Skin Anti-Aging Efficacy of Enzyme-Treated Supercritical Caviar Extract: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of CV
2.2. Free Amino Acid and Peptide Analysis
2.3. Study Design and Dose Determination
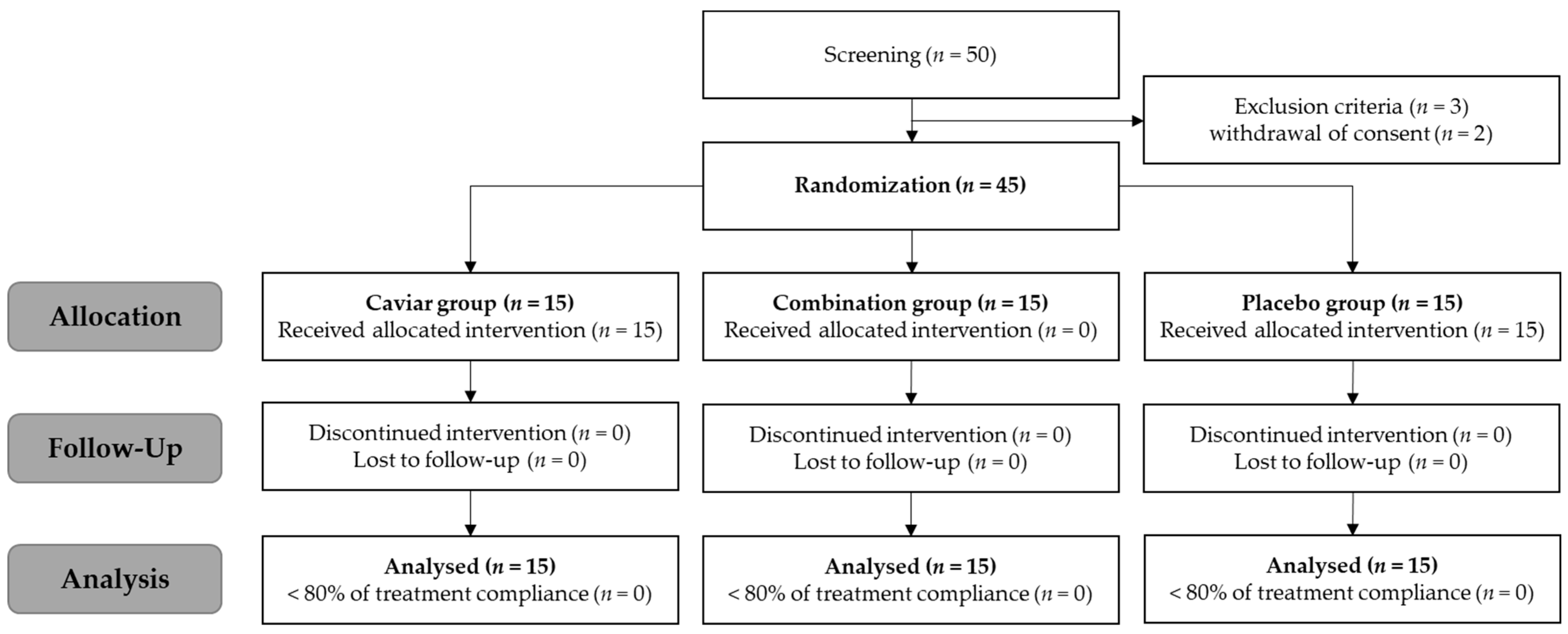
2.4. Study Participants and Schedule
2.5. Skin Hydration
2.6. Skin Elasticity
2.7. Transepidermal Water Loss (TEWL)
2.8. Skin Melanin
2.9. Skin Wrinkles
2.10. Safety Test and Adverse Reaction Test
2.11. Statistical Analysis
3. Results
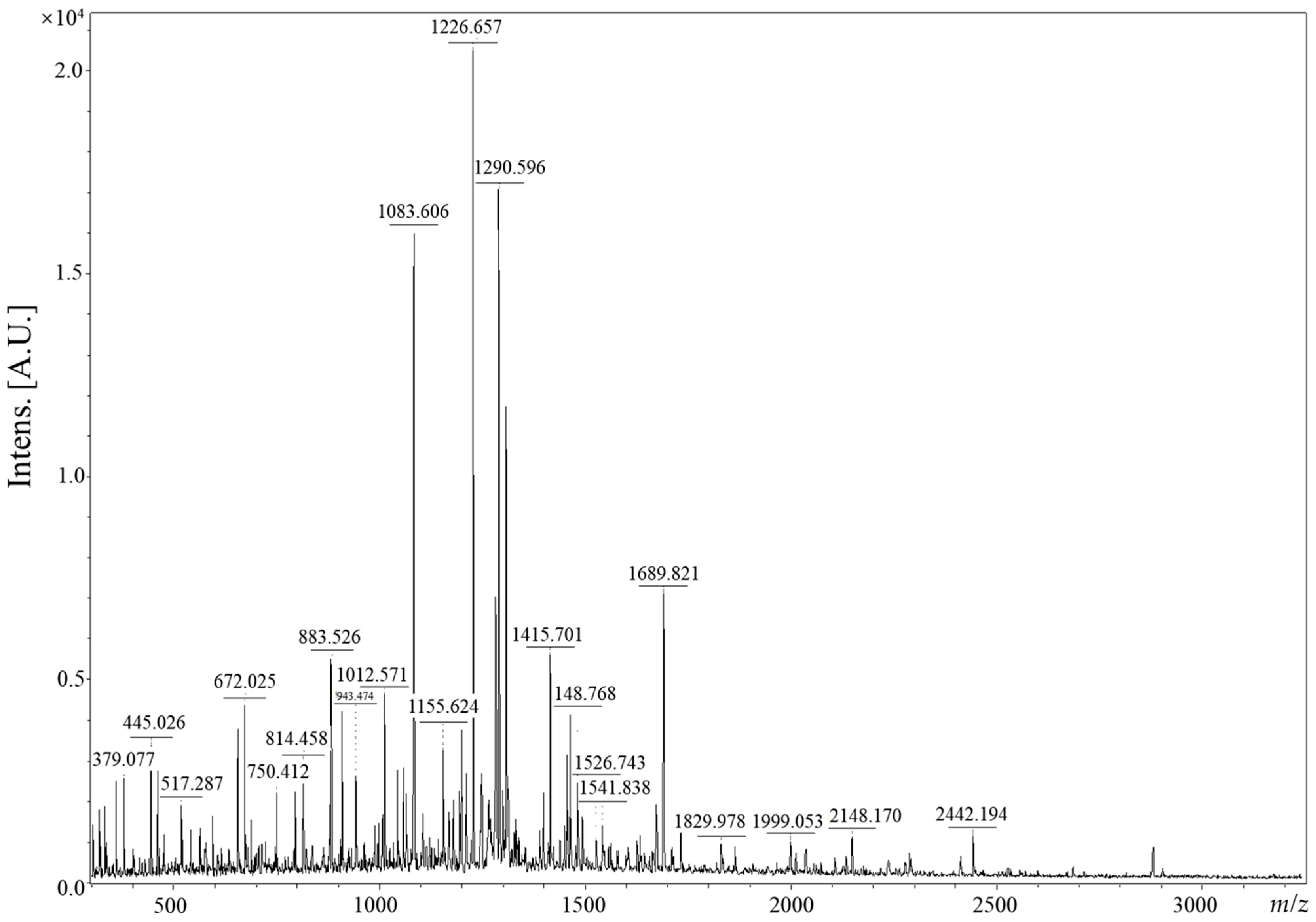
3.1. Free Amino Acid Contents and Peptide of CV
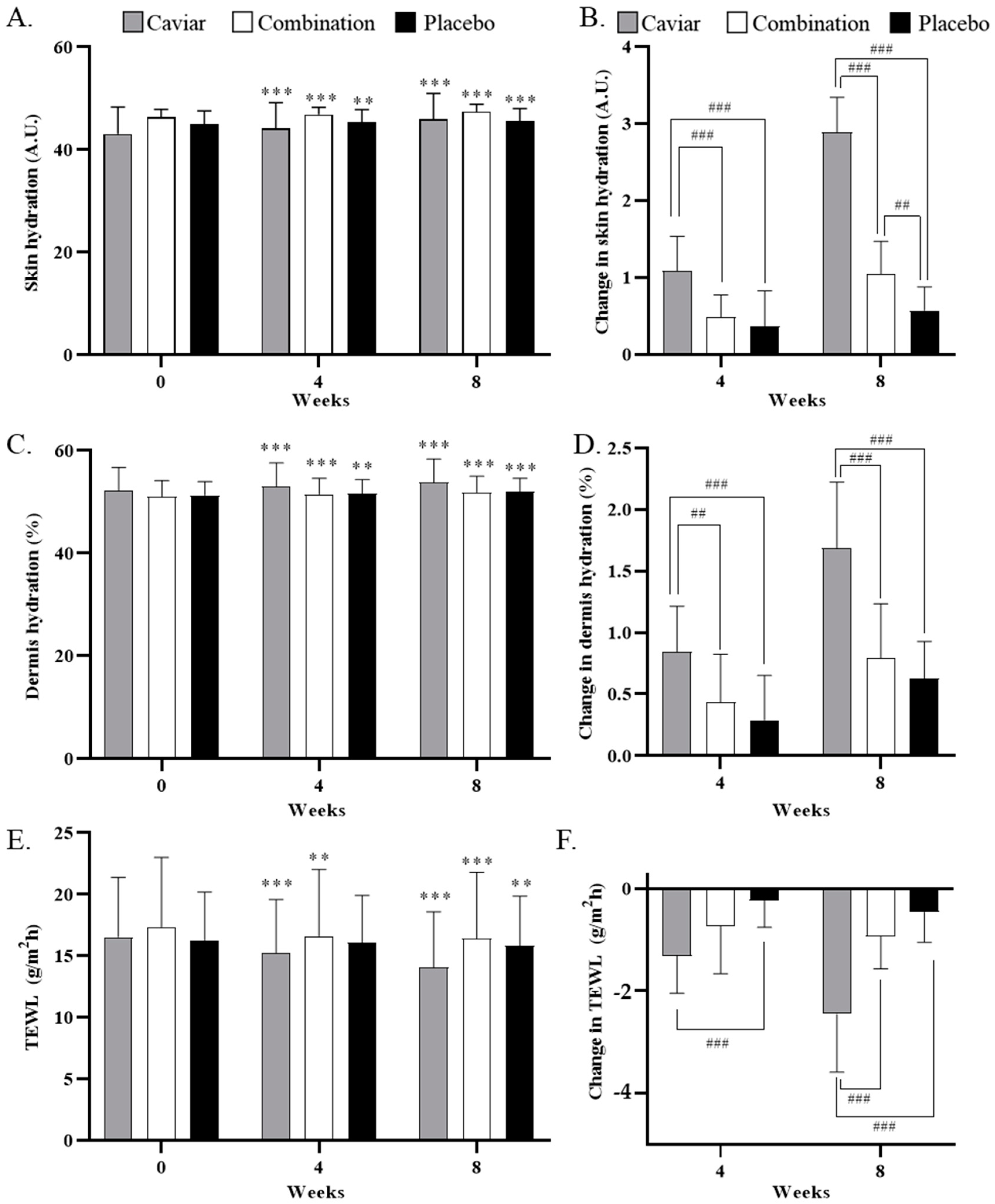
3.2. Effects of CV on Skin Hydration
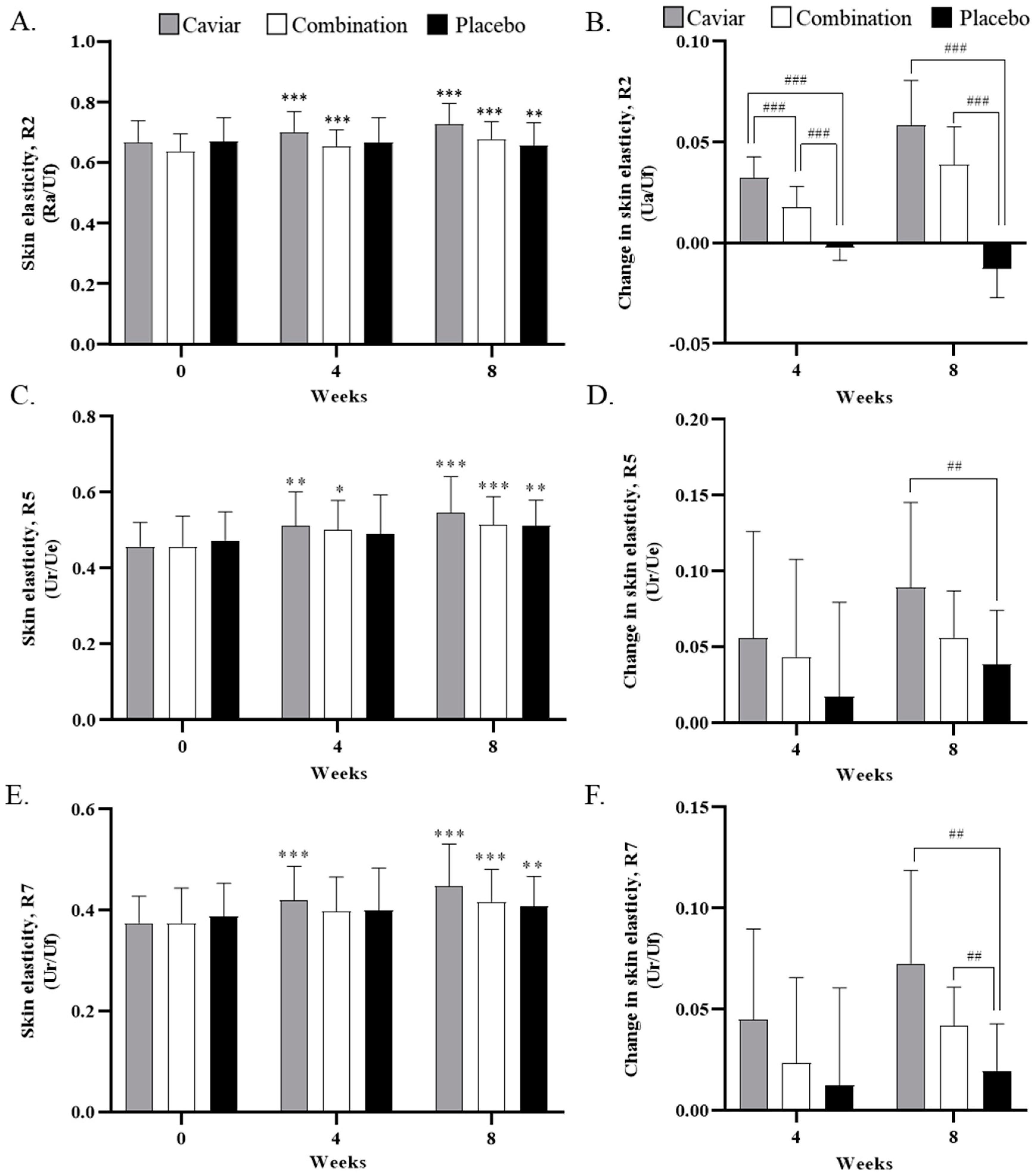
3.3. Skin Elasticity
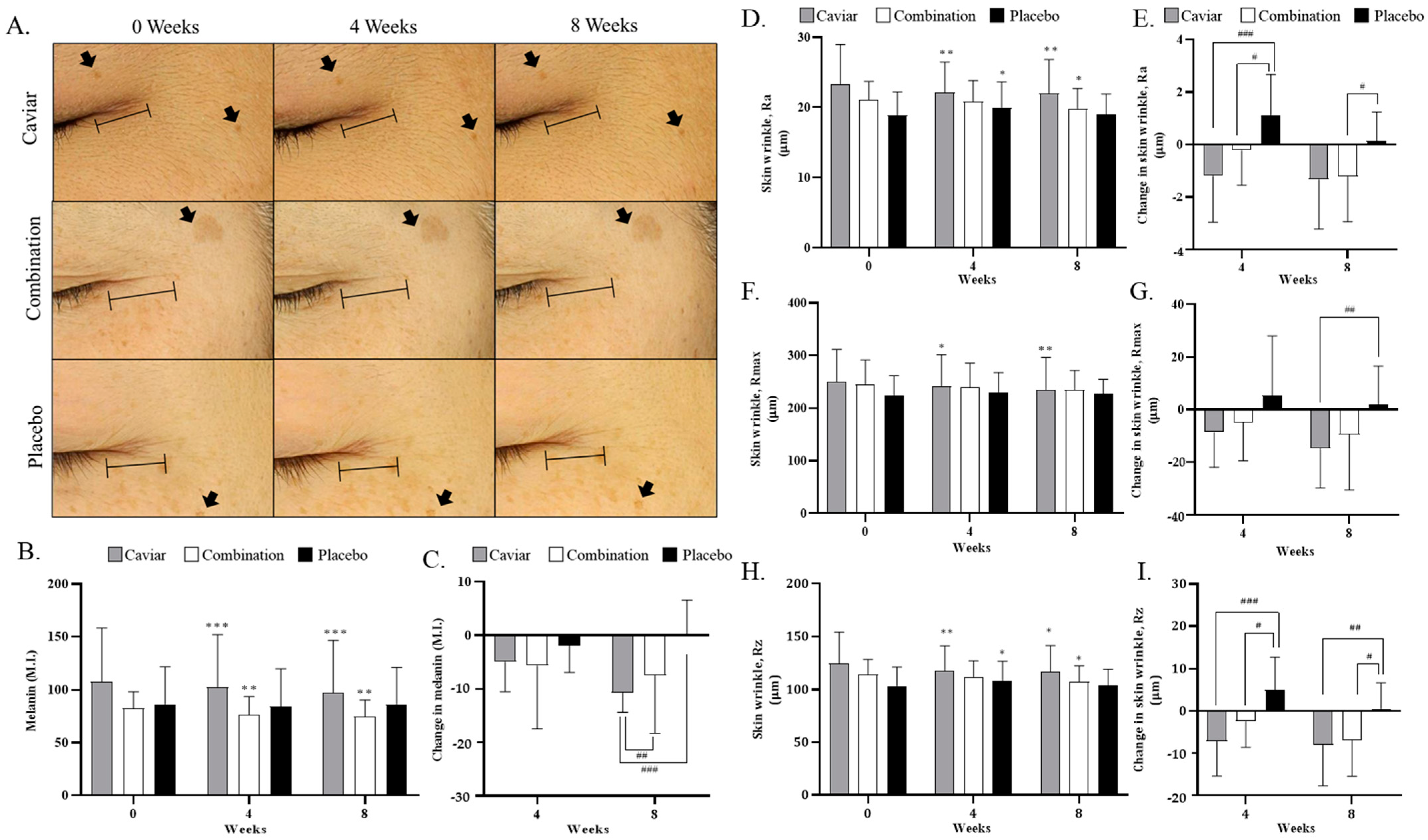
3.4. Effects of CV on Skin Melanin Index and Wrinkles
3.5. Safety and Adverse Reaction Evaluation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Wirth, M.; Kirschbaum, F.; Gessner, J.; Krüger, A.; Patriche, N.; Billard, R. Chemical and biochemical composition of caviar from different sturgeon species and origins. Nahrung/Food 2000, 44, 233–237. [Google Scholar] [CrossRef] [PubMed]
- Kieliszek, M. Selenium–fascinating microelement, properties and sources in food. Molecules 2019, 24, 1298. [Google Scholar] [CrossRef] [PubMed]
- Lorenzetti, A.; Marotta, F.; Yadav, H.; Celep, G.; Minelli, E.; Carrera-Bastos, P.; Jain, S.; Polimeni, A.; Solimene, U. Improving sperm quality and spermatogenesis through a bioactive marine compound: An experimental study. Acta Biomed. 2012, 83, 108–113. [Google Scholar] [PubMed]
