Proteomic Mediators of Overall Cardiovascular Health on All-Cause Mortality
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Cardiovascular Health
2.3. Proteomic Assessment
2.4. Statistic Analysis
3. Results
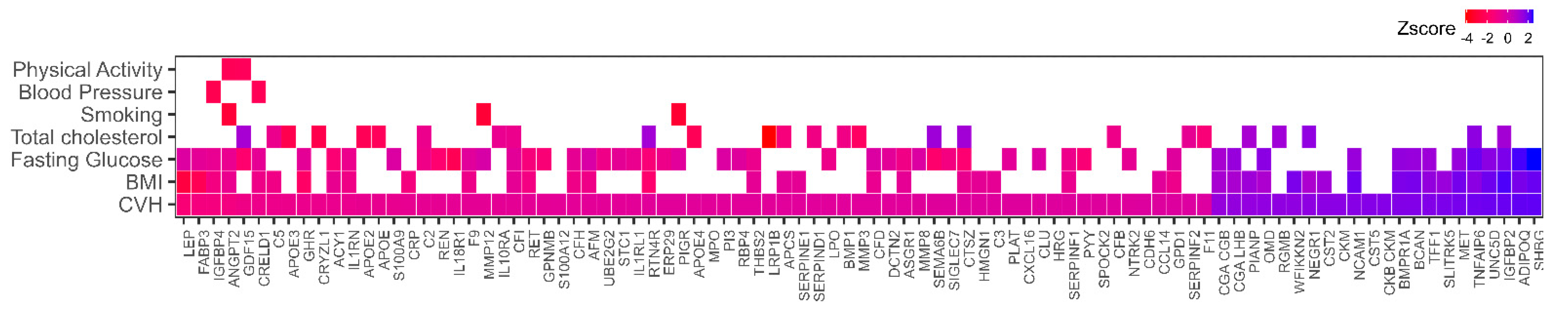
3.1. Association of Plasma Proteins with Cardiovascular Health
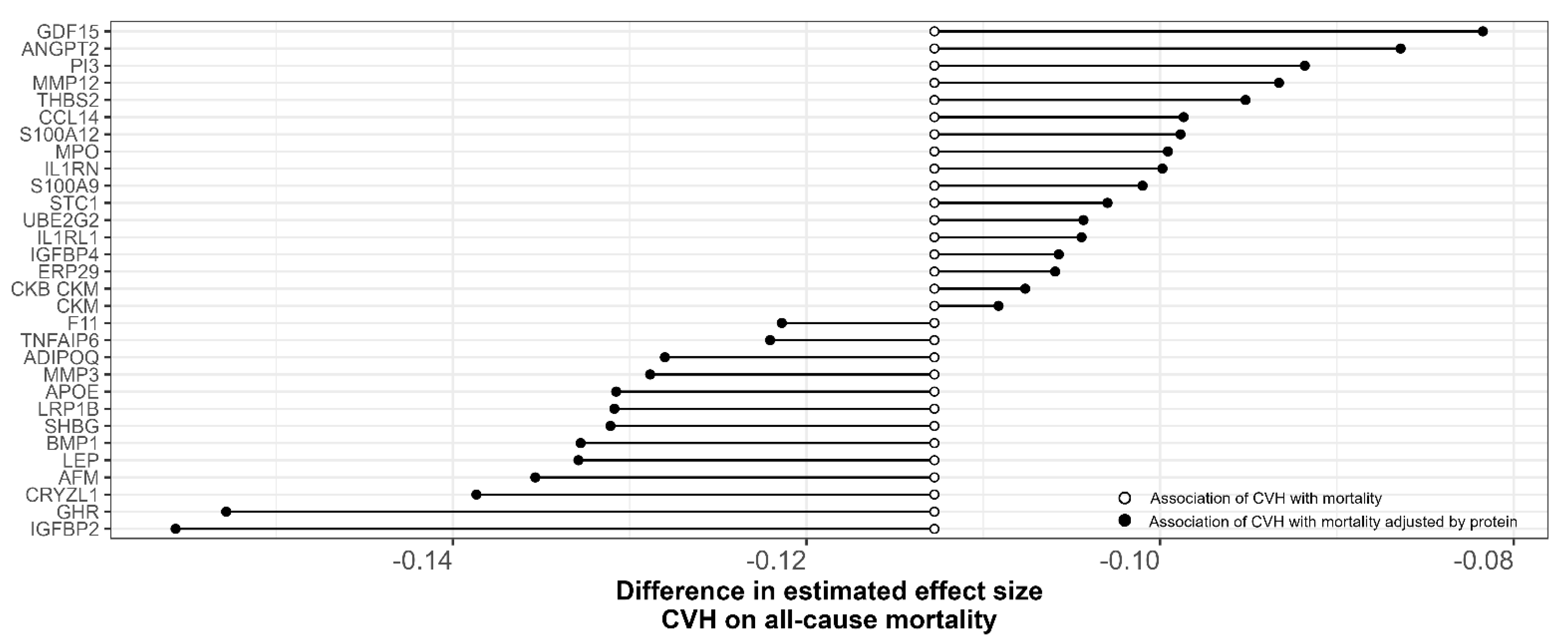
3.2. Plasma Proteins Mediate the Association between CVH and Mortality
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lloyd-Jones, D.M.; Hong, Y.; Labarthe, D.; Mozaffarian, D.; Appel, L.J.; Van Horn, L.; Greenlund, K.; Daniels, S.; Nichol, G.; Tomaselli, G.F.; et al. Defining and setting national goals for cardiovascular health promotion and disease reduction: The American Heart Association’s strategic Impact Goal through 2020 and beyond. Circulation 2010, 121, 586–613. [Google Scholar] [CrossRef] [PubMed]
- Folsom, A.R.; Yatsuya, H.; Nettleton, J.A.; Lutsey, P.L.; Cushman, M.; Rosamond, W.D.; Investigators, A.S. Community prevalence of ideal cardiovascular health, by the American Heart Association definition, and relationship with cardiovascular disease incidence. J. Am. Coll. Cardiol. 2011, 57, 1690–1696. [Google Scholar] [CrossRef] [PubMed]
- Han, L.; You, D.; Ma, W.; Astell-Burt, T.; Feng, X.; Duan, S.; Qi, L. National trends in American Heart Association Revised life’s simple 7 metrics associated with risk of mortality among US adults. JAMA Netw. Open 2019, 2, e1913131. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Cogswell, M.E.; Flanders, W.D.; Hong, Y.; Zhang, Z.; Loustalot, F.; Gillespie, C.; Merritt, R.; Hu, F.B. Trends in cardiovascular health metrics and associations with all-cause and CVD mortality among US adults. JAMA 2012, 307, 1273–1283. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, M.I.; Chevli, P.A.; Barot, H.; Soliman, E.Z. Interrelationships between american heart association’s life’s simple 7, ecg silent myocardial infarction, and cardiovascular mortality. J. Am. Heart. Assoc. 2019, 8, e011648. [Google Scholar] [CrossRef]
- Polonsky, T.S.; Ning, H.; Daviglus, M.L.; Liu, K.; Burke, G.L.; Cushman, M.; Eng, J.; Folsom, A.R.; Lutsey, P.L.; Nettleton, J.A.; et al. Association of cardiovascular health with subclinical disease and incident events: The multi-ethnic study of atherosclerosis. J. Am. Heart. Assoc. 2017, 6, e004894. [Google Scholar] [CrossRef]
- Jin, Y.; Tanaka, T.; Ma, Y.; Bandinelli, S.; Ferrucci, L.; Talegawkar, S.A. Cardiovascular Health Is Associated With Physical Function Among Older Community Dwelling Men and Women. J. Gerontol. A Biol. Sci. Med. Sci. 2017, 72, 1710–1716. [Google Scholar] [CrossRef]
