The Effect of Maternal High-Fat Diet on Adipose Tissue Histology and Lipid Metabolism-Related Genes Expression in Offspring Rats
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Body Weight Measurements
2.3. Blood Sample Collection and Evaluation of Biochemical Parameters
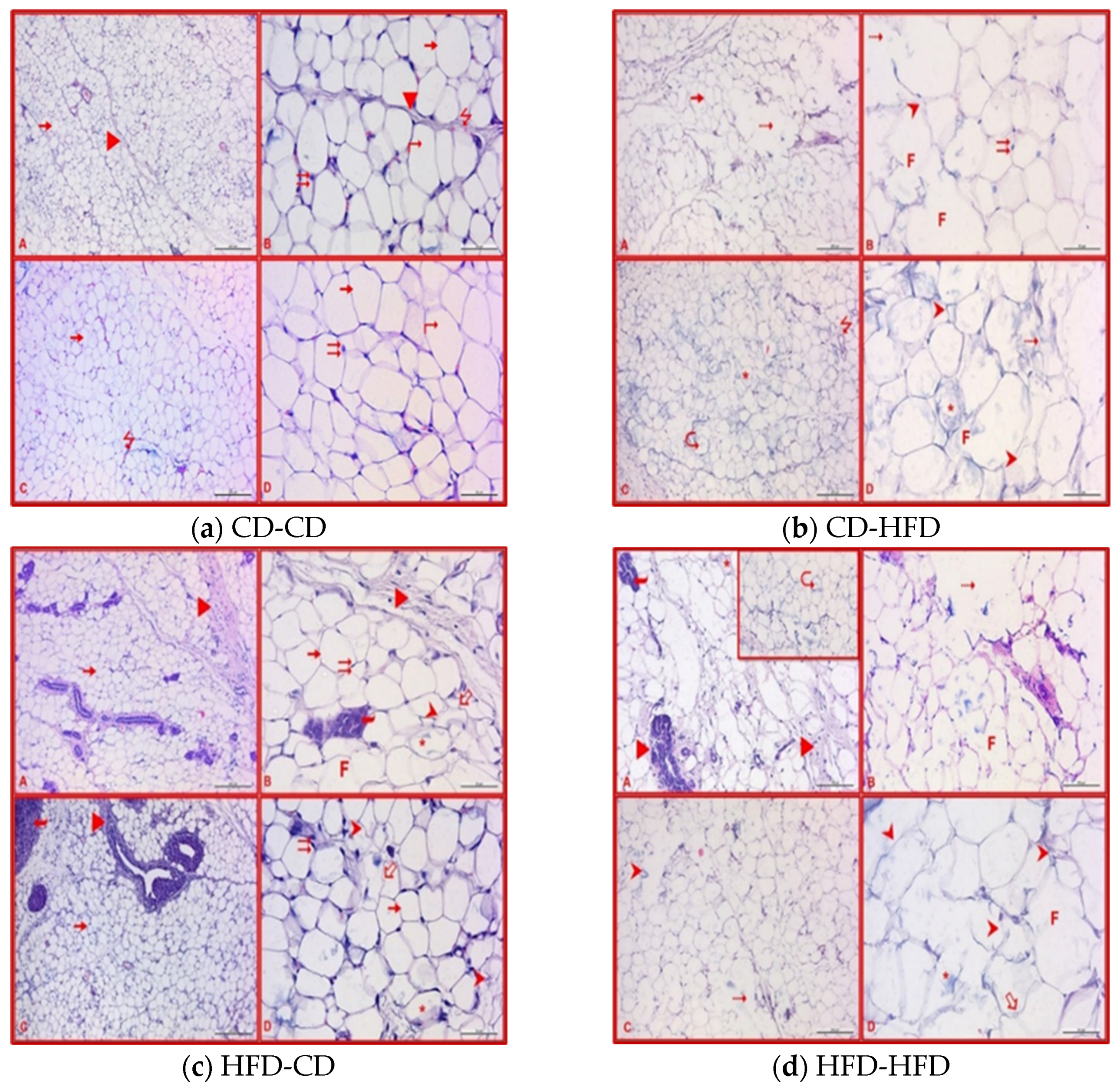
2.4. Histological Evaluation of Adipose Tissue of Offspring
2.5. Evaluation of Gene Expression Analyses of Offspring
2.6. Statistical Analysis
3. Results
3.1. Body Weights of Offspring Rats and Evaluation of Body Weight Changes
3.2. Evaluation of Biochemical Parameters in Offspring Rats
3.3. Evaluation of Adipose Tissues in Offspring Rats
3.4. Effects of Maternal Diet on Gene Expression in Offspring Rats
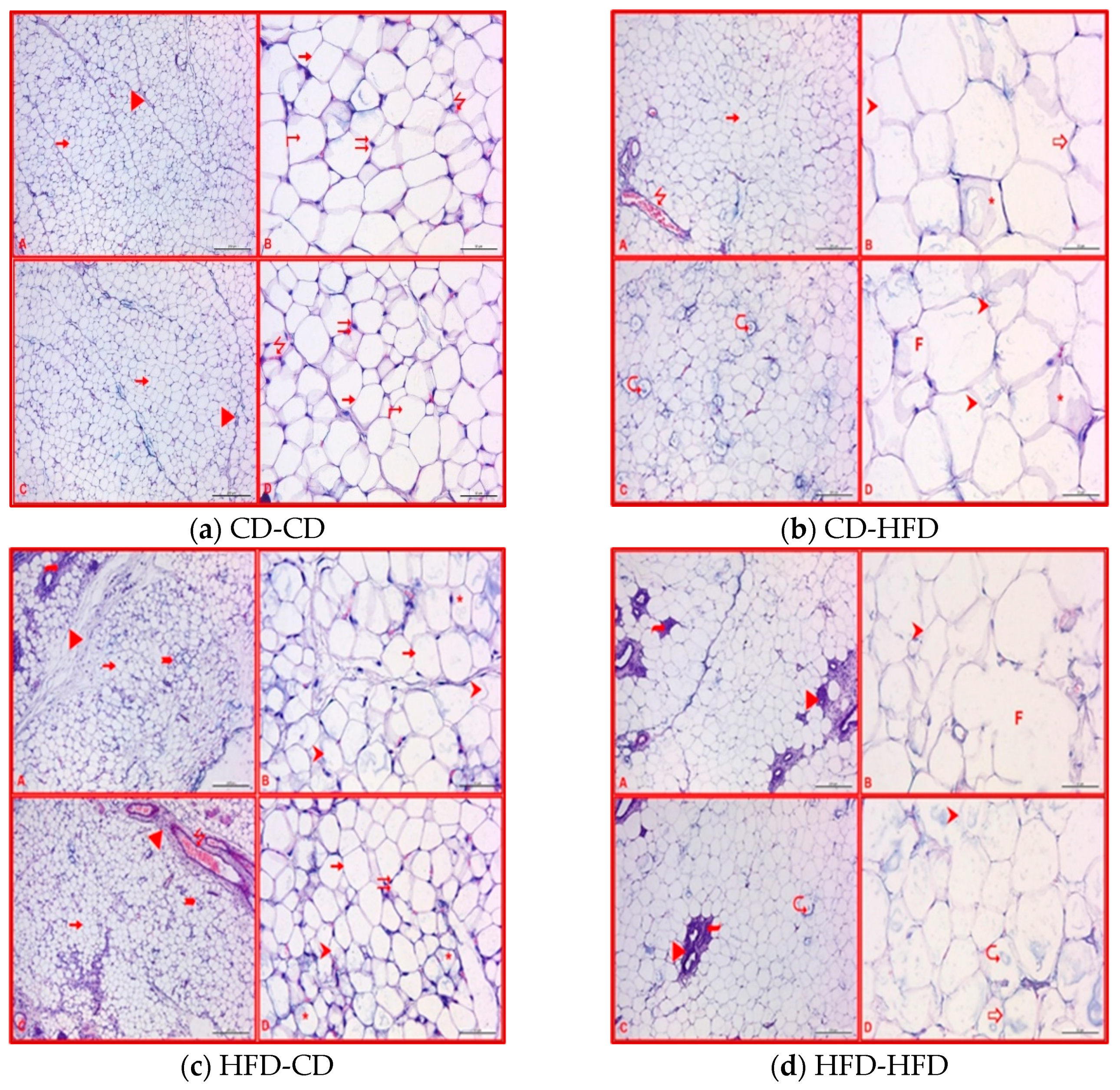
3.5. Maternal-Diet-Related Histological Analyses in Adipose Tissues of Offspring Rats
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- WHO. Obesity and Overweight. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 17 November 2023).
- Hacettepe University Institute of Population Studies. 2018 Turkey Demographic and Health Survey. Hacettepe University Institute of Population Studies, T.R. Presidency of Turkey Directorate of Strategy and Budget and TÜBİTAK, Ankara, Turkey. 2019. Available online: https://fs.hacettepe.edu.tr/hips/dosyalar/Ara%C5%9Ft%C4%B1rmalar%20-%20raporlar/2018%20TNSA/TDHS2018_mainReport_compressed.pdf (accessed on 29 November 2023).
- Turkey Nutrition and Health Survey (TNHS). General Directorate of Public Health, Ministry of Health, Republic of Turkey, Ankara. 2019. Available online: https://hsgm.saglik.gov.tr/depo/birimler/saglikli-beslenme-ve-hareketli-hayat-db/Dokumanlar/Ingilizce_Yayinlar/TBSA_RAPOR_KITAP_2017_ENG_.pdf (accessed on 29 November 2023).
