.01% and 0.1%vol/vol E. coli translocation. | Polysorbate-80 increased E. coli translocation across M cells and human Peyer’s patches. | Roberts et al., 2010 [46] |
| Component | Preclinical Models | Effects in Preclinical Models | Reference |
|---|---|---|---|
| Dietary Component | |||
| Dietary Fiber |
| Supplementation reduced bodyweight gain, fat accumulation, insulin resistance, altered gut microbiota, lowered lactulose/mannitol ratio and serum zonulin, increased mucosal GLP-2; Similar effects on fecal output and SCFA excretion; HAW-R increased fecal butyrate. | Mistry et al., 2020 [47] Camilleri et al., 2022 [21] |
| Vitamin D |
| Increased susceptibility to bacteria and LPS, weakened tight junctions. | Yamamoto et al., 2020 [49] |
| Minerals: Zinc |
| Mixed evidence on potential to upregulate “leaky” junctions in some contexts; Reduced microbial diversity, impaired nutrient uptake. | Yamamoto et al., 2020 [49] Koren et al.,2020 [51] |
| Others: polyphenols, flavanones, anthocyanins |
| Altered gut microbiota, decreased microbes associated with weight and inflammation. | Abdulrahman et al., 2021 [53] |
| Arabinoxylans | Overweight and obesity | Effects on gut health detailed in the body of the text. | |
| Probiotics | Obesity; primary sclerosing cholangitis and IBD. | ||
| Synbiotics | Indomethacin-induced increased permeability; obesity. | ||
| Prebiotics | Non-obese diabetic mice; Western diet induced increased intestinal permeability in mice; aspirin-induced intestinal permeability in humans; children with type 1 diabetes mellitus; pre-term infants; burns. | ||
| Macronutrients | |||
| Protein | Protein-restricted diet or dexamethasone-induced permeability in chickens; Dietary meat and fish (and fat) in epidemiological studies of IBD. | Increased permeability and bacterial translocation; Increased risk of IBD. | Khoshbin et al., 2020 [42] |
| Sulfur-containing AAs: cysteine & methionine | Rats infected with Salmonella enteritidis; ischemia-reperfusion injury in rats; high-fat diet with methionine restriction in mice. | Effects on gut health detailed in Figure 1. | |
| Other AAs: Glutamine, L-tryptophan, Arginine |
| Improved ileum morphology, reduced inflammation, enhanced oxidative status, Both improved colitis symptoms, with Ala-Gln showing superior efficacy; Evidence suggests benefits in gut health and barrier function. | Xu et al., 2021 [54] Xu et al., 2021 [55] |
| Fat: | Fat-restricted diets. | Alterations in microbiota and further explanations in Table 1. | |
| Alcohol | Alcohol abuse and alcoholic liver disease. | Increased permeability and bacterial translocation. | Khoshbin et al., 2020 [42] |
| Intraluminal Emulsifiers | |||
| Endogenous bile acid | Mammalian colon | Detergent effects with increased permeability measured by PEG 400 and scanning EM. | Khoshbin et al., 2020 [42] |
| Dietary emulsifiers | Mouse colon permeability and inflammatory changes. | Effects on gut health detailed in the text. | |
| Receptors Associated with Intraluminal Factors | |||
| Aryl hydrocarbon receptor | Diverse animal models of immune diseases. | Anti-inflammatory | Khoshbin et al., 2020 [42] |
| Component | Human Diseases | Effects in Human Diseases | Reference |
|---|---|---|---|
| Dietary Component | |||
| Dietary Fiber | Effect on metabolic markers and gut health detailed in text. | Gondalia et al., 2022 [48] | |
| Vitamin D | In vivo study with high levels of active vitamin D. | Associated with gut bacteria producing butyrate, enhancing intestinal barrier; Mixed evidence on potential to upregulate “leaky” junctions in some contexts. | Thomas et al., 2020 [50] Yamamoto et al., 2020 [49] |
| Minerals: Zinc | Zinc deficiency linked to gastrointestinal disorders; appropriate dosing improves gut bacteria biodiversity and barrier integrity. | Increased oxidative stress, compromised barrier integrity. | Skalny et al., 2021 [52] |