- Landi, F.; Calvani, R.; Picca, A.; Tosato, M.; D’Angelo, E.; Martone, A.M.; Serafini, E.; Ortolani, E.; Savera, G.; Salini, S.; et al. Relationship between cardiovascular health metrics and physical performance in community-living people: Results from the Longevity check-up (Lookup) 7+ project. Sci. Rep. 2018, 8, 16353. [Google Scholar] [CrossRef]
- Guo, J.; Brickman, A.M.; Manly, J.J.; Reitz, C.; Schupf, N.; Mayeux, R.P.; Gu, Y. Association of Life’s Simple 7 with incident dementia and its modification by the apolipoprotein E genotype. Alzheimer’s Dement. J. Alzheimer’s Assoc. 2021, 17, 1905–1913. [Google Scholar] [CrossRef]
- Wei, J.; Wang, L.; Kulshreshtha, A.; Xu, H. Adherence to life’s simple 7 and cognitive function among older adults: The national health and nutrition examination survey 2011 to 2014. J. Am. Heart Assoc. 2022, 11, e022959. [Google Scholar] [CrossRef]
- Speh, A.; Wang, R.; Winblad, B.; Kramberger, M.G.; Backman, L.; Qiu, C.; Laukka, E.J. The relationship between cardiovascular health and rate of cognitive decline in young-old and old-old adults: A population-based study. J. Alzheimers Dis. 2021, 84, 1523–1537. [Google Scholar] [CrossRef] [PubMed]
- Palta, P.; Griswold, M.; Ranadive, R.; Bandeen-Roche, K.; Folsom, A.R.; Petruski-Ivleva, N.; Burgard, S.; Kucharska-Newton, A.; Windham, B.G. Midlife cardiovascular health and robust versus frail late-life status: The atherosclerosis risk in communities study. J. Gerontol. A. Biol. Sci. Med. Sci. 2022, 77, 1222–1229. [Google Scholar] [CrossRef] [PubMed]
- Langford, A.T.; Butler, M.; Booth, J.N.; Jin, P.; Bress, A.P.; Tanner, R.M.; Kalinowski, J.; Blanc, J.; Seixas, A.; Shimbo, D.; et al. Stress and depression are associated with life’s simple 7 among african americans with hypertension: Findings from the jackson heart study. Am. J. Hypertens. 2021, 34, 1311–1321. [Google Scholar] [CrossRef] [PubMed]
- Park, J.W.; Dulin, A.J.; Needham, B.L.; Sims, M.; Loucks, E.B.; Fava, J.L.; Dionne, L.A.; Scarpaci, M.M.; Eaton, C.B.; Howe, C.J. Examining optimism, psychosocial risks, and cardiovascular health using life’s simple 7 metrics in the multi-ethnic study of atherosclerosis and the jackson heart study. Front Cardiovasc Med. 2021, 8, 788194. [Google Scholar] [CrossRef]
- Sims, M.; Glover, L.M.; Norwood, A.F.; Jordan, C.; Min, Y.I.; Brewer, L.C.; Kubzansky, L.D. Optimism and cardiovascular health among African Americans in the Jackson Heart Study. Prev. Med. 2019, 129, 105826. [Google Scholar] [CrossRef]
- Lemke, E.; Vetter, V.M.; Berger, N.; Banszerus, V.L.; Konig, M.; Demuth, I. Cardiovascular health is associated with the epigenetic clock in the Berlin Aging Study II (BASE-II). Mech. Ageing Dev. 2022, 201, 111616. [Google Scholar] [CrossRef]