- Lowensohn, R.I.; Stadler, D.D.; Naze, C. Current concepts of maternal nutrition. Obstet. Gynecol. Surv. 2016, 71, 413. [Google Scholar] [CrossRef] [PubMed]
- Alexandre-Gouabau, M.-C.; David-Sochard, A.; Royer, A.-L.; Parnet, P.; Paillé, V. Moderate high caloric maternal diet impacts dam breast milk metabotype and offspring lipidome in a sex-specific manner. Int. J. Mol. Sci. 2020, 21, 5428. [Google Scholar] [CrossRef] [PubMed]
- Turkey Childhood (Primary School, 2nd Grade Students) Obesity Surveillance Initiative COSI-TUR 2016. Ministry of Health—General Directorate of Public Health, Ministry of National Education, World Health Organization Regional Office for Europe, Ministry of Health Publication No: 1126 Ankara. 2019. Available online: https://hsgm.saglik.gov.tr/depo/birimler/saglikli-beslenme-ve-hareketli-hayat-db/Dokumanlar/Ingilizce_Yayinlar/Turkey_Childhood_Obesity_Cosi_tur_2016_Basic_Findings.pdf (accessed on 29 November 2023).
- Turkey Childhood (Ages 7–8) Obesity Surveillance Initiative (COSI-TUR) Republic of Turkey Ministry of Health, Republic of MEB, Hacettepe University, 922, Ankara. 2014. Available online: https://hsgm.saglik.gov.tr/depo/birimler/saglikli-beslenme-ve-hareketli-hayat-db/Dokumanlar/Ingilizce_Yayinlar/COSI_TUR_2013.pdf (accessed on 29 November 2023).
- WHO. WHO European Regional Obesity Report 2022; WHO Regional Office for Europe: Copenhagen, Denmark, 2022; Available online: https://iris.who.int/bitstream/handle/10665/353747/9789289057738-eng.pdf?sequence=1 (accessed on 29 November 2023).