| Others: polyphenols, flavanones, anthocyanins | Limited direct studies on permeability. | Polyphenols show general gut health benefits (see text). | |
| Macronutrients | |||
| Protein | Dietary meat and fish (and fat) in epidemiological studies of IBD. | Increased risk of IBD | Khoshbin et al., 2020 [42] |
| Other AAs: Glutamine, L-tryptophan, Arginine | Limited direct studies. | Evidence suggests benefits in gut health and barrier function | Xu et al., 2021 [55] |
| Fat: | Fat-restricted diets. | Alterations in microbiota and further explanations in Table 1 | Xu et al., 2021 [54] |
| Alcohol | Alcohol abuse and alcoholic liver disease. | Increased permeability and bacterial translocation | Khoshbin et al., 2020 [42] |
| Nutrient | Proposed Mechanisms | Factors That Decrease Permeability | Factors That Increase Permeability | References |
|---|---|---|---|---|
| Dietary fiber |
|
|
| [21,47,48,56,57,58,59,60,61,62] |
| Polyphenols |
|
|
| [53,63,64,65,66,67] |
| Glutamine |
|
|
| [54,55] |
| Vitamin D |
|
|
| [49,50,68] |
| Zinc |
|
|
| [51,52,69,70] |
| SCFAs |
|
|
| [58,59,60,61,62] |
| Sample | Treatment/Duration | Main Results | Ref. |
|---|---|---|---|
| Formula-fed infants and exclusively breast-fed infants | Formula supplementation with a prebiotic mixture of galacto-oligosaccharides and inulin for 10 weeks | Reduced IP (assessed using the lactulose-to-mannitol ratio) and improved microbiota profiles | Francavilla et al., 2006 [80] |
| Preterm infants | Enteral supplementation with a prebiotic mixture (1.5 g per kg per day of small-chain galacto-oligosaccharides, long-chain fructo-oligosaccharides, and nonhuman milk acidic oligosaccharides) for 30 days | No effect on IP (L:M ratio) | Westerbeek et al., 2010 [81] |
| Children with type 1 diabetes | 8 g of oligofructose-enriched inulin (chicory root) for three months | Reduced IP (L:M ratio), increased C-peptide levels, induced favorable changes in gut microbiota composition | Ho et al., 2019 [82] |
| Adult burn patients | 6 g of oligofructose for 15 days | No effect on IP (L:M ratio and sucrose excretion) | Olguin et al., 2005 [83] |
| Healthy young men | Inulin-enriched pasta for 5 weeks | Reduced IP (L:M ratio) and decreased serum levels of zonulin and GLP-2 | Russo et al., 2012 [84] |
| Adults with overweight or obesity | Arabinoxylan (7.5 or 15 g per day) for 6 weeks | No effect on IP (multi-sugar test), but upregulation of gene transcription of tight junction proteins, decreased microbiome diversity (though without compositional change), decreased fecal pH, increased fecal concentrations of total SCFAs, acetate, propionate and butyrate, decreased TNFα production | Salden et al., 2018 [85] |
| Adults with slow gastrointestinal transit without constipation | 15 g of wheat bran extract arabinoxylan–oligosaccharide (AXOS) for 12 weeks | No effect on IP (multi-sugar test), improved stool consistency, increased Bifidobacterium levels, no effect on whole-gut transit time or energy metabolism | Müller et al., 2020 [86] |
| Elderly adults | 12 g per day oat β-glucan or wheat arabinoxylan for 6 weeks | No effect on indomethacin-induced increase in IP (multi-sugar test) or microbiota composition | Ganda Mall et al., 2020 [87] |
| Adults with overweight or obesity | 25 g of yacon flour (rich in phenolic compounds and fructooligosaccharides) plus an energy-restricted diet for 6 weeks | No effect on IP (multi-sugar test), increased plasma antioxidant capacity, deceased oxidative stress markers, no effect on fecal short-chain fatty acids or inflammation markers | Machado et al., 2020 [88] |
| Adult men with type II diabetes | 5.5 g per day of galacto-oligosaccharides for 12 weeks | No effect on IP (by urinary recovery of oral 51Cr-EDTA), glucose tolerance, or microbial community structure | Pedersen et al., 2016 [89] |
| Adults at risk for type II diabetes | 10 g per day of inulin for 6 weeks | No effect on IP (multi-sugar test), reduced fasting insulin levels, improved HOMA-IR, increased fecal Bifidobacteria, no effect on plasma endotoxin concentrations or lipopolysaccharide-binding protein concentrations | Mitchell et al., 2021 [90] |