- Lee, K.E.; Nho, Y.H.; Yun, S.K.; Park, S.M.; Kang, S.; Yeo, H. Caviar extract and its constituent DHA Inhibits UVB-Irradiated skin aging by inducing adiponectin production. Int. J. Mol. Sci. 2020, 21, 3383. [Google Scholar] [CrossRef] [PubMed]
- Farris, P.; Draelos, Z.D.; Felipe de Oliveira Stehling, L. Novel facial treatment regimen improves aging skin appearance. J. Drugs Dermatol. 2021, 20, 274–278. [Google Scholar] [CrossRef]
- Baroni, A.; Buommino, E.; De Gregorio, V.; Ruocco, E.; Ruocco, V.; Wolf, R. Structure and function of the epidermis related to barrier properties. Clin. Dermatol. 2012, 30, 257–262. [Google Scholar] [CrossRef]
- Boer, M.; Duchnik, E.; Maleszka, R.; Marchlewicz, M. Structural and biophysical characteristics of human skin in maintaining proper epidermal barrier function. Postepy Dermatol. Alergol. 2016, 33, 1–5. [Google Scholar] [CrossRef]
- Farage, M.A.; Miller, K.W.; Elsner, P.; Maibach, H.I. Intrinsic and extrinsic factors in skin ageing: A review. Int. J. Cosmet. Sci. 2008, 30, 87–95. [Google Scholar] [CrossRef]
- Cavinato, M.; Jansen-Dürr, P. Molecular mechanisms of UVB-induced senescence of dermal fibroblasts and its relevance for photoaging of the human skin. Exp. Gerontol. 2017, 94, 78–82. [Google Scholar] [CrossRef]
- Mesa-Arango, A.; Flórez-Muñoz, S.; Sanclemente, G. Mechanisms of skin aging. IATREIA 2017, 30, 160–170. [Google Scholar] [CrossRef]
- Agrawal, R.; Hu, A.; Bollag, W. The Skin and Inflamm-aging. Biology 2023, 12, 1396. [Google Scholar] [CrossRef] [PubMed]
- Videira, I.F.; Moura, D.F.; Magina, S. Mechanisms regulating melanogenesis. An. Bras. Dermatol. 2013, 88, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Rabionet, M.; Gorgas, K.; Sandhoff, R. Ceramide synthesis in the epidermis. Biochim. Biophys. Acta (BBA)-Mol. Cell Biol. Lipids 2014, 1841, 422–434. [Google Scholar] [CrossRef] [PubMed]