- Joyce, B.T.; Gao, T.; Zheng, Y.; Ma, J.; Hwang, S.J.; Liu, L.; Nannini, D.; Horvath, S.; Lu, A.T.; Bai Allen, N.; et al. Epigenetic Age Acceleration reflects long-term cardiovascular health. Circ. Res. 2021, 129, 770–781. [Google Scholar] [CrossRef]
- Gold, L.; Ayers, D.; Bertino, J.; Bock, C.; Bock, A.; Brody, E.N.; Carter, J.; Dalby, A.B.; Eaton, B.E.; Fitzwater, T.; et al. Aptamer-based multiplexed proteomic technology for biomarker discovery. PLoS ONE 2010, 5, e15004. [Google Scholar] [CrossRef]
- Ferrucci, L.; Bandinelli, S.; Benvenuti, E.; Di Iorio, A.; Macchi, C.; Harris, T.B.; Guralnik, J.M. Subsystems contributing to the decline in ability to walk: Bridging the gap between epidemiology and geriatric practice in the InCHIANTI study. J. Am. Geriatr. Soc. 2000, 48, 1618–1625. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Costacou, T.; Bamia, C.; Trichopoulos, D. Adherence to a Mediterranean diet and survival in a Greek population. N. Engl. J. Med. 2003, 348, 2599–2608. [Google Scholar] [CrossRef]
- Tanaka, T.; Biancotto, A.; Moaddel, R.; Moore, A.Z.; Gonzalez-Freire, M.; Aon, M.A.; Candia, J.; Zhang, P.; Cheung, F.; Fantoni, G.; et al. Plasma proteomic signature of age in healthy humans. Aging Cell 2018, 17, e12799. [Google Scholar] [CrossRef] [PubMed]
- Candia, J.; Cheung, F.; Kotliarov, Y.; Fantoni, G.; Sellers, B.; Griesman, T.; Huang, J.; Stuccio, S.; Zingone, A.; Ryan, B.M.; et al. Assessment of Variability in the SOMAscan Assay. Sci. Rep. 2017, 7, 14248. [Google Scholar] [CrossRef] [PubMed]
- Candia, J.; Daya, G.N.; Tanaka, T.; Ferrucci, L.; Walker, K.A. Assessment of variability in the plasma 7k SomaScan proteomics assay. Sci. Rep. 2022, 12, 17147. [Google Scholar] [CrossRef]
- Jin, Y.; Tanaka, T.; Bandinelli, S.; Ferrucci, L.; Talegawkar, S.A. Overall cardiovascular health is associated with all-cause and cardiovascular disease mortality among older community-dwelling men and women. J. Aging Health. 2017, 29, 437–453. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Basisty, N.; Fantoni, G.; Candia, J.; Moore, A.Z.; Biancotto, A.; Schilling, B.; Bandinelli, S.; Ferrucci, L. Plasma proteomic biomarker signature of age predicts health and life span. Elife 2020, 9, e61073. [Google Scholar] [CrossRef] [PubMed]
- Markiewski, M.M.; Nilsson, B.; Ekdahl, K.N.; Mollnes, T.E.; Lambris, J.D. Complement and coagulation: Strangers or partners in crime? Trends Immunol. 2007, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Egerstedt, A.; Berntsson, J.; Smith, M.L.; Gidlof, O.; Nilsson, R.; Benson, M.; Wells, Q.S.; Celik, S.; Lejonberg, C.; Farrell, L.; et al. Profiling of the plasma proteome across different stages of human heart failure. Nat. Commun. 2019, 10, 5830. [Google Scholar] [CrossRef] [PubMed]