- Barker, D.J. In utero programming of chronic disease. Clin. Sci. (Lond. Engl. 1979) 1998, 95, 115–128. [Google Scholar] [CrossRef]
- Vanhees, K.; Vonhögen, I.G.; van Schooten, F.J.; Godschalk, R.W. You are what you eat, and so are your children: The impact of micronutrients on the epigenetic programming of offspring. Cell. Mol. Life Sci. 2014, 71, 271–285. [Google Scholar] [CrossRef] [PubMed]
- Masuyama, H.; Hiramatsu, Y. Effects of a high-fat diet exposure in utero on the metabolic syndrome-like phenomenon in mouse offspring through epigenetic changes in adipocytokine gene expression. Endocrinology 2012, 153, 2823–2830. [Google Scholar] [CrossRef] [PubMed]
- Desai, M.; Jellyman, J.K.; Han, G.; Beall, M.; Lane, R.H.; Ross, M.G. Maternal obesity and high-fat diet program offspring metabolic syndrome. Am. J. Obstet. Gynecol. 2014, 211, 237.e1–237.e13. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Simar, D.; Lambert, K.; Mercier, J.; Morris, M.J. Maternal and postnatal overnutrition differentially impact appetite regulators and fuel metabolism. Endocrinology 2008, 149, 5348–5356. [Google Scholar] [CrossRef]
- Rajia, S.; Chen, H.; Morris, M.J. Maternal overnutrition impacts offspring adiposity and brain appetite markers-modulation by postweaning diet. J. Neuroendocrinol. 2010, 22, 905–914. [Google Scholar] [CrossRef]
- Moreno-Mendez, E.; Quintero-Fabian, S.; Fernandez-Mejia, C.; Lazo-de-la-Vega, M.-L. Early-life programming of adipose tissue. Nutr. Res. Rev. 2020, 33, 244–259. [Google Scholar] [CrossRef]
- Benkalfat, N.B.; Merzouk, H.; Bouanane, S.; Merzouk, S.-A.; Bellenger, J.; Gresti, J.; Tessier, C.; Narce, M. Altered adipose tissue metabolism in offspring of dietary obese rat dams. Clin. Sci. 2011, 121, 19–28. [Google Scholar] [CrossRef]
- Chechi, K. Dietary Fat Intake during Pre-And Post-Weaning Time Period and Its Association with the Onset of Cardiovascular diseas in the Offspring. Ph.D. Thesis, Memorial University of Newfoundland, St. John’s, NL, Canada, 2010. [Google Scholar]
- Clarke, S.D. Regulation of fatty acid synthase gene expression: An approach for reducing fat accumulation. J. Anim. Sci. 1993, 71, 1957–1965. [Google Scholar] [CrossRef] [PubMed]
- Cerf, M.E.; Herrera, E. High fat diet administration during specific periods of pregnancy alters maternal fatty acid profiles in the near-term rat. Nutrients 2016, 8, 25. [Google Scholar] [CrossRef] [PubMed]
- Desai, M.; Ross, M.G. Maternal-infant nutrition and development programming of offspring appetite and obesity. Nutr. Rev. 2020, 78, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Desai, M.; Ferrini, M.G.; Han, G.; Narwani, K.; Ross, M.G. Maternal high fat diet programs male mice offspring hyperphagia and obesity: Mechanism of increased appetite neurons via altered neurogenic factors and nutrient sensor AMPK. Nutrients 2020, 12, 3326. [Google Scholar] [CrossRef] [PubMed]
- Bernardis, L.; Patterson, B. Correlation between ‘Lee index’ and carcass fat content in weanling and adult female rats with hypothalamic lesions. J. Endocrinol. 1968, 40, 527–528. [Google Scholar] [CrossRef] [PubMed]
- Ellsworth, L.; Harman, E.; Padmanabhan, V.; Gregg, B. Lactational programming of glucose homeostasis: A window of opportunity. Reproduction 2018, 156, R23–R42. [Google Scholar] [CrossRef] [PubMed]
- Daniel, Z.C.; Akyol, A.; McMullen, S.; Langley-Evans, S.C. Exposure of neonatal rats to maternal cafeteria feeding during suckling alters hepatic gene expression and DNA methylation in the insulin signalling pathway. Genes Nutr. 2014, 9, 365. [Google Scholar] [CrossRef]