- Rinnerthaler, M.; Bischof, J.; Streubel, M.; Trost, A.; Richter, K. Oxidative Stress in Aging Human Skin. Biomolecules 2015, 5, 545–589. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Kim, D.; Lee, M.; Han, S.; Jun, W.; Jung, H.M.; Koo, Y.K.; Na, G.H.; Han, S.H.; Han, J.; et al. Enzyme-treated caviar prevents UVB irradiation-induced skin photoaging. Mar. Drugs 2022, 20, 685. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Na, G.H.; Jung, H.M.; Han, S.H.; Han, J.; Koo, Y.K. Enzyme-treated caviar extract ameliorates melanogenesis in UVB-induced SKH-1 hairless mice. Biochem. Biophys. Res. Commun. 2023, 673, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Dai, J.; Zhang, S.; Qiao, J.; Lian, H.; Mao, L. Identification of characteristic peptides of casein in cow milk based on MALDI-TOF MS for direct adulteration detection of goat milk. Foods 2023, 12, 1519. [Google Scholar] [CrossRef] [PubMed]
- Center for Drug Evaluation and Research. Guidance for Industry: Estimating the Maximum Safe Starting Dose in Initial Clinical Trials for Therapeutics in Adult Healthy Volunteers; Pharmacology and Toxicology; Center for Drug Evaluation and Research, Food and Drug Administration: Rockville, MD, USA, 2005. [Google Scholar]
- Hamzeh, A.; Moslemi, M.; Karaminasab, M.; Khanlar, M.A.; Faizbakhsh, R.; Navai, M.B.; Tahergorabi, R. Amino acid composition of roe from wild and farmed beluga sturgeon (Huso huso). J. Agric. Sci. Technol. 2015, 17, 357–364. [Google Scholar]
- Mol, S.; Turan, S. Comparison of proximate, fatty acid and amino acid compositions of various types of fish roes. Int. J. Food Prop. 2008, 11, 669–677. [Google Scholar] [CrossRef]