- Bots, M.L.; Breteler, M.M.; van Kooten, F.; Haverkate, F.; Meijer, P.; Koudstaal, P.J.; Grobbee, D.E.; Kluft, C. Coagulation and fibrinolysis markers and risk of dementia. The dutch vascular factors in dementia study. Haemostasis 1998, 28, 216–222. [Google Scholar] [CrossRef]
- van Oijen, M.; Witteman, J.C.; Hofman, A.; Koudstaal, P.J.; Breteler, M.M. Fibrinogen is associated with an increased risk of Alzheimer disease and vascular dementia. Stroke 2005, 36, 2637–2641. [Google Scholar] [CrossRef]
- Gailani, D.; Broze, G.J., Jr. Factor XI activation in a revised model of blood coagulation. Science 1991, 253, 909–912. [Google Scholar] [CrossRef]
- Preis, M.; Hirsch, J.; Kotler, A.; Zoabi, A.; Stein, N.; Rennert, G.; Saliba, W. Factor XI deficiency is associated with lower risk for cardiovascular and venous thromboembolism events. Blood 2017, 129, 1210–1215. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.T.; Flanders, M.M.; Kim, H.; Rodgers, G.M. Elevated factor XI activity levels are associated with an increased odds ratio for cerebrovascular events. Am. J. Clin. Pathol. 2006, 126, 411–415. [Google Scholar] [CrossRef] [PubMed]
- Kyrle, P.A.; Eischer, L.; Sinkovec, H.; Eichinger, S. Factor XI and recurrent venous thrombosis: An observational cohort study. J. Thromb. Haemost. 2019, 17, 782–786. [Google Scholar] [CrossRef]
- Landino, K.; Tanaka, T.; Fantoni, G.; Candia, J.; Bandinelli, S.; Ferrucci, L. Characterization of the plasma proteomic profile of frailty phenotype. Geroscience 2021, 43, 1029–1037. [Google Scholar] [CrossRef] [PubMed]
- Junnila, R.K.; List, E.O.; Berryman, D.E.; Murrey, J.W.; Kopchick, J.J. The GH/IGF-1 axis in ageing and longevity. Nat. Rev. Endocrinol. 2013, 9, 366–376. [Google Scholar] [CrossRef] [PubMed]
- Milman, S.; Huffman, D.M.; Barzilai, N. The Somatotropic Axis in Human Aging: Framework for the Current State of Knowledge and Future Research. Cell Metab. 2016, 23, 980–989. [Google Scholar] [CrossRef]
- Liu, H.; Bravata, D.M.; Olkin, I.; Nayak, S.; Roberts, B.; Garber, A.M.; Hoffman, A.R. Systematic review: The safety and efficacy of growth hormone in the healthy elderly. Ann. Intern. Med. 2007, 146, 104–115. [Google Scholar] [CrossRef] [PubMed]
- van der Spoel, E.; Rozing, M.P.; Houwing-Duistermaat, J.J.; Slagboom, P.E.; Beekman, M.; de Craen, A.J.; Westendorp, R.G.; van Heemst, D. Association analysis of insulin-like growth factor-1 axis parameters with survival and functional status in nonagenarians of the Leiden Longevity Study. Aging 2015, 7, 956–963. [Google Scholar] [CrossRef]