| Adults with metabolic syndrome | Dietary Guidelines for Americans (DGA)-based diet incorporating potatoes (17.5 g per day of resistant starch) for 2 weeks | Reduced small IP (L:M ratio) and postprandial endotoxemia, no changes in cardiometabolic markers | Cao et al., 2022 [91] |
| Women with obesity and major depressive disorder | 10 g per day of inulin | No effect on IP, depressive symptoms, inflammatory biomarkers, or weight | Vaghef-Mehrabani et al., 2023 [92] |
| Systematic review | Chicory inulin | Reduced IP | Nascimento et al., 2024 [3] |
| Sample | Treatment/Duration | Main Results | Ref. |
|---|---|---|---|
| Preterm Infants | Fortification of formula with Bifidobacter lactis (2 × 107 cfu/g of dry milk) for 30 days | Reduced IP (L:M ratio) and increased head growth | Stratiki et al., 2007 [100] |
| Children with rotavirus or cryptosporidial diarrhea | Culturelle Lacticaseibacillus rhamnosus GG (ATCC 53103) (1 × 1010 CFU) for 4 weeks | Reduced IP (L:M ratio) and improved clinical outcomes | Sindhu et al., 2014 [101] |
| Children with Crohn disease | Valio LGG (1 × 1010 CFU twice daily) for 6 months | Reduced IP (double sugar test) and improved Crohn disease severity scores | Gupta et al., 2000 [102] |
| Children with atopic dermatitis | Lactobacillus rhamnosus 19070-2 (1 × 1010 CFU) and L reuteri DSM 12246 (1 × 1010 CFU) for 6 weeks | Reduced IP (L:M ratio), improved gastrointestinal symptoms; association between improved permeability and reduced eczema. | Rosenfeldt et al., 2004 [103] |
| Children with short bowel syndrome | Culturelle LGG (1 × 109 CFU) for 4 weeks | No effect on IP (L:M ratio), fecal Lactobacillus colonization, or small intestinal bacterial overgrowth | Sentongo et al., 2008 [104] |
| Healthy adults | Dairy product containing live or heat-inactivated Lactobacillus GG (>107/mL), Lactobacillus helveticus (>107/mL), and Lactobacillus acidophilus (>107/mL) (2.4 × 109 CFU) for 10 days | Live but not heat-killed probiotics reduced indomethacin-induced alterations in GP (urinary excretion of sucrose and L:M ratio) but did not affect IP | Gotteland, Cruchet, and Verbeke 2008 [105] |
| Healthy adults | De Simone formulation probiotic (900 billion bacteria per day) for 21 days | Reduced fecal calprotectin concentrations during indomethacin therapy | Montalto et al., 2010 [106] |
| Healthy adults | Yogurt enriched with Lactobacillus gasseri OLL2716 (1 × 109 CFU) for 16 weeks | Reduced IP (urinary sucrose excretion) and the likelihood of a positive fecal occult blood test after both high and low doses of aspirin | Akama et al., 2011 [107] |
| Healthy adults | Lallemand Lacticaseibacillus rhamnosus R0011 and Lactobacillus helveticus R0052 (at least 4 billion CFU) | Did not prevent the increase in gastroduodenal permeability (urinary sucrose excretion) induced by an acute aspirin challenge | Judkins et al., 2024 [108] |
| Adults with Crohn disease in remission | Floratil Saccharomyces boulardii (4 × 108 CFU 3 times per day) for three months | Reduced IP (L:M ratio), although did not normalize it | Garcia Vilela et al., 2008 [109] |
| Adults with ulcerative colitis in remission | Ecologic 825 formulation (1.5 × 1010 CFU) for 12 weeks | No effect on IP (multi-sugar test), serum zonulin, fecal zonulin, inflammation markers, or stool characteristics | Wegh et al., 2019 [110] |
| Adults with diarrhea-predominant IBS | Probiotic fermented milk for 4 weeks | Reduced small IP (L:M ratio) and reduced IBS symptoms, though did not affect colonic permeability (sucralose excretion) | Zeng et al., 2008 [111] |
| Adults with IBS | Zircombi Bifidobacterium longum BB536 (4 billion CFU), Lactobacillus rhamnosus HN001 (1 billion CFU), and vitamin B6 for 30 days | Reduced IP (sucralose excretion), improved IBS symptoms, and favorably modified the gut microbiota and metabolome | Bonfrate et al., 2020 [112] |