- Park, K.S.; Kang, K.H.; Bae, E.Y.; Baek, K.A.; Shin, M.H.; Kim, D.U.; Kang, H.K.; Kim, K.J.; Choi, Y.J.; Im, J.S. General and biochemical composition of caviar from Sturgeon (Acipenser ruthenus) farmed in Korea. Int. Food Res. J. 2015, 22, 777–781. [Google Scholar]
- Weijzen, M.E.G.; van Gassel, R.J.J.; Kouw, I.W.K.; Trommelen, J.; Gorissen, S.H.M.; van Kranenburg, J.; Goessens, J.P.B.; van de Poll, M.C.G.; Verdijk, L.B.; van Loon, L.J.C. Ingestion of free amino acids compared with an equivalent amount of intact protein results in more rapid amino acid absorption and greater postprandial plasma amino acid availability without affecting muscle protein synthesis rates in young adults in a double-blind randomized trial. J. Nutr. 2022, 152, 59–67. [Google Scholar] [PubMed]
- Czajka, A.; Kania, E.M.; Genovese, L.; Corbo, A.; Merone, G.; Luci, C.; Sibilla, S. Daily oral supplementation with collagen peptides combined with vitamins and other bioactive compounds improves skin elasticity and has a beneficial effect on joint and general wellbeing. Nutr. Res. 2018, 57, 97–108. [Google Scholar] [CrossRef] [PubMed]
- Borumand, M.; Sibilla, S. Effects of a nutritional supplement containing collagen peptides on skin elasticity, hydration and wrinkles. J. Med. Nutr. Nutraceuticals 2015, 4, 47–53. [Google Scholar]
- Sinova, R.; Pavlik, P.; Ondrej, M.; Velebny, K. Hyaluronan: A key player or just a bystander in skin photoaging? Clin. Exp. Dermatol. 2022, 31, 442–458. [Google Scholar]