- Svensson, J.; Carlzon, D.; Petzold, M.; Karlsson, M.K.; Ljunggren, O.; Tivesten, A.; Mellstrom, D.; Ohlsson, C. Both low and high serum IGF-I levels associate with cancer mortality in older men. J. Clin. Endocrinol. Metab. 2012, 97, 4623–4630. [Google Scholar] [CrossRef]
- Tumati, S.; Burger, H.; Martens, S.; van der Schouw, Y.T.; Aleman, A. Association between Cognition and Serum Insulin-Like Growth Factor-1 in Middle-Aged & Older Men: An 8 Year Follow-Up Study. PLoS ONE 2016, 11, e0154450. [Google Scholar] [CrossRef]
- Saydah, S.; Graubard, B.; Ballard-Barbash, R.; Berrigan, D. Insulin-like growth factors and subsequent risk of mortality in the United States. Am. J. Epidemiol. 2007, 166, 518–526. [Google Scholar] [CrossRef] [PubMed]
- Green, C.J.; Holly, J.M.; Bayer, A.; Fish, M.; Ebrahim, S.; Gallacher, J.; Ben-Shlomo, Y. The role of IGF-I, IGF-II, and IGFBP-3 in male cognitive aging and dementia risk: The caerphilly prospective study. J. Alzheimers. Dis. 2014, 41, 867–875. [Google Scholar] [CrossRef] [PubMed]
- Andreassen, M.; Raymond, I.; Kistorp, C.; Hildebrandt, P.; Faber, J.; Kristensen, L.O. IGF1 as predictor of all cause mortality and cardiovascular disease in an elderly population. Eur. J. Endocrinol. 2009, 160, 25–31. [Google Scholar] [CrossRef]
- Westwood, A.J.; Beiser, A.; Decarli, C.; Harris, T.B.; Chen, T.C.; He, X.M.; Roubenoff, R.; Pikula, A.; Au, R.; Braverman, L.E.; et al. Insulin-like growth factor-1 and risk of Alzheimer dementia and brain atrophy. Neurology 2014, 82, 1613–1619. [Google Scholar] [CrossRef] [PubMed]
- Adams, E.R.; Nolan, V.G.; Andersen, S.L.; Perls, T.T.; Terry, D.F. Centenarian offspring: Start healthier and stay healthier. J. Am. Geriatr. Soc. 2008, 56, 2089–2092. [Google Scholar] [CrossRef]
- Batsis, J.A.; Sahakyan, K.R.; Singh, P.; Bartels, S.J.; Somers, V.K.; Lopez-Jimenez, F. Leptin, adiposity, and mortality: Results from the National Health and Nutrition Examination Survey III, 1988 to 1994. Mayo Clin. Proc. 2015, 90, 481–491. [Google Scholar] [CrossRef]
- Yang, H.; Guo, W.; Li, J.; Cao, S.; Zhang, J.; Pan, J.; Wang, Z.; Wen, P.; Shi, X.; Zhang, S. Leptin concentration and risk of coronary heart disease and stroke: A systematic review and meta-analysis. PLoS ONE 2017, 12, e0166360. [Google Scholar] [CrossRef]
- Lloyd-Jones, D.M.; Allen, N.B.; Anderson, C.A.M.; Black, T.; Brewer, L.C.; Foraker, R.E.; Grandner, M.A.; Lavretsky, H.; Perak, A.M.; Sharma, G.; et al. Life’s Essential 8: Updating and Enhancing the American Heart Association’s Construct of Cardiovascular Health: A Presidential Advisory From the American Heart Association. Circulation 2022, 146, e18–e43. [Google Scholar] [CrossRef]