| Adults with IBS | De Simone formulation (900 billion CFU) for 4 or 8 weeks | Reduced IP (multi-sugar test) and improved IBS symptoms | Boonma et al., 2021 [113] |
| Adults with IBS-D and confirmed increased permeability | Lactibiane Tolérance formulation (1 × 1010 CFU twice per day) for 30 days | Reduced (81.5%) or normalized (37%) IP (radionucleotide tracers) and improved IBS symptoms, including abdominal pain and stool consistency, while also enhancing QOL | Ait Abdellah et al., 2023 [114] |
| Adults with IBS-D | Lactibiane Tolérance formulation (10 × 109 CFU twice daily) for 4 weeks | While IP (urinary lactulose excretion) remained unchanged, the probiotic improved IBS symptoms, QOL, and gut microbiota. In responders with reduced diarrhea, transcellular permeability significantly decreased, and paracellular permeability trended toward a decrease, though overall permeability showed no significant change. | Marchix et al., 2023 [115] |
| Meta-analysis of patients with small intestinal bacterial overgrowth | Various | No effect on intestinal permeability | Zhong et al., 2017 [116] |
| Adults with metabolic syndrome | Yakult (milk beverage fortified with 108 CFU/mL Lactobacillus casei Shirota) three bottles per day for 3 months | No effect on intestinal permeability (sugar absorption test and diaminooxidase serum levels), endotoxin levels, or neutrophil function | Leber et al., 2012 [117] |
| Systematic review of 12 animal and 14 clinical studies in obesity | Various, especially with Bifidobacterium, Lactobacillus, and Akkermansia | Often reduces IP in obesity, although clinical results are inconsistent | DiMattia et al., 2014 [118] |
| Adult patients with cirrhosis and a hepatic venous pressure gradient > 10 | De Simone formulation (3600 billion bacteria daily) for 2 months | No effect on IP (L:M ratio, sucrose and sucralose tests) or portal pressure | Tandon et al., 2009 [119] |
| No effect on IP (endotoxin levels), cytokine levels, microbiota or HVPG, but decrease in aldosterone | Jayakumar et al., 2013 [120] | ||
| Adults with chronic liver disease | Duolac Gold formulation (5 × 109 CFU twice daily) for 4 weeks | No effect on IP (L:M ratio), alleviated SIBO, improved GI symptoms | Kwak et al., 2014 [121] |
| Adults with cirrhosis primary and secondary analysis | Ecologic Barrier formulation (1.5 × 1010 CFU per day) for 6 months | No effect on IP (L:M ratio) or bacterial translocation, but did improve some immune and liver function, seen by increased serum neopterin levels and enhanced neutrophil reactive oxygen species production | Horvath et al., 2016 [122] |
| Reduced IP (increased neopterin and decreased fecal zonulin), enriched the gut microbiota, and increased levels of beneficial bacteria | Horvath et al., 2020 [123] | ||
| Adults with metabolic dysfunction-associated steatotic liver disease | HEXBIO Microbial Cell Preparation (30 billion CFU twice daily) for 6 months | No effect on IP (zonulin and zonula occluden-1 (ZO-1) levels) or gut microbial diversity, although did induce changes in microbiota composition and decreases in IFN-γ and TNF-α | Ayob et al., 2023 [124] |
| Adults with acute pancreatitis | Enteral feeding supplemented with Lactobacillus plantarum (1 × 108 CFU) for 7 days | Reduced IP (L:M ratio) and improved clinical outcomes including reduced colonization by pathogenic organisms and fewer septic complications | Qin et al., 2008 [125] |
| Adults with severe acute pancreatitis | Ecologic 641 formulation (1 × 1010 CFU twice daily) for 28 days | No effect on IP (PEG permeability test), increased enterocyte damage and bacterial translocation among those with organ failure (a subsequent study showed increased mortality with this probiotic in acute pancreatitis) [122] | Besselink et al., 2009 [126] |