- Chavoshnejad, P.; Foroughi, A.H.; Dhandapani, N.; German, G.K.; Razavi, M.J. Effect of collagen degradation on the mechanical behavior and wrinkling of skin. Phys. Rev. E 2021, 104, 034406. [Google Scholar] [CrossRef]
- Juhl, P.; Bondesen, S.; Hawkins, C.; Karsdal, M.A.; Bay-Jensen, A.C.; Davies, M.J.; Siebuhr, A.S. Dermal fibroblasts have different extracellular matrix profiles induced by TGF-β, PDGF and IL-6 in a model for skin fibrosis. Sci. Rep. 2020, 10, 17300. [Google Scholar] [CrossRef] [PubMed]
- Clark, R.A.; Nielsen, L.D.; Welch, M.P.; McPherson, J.M. Collagen matrices attenuate the collagen-synthetic response of cultured fibroblasts to TGF-beta. J. Cell Sci. 1995, 108, 1251–1261. [Google Scholar] [CrossRef]
- Reilly, D.M.; Lozano, J. Skin collagen through the lifestages: Importance for skin health and beauty. Plast. Aesthetic Res. 2021, 8, 2. [Google Scholar] [CrossRef]
- Kang, W.; Choi, D.; Park, T. Dietary suberic acid protects against UVB-induced skin photoaging in hairless mice. Nutrients 2019, 11, 2948. [Google Scholar] [CrossRef]
- Kim, Y.I.; Kim, K.S.; Ahn, H.; Kang, I.; Shin, M.K. Reduced matrix metalloproteinase and collagen transcription mediated by the TGF-β/Smad pathway in passaged normal human dermal fibroblasts. J. Cosmet. Dermatol. 2020, 19, 1211–1218. [Google Scholar] [CrossRef]

| Composotion | Caviar Group | Combination Group | Placebo Group |
|---|---|---|---|
| Enzyme-Treated Caviar Extract | 500 mg | 10 mg | |
| Maltodextrin | 386 mg | 886 mg | |
| Silicon dioxide | 14 mg | 12 mg | 14 mg |
| Fish Collagen | 500 mg | ||
| Hyaluronic acid | 240 mg | ||
| Ascorbic acid | 125 mg | ||
| Biotin | 7.5 mg | ||
| d-α-tocopherol | 5.5 mg |
| Characteristics | Caviar Group | Combination Group | Placebo Group | |
|---|---|---|---|---|