- Akyol, A.; McMullen, S.; Langley-Evans, S.C. Glucose intolerance associated with early-life exposure to maternal cafeteria feeding is dependent upon post-weaning diet. Br. J. Nutr. 2012, 107, 964–978. [Google Scholar] [CrossRef]
- Neri, C.; Edlow, A.G. Effects of maternal obesity on fetal programming: Molecular approaches. Cold Spring Harb. Perspect. Med. 2016, 6, a026591. [Google Scholar] [CrossRef]
- Lecoutre, S.; Deracinois, B.; Laborie, C.; Eberlé, D.; Guinez, C.; Panchenko, P.E.; Lesage, J.; Vieau, D.; Junien, C.; Gabory, A. Depot-and sex-specific effects of maternal obesity in offspring’s adipose tissue. J. Endocrinol. 2016, 230, 39–53. [Google Scholar] [CrossRef]
- Christians, J.K.; Lennie, K.I.; Wild, L.K.; Garcha, R. Effects of high-fat diets on fetal growth in rodents: A systematic review. Reprod. Biol. Endocrinol. 2019, 17, 39. [Google Scholar] [CrossRef] [PubMed]
- Ribaroff, G.; Wastnedge, E.; Drake, A.; Sharpe, R.; Chambers, T. Animal models of maternal high fat diet exposure and effects on metabolism in offspring: A meta-regression analysis. Obes. Rev. 2017, 18, 673–686. [Google Scholar] [CrossRef] [PubMed]
- Masuyama, H.; Hiramatsu, Y. Additive effects of maternal high fat diet during lactation on mouse offspring. PLoS ONE 2014, 9, e92805. [Google Scholar] [CrossRef] [PubMed]
- Barrand, S.; Crowley, T.M.; Wood-Bradley, R.J.; De Jong, K.A.; Armitage, J.A. Impact of maternal high fat diet on hypothalamic transcriptome in neonatal Sprague Dawley rats. PLoS ONE 2017, 12, e0189492. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.; Yang, Q.; Zhang, L.; Maricelli, J.W.; Rodgers, B.D.; Zhu, M.-J.; Du, M. Maternal high-fat diet during lactation impairs thermogenic function of brown adipose tissue in offspring mice. Sci. Rep. 2016, 6, 34345. [Google Scholar] [CrossRef] [PubMed]
- George, G.; Draycott, S.A.; Muir, R.; Clifford, B.; Elmes, M.J.; Langley-Evans, S.C. Exposure to maternal obesity during suckling outweighs in utero exposure in programming for post-weaning adiposity and insulin resistance in rats. Sci. Rep. 2019, 9, 10134. [Google Scholar] [CrossRef] [PubMed]
- Purcell, R.H.; Sun, B.; Pass, L.L.; Power, M.L.; Moran, T.H.; Tamashiro, K.L. Maternal stress and high-fat diet effect on maternal behavior, milk composition, and pup ingestive behavior. Physiol. Behav. 2011, 104, 474–479. [Google Scholar] [CrossRef]
- Samuelsson, A.-M.; Matthews, P.A.; Argenton, M.; Christie, M.R.; McConnell, J.M.; Jansen, E.H.; Piersma, A.H.; Ozanne, S.E.; Twinn, D.F.; Remacle, C. Diet-induced obesity in female mice leads to offspring hyperphagia, adiposity, hypertension, and insulin resistance: A novel murine model of developmental programming. Hypertension 2008, 51, 383–392. [Google Scholar] [CrossRef]
- Tsuduki, T.; Kitano, Y.; Honma, T.; Kijima, R.; Ikeda, I. High dietary fat intake during lactation promotes development of diet-induced obesity in male offspring of mice. J. Nutr. Sci. Vitaminol. 2013, 59, 384–392. [Google Scholar] [CrossRef]
- Lecoutre, S.; Breton, C. Maternal nutritional manipulations program adipose tissue dysfunction in offspring. Front. Physiol. 2015, 6, 158. [Google Scholar] [CrossRef]
- Lukaszewski, M.-A.; Eberlé, D.; Vieau, D.; Breton, C. Nutritional manipulations in the perinatal period program adipose tissue in offspring. Am. J. Physiol.-Endocrinol. Metab. 2013, 305, E1195–E1207. [Google Scholar] [CrossRef] [PubMed]