| Critically ill adults | Proviva oatmeal and fruit drink containing 5 × 107 CFU/mL of Lactobacillus plantarum 299v 500 mL per day until hospital discharge or patient preference to discontinue | No effect on IP L:M ratio), gastric microbiota composition, endotoxin levels, sepsis morbidity or mortality, but reduced serum IL-6 levels | McNaught et al., 2005 [127] |
| Critically ill adults | De Simone formulation 900 billion CFU twice daily or inactivated probiotic sonicates | No effect on IP (L:M ratio), but increased systemic IgA and IgG in live but not dead bacteria group | Alberda et al., 2007 [128] |
| Adults with HIV on antiretroviral therapy | De Simone formulation 900 billion CFU once daily for 4 weeks followed by twice daily to 26 weeks | No effect on IP (assessed by colonic biopsies measuring CD4+ cells, IL-17+ cells, and myeloperoxidase), systemic inflammation, or cellular markers of inflammation, although it did alter the microbiome by increasing beneficial species and reducing Gammaproteobacteria | Presti et al., 2021 [129] |
| Pregnant women with overweight | Bifidobacterium animalis ssp. lactis 420 (1 × 1010 CFU) and Lactobacillus rhamnosus HN00 (1 × 1010 CFU) and/or LC-PUFA for 21 weeks (on average) | No protective effect on the increase in IP during pregnancy (serum zonulin and LPS) | Mokkala et al., 2018 [130] |
| Elderly adults | Lactobacillus paracasei HII01 (2 × 1010 CFU), Bifidobacterium breve (2 × 1010 CFU), and Bifidobacterium longum (1 × 1010 CFU) | Reduced IP (assessed using a colorimetric urine analysis), enhanced HDL-cholesterol levels, and positively affected obesity-related biomarkers and short-chain fatty acids | Chaiyasut et al., 2022 [131] |
| Adults with migraine | Ecologic barrier formulation 5 × 109 CFU daily for 12 weeks | No effect on IP (L:M ratio and fecal and serum zonulin levels) or migraine severity or frequency | de Roos et al., 2017 [132] |
| Sample | Treatment/Duration | Main Results | Ref. |
|---|---|---|---|
| Critically ill adults | Trevis probiotic (4 × 109 of Lactobacillus acidophilus La5, Bifidobacterium lactis Bb 12, Streptococcus thermophilus, Lactobacillus bulgaricus) plus 7.5 g of oligofructose twice daily for 8 days | No effect on IP (L:M ratio), septic complications, or mortality, but reduced the incidence of potentially pathogenic bacteria in nasogastric aspirates. | Jain et al., 2004 [90] |
| Adults undergoing elective colorectal surgery | Mechanical bowel preparation, neomycin, and Trevis probiotic three times per day plus 15 g oligofructose twice daily | No effect on small IP (L:M ratio), whole gut permeability (sucralose excretion), inflammatory response, or sepsis morbidity, but there was significantly reduced fecal Enterobacteriaceae and bacterial translocation. | Reddy et al., 2007 [135] |
| Adult critically ill trauma patients | Synbiotic 2000 (4 × 1010 CFU) plus 2.5 g of each of β glucan, inulin, pectin, and resistant starch for 7 days | Reduced IP (L:M ratio) and incidence of infections, including pneumonia. | Spindler-Vesel et al., 2007 [136] |
| Adults with acute pancreatitis | 2.5 billion CFU of Lactobacillus acidophilus, Bifidobacterium longus, Bifidobacterium bifidum, and Bifidobacterium infantalis with 25 mg of fructo-oligosaccharide for 7 days | No effect on IP (L:M ratio), endotoxemia, duration of hospital or intensive care unit stay, or mortality, but decreased levels of C-reactive protein and immunoglobulins. | Sharma et al., 2011 [137] |
| Healthy adults | Biosource Gut Balance (2 × 109 CFU) plus 200 mg immunoglobulin, 50 mg lactoferrin, 90 mg raftiline, and 10 mg raftilose ) for 21 days | No effect on IP (uncertain how this was assessed apart from some measure via fecal and urine samples), fecal SCFAs, or markers of mucosal immunity. | West et al., 2012 [138] |
| Adults with esophageal cancer undergoing esophagectomy | Yakult 400 (at least 4 × 1010 Lactobacillus casei Shirota) Yakult Honsha (at least 1 × 1010 Bifidobacterium breve Yakult), and 15 g galacto-oligosaccharides for 7 days following surgery | Reduced IP (indirectly assessed via reduced bacterial translocation to mesenteric lymph nodes and reduced incidence of postoperative bacteremia). | Yokoyama et al., 2014 [139] |