| Age | 47.87 ± 8.27 | 48.07 ± 8.25 | 48.07 ± 6.84 | |
| Sex | Male | 3 | 1 | 2 |
| Female | 12 | 14 | 13 | |
| Skin wrinkles (grade) | 4.13 ± 0.83 | 3.93 ± 1.10 | 4.267 ± 1.03 | |
| Free Amino Acid | Supercritical CO2 Caviar Extract (mg/g) | Enzyme-Treated Supercritical Caviar Extract (mg/g) |
|---|---|---|
| Leucine | 0.73 | 10.32 |
| Phenylalanine | 0.33 | 3.63 |
| Tyrosine | 0.15 | 5.78 |
| Alanine | 0.33 | 2.49 |
| Isoleucine | 0.4 | 3.49 |
| Valine | 0.28 | 1.86 |
| Arginine | 0.14 | 1.51 |
| Methionine | 0.11 | 2.18 |
| Lysine | 0.34 | 3.6 |
| Serine | 0.23 | 1.67 |
| Threonine | 0.17 | 1.34 |
| Glutamic acid | 0.3 | 1.9 |
| Histidine | 0.05 | 0.59 |
| Aspartic acid | 0.31 | 0.53 |
| Threonine | 0.11 | 0.86 |
| Glycine | 0.07 | 0.42 |
| Proline | 0.23 | 0.66 |
| Cystine | - | 0.41 |
| Parameter | Caviar Group | Combination Group | Placebo Group | |||
|---|---|---|---|---|---|---|
| Hematological test (1) | 0 weeks | 8 weeks | 0 weeks | 8 weeks | 0 weeks | 8 weeks |
| WBC (109/L) | 5.61 ± 1.67 | 5.40 ± 1.340 | 4.75 ± 1.24 | 4.26 ± 1.35 | 5.24 ± 1.20 | 4.70 ± 1.23 |
| RBC (1012/L) | 4.63 ± 0.46 | 4.54 ± 0.43 | 4.37 ± 0.40 | 4.30 ± 0.32 | 4.24 ± 0.32 | 4.19 ± 0.25 |
| Hb (g/dL) | 13.78 ± 1.78 # | 13.45 ± 1.65 * | 11.91 ± 2.52 | 11.64 ± 2.39 | 12.61 ± 1.60 | 12.27 ± 1.40 |
| Hct (%) | 42.83 ± 4.63 # | 41.49 ± 4.32 | 38.30 ± 6.19 | 37.30 ± 5.71 | 39.31 ± 3.93 | 38.35 ± 3.31 |
| Platelet (109/L) | 250.67 ± 55.39 | 258.80 ± 55.30 | 280.80 ± 58.75 | 280.53 ± 49.41 | 279.20 ± 51.99 | 268.40 ± 57.70 |
| Seg. Neutrophil (%) | 55.27 ± 9.46 | 52.19 ± 8.77 | 55.07 ± 8.91 | 51.99 ± 8.64 | 52.95 ± 8.18 | 50.79 ± 9.18 |