- Murabayashi, N.; Sugiyama, T.; Zhang, L.; Kamimoto, Y.; Umekawa, T.; Ma, N.; Sagawa, N. Maternal high-fat diets cause insulin resistance through inflammatory changes in fetal adipose tissue. Eur. J. Obstet. Gynecol. Reprod. Biol. 2013, 169, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Gawlińska, K.; Gawliński, D.; Filip, M.; Przegaliński, E. Relationship of maternal high-fat diet during pregnancy and lactation to offspring health. Nutr. Rev. 2021, 79, 709–725. [Google Scholar] [CrossRef] [PubMed]
- Saad, S.; Tan, Y.; Pollock, C.; Chen, H. Maternal high-fat diet induces metabolic stress response disorders in offspring hypothalamus. J. Mol. Endocrinol. 2017, 59, 81–92. [Google Scholar]
- Saengnipanthkul, S.; Noh, H.L.; Friedline, R.H.; Suk, S.; Choi, S.; Acosta, N.K.; Tran, D.A.; Hu, X.; Inashima, K.; Kim, A.M. Maternal exposure to high-fat diet during pregnancy and lactation predisposes normal weight offspring mice to develop hepatic inflammation and insulin resistance. Physiol. Rep. 2021, 9, e14811. [Google Scholar] [CrossRef] [PubMed]
- Kislal, S.; Jin, W.; Maesner, C.; Edlow, A.G. Mismatch between obesogenic intrauterine environment and low-fat postnatal diet may confer offspring metabolic advantage. Obes. Sci. Pract. 2021, 7, 450–461. [Google Scholar] [CrossRef] [PubMed]
- Bayol, S.A.; Simbi, B.H.; Bertrand, J.; Stickland, N.C. Offspring from mothers fed a ‘junk food’diet in pregnancy and lactation exhibit exacerbated adiposity that is more pronounced in females. J. Physiol. 2008, 586, 3219–3230. [Google Scholar] [CrossRef]
- Gugusheff, J.R.; Vithayathil, M.; Ong, Z.; Muhlhausler, B. The effects of prenatal exposure to a ‘junk food’diet on offspring food preferences and fat deposition can be mitigated by improved nutrition during lactation. J. Dev. Orig. Health Dis. 2013, 4, 348–357. [Google Scholar] [CrossRef]
- Tsuduki, T.; Yamamoto, K.E.S.; Hatakeyama, Y.; Sakamoto, Y. High dietary fat intake during lactation promotes the development of social stress-induced obesity in the offspring of mice. Nutrients 2015, 7, 5916–5932. [Google Scholar] [CrossRef]
- Way, J.M.; Harrington, W.W.; Brown, K.K.; Gottschalk, W.K.; Sundseth, S.S.; Mansfield, T.A.; Ramachandran, R.K.; Willson, T.M.; Kliewer, S.A. Comprehensive messenger ribonucleic acid profiling reveals that peroxisome proliferator-activated receptor γ activation has coordinate effects on gene expression in multiple insulin-sensitive tissues. Endocrinology 2001, 142, 1269–1277. [Google Scholar] [CrossRef]
- Muhlhausler, B.S.; Gugusheff, J.R.; Langley-Evans, S.C. Maternal junk food diets: The effects on offspring fat mass and food preferences. In Diet, Nutrition, and Fetal Programming; Rajendram, R., Preedy, V., Patel, V., Eds.; Nutrition and Health; Humana Press: Cham, Switzerland, 2017. [Google Scholar]
- Muhlhausler, B.S.; Duffield, J.; McMillen, I. Increased maternal nutrition stimulates peroxisome proliferator activated receptor-γ, adiponectin, and leptin messenger ribonucleic acid expression in adipose tissue before birth. Endocrinology 2007, 148, 878–885. [Google Scholar] [CrossRef] [PubMed]
- Shimano, H. Sterol regulatory element-binding proteins (SREBPs): Transcriptional regulators of lipid synthetic genes. Prog. Lipid Res. 2001, 40, 439–452. [Google Scholar] [CrossRef] [PubMed]
- Dearden, L.; Balthasar, N. Sexual dimorphism in offspring glucose-sensitive hypothalamic gene expression and physiological responses to maternal high-fat diet feeding. Endocrinology 2014, 155, 2144–2154. [Google Scholar] [CrossRef] [PubMed]
- Kruse, M.; Seki, Y.; Vuguin, P.M.; Du, X.Q.; Fiallo, A.; Glenn, A.S.; Singer, S.; Breuhahn, K.; Katz, E.B.; Charron, M.J. High-fat intake during pregnancy and lactation exacerbates high-fat diet-induced complications in male offspring in mice. Endocrinology 2013, 154, 3565–3576. [Google Scholar] [CrossRef]