| Healthy adults | Ecologic 825 (6 g per day) combined with 10 g per day fructooligosaccharides for 2 weeks | No effect on IP (multi-sugar test) at baseline or after indomethacin, though stool frequency increased. | Wilms et al., 2016 [140] |
| Adults with overweight | Bifidobacterium animalis ssp. lactis 420 B420 1 × 1010 CFU per day combined with 12 g of ultra polydextrose for 6 months | Reduced effect on IP (plasma zonulin levels; reduction in zonulin correlated with reductions in trunk fat mass), reduced body fat mass, waist circumference, and energy intake, increased fecal SCFAs. | Stenman et al., 2016 [141] |
| Adults with MASLD | Nestle Fiber Mais Flora Lactobacillus reuteri 1 × 108 CFU plus 4 g of guar gum and inulin twice daily combined with nutritional counseling for three months | No effect on IP (L:M ratio) or LPS levels, but reduced hepatic steatosis, weight, BMI, and waist circumference. | Ferolla et al., 2016 [142] |
| Adults with IBS-D | OMNi-BiOTiC Stress Repair (6 × 1010 CFU) plus corn starch, maltodextrin, inulin, fructooligosaccharides, and amylases) for 4 weeks | Reduced IP (fecal zonulin levels), improved IBS symptoms, increased microbial diversity in the upper GI tract, reduced CD4+ T cells in the ascending colon, and elevated fecal SCFAs. | Moser et al., 2018 [143] |
| Adults on long-term proton pump inhibitor therapy | OMNi-BiOTiC PPI (24 × 109 CFU) in a prebiotic matrix (composed of corn starch, maltodextrin, fructo-oligosaccharide P6, inulin P2, and vegetable protein) for 3 months | Reduced IP (reflected by a reduction in fecal zonulin) in those with increased permeability at baseline, which was maintained three months post-intervention; no significant changes were observed in other markers of IP, including lipopolysaccharide, lipopolysaccharide-binding protein, and sCD14 levels. | Horvath et al., 2020 [144] |
| Adults with obesity and type II diabetes | Ecologic barrier formulation with fructo-oligosaccharides P6, and konjac glucomannan P13 for 6 months | Reduced IP (serum zonulin), lipoprotein (a), and hip circumference, with no changes in glucose metabolism. | Horvath et al., 2020 [145] |
| Adults with stage III-IV chronic kidney disease | NATUREN G (4.8 × 109 CFU), 2.5 g fructoligosaccharides, 2.5 g inulin, and antioxidants quercetin, resveratrol, and proanthocyanidins for 2 months | Reduced small IP (assessed using urinary recovery of oligosaccharide probes after oral administration), alleviated GI symptoms, and reduced free serum indoxyl sulfate.. | Cosola et al., 2021 [146] |
| Adult women with migraine | Familact (1 × 109 CFU) plus 21 mg fructooligosaccharides for 12 weeks | Reduced IP (serum zonulin), frequency of migraine attacks, painkiller use, GI symptoms, and CRP. | Ghavami et al., 2021 [147] |
| Adults with coronary artery disease | Inulin 15 mg or LGG 1.9 × 109 CFU or their combination for 60 days | Reduced IP (LPS), IL-6, and TLR-4 levels while increasing serum total antioxidant capacity. | Liu et al., 2024 [148] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matar, A.; Damianos, J.A.; Jencks, K.J.; Camilleri, M. Intestinal Barrier Impairment, Preservation, and Repair: An Update. Nutrients 2024, 16, 3494. https://doi.org/10.3390/nu16203494
Matar A, Damianos JA, Jencks KJ, Camilleri M. Intestinal Barrier Impairment, Preservation, and Repair: An Update. Nutrients. 2024; 16(20):3494. https://doi.org/10.3390/nu16203494
Chicago/Turabian StyleMatar, Ayah, John A. Damianos, Kara J. Jencks, and Michael Camilleri. 2024. "Intestinal Barrier Impairment, Preservation, and Repair: An Update" Nutrients 16, no. 20: 3494. https://doi.org/10.3390/nu16203494
APA StyleMatar, A., Damianos, J. A., Jencks, K. J., & Camilleri, M. (2024). Intestinal Barrier Impairment, Preservation, and Repair: An Update. Nutrients, 16(20), 3494. https://doi.org/10.3390/nu16203494