| Lymphocyte (%) | 35.49 ± 8.70 | 37.00 ± 7.95 | 35.97 ± 8.67 | 37.55 ± 7.70 | 36.77 ± 7.30 | 38.40 ± 8.44 |
| Monocyte (%) | 6.68 ± 1.28 | 7.45 ± 1.50 * | 6.51 ± 1.48 | 7.14 ± 1.78 | 6.98 ± 1.58 | 7.64 ± 2.09 |
| Eosinophil (%) | 1.87 ± 0.87 | 2.59 ± 1.41 * | 1.64 ± 0.83 | 2.41 ± 2.37 | 2.41 ± 2.11 | 2.41 ± 1.56 |
| Basophil (%) | 0.69 ± 0.24 | 0.73 ± 0.33 | 0.81 ± 0.49 | 0.91 ± 0.51 | 0.89 ± 0.40 | 0.77 ± 0.43 |
| Biochemical test (2) | ||||||
| Glucose (mg/dL) | 91.20 ± 12.57 | 92.07 ± 8.88 | 89.93 ± 5.42 | 88.67 ± 7.07 | 88.67 ± 7.07 | 88.47 ± 7.74 |
| T.Protein (g/dL) | 6.96 ± 0.40 | 7.03 ± 0.37 | 6.86 ± 0.37 * | 6.97 ± 0.41 * | 6.85 ± 0.50 | 6.90 ± 0.42 |
| Albumin (mg/dL) | 0.67 ± 0.19 | 0.63 ± 0.24 | 0.70 ± 0.33 | 0.73 ± 0.34 | 0.65 ± 0.16 | 0.69 ± 0.15 |
| BUN (mg/dL) | 12.40 ± 3.29 | 13.47 ± 3.58 | 12.07 ± 2.94 | 12.00 ± 2.98 | 13.67 ± 3.98 | 14.07 ± 5.18 |
| AST (U/L) | 22.00 ± 7.41 | 21.40 ± 6.13 | 19.87 ± 10.11 | 19.67 ± 7.00 | 20.60 ± 4.93 | 20.80 ± 4.86 |
| ALT (U/L) | 25.14 ± 15.98 | 23.20 ± 16.01 | 17.36 ± 13.07 | 18.39 ± 10.81 | 13.93 ± 5.86 | 15.13 ± 4.78 |
| γ-GTP (U/L) | 25.27 ± 12.93 | 23.33 ± 11.48 | 23.33 ± 25.21 | 19.00 ± 8.61 | 18.47 ± 7.31 | 18.00 ± 8.34 |
| Creatinine (mg/dL) | 0.68 ± 0.15 | 0.67 ± 0.15 | 0.64 ± 0.13 | 0.63 ± 0.12 | 0.67 ± 0.16 | 0.66 ± 0.123 |
| Total Cholesterol (mg/dL) | 197.40 ± 31.30 | 203.27 ± 32.13 | 191.20 ± 34.55 | 193.20 ± 34.97 | 200.40 ± 34.77 | 194.13 ± 33.12 |
| HDL-Cholesterol (mg/dL) | 61.07 ± 15.16 | 60.40 ± 16.49 | 61.07 ± 14.21 | 60.20 ± 13.36 | 64.07 ± 12.38 | 61.13 ± 11.63 |
| LDL- Cholesterol (mg/dL) | 119.13 ± 22.06 | 121.47 ± 25.07 | 111.00 ± 29.81 | 108.73 ± 28.78 | 119.33 ± 29.04 | 113.47 ± 27.90 |