- Choi, J.S. Effects of maternal and post-weaning high-fat diet on leptin resistance and hypothalamic appetite genes in sprague dawley rat offspring. Clin. Nutr. Res. 2018, 7, 276–290. [Google Scholar] [CrossRef] [PubMed]
- Chang, E.; Hafner, H.; Varghese, M.; Griffin, C.; Clemente, J.; Islam, M.; Carlson, Z.; Zhu, A.; Hak, L.; Abrishami, S. Programming effects of maternal and gestational obesity on offspring metabolism and metabolic inflammation. Sci. Rep. 2019, 9, 16027. [Google Scholar] [CrossRef]
- Shah, N.R.; Braverman, E.R. Measuring adiposity in patients: The utility of body mass index (BMI), percent body fat, and leptin. PLoS ONE 2012, 7, e33308. [Google Scholar] [CrossRef]
- Takasaki, M.; Honma, T.; Yanaka, M.; Sato, K.; Shinohara, N.; Ito, J.; Tanaka, Y.; Tsuduki, T.; Ikeda, I. Continuous intake of a high-fat diet beyond one generation promotes lipid accumulation in liver and white adipose tissue of female mice. J. Nutr. Biochem. 2012, 23, 640–645. [Google Scholar] [CrossRef]
- Zheng, J.; Zhang, L.; Wang, Z.; Zhang, J. Maternal high-fat diet regulates glucose metabolism and pancreatic β cell phenotype in mouse offspring at weaning. PeerJ 2020, 8, e9407. [Google Scholar] [CrossRef]
- Lima, M.; Perez, G.; Morais, G.; Santos, L.; Cordeiro, G.; Couto, R.; Deiró, T.; Leandro, C.; Barreto-Medeiros, J. Effects of maternal high fat intake during pregnancy and lactation on total cholesterol and adipose tissue in neonatal rats. Braz. J. Biol. 2018, 78, 615–618. [Google Scholar] [CrossRef]
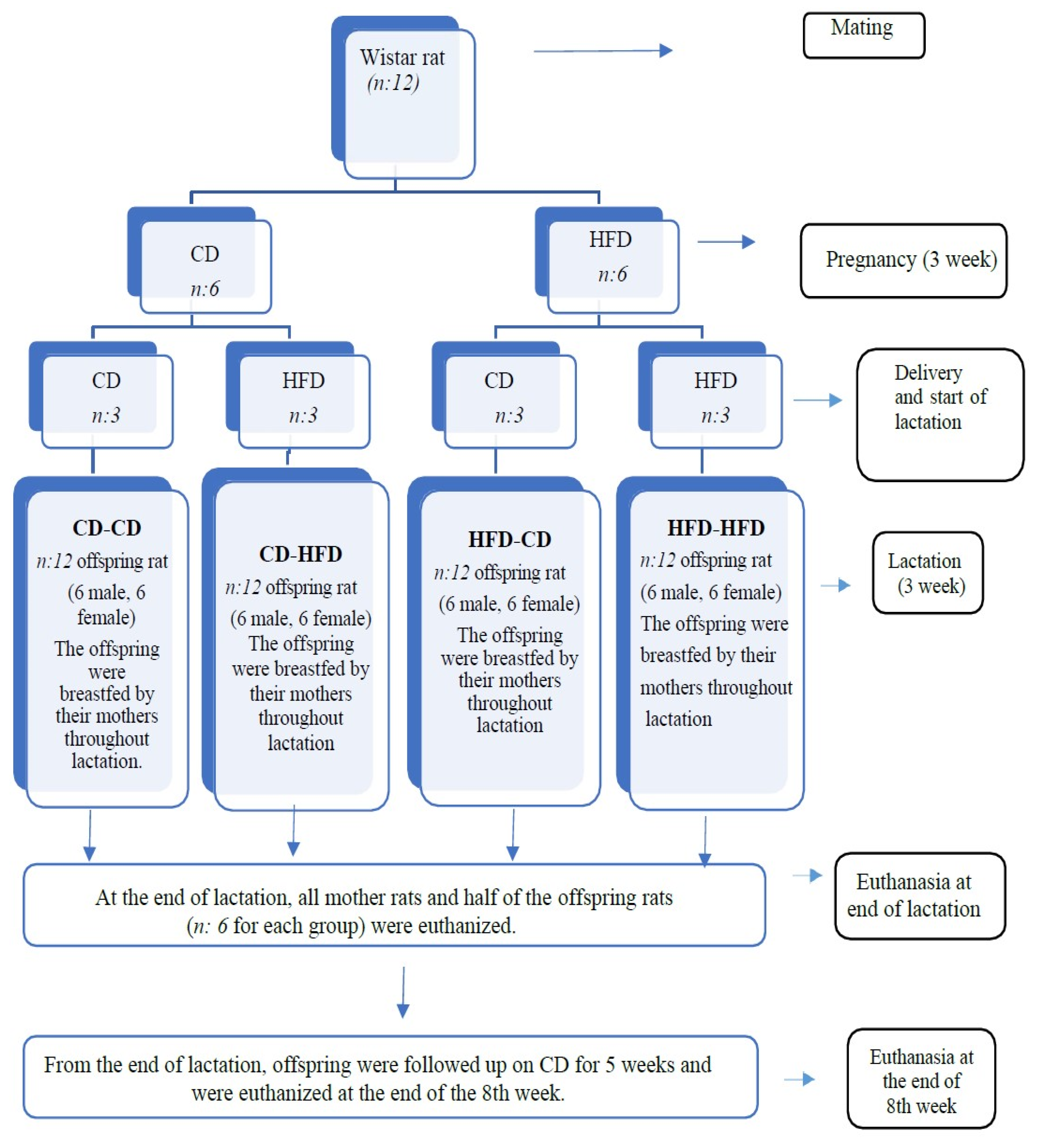

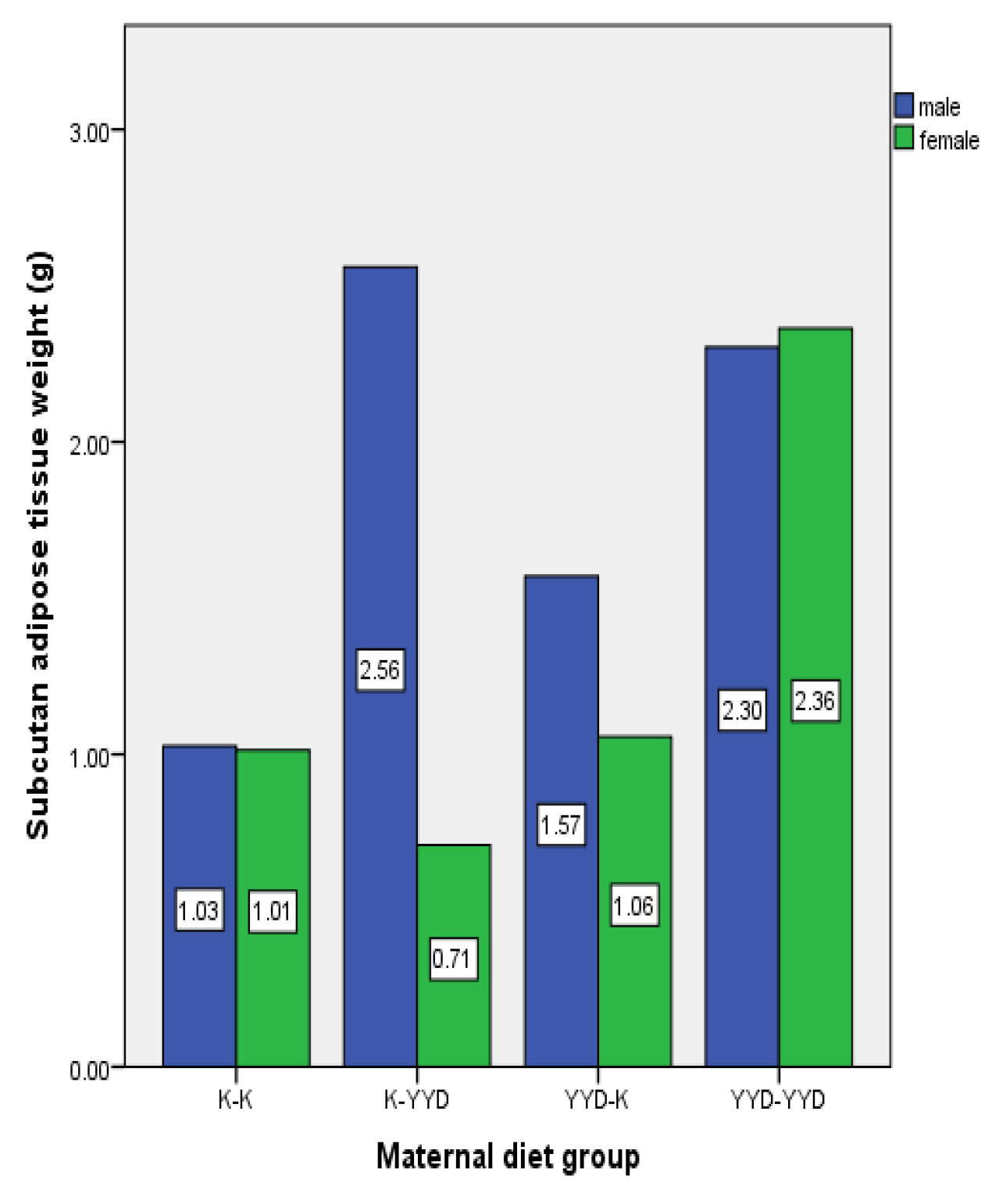
| Group | Pregnancy Period | Lactation Period |
|---|---|---|
| Group 1: CD *-CD | Control Diet | Control Diet |
| Group 2: CD-HFD ** | Control Diet | High-Fat Diet |
| Group 3: HFD-CD | High-Fat Diet | Control Diet |
| Group 4: HFD-HFD | High-Fat Diet | High-Fat Diet |
| Maternal Diet Group | Body Weights of Offspring | |||
|---|---|---|---|---|
| End of 1st Week | End of 2nd Week | End of 3rd Week | ||
| ± SD | ± SD | ± SD | p * | |
| CD-CD (n: 12) | 11.7 ± 1.74 ac | 23.0 ± 3.20 | 45.9 ± 2.34 | <0.001 *** |
| CD-HFD (n: 12) | 16.6 ± 0.88 ab | 29.5 ± 2.14 abcd | 55.0 ± 1.92 | <0.001 *** |
| HFD-CD (n: 12) | 16.4 ± 1.45 a | 25.5 ± 2.43 c | 50.7 ± 4.28 | <0.001 *** |
| HFD-HFD (n: 12) | 15.8 ± 1.14 a | 30.2 ± 4.11 a | 54.4 ± 2.27 | <0.001 *** |
| p ** | <0.001 *** | <0.001 *** | <0.001 *** | |
| Male | ||||
| CD-CD (n: 6) | 12.4 ± 1.71 a | 25.3 ± 2.60 a | 46.4 ± 1.28 | <0.001 *** |
| CD-HFD (n: 6) | 16.0 ± 0.45 b | 28.4 ± 2.69 b | 54.5 ± 1.34 a | <0.001 *** |
| HFD-CD (n: 6) | 17.0 ± 1.89 b | 27.2 ± 2.33 | 52.7 ± 3.38 b | <0.001 *** |
| HFD-HFD (n: 6) | 16.7 ± 0.65 b | 32.2 ± 4.90 | 55.0 ± 3.00 | <0.001 *** |
| p ** | 0.022 *** | 0.022 *** | 0.04 *** | |
| Female | ||||
| CD-CD (n: 6) | 10.9 ± 1.72 a | 20.6 ± 1.60 b | 45.3 ± 3.35 b | <0.001 *** |
| CD-HFD (n: 6) | 17.3 ± 0.58 b | 30.6 ± 0.86 a | 55.6 ± 2.55 a | <0.001 *** |
| HFD-CD (n: 6) | 15.8 ± 0.69 b | 23.8 ± 1.01 b | 48.7 ± 4.75 b | <0.001 *** |
| HFD-HFD (n: 6) | 15.0 ± 0.87 | 28.2 ± 2.61 b | 53.9 ± 1.73 b | <0.001 *** |
| p ** | <0.001 *** | <0.001 *** | <0.001 *** | |
| Maternal Diet Group | Post-Weaning Body Weights of Eight-Week-Old Rats | |||||
|---|---|---|---|---|---|---|
| End of 4th Week | End of 5th Week | End of 6th Week | End of 7th Week | End of 8th Week | ||
| ± SD | ± SD | ± SD | ± SD | ± SD | p * | |
| CD-CD (n: 6) | 69.5 ± 6.76 | 92.0 ± 5.64 | 120.3 ± 8.18 | 133.4 ± 6.66 | 159.0 ± 11.64 | <0.001 † |