| All | Tertile 1 Score 0–6 | Tertile 2 Score 7–8 | Tertile 3 Score 9–12 | p | |
|---|---|---|---|---|---|
| n | 703 | 191 | 275 | 237 | |
| Age (years) | 73.3 (6.24) | 73.6 (6.35) | 73.5 (6.20) | 72.9 (6.21) | 0.479 |
| Female (%) | 54.2 | 56.0 | 56.4 | 50.2 | 0.318 |
| Bagno a Ripoli (%) | 52.5 | 52.4 | 51.6 | 53.6 | 0.907 |
| Creatinine clearance (mL/minute) | 77.8 (24.9) | 75.9 (24.2) | 76.9 (23.5) | 80.5 (26.8) | 0.123 |
| Cardiovascular Health (CVH score) | 7.61 (1.90) | 5.25 (1.04) | 7.48 (0.50) | 9.67 (0.85) | <0.001 |
| BMI (kg/m2) | 27.5 (4.03) | 29.6 (4.27) | 27.7 (3.94) | 25.6 (2.93) | <0.001 |
| Fasting Glucose (mg/dL) | 95.4 (24.3) | 109.3 (32.2) | 92.8 (21.6) | 87.3 (12.3) | <0.001 |
| Total cholesterol (mg/dL) | 219.9 (38.3) | 234.0 (38.38) | 223.9 (35.2) | 204.0 (36.0) | <0.001 |
| Mediterranean diet score | 4.52 (1.63) | 3.79 (1.58) | 4.47 (1.56) | 5.17 (1.48) | <0.001 |
| Systolic blood pressure (mmHg) | 150.0 (19.3) | 154.3 (16.0) | 152.1 (19.2) | 144.0 (20.4) | <0.001 |
| Diastolic blood pressure (mmHg) | 84.0 (8.45) | 85.9 (7.80) | 84.4 (8.15) | 82.0 (8.90) | <0.001 |
| Physical activity (%) | <0.001 | ||||
| Low | 15.2 | 32.5 | 14.2 | 2.5 | |
| Medium | 45.1 | 53.4 | 51.6 | 30.8 | |
| High | 39.7 | 14.1 | 34.2 | 66.7 | |
| Smoking (%) | <0.001 | ||||
| Never smoked | 57.2 | 42.4 | 57.1 | 69.2 | |
| Former smoker | 28.2 | 30.4 | 29.5 | 24.9 | |
| Current smoker | 14.7 | 27.2 | 13.5 | 5.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tanaka, T.; Talegawkar, S.A.; Jin, Y.; Candia, J.; Fantoni, G.; Bandinelli, S.; Ferrucci, L. Proteomic Mediators of Overall Cardiovascular Health on All-Cause Mortality. Nutrients 2023, 15, 781. https://doi.org/10.3390/nu15030781
Tanaka T, Talegawkar SA, Jin Y, Candia J, Fantoni G, Bandinelli S, Ferrucci L. Proteomic Mediators of Overall Cardiovascular Health on All-Cause Mortality. Nutrients. 2023; 15(3):781. https://doi.org/10.3390/nu15030781
Chicago/Turabian StyleTanaka, Toshiko, Sameera A. Talegawkar, Yichen Jin, Julián Candia, Giovanna Fantoni, Stefania Bandinelli, and Luigi Ferrucci. 2023. "Proteomic Mediators of Overall Cardiovascular Health on All-Cause Mortality" Nutrients 15, no. 3: 781. https://doi.org/10.3390/nu15030781
APA StyleTanaka, T., Talegawkar, S. A., Jin, Y., Candia, J., Fantoni, G., Bandinelli, S., & Ferrucci, L. (2023). Proteomic Mediators of Overall Cardiovascular Health on All-Cause Mortality. Nutrients, 15(3), 781. https://doi.org/10.3390/nu15030781