| Triglyceride (mg/dL) | 92.53 ± 59.70 | 89.27 ± 40.26 | 87.00 ± 51.61 | 102.00 ± 69.71 | 79.60 ± 27.09 | 77.80 ± 28.08 |
| Urine test (abnormal personnel) | ||||||
| Specific gravity | 0 | 0 | 0 | 0 | 0 | 0 |
| pH | 2 | 0 | 2 | 0 | 1 | 0 |
| Protein | 1 | 0 | 2 | 2 | 1 | 2 |
| Glucose | 0 | 0 | 0 | 0 | 0 | 0 |
| Ketone | 0 | 0 | 0 | 0 | 0 | 0 |
| Bilirubin | 0 | 0 | 0 | 0 | 0 | 0 |
| Urobilinogen | 0 | 0 | 0 | 0 | 0 | 0 |
| Nitrite | 0 | 0 | 0 | 1 | 0 | 0 |
| RBC | 1 | 0 | 2 | 1 | 0 | 0 |
| WBC | 0 | 1 | 0 | 4 | 2 | 1 |
| Number of adverse reactions (persons) | 0 | 0 | 0 | 0 | 0 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Na, G.H.; Kim, S.; Jung, H.M.; Han, S.H.; Han, J.; Koo, Y.K. Skin Anti-Aging Efficacy of Enzyme-Treated Supercritical Caviar Extract: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. Nutrients 2024, 16, 137. https://doi.org/10.3390/nu16010137
Na GH, Kim S, Jung HM, Han SH, Han J, Koo YK. Skin Anti-Aging Efficacy of Enzyme-Treated Supercritical Caviar Extract: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. Nutrients. 2024; 16(1):137. https://doi.org/10.3390/nu16010137
Chicago/Turabian StyleNa, Gwi Hwan, SukJin Kim, Hyun Mook Jung, Sang Hun Han, Jehee Han, and Yean Kyoung Koo. 2024. "Skin Anti-Aging Efficacy of Enzyme-Treated Supercritical Caviar Extract: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial" Nutrients 16, no. 1: 137. https://doi.org/10.3390/nu16010137
APA StyleNa, G. H., Kim, S., Jung, H. M., Han, S. H., Han, J., & Koo, Y. K. (2024). Skin Anti-Aging Efficacy of Enzyme-Treated Supercritical Caviar Extract: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. Nutrients, 16(1), 137. https://doi.org/10.3390/nu16010137