| CD-HFD (n: 6) | 77.5 ± 3.23 | 106.8 ± 18.11 | 141.3 ± 35.10 | 169.4 ± 48.36 | 200.0 ± 61.37 | <0.001 † |
| HFD-CD (n: 6) | 78.2 ± 6.03 | 105.0 ± 14.90 | 129.5 ± 20.53 | 163.0 ± 22.60 | 201.8 ± 32.71 | <0.001 † |
| HFD-HFD (n: 6) | 76.7 ± 7.07 | 107.7 ± 13.34 | 144.1 ± 9.98 | 181.5 ± 24.28 | 226.5 ± 39.11 | <0.001 † |
| p ** | 0.069 | 0.199 | 0.221 | 0.061 | 0.061 | |
| Male *** | ||||||
| CD-CD (n: 3) | 73.7 ± 6.29 | 95.6 ± 2.70 | 123.2 ± 10.89 | 133.9 ± 10.01 | 165.3 ± 13.57 | <0.001 † |
| CD-HFD (n: 3) | 79.4 ± 2.65 | 118.6 ± 19.85 | 168.3 ± 29.83 | 209.6 ± 30.98 | 255.6 ± 10.40 | <0.001 † |
| HFD-CD (n: 3) | 81.7 ± 7.01 | 111.5 ± 19.48 | 138.6 ± 26.50 | 179.3 ± 16.01 | 219.0 ± 33.15 | <0.001 † |
| HFD-HFD (n: 3) | 79.1 ± 8.34 | 100.1 ± 13.07 | 143.3 ± 15.30 | 188.3 ± 30.66 | 232.0 ± 47.75 | <0.001 † |
| p **** | 0.459 | 0.361 | 0.282 | 0.066 | 0.066 | |
| Female *** | ||||||
| CD-CD (n: 3) | 65.3 ± 4.72 | 88.4 ± 5.81 | 145.0 ± 3.60 | 132.9 ± 3.16 b | 152.6 ± 5.85 a | <0.001 † |
| CD-HFD (n: 3) | 75.6 ± 2.85 | 94.9 ± 1.90 | 120.5 ± 10.21 | 129.1 ± 5.34 c | 144.3 ± 3.51 a | <0.001 † |
| HFD-CD (n: 3) | 74.8 ± 2.43 | 98.6 ± 7.24 | 114.4 ± 3.50 | 146.6 ± 14.84 a | 184.6 ± 26.31 a | <0.001 † |
| HFD-HFD (n: 3) | 74.4 ± 6.23 | 115.2 ± 10.12 | 117.4 ± 4.84 | 174.8 ± 19.93 a | 221.0 ± 38.11 b | <0.001 † |
| p **** | 0.147 | 0.055 | 0.077 | 0.039 † | 0.026 † | |
| CD-CD | CD-HFD | HFD-CD | HFD-HFD | ||
|---|---|---|---|---|---|
| ± SD | ± SD | ± SD | ± SD | p | |
| 3-week-old rats (n: 12) | |||||
| BWG ** | 51.2 ± 4.97 ac | 52.2 ± 4.50 ac | 37.6 ± 2.10 b,c | 38.8 ± 2.36 b,c | <0.001 * |
| Lee index ** | 0.26 ± 0.004 a | 0.28 ± 0.225 a | 0.30 ± 0.008 a,b | 0.29 ± 0.008 a,b | 0.001 * |
| Male (n: 6) ** | |||||
| BWG-3w | 51.7 ± 11.74 a | 57.0 ± 14.15 b | 40.1 ± 4.84 bc | 42.1 ± 3.97 a | 0.021 * |
| Lee index-3w | 0.27 ± 0.005 | 0.27 ± 0.02 | 0.30 ± 0.01 | 0.29 ± 0.01 | 0.190 |
| Female (n: 6) ** | |||||
| BWG-3w | 42.3 ± 7.71 a | 49.9 ± 6.68 b | 37.5 ± 4.61 bc | 42.5 ± 7.25 d | 0.033 * |
| Lee index-3w | 0.27 ± 0.000 | 0.29 ± 0.011 | 0.30 ± 0.005 | 0.29 ± 0.005 | 0.056 |
| 8-week-old-rats (n: 6) | |||||
| BWG ** | 152.9 ± 11.72 | 189.9 ± 58.9 | 191.9 ± 31.30 | 214.0 ± 36.89 | 0.081 |
| Lee index ** | 0.30 ± 0.017 | 0.31 ± 0.005 | 0.29 ± 0.05 | 0.30 ± 0.005 | 0.146 |
| Male (n: 3) *** | |||||
| BWG-8w | 159.4 ± 13.45 | 243.2 ± 10.03 | 207.8 ± 31.74 | 218.4 ± 45.27 | 0.082 |
| Lee index-8w | 0.31 ± 0.010 | 0.30 ± 0.005 | 0.29 ± 0.005 | 0.31 ± 0.005 | 0.122 |
| Female (n: 3) *** | |||||
| BWG-8w | 146.3 ± 5.90 | 136.5 ± 3.91 | 176.0 ± 26.17 a | 209.7 ± 36.00 b | 0.026 * |
| Lee index-8w | 0.29 ± 0.017 | 0.31 ± 0.005 | 0.29 ± 0.005 | 0.30 ± 0.005 | 0.120 |
| CD-CD | CD-HFD | HFD-CD | HFD-HFD | p | |
|---|---|---|---|---|---|
| ± SD (n: 6) | ± SD (n: 6) | ± SD (n: 6) | ± SD (n: 6) | ||
| 3-week-old rats | |||||
| Glucose (mg/dL) | 88.4 ± 5.83 | 99.7 ± 2.77 | 98.3 ± 9.65 | 103.9 ± 8.26 | 0.009 * |
| Insulin (mlU/L) | 5.53 ± 0.50 | 5.88 ± 1.25 | 6.45 ± 1.42 | 5.63 ± 0.61 | 0.429 |
| Triglyceride (mmol/L) | 494.0 ± 26.24 a | 478.4 ± 25.39 b | 472.2 ± 20.01 b | 439.3 ± 27.31 c | 0.009 * |
| Total-cholesterol (mmol/L) | 1.58 ± 0.08 | 1.67 ± 0.12 | 1.58 ± 0.21 | 1.60 ± 0.21 | 0.794 |
| Leptin (ng/L) | 1.98 ± 0.24 | 2.27 ± 0.18 | 2.20 ± 0.21 | 2.15 ± 0.18 | 0.126 |
| 8-week-old rats | |||||
| Glucose (mg/dL) | 92.3 ± 5.31 | 94.9 ± 7.42 | 92.1 ± 4.39 | 93.5 ± 4.47 | 0.811 |
| Insulin (mlU/L) | 5.93 ± 0.49 b | 5.25 ± 0.62 a | 6.42± 1.01 c | 5.25 ± 0.37 c | 0.016 * |
| Triglyceride (mmol/L) | 500.8 ± 25.88 | 487.5 ± 19.74 | 466.9 ± 29.38 | 469.8 ± 18.6 | 0.066 |
| Total cholesterol (mmol/L) | 1.53 ± 1.63 | 1.35 ± 1.88 | 1.47 ± 0.22 | 1.42 ± 1.48 | 0.368 |
| Leptin (ng/L) | 2.07 ± 0.12 | 1.68 ± 0.39 | 1.77 ± 0.27 | 1.82 ± 0.22 | 0.116 |
| Gene Expression | CD-CD ± SD | CD-HFD ± SD | HFD-CD ± SD | HFD-HFD | p |
|---|---|---|---|---|---|
| 3-week-old rats (n: 6) | |||||
| FAS | 1.634 ± 2.226 | 0.898 ± 0.571 | 0.158 ± 0.121 | 0.547 ± 0.440 | 0.195 |
| SREBP-1c | 1.919 ± 2.597 | 0.530 ± 0.405 | 0.417 ± 0.352 | 1.884 ± 1.756 | 0.217 |
| PPAR-γ | 4.197 ± 5.295 | 0.584 ± 0.327 | 0.800 ± 0.940 | 1.028 ± 1.882 | 0.127 |
| Male 3-week-old (n: 3) | |||||
| FAS | 2.395 ± 3.255 | 0.921 ± 0.339 | 0.204 ± 0.166 | 0.730 ± 0.594 | 0.460 |
| SREBP-1c | 3.330 ± 3.284 | 0.758 ± 0.431 | 0.523 ± 0.378 | 2.403 ± 2.122 | 0.316 |
| PPAR-γ | 2.826 ± 4.720 | 0.646 ± 0.220 | 1.122 ± 1.363 | 1.682 ± 2.676 | 0.799 |
| Female 3-week-old (n: 3) | |||||
| FAS | 0.873 ± 0.244 | 0.876 ± 0.835 | 0.112 ± 0.052 | 0.364 ± 1.745 | 0.363 |
| SREBP-1c | 0.508 ± 0.324 | 0.304 ± 0.265 | 0.310 ± 0.365 | 1.365 ± 1.548 | 0.825 |
| PPAR-γ | 5.568 ± 6.495 | 0.523 ± 0.456 | 0.478 ± 0.208 | 0.373 ± 0.640 | 0.176 |
| 8-week-old rats (n: 6) | |||||
| FAS | 2.063 ± 2.313 | 3.171 ± 6.437 | 2.073 ± 1.683 | 3.603 ± 6.477 | 0.922 |
| SREBP-1c | 1.909 ± 1.735 | 0.918 ± 0.364 | 2.265 ± 2.01 | 1.768 ± 3.175 | 0.718 |
| PPAR-γ | 1.878 ± 1.528 | 0.220 ± 0.197 | 3.409 ± 6.725 | 1.465 ± 2.390 | 0.520 |
| Male 8-week-old (n: 3) | |||||
| FAS | 2.426 ± 2.518 | 5.960 ± 8.958 | 1.809 ± 1.837 | 6.123 ± 9.142 | 0.164 |
| SREBP-1c | 2.357 ± 2.119 | 0.927 ± 0.524 | 1.313 ± 1.100 | 0.642 ± 0.521 | 0.393 |
| PPAR-γ | 1.496 ± 1.728 | 0.261 ± 0.260 | 0.628 ± 2.413 | 2.369 ± 3.437 | 0.220 |
| Female 8-week-old (n: 3) | |||||
| FAS | 1.701 ± 2.576 | 0.383 ± 0.132 | 2.338 ± 1.870 | 1.084 ± 1.509 | 0.591 |
| SREBP-1c | 1.462 ± 1.561 | 0.909 ± 0.236 | 3.217 ± 2.492 | 2.894 ± 2.492 | 0.695 |
| PPAR-γ | 2.261 ± 1.552 | 0.181 ± 0.157 | 6.191 ± 9.477 | 0.561 ± 0.157 | 0.448 |
| 3 Weeks Old | 8 Weeks Old | |||||||
|---|---|---|---|---|---|---|---|---|
| CD-CD (n: 6) | CD-HFD (n: 6) | HFD-CD (n: 6) | HFD-HFD (n: 6) | CD-CD (n: 6) | CD-HFD (n: 6) | HFD-CD (n: 6) | HFD-HFD (n: 6) | |
| Deformation of adipocytes | ++ | + | ++ | +++ | + | +++ | ||
| Fusion of adipocytes | + | ++ | ++ | + | +++ | |||
| Lipid disturbance and myelin formation in adipocytes | ++ | + | ++ | ++ | + | +++ | ||
| Density of brown adipose tissue cells | + | + | ||||||
| Increase in connective tissue | ++ | ++ | +++ | ++ | ||||
| Dilation in blood vessels | + | ++ | + | ++ | ++ | +++ | ||
| Infiltration | ++ | ++ | ++ | ++ | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arslan, S.; Yıldıran, H.; Seymen, C.M. The Effect of Maternal High-Fat Diet on Adipose Tissue Histology and Lipid Metabolism-Related Genes Expression in Offspring Rats. Nutrients 2024, 16, 150. https://doi.org/10.3390/nu16010150
Arslan S, Yıldıran H, Seymen CM. The Effect of Maternal High-Fat Diet on Adipose Tissue Histology and Lipid Metabolism-Related Genes Expression in Offspring Rats. Nutrients. 2024; 16(1):150. https://doi.org/10.3390/nu16010150
Chicago/Turabian StyleArslan, Sabriye, Hilal Yıldıran, and Cemile Merve Seymen. 2024. "The Effect of Maternal High-Fat Diet on Adipose Tissue Histology and Lipid Metabolism-Related Genes Expression in Offspring Rats" Nutrients 16, no. 1: 150. https://doi.org/10.3390/nu16010150
APA StyleArslan, S., Yıldıran, H., & Seymen, C. M. (2024). The Effect of Maternal High-Fat Diet on Adipose Tissue Histology and Lipid Metabolism-Related Genes Expression in Offspring Rats. Nutrients, 16(1), 150. https://doi.org/10.3390/nu16010150







